Abstract
Background
The aim of the present study was to evaluate the uptake of F18-NaF by the arterial wall in patients with high cardiovascular (CV) risk profile. The tracer uptake was assessed in relation to gender and the number of CV risk factors.
Methods and Results
25 patients without known CV disease were included and evaluated by PET-CT with F18-NaF: 14 (56%) men and 11 (44%) women. The mean target-to-background ratio (TBR: max SUV/mean blood-pool SUV) but not the corrected uptake per lesion (CUL: max SUV – mean blood-pool SUV) was higher in men than women (TBR: 1.8 ± 0.6 vs 1.7 ± 0.2; P = 0.04; CUL: 0.7 ± 0.3 vs W 0.6 ± 0.1; P = 0.4). Patients with >3 CV risk factors had higher CUL (0.8 ± 0.1 vs 0.6 ± 0.2; P = 0.01) but not TBR (1.8 ± 0.2 vs 1.7 ± 0.6; P = 0.7) than patients with <3 risk factors.
Conclusions
The TBR but not CUL is higher in men than women while the CUL but not TBR is related to the number of CV risk factors. These results are hypothesis—generating and require validation in larger studies.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Background
Atherosclerosis is a chronic inflammatory condition that affects the arterial wall and is characterized by the formation of atherosclerotic plaques. These are the sites where thrombotic events occur, and for those, neither the size nor the degree of luminal narrowing, imposed by the plaques, are as important as their biological content and inflammatory activity.1,2,3
There has been a growing interest in predicting noninvasively, the risk of events, by analyzing the degree of calcification and inflammation of the plaques. For that purpose, positron emission computed tomography (PET), multi-slice computed tomography (CT), and cardiac magnetic resonance imaging have played major roles.4,5,6
For a few years, PET-CT, with fluorine-18 fluorodeoxyglucose (F18-FDG), has been the imaging method of choice in the identification of sites of vascular inflammation especially in the aortic and carotid territories. The results show that the F18-FDG uptake is a marker of inflammation and is related to future cardiovascular (CV) events.7 The major limitation of this tracer is its strong uptake by the normal myocardium, which makes it difficult to evaluate the coronary uptake.8,9,10,11,12,13
Fluorine-18 sodium fluoride (F18-NaF) is used to detect bone metastases. Retrospective studies showed that this tracer is also taken by the vascular wall and its uptake level has been associated with atherosclerotic risk factor burden.14,15 The F18-NaF uptake seems to signal active calcification which is a cellular response to a chronic inflammatory state. The hydroxyapatite crystal is the major component of vascular calcification in early stages. Fluoride ions are incorporated into the crystal by ion exchange with hydroxyl groups.16,17 According to recent studies, this marker seems to identify higher risk plaques.18,19
The aim of the present study was to evaluate the uptake of F18-NaF in the arterial wall in patients at high CV risk. The uptake of the tracer was assessed in relation to gender and the number of CV risk factors.
Methods
Population and Study Design
Patients aged >40 years and considered of being of high CV risk according to the European Society of Cardiology (ESC) Guidelines were included based on their acceptance in participating in this prospective and exploratory study between May 1, 2014 and June 1, 2015. These patients were being followed in the outpatient clinic, of a tertiary facility university hospital, due to hypertension.
Inclusion Criteria
For the purpose of this study, we included individuals with a predicted fatal CV event rate at 10 years ≥5% (ESC SCORE tables for low-risk countries),20 chronic kidney disease with glomerular filtration rate under 60 mL·min−1 (Modification of Diet in Renal Disease equation), diabetes mellitus (type 1 or 2), or markedly abnormal single-risk factor.
Exclusion Criteria
Patients <40 years old and women of childbearing age were excluded. For the purpose of this study, patients with known CV disease such as coronary artery disease, cerebral vascular disease, or peripheral arterial disease—or with symptoms suggestive of disease (angina, neurological complaints or claudication)—patients with severe renal impairment (GFR < 30 mL·min−1), moderate to severe hepatic failure (Child-Pugh class B or C), chronic inflammatory diseases, and neoplastic disease (in the last 5 years) were also excluded.
All patients underwent a baseline clinical evaluation where the CV risk profile was assessed. For that purpose, a blood sample was drawn to measure the levels of total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, fasting glucose, hemoglobin A1c (HbA1c), creatinine, and C-reactive protein.
A F18-NaF PET-CT study was performed, to assess the uptake of the tracer in the carotid arteries, coronary arteries, and aorta, in the same day of clinical and biochemical evaluation.
All the procedures were done with the approval of the ethics committee of the Faculty of Medicine of the University of Coimbra, in accordance with the Declaration of Helsinki and with the written informed consent of each participant.
Image Acquisition, Data Processing, and Calculation of Outcome Measures
A Gemini GXL Philips 16 PET/CT system was used, and image acquisition was started 60 minutes after intravenous administration of 185 MBq of F18-NaF (5 mCi).
The enrolled patients underwent a cardiac PET-CT imaging of the heart. An attenuation correction CT scan (non-enhanced 120 kV and 50 mA) was performed, followed by an electrocardiogram-gated 10-min acquisition. An iterative reconstruction of cardiac PET scan was accomplished in multiple phases. For analysis, the diastolic phase between 50% and 75% was used.
To assess aorta and carotid vessels, a whole-body PET-CT acquisition was performed. The corresponding bounding box was defined by the base of the skull, the coccygeal bone, the sternum, the spinous process of L5, and the sacroiliac joints.
Imaging started with a non-enhanced, low-dose CT scan (120 kV and 50 mA). PET images were acquired, in list mode, taking 5 minutes per bed position and an overlap between consecutive bed positions of 50%.
Transversal PET slices were reconstructed into a 144 × 144 matrix. Voxel size was of 4 × 4 × 4 mm, and the spatial resolution was about 8 mm. Low-dose CT imaging was performed for attenuation correction and anatomical orientation.
Arterial uptake of the tracer was evaluated, after qualitative review of the images in the coronal, axial, and sagittal PET reconstructions. The localization of areas of focal uptake, in relation to the vascular wall, was determined by PET/CT fusion images. Semi-quantitative analysis was accomplished using the maximum standardized uptake value (SUV max) by visually placing an individual region of interest (ROI) around the lesion. Blood-pool SUV was estimated as the mean of five ROI in the mid lumen of superior vena cava. The SUV max was then corrected by the blood-pool SUV, dividing SUV max by the blood-pool SUV, thereby obtaining the target-to-background ratio (TBR). SUV max was also corrected by the blood-pool SUV subtracting the blood-pool SUV from the SUV max, resulting in the corrected uptake per lesion (CUL). For the purpose of this study, the uptake of F18-NaF in each vessel territory (aortic, carotid, coronary, and all vascular territories) was described as the mean of SUV max, TBR, and CUL obtained by the sum of the SUV max, TBR, and CUL of each lesion divided by the number of lesions. All steps were reviewed by 3 different readers.
Statistical Analysis
Continuous data were tested for normality with the Shapiro-Wilks test and was represented by their mean, median, and interquartile range. Student’s t test and the Mann-Whitney test were applied to compare continuous variables as appropriate. Categorical variables were represented by their frequency and compared using Fisher’s exact test or the χ 2-test. Data analysis was performed by StatView 5.0.1, version for Macintosh and Windows, SAS Institute.
Results
Twenty-five patients were included: 14 (56%) men and 11 (44%) women with a mean age of 63.9 ± 8.6 years. The clinical data of the studied patients are shown in Tables 1 and 2.
As these patients have a high CV risk, the prevalence of risk factors such as diabetes and hyperlipidemia was very high among the studied group. Considering the overall BMI, overweight and obese patients were included, and as expected, high mean values of systolic blood pressure were found. More women than men had a positive family history of CV disease. All the other compared variables showed no statistically significant differences. There were 7 (28%) patients with >3 risk factors. Biochemical measurements are presented in Table 3. There was a trend for higher values in women.
The medications at time of enrollment are described in Table 4. Most patients were on one or more medication but there were no significant differences between men and women.
Imaging Results
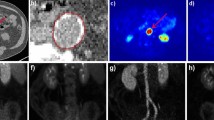
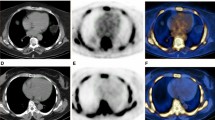
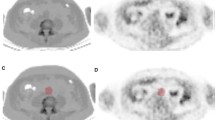
One hundred and thirty-one lesions with F18-NaF uptake were identified in 24 patients, and 71 were also calcified (54.2%). One patient had no F18-NaF uptake. Figure 1 shows the number of observed lesions per arterial territory, in the overall population and in both genders. PET-CT images of a calcified and a non-calcified lesion, with F18-NaF uptake, could be seen in Figure 2.
The characterization of F18-NaF uptake according to the mean number of lesions and its quantification, considering the different correction methods for blood-pool activity, is shown in Table 5. The number of F18-NaF lesions and their quantification according to gender are shown in Table 6.
The number of lesions and consequently the tracer uptake were higher in the aorta. There was no difference in the number of lesions with tracer uptake between men and women. The aorta and the overall F18-NaF uptake, as measured by the TBR, were higher in men than women; the SUV max and CUL were not different, however. Carotid and coronary uptake were similar in both genders.
The uptake of F18-NaF was also examined in relation to the number of CV risk factors (Table 7). Those with >3 risk factors had a higher F18-NaF uptake (SUV max and CUL) than patients with <3 risk factors. These differences could be seen in the overall uptake, in the aorta and coronary vessels. In the overall arterial territories, these patients had also a higher number of lesions with tracer uptake.
Discussion
Recent publications showed that sodium fluoride seems to have the ability to identify high-risk atherosclerotic plaques.18 This was the main conclusion of the study by Joshi et al, in 2014.18 In that study, PET-CT was performed in patients with an acute myocardial infarction, with stable angina and with carotid disease. F18-NaF had the highest uptake in culprit plaques, in ruptured carotid plaques, associated with histological evidence of active calcification, and in coronary plaques related with high-risk features by intravascular ultrasound. Myocardial uptake of F18-FDG interfered with the visualization of coronary uptake.18
Before this landmark study, there were a few publications that addressed this subject mainly in oncologic patients. From these studies, it has become clear that F18-FDG and F18-NaF identify different pathophysiologic processes in atherosclerotic lesions and both correlated with the presence of CV risk factors.14,15,18,21
The present study was an exploratory hypothesis generating, prospective study, that involves asymptomatic patients who have a high CV risk profile but without known CV disease. The uptake of F18-NaF was quantified and examined in relation to gender and the number of CV risk factors.
Our results showed that 96% of the studied patients had at least one arterial lesion with F18-NaF uptake. Compared with previous studies,14,15,21 we found higher rates of tracer uptake probably related to the high CV risk profile of the studied population.
To assess F18-NaF uptake, TBR and CUL were used. Quantification of F18-NaF uptake is still controversial. Our option, in using two different methods of correction for blood-pool activity, was supported by the results of a study by Blomberg et al, which described a higher influence of blood-pool activity in the estimation of arterial TBR compared to arterial CUL.22
The results of F18-NaF uptake were different when TBR or CUL were considered. A higher overall and aortic uptake was observed in men, when TBR was used, while no significant differences were seen when CUL was used. F18-NaF uptake correlated with the number of risk factors; higher number of lesions with significantly higher values of SUV max and CUL in the aorta, coronary, and in the overall arterial territories, in patients with >3 RF than <3 risk factors.
In this asymptomatic population with a high CV risk profile, the presence of areas, in the vessel wall, with active calcification is almost similar in both genders, despite slight differences in TBR which could be due to the influence of blood-pool activity.22
All the studied patients had high CV risk profile and, in spite of the 10 years gap, between men and women, in what concerns CV disease,20 no significant differences were seen between men and women in carotid and coronary uptake.
The relation between CUL and the number of risk factors, even in the coronary territory, where only a few lesions were seen, may suggest a different performance than TBR.22
Calcification was seen in ~54% of the lesions with fluoride uptake. Studies show that non-calcified plaque burden is seen in patients with CV risk factors and calcification seems to result in plaque stabilization, appearing as a natural consequence of inflammation and active microcalcification; a biological process nicely depicted by PET-CT with F18-NaF.23,24,25,26 So it was not surprising that about half of the lesions with F18-NaF uptake were also calcified, and it seems that with this imaging technique, a different stage of atherosclerotic disease could be identified.
A few considerations, regarding the technical aspects of the imaging must be clarified.
In this study, a dose of 185 MBq of F18-NaF (mean of 2.5 MBq·kg−1 per patient) were administered, which was lower than the dose used in some studies14,15 and the lowest recommended dose for bone scans.27 In image acquisition, the extended time per bed, higher than the time described by others,14,15 was a logical compensation for the reduced dose. Correction for blood-pool activity was performed drawing five ROI in the superior vena cava. Recent reports questioned the stability of blood-pool measurements in this vessel and referred that its small diameter could cause a higher susceptibility to partial volume effects.28 Sampling blood-pool from the right atrium seemed to overcome this limitation but it was not our option in this study.29
New Knowledge Gained
As far as we know, this was the first attempt to characterize, in what concerns F18-NaF uptake, an asymptomatic high cardiovascular risk population. The uptake was quantified concerning gender and the number of CV risk factors. Besides the TBR, another outcome measure was used in this study, the CUL, and the results, although limited by the relatively small sample size, raise questions about the ideal way of correcting tracer uptake to blood-pool activity.
Limitations
The number of patients studied was relatively small, and so, our results and conclusions should be considered hypothesis generating in need to be validated in a larger population. In order to evaluate cardiovascular outcome, a long follow-up is required.
The ideal dose of F18-NaF for these kinds of studies remains to be established. Quantification of sodium fluoride uptake in the vascular wall is still controversial and the choice of the site for blood-pool activity evaluation, and the used outcome measure could influence conclusions.
Abbreviations
- CT:
-
Computed tomography
- CV:
-
Cardiovascular
- CUL:
-
Corrected uptake per lesion
- F18-FDG:
-
Fluorine-18 fluoro-deoxy-glucose
- F18-NaF:
-
Fluorine-18 sodium fluoride
- PET:
-
Positron emission computed tomography
- ROI:
-
Region of interest
- SUV:
-
Standardized uptake value
References
Bentzon JF, Otsuka F, Virmani R, Falk E. Mechanisms of plaque formation and rupture. Circ Res. 2014;114:1852-66.
Libby P, Tabas I, Fredman G, Fisher EA. Inflammation and its resolution as determinants of acute coronary syndromes. Circ Res. 2014;114:1867-79.
Otsuka F, Joner M, Prati F, Virmani R, Narula J. Clinical classification of plaque morphology in coronary disease. Nat Rev Cardiol. 2014;11:379-89.
Beller GA. Recent advances and future trends in multimodality cardiac imaging. Heart Lung Circ. 2010;19:193-209.
Tahara N, Tahara A, Honda A, Nitta Y, Kodama N, Yamagishi S, et al. Molecular imaging of vascular inflammation. Curr Pharm Des. 2014;20:2439-47.
Tarkin JM, Rudd JH. Techniques for noninvasive molecular imaging of atherosclerotic plaque. Nat Rev Cardiol. 2015;12:79.
Figueroa AL, Abdelbaky A, Truong QA, Corsini E, MacNabb MH, Lavender ZR, et al. Measurement of arterial activity on routine FDG PET/CT images improves prediction of risk of future CV events. JACC Cardiovasc. Imaging. 2013;6:1250-9.
Ben-Haim S, Kupzov E, Tamir A, Israel O. Evaluation of 18F-FDG uptake and arterial wall calcifications using 18F-FDG PET/CT. J Nucl Med. 2004;45:1816-21.
Chen W, Bural GG, Torigian DA, Rader DJ, Alavi A. Emerging role of FDG-PET/CT in assessing atherosclerosis in large arteries. Eur J Nucl Med Mol Imaging. 2009;36:144-51.
Wasselius JA, Larsson SA, Jacobsson H. Fdg-accumulating atherosclerotic plaques identified with 18F-FDG-PET/CT in 141 patients. Mol Imaging Biol. 2009;11:455-9.
Sheikine Y, Akram K. Fdg-pet imaging of atherosclerosis: Do we know what we see? Atherosclerosis. 2010;211:371-80.
Meirelles GS, Gonen M, Strauss HW. 18F-Fdg uptake and calcifications in the thoracic aorta on positron emission tomography/computed tomography examinations: Frequency and stability on serial scans. J Thorac Imaging. 2011;26:54-62.
Hetterich H, Rominger A, Walter L, Habs M, Volpers S, Hacker M, et al. Natural history of atherosclerotic disease progression as assessed by (18)F-FDG PET/CT. Int J Cardiovasc Imaging. 2016;32:49-59.
Derlin T, Richter U, Bannas P, Begemann P, Buchert R, Mester J, et al. Feasibility of 18F-sodium fluoride PET/CT for imaging of atherosclerotic plaque. J Nucl Med. 2010;51:862-5.
Derlin T, Wisotzki C, Richter U, Apostolova I, Bannas P, Weber C, et al. In vivo imaging of mineral deposition in carotid plaque using 18F-sodium fluoride PET/CT: Correlation with atherogenic risk factors. J Nucl Med. 2011;52:362-8.
Dweck MR, Chow MW, Joshi NV, Williams MC, Jones C, Fletcher AM, et al. Coronary arterial 18F-sodium fluoride uptake: A novel marker of plaque biology. J Am Coll Cardiol. 2012;59:1539-48.
Adamson PD, Vesey AT, Joshi NV, Newby DE, Dweck MR. Salt in the wound: (18)F-fluoride positron emission tomography for identification of vulnerable coronary plaques. Cardiovasc Diagn Ther. 2015;5:150-5.
Joshi NV, Vesey AT, Williams MC, Shah AS, Calvert PA, Craighead FH, et al. 18F-fluoride positron emission tomography for identification of ruptured and high-risk coronary atherosclerotic plaques: A prospective clinical trial. Lancet. 2014;383:705-13.
Joshi NV, Vesey A, Newby DE, Dweck MR. Will 18F-sodium fluoride PET-CT imaging be the magic bullet for identifying vulnerable coronary atherosclerotic plaques? Curr Cardiol Rep. 2014;16:521.
Piepoli MF, Hoes AW, Agewall S, Albus C, Brotons C, Catapano AL, et al. European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of cardiology and other societies on cardiovascular disease prevention in clinical practice (constituted by representatives of 10 societies and by invited experts): Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur Heart J. 2016;37:2315-81.
Derlin T, Toth Z, Papp L, Wisotzki C, Apostolova I, Habermann CR, et al. Correlation of inflammation assessed by 18F-FDG PET, active mineral deposition assessed by 18F-fluoride pet, and vascular calcification in atherosclerotic plaque: A dual-tracer PET/CT study. J Nucl Med. 2011;52:1020-7.
Blomberg BA, Thomassen A, de Jong PA, Simonsen JA, Lam MG, Nielsen AL, et al. Impact of personal characteristics and technical factors on quantification of sodium 18F-fluoride uptake in human arteries: Prospective evaluation of healthy subjects. J Nucl Med. 2015;56:1534-40.
Gottlieb I, Miller JM, Arbab-Zadeh A, Dewey M, Clouse ME, Sara L, et al. The absence of coronary calcification does not exclude obstructive coronary artery disease or the need for revascularization in patients referred for conventional coronary angiography. J Am Coll Cardiol. 2010;55:627-34.
New SE, Aikawa E. Molecular imaging insights into early inflammatory stages of arterial and aortic valve calcification. Circ Res. 2011;108:1381-91.
Otsuka F, Sakakura K, Yahagi K, Joner M, Virmani R. Has our understanding of calcification in human coronary atherosclerosis progressed? Arterioscler Thromb Vasc Biol. 2014;34:724-36.
Puri R, Nicholls SJ, Shao M, Kataoka Y, Uno K, Kapadia SR, et al. Impact of statins on serial coronary calcification during atheroma progression and regression. J Am Coll Cardiol. 2015;65:1273-82.
Segall G, Delbeke D, Stabin MG, Even-Sapir E, Fair J, Sajdak R, et al. Snm practice guideline for sodium 18F-fluoride PET/CT bone scans 1.0. J Nucl Med. 2010;51:1813-20.
Chen W, Dilsizian V. Pet assessment of vascular inflammation and atherosclerotic plaques: SUV or TBR? J Nucl Med. 2015;56:503-4.
Pawade TA, Cartlidge TR, Jenkins WS, Adamson PD, Robson P, Lucatelli C, et al. Optimization and reproducibility of aortic valve 18F-fluoride positron emission tomography in patients with aortic stenosis. Circ Cardiovasc Imaging. 2016. doi:10.1161/CIRCIMAGING.116.005131.
Disclosure
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ferreira, M.J.V., Oliveira-Santos, M., Silva, R. et al. Assessment of atherosclerotic plaque calcification using F18-NaF PET-CT. J. Nucl. Cardiol. 25, 1733–1741 (2018). https://doi.org/10.1007/s12350-016-0776-9
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12350-016-0776-9