Summary
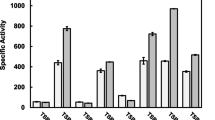
Kinetic properties of extracellular β-glucosidase from Aspergillus ornatus were determined. The pH and temperature optima for the enzyme were found to be 4.6 and 60°C, respectively. Under these conditions, the enzyme exhibited a K m (p-nitrophenyl-β-glucoside) value of 0.76±0.11 mM. The activation energy for the enzyme was 11.8 kcal/mol. Several divalent metal ions inhibited β-glucosidase activity, some of which showed inhibition of enzyme activity only at higher concentrations. Ag2+ was the most potent inhibitor. A metal chelating agent, EDTA, also inhibited β-glucosidase activity. Except for trehalose, glucose, glucono-δ-lactone, cellobiose, gentiobiose, laminaribiose, maltose and isomaltose inhibited β-glucosidase activity. Glucose was found to be a competitive inhibitor, whereas glucono-δ-lactone and other β-linked disaccharides were noncompetitive (mixed) inhibitors of the enzyme.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Engel PC (1981) Enzyme kinetics. The steady-state approach. 2nd edn. Chapman and Hall, London, p 26–36
Evans CS (1985) Properties of the β-d-glucosidase (cellobiase) from the wood-rotting fungus, Coriolus versicolor. Appl Microbiol Biotechnol 22:128–131
Hu ASL, Epstein R, Halvorson HO, Bock RM (1960) Yeast β-glucosidase: Comparison of the physical-chemical properties of purified constitutive and inducible enzyme. Arch Biochem Biophys 91:210–219
Kohchi C, Hayashi M, Nagai S (1985) Purification and properties of β-glucosidase from Candida pelliculosa var. acetaetherius. Agri Biol Chem 49:779–784
Miller GL (1959) Protein determination for large numbers of samples. Anal Chem 31:964
Okada G (1985) Purification and properties of a cellulase from Aspergillus niger. Agri Biol Chem 49:1257–1265
Parr SR (1983) Some kinetic properties of the β-d-glucosidase (cellobiase) in a commercial cellulase product from Penicillium funiculosum and its relevance in the hydrolysis of cellulose. Enzyme Microb Technol 5:457–462
Ryu DDY, Mandels M (1980) Cellulase: biosynthesis and applications. Enzyme Microb Technol 2:91–102
Sternberg D, Mandels GR (1982) β-glucosidase induction and repression in the cellulolytic fungus, Trichoderma reesei. Expt Mycology 6:115–124
Tan TK, Yeoh HH, Tian KE (1985) Cellulolytic fungi isolated from wood shavings. Mycopathologia 90:97–99
Uziie M, Matsuo M, Yasui T (1985) Possible identity of β-xylosidase and β-glucosidase of Chaetomium trilaterale. Agri Biol Chem 4:1167–1173
Wilkinson GN (1961) Statistical estimations in enzyme kinetics. Biochem J 80:324–332
Woodward J, Wiseman A (1982) Fungal and other β-d-glucosidases — their properties and applications. Enzyme Microb Technol 4:73–79
Workman WE, Day DF (1982) Purification and properties of β-glucosidase from Aspergillus terreus. Appl Environ Microbiol 44:1289–1295
Yeoh HH, Tan TK, Tian KE (1984) Cellulolytic enzymes of fungi isolated from wood materials. Mycopathologia 87:51–55
Zhu YS, Wu YQ, Chen W, Tan C, Gao JH, Fei JX, Shih CN (1982) Induction and regulation of cellulose synthesis in Trichoderma pseudokoningii mutants EA3-867 and N2-78. Enzyme Microb Technol 4:3–12
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yeoh, H.H., Tan, T.K. & Koh, S.K. Kinetic properties of β-glucosidase from Aspergillus ornatus . Appl Microbiol Biotechnol 25, 25–28 (1986). https://doi.org/10.1007/BF00252507
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00252507