Abstract
Low-cost plant and algal biomass are increasingly demanded as a source of proteins, and also, peptides and amino acids are gaining interest for their biological and functional properties. The reduction in chemicals, time and energy, and the integral valorization of the raw materials in the framework of biorefineries are major concerns in the development of environmentally friendly processes. Subcritical water technology is an efficient green technique useful both for extraction and for hydrolysis of protein and other fractions (lipid, carbohydrates, phenolics). However, adequate selection of operational conditions is needed in order both to maximize their extraction yield and to avoid degradation into monomeric units and decomposition products. This review summarizes the major features of subcritical water–based processes for the extraction/hydrolysis of protein. In order to valorizate other valuable fractions from agro-food wastes and algal biomass, optimal conditions should be established as a compromise solution. Alternatively, stagewise operation to sequentially obtain the target fractions could be desirable.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
In a scenery where the need for renewable and sustainable sources of proteins is growing, but the animal protein production has a high environmental impact, the vegetarian population increases, and some alternative sources such as food grade insects are not universally accepted (Pojić et al. 2018), plant and algal proteins are increasingly demanded (Du et al. 2020). Agro-food wastes are generated worldwide and their valorization offers not only a low-cost protein source, but also a solution to an environmental problem. Micro- and macroalgae are advantageous in relation to their rapid growth without requiring land and fertilizers.
The term peptides refer to those compounds with 2–20 amino acid residues, which in addition to their nutritional value present a variety of physiological activities and are named bioactive peptides (Hou et al. 2017; Baig et al. 2018; Bhandari et al. 2020). Bioactive peptides are more bioavailable and less allergenic when compared with total proteins, or high-molecular-weight polypeptides, usually with more than 70 amino acid residues. Their pharmacological use, either purified or in mixtures, for the treatment and prevention of various diseases also depends on their absorption and bioavailability (Udenigwe and Aluko 2012) and on the different regulations found in different territories (Chalamaiah et al. 2019). Amino acids also present applications in nutraceuticals and ingredients for functional foods and feeds, and biostimulants in agriculture (Montesano et al. 2020).
Numerous health benefits have been attributed to protein hydrolyzates (Ashaolu 2020) and bioactive peptides, for the risk reduction, treatment, and management of chronic and degenerative diseases (Baig et al. 2018; Katayama and Nakamura 2019; Lammi et al. 2019; Mada et al. 2019; Patel et al. 2019; Montesano et al. 2020). Their different actions include antihypertensive (Abdelhedi and Nasri 2019; Premkumar et al. 2019; Jahandideh and Wu 2020), antimicrobial (Baig et al. 2018; Bártová et al. 2019; Oshiro et al. 2019), antiinflammatory (Ahn et al. 2017; Fernández-Tomé et al. 2019; Guha and Majumder 2019; Reyes-Díaz et al. 2019), antioxidant (Fernández-Tomé et al. 2019), immunomodulatory (Fernández-Tomé et al. 2019), cytotoxicity against different cancer cells (Baig et al. 2018; Marquez-Rios and Del-Toro-Sánchez 2018; Aaghaz et al. 2019; Guidotti et al. 2019), antiplatelet aggregation (Rengasamy et al. 2019), antidiabetes (Baig et al. 2018; Kehinde and Sharma 2018; Hu et al. 2019; Moreno-Valdespino et al. 2020), antiobesity (Lee et al. 2017), and bone health properties (Ahn and Je 2019). Their inclusion as protein hydrolyzates in diets can ensure desirable rates of growth performance and feed efficiency in terrestrial animals and fish (Hou et al. 2017). Some of these peptides are multifunctional (Li et al. 2019a), affecting more than one physiological function, with low negative side effects and reduced costs (Lammi et al. 2019). Bioactive peptides can be used in cosmeceuticals (Lima and Moraes 2018), based on their properties in relation to slow aging (Ahsan 2019; Apone et al. 2019; Bhullar and Wu 2020), attenuation or prevention of different skin dysfunctions (Lima and Moraes 2018) and for conferring moisturizing properties (Ahsan 2019; Aguilar-Toalá et al. 2019). These peptides also possess functional properties determined by the protein structure and interactions with other molecules and contributing to the physical properties of the food or cosmetic product. The most important are water-holding capacity and solubility, foaming capacity, emulsifying, and gel forming properties.
A number of recent reviews on the potential of bioactive peptides can be found, including purification, identification, bioavailability, and safety aspects (Hajfathalian et al. 2018; Chakrabarti et al. 2018; Wang et al. 2018; Bhandari et al. 2020; Ghanbari 2019), the techniques for protein extraction, the production methods of hydrolyzates and peptides (Hajfathalian et al. 2018; Zamora-Sillero et al. 2018; Li et al. 2019a; Bhandari et al. 2020), in biorefineries (Pojić et al. 2018; Du et al. 2020). Among emerging extraction techniques, subcritical water (SW) is an environmentally friendly technology (King 2006) valid for the extraction of bioactives (Cervantes-Cisneros et al. 2017; Youssef et al. 2019; Zhang et al. 2020). It offers advantages to extract protein from food industry wastes, including plant and animal sources, and for the preparation of hydrolyzates and peptides with functional and antioxidant properties (Marcet et al. 2016), being an alternative to conventional methods using chemicals or enzymes (Zhu et al. 2015; Powell et al. 2016). Ziero et al. (2020) discussed the economics, extraction routes, conditions, and reactor configurations for protein extraction and hydrolysis during subcritical and supercritical water processing of proteic materials with the aim of obtaining amino acids.
The present review presents the potential of subcritical water as a tool for the extraction and hydrolysis of protein in the context of biorefineries with the aim of achieving an integral valorization of biomass. If the final application of hydrolyzates and peptides is related to food, nutraceutical, and cosmeceutical applications, the consumer preferences prioritize the utilization of plant and algal sources. Since subcritical water processing can simultaneously fractionate and hydrolyze the different components of the raw material, the need of a careful selection of operational conditions to attain an optimal yield of the target products is discussed.
Sources
Bioactive peptides and hydrolyzates have been initially produced from food (Chakrabarti et al. 2018; Udenigwe and Aluko 2012; Premkumar et al. 2019; Priya 2019; Liu et al. 2020; Montesano et al. 2020), including legumes (Reyes-Díaz et al. 2019; Moreno-Valdespino et al. 2020), potato (Bártová et al. 2019), defatted soy meal (Ashaolu 2020), and byproducts from terrestrial animals, and fish and their byproducts (Zamora-Sillero et al. 2018; Wang et al. 2019a). Despite hydrolyzates and peptides from food sources are generally considered more “natural,” impart less adverse effects, and are more accepted (Chakrabarti et al. 2018; Kehinde and Sharma 2018), some of them are new without a history of safe use and may present allergic or toxic concerns (Liu et al. 2020).
Peptides can also be obtained by chemical synthesis, requiring amino acid mixtures as starting material and following the rational design, directed evolution, or computational methods to improve the properties (Adhikari et al. 2020). However, both synthetic and animal-derived peptides are progressively less accepted, due to the presence of toxic solvent residues after the chemical synthesis or the risk of infections from pathogenic virus as well as the presence of dysfunctional protein aggregates in the second case (Apone et al. 2019). Therefore, the increasing consumer demand for less allergenic and vegan ingredients (Apone et al. 2019) favors the utilization of alternative sources for the production of bioactive peptides (Montesano et al. 2020). Furthermore, the valorization of wastes from agro-industrial activities (Marciniak et al. 2018) in cost-effective, sustainable, and environmentally friendly processes tends to the circular economy and zero waste (Chakrabarti et al. 2018; Pojić et al. 2018; Montesano et al. 2020).
Agro-food wastes contain not only 10–40% protein, but also other components (Table1). Defatted meal remaining after oil extraction is an excellent protein source, soymeal was initially the byproduct of oil extraction, but both the quality and content in the defatted meal makes soybean protein a valuable product (Khuwijitjaru et al. 2011). It is mentioned here as a model, more than a waste and underused source. Micro- and macroalgae are attractive sources of protein hydrolyzates and peptides (Li et al. 2019a), since they are safe, well tolerated (Apone et al. 2019; Abdelhedi and Nasri 2019; Hu et al. 2019), and increasingly demanded (Soto-Sierra et al. 2018).
Microalgae, with a high content of nutritious and low allergenicity protein, contain other valuable compounds, including carbohydrates, lipids with high omega-3 content, and carotenes. They present a rapid growth from a variety of low-cost nutrients, do not compete with food production, and can be a raw material for food, fuels, and chemicals (Garcia-Moscoso et al. 2013; Soto-Sierra et al. 2018).
Red seaweeds possess a higher protein content than other groups, up to 50% dry weight (d.w.), but macroalgae contain an important fraction of polysaccharides or hydrocolloids and also possess high levels of minerals and vitamins, which makes them an interesting source for biorefining. Compared with terrestrial biomass, seaweeds offer advantages since they do not need land or freshwater and have a rapid growth and biomass yield. Another low-cost source susceptible of valorization is macroalgal blooms (Torres et al. 2019a, b). Marine plants, such as mangroves, are used traditionally as herbal medicine (Pangestuti et al. 2020).
Protein Extraction and Hydrolysis Techniques
Conventional extraction of protein from biomass relies on the use of chemicals (acid, alkali) (Garcia-Moscoso et al. 2013; Du et al. 2020). The further production of protein hydrolyzates, composed of large peptides, small peptides, and free amino acids, can be addressed using chemical synthesis or by partial digestion of proteins, by chemical, enzymatic, or microbial hydrolysis (Mirmoghtadaie et al. 2016).
Acid hydrolysis offers the advantage of low cost, but results in the complete destruction of tryptophan, a partial loss of methionine, and the conversion of glutamine into glutamate and asparagine into aspartate (Hou et al. 2017). Alkaline hydrolysis with calcium, sodium, or potassium hydroxide has the advantage of a low cost and a complete recovery of tryptophan. However, due to the complete destruction of most amino acids, it is not widely used in the food industry (Hou et al. 2017). Some amino acids can undergo chemical alterations during processing such as decomposition, racemization, dehydration, and glycation. Heat- and alkali-treated proteins often induce formation of cross-linked amino acids, and undesirable products can also be generated in the presence of other components of the solid matrix, such as carbohydrates, lipids, or secondary metabolites, susceptible of interacting with proteins (Liu et al. 2020).
The enzymatic hydrolysis is valid for different protein sources (Montesano et al. 2020) and can be performed under mild conditions with precise control of the degree of hydrolysis, minimizing side reactions and the presence of toxic chemicals in the products. The most frequently used enzymes are from plant, from animal, and from microbial origin, which have low production cost and good stability and specificity (dos Santos Aguilar and Sato 2018; Mazorra-Manzano et al. 2018). The relatively high cost and the potential presence of enzyme inhibitors in the raw protein materials are major disadvantages (Hou et al. 2017). Microorganisms produce proteases that can hydrolyze proteins into peptides and free amino acids, and other enzymes that can degrade complex carbohydrates and lipids. One major advantage of fermentation is the possibility of removing hyper-allergic or antinutritional factors, but microbial hydrolysis presents relatively high costs (Hou et al. 2017).
Another challenge to be considered for the production of protein hydrolyzates and peptides is the development of efficient and scalable methods, demanded to avoid chemicals or the need of costly enzymes and prolonged times for modifying functionality (Mirmoghtadaie et al. 2016; Hou et al. 2017; Chakrabarti et al. 2018; Soto-Sierra et al. 2018; Montesano et al. 2020). Among them are microwave, ultrasound, high-voltage pulsed electric field, high hydrostatic pressure–assisted enzyme hydrolysis (Ozuna et al. 2015; Marciniak et al. 2018; Liu et al. 2020), and water under subcritical conditions (Ma et al. 2018; Zhang et al. 2019a, b; Wang et al. 2019b).
Subcritical Water Processing
Potential for Extraction and Hydrolysis
Subcritical water (SW) is found at 100–374 °C, during operation under pressures up to 22 MPa in order to maintain its liquid state. Under these conditions, water presents unique properties, the viscosity and density decrease, but the compressibility is low. The dielectric constant decreases and the hydrogen bonding structure is weakened, facilitating the solubilization of apolar components. Furthermore, the greater ionic product, compared with that of water at atmospheric conditions, favors the catalytic action of SW and enhances depolymerization of polysaccharides to oligomers and to monomeric units, as well as the generation of smaller soluble protein fractions and amino acids, which could be further degraded (Quitain et al. 2006; Hata et al. 2008; Sereewatthanawut et al. 2008; Zainan et al. 2019). The possibility of tuning the properties of water at elevated temperatures and pressures makes it a promising reaction medium in short residence times and with high rates of conversion (Rogalinski et al. 2008; Brunner 2009). Alternative names used for subcritical water processing include hot compressed water, autohydrolysis, steam explosion, pressurized low polarity water, or liquid hot water (Garrote et al. 1999; King 2006; Ho et al. 2007; Kazan et al. 2015).
Subcritical water extraction is a technically efficient, inexpensive, scalable, and environmentally friendly technology. Compared with operation at atmospheric conditions, SW extraction is faster and more efficient for extraction and depolymerization. Furthermore, the possibility of avoiding the drying stages is particularly useful for algal biomass (Garcia-Moscoso et al. 2013; Saravana et al. 2016a, b), but has also been reported for plants and mushrooms (Garrote et al. 1999; Marcet et al. 2016; Powell et al. 2016; Gallego et al. 2019).
Subcritical water hydrolysis can be an alternative, efficient, cheap, and fast alternative to enzymatic digestion for the production of peptides and amino acids (Aida et al. 2017). The secondary reactions involving decarboxylation, deamination, transamination, and oxidation lead to different amino acid profiles (Pińkowska and Oliveros 2014; Watchararuji et al. 2008). Based on studies with model animal proteins, Powell et al. (2016) observed that SW allows more than 80% peptide production, comparable to trypsin hydrolysis. However, other hydrolysis products can be found due to modifications of amino acid side chains, an effect highly influenced by temperature and heating modes (Fan et al. 2020). Usually irreversible first-order kinetics is used to describe the protein hydrolysis to amino acids (Marcet et al. 2016), but also, a simplified mechanism of protein aggregation with zero-order kinetics and next disaggregation and decomposition following second-order and zero-order kinetics, respectively, were proposed for the production of polypeptides and amino acids (Sunphorka et al. 2012a) or a second-order reaction kinetics for the hydrolysis to peptides and zeroth order for the production of amino acids (Garcia-Moscoso et al. 2013).
The reader can find excellent compilations of the conditions defined in different studies with subcritical water for the extraction of protein and the further hydrolysis to peptides and amino acids from different plant and animal sources (Marcet et al. 2016; Lamp et al. 2020). When different components are desired, the optimal yields for each of them have been reported at different temperatures, as summarized in Table 2 for some selected examples.
Simultaneous or Cascade Extraction in the Context of Biorefineries
The plant and algal materials used as protein sources have a complex composition. Usually other fractions with commercial value can be obtained from different protein sources, i.e., cellulose, hemicelluloses, lignin, and free phenolics from plant residues (Viriya-Empikul et al. 2012; Getachew and Chun 2017) and food wastes (Wiboonsirikul et al. 2013); oil, carbohydrates, and phenolics from oil seeds (Ho et al. 2007; Ndlela et al. 2012; Ravber et al. 2015a, b; Pińkowska et al. 2019); and lipids, pigments, and polysaccharides from algae (Fu et al. 2018). Optimal operational conditions should consider their simultaneous or their sequential extraction. The simultaneous extraction of other components provides the hydrolyzates interesting biological and functional properties, particularly derived from the enrichment in phenolics and sugars (Watchararuji et al. 2008; Park et al. 2019), and the stronger hydrophobic interactions due to the accumulation of flavonoids at the interface enhance the physical and oxidative stability of emulsions (Chen et al. 2016).
Different conditions are required due to different susceptibility of the components. In a study on the hydrolysis kinetics of model biopolymers, starch, cellulose, and bovine serum albumin, in a continuous-flow reactor, Rogalinski et al. (2008) confirmed that the peptide bonds in proteins exhibited higher stability compared with the β-1,4- and β-1,6-glycosidic linkages in cellulose and starch, respectively. Operating in batch mode during a fixed time, a slightly lower temperature was preferred for the extraction of carbohydrates than for protein. Wiboonsirikul et al. (2007a) found for black rice bran maximum carbohydrate yield at 200 °C, and protein at 220 °C, but the phenolic compound maximum occurred at a higher value; Chiou et al. (2010) selected 180 °C for carbohydrate and 210 °C for protein from rice bran, but for the extraction of antioxidant compounds, 20% ethanol at 237 °C was more efficient than SW (Chiou et al. 2012). Kataoka et al. (2008) found the highest yield and saccharide content at 200 °C for wheat bran, whereas the protein, total phenolic, hydroxymethylfurfural, and furfural contents were the highest at 240 °C, as well as the radical scavenging activity and the protective action against linoleic acid oxidation at 250 °C. Khuwijitjaru et al. (2011) reported maximum carbohydrate and protein contents from defatted soybean meal at 175 °C and 225 °C, respectively. Wiboonsirikul et al. (2013) obtained the highest carbohydrate content from okara at 170 °C, the highest protein content at 240–260 °C, and the radical scavenging activity at 240 °C. Nomura et al. (2019) reported the highest content of carbohydrates at 180 °C, protein at 200 °C, and the antiinflamamtory apocynin at 180–260 °C from Mentha arvensis leaves. Ho and Chun (2019) found the maximum amount of sugars and flavonoids from Pseuderanthemum palatiferum at 130 °C, but for saponin, phenolics, and protein at 170 °C, 190 °C, and 230 °C, respectively. SW extraction of black rice bran provided maximum yields, carbohydrate and protein contents at 200 °C and antioxidant properties at 260 °C (Wiboonsirikul et al. 2007b).
Phusunti et al. (2017) reported the highest carbohydrate removal from microalgae at 180 °C and protein removal at 200 °C. Gereniu et al. (2017) proposed the operation at 150 °C to maximize sugars from Kappaphycus alvarezii, whereas proteins and phenolics and antioxidant properties were best extracted at 270 °C. Pangestuti et al. (2019) reported the highest sugar content from Hypnea musciformis at 120–180 °C, whereas the highest protein and phenolic yields were obtained at 210 °C. Park et al. (2019) found optimal overall hydrolysis efficiency and protein yield of Pyropia yezoensis at 180 °C, whereas the maximal phenolics and antiradical properties were found at 210 °C, and the maximum total sugar content at 120 °C and the reducing sugar content at 180 °C; also, different optimal temperatures for essential and non-essential amino acids were reported. Saravana et al. (2016a) found the maximum protein content at 170 °C for Saccharina japonica (S. japonica). However, the maximal crude fucoidan and phlorotannin contents were found at 140 °C. Saravana et al. (2016b) also found the maximum for the recovery of sugars and polyols from S. japonica at 180 °C, and also for total amino acids, the non-essential showing higher yield than essential amino acids. However, the protein and mineral (Mg, Ca, K, Na) contents occurred at temperature higher than 300 °C. Flórez-Fernández et al. (2019) applied non-isothermal heating up to 220 °C to Laminaria ochroleuca, to obtain the highest extraction yield, phenolic and protein contents, and antioxidant capacity, but the maximal alginate extraction and viscoelastic properties were achieved at 160 °C and the fucose and sulfate content at 180 °C.
Simultaneous extraction of different biomass components is possible when the required conditions are coincident or if compromise conditions are established. These have been reported at short times for the recovery of protein and carbohydrates from defatted rice bran at 200 °C (Wiboonsirikul et al. 2007b; Sereewatthanawut et al. 2008) and 160 °C (Viriya-Empikul et al. 2012), for protein and for radical scavenging properties from okara treated at 240 °C (Wiboonsirikul et al. 2013), or at 120 °C for protein and phenolics from mangroves (Pangestuti et al. 2020). Awaluddin et al. (2016) proposed more moderated conditions for the joint extraction of carbohydrates and protein from Chlorella vulgaris compared with those leading to maximal protein yields.
However, when different conditions are needed for different components and the valorization of all of them is desired, cascade operation seems preferable to increase the yields and to substantially reduce the formation of undesired degradation products (Reisinger et al. 2018). The integral valorization of agro-industrial, food, forest, and marine sources could be addressed with an initial subcritical water extraction stage. The extraction could be defined to obtain initially carbohydrates, then protein and oil in severity growing sequence (Ravber et al. 2015a, b). When a higher quality oil is desired, a different sequence is proposed, i.e., extraction with supercritical carbon dioxide and further extraction of proteins with pressurized hot water from the olive pomace oil, followed by more severe conditions to obtain fermentable sugars (Kazan et al. 2015). SW serves as a pretreatment to obtain a microalgal solid with less N-containing compounds and higher long-chain fatty acid content, facilitating the further pyrolysis regarding the energy consumption and the quality of the biooil (Phusunti et al. 2017; Thiruvenkadam et al. 2015). Operation at higher temperatures (350 °C) could be of interest for the energetic valorization from algal components (Cui et al. 2020).
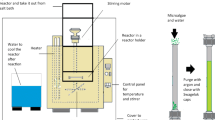
Figure 1 illustrates a very simple flow diagram of general biorefinery processes, showing in gray areas the stages that could be replaced by subcritical water processing for both the extraction and the production of protein hydrolyzates and peptides from agro-food wastes and micro- and macroalgal biomass
Simplified flow diagrams of the processes for the production of protein hydrolyzates and peptides from agro-food wastes, microalgae, and macroalgae in the framework of a biorefinery to obtain other valuable fractions. Note that not all stages are always used during conventional processing, i.e., solvent extraction or hydrolysis
Modes of Subcritical Water Operation
The efficiency of the SW process is determined by different operational variables that should be independently optimized for each raw material and final product. Most studies have been performed in batch-type stainless steel vessel, but also in semicontinuous and in continuous operation in a range from lab to bench scale (Khuwijitjaru et al. 2011; Sunphorka et al. 2012b; Wiboonsirikul et al. 2013; Hwang et al. 2015; Saravana et al. 2016b; Zainan et al. 2019). Before operation, water can be sonicated or bubbled with nitrogen gas to increase the reactor pressure and to purge and displace oxygen avoiding oxidation reactions (Plaza et al. 2010; Khuwijitjaru et al. 2011; Viriya-Empikul et al. 2012; Saravana et al. 2016a; Phusunti et al. 2017; Park et al. 2019). During semicontinuous operation, with the packed raw material, a sequence of fractions with different compositions can be eluted; increased efficiency was reported using co-packing materials (such as glass beads) (Ho et al. 2007). A preliminary static stage before dynamic operation has been described; in this case, the influence of the flow rates could be attenuated (Viriya-Empikul et al. 2012). Continuous operation provides higher productivity than batch one, but during continuous operation (Saravana et al. 2016b; Du et al. 2020), but both the radial temperature distribution of slurries (Fu et al. 2018) and the rheological properties of the microalgal suspension, affected by the starch gelatinization and protein denaturation (Zhang et al. 2018), can influence the yield of carbohydrates and proteins.
One strategy suitable for materials containing components with different depolymerization kinetics in SW, and for working in continuous-flow scalable systems, is flash hydrolysis. Several operations (centrifugation, concentration, and drying) could be avoided, and the residence time can be precisely controlled to several seconds avoiding prolonged preheating and cooling times (Garcia-Moscoso et al. 2013). It has been reported for protein extraction from microalgae (Asiedu et al. 2018), and to selectively remove inorganic elements, hydrolyze proteins, and preserve lipids of microalgae, proposed for biofuel production (Garcia-Moscoso et al. 2013, 2015).
A modification of SW extraction consists on the steam explosion, based on exposing the biomass to pressurized steam, followed by a sudden decompression that breaks the internal structure during the rapid expansion of the biomass. The thermal and physical changes during steam explosion not only enhance the extraction yield of the protein, some covalent bonds in the polypeptidic chains can be broken, and the further recombination forming new bonds can modify the final structure and surface morphology of proteins. Zhang et al. (2019a, b) applied it before the oil extraction from camellia seed and found a decrease in the simple sugars, free amino acids, and lightness of the products, but the foaming and emulsifying properties increased compared with the untreated ones. Li et al. (2019b) observed lowered molecular weight of okara polysaccharides and proteins, with increased water solubility, probably by loosening and degrading the fiber structure, exposing more hydrophilic groups. Steam explosion pretreatment enhanced the fermentation of sorghum by increasing the sugar content and changing the physicochemical properties (Zhao et al. 2020). Despite acid soaking could lower the purity of the product, it can increase the yields (Zhang et al. 2013) and facilitate the hydrolysis of carbohydrates and proteins as well as the further fractionation stages and the better solvent access to lipids (Lorente et al. 2018).
Effect of Operational Conditions During Subcritical Water Operation
Effect of Pretreatment
Drying and grinding are the usual pretreatment stages, applied with the aim of stabilizing the raw material during storage and providing higher solvent accessibility to solutes, respectively. The type of mechanical pretreatment (flaking, extrusion) (Ndlela et al. 2012) and defatting (Watchararuji et al. 2008) also influence the extraction yields. Previous deoiling allowed lower temperatures for soybean, but not for rice bran (Sereewatthanawut et al. 2008; Watchararuji et al. 2008).
Effect of Liquid-to-Solid Ratio
The optimal liquid-to-solid ratio has been usually selected as a function of the raw material and particle size (Awaluddin et al. 2016) and according to the desired product (Awaluddin et al. 2016). Values in the range 3–12 are frequent for the oil and protein extraction from soybean (Ndlela et al. 2012) and 5–40 for soybean meal (Khuwijitjaru et al. 2011; Ndlela et al. 2012; Pińkowska and Oliveros 2014; Hwang et al. 2015), 5–50 for brans (Watchararuji et al. 2008; Wiboonsirikul et al. 2013), and up to 210 for flaxseed (Ho et al. 2007). Lower ratios, such as 2.5 tried for deoiled rice bran, could be limiting since poor mixing makes external mass transfer difficult (Watchararuji et al. 2008). Comparable values, 10–50, have been reported for seaweeds (Santoyo et al. 2011; Park et al. 2019; Saravana et al. 2016b; Pangestuti et al. 2019), whereas a wider range has been applied to microalgae, from 6 to 110, including batch and continuously processed slurries (Garcia-Moscoso et al. 2013; Kumar et al. 2014; Garcia-Moscoso et al. 2013; Phusunti et al. 2017; Zainan et al. 2019; Fan et al. 2020).
Effect of Operation Severity
Pressure is a key variable to maintain the solvent in liquid state, but hardly influences its properties. The hardness of the process is frequently measured with the temperature and with time (Awaluddin et al. 2016; Pangestuti et al. 2019), or with a combination of both in a single parameter, which allows comparison of different studies. A useful one is the severity factor log R0, defined by Overend and Chornet (1987) for the solubilization of hemicelluloses as:
where R0 is the reaction ordinate (min), T is the temperature of the process (°C), and t is the reaction time under isothermal operation (min). It has also been applied to other fractions from lignocellulosics (Garrote et al. 2017) and to protein solubilization (Garcia-Moscoso et al. 2015; Lamp et al. 2020). A combined factor, considering the influence of pH, has been considered (Lamp et al. 2020). When operation is performed under non-isothermal conditions, Eq. (2) can be used:
where T(t) is the temperature versus time profile.
During isothermal extraction of protein, a previous heating (5–30 min) and a subsequent cooling (5–30 min) stages are needed (Watchararuji et al. 2008; Khuwijitjaru et al. 2011; Plaza et al. 2010; Wiboonsirikul et al. 2013). More prolonged heating time has been also described (50–105 min) (Saravana et al. 2016b; Park et al. 2019). The severity during these periods can be defined according to Eq. (2).
Temperature and time should be optimized simultaneously (Wiboonsirikul et al. 2013). In continuous operation, the flow rate determines the residence time. Initially higher protein yield could also be obtained by increasing the hydrolysis time and/or temperature, but protein denatures at high temperatures, due to destruction of the protein tertiary structure through the cleavage of its hydrogen bonds (Awaluddin et al. 2016) and also is decomposed to amino acids and to organic acids (Sereewatthanawut et al. 2008; Watchararuji et al. 2008), Similarly, other components are degraded (polysaccharides, phenolics). When the proteins are strongly bound to the cell structure and require a higher severity to be released, amino acid degradation could already occur (Plaza et al. 2010; Lamp et al. 2020). Usually, this has been observed at temperatures in the range 190–270 °C in 15–50 min (Sato et al. 2004; Hata et al. 2008; Sereewatthanawut et al. 2008; Pińkowska et al. 2013; Hwang et al. 2015; Ramachandraiah et al. 2017; Zainan et al. 2019; Lamp et al. 2020).
Based on the compilation of data in Fig. 2, summarizing the influence of the heating temperature on the extraction yield and protein content in the extracts, the optimal values can be attained operating close to 200 °C. In the general reaction of Fig. 3, they are represented by the oligomeric fraction, which can be further degraded to amino acids and to decomposition products. The scheme is valid also for the saccharide and phenolic fraction, which can also be present in the raw material. However, the temperature and/or time conditions leading to maximal yield/content differ for each fraction.
Addition of Chemicals
Integration of alkaline and subcritical water treatment succeeds for the optimization of protein extraction from biomass. Alkaline treatment led to high protein extraction yield, but some unextracted protein remained in the residue, and a further SW treatment might further destroy the lignocellulosic structure of the residue and promotes protein extraction efficiency (Pangestuti et al. 2019; Du et al. 2020). The protein content from S. japonica was higher during operation at 140 °C with the addition of 0.1% formic acid or 0.1% NaOH (Saravana et al. 2016a, b). The maximum carbohydrate recovery from defatted flaxseed meal was obtained at 150 °C and pH 4, whereas the maximum yield of proteins and lignin was attained at 160 °C and lignans 170 °C, respectively, in both cases at pH 9 (Ho et al. 2007). Supawong et al. (2019) prepared hydrolyzates from hexane defatted rice bran treated with subcritical alkaline water extraction and further enzymatic hydrolysis, and the product reduced fat uptake in fried fish cakes and protected surimi seafood against lipid oxidation. Addition of acid could make the conditions more severe and cause undesirable degradation, but it is chosen when the objective is to produce monomeric sugars, which can further be used as a carbon source for ethanol production. In order to avoid excessive degradation, Reisinger et al. (2018) proposed a cascade process with stepwise separation of the liquid phase, to increase the yields and to reduce the formation of excessive amounts of sugar monomers and undesired degradation products from destarched wheat bran.
Combination with Other Extraction Techniques
The selectivity of subcritical water extraction can be modified by altering the operational conditions, and the process performance can be enhanced by combination with other techniques (King 2006; Zhang et al. 2020), such as pretreatments with microwave and ultrasonics or with modifiers such as nitrogen and carbon dioxide on subcritical water (Getachew and Chun 2017). Coupling of ultrasound and SW extraction to Spirulina platensis provided higher protein extraction yield in 1 h than the traditional ultrasound and freeze-thaw method in 16 h. The protein can be released by the high frequency vibration of ultrasound and the degradation of cell wall induced by the high pressure gradients and shear forces induced by cavitation. Combination of ultrasound with SW was proposed to obtain antidiabetes peptides (Hu et al. 2019) and this strategy can also accelerate the degradation of proteins, resulting in a molecular weight distribution in the range 0.18–5 kDa, whereas from the combination of ultrasound with freeze-thaw were > 5 kDa (Fan et al. 2020). Microwave-assisted SW extraction lowered processing times compared with the conventional heating method. Operating in non-isothermal heating up to 150 °C provided maximum for protein content from Mastocarpus stellatus, but the highest phenolic content, antiradical capacity, and sulfate content were observed at 190 °C, and strongest hydrogels of hybrid carrageenans were found at 170 °C for 6 min (Ponthier et al. 2020).
The SW processing can be applied before or simultaneously with the enzymatic hydrolysis or can replace the hydrolysis with enzymes. A previous SW processing stage can enhance the enzymatic susceptibility (Hwang et al. 2017; Zhang et al. 2019a, b; Wang et al. 2017). Alternatively, prehydrolysis with an Aspergillus oryzae protease before SW extraction of protein from soy meal enhanced the extraction yield, but lowered selectivity. The increased surface hydrophobicity, caused by protein unfolding, and the formation of small aggregates enhanced the emulsifying capacity and emulsion stability. The presence of secondary activities, such as β-glucosidase, can enhance the antioxidant capacity, due to the formation of Maillard reaction products and the release of flavonoid aglycones (Lu et al. 2015). However, SW treatment causes heat-induced conformational changes that could decrease the enzymatic susceptibility. After SW treatment at 220 °C for 20 min, protein solubility increased but the degradation, deamidation, and small molecular size caused that many hydrolysis sites disappear and were difficult to access by protease (Hwang et al. 2017).
Effect of the Operation Conditions on the Properties of the Products
The antioxidant activity of protein and peptides is closely related to their structure, those with lower molecular weight and high degree of hydrophobicity showing more potent antioxidant activity (Pangestuti et al. 2019). The optimal conditions for phenolics and proteins are sometimes coincident, thus leading to high antioxidant properties, since both contents could positively correlates with the activity of the extracts (Wiboonsirikul et al. 2013). The increased antiradical properties observed at higher temperatures than the maximal for protein and phenolics would also suggest the relevance of the products from the carbohydrate degradation and proteins, both the Maillard browning and the caramelization reactions (Plaza et al. 2010; Wiboonsirikul et al. 2013; Saravana et al. 2016b; Gereniu et al. 2017). These reactions also induce color changes from colorless to slight yellow and to dark brown (Wiboonsirikul et al. 2007a, b; Watchararuji et al. 2008; Khuwijitjaru et al. 2011; Ramachandraiah et al. 2017; Pangestuti et al. 2019; Park et al. 2019).
Also, the color of the extracts changed depending on the conditions, being a result of the extraction of different components, i.e., pigments from algae, or the formation of new colored compounds. The addition of soy protein hydrolyzates obtained by SW processing at 180 °C can confer darker color to pork patties, particularly when used at higher doses; the water holding capacity and hardness was not affected and the addition at 0.5–1.5% suppressed oxidative deterioration of fat-containing meat products during chilled storage (Lee et al. 2015). Also, a toasty aroma at the lower temperatures became more pungent with an increase in temperature and modifications in the particle size and turbidity occur due to the aggregation and degradation of protein (Zhang et al. 2015; Saravana et al. 2016b; Gereniu et al. 2017).
The use of excessively high temperatures could cause a reduction in the high-molecular-mass polysaccharides and peptides and also some functional properties, i.e., emulsion stability (Wiboonsirikul et al. 2007a, b).
Subcritical water treatment increased the functionality, solubility, emulsifying, and foaming properties of protein and improved the thermal stability (Zhang et al. 2015, 2018; Park et al. 2019; Du et al. 2020). Foaming and emulsifying properties are highly influenced by interactions with polysaccharides, and pH. The emulsification properties were less affected by the extraction temperature; the foaming capacity and stability were more affected for soy protein from soybean meal (Khuwijitjaru et al. 2011). Gereniu et al. (2017) found optimal foaming properties when Kappaphycus alvarezii was extracted at 150 °C and optimal emulsifying properties at 270 °C, for red and for brown seaweeds. However, the emulsifying properties were usually optimal at conditions less severe than those providing maximal of protein, and closer to the optimal for carbohydrates (Hata et al. 2008; Kataoka et al. 2008; Wiboonsirikul et al. 2007b). SW treatment of soy protein isolate at 120 °C induced the formation of larger aggregates with higher surface activity at the air-water and oil-water interface than native protein due to lower aggregation degree and more flexible conformation, which improved storage and freeze-thaw stability against emulsion coalescence due to the formation of a thicker multilayer (Wang et al. 2018).
Conclusions
In summary, compared with conventional technologies, subcritical water provides comparable or higher yields of protein and amino acids than conventional alkali or enzymatic hydrolysis in shorter times. Furthermore, the content of bioactives was higher in extracts from SW compared with conventional solvents and exhibited higher antioxidant and antimicrobial activity and enhanced functional properties. However, the safety of compounds produced during protein or peptide processing should be tested to avoid the possible formation of allergenic and toxic peptides and hazardous compounds during protein extraction and hydrolysis. Furthermore, if these peptides are destined for functional food ingredients and/or nutraceuticals, their safety in humans should be evaluated prior to commercialization. In the current scenario demanding lower cost and non-animal protein sources, a variety of possibilities should be considered for their integral utilization. Subcritical water processing is a “green” solvent for both the extraction of bioactive compounds and is attracting interest regarding waste and biomass conversion. This is an efficient and clean scalable method for the extraction and hydrolysis of proteins that should be considered for the integral valorization of renewable resources. However, for this latter purpose and considering the ability of this technology to extract and depolymerize other fractions with interesting properties, careful selection of operational conditions in either simultaneously or in a sequential process is recommended.
References
Aaghaz, S., Gohel, V., & Kamal, A. (2019). Peptides as potential anticancer agents. Current Topics in Medicinal Chemistry, 19(17), 1491–1511. https://doi.org/10.2174/1568026619666190125161517.
Abdelhedi, O., & Nasri, M. (2019). Basic and recent advances in marine antihypertensive peptides: production, structure-activity relationship and bioavailability. Trends in Food Science and Technology, 88, 543–557. https://doi.org/10.1016/j.tifs.2019.04.002.
Adhikari, S., Leissa, J. A., & Karlsson, A. J. (2020). Beyond function: engineering improved peptides for therapeutic applications. AICHE Journal, 66(3), e16776. https://doi.org/10.1002/aic.16776.
Aguilar-Toalá, J. E., Hernández-Mendoza, A., González-Córdova, A. F., Vallejo-Cordoba, B., & Liceaga, A. M. (2019). Potential role of natural bioactive peptides for development of cosmeceutical skin products. Peptides, 122, 170170. https://doi.org/10.1016/j.peptides.2019.170170.
Ahn, C.-B., & Je, J.-Y. (2019). Bone health-promoting bioactive peptides. Journal of Food Biochemistry, 43(1), e12529. https://doi.org/10.1111/jfbc.12529.
Ahn, M. Y., Hwang, J. S., Ham, S. A., Hur, J., Jo, Y., Lee, S., Choi, M.-J., Han, S. G., & Seo, H. G. (2017). Subcritical water-hydrolyzed fish collagen ameliorates survival of endotoxemic mice by inhibiting HMGB1 release in a HO-1-dependent manner. Biomedicine and Pharmacotherapy, 93, 923–930. https://doi.org/10.1016/j.biopha.2017.07.041.
Ahsan, H. (2019). Immunopharmacology and immunopathology of peptides and proteins in personal products. Journal of Immunoassay and Immunochemistry, 40(4), 439–447. https://doi.org/10.1080/15321819.2019.1629590.
Aida, T. M., Oshima, M., & Smith, R. L. (2017). Controlled conversion of proteins into high-molecular-weight peptides without additives with high-temperature water and fast heating rates. ACS Sustainable Chemistry & Engineering, 5(9), 7709–7715. https://doi.org/10.1021/acssuschemeng.7b01146.
Apone, F., Barbulova, A., & Colucci, M. G. (2019). Plant and microalgae derived peptides are advantageously employed as bioactive compounds in cosmetics. Frontiers in Plant Science, 10, 756. https://doi.org/10.3389/fpls.2019.00756.
Ashaolu, T. J. (2020). Health applications of soy protein hydrolysates. International Journal of Peptide Research and Therapeutics. https://doi.org/10.1007/s10989-020-10018-6.
Asiedu, A., Ben, S., Resurreccion, E., & Kumar, S. (2018). Techno-economic analysis of protein concentrate produced by flash hydrolysis of microalgae. Environmental Progress & Sustainable Energy, 37(2), 881–890. https://doi.org/10.1002/ep.12722.
Awaluddin, S. A., Thiruvenkadam, S., Izhar, S., Hiroyuki, Y., Danquah, M. K., & Harun, R. (2016). Subcritical water technology for enhanced extraction of biochemical compounds from Chlorella vulgaris. BioMed Research International, 2016, 5816974. https://doi.org/10.1155/2016/5816974.
Baig, M. H., Ahmad, K., Saeed, M., Alharbi, A. M., Barreto, G. E., Ashraf, G. M., & Choi, I. (2018). Peptide based therapeutics and their use for the treatment of neurodegenerative and other diseases. Biomedicine and Pharmacotherapy, 103, 574–581. https://doi.org/10.1016/j.biopha.2018.04.025.
Bártová, V., Bárta, J., & Jarošová, M. (2019). Antifungal and antimicrobial proteins and peptides of potato (Solanum tuberosum L.) tubers and their applications. Applied Microbiology and Biotechnology, 103(14), 5533–5547. https://doi.org/10.1007/s00253-019-09887-9.
Bhandari, D., Rafiq, S., Gat, Y., Gat, P., Waghmare, R., & Kumar, V. (2020). A review on bioactive peptides: physiological functions, bioavailability and safety. International Journal of Peptide Research and Therapeutics, 26(1), 139–150. https://doi.org/10.1007/s10989-019-09823-5.
Bhullar, K. S., & Wu, J. (2020). Dietary peptides in aging: evidence and prospects. Food Science and Human Wellness, 9(1), 1–7. https://doi.org/10.1016/j.fshw.2020.01.001.
Brunner, G. (2009). Near critical and supercritical water. Part I. Hydrolytic and hydrothermal processes. The Journal of Supercritical Fluids, 47(3), 373–381. https://doi.org/10.1016/j.supflu.2008.09.002.
Cernadas, H., Flórez-Fernández, N., González-Muñoz, M. J., Domínguez, H., & Torres, M. D. (2019). Retrieving of high-value biomolecules from edible Himanthalia elongata brown seaweed using hydrothermal processing. Food and Bioproducts Processing, 117, 275–286. https://doi.org/10.1016/j.fbp.2019.07.015.
Cervantes-Cisneros, D. E., Arguello-Esparza, D., Cabello-Galindo, A., Picazo, B., Aguilar, C. N., Ruiz, H. A., & Rodríguez-Jasso, R. M. (2017). Hydrothermal processes for extraction of macroalgae high value-added compounds. Production of bioethanol and high added-value compounds of second and third generation biomass. In H. A. Ruiz, M. H. Thomsen, & H. L. Trajano (Eds.), Hydrothermal processing in biorefineries. Gewerbestrasse: Springer International Publishing AG.
Chakrabarti, S., Guha, S., & Majumder, K. (2018). Food-derived bioactive peptides in human health: challenges and opportunities. Nutrients, 10(11), 1738. https://doi.org/10.3390/nu10111738.
Chalamaiah, M., Keskin Ulug, S., Hong, H., & Wu, J. (2019). Regulatory requirements of bioactive peptides (protein hydrolysates) from food proteins. Journal of Functional Foods, 58, 123–129. https://doi.org/10.1016/j.jff.2019.04.050.
Chen, X.-W., Wang, J.-M., Yang, X.-Q., Qi, J.-R., & Hou, J.-J. (2016). Subcritical water induced complexation of soy protein and rutin: improved interfacial properties and emulsion stability. Journal of Food Science, 81(9), C2149–C2157. https://doi.org/10.1111/1750-3841.13403.
Chiou, T.-Y., Neoh, T. L., Kobayashi, T., & Adachi, S. (2010). Antioxidative ability of defatted rice bran extract obtained by subcritical water extraction in bulk oil and aqueous dispersion systems. Japan Journal of Food Engineering, 12(4), 147–154. https://doi.org/10.11301/jsfe.12.147.
Chiou, T.-Y., Neoh, T. L., Kobayashi, T., & Adachi, S. (2012). Properties of extract obtained from defatted rice bran by extraction with aqueous ethanol under subcritical conditions. Food Science and Technology Research, 18(1), 37–45. https://doi.org/10.3136/fstr.18.37.
Cian, R. E., Drago, S. R., Sáncez de Medina, F., & Martínez-Augustin, O. (2015). Proteins and carbohydrates from red seaweeds: evidence for beneficial effect on gut function and microbiota. Marine Drugs, 13(8), 5358–5383. https://doi.org/10.3390/md13085358.
Cui, Z., Cheng, F., Jarvis, J. M., Dungan, B., Brewer, C. E., & Jena, U. (2020). Roles of co-solvents in hydrothermal liquefaction of protein-rich algae. Bioresource Technology, 310, 123454. https://doi.org/10.13031/aim.201900360.
dos Santos Aguilar, J. G., & Sato, H. H. (2018). Microbial proteases: production and application in obtaining protein hydrolysates. Food Research International, 103, 253–262. https://doi.org/10.1016/j.foodres.2017.10.044.
Du, L., Arauzo, P. J., Meza Zavala, M. F., Cao, Z., Olszewski, M. P., & Kruse, A. (2020). Towards the properties of different biomass-derived proteins via various extraction methods. Molecules, 25(3), 488. https://doi.org/10.3390/molecules25030488.
El-Baky, H. H. A., El-Baz, F. K., & El-Baroty, G. S. (2009). Natural preservative ingredient from marine alga Ulva lactuca L. International Journal of Food Science and Technology, 44, 1688–1695. https://doi.org/10.1111/j.1365-2621.2009.01926.x.
Fan, X., Hu, S., Wang, K., Yang, R., & Zhang, X. (2020). Coupling of ultrasound and subcritical water for peptides production from Spirulina platensis. Food and Bioproducts Processing, 121, 105–112. https://doi.org/10.1016/j.fbp.2020.01.012.
Fernández-Tomé, S., Hernández-Ledesma, B., Chaparro, M., Indiano-Romacho, P., Bernardo, D., & Gisbert, J. P. (2019). Role of food proteins and bioactive peptides in inflammatory bowel disease. Trends in Food Science and Technology, 88, 194–206. https://doi.org/10.1016/j.tifs.2019.03.017.
Flórez-Fernández, N., Torres, M. D., González-Muñoz, M. J., & Domínguez, H. (2019). Recovery of bioactive and gelling extracts from edible brown seaweed Laminaria ochroleuca by non-isothermal autohydrolysis. Food Chemistry, 277, 353–361. https://doi.org/10.1016/j.foodchem.2018.10.096.
Fu, Q., Zhang, H., Chen, H., Liao, Q., Xia, A., Huang, Y., Zhu, X., Reungsang, A., & Liu, Z. (2018). Hydrothermal hydrolysis pretreatment of microalgae slurries in a continuous reactor under subcritical conditions for large–scale application. Bioresource Technology, 266, 306–314. https://doi.org/10.1016/j.biortech.2018.06.088.
Gallego, R., Bueno, M., & Herrero, M. (2019). Sub- and supercritical fluid extraction of bioactive compounds from plants, food-by-products, seaweeds and microalgae – an update. TrAC, Trends in Analytical Chemistry, 116, 198–213. https://doi.org/10.1016/j.trac.2019.04.030.
Garcia-Moscoso, J. L., Obeid, W., Kumar, S., & Hatcher, P. G. (2013). Flash hydrolysis of microalgae (Scenedesmus sp.) for protein extraction and production of biofuels intermediates. Journal of Supercritical Fluids, 82, 183–190. https://doi.org/10.1016/j.supflu.2013.07.012.
Garcia-Moscoso, J. L., Teymouri, A., & Kumar, S. (2015). Kinetics of peptides and arginine production from microalgae (Scenedesmus sp.) by flash hydrolysis. Industrial and Engineering Chemistry Research, 54(7), 2048–2058. https://doi.org/10.1021/ie5047279.
Garrote, G., Domínguez, H., & Parajó, J. C. (1999). Hydrothermal processing of lignocellulosic materials. Holz als Roh und Werkstoff, 57(3), 191–202. https://doi.org/10.1007/s001070050039.
Garrote, G., Moure, A., & Domínguez, H. (2017). Effect of hydrothermal pretreatment on lignin and antioxidant activity. In H. A. Ruiz, M. H. Thomsen, & H. L. Trajano (Eds.), Hydrothermal processing in biorefineries. Production of bioethanol and high-added value compounds of second and third generation biomass. Boca Raton: Springer.
Gereniu, C. R. N., Saravana, P. S., Getachew, A. T., & Chun, B.-S. (2017). Characteristics of functional materials recovered from Solomon Islands red seaweed (Kappaphycus alvarezii) using pressurized hot water extraction. Journal of Applied Phycology, 29(3), 1609–1621. https://doi.org/10.1007/s10811-017-1052-3.
Getachew, A. T., & Chun, B. S. (2017). Influence of pretreatment and modifiers on subcritical water liquefaction of spent coffee grounds: a green waste valorization approach. Journal of Cleaner Production, 142, 3719–3727. https://doi.org/10.1016/j.jclepro.2016.10.096.
Ghanbari, R. (2019). Review on the bioactive peptides from marine sources: indication for health effects. International Journal of Peptide Research and Therapeutics, 25(3), 1187–1199. https://doi.org/10.1007/s10989-018-9766-x.
Guha, S., & Majumder, K. (2019). Structural-features of food-derived bioactive peptides with anti-inflammatory activity: a brief review. Journal of Food Biochemistry, 43(1), e12531. https://doi.org/10.1111/jfbc.12531.
Guidotti, G., Brambilla, L., & Rossi, D. (2019). Peptides in clinical development for the treatment of brain tumors. Current Opinion in Pharmacology, 47, 102–109. https://doi.org/10.1016/j.coph.2019.02.007.
Hajfathalian, M., Ghelichi, S., García-Moreno, P. J., Moltke Sørensen, A.-D., & Jacobsen, C. (2018). Peptides: production, bioactivity, functionality, and applications. Critical Reviews in Food Science and Nutrition, 58(18), 3097–3129. https://doi.org/10.1080/10408398.2017.1352564.
Hata, S., Wiboonsirikul, J., Maeda, A., Kimura, Y., & Adachi, S. (2008). Extraction of defatted rice bran by subcritical water treatment. Biochemical Engineering Journal, 40(1), 44–53. https://doi.org/10.1016/j.bej.2007.11.016.
Ho, T. C., & Chun, B.-S. (2019). Extraction of bioactive compounds from Pseuderanthemum palatiferum (Nees) Radlk. using subcritical water and conventional solvents: a comparison study. Journal of Food Science, 84(5), 1201–1207. https://doi.org/10.1111/1750-3841.14501.
Ho, C. H. L., Cacace, J. E., & Mazza, G. (2007). Extraction of lignans, proteins and carbohydrates from flaxseed meal with pressurized low polarity water. LWT - Food Science and Technology, 40(9), 1637–1647. https://doi.org/10.1016/j.lwt.2006.12.003.
Holdt, S. L., & Kraan, S. (2011). Bioactive compounds in seaweed: functional food applications and legislation. Journal of Applied Phycology, 23(3), 543–597. https://doi.org/10.1007/s10811-010-9632-5.
Hou, Y., Wu, Z., Dai, Z., Wang, G., & Wu, G. (2017). Protein hydrolysates in animal nutrition: industrial production, bioactive peptides, and functional significance. Journal of Animal Science and Biotechnology, 8(1), 24. https://doi.org/10.1186/s40104-017-0153-9.
Hu, S., Fan, X., Qi, P., & Zhang, X. (2019). Identification of anti-diabetes peptides from Spirulina platensis. Journal of Functional Foods, 56, 333–341. https://doi.org/10.1016/j.jff.2019.03.024.
Hwang, Y. H., Cho, H.-Y., Kim, K.-R., Lee, S. H., & Choi, M.-J. (2015). Hydrolysis of isolate soybean protein using subcritical water. Korean Journal of Food Science and Technology, 47(6), 772–778. https://doi.org/10.9721/KJFST.2015.47.6.772.
Hwang, Y.-H., Kim, G., Shin, J.-K., Lee, S., Pyun, Y. R., & Cho, H.-Y. (2017). Enzymatic susceptibility of wheat gluten after subcritical water treatment. Food Science and Biotechnology, 26(6), 1545–1553. https://doi.org/10.1007/s10068-017-0214-z.
Jahandideh, F., & Wu, J. (2020). Perspectives on the potential benefits of antihypertensive peptides towards metabolic syndrome. International Journal of Molecular Sciences, 21(6), 2192. https://doi.org/10.3390/ijms21062192.
Jiménez-Escrig, A., Gómez-Ordóñez, E., & Rupérez, P. (2012). Brown and red seaweeds as potential sources of antioxidant nutraceuticals. Journal of Applied Phycology, 24, 1123–1132. https://doi.org/10.1007/s10811-011-9742-8.
Kataoka, M., Kimura, Y., Adachi, S., & Wiboonsirikul, J. (2008). Properties of extracts from wheat bran by subcritical water treatment. Food Science and Technology Research, 14(6), 553–556. https://doi.org/10.3136/fstr.14.553.
Katayama, S., & Nakamura, S. (2019). Emerging roles of bioactive peptides on brain health promotion. International Journal of Food Science and Technology, 54(6), 1949–1955. https://doi.org/10.1111/ijfs.14076.
Kazan, A., Celiktas, M. S., Sargin, S., & Yesil-Celiktas, O. (2015). Bio-based fractions by hydrothermal treatment of olive pomace: process optimization and evaluation. Energy Conversion and Management, 103, 366e373. https://doi.org/10.1016/j.enconman.2015.06.084.
Kehinde, B. A., & Sharma, P. (2018). Recently isolated antidiabetic hydrolysates and peptides from multiple food sources: a review. Critical Reviews in Food Science and Nutrition, 60(2), 322–340. https://doi.org/10.1080/10408398.2018.1528206.
Khuwijitjaru, P., Anantanasuwong, S., & Adachi, S. (2011). Emulsifying and foaming properties of defatted soy meal extracts obtained by subcritical water treatment. International Journal of Food Properties, 14(1), 9–16. https://doi.org/10.1080/10942910903112118.
King, J. W. (2006). Pressurized water extraction: resources and techniques for optimizing analytical applications. ACS Symposium Series, 926, 79–95. https://doi.org/10.1021/bk-2006-0926.ch006.
Kumar, S., Hablot, E., Moscoso, J. L. G., Obeid, W., Hatcher, P. G., Duquette, B. M., Graiver, D., Narayan, R., & Balan, V. (2014). Polyurethanes preparation using proteins obtained from microalgae. Journal of Materials Science, 49(22), 7824–7833. https://doi.org/10.1007/s10853-014-8493-8.
Lammi, C., Aiello, G., Boschin, G., & Arnoldi, A. (2019). Multifunctional peptides for the prevention of cardiovascular disease: a new concept in the area of bioactive food-derived peptides. Journal of Functional Foods, 55, 135–145. https://doi.org/10.1016/j.jff.2019.02.016.
Lamp, A., Kaltschmitt, M., & Lüdtke, O. (2020). Protein recovery from bioethanol stillage by liquid hot water treatment. Journal of Supercritical Fluids, 155, 104624. https://doi.org/10.1016/j.supflu.2019.104624.
Lee, Y.-K., Ko, B.-B., Min, S.-G., & Hong, G.-P. (2015). Effect of soy protein hydrolysates prepared by subcritical water processing on the physicochemical properties of pork patty during chilled storage. Korean Journal for Food Science of Animal Resources, 35(4), 557–563. https://doi.org/10.5851/kosfa.2015.35.4.557.
Lee, E. J., Hur, J., Ham, S. A., Jo, Y., Lee, S., Choi, M.-J., & Seo, H. G. (2017). Fish collagen peptide inhibits the adipogenic differentiation of preadipocytes and ameliorates obesity in high fat diet-fed mice. International Journal of Biological Macromolecules, 104, 281–286. https://doi.org/10.1016/j.ijbiomac.2017.05.151.
Li, Y., Lammi, C., Boschin, G., Arnoldi, A., & Aiello, G. (2019a). Recent advances in microalgae peptides: cardiovascular health benefits and analysis. Journal of Agricultural and Food Chemistry, 67(43), 11825–11838. https://doi.org/10.1021/acs.jafc.9b03566.
Li, B., Yang, W., Nie, Y., Kang, F., Goff, H. D., & Cui, S. W. (2019b). Effect of steam explosion on dietary fiber, polysaccharide, protein and physicochemical properties of okara. Food Hydrocolloids, 94, 48–56. https://doi.org/10.1016/j.foodhyd.2019.02.042.
Lima, T. N., & Moraes, C. A. P. (2018). Bioactive peptides: applications and relevance for cosmeceuticals. Cosmetics, 5(1), 21. https://doi.org/10.3390/cosmetics5010021.
Liu, L., Li, S., Zheng, J., Bu, T., He, G., & Wu, J. (2020). Safety considerations on food protein-derived bioactive peptides. Trends in Food Science and Technology, 96, 199–207. https://doi.org/10.1016/j.tifs.2019.12.022.
Lorente, E., Hapońska, M., Clavero, E., Torras, C., & Salvadó, J. (2018). Steam explosion and vibrating membrane filtration to improve the processing cost of microalgae cell disruption and fractionation. Processes, 6(4), 28. https://doi.org/10.3390/pr6040028.
Lu, W., Ding, J., Guan, Y.-H., Wang, J.-M., & Yang, X.-Q. (2015). Extraction and properties of soy protein from heat-denatured soy meal using enzyme-assisted subcritical water treatment. Modern Food Science and Technology, 31(1), 126–130. https://doi.org/10.13982/j.mfst.1673-9078.2015.1.023.
Ma, M., Ren, Y., Xie, W., Zhou, D., Tang, S., Kuang, M., Wang, Y., & Du, S.-K. (2018). Physicochemical and functional properties of protein isolate obtained from cottonseed meal. Food Chemistry, 240, 856–862. https://doi.org/10.1016/j.foodchem.2017.08.030.
Mada, S. B., Ugwu, C. P., & Abarshi, M. M. (2019). Health promoting effects of food-derived bioactive peptides: a review. International Journal of Peptide Research and Therapeutics. https://doi.org/10.1007/s10989-019-09890-8.
Marcet, I., Álvarez, C., Paredes, B., & Díaz, M. (2016). The use of sub-critical water hydrolysis for the recovery of peptides and free amino acids from food processing wastes. Review of sources and main parameters. Waste Management, 49, 364–371. https://doi.org/10.1016/j.wasman.2016.01.009.
Marciniak, A., Suwal, S., Naderi, N., Pouliot, Y., & Doyen, A. (2018). Enhancing enzymatic hydrolysis of food proteins and production of bioactive peptides using high hydrostatic pressure technology. Trends in Food Science and Technology, 80, 187–198. https://doi.org/10.1016/j.tifs.2018.08.013.
Marquez-Rios, E., & Del-Toro-Sánchez, C. L. (2018). Antioxidant peptides from terrestrial and aquatic plants against cancer. Current Protein and Peptide Science, 19(4), 368–379. https://doi.org/10.2174/1389203718666170111120527.
Mazorra-Manzano, M. A., Ramírez-Suarez, J. C., & Yada, R. Y. (2018). Plant proteases for bioactive peptides release: a review. Critical Reviews in Food Science and Nutrition, 58(13), 2147–2163. https://doi.org/10.1080/10408398.2017.1308312.
Mirmoghtadaie, L., Shojaee Aliabadi, S., & Hosseini, S. M. (2016). Recent approaches in physical modification of protein functionality. Food Chemistry, 199, 619–627. https://doi.org/10.1016/j.foodchem.2015.12.067.
Montesano, D., Gallo, M., Blasi, F., & Cossignani, L. (2020). Biopeptides from vegetable proteins: new scientific evidences. Current Opinion in Food Science, 31, 31–37. https://doi.org/10.1016/j.cofs.2019.10.008.
Moreno-Valdespino, C. A., Luna-Vital, D., Camacho-Ruiz, R. M., & Mojica, L. (2020). Bioactive proteins and phytochemicals from legumes: mechanisms of action preventing obesity and type-2 diabetes. Food Research International, 130, 108905. https://doi.org/10.1016/j.foodres.2019.108905.
Ndlela, S. C., De Moura, J. M. L. N., Olson, N. K., & Johnson, L. A. (2012). Aqueous extraction of oil and protein from soybeans with subcritical water. Journal of the American Oil Chemists' Society, 89(6), 1145–1153. https://doi.org/10.1007/s11746-011-1993-7.
Neto, R. T., Marçal, C., Queirós, A. S., Abreu, H., Silva, A. M. S., & Cardoso, S. M. (2018). Screening of Ulva rigida, Gracilaria sp., Fucus vesiculosus and Saccharina latissima as functional ingredients. International Journal of Molecular Sciences, 19, 2987. https://doi.org/10.3390/ijms19102987.
Nomura, S., Lee, W.-J., Konishi, M., Saitoh, T., Murata, M., Ohtsu, N., Shimotori, Y., Kohari, Y., Nagata, Y., & Chiou, T.-Y. (2019). Characteristics of Japanese mint extracts obtained by subcritical-water treatment. Food Science and Technology Research, 25(5), 695–703. https://doi.org/10.3136/fstr.25.695.
Oshiro, K. G. N., Rodrigues, G., Monges, B. E. D., Cardoso, M. H., & Franco, O. L. (2019). Bioactive peptides against fungal biofilms. Frontiers in Microbiology, 10, 2169. https://doi.org/10.3389/fmicb.2019.02169.
Overend, R. P., & Chornet, E. (1987). Fractionation of lignocellulosics by steam-aqueous pretreatments. Philosophical Transactions of the Royal Society A, 321, 523–536.
Ozuna, C., Paniagua-Martínez, I., Castaño-Tostado, E., Ozimek, L., & Amaya-Llano, S. L. (2015). Innovative applications of high-intensity ultrasound in the development of functional food ingredients: production of protein hydrolysates and bioactive peptides. Food Research International, 77, 685–696. https://doi.org/10.1016/j.foodres.2015.10.015.
Pangestuti, R., Getachew, A. T., Siahaan, E. A., & Chun, B.-S. (2019). Characterization of functional materials derived from tropical red seaweed Hypnea musciformis produced by subcritical water extraction systems. Journal of Applied Phycology, 31(4), 2517–2528. https://doi.org/10.1007/s10811-019-1754-9.
Pangestuti, R., Getachew, A. T., Siahaan, E. A., & Chun, B. S. (2020). Characteristics of functional materials recovered from Indonesian mangroves (Sonneratia alba and Rhizhophora mucronata) using subcritical water extraction. E3S Web of Conferences, 147, 03013. https://doi.org/10.1051/e3sconf/202014703013.
Park, J.-S., Jeong, Y.-R., & Chun, B.-S. (2019). Physiological activities and bioactive compound from laver (Pyropia yezoensis) hydrolysates by using subcritical water hydrolysis. Journal of Supercritical Fluids, 148, 130–136. https://doi.org/10.1016/j.supflu.2019.03.004.
Patel, R., Santhosh, M., Dash, J. K., Karpoormath, R., Jha, A., Kwak, J., Patel, M., & Kim, J. H. (2019). Ile-Lys-Val-ala-Val (IKVAV) peptide for neuronal tissue engineering. Polymers for Advanced Technologies, 30(1), 4–12. https://doi.org/10.1002/pat.4442.
Phusunti, N., Phetwarotai, W., Tirapanampai, C., & Tekasakul, S. (2017). Subcritical water hydrolysis of microalgal biomass for protein and pyrolytic bio-oil recovery. Bioenergy Research, 10(4), 1005–1017. https://doi.org/10.1007/s12155-017-9859-y.
Pińkowska, H., & Oliveros, E. (2014). Application of the doehlert matrix for the determination of the optimal conditions of hydrolysis of soybean protein in subcritical water. Industrial and Engineering Chemistry Research, 53(4), 1320–1326. https://doi.org/10.1021/ie403451b.
Pińkowska, H., Wolak, P., & Oliveros, E. (2013). Application of Doehlert matrix for determination of the optimal conditions of hydrothermolysis of rapeseed meal in subcritical water. Fuel, 106, 258–264. https://doi.org/10.1016/j.fuel.2012.12.023.
Pińkowska, H., Krzywonos, M., & Wolak, P. (2019). Valorization of rapeseed meal by hydrothermal treatment – effect of reaction parameters on low molecular products distribution. Cellulose Chemistry and Technology, 53(7–8), 755–765. https://doi.org/10.35812/CelluloseChemTechnol.2019.53.74.
Plaza, M., Amigo-Benavent, M., del Castillo, M. D., Ibáñez, E., & Herrero, M. (2010). Facts about the formation of new antioxidants in natural samples after subcritical water extraction. Food Research International, 43, 2341–2348. https://doi.org/10.1016/j.foodres.2010.07.036.
Pojić, M., Mišan, A., & Tiwari, B. (2018). Eco-innovative technologies for extraction of proteins for human consumption from renewable protein sources of plant origin. Trends in Food Science and Technology, 75, 93–104. https://doi.org/10.1016/j.tifs.2018.03.010.
Ponthier, E., Domínguez, H., & Torres, M.D. (2020). The microwave assisted extraction sway on the features of antioxidant compounds and gelling biopolymers from Mastocarpus stellatus. https://doi.org/10.1016/j.algal.2020.102081.
Powell, T., Bowra, S., & Cooper, H. J. (2016). Subcritical water processing of proteins: an alternative to enzymatic digestion? Analytical Chemistry, 88(12), 6425–6432. https://doi.org/10.1021/acs.analchem.6b01013.
Premkumar, J., Malini, M., & Arun Joshy, V. (2019). A critical review on food protein derived antihypertensive peptides. Drug Invention Today, 12(3), 474–479.
Priya, S. (2019). Therapeutic perspectives of food bioactive peptides: a mini review. Protein and Peptide Letters, 26(9), 664–675. https://doi.org/10.2174/0929866526666190617092140.
Quitain, A.T., Daimon, H., Fujie, K., Katoh, S., & Moriyoshi, T. (2006). Microwave-assisted hydrothermal degradation of silk protein to amino acids. Industrial and Engineering Chemistry Research, 45(13), 4471–4474.
Ramachandraiah, K., Koh, B.-B., Davaatseren, M., & Hong, G.-P. (2017). Characterization of soy protein hydrolysates produced by varying subcritical water processing temperature. Innovative Food Science and Emerging Technologies, 43, 201–206. https://doi.org/10.1016/j.ifset.2017.08.011.
Ravber, M., Knez, Ž., & Škerget, M. (2015a). Simultaneous extraction of oil- and water-soluble phase from sunflower seeds with subcritical water. Food Chemistry, 166, 316–323. https://doi.org/10.1016/j.foodchem.2014.06.025.
Ravber, M., Knez, Z., & Škerget, M. (2015b). Hydrothermal degradation of fats, carbohydrates and proteins in sunflower seeds after treatment with subcritical water. Chemical and Biochemical Engineering Quarterly, 29(3), 351–355. https://doi.org/10.15255/CABEQ.2015.2193.
Reisinger, M., Tirpanalan, Ö., Pruksasri, S., Kneifel, W., & Novalin, S. (2018). Disintegration of the agricultural by-product wheat bran under subcritical conditions. Journal of the Science of Food and Agriculture, 98(11), 4296–4303. https://doi.org/10.1002/jsfa.8952.
Rengasamy, K. R. R., Khan, H., Ahmad, I., Lobine, D., Mahomoodally, F., Suroowan, S., Hassan, S. T. S., Xu, S., Patel, S., Daglia, M., Nabavi, S. M., & Pandian, S. K. (2019). Bioactive peptides and proteins as alternative antiplatelet drugs. Medicinal Research Reviews, 39(6), 2153–2171. https://doi.org/10.1002/med.21579.
Reyes-Díaz, A., Del Toro-Sánchez, C. L., Rodríguez-Figueroa, J. C., Valdéz-Hurtado, S., Womg-Corral, F. J., Borboa-Flores, J., González-Osuna, M. F., Perez-Perez, L. M., & González-Vega, R. I. (2019). Legume proteins as a promising source of anti-inflammatory peptides. Current Protein and Peptide Science, 20(12), 1204–1217. https://doi.org/10.2174/1389203720666190430110647.
Rogalinski, T., Liu, K., Albrecht, T., & Brunner, G. (2008). Hydrolysis kinetics of biopolymers in subcritical water. Journal of Supercritical Fluids, 46(3), 335–341. https://doi.org/10.1016/j.supflu.2007.09.037.
Santoyo, S., Plaza, M., Jaime, L., Ibañez, E., Reglero, G., & Señorans, J. (2011). Pressurized liquids as an alternative green process to extract antiviral agents from the edible seaweed Himanthalia elongata. Journal of Applied Phycology, 23(5), 909–917.
Saravana, P. S., Cho, Y.-J., Park, Y.-B., Woo, H.-C., & Chun, B.-S. (2016a). Structural, antioxidant, and emulsifying activities of fucoidan from Saccharina japonica using pressurized liquid extraction. Carbohydrate Polymers, 153, 518–525. https://doi.org/10.1016/j.carbpol.2016.08.014.
Saravana, P. S., Choi, J. H., Park, Y. B., Woo, H. C., & Chun, B. S. (2016b). Evaluation of the chemical composition of brown seaweed (Saccharina japonica) hydrolysate by pressurized hot water extraction. Algal Research, 13, 246–254. https://doi.org/10.1016/j.algal.2015.12.004.
Sato, N., Quitain, A. T., Kang, K., Daimon, H., & Fujie, K. (2004). Reaction kinetics of amino acid decomposition in high-temperature and high-pressure water. Industrial and Engineering Chemistry Research, 43(13), 3217–3222. https://doi.org/10.1021/ie020733n.
Sereewatthanawut, I., Prapintip, S., Watchiraruji, K., Goto, M., Sasaki, M., & Shotipruk, A. (2008). Extraction of protein and amino acids from deoiled rice bran by subcritical water hydrolysis. Bioresource Technology, 99(3), 555–561. https://doi.org/10.1016/j.biortech.2006.12.030.
Soto-Sierra, L., Stoykova, P., & Nikolov, Z. L. (2018). Extraction and fractionation of microalgae-based protein products. Algal Research, 36, 175–192. https://doi.org/10.1016/j.algal.2018.10.023.
Sunphorka, S., Chavasiri, W., Oshima, Y., & Ngamprasertsith, S. (2012a). Kinetic studies on rice bran protein hydrolysis in subcritical water. Journal of Supercritical Fluids, 65, 54–60. https://doi.org/10.1016/j.supflu.2012.02.017.
Sunphorka, S., Chavasiri, W., Oshima, Y., & Ngamprasertsith, S. (2012b). Protein and sugar extraction from rice bran and de-oiled rice bran using subcritical water in a semi-continuous reactor: optimization by response surface methodology. International Journal of Food Engineering, 8(3), 26. https://doi.org/10.1515/1556-3758.2262.
Supawong, S., Park, J. W., & Thawornchinsombut, S. (2019). Effect of rice bran hydrolysates on physicochemical and antioxidative characteristics of fried fish cakes during repeated freeze-thaw cycles. Food Bioscience, 32(2), 100471. https://doi.org/10.1016/j.fbio.2019.100471.
Susanto, E., Fahmi, A. S., Abe, M., Hosokawa, M., & Miyashita, K. (2016). Lipids, fatty acids and fucoxanthin content from temperate and tropical brown seaweeds. Aquatic Procedia, 7, 66–75. https://doi.org/10.1016/j.aqpro.2016.07.009.
Thiruvenkadam, S., Izhar, S., Yoshida, H., Danquah, M. K., & Harun, R. (2015). Process application of subcritical water extraction (SWE) for algal bio-products and biofuels production. Applied Energy, 154, 815–828. https://doi.org/10.1016/j.apenergy.2015.05.076.
Torres, M. D., Flórez-Fernández, N., & Domínguez, H. (2019a). Integral utilization of red seaweed for bioactive production. Marine Drugs, 17, 314. https://doi.org/10.3390/md17060314.
Torres, M. D., Kraan, S., & Domínguez, H. (2019b). Seaweed biorefinery. Reviews in Environmental Science and Biotechnology, 18, 335–388. https://doi.org/10.1007/s11157-019-09496-y.
Udenigwe, C. C., & Aluko, R. E. (2012). Food protein-derived bioactive peptides: production, processing, and potential health benefits. Journal of Food Science, 77(1), R11–R24. https://doi.org/10.1111/j.1750-3841.2011.02455.x.
Viriya-Empikul, N., Wiboonsirikul, J., Kobayashi, T., & Adachi, S. (2012). Effects of temperature and flow rate on subcritical-water extraction from defatted rice bran. Food Science and Technology Research, 18(3), 333–340. https://doi.org/10.3136/fstr.18.333.
Wang, M.-P., Lu, W., Yang, J., Wang, J.-M., & Yang, X.-Q. (2017). Preparation and characterisation of isoflavone aglycone-rich calcium-binding soy protein hydrolysates. International Journal of Food Science and Technology, 52(10), 2230–2237. https://doi.org/10.1111/ijfs.13502.
Wang, Y., Huang, Q., Kong, D., & Xu, P. (2018). Production and functionality of food-derived bioactive peptides: a review. Mini-Reviews in Medicinal Chemistry, 18(18), 1524–1535. https://doi.org/10.2174/1389557518666180424110754.
Wang, C.-H., Doan, C. T., Nguyen, V. B., Nguyen, A. D., & Wang, S.-L. (2019a). Reclamation of fishery processing waste: a mini-review. Molecules, 24(12), 2234. https://doi.org/10.3390/molecules24122234.
Wang, M.-P., Chen, X.-W., Guo, J., Yang, J., Wang, J.-M., & Yang, X.-Q. (2019b). Stabilization of foam and emulsion by subcritical water-treated soy protein: effect of aggregation state. Food Hydrocolloids, 87, 619–628. https://doi.org/10.1016/j.foodhyd.2018.08.047.
Watchararuji, K., Goto, M., Sasaki, M., & Shotipruk, A. (2008). Value-added subcritical water hydrolysate from rice bran and soybean meal. Bioresource Technology, 99(14), 6207–6213. https://doi.org/10.1016/j.biortech.2007.12.021.
Wiboonsirikul, J., Hata, S., Tsuno, T., Kimura, Y., & Adachi, S. (2007a). Production of functional substances from black rice bran by its treatment in subcritical water. LWT - Food Science and Technology, 40(10), 1732–1740. https://doi.org/10.1016/j.lwt.2007.01.003.
Wiboonsirikul, J., Kimura, Y., Kadota, M., Morita, H., Tsuno, T., & Adachi, S. (2007b). Properties of extracts from defatted rice bran by its subcritical water treatment. Journal of Agricultural and Food Chemistry, 55(21), 8759–8765. https://doi.org/10.1021/jf072041l.
Wiboonsirikul, J., Mori, M., Khuwijitjaru, P., & Adachi, S. (2013). Properties of extract from okara by its subcritical water treatment. International Journal of Food Properties, 16(5), 974–982. https://doi.org/10.1080/10942912.2011.573119.
Youssef, F. S., Ashour, M. L., Singab, A. N. B., & Wink, M. (2019). A comprehensive review of bioactive peptides from marine fungi and their biological significance. Marine Drugs, 17(10), 559. https://doi.org/10.3390/md17100559.
Zainan, N. H., Sapardi, M. A. M., Ho, B. C. H., Siajam, S. I., Kamal, S. M. M., Danquah, M. K., & Harun, R. (2019). Kinetic and thermodynamic characterization of amino acids generation via subcritical water reaction of microalgae Nannochloropsis sp. biomass. Biomass Conversion and Biorefinery. https://doi.org/10.1007/s13399-019-00538-7.
Zainan, N. H., Thiruvenkadam, S., Danquah, M. K., & Harun, R. (2020). Biochemical analysis and potential applications of aqueous and solid products generated from subcritical water extraction of microalgae Chlorella pyrenoidosa biomass. Journal of Applied Phycology, 32(1), 111–126. https://doi.org/10.1007/s10811-019-01960-0.
Zamora-Sillero, J., Gharsallaoui, A., & Prentice, C. (2018). Peptides from fish by-product protein hydrolysates and its functional properties: an overview. Marine Biotechnology, 20(2), 118–130. https://doi.org/10.1007/s10126-018-9799-3.
Zhang, Y., Zhao, W., Yang, R., Ahmed, M. A., Hua, X., Zhang, W., & Zhang, Y. (2013). Preparation and functional properties of protein from heat-denatured soybean meal assisted by steam flash-explosion with dilute acid soaking. Journal of Food Engineering, 119(1), 56–64. https://doi.org/10.1016/j.jfoodeng.2013.05.008.
Zhang, Q.-T., Tu, Z.-C., Wang, H., Huang, X.-Q., Fan, L.-L., Bao, Z.-Y., & Xiao, H. (2015). Functional properties and structure changes of soybean protein isolate after subcritical water treatment. Journal of Food Science and Technology, 52(6), 3412–3421. https://doi.org/10.1007/s13197-014-1392-9.
Zhang, H., Liao, Q., Fu, Q., Chen, H., Huang, Y., Xia, A., Zhu, X., Reungsang, A., Liu, Z., & Li, J. (2018). Rheological properties of microalgae slurry under subcritical conditions for hydrothermal hydrolysis systems. Algal Research, 3, 78–83. https://doi.org/10.1016/j.algal.2018.04.026.
Zhang, J., Wen, C., Li, C., Duan, Y., Zhang, H., & Ma, H. (2019a). Antioxidant peptide fractions isolated from wheat germ protein with subcritical water extraction and its transport across Caco-2 cells. Journal of Food Science, 84(8), 2139–2146. https://doi.org/10.1111/1750-3841.14720.
Zhang, S., Zheng, L., Zheng, X., Ai, B., Yang, Y., Pan, Y., & Sheng, Z. (2019b). Effect of steam explosion treatments on the functional properties and structure of camellia (Camellia oleifera Abel.) seed cake protein. Food Hydrocolloids, 93, 189–197. https://doi.org/10.1016/j.foodhyd.2019.02.017.
Zhang, J., Wen, C., Zhang, H., Duan, Y., & Ma, H. (2020). Recent advances in the extraction of bioactive compounds with subcritical water: a review. Trends in Food Science and Technology, 95, 183–195. https://doi.org/10.1016/j.tifs.2019.11.018.
Zhao, G., Kuang, G., Wang, Y., Yao, Y., Zhang, J., & Pan, Z.-H. (2020). Effect of steam explosion on physicochemical properties and fermentation characteristics of sorghum (Sorghum bicolor (L.) Moench). LWT- Food Science and Technology, 129, 109579. https://doi.org/10.1016/j.lwt.2020.109579.
Zhu, G., Zhu, X., Xiao, Z., Zhou, R., Feng, N., & Niu, Y. (2015). A review of amino acids extraction from animal waste biomass and reducing sugars extraction from plant waste biomass by a clean method. Biomass Conversion and Biorefinery, 5(3), 309–320. https://doi.org/10.1007/s13399-014-0153-3.
Ziero, H. D. D., Buller, L. S., Mudhoo, A., Ampese, L. C., Mussatto, S. I., & Carneiro, T. F. (2020). An overview of subcritical and supercritical water treatment of different biomasses for protein and amino acids production and recovery. Journal of Environmental Chemical Engineering, 8(5), 104406. https://doi.org/10.1016/j.jece.2020.104406.
Funding
This research was funded by the Ministry of Science, Innovation and Universities of Spain, grant number RTI2018-096376-B-I00.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Álvarez-Viñas, M., Rodríguez-Seoane, P., Flórez-Fernández, N. et al. Subcritical Water for the Extraction and Hydrolysis of Protein and Other Fractions in Biorefineries from Agro-food Wastes and Algae: a Review. Food Bioprocess Technol 14, 373–387 (2021). https://doi.org/10.1007/s11947-020-02536-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11947-020-02536-4