Abstract
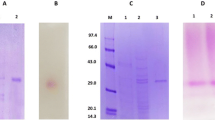
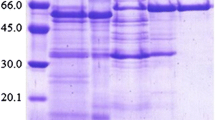
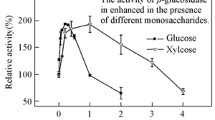
An extracellular β-glucosidase was extracted from the culture filtrate of Aspergillus niger No. 5.1 and purified to homogeneity by using ammonium sulfate precipitation, Chitopearl-DEAE chromatography, and Sephadex G-100 chromatography. The specific activity of the enzyme was enriched 6.33-fold, with a recovery of 11.67%. The enzyme was a monomer and the molecular mass was 67.5 kDa by sodium dodecyl sulfate polyacrylamide gel electrophoresis and 66.5 kDa by gel-filtration chromatography. The enzyme had optimum activity at pH 6.0 and 60°C and was stable over the pH range of 3.0–9.0. It showed specificity of hydrolysis for p-nitrophenyl-β-d-glucoside and cellobiose. The K m and V max values of the enzyme for cellobiose and salicin were 5.34 mM, 2.57 µmol/(mL·s), and 3.09 mM, 1.34 µmol/(mL·s), respectively. Both amino acid composition and N-terminal amino acid sequence of the enzyme were determined, which provides useful information for cloning of this enzyme.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
International Union of Biochemistry. (1984), in Enzyme Nomenclature, Webb, E. C., ed., Academic, Orlando, FL, pp. 310, 311.
Shewale, J. G. (1982), Int. J. Biochem. 14, 435–443.
Esen, A. (1993), in β-Glucosidases—Biochemistry and Molecular Biology, ACS Symposium Series 533, Esen, A., ed., American Chemical Society, Washington, DC, pp. 1–14.
Christakopoulos, P., Goodenough, P. W., Kekos, D., Macris, B. J., Claeyssens, M., and Bhat, M. K. (1994), Eur. J. Biochem. 224, 379–385.
Fujimoto, H., Nishida, H., and Ajisaka, K. (1988), Agric. Biol. Chem. 52, 1345–1351.
Sasaki, K., Tachiki, T., and Tochikura, T. (1989), Agric. Biol. Chem. 53, 313–318.
Shinoyama, H., Takei, K., Ando, A., Fujii, T., Sasaki, M., and Doi, Y. (1991), Agric. Biol. Chem. 55, 1679–1681.
Cappellini, R. A. and Peterson, J. L. (1965), Mycologia 57, 962–966.
Miller, G. L. (1959), Anal. Chem. 31, 426–429.
Bradford, M. M. (1976), Anal. Biochem. 72, 248–254.
Laemmli, U. K. (1970), Nature 227, 680–685.
Heupel, C., Schlochtermeier, A., and Schrempf, H. (1993), Enzyme Microb. Technol. 15, 127–132.
Ohmiya, K., Shirai, M., Kurachi, Y., and Shimizu, S. (1985), J. Bacteriol. 161, 432–434.
Deshpande, V., Eriksson, K. E., and Pettersson, B. (1978), Eur. J. Biochem. 90, 191–198.
Himmel, M. E., Adney, W. S., Fox, J. W., Mitchell, D. J., and Baker, J. O. (1993), Appl. Biochem. Biotechnol. 39/40, 213–225.
Hoh, Y. K., Yeoh, H. H., and Tan, T. K. (1992), Appl. Microbiol. Biotechnol. 37, 590–593.
Yan, T. R., Lin, Y. H., and Lin, C. L. (1998), J. Agric. Food Chem. 46, 431–437.
Sano, K., Amemura, A., and Harada, T. (1975), Biochim. Biophys. Acta 377, 410–420.
Yeoh, H. H., Tan, T. K., and Koh, S. K. (1986), Appl. Microbiol. Biotechnol. 25, 25–28.
Yan, T. R. and Lin, C. L. (1997), Biosci. Biotech. Biochem. 61, 965–970.
Kitpreechavanich, V., Hayashi, M., and Nagai, S. (1986), Agric. Biol. Chem. 50, 1703–1711.
Sakamoto, R., Kanamoto, J., Arai, M., and Murao, S. (1985), Agric. Biol. Chem. 49, 1275–1281.
Mandels, M. and Reese, E. T. (1963), in Advances in Enzymic Hydrolysis of Cellulases and Related Materials, Reese, E. T., ed., Pergamon, London, pp. 115–157.
Sanyal, A., Kundu, R. K., Dube, S., and Dube, D. K. (1988), Enzyme Microb. Technol. 10, 91–99.
Watanabe, T., Sato, T., Yoshioka, S., Koshijima, T., and Kuwahara, M. (1992), Eur. J. Biochem. 209, 651–659.
Workman, W. E. and Day, D. F. (1982), Appl. Environ. Microbiol. 44, 1289–1295.
Bause, E. and Legler, G. (1980), Biochim. Biophys. Acta 626, 459–465.
Machida, M., Ohtsuki, I., Fukui, S., and Yamashita, I. (1988), Appl. Environ. Microbiol. 54, 3147–3155.
Moranelli, F., Barbier, J. R., Dove, M. J., Mackay, R. M., Seligy, V. L., Yaguchi, M., and Willick, G. E. (1986), Biochem. Int. 12, 905–912.
Li, C. P., Swain, E., and Poulton, J. E. (1992), Plant Physiol. 100, 282–290.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xie, Y., Gao, Y. & Chen, Z. Purification and characterization of an extracellular β-glucosidase with high transglucosylation activity and stability from Aspergillus niger No. 5.1. Appl Biochem Biotechnol 119, 229–240 (2004). https://doi.org/10.1007/s12010-004-0004-y
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s12010-004-0004-y