Summary
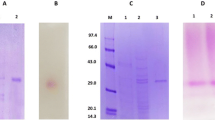
Two extracellular β-glucosidases (EC 3.2.1.21) were isolated from Aspergillus niger USDB 0827 and A. niger USDB 0828, and their physical and kinetic properties studied. Both enzymes were very similar in terms of molecular size (230000 Da), pH optimum (pH 4.6), temperature optimum (65° C), stability at high temperatures and substrate preferences. They were capable of hydrolysing β-linked disaccharides, phenyl β-d-glucoside, p-nitrophenyl β-d-glucoside (PNPG), o-nitrophenyl β-d-glucoside, salicin and methyl β-d-glucoside but lacked activity towards α-linked disaccharides, a range of p-nitrophenyl monoglycosides and p-nitrophenyl diglycosides. Both β-glucosidases were better at hydrolysing cellobiose than cellotriose, cellotetraose or cellopentaose. For both enzymes, glucose showed competitive inhibition with PNPG as substrate but had no effect with cellobiose. However, the two β-glucosidases differed in inhibition by glucono-1,5-lactone and affinity for cellobiose. β-Glucosidase from A. niger USDB 0827 also gave lower specific activity, and was more susceptible to metal ions (Ag+, Fe2+ and Fe3+) inhibition than that of A. niger USDB 0828.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Berghem LER, Petterson LG (1974) The mechanism of enzymatic cellulose degradation. Isolation and some properties of a β-glucosidase from Trichoderma viride. Eur J Biochem 46:295–305
Deshpande V, Eriksson KE, Pettersson B (1978) Production, purification and partial characterization of 1,4-β-glucosidase enzymes from Sporotrichum pulverulentum. Eur J Biochem 90:191–198
Gong CS, Ladisch MR, Tsao GT (1977) Cellobiase from Trichoderma viride: purification, properties, kinetics and mechanism. Biotechnol Bioeng 19:959–981
Hedrick JL, Smith AJ (1968) Size and charge isomer separation and estimation of molecular weights of proteins by disc gel electrophoresis. Arch Biochem Biophys 126:155–164
Lo AC, Barbier JR, Willick GE (1990) Kinetics and specificities of two closely related β-glucosidases secreted by Schizophyllum commune. Eur J Biochem 192:175–181
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Pontecorvo G, Roper JA, Hemmons LM, McDonald KD, Bufton AWJ (1953) The genetics of Aspergillus nidulans. Adv Genet 5:141–238
Raabo E, Terkildsen TC (1960) On the enzymatic determination of blood glucose. Scand J Clin Lab Invest 12:402–407
Saha SC, Sanyal A, Kundu RK, Dube S, Dube DK (1981) Purification and characterization of two forms of extracellular β-glucosidase from jute pathogenic fungus Macrophimina phaseolina. Biochim Biophys Acta 662:22–29
Sakamoto R, Arai M, Murao S (1985) Enzymic properties of three β-glucosidases from Aspergillus aculeatus no. F-50. Agric Biol Chem 49:1283–1290
Sanyal A, Kundu RK, Dube S, Dube DK (1988) Extracellular cellulolytic enzyme system of Aspergillus japonicus. 2. Purification and characterization of an inducible extracellular β-glucosidase. Enzyme Microb Technol 10:91–99
Sharma S, Sandhu DK, Bagga PS (1991) Physical characterization of isozymes of endo-β-1,4-glucanase and β-1,4-glucasidase from Aspergillus species. FEMS Microbiol Lett 79:99–104
Shewale JG (1982) β-Glucosidase: its role in cellulase synthesis and hydrolysis of cellulose. Int J Biochem 14:435–443
Srivastava SK, Gopalkrishnan KS, Ramachandran KB (1984) Kinetic characterization of a crude β-d-glucosidase from Aspergillus wentii Pt 2804. Enzyme Microb Technol 6:508–512
Tan TK, Yeoh HH, Paul K (1986) Cellulolytic activities of Trichoderma hamatum grown on different carbon substrates. Mircen J Appl Microbiol Biotechnol 2:467–472
Tavobilov IM, Rodionova NA, Bezborodov AM (1984) Substrate specificity and some properties of β-glucosidase from Aspergillus niger 15. Biochem SSR 49:847–853
Wilkinson GN (1961) Statistical estimations in enzyme kinetics. Biochem J 80:324–332
Woodward J, Wiseman A (1982) Fungal and other β-d-glucosidases — their properties and applications. Enzyme Microb Technol 4:73–79
Workman WE, Day DF (1982) Purification and properties of β-glucosidase from Aspergillus terreus. Appl Environ Microbiol 44:1289–1295
Yeoh HH, Tan TK, Koh SK (1986) Kinetic properties of β-glucosidase from Aspergillus ornatus. Appl Microbiol Biotechnol 25:25–28
Yeoh HH, Tan TK, Chua SL, Lim G (1988) Properties of β-glucosidase purified from Aspergillus niger. Mircen J Appl Microbiol Biotechnol 4:425–430
Zhu YS, Wu YQ, Chen W, Tan C, Gao JH, Fei JX, Shih CN (1982) Induction and regulation of cellulase synthesis in Trichoderma pseudokoningii mutants EA3–867 and N2–78. Enzyme Microb Technol 4:3–12
Author information
Authors and Affiliations
Additional information
Correspondence to: Y. K. Hoh
Rights and permissions
About this article
Cite this article
Hoh, Y.K., Yeoh, HH. & Tan, T.K. Properties of β-glucosidase purified from Aspergillus niger mutants USDB 0827 and USDB 0828. Appl Microbiol Biotechnol 37, 590–593 (1992). https://doi.org/10.1007/BF00240731
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00240731