Abstract
Exercise prescribed according to relative intensity is a routine feature in the exercise science literature and is intended to produce an approximately equivalent exercise stress in individuals with different absolute exercise capacities. The traditional approach has been to prescribe exercise intensity as a percentage of maximal oxygen uptake (VO2max) or maximum heart rate (HRmax) and these methods remain common in the literature. However, exercise intensity prescribed at a %VO2max or %HRmax does not necessarily place individuals at an equivalent intensity above resting levels. Furthermore, some individuals may be above and others below metabolic thresholds such as the aerobic threshold (AerT) or anaerobic threshold (AnT) at the same %VO2max or %HRmax. For these reasons, some authors have recommended that exercise intensity be prescribed relative to oxygen consumption reserve (VO2R), heart rate reserve (HRR), the AerT, or the AnT rather than relative to VO2max or HRmax. The aim of this review was to compare the physiological and practical implications of using each of these methods of relative exercise intensity prescription for research trials or training sessions. It is well established that an exercise bout at a fixed %VO2max or %HRmax may produce interindividual variation in blood lactate accumulation and a similar effect has been shown when relating exercise intensity to VO2R or HRR. Although individual variation in other markers of metabolic stress have seldom been reported, it is assumed that these responses would be similarly heterogeneous at a %VO2max, %HRmax, %VO2R, or %HRR of moderate-to-high intensity. In contrast, exercise prescribed relative to the AerT or AnT would be expected to produce less individual variation in metabolic responses and less individual variation in time to exhaustion at a constant exercise intensity. Furthermore, it would be expected that training prescribed relative to the AerT or AnT would provide a more homogenous training stimulus than training prescribed as a %VO2max. However, many of these theoretical advantages of threshold-related exercise prescription have yet to be directly demonstrated. On a practical level, the use of threshold-related exercise prescription has distinct disadvantages compared to the use of %VO2max or %HRmax. Thresholds determined from single incremental tests cannot be assumed to be accurate in all individuals without verification trials. Verification trials would involve two or three additional laboratory visits and would add considerably to the testing burden on both the participant and researcher. Threshold determination and verification would also involve blood lactate sampling, which is aversive to some participants and has a number of intrinsic and extrinsic sources of variation. Threshold measurements also tend to show higher day-to-day variation than VO2max or HRmax. In summary, each method of prescribing relative exercise intensity has both advantages and disadvantages when both theoretical and practical considerations are taken into account. It follows that the most appropriate method of relative exercise intensity prescription may vary with factors such as exercise intensity, number of participants, and participant characteristics. Considering a method’s limitations as well as advantages and increased reporting of individual exercise responses will facilitate accurate interpretation of findings and help to identify areas for further study.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
It is widely understood that exercise standardized according to an absolute external workload may produce large differences in internal cardiovascular and metabolic stress between individuals. For this reason, it is more common to “individualize” exercise prescription according to relative intensity [1–3]. This approach is intended to account for differences in physiological and functional capacity, to produce an approximately equivalent exercise stress among individuals despite differences in phenotype. For research purposes, controlling the relative intensity of an exercise bout allows for the interpretation of other exercise-related responses, while for health and performance purposes prescribing training according to relative intensity allows for more predictable adaptive responses.
The traditional approach when prescribing relative intensity has been to use a percentage of maximal oxygen consumption (VO2max) or maximal heart rate (HRmax) and many recent publications continue to favor these methods [4–11]. However, a number of authors have argued against the use of %VO2max or %HRmax for exercise intensity prescription, recommending other methods as more meaningful for equating exercise stress [12–19]. For example, Swain et al. [12–14] have reasoned that the use of %VO2max does not account for individual differences in resting metabolic rate and that it is preferable to prescribe exercise relative to an individual’s oxygen consumption reserve (VO2R) [VO2max minus resting oxygen consumption (VO2)]. Use of %VO2R has the advantage of placing individuals at an equivalent intensity above resting levels. Furthermore, several studies have found that %VO2R and percent heart rate reserve (HRR) [HRmax minus resting heart rate (HR)] can be considered equivalent methods of exercise intensity prescription, whereas %VO2max and %HRR may differ noticeably at lower exercise intensities [13, 14, 20]. Based on the convenience of the %VO2R–%HRR relationship for HR-based monitoring of training, these methods of exercise intensity prescription were recommended by the American College of Sports Medicine (ACSM) in 1998 [21]. However, the %VO2R–%HRR relationship has since been questioned [22–25] and the 2011 ACSM guidelines included %VO2max and %HRmax along with %VO2R and %HRR as recommended methods of exercise intensity prescription [3].
A separate criticism of the use of %VO2max or %HRmax for exercise intensity prescription is that these methods fail to account for differences in metabolic stress [15–19]. Authors highlighting this discrepancy have advocated the use of metabolic thresholds such as the aerobic threshold (AerT) and anaerobic threshold (AnT) as preferable “anchors” for relative exercise intensity prescription and there are indeed numerous examples of this approach [26–32]. However, there has been little consistency in methods of threshold calculation and the theoretical basis of the thresholds remains controversial [33–35].
In summary, there appear to be some discrepancies in the methods of relative exercise intensity prescription recommended by different authors. Furthermore, there are discrepancies between methods of exercise intensity prescription that have been recommended and those methods that continue to be used by researchers. Although different methods of relative exercise intensity prescription have been reviewed on previous occasions, Hills et al. [1] focused on the development of equations for prescribing exercise intensity and Carvalho et al. [2] focused on the implications of prescribing exercise intensity in clinical populations. In contrast, the aim of the current review was to compare the physiological and practical implications of prescribing exercise relative to VO2max, HRmax, VO2R, or HRR, the AerT or the AnT in healthy, active, or athletic populations.
2 Physiological Responses to Exercise Standardized by Relative Intensity
2.1 Acute Exercise Responses
2.1.1 Systemic Responses
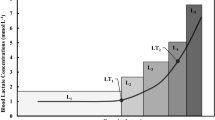
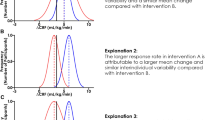
Most authors citing poorly standardized metabolic stress at a fixed %VO2max or %HRmax have based this view on the individual variation in blood lactate accumulation that may occur when using these methods [15–19]. For example, Dwyer and Bybee [16] observed that for any intensity between 58 and 75 % VO2max, some of their participants were below, and others above the AnT. Conversely, Meyer et al. [17] showed that the workload associated with 75 % VO2max corresponded to 86–118 % of the individual anaerobic threshold and blood lactate concentrations of 1.4–4.6 mmol/l in different individuals following an incremental test. Scharhag-Rosenberger et al. [18] reported similar findings with 4 and 14 of 18 participants exceeding the individual anaerobic threshold during constant-intensity exercise at 60 and 75 % of VO2max, respectively. Furthermore, in comparing the blood lactate ranges at 60 % VO2max (0.7–5.6 mmol/l) and 75 % VO2max (2.2–8.0 mmol/l), Scharhag-Rosenberger et al. demonstrated increased variation in blood lactate response with increasing exercise intensity by %VO2max as well as increased variation in blood lactate response in a heterogeneous group when compared to the more homogenous group of Meyer et al. [17] at the same intensity. Although blood lactate variation at a given %VO2max would be expected to be larger in heterogeneous groups, some variation may occur even when individuals have a similar VO2max [36].
Even though exercise prescribed as a %VO2R or %HRR may place individuals at a similar intensity above resting metabolism, these methods have also been linked to individual variation relative to threshold concepts [18, 37–40]. For example, Scharhag-Rosenberger et al. [18] reported that some participants were above and others below the AnT at an intensity corresponding to 71 ± 1 % of VO2R and Acevedo et al. [40] reported that the AerT occurred at 70 ± 10 % HRR in men of high cardiorespiratory fitness, implying that a fixed %HRR could be above the AerT for some individuals and below the AerT for others.
Notably, it is individuals exercising in different exercise intensity domains at the same %VO2max, %HRmax, %VO2R, or %HRR that may be cause for concern rather than heterogeneous blood lactate concentrations; it has been documented that the AnT can be associated with blood lactate concentrations of 2–9 mmol/l in different individuals [41–43]. As is well known, different exercise intensity domains are associated not only with a shift in blood lactate responses but also with changes in ventilation [44], oxygen uptake kinetics [45], and catecholamine responses [46, 47]. For example, constant-intensity exercise within the “severe” exercise intensity domain (>AnT) is characterized by a continuous increase in ventilation and VO2, progressive acidosis, and metabolite accumulation, whereas constant-intensity exercise equal to or below the AnT is associated with a physiological steady state [48–51]. The metabolic characteristics of the AnT are analogous to those of critical power in that both measurements are intended to represent the highest workload at which it is possible to achieve a steady state [48, 52]. It follows that many of the discussion points regarding exercise prescription relative to the AnT also apply when prescribing exercise intensity relative to critical power. On a practical level, however, the AnT has been associated with a significantly lower workload and increased time to exhaustion when compared to critical power [42, 48, 53].
2.1.2 Time to Exhaustion During Constant-Intensity Exercise and Correlations with Performance
If exercise intensity prescribed as a %VO2max, %HRmax, %VO2R, or %HRR results in different metabolic and respiratory profiles among individuals, these differences would be expected to contribute to individual variation in the time to exhaustion at a constant exercise intensity. For example, those exercising above the AnT might be expected to terminate exercise earlier than those exercising below the AnT at the same %VO2max due to an increased depletion of anaerobic energy reserves and an increased accumulation of metabolites [54]. However, Scharhag-Rosenberger et al. [18] found that exercise above or below the AnT did not appear to explain premature exercise cessation when individuals of varied aerobic capacity attempted 60 min of exercise at 75 % VO2max. This finding highlights both the complex nature of fatigue [55] and the discrepancies that can exist between theoretical expectations and individual responses in practice. Nevertheless, at the cross-sectional level, lactate threshold concepts have been related to metabolic activity in the muscle through significant correlations with muscle capillarization (r = 0.59–0.77) [56, 57], percentage of slow twitch fibers (r = 0.74–0.78 [57, 58]), oxidative capacity (r = 0.94 [58]), and muscle enzyme activity (r = 0.54–0.68 [59, 60], and it is not surprising that blood lactate markers have been shown to explain more variation in performance than is explained by VO2max; for example, r = 0.83 vs 0.91 [61], r = 0.55 vs 0.61–0.84 [62], and r = 0.51 vs 0.76 [63].
2.1.3 Molecular Responses
While it is clear that the skeletal muscle’s transcriptional response to exercise is sensitive to increases in relative exercise intensity [9, 64, 65], the effect of different methods of relative exercise intensity prescription does not appear to have been directly investigated. The biochemical signals that activate adaptive cellular pathways include an increased ratio of adenosine monophosphate (AMP) to adenosine triphosphate (ATP), increased levels of reactive species of oxygen and nitrogen, depleted levels of muscle glycogen, and decreased oxygen tension [66–68]; it could be speculated that changes in blood lactate accumulation would be associated with some of these changes. Nevertheless, without evidence, it is not clear whether exercise above and below a threshold concept would be more significant for training adaptation than any other increase in exercise intensity. Future investigations could address this topic by comparing transcriptional and translation responses of individuals exercising above and below a threshold measurement but at the same %VO2max.
2.2 Training Responses
2.2.1 Individual Variation in Training Responses
Adaptation to training can be understood as the accumulated effect of micro-adaptations that occur in response to the stimulus of each training session [68]. With this in mind, it has been suggested that differences in acute metabolic stress during exercise prescribed relative to %VO2max (i.e., individuals exercising above and below threshold levels) may explain the large interindividual variation in response that has been reported following training programs using this method of exercise prescription [17, 18, 69]. Although many studies allude to variation in response by way of the standard deviation around the mean response, only a small number of studies deliberately highlight these individual differences. One prominent study that highlighted individual differences is the HERITAGE Family study where 20 weeks of endurance training standardized by %VO2max produced a mean increase in VO2max of 384 ml O2 with a range of ~0 to 1,000 ml O2 for individual responses [70]. Large ranges in individual VO2max response have also been reported following other training programs based on %VO2max [19, 71] as well as following training programs prescribed by %HRmax [72, 73] and %HRR [74]. Furthermore, a range of individual responses following training based on maximal measurements is not restricted to VO2max but includes other measurements such as submaximal HR [19, 70], the AerT [74], the AnT [74], submaximal blood lactate concentration [19], muscle glycogen [19], muscle enzyme activity [8, 19], and performance [19]. While Bouchard and Rankinen [70] are confident that individual differences in training responses are “biologically meaningful,” it is rare for authors to discuss individual variation in response in the context of within-subject variability [75]. It could be argued that only individual differences in response that exceed the biological variation of a measurement represent differences that are truly meaningful.
If different individual responses following training at a %VO2max, %HRmax, or %HRR can be explained by differences in metabolic stress, it follows that training standardized with respect to metabolic stress should produce more homogenous individual responses. However, it is rare to find studies addressing individual variation when exercise has been prescribed relative to a threshold concept. Karavirta et al. [76] demonstrated substantial individual variation in VO2max responses (approximately −10 to +58 %) following 21 weeks of endurance training prescribed relative to threshold concepts. However, the authors described the prescribed intensities as simply “below,” “between,” or “above” the aerobic and anaerobic thresholds, and it is unclear whether inadequate standardization of the exercise prescription may have contributed to the heterogeneous responses. In another example, McLellan and Skinner [69] investigated whether interindividual variability in response would be reduced in a group trained relative to the AerT compared to a group trained relative to VO2max following 8 weeks of cycling training. Contrary to their expectations, no group differences in individual variation were observed; however, the small participant numbers (n = 6 and 8 per group) and the manipulation of individual training loads to match overall intensity between the groups (%VO2max group = 50–58 %VO2max and +3.6 to +8.5 % AerT) make these results similarly difficult to interpret. Until a training study strictly standardized relative to a threshold measurement clarifies the individual responses, it remains an assumption that the use of threshold-related training will produce less individual variability in training effects than training prescribed by %VO2max.
2.2.2 Contribution of Genetics to Individual Variation in Training Response
An individual’s response to exercise training is determined not only by the physiological stress of the exercise bouts but also by the individual’s genotype. This is an important reason why reduced individual variation following threshold-related training would need to be demonstrated, rather than assumed. A detailed discussion of genomic predictors of trainability is beyond the scope of the present review and can be found elsewhere [77]. However, by way of illustration, the HERITAGE Family study reported that the VO2max training response showed 2.5 times more variance between families than within families following 20 weeks of endurance training [78]. Hereditary factors explained up to 47 % of VO2max “trainability” in Caucasian participants [78] and a subsequent genome-wide analysis identified 21 single-nucleotide polymorphisms that explained 49 % of the variance in VO2max response [79]. In a similar sample, the hereditary contribution to submaximal heart rate response was calculated to be about 30 % [80] and very significant familial aggregation has also been reported for the training response of enzymes in the phosphagen, glycolytic, and oxidative pathways [81]. Genetic factors may also affect changes in body temperature, norepinephrine, and blood lactate during exercise among untrained individuals [82]. It is not yet known how the amount of variance explained by hereditary factors is affected by differences in exercise intensity or exercise mode [77]. Nevertheless, it is possible that genotype may account for a significant proportion of the variation following training studies based on %VO2max. The relative contribution of hereditary factors versus differences in metabolic stress during exercise is some way from being understood and provides a large scope for further study.
3 Practical Advantages and Disadvantages of Different Anchor Measurements
When making the case for one method of relative exercise prescription over another, it is typical for the recommendation to center on the physiological basis of each method. Often, little consideration is given to the day-to-day application of each method. It follows that an essential aspect of comparing and contrasting the aforementioned anchor measurements is to consider questions such as those addressed below.
3.1 Can the Measurement be Verified?
Although the measurements under discussion are routinely determined, the characteristics of the incremental protocol may differ depending on the requirements of the study and the preferences of the research group. In a similar way, there may also be variation in the method of data processing, and, where appropriate, the model of graphical analysis. These differences in the nature of the protocol and method of graphical analysis can have a large effect on the measurement value under some circumstances [83]. It follows that if the measurement is to be used as the basis for prescribing relative intensity or to monitor changes in performance, verifying the measurement value would be an important precaution.
3.1.1 VO2max and HRmax Verification
Failure of participants to reach maximal exertion would result in VO2max or HRmax being underestimated and it is common to apply certain checks to evaluate whether the measurement was truly maximal. For example, criteria such as respiratory exchange ratio (RER) > 1.1 or 1.15, a “plateau” in VO2 with an increased workload, and blood lactate >8 mmol/l are often used to verify a true VO2max. However, using these criteria not only allows for the significant underestimation of VO2max [84] but may also result in some genuinely maximal efforts being discounted [84–86]. In recent years, a solution to the limitations of these secondary criteria has emerged in the form of a square wave “verification” bout, performed to exhaustion shortly after the graded exercise protocol [87]. There is, as yet, no consensus as to the duration of the rest interval nor the use of a submaximal or supramaximal workload, and a recent report has suggested that these factors might be adjusted based on the length of the preceding protocol [88]. However, it would appear that a 10-min rest period prior to a supramaximal verification bout is sufficient [89, 90]. The duration of the supramaximal verification bout itself is reported to be approximately 2 min with the verification VO2max expected to differ from the incremental VO2max by no more than 3 % [85, 91, 92] or 5.5 % [90] according to different recommendations.
A HRmax is generally considered accurate if it falls within 10 beats/min of the “220 minus age” predicted HRmax. This formula is so widely used that an original reference is very rarely cited but Robergs and Landwehr [93] have attributed the original reference to Fox et al. [94]. Robergs and Landwehr calculated that the standard error of the estimate around Fox’s original data was certainly greater than 10 beats/min, thus proximity to that particular age-predicted HRmax does not constitute a meaningful criterion for having attained HRmax. Although other more accurate formulas have been developed, the use of any standard formula does not consider the variation in HRmax that can occur across different exercise modes. As an alternative, Midgley et al. [89] incorporated HRmax verification within the VO2max verification bout and proposed a verification criterion of ≤4 beat/min difference between the initial HRmax and the verification HRmax of an individual. Notably, the protocol from which this criterion was developed involved 3 min of submaximal exercise before the supramaximal workload, resulting in a total verification exercise time of ±4.5 min. It follows that this criterion may not be appropriate for shorter verification trials that allow less time for heart rate to reach maximum.
3.1.2 VO2R and HRR Verification
Given that VO2R is based on both resting VO2 and VO2max, verifying both of these measurements should ensure that a true VO2R value has been obtained. Although many authors take precautions of some form to verify that a true VO2max has been obtained (see Sect. 3.1.1), a recent review highlighted poor standardization of resting VO2 measurements and the error that variation in resting VO2 can introduce into %VO2R exercise prescription [23]. A number of studies reporting VO2R have inferred a standard resting VO2 of 3.5 ml/kg per min for all individuals [25, 95, 96], whereas others have determined resting VO2 individually but have failed to fulfill “best-practice” criteria for resting VO2 measurements [13, 14, 20]. These best-practice criteria for resting metabolic measurements are based on a systematic review by Compher et al. [97] and include a ≥5-h fast and restrictions on physical exertion in the hours prior to the assessment among other evidence-based recommendations. Recent studies reporting VO2R have adopted these best-practice criteria for determining resting VO2, but did not verify VO2max at supramaximal intensities [22, 24]. An approach incorporating both of these elements might reasonably be assumed to produce an accurate VO2R, just as the corresponding values for resting heart rate and HRmax might reasonably be assumed to produce an accurate HRR.
3.1.3 AerT and AnT Verification
Although it is possible to verify AerT and AnT, doing so would require a minimum of two subsequent laboratory visits and several blood lactate measurements. For AerT, workloads less than or equal to the AerT would be expected to produce blood lactate concentrations not different from baseline, whereas workloads greater than AerT would be expected to produce a blood lactate concentration that was significantly elevated above baseline but stable [98].
Similarly, the AnT could be verified by demonstrating that workloads less than or equal to the AnT workload produced blood lactate responses that were elevated but stable, perhaps showing a slight decrease toward the end of the exercise bout [48, 98], whereas workloads greater than AnT should result in a progressive increase in blood lactate concentration [48, 98, 99]. In essence, verifying the AnT would involve showing that an AnT workload calculated from an incremental test was, in fact, the maximal lactate steady state (MLSS). Detection of the MLSS is, in turn, influenced by the length of the constant-load exercise and the maximum acceptable increase in blood lactate that is applied [100]. The generally accepted [48, 101] standard appears to be an increase in blood lactate of no more than 1 mmol/l after 10 min of an exercise bout at least 30 min in duration [100, 102].
Although some studies have reported stable blood lactate concentrations at the workload associated with the individual anaerobic threshold (IAT) [41, 103], they did not demonstrate the accumulation of lactate at higher workloads and it is possible that the MLSS was underestimated. In instances where the IAT was indeed verified using the blood lactate response during two to three 30-min exercise bouts, the IAT overestimated the MLSS at a group level [104], and in some individuals [51], respectively. Another study [43] found no significant group mean differences between the workload at the MLSS, at 4 mmol/l blood lactate, and at the AnT determined according to Cheng’s “D max” method [105]. However, the authors concluded that neither of the single-effort methods was sufficiently precise to identify the MLSS on an individual level and recommended constant-load verification trials if the MLSS was to be considered valid.
3.2 Is the Measurement Reliable?
If a measurement has been verified as accurate on one particular day, would a subsequent verification produce a different result? It is to be expected that each of the anchor measurements will have a certain amount of day-to-day variation as a result of biological variation, equipment error, and reproducibility of the testing protocol. However, the smaller the variation on a day-to-day basis, the more reliable the measurement and the more likely that exercise bouts determined relative to the measurement will have the anticipated physiological effect.
The literature contains a variety of reliability measurements including absolute and relative measures of within-participant variation and measures of between-participant variation by way of test–retest correlations. Absolute within-participant variation may be influenced by the absolute magnitude of the participant’s measurements, while the between-participant variation is influenced by the degree of heterogeneity within the participant group. Therefore, the most reasonable basis for comparing the reliability of different anchor measurements may be the relative within-participant reliability or coefficient of variation (CV). The CV represents the measurement error as a percentage of the measurement mean and the present discussion includes CVs calculated in the following ways (a) \( {\text{CV}} = \left( {\surd (\sum d^{ 2} / 2n)} \right)/{\text{sample mean}} \) where d is the between-trial difference and n is the number of participants [106], (b) \( {\text{CV}} = ({\text{SD of difference}}/\surd 2)/{\text{sample mean,}} \) referring to the standard deviation of the between-trial differences [107], (c) \( {\text{CV}} = ({\text{SD}}\surd ( 1- {\text{r))}}/{\text{sample mean}}, \) referring to the average standard deviation of the repeated trials and Pearson’s correlation coefficient or intraclass correlation coefficient [108], and (d) \( {\text{CV = SD of the test-retest differences/sample mean}} \) [109].
3.2.1 Relative Reliability of VO2max, HRmax, and Threshold Measurements
Variation in the statistical basis of the CV calculation may affect the reported measurement reliability, along with factors such as the training status of the participants [110], exercise mode, equipment used, and the time period between repeated trials. Therefore, this review focused on studies reporting the reliability of at least two anchor measurements such that their relative reliability could be compared on an equal basis. For example, of six studies reporting the reliability of VO2max, HRmax, and one or more threshold measurements, five studies found HRmax to be the most reliable measurement (HRmax CV = 1.0–3.2 %) [109, 111–114] and one study found it to be approximately equivalent to the most reliable measurement (AnT HR CV = 1.2 %, HRmax CV = 1.3 %) [115] (Table 1). There was a lack of agreement over which was the next most reliable measurement with three studies reporting VO2max as more reliable than AerT [112, 114] or AerT and AnT [109], two studies reporting threshold measurements as more reliable than VO2max [113, 115], and one study reporting very similar CV values for VO2max and AerT (CV = 3.5 vs 3.8 %) [111].
Those studies that found VO2max was more reliable than threshold measurements reported VO2max CVs of 1.9, 2.0, and 4.0 %, which are in keeping with the 2.2–2.7 % VO2max CVs reported elsewhere [106, 116, 117]. Studies that found threshold measurements to be more reliable than VO2max appeared to have somewhat higher VO2max CV values of 4.7 % [115] and 8.5 % [113]. The VO2max CV of 8.5 %, reported by Lourenco et al. [113], was calculated from four maximal efforts ≥48 h apart and there was a decrease of 2.9 ml/kg per min in the group mean VO2max between the second and fourth trials. This suggests that the abnormally high variation in VO2max in that study could be attributed to accumulated fatigue and that participants did not complete all four trials in an equivalent physiological state. Were the findings of Lourenco et al. [113] to be set aside, it can be concluded that VO2max shows a typical CV of 1.9–4.7 % and that VO2max is more reliable than threshold measurements on most, but not all, occasions.
3.2.2 Relative Reliability of AerT and AnT
In comparison to HRmax and VO2max, the range of CVs reported for threshold measurements is large, spanning 1.5–10.4 % for AerT and 1.2–11.9 % for AnT (Table 1). This may be partly attributed to differences in protocol and study design, including, in some cases, the reliability with which investigators are able to identify threshold measurements by visual inspection [118, 119]. It is also apparent that the reliability of threshold measurements varies according to whether the threshold is reported as a workload, an HR, a VO2, or a blood lactate concentration. Of six examples [109, 111, 112, 115, 120] of a threshold reported according to the corresponding HR, speed or power output, and VO2, four found threshold HR to be the most reliable with CVs of 1.5–3.8 % [109, 111, 112, 115]. Furthermore, of nine examples [106, 109, 111–115, 117] reporting both threshold speed or power output and threshold VO2, six reported speed or power output to be the more reliable threshold measurement with speed or power output CVs of 1.7–5.9 % [106, 109, 111, 113, 115, 117]. When comparing the relative reliability of the AerT or AnT on the basis of the associated workload, Aunola and Rusko [111], Weltman et al. [115], and Dickhuth et al. [121] all found AnT to be the more reliable threshold measure with AnT versus AerT CV differences of 3.0 vs 3.5 %, 1.7 vs 3.0 %, and 2.6 % vs 5.3–5.6 %, respectively.
3.2.3 Relative Reliability of Blood Lactate Samples
Given that several of the methods of AerT and AnT determination or verification involve measuring blood lactate, it is important to mention the many sources of variation in blood lactate measurements. Blood lactate responses during exercise may be affected by factors such as prior exercise [120], the glycogen status of the participant [122], and ambient temperature [123, 124]. Furthermore, the lactate concentration measured may vary depending on the sampling site [125–127], sweat contamination, and the accuracy of the lactate analyzer. The portable Accusport® analyzer (Boehringer-Mannheim), for example, has a standard error of measurement of 0.3–0.5 mmol/l for duplicate samples during a single trial and a day-to-day standard error of measurement of 0.4 mmol/l when measuring the same lactate solution [128]. Random measurement error of this magnitude could have a significant effect on the identification of the MLSS (<1 mmol/l change in blood lactate between 10 min and the end of the exercise). As a measure of systematic error, the limits of agreement for measurements made using the Accusport® analyzer and a criterion measure of lactate concentration were +1.9 to −2.2 mmol/l [128]. The errors associated with measuring blood lactate are discussed in greater detail in other reviews [129–131].
3.2.4 Reliability of VO2R and HRR
The relative reliability of VO2R and HRR does not appear to have been directly investigated. Nevertheless, the influence of variation in both resting and maximal measurements on repeated determinations of VO2R and HRR would be expected to result in higher variation than that of VO2max or HRmax. In other words, VO2R reliability would incorporate both VO2max reliability (CV = 2–5 %; Table 1) and resting VO2 reliability (CV = 10%) [97], whereas HRR would incorporate both HRmax reliability (CV = 1–2 %; Table 1) and resting HR reliability (CV = 7–8 %) [132].
Another factor to consider is the reliability of the %VO2R–%HRR relationship given that these methods of exercise intensity prescription could be assumed to be equivalent based on previous findings [13, 14, 20]. In a recent review, da Cunha et al. [23] raised a number of evidence-based concerns for prescribing exercise intensity based on this relationship, including the influence of the incremental protocol on the %VO2R–%HRR relationship, the influence of resting VO2 measurements on the %VO2R–%HRR relationship, the stability of the %VO2R–%HRR relationship during prolonged exercise, and the consistency of the %VO2R–%HRR relationship across different exercise modes. The authors clearly demonstrated that the %VO2R–%HRR is not consistently reliable and researchers should consider verifying the %VO2R–%HRR relationship within their own context of exercise prescription.
3.2.5 Reliably Targeting a Relative Response
In a final word on reliability, it is advantageous to be able to monitor, during a particular exercise bout, whether the measured VO2, HR, or lactate response is in fact the target response prescribed for that bout. For example, 60 % of a 60-ml/kg per min VO2max would be a target VO2 of 36 ml/kg per min, and exercise at the MLSS workload would be expected to produce a stable blood lactate concentration rather than a blood lactate concentration that increases. With this in mind, both VO2 and HR can be monitored continuously and noninvasively during single exercise bouts, although only HR measurements would be practical for use in regular training sessions. The effect of small adjustments in workload can be observed within a short period and as a result it is comparatively easy to match the measured exercise intensity to the target exercise intensity. This is not the case when the target intensity is prescribed relative to a threshold concept. Blood lactate is monitored at discrete time points rather than continuously, and a longer period is required to observe the blood lactate response at a particular workload. If a threshold measurement has been verified, it might be argued that blood lactate monitoring is not necessary. However, the ability to verify both the anchor measurement and the target exercise intensity are practical points to consider for study design.
4 Summary and Conclusion
Based on a theoretical understanding of each anchor measurement, training prescribed relative to threshold measurements would be expected to elicit more comparable metabolic and respiratory responses between individuals than exercise prescribed relative to VO2max, HRmax, VO2R, or HRR. Possible consequences of comparable metabolic and respiratory responses include less variation in time to exhaustion during constant-intensity exercise, a more homogenous exercise stimulus at the molecular level, and less individual variation in the adaptive responses following a training program. However, many of these theoretical expectations have yet to be directly demonstrated. For example, there do not appear to be any studies describing individual variation in response to training prescribed relative to a threshold concept. In a similar way, we are not aware of any studies comparing the effect of method of relative exercise intensity prescription on the transcriptional and translation responses to single exercise bouts.
While there is a strong theoretical basis for using threshold-based exercise prescription, the challenges of determining thresholds in practice may partially explain why many researchers continue to favor the use of %VO2max, %HRmax, %VO2R, or %HRR. For instance, when derived from a blood lactate curve, neither the AerT nor the AnT can be assumed to pinpoint the true thresholds of metabolic response in all individuals without verification. Verification of threshold measures on an individual basis would require two or three additional visits to the laboratory and is highly uncommon. Nevertheless, failure to verify threshold measurements may create the same individual variation in blood lactate accumulation for which %VO2max and %HRmax have been criticized. It follows that VO2max and HRmax, which can be measured and verified within a single laboratory visit, have a definite practical, if not theoretical, advantage over threshold measurements for prescribing exercise intensity.
It can be concluded that none of the methods of relative exercise intensity prescription under discussion are without limitations and the most appropriate measurement for a research study may differ according to factors such as exercise intensity, number of participants, participant characteristics, and laboratory resources. For example, exercise prescribed at a %VO2max of moderate intensity would be less likely to elicit individual variation in blood lactate accumulation than exercise prescribed at a high %VO2max. Therefore, studies involving only moderate exercise intensity (e.g., ≤60 % VO2max) might reasonably choose %VO2max, %HRmax, %VO2R, or %HRR over threshold-based relative exercise intensity prescription. Furthermore, studies involving moderate-intensity exercise might favor %VO2R over %VO2max in order to place individuals at an equivalent intensity above rest—%VO2R and %VO2max go on to converge as exercise intensity increases. At higher exercise intensities, the importance of accounting for threshold measurements becomes more important. Nevertheless, it could be argued that it is more important to control exercise intensity relative to threshold measurements in participants with a range of exercise capacities compared to participants with similar exercise capacities. In terms of threshold verification, it could be argued that exercise intensity prescribed relative to unverified threshold measurements would be more likely to impact results in a study with a small number of participants compared to a study with a large number of participants. Furthermore, it is acknowledged that in some cases the time frame for a research study, the available resources, and other practical constraints may not allow for the most appropriate method of exercise intensity prescription. For example, it may not be feasible to verify threshold measurements for threshold measurement-based exercise prescription due to the overall testing burden on the participants. In these cases, the possibility of individuals exercising above and below threshold measurements could at least be acknowledged and considered when interpreting the study results.
As a final comment, even a brief overview of individual responses within a study allows for more balanced data interpretation and provides useful insight for subsequent studies. Therefore, irrespective of the method of relative intensity prescription used, future studies should emphasize the reporting of individual responses, particularly for studies involving small sample sizes.
References
Hills AP, Byrn NM, Ramage AJ. Submaximal markers of exercise intensity. J Sport Sci. 1998;16:S71–6.
Carvalho VO, Mezzani A. Aerobic exercise training intensity in patients with chronic heart failure: principles of assessment and prescription. Eur J Cardiovasc Prev and Rehabil. 2011;18(1):5–14.
Garber CE, Blissmer B, Deschenes MR, et al. American College of Sports Medicine position stand. Quantity and quality of exercise for developing and maintaining cardiorespiratory, musculoskeletal, and neuromotor fitness in apparently healthy adults: guidance for prescribing exercise. Med Sci Sports Exerc. 2011;43(7):1334–59.
Sedlock DA, Lee M-G, Flynn MG, et al. Excess postexercise oxygen consumption after aerobic exercise training. Int J Sport Nutr Exerc Metab. 2010;20(4):336–49.
Killgore GL, Coste SC, O’ Meara SE, et al. A comparison of the physiological exercise intensity differences between shod and barefoot submaximal deep-water running at the same cadence. J Strength Cond Res. 2010;24(12):3302–12.
Ferguson-Stegall L, McCleave E, Ding Z, et al. (2011) Aerobic exercise training adaptations are increased by postexercise carbohydrate-protein supplementation. J Nutr Metab. (epub 2011 June 9).
Van Proeyen K, Szlufcik K, Nielens H, et al. Beneficial metabolic adaptations due to endurance exercise training in the fasted state. J Appl Physiol. 2011;110(1):236–45.
McPhee JS, Williams AG, Perez-Schindler J, et al. Variability in the magnitude of response of metabolic enzymes reveals patterns of co-ordinated expression following endurance training in women. Exp Physiol. 2011;96(7):699–707.
Nordsborg NB, Lundby C, Leick L, et al. Relative workload determines exercise-induced increases in PGC-1alpha mRNA. Med Sci Sports Exerc. 2010;42(8):1477–84.
Katayama K, Goto K, Ishida K, et al. Substrate utilization during exercise and recovery at moderate altitude. Metabolism. 2010;59(7):959–66.
Donges CE, Duffield R, Drinkwater EJ. Effects of resistance or aerobic exercise training on interleukin-6, C-reactive protein, and body composition. Med Sci Sports Exerc. 2010;42(2):304–13.
Swain DP, Abernathy KS, Smith CS, et al. Target heart rates for the development of cardiorespiratory fitness. Med Sci Sports Exerc. 1994;26(1):112–6.
Swain DP, Leutholtz BC. Heart rate reserve is equivalent to %VO2 reserve, not to %VO2max. Med Sci Sports Exerc. 1997;29(3):410–4.
Swain DP, Leutholtz BC, King ME, et al. Relationship between % heart rate reserve and %VO2 reserve in treadmill exercise. Med Sci Sports Exerc. 1998;30(2):318–21.
Katch V, Weltman A, Sady S, et al. Validity of the relative percent concept for equating training intensity. Eur J Appl Physiol Occup Physiol. 1978;39(4):219–27.
Dwyer J, Bybee R. Heart rate indices of the anaerobic threshold. Med Sci Sports Exerc. 1983;15(1):72–6.
Meyer T, Gabriel HH, Kindermann W. Is determination of exercise intensities as percentages of VO2max or HRmax adequate? Med Sci Sports Exerc. 1999;31(9):1342–5.
Scharhag-Rosenberger F, Meyer T, Gässler N, et al. Exercise at given percentages of VO2max: heterogeneous metabolic responses between individuals. J Sci Med Sport. 2010;13(1):74–9.
Vollaard NBJ, Constantin-Teodosiu D, Fredriksson K, et al. Systematic analysis of adaptations in aerobic capacity and submaximal energy metabolism provides a unique insight into determinants of human aerobic performance. J Appl Physiol. 2009;106(5):1479–86.
Lounana J, Campion F, Noakes TD, et al. Relationship between %HRmax, %HR reserve, %VO2max, and %VO2 reserve in elite cyclists. Med Sci Sports Exerc. 2007;39(2):350–7.
Pollock ML, Gaesser GA, Butcher JD, et al. American College of Sports Medicine Position Stand. The recommended quantity and quality of exercise for developing and maintaining cardiorespiratory and muscular fitness, and flexibility in healthy adults. Med Sci Sports Exerc. 1998;30(6):975–91.
Cunha FA, Midgley AW, Monteiro WD, et al. The relationship between oxygen uptake reserve and heart rate reserve is affected by intensity and duration during aerobic exercise at constant work rate. Appl Physiol Nutr Metab. 2011;36(6):839–47.
Da Cunha FA, Farinatti PDTV, Midgley AW. Methodological and practical application issues in exercise prescription using the heart rate reserve and oxygen uptake reserve methods. J Sci Med Sport; 2011; 14(1):46–57.
Cunha FA, Midgley AW, Monteiro WD, et al. Influence of cardiopulmonary exercise testing protocol and resting VO(2) assessment on %HR(max), %HRR, %VO(2max) and %VO(2)R relationships. Int J Sports Med. 2010;31(5):319–26.
Gaskill SE, Bouchard C, Rankinen T, et al. %Heart rate reserve is better related to %VO2max than to %VO2 reserve: the HERITAGE Family Study. Med Sci Sport Exerc. 2004;36(5):S3.
Aellen R, Hollmann W, Boutellier U. Effects of aerobic and anaerobic training on plasma lipoproteins. Int J Sports Med. 1993;14(7):396–400.
Jenkins DG, Quigley BM. Endurance training enhances critical power. Med Sci Sports Exerc. 1992;24(11):1283–9.
Billat VL, Sirvent P, Lepretre P-M, et al. Training effect on performance, substrate balance and blood lactate concentration at maximal lactate steady state in master endurance-runners. Pflügers Archiv. 2004;447(6):875–83.
Vanhatalo A, Doust JH, Burnley M. A 3-min all-out cycling test is sensitive to a change in critical power. Med Sci Sports Exerc. 2008;40(9):1693–9.
Casaburi R, Storer TW, Sullivan CS, et al. Evaluation of blood lactate elevation as an intensity criterion for exercise training. Med Sci Sports Exerc. 1995;27(6):852–62.
Yoshida T, Suda Y, Takeuchi N. Endurance training regimen based upon arterial blood lactate: effects on anaerobic threshold. Eur J Appl Physiol Occup Physiol. 1982;49(2):223–30.
Morton JP, MacLaren DPM, Cable NT, et al. Time course and differential responses of the major heat shock protein families in human skeletal muscle following acute nondamaging treadmill exercise. J Appl Physiol. 2006;101(1):176–82.
Myers J, Ashley E. Dangerous curves. A perspective on exercise, lactate, and the anaerobic threshold. Chest. 1997;111(3):787–95.
Hopker JG, Jobson SA, Pandit JJ. Controversies in the physiological basis of the “anaerobic threshold” and their implications for clinical cardiopulmonary exercise testing. Anaesthesia. 2011;66(2):111–23.
Whipp BJ, Ward SA. The physiological basis of the “anaerobic threshold” and implications for clinical cardiopulmonary exercise testing. Anaesthesia. 2011; 66(11):1048–1049 (author reply 1049–50).
Coyle EF, Coggan AR, Hopper MK, et al. Determinants of endurance in well-trained cyclists. J Appl Physiol. 1988;64(6):2622–30.
Weltman A, Weltman J, Rutt R, et al. Percentages of maximal heart rate, heart rate reserve, and VO2peak for determining endurance training intensity in sedentary women. Int J Sports Med. 1989;10(3):212–6.
Weltman A, Snead D, Seip R, et al. Percentages of maximal heart rate, heart rate reserve and VO2max for determining endurance training intensity in male runners. Int J Sports Med. 1990;11(3):218–22.
Skinner JS, Gaskill SE, Rankinen T, et al. Evaluation of ACSM guidelines on prescribing exercise intensity for “quite unfit”: the HERITAGE Family Study. Med Sci Sports Exerc. 2004;36(5):S3.
Azevedo LF, Perlingeiro PS, Brum PC, et al. Exercise intensity optimization for men with high cardiorespiratory fitness. J Sports Sci. 2011;29(6):555–61.
Schnabel A, Kindermann W, Schmitt WM, et al. Hormonal and metabolic consequences of prolonged running at the individual anaerobic threshold. Int J Sports Med. 1982;3(3):163–8.
McLellan TM, Cheung KS. A comparative evaluation of the individual anaerobic threshold and the critical power. Med Sci Sports Exerc. 1992;24(5):543–50.
Van Schuylenbergh R, Vanden Eynde B, Hespel P. Correlations between lactate and ventilatory thresholds and the maximal lactate steady state in elite cyclists. Int J Sports Med. 2004;25(6):403–8.
Skinner JS, McLellan TH. The transition from aerobic to anaerobic metabolism. Res Q Exerc Sport. 1980;51(1):234–48.
Jones AM, Poole DC. Oxygen uptake dynamics: from muscle to mouth—an introduction to the symposium. Med Sci Sports Exerc. 2005;37(9):1542–50.
Mazzeo RS, Marshall P. Influence of plasma catecholamines on the lactate threshold during graded exercise. J Appl Physiol. 1989;67(4):1319–22.
Urhausen A, Weiler B, Coen B, et al. Plasma catecholamines during endurance exercise of different intensities as related to the individual anaerobic threshold. Eur J Appl Physiol Occup Physiol. 1994;69(1):16–20.
Pringle JSM, Jones AM. Maximal lactate steady state, critical power and EMG during cycling. Eur J Appl Physiol. 2002;88(3):214–26.
Jones AM, Wilkerson DP, DiMenna F, et al. Muscle metabolic responses to exercise above and below the “critical power” assessed using 31P-MRS. Am J Physiol Regul Integr Comp Physiol. 2008;294(2):R585–93.
Chwalbinska-Moneta J, Robergs RA, Costill DL, et al. Threshold for muscle lactate accumulation during progressive exercise. J Appl Physiol. 1989;66(6):2710–6.
McLellan TM, Jacobs I. Reliability, reproducibility and validity of the individual anaerobic threshold. Eur J Appl Physiol Occup Physiol. 1993;67(2):125–31.
Poole DC, Ward SA, Gardner GW, et al. Metabolic and respiratory profile of the upper limit for prolonged exercise in man. Ergonomics. 1988;31(9):1265–79.
Dekerle J, Baron B, Dupont L, et al. Maximal lactate steady state, respiratory compensation threshold and critical power. Eur J Appl Physiol. 2003;89(3–4):281–8.
Allen DG, Lamb GD, Westerblad H. Skeletal muscle fatigue: cellular mechanisms. Physiol Rev. 2008;88(1):287–332.
Abbiss CR, Laursen PB. Models to explain fatigue during prolonged endurance cycling. Sports Med. 2005;35(10):865–98.
Sjödin B, Jacobs I. Onset of blood lactate accumulation and marathon running performance. Int J Sports Med. 1981;2(1):23–6.
Komi PV, Ito A, Sjödin B, et al. Muscle metabolism, lactate breaking point, and biomechanical features of endurance running. Int J Sports Med. 1981;2(3):148–53.
Ivy JL, Withers RT, Van Handel PJ, et al. Muscle respiratory capacity and fiber type as determinants of the lactate threshold. J Appl Physiol. 1980;48(3):523–7.
Sjödin B, Jacobs I, Karlsson J. Onset of blood lactate accumulation and enzyme activities in m. vastus lateralis in man. Int J Sports Med. 1981;2(3):166–70.
Rusko H, Rahkila P, Karvinen E. Anaerobic threshold, skeletal muscle enzymes and fiber composition in young female cross-country skiers. Acta Physiol Scand. 1980;108(3):263–8.
Farrell PA, Wilmore JH, Coyle EF, et al. Plasma lactate accumulation and distance running performance. Med Sci Sports. 1979;11(4):338–44.
Bishop D, Jenkins DG, Mackinnon LT. The relationship between plasma lactate parameters, W peak and 1-h cycling performance in women. Med Sci Sports Exerc. 1998;30(8):1270–5.
Stratton E, O’Brien B, Harvey J, et al. Treadmill velocity best predicts 5000-m run performance. Int J Sports Med. 2008;30(1):40–5.
Hildebrandt AL, Pilegaard H, Neufer PD. Differential transcriptional activation of select metabolic genes in response to variations in exercise intensity and duration. Am J Physiol Endocrinol Metab. 2003;285(5):E1021–7.
Chen Z-P, Stephens TJ, Murthy S, et al. Effect of exercise intensity on skeletal muscle AMPK signaling in humans. Diabetes. 2003;52(9):2205–12.
Baar K. The signaling underlying FITness. Appl Physiol Nutr Metab. 2009;34(3):411–9.
Coffey VG, Hawley JA. The molecular bases of training adaptation. Sports Med. 2007;37(9):737–63.
Flück M. Functional, structural and molecular plasticity of mammalian skeletal muscle in response to exercise stimuli. J Exp Biol. 2006;209(Pt 12):2239–48.
McLellan TM, Skinner JS. The use of the aerobic threshold as a basis for training. Can J Appl Sport Sci. 1981;6(4):197–201.
Bouchard C, Rankinen T. Individual differences in response to regular physical activity. Med Sci Sports Exerc. 2001;33(6 Suppl):S446–51.
Sisson SB, Katzmarzyk PT, Earnest CP, et al. Volume of exercise and fitness nonresponse in sedentary, postmenopausal women. Med Sci Sports Exerc. 2009;41(3):539–45.
Kohrt WM, Malley MT, Coggan AR, et al. Effects of gender, age, and fitness level on response of VO2max to training in 60–71 year olds. J Appl Physiol. 1991;71(5):2004–11.
Hautala AJ, Kiviniemi AM, Mäkikallio TH, et al. Individual differences in the responses to endurance and resistance training. Eur J Appl Physiol. 2006;96(5):535–42.
Prud’homme D, Bouchard C, Leblanc C, et al. Sensitivity of maximal aerobic power to training is genotype-dependent. Med Sci Sports Exerc. 1984;16(5):489–493.
Scharhag-Rosenberger F, Walitzek S, Kindermann W, et al. Differences in adaptations to 1 year of aerobic endurance training: individual patterns of nonresponse. Scand J Med Sci Sports. 2010;22(1):113–8.
Karavirta L, Häkkinen K, Kauhanen A, et al. Individual responses to combined endurance and strength training in older adults. Med Sci Sports Exerc. 2010;31:484–90.
Bouchard C. Genomic predictors of trainability. Exp Physiol. 2012;97(3):347–52.
Bouchard C, An P, Rice TK, et al. Familial aggregation of VO2max response to exercise training: results from the HERITAGE Family Study. J Appl Physiol. 1999;87(3):1003–8.
Bouchard C, Sarzynski MA, Rice TK, et al. Genomic predictors of the maximal O2 uptake response to standardized exercise training programs. J Appl Physiol. 2011;110(5):1160–70.
Rice TK, An P, Gagnon J, et al. Heritability of HR and BP response to exercise training in the HERITAGE Family Study. Med Sci Sports Exerc. 2002;34(6):972–9.
Rico-Sanz J, Rankinen T, Joanisse DR, et al. Familial resemblance for muscle phenotypes in the HERITAGE Family Study. Med Sci Sports Exerc. 2003;35(8):1360–6.
Karoly HC, Stevens CJ, Magnan RE, et al. Genetic influences on physiological and subjective responses to an aerobic exercise session among sedentary adults. J Cancer Epidemiol. (epub 2012 Jul 29).
Bentley DJ, Newell J, Bishop D. Incremental exercise test design and analysis: implications for performance diagnostics in endurance athletes. Sports Med. 2007;37(7):575–86.
Poole DC, Wilkerson DP, Jones AM. Validity of criteria for establishing maximal O2 uptake during ramp exercise tests. Eur J Appl Physiol. 2008;102(4):403–10.
Kirkeberg JM, Dalleck LC, Kamphoff CS, et al. Validity of 3 protocols for verifying VO2 max. Int J Sports Med. 2011;32(4):266–70.
Day JR, Rossiter HB, Coats EM, et al. The maximally attainable VO2 during exercise in humans: the peak vs. maximum issue. J Appl Physiol. 2003;95(5):1901–7.
Midgley AW, Carroll S. Emergence of the verification phase procedure for confirming “true” VO2max. Scand J Med Sci Sports. 2009;19(3):313–22.
Pettitt RW, Clark IE, Ebner SM, et al. Gas exchange threshold and VO2max testing for athletes: an update. J Strength Cond Res. 2013;27(2):549–55.
Midgley AW, Carroll S, Marchant D, et al. Evaluation of true maximal oxygen uptake based on a novel set of standardized criteria. Appl Physiol Nutr Metab. 2009;34(2):115–23.
Scharhag-Rosenberger F, Carlsohn A, Cassel M, et al. How to test maximal oxygen uptake: a study on timing and testing procedure of a supramaximal verification test. Appl Physiol Nutr Metab. 2011;36(1):153–60.
Astorino TA, White AC. Assessment of anaerobic power to verify VO2max attainment. Clin Physiol Funct Imaging. 2010;30(4):294–300.
Dalleck LC, Astorino TA, Erickson RM, et al. Suitability of verification testing to confirm attainment of VO2max in middle-aged and older adults. Res Sport Med. 2012;20(2):118–28.
Robergs RA, Landwehr R. The surprising history of the “HRmax = 220 – age” equation. J Exerc Physiol. 2002;5(2):1–10.
Fox SM, Naughton JP, Haskell WL. Physical activity and the prevention of coronary heart disease. Ann Clin Res. 1971;3(6):404–32.
Swain DP, Franklin BA. VO(2) reserve and the minimal intensity for improving cardiorespiratory fitness. Med Sci Sports Exerc. 2002;34(1):152–7.
Brawner CA, Keteyian SJ, Ehrman JK. The relationship of heart rate reserve to VO2 reserve in patients with heart disease. Med Sci Sports Exerc. 2002;34(3):418–22.
Compher C, Frankenfield D, Keim N, et al. Best practice methods to apply to measurement of resting metabolic rate in adults: a systematic review. J Am Diet Assoc. 2006;106(6):881–903.
Rusko H, Luhtanen P, Rahkila P, et al. Muscle metabolism, blood lactate and oxygen uptake in steady state exercise at aerobic and anaerobic thresholds. Eur J Appl Physiol Occup Physiol. 1986;55(2):181–6.
Aunola S, Rusko H. Does anaerobic threshold correlate with maximal lactate steady-state? J Sports Sci. 1992;10(4):309–23.
Beneke R. Methodological aspects of maximal lactate steady state—implications for performance testing. Eur J Appl Physiol. 2003;89(1):95–9.
Smith CGM. The relationship between critical velocity, maximal lactate steady-state velocity and lactate turnpoint velocity in runners. Eur J Appl Physiol. 2001;85:19–26.
Beneke R, Von Duvillard SP. Determination of maximal lactate steady state response in selected sports events. Med Sci Sports. 1996;28(2):241–6.
McLellan TM, Cheung KS, Jacobs I. Incremental test protocol, recovery mode and the individual anaerobic threshold. Int J Sports Med. 1991;12(2):190–5.
Beneke R. Anaerobic threshold, individual anaerobic threshold, and maximal lactate steady state in rowing. Med Sci Sports Exerc. 1995;27(6):863–7.
Cheng B, Kuipers H, Snyder AC, et al. A new approach for the determination of ventilatory and lactate thresholds. Int J Sports Med. 1992;13(7):518–22.
Zhou S, Weston SB. Reliability of using the D-max method to define physiological responses to incremental exercise testing. Physiol Meas. 1997;18(2):145–54.
Hopkins WG. Measures of reliability in sports medicine and science. Sports Med. 2000;30(1):1–15.
Hopkins WG, Schabort EJ, Hawley JA. Reliability of power in physical performance tests. Sports Med. 2001;31(3):211–34.
Jensen K, Johansen L. Reproducibility and validity of physiological parameters measured in cyclists riding on racing bikes placed on a stationary magnetic brake. Scand J Med Sci Sports. 1998;8(1):1–6.
Bingisser R, Kaplan V, Scherer T, et al. Effect of training on repeatability of cardiopulmonary exercise performance in normal men and women. Med Sci Sports Exerc. 1997;29(11):1499–504.
Aunola S, Rusko H. Reproducibility of aerobic and anaerobic thresholds in 20–50 year old men. Eur J Appl Physiol Occup Physiol. 1984;53(3):260–6.
Weston SB, Gabbett TJ. Reproducibility of ventilation of thresholds in trained cyclists during ramp cycle exercise. J Sci Med Sport. 2001;4(3):357–66.
Lourenço TF, Martins LEB, Tessutti LS, et al. Reproducibility of an incremental treadmill VO2max test with gas exchange analysis for runners. J Strength Cond Res. 2011;25(7):1994–9.
Wisén AGM, Wohlfart B. A refined technique for determining the respiratory gas exchange responses to anaerobic metabolism during progressive exercise—repeatability in a group of healthy men. Clin Physiol Funct Imaging. 2004;24(1):1–9.
Weltman A, Snead D, Stein P, et al. Reliability and validity of a continuous incremental treadmill protocol for the determination of lactate threshold, fixed blood lactate concentrations, and VO2max. Int J Sports Med. 1990;11(1):26–32.
Lamberts RP, Swart J, Richard W, et al. Measurement error associated with performance testing in well-trained cyclists: application to the precision of monitoring changes in training status. Int Sports Med J. 2009;10(1):33–44.
Amann M, Subudhi AW, Walker J, et al. An evaluation of the predictive validity and reliability of ventilatory threshold. Med Sci Sports Exerc. 2004;36(10):1716–22.
Yeh MP, Gardner RM, Adams TD, et al. “Anaerobic threshold”: problems of determination and validation. J Appl Physiol. 1983;55(4):1178–86.
Gladden LB, Yates JW, Stremel RW, et al. Gas exchange and lactate anaerobic thresholds: inter- and intraevaluator agreement. J Appl Physiol. 1985;58(6):2082–9.
Coen B, Urhausen A, Kindermann W. Individual anaerobic threshold: methodological aspects of its assessment in running. Int J Sports Med. 2001;22(1):8–16.
Dickhuth HH, Yin L, Niess A, et al. Ventilatory, lactate-derived and catecholamine thresholds during incremental treadmill running: relationship and reproducibility. Int J Sports Med. 1999;20(2):122–7.
Podolin DA, Munger PA, Mazzeo RS. Plasma catecholamine and lactate response during graded exercise with varied glycogen conditions. J Appl Physiol. 1991;71(4):1427–33.
Parkin JM, Carey MF, Zhao S, et al. Effect of ambient temperature on human skeletal muscle metabolism during fatiguing submaximal exercise. J Appl Physiol. 1999;86(3):902–8.
Flore P, Therminarias A, Oddou-Chirpaz MF, et al. Influence of moderate cold exposure on blood lactate during incremental exercise. Eur J Appl Physiol Occup Physiol. 1992;64(3):213–7.
Dassonville J, Beillot J, Lessard Y, et al. Blood lactate concentrations during exercise: effect of sampling site and exercise mode. J Sports Med Phys Fitness. 1998;38(1):39–46.
el-Sayed MS, George KP, Dyson K. The influence of blood sampling site on lactate concentration during submaximal exercise at 4 mmol/l lactate level. Eur J Appl Physiol Occup Physiol. 1993;67(6):518–22.
Robergs RA, Chwalbinska-Moneta J, Mitchell JB, et al. Blood lactate threshold differences between arterialized and venous blood. Int J Sports Med. 1990;11(6):446–51.
Bishop D. Evaluation of the Accusport lactate analyser. Int J Sports Med. 2001;22(7):525–30.
Jacobs I. Blood lactate. Implications for training and sports performance. Sports Med. 1986;3(1):10–25.
Swart J, Jennings C. Use of blood lactate concentration as a marker of training. SA J Sports Med. 2004;16(3):1–5.
Beneke R, Leithäuser RM, Ochentei O. Blood lactate diagnostics in exercise testing and training. Int J Sports Physiol Perform. 2011;6:8–24.
Stanforth PR, Gagnon J, Rice T, et al. Reproducibility of resting blood pressure and heart rate measurements. The HERITAGE Family Study. Ann Epidemiol. 2000;10(5):271–7.
Acknowledgments
This research was supported financially by the Deutscher Akademischer Austausch Dienst (DAAD), the Ernst and Ethel Eriksen Trust, and the University of Cape Town. The authors declare that there was no conflict of interest in the preparation of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mann, T., Lamberts, R.P. & Lambert, M.I. Methods of Prescribing Relative Exercise Intensity: Physiological and Practical Considerations. Sports Med 43, 613–625 (2013). https://doi.org/10.1007/s40279-013-0045-x
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40279-013-0045-x