Abstract
Cold plasma (CP) is a novel non-thermal technology and has marked a new trend in both the sectors of agriculture and food processing for their safety and quality. This review describes an overview on the effects of CP with respect to microbial decontamination, enzyme denaturation, pesticide degradation, food allergens, polyphenols, food packaging, and many other physiological processes. Furthermore, mechanisms and applications involving different aspects related to cold plasma are discussed. The recent studies on cold plasma referred mainly with the interactions of reactive species and target food commodity. Finally, the future prospects and challenges that could help in rendering substantial benefits of CP to the food industries and researchers, particularly in upscaling this eco-friendly technology, are discussed.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over a decade, non-thermal plasma has gained interest by food processing researchers. The reason for this could be its economical, eco-friendly, and versatile performance. At present, food safety is a major concern, and this could be more useful if maintained without affecting its nutritional, sensorial, and shelf-life properties. In this regard, non-thermal plasma possesses all the features to process the food economically without affecting their properties [1]. Let us go to the basics of plasma. When a gaseous substance is subjected to a high level of energy, it transforms into an ionized state of matter and is known as “plasma.” The term “plasma” was first coined by Irving Langmuir in 1927 [2]. All types of plasma are basically ionized gaseous entity consisting of variety of elements, such as electrons, photons, ions, and free radicals. Elements like photons and electrons constitute the “light” species, and the rest of the elements are considered as “heavy” species [3,4,5]. On the basis of the temperature, there are two types of plasmas: non-thermal and thermal. Thermal plasma is generated under high power (≈50 MW) and extremely high pressures (≥ 105 Pa). Thermal plasma exhibits a thermodynamic equilibrium between the “heavy” species and the electrons. It ensures an overall consistent plasma temperature [4, 6]. On the other hand, non-thermal plasma can be obtained under low pressures and low power. These plasmas are also called non-equilibrium plasma in which the electrons exhibit much higher temperature compared to the gas at macroscopic level [4]. Therefore, no thermodynamic equilibrium is observed between the “heavy” species and the electrons. In this review, cold plasma and non-thermal plasma have been used interchangingly for low-temperature plasma.
Food consumption provides nutrition and development to an organism. In the current situation, food quality and safety have been a major concern for the food industry and food researchers. Therefore, tackling some food-related issues is the need of hour-like microbial contamination or any kind of enzymatic degradation in perishable food products [7, 8]. Moreover, there have been several studies on the existence of various types of food contaminants, such as mycotoxin [9,10,11], heavy metals [12, 13], and polycyclic aromatic hydrocarbons [14]. In this regard, pesticides and allergens deserve a special note on the account of exhibiting the most prominent effects among all the food contaminants [15,16,17,18,19]. Previously, several thermal techniques have been used for food processing and sterilizing, such as freezing, drying, and heating [20,21,22]. Other sterilization approaches, such as chemical treatments, are often tedious and leave behind the toxic residues [23]. Furthermore, in terms of food packaging, several conventional substances such as papers and metals have been replaced by polymers [24]. In addition to providing functional characteristics similar to those of the conventional substances, the polymeric substances give more flexibility, are essentially inert, much cheaper, and exhibit lower specific weight. For food packaging, multi-layered materials are in high demand. However, the production of multi-layered polymers necessitates the surface sterilization or modification of these materials. The surfaces of polymeric materials are usually hydrophobic and have low surface energy [25]. This characteristic renders the conventional strategies useless during surface sterilization of the polymers.
Cold plasma (CP) technique employs versatile application. CP has grabbed lot of awareness in the field of food safety and processing. It was initially used for other industries such as electronic and polymer industries. CP was used to enhance the surface energy of the materials, properties of paper and glass, and printing and adhesion properties of polymers [4, 26]. However, it has recently been recognized as a potentially useful in the domain of food safety and processing. It offers wide range of applications along with very short processing time without leaving any toxic residues [27]. Several researchers have shown that the presence of reactive species within the plasma makes it a good medium for the inactivation of microbial species. Therefore, sterilization using plasma would not only reduce the surface contamination of the food products but also increases their shelf-life [8, 28,29,30,31,32,33,34,35]. There are handful of studies describing the applications of cold plasma to imrpove food quality through enzyme degradation [36,37,38,39,40]. In addition, CP has been reported to enhance the characteristics of various food packaging materials [24, 41,42,43]. CP has also been reported to enhance several functional aspects of food products, such as seed germination [44,45,46], physico-chemical properties of grains [47, 48], hydrogenation of vegetable oils [49, 50], inactivation of anti-nutritional factors [51], and high-quality mung bean sprout [52].
Therefore, this article gives a synopsis on recent developments in the application of cold plasma (CP) for food processing industry in terms of food safety and quality maintenance. It includes background of plasma generation and its sources as well as types of setups and apparatus in current use. Mechanism and application of non-thermal plasma in different approaches of food processing are discussed for different targets. Furthermore, limitations and future prospects of CP have also been discussed. The recent understanding and potential use of CP, a green and eco-friendly technology, will provide new opportunities for boosting and sustaining food sector.
Basics of Cold Plasma Technology
Plasma Generation
Usually, plasma is produced by the application of energy to a gaseous substance. The energy can be applied via different approaches, such as electric current (direct or alternating), thermal energy, magnetic fields, microwave, or radiofrequencies. Such energy sources can impart energy to the gaseous atoms and molecules by, essentially, increasing the kinetic energy of the electrons within them. Such an increase in the kinetic energies of the electrons causes them to move from lower energy states to higher energy states, which leads to the generation of ions. In addition, it results in more frequent collisions between the electrons and the “heavy species” that release several types of radiations [53]. Although several sources could be employed for the energy input in plasma generation for food processing, an electrical discharge is preferred to produce the non-thermal plasma [54]. This is because the electric discharge approach helps in better regulation of the plasma temperature. Low temperature is the primary requirement of the plasma for use in food products processing [4]. The type of gas employed for plasma production as well as the frequency and magnitude of the electric current chiefly determine the type of active species present in it [55]. Plasma can be produced via various sources, such as corona discharge, dielectric barrier discharge (DBD), gliding arc discharge (GAD), microwave frequency, and radio frequency. However, with respect to food products and food processing industry, jet plasma and the plasma generated from DBD are the most commonly used plasmas [56].
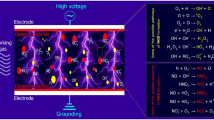
Broadly, plasma works on the principles of ions collision in the substance without generating heat. On the application of electric energy between two electrodes, the kinetic energy of the gas molecules is increased. During the process, electrons are released from the cathode surface and accelerated towards the anode. Hence, ionization of gas molecules continues till the inelastic collision reached. In the process, dissociation of molecules may happen and releases the ions (Fig. 1). Hence, the process would bring out the changes in the materials under consideration.
Plasma Characteristics
As already mentioned, the intensity and the frequency of the electric current primarily determine the energy of the plasma and the type of active species present in it [1, 4]. Another important factor involved in the determination of the plasma characteristics is the composition of the source gas. The type of gas used decides the type of active species produced. Modified atmospheric packaging is extensively used to avoid food contamination. Misra et al. [57] reported a 3-log reduction in strawberries upon plasma treatment in sealed package containing oxygen, nitrogen, and carbon-dioxide. Moreover, type of exposure (direct or indirect) also defines the effect of generated reactive species [4, 55].
Target Characteristics
With respect to food safety, the antimicrobial effects of plasma have been studied extensively. The internal characteristics of the microbes are majorly influencing the efficiency of the plasma treatment. Different species and strains of microbes exhibit variable sensitivities to plasma exposure [58, 59]. Furthermore, gram-positive bacteria exhibit higher resistance to plasma treatment compared to gram-negative bacteria [58, 59]. Sporulated bacteria also exhibit higher resistance against the plasma treatment. Liang et al. [60] reported higher resistance of fungi to plasma because of the chitin present in their cell walls, and is imparting them more rigidity.
Other Factors
Several surrounding physical factors also determine the efficiency of plasma treatment, including the pH of the medium, temperature, relative humidity, and treatment time. The presence of humidity usually enhances the impact of plasma due to the formation of higher levels of hydroxyl radicals. Furthermore, solid and liquid matrices react differently against the plasma treatment. In fact, different matrices of the same state show variable interactions with the plasma species. For instance, plasma treatment can decontaminate an agar plate or a filter membrane more rapidly compared to a fruit surface [59].
Augmentation of Plasma Efficiency
All the above factors seem to be affecting the efficiency of plasma treatment and open up a wide domain of approaches to enhance the effect of plasma. Moreover, several techniques could be used in conjugation with plasma treatment to further improve the impact of plasma.
In general, food products are packaged under different mixtures of gases, which help in enhancing the shelf life of food products, minimize microbial contamination, and act as a preservative. Since the surrounding environment of the target plays an important role during plasma interaction, exposure to CP in the presence of these mixtures of gases has been exploited and found to enhance the sterilization efficiency of plasma [57, 61, 62]. Furthermore, the pH of the medium could be altered to enhance the plasma sterilization efficiency. For instance, use of sanitizers, plasma-activated water (PAW), and essential oils, in conjugation with CP treatment, has been shown to enhance CP efficacy [63, 64]. Moreover, Mehta et al. [62] also reported enhancement in polyphenolic components in strawberry juice when cold plasma processing was coupled with hydrothermal treatment. Furthermore, external magnetic fields have also been reported to enhance the efficacy of plasma by increasing its density [65].
Types of Plasma
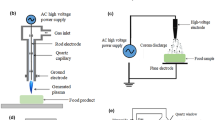
Plasma consists of several species that are present either in excited or in their fundamental state but with an overall neutral charge [66]. CP is a type of non-thermal plasma that can be generated via various approaches at both atmospheric pressure and reduced pressure. For utilization in the food industry, CP at atmospheric pressure is preferred to minimize the risk of degradation of food products. Schematic diagram of different types of plasma is shown in Fig. 2.
Dielectric Barrier Discharge (DBD)
DBD setup includes two metal electrodes kept close to each other and separated by a dielectric material. Commonly used dielectric materials include quartz, ceramic, polymer, or plastic [67]. The gap between the discharge material ranges from 100 mm to a few centimeters. An alternating current is emitted by the electrodes to generate plasma [68]. The dielectric barrier essentially acts as a stabilizer that helps to create several micro-discharges [56]. Recently, a novel model of DBD plasma, known as “in-house” or “encapsulated” plasma has been employed. In the latter, the food for sterilization is enclosed in a plastic packaging material and kept in place of the dielectric materials [59, 69, 70]. DBD approach offers several advantages such as simple design, flexibility in terms of choice of gas and size of electrodes, and uniform discharge ignition. However, DBD plasma is produced at high ignition voltage of 10 kV and, hence, requires technical expertise and caution. A high level of energy is required to sustain DBD plasma, produced at atmospheric pressure. DBD plasma has also been used for UV generation, CO2 lasers, and ozone generation [4].
The breakdown voltage as a function of pressure and distance between the electrode is represented by Eq. (1).
where \(C=log\left\{\frac{A}{log\ \left(1+1/y\right)}\right\}\) and VB = Ed, the total voltage applied between two electrodes. A and B are the constants depending on the gas used. The secondary emission coefficient (γ) depends on the material of cathode, the state of its surface, type of gas, and reduced electric field.
Gliding Arc Discharge (GAD)
GAD is created using two or more diverging metal electrodes and operates at a high potential difference (~9 kV). Approximately, 100 mA of electric current is passed through the electrodes, which leads to an arc formation in the narrower region between the electrodes. The inlet gas, carrying the humid air, flows from the narrow to the wider region between the electrodes and causes the arc to glide along with it. Based on the conditions, GAD could produce both thermal and non-thermal plasma at atmospheric pressure. GAD setup could be used to produce a high concentration of short-lived active species by applying high electric power [71]. GAD can also be used to produce high density plasma. Since there is no thermodynamic equilibrium between electrons and other plasma components, it is classified as non-thermal plasma [3]. GAD plasma can be used for both liquid and surface sterilization. It has previously been used for organic compounds degradation, bacterial decontamination, and water purification [4, 72]. The Arrhenius equation used to express the radical reaction rate and describe the effective rate of associative ionization in the plasma is represented by Eq. (2).
where τi is a pre-exponential factor, Ea is the effective activation energy, Ru is the universal gas constant, and Tg is the gas temperature.
Corona Discharge Plasma
Corona discharge plasma refers to the plasma that is produced when an ample amount of electric field around a sharp electrode ionizes the electrons in the atoms or molecules of surrounding gases [6]. It is only produced at high voltage, and its applications are limited to non-homogenous medium. However, its design is simple and relatively less costly. This technique has been employed previously for the surface sterilization, microbe elimination, and electro-precipitation [4, 73]. Corona discharge gives the current in the gas, and the total current flowing between the electrodes in the discharge region is described by Eq. (3).
where Iion is the ionic current component, and Idisp is the displacement current component.
Plasma Jet
The plasma jet device consists of two concentric electrodes, and a gas or a mixture of gases is flown between the electrodes. The inner electrode is subjected to a high radio frequency (~13.56 MHz), and a potential difference of 100–250 V is maintained between the electrodes that leads to ionization of the gas. The ionized gas molecules are directed out through a nozzle towards the surface of a food product located at a distance of few millimeters [28, 56, 74]. The ionized gas flows out through the nozzle in the form of a jet and hence, the name plasma jet. Plasma jet may also be generated by applying low-frequency kHz, nanosecond pulses, etc.
Capacitively Coupled Plasma (CCP)
CCP is most commonly used at the industrial level. CCP setup consists of two closely placed metal electrodes in a gas-filled chamber. One of the electrodes is attached to a radio frequency power supply and the other is grounded. The potential difference between the electrodes leads to the ionization of the gas. Since this setup resembles a capacitor, the plasma produced by it is termed as capacitively coupled plasma [75, 76]. CCP has a wide range of applications [24, 77].
Others
Inductively Coupled Plasma (ICP)
ICP refers to the plasma that is produced by the ionization of gas using the energy imparted by the electric current which is produced via electromagnetic induction [78]. Structurally, ICP setup is broadly classified into two categories: cylindrical and planar. In planar design, a spiral-shaped coil of flat metal is used as an electrode. In cylindrical design, the metal electrode is in the shape of a helical spring. With the passing of an electric current through the electrode, a magnetic field is created that induces the electric current into the target gas and ionizes it into plasma. Since the electric current is produced in the gas via a magnetic field, there is no need for the electrodes to be in direct contact with the gas and, hence, the electrodes are placed outside the reaction chamber [24].
Microwave Plasma/Electron Cyclotron Resonance Plasma (MP/ECR Plasma)
The MP plasma is generated using electromagnetic waves with high frequencies. Contrary to the electrode-based methods, a magnetron is used to produce microwave discharges in a process chamber. The heat produced by the microwaves causes ionization of the gas molecules. The major advantage of MP is the non-requirement of electrodes that allows ionization of the gas in free air and reduced the level of gas required to produce the plasma. However, the main limitation of ECR is that it cannot be used over a large area unless an array of discharges is used [4, 79]. Some basic magnetic circuits can be used are horseshoe magnet with iron keeper (low-reluctance circuit), horseshoe magnet with no keeper (high-reluctance circuit), electric motor (variable-reluctance circuit), and pickup cartridge (variable-reluctance circuits).
ECR plasma is a type of MP, produced by using microwaves with 2.45 GHz [80]. Electrons trajectory is in the form of a vertical spiral along the magnetic field lines. In ECR design, the electrons flow along the magnetic lines in a vertical spiral and ionize the gas molecules to produce plasma. This type of plasma is more efficient for surface treatments, such as surface deposition, surface functionalization, and surface etching [24, 81, 82].
Basically, an ECR and microwave ion source comprises of a multimode cavity. This cavity serves as the plasma generation and containment cavity. In the plasma drifts down the axial magnetic field gradient, the electrons are resonantly excited by the high-frequency field at a frequency explained through the following Eq. (4).
In this, ωRF is the excitation frequency, ωc is the electroncyclotron frequency, e is the magnitude of the electronic charge, B is the magnetic flux density, and me is the electron mass.
For more complete description of these phenomena, please see the following articles [83,84,85].
Mechanism and Applications of Plasma
All the entities present in the plasma, whether charged or neutral, play a significant role in its action. Apart from physical particles (ions, free radicals, electrons), plasma also comprises of radiations, such as UV. For various applications of the plasma, different plasma components are needed. Various hypotheses have been given to explain the mechanism of action by reactive species generated by cold plasma. For instance, cold plasma generates reactive and charged particles that induce numerous chemical reactions owing to the possession of sufficient electrical energy to break covalent bonds that lead to the breakdown of cell membrane via hydrolysis [86]. Another mechanism could involve the erosion of tissues and release of the bioactives accumulated in central vacuoles of guard cells under the effect of reactive oxygen species such as ·OH, O, and O2 [62, 87]. However, the mechanism of action of each plasma component remains more or less similar (Fig. 3). Applications of cold plasma are discussed under the following headings, viz, microbial inactivation, enzymes inactivation, stability of polyphenols, pesticide degradation, food allergen degradation, food packaging, effect of CP on starch, vitamins, and lipids, and seed germination.
Microbial Inactivation
During microbial decontamination, the cytoplasmic membrane of the microbe plays a crucial role. The efficiency of sterilizing agents and techniques majorly depends on their ability to penetrate the cytoplasmic membrane of the microbes and eliminate them. Interestingly, in terms of disruption of the cytoplasmic membrane, the oxidizing potential of the sterilization agent has been shown to be very crucial [33]. Therefore, the oxidizing agents in the plasma play a critical role in eliminating the microbes. In addition, several mechanical effects of plasma have also been reported to be mediated by the free radicals present in it, such as OH• and NO• radicals [53, 88, 89]. During plasma treatment, several such free radicals interact with the microbial cells and cause surface lesions. Due to the high frequency of the formation of these lesions, the cell does not get enough time to repair itself and get destroyed. This process is termed as “etching” [90]. The most widely accepted mechanism of plasma-induced DNA damage includes the formation of reactive oxidative species (ROS) by plasma elements in the microbial cells that damage their DNA molecules. The major plasma-induced ROS include H2O2, superoxide anion, and hydroxyl radicals [53]. The most prominent mechanism of plasma-induced microbial destruction includes the reaction of plasma elements with the water molecules inside the microbial cells that leads to the generation of hydroxyl radicals and responsible for most of the DNA damage [53]. Studies have also proposed that the accumulation of charged particles within the microbial cells induces the apoptosis, electroporation, and electrostatic disruption [31, 58].
Several studies have demonstrated the antimicrobial effects of CP [34, 91,92,93,94]. Recently, CP treatment has been reported for decontamination of tofu [28] and black pepper grain [91]. Moreover, reduction in microbial content has also been reported in chicken breast [70] and pork jerky [35]. Also, decontamination of dried squid has been performed using corona discharge plasma [73]. A study has also revealed the decontaminating effects of microwave-induced CP on onion powder without any visible effects on their sensory characteristics [95]. Wan et al. [96] have successfully used the CP technique for the decontamination of egg shells. Similarly, microwave-induced CP has been used to eliminate E. coli from lettuce with a 90% efficiency [97] and from radish [98].
As mentioned above, the efficiency of the plasma depends on the sample characteristics as well. CP is much more effective against Botrytis cinerea compared to ozone or UV-C exposure [99]. CP showed effective decontamination of fresh produce within 10 s of exposure, while the same efficacy of CP was attained for decontamination of strawberries after 300 s of exposure [100]. The in-package sterilization technique has been shown to be highly advantageous because the sealed sample can be directly exposed to the plasma without any leftover contamination after the process [33, 101, 102]. Indirect air plasma, such as plasma jet, has been used to create plasma-activated water (PAW) that has an acidic pH and contains several active species [103]. PAW, in turn, has been shown to exhibit antimicrobial effects [104]. Synergistic effects of pH and plasma species have been proposed to be responsible for the non-thermal sterilization by PAW [105]. Application of plasma-activated water (PAW) has been documented for food decontamination [106]. Some antimicrobial effects of CP are listed in Table 1.
Enzymes Inactivation
Majority of enzymes found in the food products are proteinaceous in nature. Hence, the mechanisms of action of CP on enzymes and other proteins are similar. Plasma-induced changes in the protein structure are primarily attributed to the active plasma species. For instance, protein inactivation induced by helium/oxygen plasma is mainly due to nitric oxide and atomic oxygen. Atomic oxygen in the plasma is able to remove the hydrogen from the protein backbone. This has led to the generation of radical sites and hence the cleavage of polypeptide chains [38]. Hydroxyl, hydroperoxy (HO2), nitric oxide, and superoxide anion radicals in the plasma could also modify the side-chains and aromatic rings of amino acids, leading to enzyme inactivation [107]. While the oxygen-based plasma species can cause structural changes in protein by converting C–N, N–H, and C–H bonds to NO2, H2O, and CO2, respectively. Plasma species such as O2−, H3O+, and O2+ are able to increase the polarity of zein molecules, a major protein found in maize [108].
Enzymatic browning has been a major problem in the food industry. CP has been used for the inactivation of several enzymes, such as polyphenol oxidase (PPO), pectin methylesterase (PME), superoxide dismutase (SOD), peroxidase (POD), catalase (CAT), lysozyme, alkaline phosphatase, a-chymotrypsin, and lipase [7]. Recently, inactivation of peroxidase enzyme in green coconut juice and amazonia juice has been reported upon CP treatment [38, 109, 110]. Moreover, lipase and lipoxygenase enzyme in wheat germ have been reported to be inactivated by CP [40]. Also, CP can be used for the extension of the storage time of potatoes and apples by inactivation of POD and PPO enzymes [36]. Furthermore, mild inactivation of PME, a cell wall-bound enzyme, has been documented using DBD in fresh-cut melon [39]. Their results have concluded that different enzymes are exhibiting different sensitivities to CP based on their structures. A significant decrease in alkaline phosphatase activity was observed within seconds after treatment with CP [111]. Similarly, a 70% and 10% decrease in PPO activity has been documented in the CP-treated guava pulp and whole fruit, respectively [53]. However, CP treatment is not effective for all types of enzymes. CAT and SOD activities were increased upon CP treatment in blueberries [99], while PAW was inefficient for the inactivation of the SOD enzyme in button mushrooms [112]. Some food enzymes affected by CP treatment are listed in Table 2.
Stability of Polyphenols
Antioxidants pose a problem during plasma treatment owing to the fact that they exhibit the opposite action to that of free radicals. They protect the cells against reactive oxygen species (ROS). However, the ozone and hydroxyl radicals present in the plasma could degrade flavonoid compounds [62]. Similarly, an increase in the level of polyphenols was observed on the treatment of sour cherry marasca juice with CP [87].
Studies have shown a decrease in the level of phenolic compounds in various food products such as orange juice, lamb’s lettuce, and white grape juice after treatment with CP [113, 114]. In contrast, de Castro et al. [110] and Rana et al. [115] applied cold plasma and reported an increase in phenolic compounds in camu-camu juice and strawberry fruit, respectively. Also, cold plasma has enhanced the phenolic compounds in tomato-based beverage, siriguela juice, and blueberry juice [62, 74, 86]. Moreover, Rodríguez et al. [116] and Sarangapani et al. [117] reported an increase in the content of phenols upon CP treatment in cashew apple juice and blueberries, respectively. These varied results have necessitated further research on the CP effects and mechanism of actions of different phenolic compounds. Thus, the plasma source, the food products, mode of exposure, and other treatment parameters have been observed to be critical for the impact of CP on polyphenolic compounds. Effects of CP application on the polyphenols/antioxidant components of food matrix are tabulated in Table 3.
Pesticide Degradation
Pesticides present in the food stuff are difficult to degrade and eliminate. As a result, they enter the food chain and accumulate in the bodies of the animals, soil, and water bodies. This process is termed as “bioaccumulation” and is regarded as an ecological hazard due to the toxic nature of the pesticides. Importantly, the source gas composition and the overall plasma energy are critical to be involved in plasma-mediated pesticide degradation [117]. Organic molecules of the pesticides possessed similar ionization and dissociation energies to those of the electrons in CP. Therefore, they are easily dissociated upon plasma applications [118, 119]. In addition, the high oxidizing potential of free radicals in the plasma also makes them to participate in the degradation of pesticide molecules, and releasing fewer toxic compounds [118].
Different plasma species show varied efficacies on different types of pesticides. For instance, in the degradation of dichlorvos and organophosphorus pesticides, free radicals and electrons have been deemed to be most effective [118,119,120]. Several methods of pesticide degradation, such as photocatalysis, adsorption, ultrasound, Fenton oxidation, and membrane filtration, have been explored [121,122,123,124,125]. However, these methods are not feasible for industrial applications due to either incomplete pesticide degradation or generation of undesirable by-products. Recently, the focus has been turned in this regard to the plasma technique.
The use of “plasma-activated water” (PAW) has been reported effective in the degradation of pesticides on tangerine and grapes [119, 126]. Also, the reduction of pesticides like chloropyrifos, carbaryl, and cypermethrin has been documented by DBD plasma [71, 127, 128]. Furthermore, Mousavi et al. [129] showed a complete degradation of organophosphorus pesticides in cucumbers and apples using CP treatment. Similarly, use of high efficiency of CP at atmospheric pressure has been suggested in the degradation of paraoxon and parathion [130]. It has been suggested that hydroxide radical, molecular nitrogen, and atomic oxygen are the primary plasma species responsible for achieving such effects. A mixture of fludioxonil, pyriproxyfen, cyprodinil, and azoxystrobin pesticides applied on strawberries and subjected them to in-package plasma exposure at various voltages and treatment durations. The plasma treatment of 80 kV for 5 min could effectively decrease the fludioxonil, azoxystrobin, pyriproxyfen, and cyprodinil by 71%, 69%, 56%, and 45% respectively [118]. Sarangapani et al. [117] achieved an 80.18% and 75.62% decomposition of boscalid and imidacloprid, respectively, in blueberries upon DBD plasma treatment without any changes in their color or other physical attributes. Similarly, the efficacy of CP in degradation of several pesticides, including 17β-Estradiol, endosulfan, organophosphate, and dichlorvos/omethoate, has been revealed [131, 132].
All these reports have suggested that plasma-triggered pesticide degradation is mainly affected by gas composition, input voltage, treatment duration, and plasma power. Some of the studies reporting the effects of CP on different pesticides are listed in Table 4.
Food Allergen Degradation
After pesticides, food allergens are considered the most hazardous contaminants of food products. Food allergens are the compounds that are naturally present in food products but induce the allergic reactions in certain individuals [16]. Some of the most commonly consumed food products that have been reported to elicit allergic reactions include milk, egg, soybean, nuts, and fish. However, the tricky part of the management of food allergens is that the food products inducing allergic reactions in some individuals may be highly beneficial in some other aspects like soybean is rich in protein. However, such allergens are difficult to be eliminated using conventional techniques. In addition, the use of conventional treatments often leads to undesirable changes in the food product itself. Similar to the microbial decontamination and pesticide degradation, the composition of gas used to produce plasma is the key factor that determines the efficacy of plasma in allergen removal [133, 134]. Changes in the linear and the conformational epitopes of the allergen and their reactivity have been proposed to be the major mechanisms of plasma-induced allergen inactivation. The reactive species in the CP degrade the linear epitopes and trigger the structural changes in the proteinaceous conformational epitopes that cause the deactivation of allergens [135]. Furthermore, various oxygen, nitrogen, and hydroxyl radicals have been shown to adversely affect the protein structure that renders the allergen molecules ineffective [1, 136].
Recently, effects of CP have been studied against several allergens, such as glycinin, conglycinin, β-conglycinin, α-lactalbumin, and β-lactoglobulin [51, 136, 137]. Degradation of food allergens by cold plasma like tropomyosin in prawns and anacardic acids in cashew nuts has been documented [51, 138]. Moreover, Venkataratnam et al. [139] reported the decrease in allergenicity of peanut after cold plasma treatment. CP exposure has completely eliminated the primary allergenic components of soy protein, glycinin, and β-conglycinin by converting them into insoluble aggregates [136, 140]. In soymilk, the CP-triggered oxidative reactions and conformational changes led to an 86% decrease in the activity of soybean trypsin inhibitor [137]. Similarly, CP effect has also been studied on allergenic compounds present in milk, such as α-lactalbumin, α-casein, and β-lactoglobulin [141]. Table 5 presents the effect of CP on food allergens.
Food Packaging
Packaging of food materials has always been an important part of food industries. Efficient food packaging is crucial to enhance the shelf life, while maintaining physical and chemical attributes and preventing microbial contamination of food products. In the past few decades, the conventional packing materials have been replaced by polymeric materials that offer more advantages in terms of functionality and stability of food stuffs. However, the polymeric materials are often hydrophobic in nature and have low surface energies [24]. Plasma-induced changes in packaging films are classified in two mechanisms: surface activation and surface deposition [142].
During surface activation, plasma modifies the barrier properties of the polymeric film that essentially reduces the hydrophobicity and permeability of the film. This, in turn, increases the shelf-life of the food product while decreasing the chances of contamination during storage and transport. In this direction, the plasma exposure causes the formation of cross-linked or polar groups on the film surface. During surface deposition, the packing film is coated with desired elements that alter its thickness as well as affect its properties. As with other substances, the plasma-induced changes in the film properties are largely dependent on the type of plasma, gas source, gas composition, plasma power, and treatment duration. On the contrary, a study has reported no effect of plasma-induced changes in the packaging films on shelf-life improvement upon extended periods of storage [143].
Recently, CP technique is gaining immense interest in the sterilization and enhancement of the functionality of food packaging materials. The main advantage of CP is its effect on the entire surface of the packing material and, therefore, reduces the chance of the shadow effect or non-exposure of parts of material to sterilizing agent [53]. CP has also reported changes in crystal structure, improvement in barrier properties, and mechanical strength in casein- or protein-based edible films [43, 144]. Also, enhancement in surface roughness and microorganism inhibition properties in new kind of antimicrobial active packaging has been observed after application of capacitively coupled plasma treatment [76]. Plasma treatment is found to be partially cleaved acyloamino groups of zein film, making it more tensile and robust film [145]. Application of CP on fish protein films has improved their color, mechanical, and barrier properties [146, 147]. CP has also decreased the sensitivity of films to water and decreased water vapor permeability and their solubility. Furthermore, surface activation has been defined as the formation of polar groups or cross-linked molecules that enhance the properties of the packaging film’s surface, such as hydrophobicity and oxygen and moisture permeability [148]. An increase in surface hydrophobicity and antimicrobial characteristics of films has been reported following treatment with DBD plasma in the presence of ZnO. This could be due to incorporation of oxygen-containing functional groups on the surface [149]. Some of the studies focused on effects of CP of most commonly used food packaging films are tabulated (Table 6).
Effect of CP on Starch, Vitamins, and Lipids
Carbohydrates and lipids are among the most important biomolecules present in food products. Ozonolysis caused by the plasma species has been considered as the major pathway of breakdown of the glycosidic bonds that led to the depolymerization of the carbohydrate molecules and subsequent oxidation of its breakdown products to form CO2, carboxyl and carbonyl compounds, lactones, and hydroperoxides [113]. Plasma-induced structural modifications in starch molecules have altered their properties, such as pasting characteristics, swelling power, water absorption, solubility, and enzyme susceptibility [48, 150, 151]. These changes have been attributed to cross linking, increase in surface energy, depolymerization, change in hydrophilic nature, and incorporation of functional groups [48, 150, 152].
Nowadays, modified starch is used as a prominent food additive and CP is being employed to modify starch. Starch is usually modified via cross-linking and depolymerization. Changes in gelatinization and crystallinity of starch have been reported upon CP treatment [48]. Moreover, improvement in paste-cooling stability and reduced retrogradation in corn starch has also been reported on cold plasma treatment [153]. CP treatment enhanced the water uptake rate in black gram. It has been suggested that CP treatment increased the number of water binding sites and surface etching because of protein and starch fragmentation [47]. CP treatment has also been reported to reduce the cooking time of brown rice due to inclusion of polar groups between starch molecules [154]. In rice, CP treatment modified the starch that led to decrease in gelatinization and pasting temperatures, degree of hydrolysis, amylose content, and retrogradation tendency [155].
In lipids, the major effect of plasma has been observed for their oxidation. However, plasma effects on lipid oxidation are variable and inconclusive. Few studies have observed no effect of plasma on oxidation of lipid molecules [156, 157]. Nevertheless, CP has been employed for the production of partially hydrogenated oils or trans-free oils. Hence, more studies are needed to further elucidate the exact mechanism of CP on lipids and the utility of plasma-generated partially hydrogenated oils. With respect to food products, lipid oxidation poses a major problem as it leads to changes in the shelf-life, odor, and taste. Hence, it is important to elucidate the effects of oxidizing elements of CP with respect to lipids present in the food items. Previous studies have reported no marked changes in the lipid oxidation status of CP-treated food items, including sushi, raw pork, fresh pork, and beef jerky [73, 158]. On the contrary, a significant lipid oxidation has been reported in CP-treated mackerel fillets [159]. Also, lipid oxidation has been documented in several CP-treated dairy and meat fats [160]. A new technique of producing partially hydrogenated soybean oil has been devised using hydrogen plasma [50]. This study has opened up a new domain of CP application in hydrogenation of oils and considered to be a desirable characteristic as performed at atmospheric pressure in the absence of a catalyst. Hence, more experimental evidences are required for a conclusive effect of CP on lipids of food stuffs. Some of the effects of CP on food characteristics are tabulated (Table 7).
Among vitamins, the effect of CP has been evaluated on the stability and levels of vitamin C in food products. Analysis on whole vegetables and fruits, such as radish sprout, lettuce, and kiwifruit, has shown no significant impact of CP on their vitamin C content [97, 98]. On the contrary, Hosseini et al. [161] reported reduction in the level of vitamin C in sour cherry juice after treatment with plasma. A few other studies also reported the reduction in vitamin C content of cashew apple juice and orange juice upon CP treatment [94, 116]. Decrease in vitamin C content is attributed to the interaction with oxidizing species of plasma. In addition, few studies have shown enhancement in other vitamins after cold plasma treatment [86, 162].
Seed Germination
An enhanced rate of seed germination has been observed following plasma treatment. The reactive species of plasma are known to be able to penetrate the seed coat and directly influence the cells inside. In addition, exposure to plasma leads to surface ablation on the seed coat that promotes the penetration of moisture and oxygen to the embryo and enhances seed germination [53, 163]. Exposure to plasma has also been observed for the disruption of the cell wall, and affects on the enzyme activity that brings the seed out of the dormant phase and promotes germination.
Plasma treatment has generally been associated with a more rapid germination of seeds owing to several factors. Recently, Dawood [164] and Billah et al. [165] have reported enhanced seed germination in moringa seeds and black gram seeds, respectively, after cold plasma treatment. Moreover, plasma-activated water (PAW) also showed positive effect on seed germination in black gram, radish, tomato, and sweet pepper [46, 166]. Fenugreek seeds and wheat seeds have been observed for enhanced germination rate upon argon plasma jet [167, 168]. Also, CP treatment has increased the germination rate (102%), seed conductivity (20%), and radical root (36.2%) of mung bean seeds [169]. These findings have suggested that CP treatment enhances the water inhibition capacity of seeds, which not only enhances the germination rate but also inhibits the growth of microbes and enhances other growth aspects. An increase in the demethylation levels of ATP, growth regulators, and rapamycin has been observed in argon plasma-treated soybean sprouts [170]. Also, an 80% increase in germination rate of radish and enhancement in stem elongation by 60% in tomatoes have been reported after treatment with DBD PAW [46]. Few studies have also observed negligible effect of cold plasma on seed germination of mung beans [44] and grain seeds [171]. Some studies reporting the effect of cold plasma on seed germination are tabulated for reference (Table 8).
Limitations and Future of Non-thermal Plasma
From the discussion, cold plasma has been observed as a disruptive technology for the many food processes. However, adoption of cold plasma for food-manufacturing and food-processing might face one of more of the followings major challenges like design and source of plasma, control of cold plasma process, and regulatory aspects. Though the conditions of cold plasma process can be tuned to maximize the production of desired component from the agricultural or food commodities, focus is required to design the cold plasma for scale-up as well as on commercialization of the in-package plasma technology.
Even with a plethora of studies, several aspects of the CP technique with respect to food industry are still not known. For instance, there are still some gaps in the research concerning the effects of CP on allergens and antioxidants. Moreover, studies need to be conducted on the safety, toxicity, and/or health effects of CP-treated food products in humans. Furthermore, the applicability of CP technique on fatty products is still unresolved, owing to the possibility of increased oxidation in such products and subsequent decrease of their nutritional value.
Due to variable effects of different plasma components on different food products, optimization studies are needed with respect to the type, intensity, and duration of plasma treatments as well as the food types. Recent few studies have reported the effect of CP on firmness, discoloration, and pH of vegetables and fruits.
One of the major limitations of CP efficiency is its dependency on the surface topography of the target. At present, the cold plasma technology has limited penetration depth, and in order to maintain the product quality, interactions between product and processes should be minimum. Cold plasma technology has recently gained the pace in the improvement of food processing and food quality. Cold plasma has already crossed the technology readiness level (TRL5). Also, present scale of operation is at gram or kilogram scale. However, further work is essential to design systems that are scalable to industrial requirements. Hence, efforts need to be initiated for the scale-up of CP techniques for larger food commodity at industrial scale. For this, more intensive and focused efforts are required to exploit the use of plasma technique at commercial level.
Conclusions
Cold plasma (CP) technique is a non-conventional, non-thermal technique that has been demonstrated to exhibit high potential in enhancing food quality, ensuring better food safety, and facilitating better packaging of food products. CP technique has been shown to be extremely beneficial in the areas where the applications of conventional techniques are not feasible. Since CP technique can be implemented at atmospheric pressure and ambient temperature, its use could be safe on food products without or minimal loss of their nutritional and sensory characteristics. Food processing sector and agricultural industries need to understand the importance of CP and make sincere efforts for its applicability at commercial level for the benefit of society in large.
References
Gavahian M, Khaneghah AM (2020) Cold plasma as a tool for the elimination of food contaminants: recent advances and future trends. Crit Rev Food Sci Nutr 60(9):1581–1592. https://doi.org/10.1080/10408398.2019.1584600
Langmuir I (1928) Oscillations in ionized gases. Proc Natl Acad Sci USA 14(8):627. https://doi.org/10.1073/pnas.14.8.627
Dasan BG, Onal-Ulusoy B, Pawlat J, Diatczyk J, Sen Y, Mutlu M (2017) A new and simple approach for decontamination of food contact surfaces with gliding arc discharge atmospheric non-thermal plasma. Food Bioprocess Technol 10(4):650–661. https://doi.org/10.1007/s11947-016-1847-2
Ekezie FGC, Sun DW, Cheng JH (2017) A review on recent advances in cold plasma technology for the food industry: current applications and future trends. Trends Food Sci Technol 69:46–58. https://doi.org/10.1016/j.tifs.2017.08.007
Peng P, Schiappacasse C, Zhou N, Addy M, Cheng Y, Zhang Y, Anderson E, Chen D, Wang Y, Liu Y, Chen P, Ruan R (2019) Plasma in situ gas-liquid nitrogen fixation using concentrated high-intensity electric field. J Phys D Appl Phys 52:494001. https://doi.org/10.1088/1361-6463/ab3ea6
Scholtz V, Pazlarova J, Souskova H, Khun J, Julak J (2015) Nonthermal plasma–a tool for decontamination and disinfection. Biotechnol Adv 33(6):1108–1119. https://doi.org/10.1016/j.biotechadv.2015.01.002
Han Y, Cheng JH, Sun DW (2019) Activities and conformation changes of food enzymes induced by cold plasma: a review. Crit Rev Food Sci Nutr 59(5):794–811. https://doi.org/10.1080/10408398.2018.1555131
Mandal R, Singh A, Singh AP (2018) Recent developments in cold plasma decontamination technology in the food industry. Trends Food Sci Technol 80:93–103. https://doi.org/10.1016/j.tifs.2018.07.014
Bessaire T, Perrin I, Tarres A, Bebius A, Reding F, Theurillat V (2019) Mycotoxins in green coffee: occurrence and risk assessment. Food Control 96:59–67. https://doi.org/10.1016/j.foodcont.2018.08.033
Khaneghah AM, Fakhri Y, Raeisi S, Armoon B, Sant’Ana AS (2018) Prevalence and concentration of ochratoxin A, zearalenone, deoxynivalenol and total aflatoxin in cereal-based products: a systematic review and meta-analysis. Food Chem Toxicol 118:830–848. https://doi.org/10.1016/j.fct.2018.06.037
Khaneghah AM, Eş I, Raeisi S, Fakhri Y (2018) Aflatoxins in cereals: state of the art. J Food Saf 38(6):e12532. https://doi.org/10.1111/jfs.12532
Fathabad AE, Shariatifar N, Moazzen M, Nazmara S, Fakhri Y, Alimohammadi M, Azari A, Khaneghah AM (2018) Determination of heavy metal content of processed fruit products from Tehran’s market using ICP-OES: a risk assessment study. Food Chem Toxicol 115:436–446. https://doi.org/10.1016/j.fct.2018.03.044
Shahsavani A, Fakhri Y, Ferrante M, Keramati H, Zandsalimi Y, Bay A, Pouya SRH, Moradi B, Bahmani Z, Mousavi Khaneghah A (2017) Risk assessment of heavy metals bioaccumulation: fished shrimps from the Persian Gulf. Toxin Rev 36(4):322–330. https://doi.org/10.1080/15569543.2017.1312451
Yousefi M, Shemshadi G, Khorshidian N, Ghasemzadeh-Mohammadi V, Fakhri Y, Hosseini H, Khaneghah AM (2018) Polycyclic aromatic hydrocarbons (PAHs) content of edible vegetable oils in Iran: a risk assessment study. Food Chem Toxicol 118:480–489. https://doi.org/10.1016/j.fct.2018.05.063
Amirahmadi M, Kobarfard F, Pirali-Hamedani M, Yazdanpanah H, Rastegar H, Shoeibi S, Mousavi Khaneghah A (2017) Effect of Iranian traditional cooking on fate of pesticides in white rice. Toxin Rev 36(3):177–186. https://doi.org/10.1080/15569543.2017.1301956
Crevel RW, Baumert JL, Luccioli S, Baka A, Hattersley S, Hourihane JOB, Ronsmans S, Timmermans F, Ward R, Chung YJ (2014) Translating reference doses into allergen management practice: challenges for stakeholders. Food Chem Toxicol 67:277–287. https://doi.org/10.1016/j.fct.2014.01.033
Razzaghi N, Ziarati P, Rastegar H, Shoeibi S, Amirahmadi M, Conti GO, Ferrante M, Fakhri Y, Khaneghah AM (2018) The concentration and probabilistic health risk assessment of pesticide residues in commercially available olive oils in Iran. Food Chem Toxicol 120:32–40. https://doi.org/10.1016/j.fct.2018.07.002
Schaarschmidt S (2016) Public and private standards for dried culinary herbs and spices–Part I: standards defining the physical and chemical product quality and safety. Food Control 70:339–349. https://doi.org/10.1016/j.foodcont.2016.06.004
Sicherer SH, Sampson HA (2018) Food allergy: a review and update on epidemiology, pathogenesis, diagnosis, prevention, and management. J Allergy Clin Immunol 141(1):41–58. https://doi.org/10.1016/j.jaci.2017.11.003
Cheng L, Sun DW, Zhu Z, Zhang Z (2017) Emerging techniques for assisting and accelerating food freezing processes: a review of recent research progresses. Crit Rev Food Sci Nutr 57(4):769–781. https://doi.org/10.1080/10408398.2015.1004569
Pu YY, Sun DW (2017) Combined hot-air and microwave-vacuum drying for improving drying uniformity of mango slices based on hyperspectral imaging visualization of moisture content distribution. Biosys Eng 156:108–119. https://doi.org/10.1016/j.biosystemseng.2017.01.006
Yang Q, Sun DW, Cheng W (2017) Development of simplified models for nondestructive hyperspectral imaging monitoring of TVB-N contents in cured meat during drying process. J Food Eng 192:53–60. https://doi.org/10.1016/j.jfoodeng.2016.07.015
Muranyi P, Wunderlich J, Heise M (2007) Sterilization efficiency of a cascaded dielectric barrier discharge. J Appl Microbiol 103(5):1535–1544. https://doi.org/10.1111/j.1365-2672.2007.03385.x
Pankaj SK, Bueno-Ferrer C, Misra NN, Milosavljević V, O’donnell CP, Bourke P, Keener KM, Cullen PJ (2014) Applications of cold plasma technology in food packaging. Trends Food Sci Technol 35(1):5–17. https://doi.org/10.1016/j.tifs.2013.10.009
Vesel A, Mozetic M (2012) Surface modification and ageing of PMMA polymer by oxygen plasma treatment. Vacuum 86(6):634–637. https://doi.org/10.1016/j.vacuum.2011.07.005
Gavahian M, Chu YH, Khaneghah AM, Barba FJ, Misra NN (2018) A critical analysis of the cold plasma induced lipid oxidation in foods. Trends Food Sci Technol 77:32–41. https://doi.org/10.1016/j.tifs.2018.04.009
López M, Calvo T, Prieto M, Múgica-Vidal R, Muro-Fraguas I, Alba-Elías F, Alvarez-Ordóñez A (2019) A review on non-thermal atmospheric plasma for food preservation: mode of action, determinants of effectiveness, and applications. Front Microbiol 10:622. https://doi.org/10.3389/fmicb.2019.00622
Frías E, Iglesias Y, Alvarez-Ordóñez A, Prieto M, González-Raurich M, López M (2020) Evaluation of cold atmospheric pressure plasma (CAPP) and plasma-activated water (PAW) as alternative non-thermal decontamination technologies for tofu: impact on microbiological, sensorial and functional quality attributes. Food Res Int 129:108859. https://doi.org/10.1016/j.foodres.2019.108859
Hertwig C, Meneses N, Mathys A (2018) Cold atmospheric pressure plasma and low energy electron beam as alternative nonthermal decontamination technologies for dry food surfaces: a review. Trends Food Sci Technol 77:131–142. https://doi.org/10.1016/j.tifs.2018.05.011
Jayasena DD, Kim HJ, Yong HI, Park S, Kim K, Choe W, Jo C (2015) Flexible thin-layer dielectric barrier discharge plasma treatment of pork butt and beef loin: effects on pathogen inactivation and meat-quality attributes. Food Microbiol 46:51–57. https://doi.org/10.1016/j.fm.2014.07.009
Liao X, Liu D, Xiang Q, Ahn J, Chen S, Ye X, Ding T (2017) Inactivation mechanisms of non-thermal plasma on microbes: a review. Food Control 75:83–91. https://doi.org/10.1016/j.foodcont.2016.12.021
Muhammad AI, Li Y, Liao X, Liu D, Ye X, Chen S, Hu Y, Wang J, Ding T (2019) Effect of dielectric barrier discharge plasma on background microflora and physicochemical properties of tiger nut milk. Food Control 96:119–127. https://doi.org/10.1016/j.foodcont.2018.09.010
Prasad P, Mehta D, Bansal V, Sangwan RS (2017) Effect of atmospheric cold plasma (ACP) with its extended storage on the inactivation of Escherichia coli inoculated on tomato. Food Res Int 102:402–408. https://doi.org/10.1016/j.foodres.2017.09.030
Yannam SK, Estifaee P, Rogers S, Thagard SM (2018) Application of high voltage electrical discharge plasma for the inactivation of Escherichia coli ATCC 700891 in tangerine juice. LWT 90:180–185. https://doi.org/10.1016/j.lwt.2017.12.018
Yong HI, Lee SH, Kim SY, Park S, Park J, Choe W, Jo C (2019) Color development, physiochemical properties, and microbiological safety of pork jerky processed with atmospheric pressure plasma. Innov Food Sci Emerg Technol 53:78–84. https://doi.org/10.1016/j.ifset.2017.09.005
Bußler S, Ehlbeck J, Schlüter OK (2017) Pre-drying treatment of plant related tissues using plasma processed air: impact on enzyme activity and quality attributes of cut apple and potato. Innov Food Sci Emerg Technol 40:78–86. https://doi.org/10.1016/j.ifset.2016.05.007
Kang JH, Roh SH, Min SC (2019) Inactivation of potato polyphenol oxidase using microwave cold plasma treatment. J Food Sci 84(5):1122–1128. https://doi.org/10.1111/1750-3841.14601
Porto E, Filho EGA, Silva LMA, Fonteles TV, do Nascimento RBR, Fernandes FA, de Brito ES, Rodrigues S (2020) Ozone and plasma processing effect on green coconut water. Food Res Int 131:109000. https://doi.org/10.1016/j.foodres.2020.109000
Tappi S, Gozzi G, Vannini L, Berardinelli A, Romani S, Ragni L, Rocculi P (2016) Cold plasma treatment for fresh-cut melon stabilization. Innov Food Sci Emerg Technol 33:225–233. https://doi.org/10.1016/j.ifset.2015.12.022
Tolouie H, Mohammadifar MA, Ghomi H, Yaghoubi AS, Hashemi M (2018) The impact of atmospheric cold plasma treatment on inactivation of lipase and lipoxygenase of wheat germs. Innov Food Sci Emerg Technol 47:346–352. https://doi.org/10.1016/j.ifset.2018.03.002
Chen G, Dong S, Zhao S, Li S, Chen Y (2019) Improving functional properties of zein film via compositing with chitosan and cold plasma treatment. Ind Crops Prod 129:318–326. https://doi.org/10.1016/j.indcrop.2018.11.072
Cui H, Bai M, Lin L (2018) Plasma-treated poly (ethylene oxide) nanofibers containing tea tree oil/beta-cyclodextrin inclusion complex for antibacterial packaging. Carbohyd Polym 179:360–369. https://doi.org/10.1016/j.carbpol.2017.10.011
Wu X, Liu Q, Luo Y, Murad MS, Zhu L, Mu G (2020) Improved packing performance and structure-stability of casein edible films by dielectric barrier discharges (DBD) cold plasma. Food Packag Shelf Life 24:100471. https://doi.org/10.1016/j.fpsl.2020.100471
Darmanin M, Kozak D, de Oliveira Mallia J, Blundell R, Gatt R, Valdramidis VP (2020) Generation of plasma functionalized water: antimicrobial assessment and impact on seed germination. Food Control 113:107168. https://doi.org/10.1016/j.foodcont.2020.107168
Li L, Li J, Shao H, Dong Y (2018) Effects of low-vacuum helium cold plasma treatment on seed germination, plant growth and yield of oilseed rape. Plasma Sci Technol 20(9):095502. https://doi.org/10.1088/2058-6272/aac3d0
Sivachandiran L, Khacef A (2017) Enhanced seed germination and plant growth by atmospheric pressure cold air plasma: combined effect of seed and water treatment. RSC Adv 7(4):1822–1832. https://doi.org/10.1039/c6ra24762h
Sarangapani C, Devi RY, Thirumdas R, Trimukhe AM, Deshmukh RR, Annapure US (2017) Physico-chemical properties of low-pressure plasma treated black gram. LWT-Food Science and Technology 79:102–110. https://doi.org/10.1016/j.lwt.2017.01.017
Yan Y, Feng L, Shi M, Cui C, Liu Y (2020) Effect of plasma-activated water on the structure and in vitro digestibility of waxy and normal maize starches during heat-moisture treatment. Food Chem 306:125589. https://doi.org/10.1016/j.foodchem.2019.125589
Na H, Mok C, Lee J (2020) Effects of plasma treatment on the oxidative stability of vegetable oil containing antioxidants. Food Chem 302:125306. https://doi.org/10.1016/j.foodchem.2019.125306
Yepez XV, Keener KM (2016) High-voltage atmospheric cold plasma (HVACP) hydrogenation of soybean oil without trans-fatty acids. Innov Food Sci Emerg Technol 38:169–174. https://doi.org/10.1016/j.ifset.2016.09.001
Ekezie FGC, Sun DW, Cheng JH (2019) Altering the IgE binding capacity of king prawn (Litopenaeus Vannamei) tropomyosin through conformational changes induced by cold argon-plasma jet. Food Chem 300:125143. https://doi.org/10.1016/j.foodchem.2019.125143
Chou YJ, Cheng KC, Hsu FC, Wu JS, Ting Y (2021) Producing high quality mung bean sprout using atmospheric cold plasma treatment: better physical appearance and higher γ-aminobutyric acid (GABA) content. J Sci Food Agric 101(15):6463–6471. https://doi.org/10.1002/jsfa.11317
Thirumdas R, Deshmukh RR, Annapure US (2015) Effect of low temperature plasma processing on physicochemical properties and cooking quality of basmati rice. Innov Food Sci Emerg Technol 31:83–90. https://doi.org/10.1016/j.ifset.2015.08.003
Pankaj SK, Bueno-Ferrer C, Misra NN, O’Neill L, Bourke P, Cullen PJ (2017) Effects of cold plasma on surface, thermal and antimicrobial release properties of chitosan film. J Renew Mater 5(1):14–20. https://doi.org/10.7569/JRM.2016.634105
Guo J, Huang K, Wang J (2015) Bactericidal effect of various non-thermal plasma agents and the influence of experimental conditions in microbial inactivation: a review. Food Control 50:482–490. https://doi.org/10.1016/j.foodcont.2014.09.037
Pankaj SK, Wan Z, Keener KM (2018) Effects of cold plasma on food quality: a review. Foods 7(1):4. https://doi.org/10.3390/foods7010004
Misra NN, Keener KM, Bourke P, Mosnier JP, Cullen PJ (2014) In-package atmospheric pressure cold plasma treatment of cherry tomatoes. J Biosci Bioeng 118(2):177–182. https://doi.org/10.1016/j.jbiosc.2014.02.005
Lunov O, Zablotskii V, Churpita O, Jäger A, Polívka L, Syková E, Dejneka A, Kubinová Š (2016) The interplay between biological and physical scenarios of bacterial death induced by non-thermal plasma. Biomaterials 82:71–83. https://doi.org/10.1016/j.biomaterials.2015.12.027
Yong HI, Kim HJ, Park S, Alahakoon AU, Kim K, Choe W, Jo C (2015) Evaluation of pathogen inactivation on sliced cheese induced by encapsulated atmospheric pressure dielectric barrier discharge plasma. Food Microbiol 46:46–50. https://doi.org/10.1016/j.fm.2014.07.010
Liang Y, Wu Y, Sun K, Chen Q, Shen F, Zhang J, Yao M, Zhu T, Fang J (2012) Rapid inactivation of biological species in the air using atmospheric pressure nonthermal plasma. Environ Sci Technol 46(6):3360–3368. https://doi.org/10.1021/es203770q
Han L, Boehm D, Amias E, Milosavljević V, Cullen PJ, Bourke P (2016) Atmospheric cold plasma interactions with modified atmosphere packaging inducer gases for safe food preservation. Innov Food Sci Emerg Technol 38:384–392. https://doi.org/10.1016/j.ifset.2016.09.026
Mehta D, Sharma N, Bansal V, Sangwan RS, Yadav SK (2019) Impact of ultrasonication, ultraviolet and atmospheric cold plasma processing on quality parameters of tomato-based beverage in comparison with thermal processing. Innov Food Sci Emerg Technol 52:343–349. https://doi.org/10.1016/j.ifset.2019.01.015
Cui H, Wu J, Li C, Lin L (2017) Promoting anti-listeria activity of lemongrass oil on pork loin by cold nitrogen plasma assist. J Food Saf 37(2):e12316. https://doi.org/10.1111/jfs.12316
Trevisani M, Berardinelli A, Cevoli C, Cecchini M, Ragni L, Pasquali F (2017) Effects of sanitizing treatments with atmospheric cold plasma, SDS and lactic acid on verotoxin-producing Escherichia coli and Listeria monocytogenes in red chicory (radicchio). Food Control 78:138–143. https://doi.org/10.1016/j.foodcont.2017.02.056
Liu CT, Kumakura T, Ishikawa K, Hashizume H, Takeda K, Ito M, Hori M, Wu JS (2016) Effects of assisted magnetic field to an atmospheric-pressure plasma jet on radical generation at the plasma-surface interface and bactericidal function. Plasma Sources Sci Technol 25(6):065005. https://doi.org/10.1088/0963-0252/25/6/065005
Pankaj SK, Misra NN, Cullen PJ (2013) Kinetics of tomato peroxidase inactivation by atmospheric pressure cold plasma based on dielectric barrier discharge. Innov Food Sci Emerg Technol 19:153–157. https://doi.org/10.1016/j.ifset.2013.03.001
Liang Y, Jensen RE, Pappas DD, Palmese GR (2011) Toughening vinyl ester networks with polypropylene meso-fibers: interface modification and composite properties. Polymer 52(2):510–518. https://doi.org/10.1016/j.polymer.2010.12.006
O’Connor N, Milosavljević V, Daniels S (2011) Development of a real time monitor and multivariate method for long term diagnostics of atmospheric pressure dielectric barrier discharges: application to He, He/N2, and He/O2 discharges. Rev Sci Instrum 82(8):083501. https://doi.org/10.1063/1.3624743
Mehta D, Yadav SK (2020) Impact of atmospheric non-thermal plasma and hydrothermal treatment on bioactive compounds and microbial inactivation of strawberry juice: a hurdle technology approach. Food Sci Technol Int 26(1):3–10. https://doi.org/10.1177/1082013219865360
Moutiq R, Misra NN, Mendonca A, Keener K (2020) In-package decontamination of chicken breast using cold plasma technology: microbial, quality and storage studies. Meat Sci 159:107942. https://doi.org/10.1016/j.meatsci.2019.107942
Phan KTK, Phan HT, Boonyawan D, Intipunya P, Brennan CS, Regenstein JM, Phimolsiripol Y (2018) Non-thermal plasma for elimination of pesticide residues in mango. Innov Food Sci Emerg Technol 48:164–171. https://doi.org/10.1016/j.ifset.2018.06.009
Gong X, Lin Y, Li X, Wu A, Zhang H, Yan J, Du C (2020) Decomposition of volatile organic compounds using gliding arc discharge plasma. J Air Waste Manag Assoc 70(2):138–157. https://doi.org/10.1080/10962247.2019.1698476
Choi S, Puligundla P, Mok C (2017) Effect of corona discharge plasma on microbial decontamination of dried squid shreds including physico-chemical and sensory evaluation. LWT 75:323–328. https://doi.org/10.1016/j.lwt.2016.08.063
Hou Y, Wang R, Gan Z, Shao T, Zhang X, He M, Sun A (2019) Effect of cold plasma on blueberry juice quality. Food Chem 290:79–86. https://doi.org/10.1016/j.foodchem.2019.03.123
Bauer S, Schmuki P, Von Der Mark K, Park J (2013) Engineering biocompatible implant surfaces: Part I: materials and surfaces. Prog Mater Sci 58(3):261–326. https://doi.org/10.1016/j.pmatsci.2012.09.001
Wong LW, Hou CY, Hsieh CC, Chang CK, Wu YS, Hsieh CW (2020) Preparation of antimicrobial active packaging film by capacitively coupled plasma treatment. LWT 117:108612. https://doi.org/10.1016/j.lwt.2019.108612
Ryan K, O’Farrell D, Ellingboe AR (2011) Spatial structure of plasma potential oscillation and ion saturation current in VHF multi-tile electrode plasma source. Curr Appl Phys 11(5):S114–S116. https://doi.org/10.1016/j.cap.2011.05.003
Milosavljević V, Faulkner R, Hopkins MB (2007) Real time sensor for monitoring oxygen in radio–frequency plasma applications. Opt Express 15(21):13913–13923. https://doi.org/10.1364/OE.15.013913
Kim JE, Oh YJ, Song AY, Min SC (2019) Preservation of red pepper flakes using microwave-combined cold plasma treatment. J Sci Food Agric 99(4):1577–1585. https://doi.org/10.1002/jsfa.9336
Milosavljević V, MacGearailt N, Cullen PJ, Daniels S, Turner MM (2013) Phase-resolved optical emission spectroscopy for an electron cyclotron resonance etcher. J Appl Phys 113(16):163302. https://doi.org/10.1063/1.4802440
Oh YA, Roh SH, Min SC (2016) Cold plasma treatments for improvement of the applicability of defatted soybean meal-based edible film in food packaging. Food Hydrocolloids 58:150–159. https://doi.org/10.1016/j.foodhyd.2016.02.022
Song AY, Oh YA, Roh SH, Kim JH, Min SC (2016) Cold oxygen plasma treatments for the improvement of the physicochemical and biodegradable properties of polylactic acid films for food packaging. J Food Sci 81(1):E86–E96. https://doi.org/10.1111/1750-3841.13172
Laroussi M (2020) Cold plasma in medicine and healthcare: the new frontier in low temperature plasma applications. Front Phys 8:74. https://doi.org/10.3389/fphy.2020.00074
Szili EJ, Short RD, Steele DA, Bradley JW (2021) Surface modification of biomaterials by plasma polymerization. In: Williams R (ed) Woodhead Publishing Series in Biomaterials, surface modification of biomaterials. Woodhead Publishing, pp 3–39. https://doi.org/10.1533/9780857090768.1.3
Tabares FL, Junkar I (2021) Cold plasma systems and their application in surface treatments for medicine. Molecules 26:1903. https://doi.org/10.3390/molecules26071903
Paixão LM, Fonteles TV, Oliveira VS, Fernandes FA, Rodrigues S (2019) Cold plasma effects on functional compounds of siriguela juice. Food Bioprocess Technol 12(1):110–121. https://doi.org/10.1007/s11947-018-2197-z
Garofulić IE, Jambrak AR, Milošević S, Dragović-Uzelac V, Zorić Z, Herceg Z (2015) The effect of gas phase plasma treatment on the anthocyanin and phenolic acid content of sour cherry Marasca (Prunus cerasus var. Marasca) juice. LWT Food Sci Technol 62(1):894–900. https://doi.org/10.1016/j.lwt.2014.08.036
Berardinelli A, Pasquali F, Cevoli C, Trevisani M, Ragni L, Mancusi R, Manfreda G (2016) Sanitisation of fresh-cut celery and radicchio by gas plasma treatments in water medium. Postharvest Biol Technol 111:297–304. https://doi.org/10.1016/j.postharvbio.2015.09.026
Choi EJ, Yang HS, Park HW, Chun HH (2018) Inactivation of Escherichia coli O157: H7 and Staphylococcus aureus in red pepper powder using a combination of radio frequency thermal and indirect dielectric barrier discharge plasma non-thermal treatments. LWT 93:477–484. https://doi.org/10.1016/j.lwt.2018.03.081
Pelletier J (1992) La stérilisation par le procédé plasma. Agressologie (Paris) 33:105–110
Charoux CM, Free L, Hinds LM, Vijayaraghavan RK, Daniels S, O’Donnell CP, Tiwari BK (2020) Effect of non-thermal plasma technology on microbial inactivation and total phenolic content of a model liquid food system and black pepper grains. LWT 118:108716. https://doi.org/10.1016/j.lwt.2019.108716
McClurkin-Moore JD, Ileleji KE, Keener KM (2017) The effect of high-voltage atmospheric cold plasma treatment on the shelf-life of distiller’s wet grains. Food Bioprocess Technol 10(8):1431–1440. https://doi.org/10.1007/s11947-017-1903-6
Wang J, Zhuang H, Zhang J (2016) Inactivation of spoilage bacteria in package by dielectric barrier discharge atmospheric cold plasma–treatment time effects. Food Bioprocess Technol 9(10):1648–1652. https://doi.org/10.1007/s11947-016-1746-6
Xu L, Garner AL, Tao B, Keener KM (2017) Microbial inactivation and quality changes in orange juice treated by high voltage atmospheric cold plasma. Food Bioprocess Technol 10(10):1778–1791. https://doi.org/10.1007/s11947-017-1947-7
Kim JE, Oh YJ, Won MY, Lee KS, Min SC (2017) Microbial decontamination of onion powder using microwave-powered cold plasma treatments. Food Microbiol 62:112–123. https://doi.org/10.1016/j.fm.2016.10.006
Wan Z, Chen Y, Pankaj SK, Keener KM (2017) High voltage atmospheric cold plasma treatment of refrigerated chicken eggs for control of Salmonella Enteritidis contamination on egg shell. LWT Food Sci Technol 76:124–130. https://doi.org/10.1016/j.lwt.2016.10.051
Song AY, Oh YJ, Kim JE, Song KB, Oh DH, Min SC (2015) Cold plasma treatment for microbial safety and preservation of fresh lettuce. Food Sci Biotechnol 24(5):1717–1724. https://doi.org/10.1007/s10068-015-0223-8
Oh YJ, Song AY, Min SC (2017) Inhibition of Salmonella typhimurium on radish sprouts using nitrogen-cold plasma. Int J Food Microbiol 249:66–71. https://doi.org/10.1016/j.ijfoodmicro.2017.03.005
Zhou D, Wang Z, Tu S, Chen S, Peng J, Tu K (2019) Effects of cold plasma, UV-C or aqueous ozone treatment on Botrytis cinerea and their potential application in preserving blueberry. J Appl Microbiol 127(1):175–185. https://doi.org/10.1111/jam.14280
Ziuzina D, Patil S, Cullen PJ, Keener KM, Bourke P (2014) Atmospheric cold plasma inactivation of Escherichia coli, Salmonella enterica serovar Typhimurium and Listeria monocytogenes inoculated on fresh produce. Food Microbiol 42:109–116. https://doi.org/10.1016/j.fm.2014.02.007
Misra NN, Yepez X, Xu L, Keener K (2019) In-package cold plasma technologies. J Food Eng 244:21–31. https://doi.org/10.1016/j.jfoodeng.2018.09.019
Zhuang H, Rothrock MJ Jr, Hiett KL, Lawrence KC, Gamble GR, Bowker BC, Keener KM (2019) In-package air cold plasma treatment of chicken breast meat: treatment time effect. J Food Qual 2019:1837351. https://doi.org/10.1155/2019/1837351
Traylor MJ, Pavlovich MJ, Karim S, Hait P, Sakiyama Y, Clark DS, Graves DB (2011) Long-term antibacterial efficacy of air plasma-activated water. J Phys D Appl Phys 44(47):472001. https://doi.org/10.1088/0022-3727/44/47/472001
Xiang Q, Kang C, Niu L, Zhao D, Li K, Bai Y (2018) Antibacterial activity and a membrane damage mechanism of plasma-activated water against Pseudomonas deceptionensis CM2. LWT 96:395–401. https://doi.org/10.1016/j.lwt.2018.05.059
Sun P, Wu H, Bai N, Zhou H, Wang R, Feng H, Zhu W, Zhang J, Fang J (2012) Inactivation of Bacillus subtilis spores in water by a direct-current, cold atmospheric-pressure air plasma microjet. Plasma Processes Polym 9(2):157–164. https://doi.org/10.1002/ppap.201100041
Wang Q, Salvi D (2021) Recent progress in the application of plasma-activated water (PAW) for food decontamination. Curr Opin Food Sci 42:51–60. https://doi.org/10.1016/j.cofs.2021.04.012
Takai E, Kitano K, Kuwabara J, Shiraki K (2012) Protein inactivation by low-temperature atmospheric pressure plasma in aqueous solution. Plasma Processes Polym 9(1):77–82. https://doi.org/10.1002/ppap.201100063
Dong S, Gao A, Xu H, Chen Y (2017) Effects of dielectric barrier discharges (DBD) cold plasma treatment on physicochemical and structural properties of zein powders. Food Bioprocess Technol 10(3):434–444. https://doi.org/10.1007/s11947-016-1814-y
Castro DRG, Mar JM, da Silva LS, da Silva KA, Sanches EA, de Araújo Bezerra J, Rodrigues S, Fernandes FAN, Campelo PH (2020) Improvement of the bioavailability of amazonian juices rich in bioactive compounds using glow plasma technique. Food Bioprocess Technol 13:670–679. https://doi.org/10.1007/s11947-020-02427-8
de Castro DRG, Mar JM, da Silva LS, da Silva KA, Sanches EA, de Araújo Bezerra J, Rodrigues S, Fernandes FAN, Campelo PH (2020) Dielectric barrier atmospheric cold plasma applied on camu-camu juice processing: effect of the excitation frequency. Food Res Int 131:109044. https://doi.org/10.1016/j.foodres.2020.109044
Segat A, Misra NN, Cullen PJ, Innocente N (2016) Effect of atmospheric pressure cold plasma (ACP) on activity and structure of alkaline phosphatase. Food Bioprod Process 98:181–188. https://doi.org/10.1016/j.fbp.2016.01.010
Xu Y, Tian Y, Ma R, Liu Q, Zhang J (2016) Effect of plasma activated water on the postharvest quality of button mushrooms, Agaricus bisporus. Food Chem 197:436–444. https://doi.org/10.1016/j.foodchem.2015.10.144
Almeida FDL, Cavalcante RS, Cullen PJ, Frias JM, Bourke P, Fernandes FA, Rodrigues S (2015) Effects of atmospheric cold plasma and ozone on prebiotic orange juice. Innov Food Sci Emerg Technol 32:127–135. https://doi.org/10.1016/j.ifset.2015.09.001
Pankaj SK, Wan Z, Colonna W, Keener KM (2017) Effect of high voltage atmospheric cold plasma on white grape juice quality. J Sci Food Agric 97(12):4016–4021. https://doi.org/10.1002/jsfa.8268
Rana S, Mehta D, Bansal V, Shivhare US, Yadav SK (2020) Atmospheric cold plasma (ACP) treatment improved in-package shelf-life of strawberry fruit. J Food Sci Technol 57(1):102–112. https://doi.org/10.1007/s13197-019-04035-7
Rodríguez Ó, Gomes WF, Rodrigues S, Fernandes FA (2017) Effect of indirect cold plasma treatment on cashew apple juice (Anacardium occidentale L.). LWT 84:457–463. https://doi.org/10.1016/j.lwt.2017.06.010
Sarangapani C, O’Toole G, Cullen PJ, Bourke P (2017) Atmospheric cold plasma dissipation efficiency of agrochemicals on blueberries. Innov Food Sci Emerg Technol 44:235–241. https://doi.org/10.1016/j.ifset.2017.02.012
Misra NN, Moiseev T, Patil S, Pankaj SK, Bourke P, Mosnier JP, Keener KM, Cullen PJ (2014) Cold plasma in modified atmospheres for post-harvest treatment of strawberries. Food Bioprocess Technol 7(10):3045–3054. https://doi.org/10.1007/s11947-014-1356-0
Sawangrat C, Leksakul K, Bonyawan D, Anantana T, Jomjunyong S (2019) Decontamination of pesticide residues on tangerine fruit using non-thermal plasma technology. IOP Conf Ser Earth Environ Sci 347(1):012048
Zhou R, Zhou R, Yu F, Xi D, Wang P, Li J, Wang X, Zhang X, Bazaka K, Ostrikov KK (2018) Removal of organophosphorus pesticide residues from Lycium barbarum by gas phase surface discharge plasma. Chem Eng J 342:401–409. https://doi.org/10.1016/j.cej.2018.02.107
Elhussein EAA, Şahin S, Bayazit ŞS (2018) Preparation of CeO2 nanofibers derived from Ce-BTC metal-organic frameworks and its application on pesticide adsorption. J Mol Liq 255:10–17. https://doi.org/10.1016/j.molliq.2018.01.165
Jawale RH, Gogate PR (2018) Combined treatment approaches based on ultrasound for removal of triazophos from wastewater. Ultrason Sonochem 40:89–96. https://doi.org/10.1016/j.ultsonch.2017.02.019
Patil PN, Gogate PR (2016) Combined treatment processes based on ultrasound and photocatalysis for treatment of pesticide containing wastewater. Handbook of Ultrasonics and Sonochemistry. Springer, pp 901–929
Plattner J, Kazner C, Naidu G, Wintgens T, Vigneswaran S (2018) Removal of selected pesticides from groundwater by membrane distillation. Environ Sci Pollut Res 25(21):20336–20347. https://doi.org/10.1007/s11356-017-8929-1
Saini R, Kumar Mondal M, Kumar P (2017) Fenton oxidation of pesticide methyl parathion in aqueous solution: kinetic study of the degradation. Environ Prog Sustainable Energy 36(2):420–427. https://doi.org/10.1002/ep.12473
Zheng Y, Wu S, Dang J, Wang S, Liu Z, Fang J, Han P, Zhang J (2019) Reduction of phoxim pesticide residues from grapes by atmospheric pressure non-thermal air plasma activated water. J Hazard Mater 377:98–105. https://doi.org/10.1016/j.jhazmat.2019.05.058
Feng X, Ma X, Liu H, Xie J, He C, Fan R (2019) Argon plasma effects on maize: pesticide degradation and quality changes. J Sci Food Agric 99(12):5491–5498. https://doi.org/10.1002/jsfa.9810
Ranjitha Gracy TK, Gupta V, Mahendran R (2019) Influence of low-pressure nonthermal dielectric barrier discharge plasma on chlorpyrifos reduction in tomatoes. J Food Process Eng 42(6):e13242. https://doi.org/10.1111/jfpe.13242
Mousavi SM, Imani S, Dorranian D, Larijani K, Shojaee M (2017) Effect of cold plasma on degradation of organophosphorus pesticides used on some agricultural products. J Plant Prot Res 57(1):25–35. https://doi.org/10.1515/jppr-2017-0004
Heo NS, Lee MK, Kim GW, Lee SJ, Park JY, Park TJ (2014) Microbial inactivation and pesticide removal by remote exposure of atmospheric air plasma in confined environments. J Biosci Bioeng 117(1):81–85. https://doi.org/10.1016/j.jbiosc.2013.06.007
Gao L, Sun L, Wan S, Yu Z, Li M (2013) Degradation kinetics and mechanism of emerging contaminants in water by dielectric barrier discharge non-thermal plasma: the case of 17β-Estradiol. Chem Eng J 228:790–798. https://doi.org/10.1016/j.cej.2013.05.079
Reddy PMK, Mahammadunnisa S, Subrahmanyam C (2014) Catalytic non-thermal plasma reactor for mineralization of endosulfan in aqueous medium: a green approach for the treatment of pesticide contaminated water. Chem Eng J 238:157–163. https://doi.org/10.1016/j.cej.2013.08.087
Tolouie H, Mohammadifar MA, Ghomi H, Hashemi M (2017) Cold atmospheric plasma manipulation of proteins in food systems. Crit Rev Food Sci Nutr 58(15):2583–2597. https://doi.org/10.1080/10408398.2017.1335689
Wu Y, Liang Y, Wei K, Li W, Yao M, Zhang J (2014) Rapid allergen inactivation using atmospheric pressure cold plasma. Environ Sci Technol 48(5):2901–2909. https://doi.org/10.1021/es5003988
Surowsky B, Fischer A, Schlueter O, Knorr D (2013) Cold plasma effects on enzyme activity in a model food system. Innov Food Sci Emerg Technol 19:146–152. https://doi.org/10.1016/j.ifset.2013.04.002
Meinlschmidt P, Ueberham E, Lehmann J, Reineke K, Schlüter O, Schweiggert-Weisz U, Eisner P (2016) The effects of pulsed ultraviolet light, cold atmospheric pressure plasma, and gamma-irradiation on the immunoreactivity of soy protein isolate. Innov Food Sci Emerg Technol 38:374–383. https://doi.org/10.1016/j.ifset.2016.06.007
Li J, Xiang Q, Liu X, Ding T, Zhang X, Zhai Y, Bai Y (2017) Inactivation of soybean trypsin inhibitor by dielectric-barrier discharge (DBD) plasma. Food Chem 232:515–522. https://doi.org/10.1016/j.foodchem.2017.03.167
Alves Filho EG, Silva LMA, Oiram Filho F, Rodrigues S, Fernandes FA, Gallão MI, Mattison CP, de Brito ES (2019) Cold plasma processing effect on cashew nuts composition and allergenicity. Food Res Int 125:108621. https://doi.org/10.1016/j.foodres.2019.108621
Venkataratnam H, Sarangapani C, Cahill O, Ryan CB (2019) Effect of cold plasma treatment on the antigenicity of peanut allergen Ara h 1. Innov Food Sci Emerg Technol 52:368–375. https://doi.org/10.1016/j.ifset.2019.02.001
Bußler S, Steins V, Ehlbeck J, Schlüter O (2015) Impact of thermal treatment versus cold atmospheric plasma processing on the techno-functional protein properties from Pisum sativum ‘Salamanca.’ J Food Eng 167:166–174. https://doi.org/10.1016/j.jfoodeng.2015.05.036
Tammineedi CV, Choudhary R, Perez-Alvarado GC, Watson DG (2013) Determining the effect of UV-C, high intensity ultrasound and nonthermal atmospheric plasma treatments on reducing the allergenicity of α-casein and whey proteins. LWT Food Sci Technol 54(1):35–41. https://doi.org/10.1016/j.lwt.2013.05.020
Pan Y, Cheng JH, Sun DW (2019) Cold plasma-mediated treatments for shelf life extension of fresh produce: a review of recent research developments. Compr Rev Food Sci Food Saf 18(5):1312–1326. https://doi.org/10.1111/1541-4337.12474
Pankaj SK, Thomas S (2016) Cold plasma applications in food packaging. Cold Plasma in Food and Agriculture. Academic Press, pp 293–307. https://doi.org/10.1016/B978-0-12-801365-6.00012-3
Moosavi MH, Khani MR, Shokri B, Hosseini SM, Shojaee-Aliabadi S, Mirmoghtadaie L (2020) Modifications of protein-based films using cold plasma. Int J Biol Macromol 142:769–777. https://doi.org/10.1016/j.ijbiomac.2019.10.017
Li Y, Bai Y, Huang J, Yuan C, Ding T, Liu D, Hu Y (2020) Airglow discharge plasma treatment affects the surface structure and physical properties of zein films. J Food Eng 273:109813. https://doi.org/10.1016/j.jfoodeng.2019.109813
Romani VP, Olsen B, Collares MP, Oliveira JRM, Prentice-Hernández C, Martins VG (2019) Improvement of fish protein films properties for food packaging through glow discharge plasma application. Food Hydrocolloids 87:970–976. https://doi.org/10.1016/j.foodhyd.2018.09.022
Romani VP, Olsen B, Collares MP, Oliveira JRM, Prentice C, Martins VG (2019) Plasma technology as a tool to decrease the sensitivity to water of fish protein films for food packaging. Food Hydrocolloids 94:210–216. https://doi.org/10.1016/j.foodhyd.2019.03.021
Tenn N, Follain N, Fatyeyeva K, Poncin-Epaillard F, Labrugère C, Marais S (2014) Impact of hydrophobic plasma treatments on the barrier properties of poly (lactic acid) films. RSC Adv 4(11):5626–5637. https://doi.org/10.1039/c3ra45323e
Paisoonsin S, Pornsunthorntawee O, Rujiravanit R (2013) Preparation and characterization of ZnO-deposited DBD plasma-treated PP packaging film with antibacterial activities. Appl Surf Sci 273:824–835. https://doi.org/10.1016/j.apsusc.2013.03.026
Thirumdas R, Kadam D, Annapure US (2017) Cold plasma: an alternative technology for the starch modification. Food Biophys 12(1):129–139. https://doi.org/10.1007/s11483-017-9468-5
Wu TY, Sun NN, Chau CF (2018) Application of corona electrical discharge plasma on modifying the physicochemical properties of banana starch indigenous to Taiwan. J Food Drug Anal 26(1):244–251. https://doi.org/10.1016/j.jfda.2017.03.005
Banura S, Thirumdas R, Kaur A, Deshmukh RR, Annapure US (2018) Modification of starch using low pressure radio frequency air plasma. LWT 89:719–724. https://doi.org/10.1016/j.lwt.2017.11.056
Wu TY, Chang CR, Chang TJ, Chang YJ, Liew Y, Chau CF (2019) Changes in physicochemical properties of corn starch upon modifications by atmospheric pressure plasma jet. Food Chem 283:46–51. https://doi.org/10.1016/j.foodchem.2019.01.043
Thirumdas R, Saragapani C, Ajinkya MT, Deshmukh RR, Annapure US (2016) Influence of low-pressure cold plasma on cooking and textural properties of brown rice. Innov Food Sci Emerg Technol 37:53–60. https://doi.org/10.1016/j.ifset.2016.08.009
Thirumdas R, Trimukhe A, Deshmukh RR, Annapure US (2017) Functional and rheological properties of cold plasma treated rice starch. Carbohyd Polym 157:1723–1731. https://doi.org/10.1016/j.carbpol.2016.11.050
Kim HJ, Yong HI, Park S, Kim K, Choe W, Jo C (2015) Microbial safety and quality attributes of milk following treatment with atmospheric pressure encapsulated dielectric barrier discharge plasma. Food Control 47:451–456. https://doi.org/10.1016/j.foodcont.2014.07.053
Vandamme J, Nikiforov A, Dujardin K, Leys C, De Cooman L, Van Durme J (2015) Critical evaluation of non-thermal plasma as an innovative accelerated lipid oxidation technique in fish oil. Food Res Int 72:115–125. https://doi.org/10.1016/j.foodres.2015.03.037
Kulawik P, Alvarez C, Cullen PJ, Aznar-Roca R, Mullen AM, Tiwari B (2018) The effect of non-thermal plasma on the lipid oxidation and microbiological quality of sushi. Innov Food Sci Emerg Technol 45:412–417. https://doi.org/10.1016/j.ifset.2017.12.011
Albertos I, Martín-Diana A, Cullen PJ, Tiwari BK, Ojha SK, Bourke P, Álvarez C, Rico D (2017) Effects of dielectric barrier discharge (DBD) generated plasma on microbial reduction and quality parameters of fresh mackerel (Scomber scombrus) fillets. Innov Food Sci Emerg Technol 44:117–122. https://doi.org/10.1016/j.ifset.2017.07.006
Sarangapani C, Keogh DR, Dunne J, Bourke P, Cullen PJ (2017) Characterization of cold plasma treated beef and dairy lipids using spectroscopic and chromatographic methods. Food Chem 235:324–333. https://doi.org/10.1016/j.foodchem.2017.05.016
Hosseini SM, Rostami S, Hosseinzadeh Samani B, Lorigooini Z (2020) The effect of atmospheric pressure cold plasma on the inactivation of Escherichia coli in sour cherry juice and its qualitative properties. Food Sci Nutr 8(2):870–883. https://doi.org/10.1002/fsn3.1364
Fernandes FA, Santos VO, Rodrigues S (2019) Effects of glow plasma technology on some bioactive compounds of acerola juice. Food Res Int 115:16–22. https://doi.org/10.1016/j.foodres.2018.07.042
Singh R, Prasad P, Mohan R, Verma MK, Kumar B (2019) Radiofrequency cold plasma treatment enhances seed germination and seedling growth in variety CIM-Saumya of sweet basil (Ocimum basilicum L.). J Appl Res Med Aromat Plants 12:78–81. https://doi.org/10.1016/j.jarmap.2018.11.005
Dawood N (2020) Effect of RF plasma on Moringa seeds germination and growth. J Taibah Univ Sci 14(1):279–284. https://doi.org/10.1080/16583655.2020.1713570
Billah M, Sajib SA, Roy NC, Rashid MM, Reza MA, Hasan MM, Talukder MR (2020) Effects of DBD air plasma treatment on the enhancement of black gram (Vigna mungo l.) seed germination and growth. Arch Biochem Biophys 681:108253. https://doi.org/10.1016/j.abb.2020.108253
Sajib SA, Billah M, Mahmud S, Miah M, Hossain F, Omar FB, Roy NP, Hoque KMF, Talukdar MR, Kabir AH, Reza MA (2020) Plasma activated water: the next generation eco-friendly stimulant for enhancing plant seed germination, vigor and increased enzyme activity, a study on black gram (Vigna mungo L.). Plasma Chem Plasma Process 40(1):119–143. https://doi.org/10.1007/s11090-019-10028-3
Boutraa T, Fadhlalmawla SA, Almarashi JQ, Mohamed AAH (2019) Argon cold atmospheric pressure plasma jet enhancing seed germination of Fenugreek (Trigonella Foenum-Graecum). 2019 IEEE Pulsed Power and Plasma Science (PPPS). IEEE, pp 1–5. https://doi.org/10.1109/ppps34859.2019.9009694
Iqbal T, Farooq M, Afsheen S, Abrar M, Yousaf M, Ijaz M (2019) Cold plasma treatment and laser irradiation of Triticum spp. seeds for sterilization and germination. J Laser Appl 31(4):042013. https://doi.org/10.2351/1.5109764
Sadhu S, Thirumdas R, Deshmukh RR, Annapure US (2017) Influence of cold plasma on the enzymatic activity in germinating mung beans (Vigna radiate). LWT 78:97–104. https://doi.org/10.1016/j.lwt.2016.12.026
Zhang JJ, Jo JO, Mongre RK, Ghosh M, Singh AK, Lee SB, Mok YS, Hyuk P, Jeong DK (2017) Growth-inducing effects of argon plasma on soybean sprouts via the regulation of demethylation levels of energy metabolism-related genes. Sci Rep 7:41917. https://doi.org/10.1038/srep41917
Baldanov BB, Ranzhurov TV, Sordonova MN, Budazhapov LV (2020) Changes in the properties and surface structure of grain seeds under the influence of a glow discharge at atmospheric pressure. Plasma Phys Rep 46(1):110–114. https://doi.org/10.1134/S1063780X2001002X
Liao X, Li J, Muhammad AI, Suo Y, Chen S, Ye X, Liu D, Ding T (2018) Application of a dielectric barrier discharge atmospheric cold plasma (Dbd-Acp) for Eshcerichia coli inactivation in apple juice. J Food Sci 83(2):401–408. https://doi.org/10.1111/1750-3841.14045
Xiang Q, Liu X, Li J, Liu S, Zhang H, Bai Y (2018) Effects of dielectric barrier discharge plasma on the inactivation of Zygosaccharomyces rouxii and quality of apple juice. Food Chem 254:201–207. https://doi.org/10.1016/j.foodchem.2018.02.008
Won MY, Lee SJ, Min SC (2017) Mandarin preservation by microwave-powered cold plasma treatment. Innov Food Sci Emerg Technol 39:25–32. https://doi.org/10.1016/j.ifset.2016.10.021
Patange A, Boehm D, Bueno-Ferrer C, Cullen PJ, Bourke P (2017) Controlling Brochothrix thermosphacta as a spoilage risk using in-package atmospheric cold plasma. Food Microbiol 66:48–54. https://doi.org/10.1016/j.fm.2017.04.002
Moritz M, Wiacek C, Koethe M, Braun PG (2017) Atmospheric pressure plasma jet treatment of Salmonella Enteritidis inoculated eggshells. Int J Food Microbiol 245:22–28. https://doi.org/10.1016/j.ijfoodmicro.2017.01.004
Min SC, Roh SH, Niemira BA, Boyd G, Sites JE, Uknalis J, Fan X (2017) In-package inhibition of E. coli O157: H7 on bulk Romaine lettuce using cold plasma. Food Microbiol 65:1–6. https://doi.org/10.1016/j.fm.2017.01.010
Devi Y, Thirumdas R, Sarangapani C, Deshmukh RR, Annapure US (2017) Influence of cold plasma on fungal growth and aflatoxins production on groundnuts. Food Control 77:187–191. https://doi.org/10.1016/j.foodcont.2017.02.019
Dasan BG, Mutlu M, Boyaci IH (2016) Decontamination of Aspergillus flavus and Aspergillus parasiticus spores on hazelnuts via atmospheric pressure fluidized bed plasma reactor. Int J Food Microbiol 216:50–59. https://doi.org/10.1016/j.ijfoodmicro.2015.09.006
Chutia H, Kalita D, Mahanta CL, Ojah N, Choudhury AJ (2019) Kinetics of inactivation of peroxidase and polyphenol oxidase in tender coconut water by dielectric barrier discharge plasma. LWT 101:625–629. https://doi.org/10.1016/j.lwt.2018.11.071
Tappi S, Berardinelli A, Ragni L, Dalla Rosa M, Guarnieri A, Rocculi P (2014) Atmospheric gas plasma treatment of fresh-cut apples. Innov Food Sci Emerg Technol 21:114–122. https://doi.org/10.1016/j.ifset.2013.09.012
Khani MR, Shokri B, Khajeh K (2017) Studying the performance of dielectric barrier discharge and gliding arc plasma reactors in tomato peroxidase inactivation. J Food Eng 197:107–112. https://doi.org/10.1016/j.jfoodeng.2016.11.012
Chen HH, Hung CL, Lin SY, Liou GJ (2015) Effect of low-pressure plasma exposure on the storage characteristics of brown rice. Food Bioprocess Technol 8(2):471–477. https://doi.org/10.1007/s11947-014-1415-6
Illera AE, Chaple S, Sanz MT, Ng S, Lu P, Jones J, Carey E, Bourke P (2019) Effect of cold plasma on polyphenol oxidase inactivation in cloudy apple juice and on the quality parameters of the juice during storage. Food Chem X 3:100049. https://doi.org/10.1016/j.fochx.2019.100049
Amanpour A, Vandamme J, Polat S, Kelebek H, Van Durme J, Selli S (2019) Non-thermal plasma effects on the lipoxygenase enzyme activity, aroma and phenolic profiles of olive oil. Innov Food Sci Emerg Technol 54:123–131. https://doi.org/10.1016/j.ifset.2019.04.004
Tappi S, Ramazzina I, Rizzi F, Sacchetti G, Ragni L, Rocculi P (2018) Effect of plasma exposure time on the polyphenolic profile and antioxidant activity of fresh-cut apples. Appl Sci 8(10):1939. https://doi.org/10.3390/app8101939
Dasan BG, Boyaci IH (2018) Effect of cold atmospheric plasma on inactivation of Escherichia coli and physicochemical properties of apple, orange, tomato juices, and sour cherry nectar. Food Bioprocess Technol 11(2):334–343. https://doi.org/10.1007/s11947-017-2014-0
Kovačević DB, Kljusurić JG, Putnik P, Vukušić T, Herceg Z, Dragović-Uzelac V (2016) Stability of polyphenols in chokeberry juice treated with gas phase plasma. Food Chem 212:323–331. https://doi.org/10.1016/j.foodchem.2016.05.192
Shi H, Cooper B, Stroshine RL, Ileleji KE, Keener KM (2017) Structures of degradation products and degradation pathways of aflatoxin B1 by high-voltage atmospheric cold plasma (HVACP) treatment. J Agric Food Chem 65(30):6222–6230. https://doi.org/10.1021/acs.jafc.7b01604
Heidemann HM, Dotto ME, Laurindo JB, Carciofi BA, Costa C (2019) Cold plasma treatment to improve the adhesion of cassava starch films onto PCL and PLA surface. Colloids Surf A 580:123739. https://doi.org/10.1016/j.colsurfa.2019.123739
Dong S, Guo P, Chen GY, Jin N, Chen Y (2020) Study on the atmospheric cold plasma (ACP) treatment of zein film: surface properties and cytocompatibility. Int J Biol Macromol 153:1319–1327. https://doi.org/10.1016/j.ijbiomac.2019.10.268
Paneru R, Lamichhane P, Adhikari CB, Ki SH, Choi J, Kwon JS, Choi EH (2019) Surface modification of PVA thin film by nonthermal atmospheric pressure plasma for antifogging property. AIP Adv 9(7):075008. https://doi.org/10.1063/1.5100776
Dong S, Guo P, Chen Y, Chen GY, Ji H, Ran Y, Li S, Chen Y (2018) Surface modification via atmospheric cold plasma (ACP): improved functional properties and characterization of zein film. Ind Crops Prod 115:124–133. https://doi.org/10.1016/j.indcrop.2018.01.080
Honarvar Z, Farhoodi M, Khani MR, Mohammadi A, Shokri B, Ferdowsi R, Shojaee-Aliabadi S (2017) Application of cold plasma to develop carboxymethyl cellulose-coated polypropylene films containing essential oil. Carbohyd Polym 176:1–10. https://doi.org/10.1016/j.carbpol.2017.08.054
Pankaj SK, Bueno-Ferrer C, Misra NN, O’Neill L, Tiwari BK, Bourke P, Cullen PJ (2015) Dielectric barrier discharge atmospheric air plasma treatment of high amylose corn starch films. LWT Food Sci Technol 63(2):1076–1082. https://doi.org/10.1016/j.lwt.2015.04.027
Kostov KG, Nishime TMC, Castro AHR, Toth A, Hein LRDO (2014) Surface modification of polymeric materials by cold atmospheric plasma jet. Appl Surf Sci 314:367–375. https://doi.org/10.1016/j.apsusc.2014.07.009
Ulbin-Figlewicz N, Brychcy E, Jarmoluk A (2015) Effect of low-pressure cold plasma on surface microflora of meat and quality attributes. J Food Sci Technol 52(2):1228–1232. https://doi.org/10.1007/s13197-013-1108-6
Choi S, Puligundla P, Mok C (2016) Corona discharge plasma jet for inactivation of Escherichia coli O157: H7 and Listeria monocytogenes on inoculated pork and its impact on meat quality attributes. Ann Microbiol 66(2):685–694. https://doi.org/10.1007/s13213-015-1147-5
Thirumdas R, Deshmukh RR, Annapure US (2016) Effect of low temperature plasma on the functional properties of basmati rice flour. J Food Sci Technol 53(6):2742–2751. https://doi.org/10.1007/s13197-016-2246-4
Lee KH, Kim HJ, Woo KS, Jo C, Kim JK, Kim SH, Park HY, Oh SK, Kim WH (2016) Evaluation of cold plasma treatments for improved microbial and physicochemical qualities of brown rice. LWT 73:442–447. https://doi.org/10.1016/j.lwt.2016.06.055
Sarangapani C, Devi Y, Thirundas R, Annapure US, Deshmukh RR (2015) Effect of low-pressure plasma on physico-chemical properties of parboiled rice. LWT Food Sci Technol 63(1):452–460. https://doi.org/10.1016/j.lwt.2015.03.026
Thirumdas R, Sarangapani C, Annapure US (2015) Cold plasma: a novel non-thermal technology for food processing. Food Biophys 10(1):1–11. https://doi.org/10.1007/s11483-014-9382-z
Porto CL, Sergio L, Boari F, Logrieco AF, Cantore V (2019) Cold plasma pretreatment improves the germination of wild asparagus (Asparagus acutifolius L.) seeds. Sci Hortic 256:108554. https://doi.org/10.1016/j.scienta.2019.108554
Kumar R, Thakur AK, Vikram A, Vaid A, Rane R (2019) Effect of cold plasma treatment of seeds on quality of seed crop of okra. Int J Econ Plants 6(2):73–77
Yodpitak S, Mahatheeranont S, Boonyawan D, Sookwong P, Roytrakul S, Norkaew O (2019) Cold plasma treatment to improve germination and enhance the bioactive phytochemical content of germinated brown rice. Food Chem 289:328–339. https://doi.org/10.1016/j.foodchem.2019.03.061
Los A, Ziuzina D, Boehm D, Cullen PJ, Bourke P (2019) Investigation of mechanisms involved in germination enhancement of wheat (Triticum aestivum) by cold plasma: effects on seed surface chemistry and characteristics. Plasma Processes Polym 16(4):1800148. https://doi.org/10.1002/ppap.201800148
de Groot GJ, Hundt A, Murphy AB, Bange MP, Mai-Prochnow A (2018) Cold plasma treatment for cotton seed germination improvement. Sci Rep 8(1):1–10. https://doi.org/10.1038/s41598-018-32692-9
Ling LI, Jiangang LI, Minchong SHEN, Jinfeng H, Hanliang S, Yuanhua D, Jiafeng J (2016) Improving seed germination and peanut yields by cold plasma treatment. Plasma Sci Technol 18(10):1027. https://doi.org/10.1088/1009-0630/18/10/10
Acknowledgements
We are thankful to the CEO, CIAB, for his continuous support and encouragement.
Funding
This study is financially supported by the Department of Biotechnology, Government of India.
Author information
Authors and Affiliations
Contributions
DM and SKY conceived the idea. DM collected the literature and prepared the draft. SKY edited the article.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Cold plasma exhibits antimicrobial effect and enzyme denaturation property.
• Cold plasma ensures food quality, better food safety, and food packaging.
• Cold plasma is an eco-friendly solution for food and agriculture industry.
• Cold plasma-treated food retains texture, nutritional, and sensory characteristics.
• Future scope of cold plasma technology in food sector.
Rights and permissions
About this article
Cite this article
Mehta, D., Yadav, S.K. Recent Advances in Cold Plasma Technology for Food Processing. Food Eng Rev 14, 555–578 (2022). https://doi.org/10.1007/s12393-022-09317-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12393-022-09317-z