Abstract
In some Brazilian coastal cities, it is common to observe ‘black tongues’ in beaches, i.e. a mixture of urban runoff and untreated domestic sewage containing pollutants of emerging concern, namely pharmaceutical and personal care products (PPCPs), flowing into the South Atlantic Ocean. Such diffuse loads of pollutants might expose nontarget aquatic organisms to harmful compounds. In this work, the occurrence and preliminary ecological risk of 27 PPCPs of various therapeutic classes (including cocaine and its primary metabolite, benzoylecgonine) were investigated, for the first time, in seven urban drainage channels whose diffuse loads flow continuously to the beaches of Santos Bay, São Paulo, Brazil. Of these, 21 compounds were detected using liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS), and nine of them were consistently quantified in all urban channels of Santos, suggesting that those pollutants are ubiquitous in this region: caffeine (143.4–516.0 ng/L), losartan (4.2–21.8 ng/L), atenolol (1.1–18.2 ng/L), acetaminophen (1.5–13.8 ng/L), benzoylecgonine (1.0–4.8 ng/L), carbamazepine (1.1–4.0 ng/L), diclofenac (1.9–3.5 ng/L), cocaine (0.5–1.7 ng/L), and orphenadrine (0.1–0.8 ng/L). Moreover, twelve compounds were found below the limit of quantification ( <LOQ): citalopram, propranolol, diazepam, rosuvastatin, atorvastatin, midazolam, ranitidine, chlortalidone, clopidogrel, chlorpheniramine, enalapril and valsartan. According to our knowledge, this is the first report on the occurrence of midazolam, ranitidine and chlorpheniramine in surface waters in Latin America and, therefore, these compounds should be considered environmental warning signs. A preliminary ecological risk assessment revealed that caffeine, acetaminophen and losartan presented a moderate risk, and carbamazepine a low risk to sensitive aquatic organisms at maximum measured concentrations. This study provides valuable information to reinforce the importance of a continuous monitoring of the diffuse loads (containing PPCPs and illicit drugs) flowing to the coastal zones in developing countries.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
The occurrence of emerging pollutants, such as pharmaceutical and personal care products (PPCPs), in the aquatic compartment, and their potential detrimental effects on these ecosystems, have raised concerns in the coastal zones worldwide (Dafouz et al. 2018; Desbiolles et al. 2018; Ojemaye and Petrik 2018). The high urban pressure in the coastline and associated anthropogenic activities, led to the unregulated disposal of domestic sewage and urban surface runoff in different marine environmental matrices (e.g. water, sediment, and/or biota) (Cortez et al. 2018; Blackburn et al. 2019; Maasz et al. 2019). PPCPs are being continuously reported in several coastal zones worldwide, although they are not routinely monitored and not legally regulated (Pereira et al. 2016; Quadra et al. 2017; Hossain et al. 2018). However, PPCPs (e.g. antiepileptics, stimulants, analgesics/antiinflammatories, and antihypertensive drugs) and illicit drugs (e.g. cocaine) are ubiquitous, potentially hazardous and environmentally pseudo-persistent in marine areas (Wang et al. 2019; Fontes et al. 2020; Valdez-Carrillo et al. 2020). Although most of these compounds are often detected at low concentrations (generally at the ng/L or μg/L levels), they are still of major concern because they may exert deleterious effects on aquatic nontarget organisms, such as algae, crustacean, molluscs and fishes (Cortez et al. 2018; Capaldo et al. 2019; Chen et al. 2019). Some of the reported harmful effects of low concentrations of PPCPs and illicit drugs includes behavioural changes, mortality, immobilisation, growth and reproduction inhibition, endocrine disruption, genotoxicity and carcinogenicity (Godoy and Kummrow 2017; Radwan et al. 2019; Valdez-Carrillo et al. 2020).
At present, most studies concerning the occurrence and risk assessment of PPCPs in coastal zones took place on Europe (Mijangos et al. 2018; González-Mariño et al. 2019; Maasz et al. 2019), North America (Klosterhaus et al. 2013; Lara-Martín et al. 2014; Anumol and Snyder 2015) and Asia (Bayen et al. 2016; Li et al. 2016; Hossain et al. 2018). This kind of information regarding the coastal zones of the Latin America is however scarce (Starling et al. 2018; Griffero et al. 2019; Peña-Guzmán et al. 2019). Although Brazil is the ninth largest producer of pharmaceuticals in the world and approximately 50 million people live in coastal municipalities (along 8,500 km of coastline) (Quadra et al. 2017; Ibge 2019; Roveri et al. 2020b), studies reporting the detection of PPCPs in its coastline are relatively new and still limited (Pereira et al. 2016; Fontes et al. 2020; Roveri et al. 2020a, 2020b).
Most of the studies conducted in coastal zones across the world, showed that the discharge of wastewater treatment plants (WWTPs) is one of the major entry sources of PPCPs and illicit drugs into the marine environment (Desbiolles et al. 2018; Ojemaye and Petrik 2018; Fontes et al. 2020). However, the works regarding the detection of PPCPs and illicit drugs in urban drainage channels in coastal zones across the world (e.g. Brazil) are almost inexistent (Roveri et al. 2020a and 2020b). This is the case of Santos, one of the coastal cities of São Paulo, Brazil, which suffers from environmental deterioration due to several anthropogenic stressors (e.g. strong urbanisation, port activities, and seasonal tourism) (Moreira et al. 2017; SMA/CPLA 2018; Ibge 2019). Historically, the channels of Santos are responsible for carrying the diffuse load of the city (popularly known as ‘black tongues’: mixture of urban runoff and untreated domestic sewage) (Rocha et al. 2011) onto the tourist beaches (Ambrozevicius and Abessa 2008; Ferraz et al. 2012; Lamparelli et al. 2015). Some studies conducted in the channels of Santos have already identified urban runoff as a potential threat to public and environmental health, as it is responsible for introducing chemical and biological pollutants directly onto the beaches, which are areas of intense human recreation (Ferraz et al. 2012; Lamparelli et al. 2015; Gandra et al. 2020).
Recently, the occurrence of a vast mixture of 16 PPCPs and illicit drugs in urban drainage channels of Guarujá, a neighbouring city of Santos, showed that five of these compounds, namely caffeine, acetaminophen, diclofenac, losartan and valsartan, presented moderate to severe risks to the aquatic biota (Roveri et al. 2020b). This emphasises the need to screen and identify the presence of PPCPs in the urban drainage channels of Santos (a city with substantial urban development) and to determine the potential associated ecological risks.
In this context, the purpose of this study was to assess, for the first time, the occurrence and the ecological risk potential of 27 PPCPs of various therapeutic classes (including cocaine and its primary metabolite, benzoylecgonine) along the beaches of Santos, which daily receive diffuse loads from nearby neighbourhoods through seven urban drainage channels. The gathered information is crucial to monitor and to manage the diffuse loads that flow to this important Brazilian recreational area.
Materials and methods
Study site and sample collection
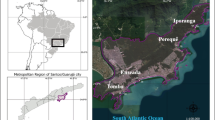
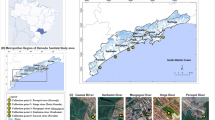
This study took place in Santos city (23°57′52″S 46°20′0″W), located within the metropolitan area of Baixada Santista, São Paulo, Brazil. Santos has a tropical climate, characterised by a mean annual precipitation and temperature of 3000 mm and 22°C, respectively. Two main annual seasons are observed in the region: a rainy (November to March) and a dry (April to October) season (SMA/CPLEA 2016; SMA/SPLA 2018). Santos has an area of 280.6 km2 of which 39.8 km2 are already completely urbanised (island area). Santos has a permanent population of approximately 430,000 inhabitants (Ibge 2019), which practically doubles during the summer, between December and February (Cetesb 2019). It is estimated that about 99.3% of the population lives in the island area (SMA/CPLEA, 2016; SMA/SPLA, 2018; Ibge, 2019). The remaining 240.8 km2 are located on the mainland, of which about 150 km2 comprises environmental preservation areas, which therefore remain uninhabited (SMA/CPLEA, 2016; SMA/SPLA, 2018; Ibge, 2019). Santos city holds the largest marine port in Latin America, but the commerce, fishing and tourism occurring along the sandy beaches are also economically important (SMA/CPLEA, 2016; SMA/SPLA, 2018). These beaches include seven urban drainage channels across the city edge, which are the natural boundaries of different neighbourhoods (José Menino; Gonzaga; Boqueirão; Embaré; Aparecida; and Ponta da Praia) (SMA/CPLEA, 2016; SMA/SPLA, 2018).
The field study was conducted along these urban drainage channels which are part of the city’s basic sanitation infrastructures (Ambrozevicius and Abessa, 2008; Cetesb, 2019; Gandra et al. 2020). Six of these channels are made of concrete and have a floodgate system with automatic mechanisms for their opening and closure. A seventh artificial channel, without floodgates, which flows continuously onto the beach also exists (Ambrozevicius and Abessa, 2008; Cetesb, 2019; Gandra et al. 2020). During storm events or periods of high rainfall, the floodgates are opened, and the diffuse loads are discharged directly onto the beaches, in an area of intense recreational activities (bathing waters) (Rocha et al. 2011; Cetesb, 2019). During the dry season or periods of low rainfall (condition observed in this study), the channel floodgates are closed and the diffuse loads drain towards the WWTP, through an underground oceanic interceptor located at the edge of the beaches (Ambrozevicius and Abessa, 2008; Cetesb, 2019; Gandra et al. 2020). However, this WWTP only performs a mechanical treatment (railing and screening for the removal of solids), followed by a chlorination step (De Souza Abessa et al. 2012; Baptistelli and Marcellino, 2016; Ortiz et al. 2016). The final destination of this preconditioned sewage is a submarine outfall 4500 m long and 10 m deep that daily disposes sewage into a low-wave energy semiclosed bay (De Souza Abessa et al. 2012; Baptistelli and Marcellino, 2016; Ortiz et al. 2016). For further details, see Fig.1.
Map of the study area showing Brazil (in grey colour), São Paulo State (A), the Metropolitan area of Baixada Santista (B) and Santos city (C). Water sampling locations (urban surface runoff) on the seven urban drainage channels of the municipality of Santos were shown in numbered yellow circles. These channels are made of concrete, and six of them (channels 1 to 6) have a floodgate system with an automatic closure (D) and opening (E) mechanism. During the dry period, the floodgates are closed and the diffuse load flows to the Santos wastewater treatment plant (WWTP), through an underground oceanic interceptor located on the edge of the beaches. However, during intense rain and floods events, the floodgates are opened and the diffuse loads are discharged directly into the bathing waters of Santos. The channel 7 does not have a floodgate system and therefore all the diffuse loads flow directly to Santos Bay (South Atlantic Ocean) throughout the year. All water samples were collected upstream the floodgates
A sampling point was selected at each channel, located at the mouth of the channel (upstream of the gate system) and therefore, unaffected by the tidal regime. The water samples were collected during the dry season and low tourist season, and weekends were avoided, to include only the disposal of PPCPs and illicit drugs by the resident population of Santos (Pereira et al. 2016; Fontes et al. 2019; Roveri et al. 2020b). A single water sample (1L) was collected on Wednesday, 21st October of 2020, from each location and packed into pre-cleaned amber glass bottles. All samples were kept at 4°C, and target PPCPs were extracted from water samples within 4 days of collection (USEPA, 2007).
Preparation and analysis of pharmaceutical compounds
Chemical and standards
Chemicals and analytical reagents such as nitric acid and sulphuric acid were purchased from Merck (Darmstadt, Germany). Grade solvents used in HPLC and LC-MS, such as acetonitrile, methanol and isopropanol, were acquired from Sigma-Aldrich (Massachusetts, USA). Mobile phase additives, LC-MS grade formic acid and ammonium acetate, were acquired from Sigma-Aldrich and Merck, respectively. Analytical standards of acetaminophen, atenolol, bromazepam, caffeine, carbamazepine, cyproterone, clonazepam, clopidogrel, diclofenac, enalapril, loratadine, losartan, midazolam orphenadrine, propranolol, sildenafil, atorvastatin, ranitidine, diazepam, chlorpheniramine and valsartan were acquired from Sigma-Aldrich. Cocaine and benzoylecgonine were acquired from Cerillant (Texas, USA). The other pharmaceuticals were bought in several suppliers: citalopram (Alcytam®: Torrent, Brazil), chlortalidone (Higroton®: Novartis, Swiss), rosuvastatin (Crestor®: AstraZeneca, UK) and generic paroxetine medication (Medley, Brazil). Analytical standards with purity grade > 98%.
Sample preparation
The extraction technique used was adopted from Roveri et al. (2020a). Prior to extraction, the following procedures were adopted: (i) the pH of each sample was adjusted to 7.0 ± 0.5 using a HCl solution (1 M); (ii) samples were filtered through a cellulose filter paper (Whatman® GF/C Glass microfiber filters, diameter 47 mm, particle retention 1.2 μm; Merck,Darmstadt, German); (iii) the filters were washed with 2 mL of methanol (Sigma-Aldrich, St. Louis, USA); and (iv) at the end, the methanol extract collected was then combined to the filtered sample. The solid-phase extraction was performed using SPE Chromabond HR-X cartridges (200 mg, 3 mL; Marcherey-Nagel, Duren, Germany). The cartridges were preconditioned with methanol (5 mL) and ultrapure water (5 mL) (Milli-Q®-Merck, Darmstadt, Germany). Thereafter, there were loaded with 1L of the filtered sample combined with the methanol from filter washings. The cartridges were then dried under vacuum for 30 min, and the elution was performed twice using 5 mL of methanol and 5 mL of acetone. After the extraction, the samples were dried under a nitrogen flow (at 50°C) and eluted with water/acetonitrile (95:5 v/v) prior to mass spectrometry analysis. In the laboratory each water sample was analysed in triplicate using liquid chromatography coupled with tandem mass spectrometry (LC–MS/MS). A concentration factor (1/1000) was used to obtain the final concentrations, and individual average results were expressed in ng/L (Table 1).
LC–MS/MS analysis
The hereby 27 PPCPs (Table S1a) screened were selected taking into the reported annual consumption, expected toxicity, environmental persistence and the previous results from other regional studies (Cemed, 2019; Roveri et al. 2020a, 2020b). LC-MS/MS analytical procedures were validated by Shihomatzu (2015) and fully described by Roveri et al. (2020a). Briefly, an aliquot of 10 μL of sample that was subjected to analysis by HPLC Agilent 1260 (Agilent™, Germany) combined with a mass spectrometer hybrid triple quadrupole/LIT instrument (3200QTRAP®-linear ion trap) (ABSciex, Ontario, Canada). The samples were analysed using an Agilent Zorbax Eclipse XDB–C18 column (50 × 4.6 mm ID, 1.8 μm column at 25°C). The eluent flow was 0.7 mL/min and the mobile phase for positive mode analysis was 0.1% formic acid (Sigma-Aldrich; LC–MS Grade) in water (solvent A) and acetonitrile (solvent B) (J.T. Baker, Philipsburg, NJ, USA). A linear gradient of 0.7 mL/min was used, starting with a mixture of solvent A (95%) and solvent B (5%). The percentage of solvent A was decreased linearly from 95% to 5% over 5 min, and this condition was maintained for 1 min. This mixture was then returned to initial conditions over 2 min and the analytes were detected and quantified using the electrospray ionisation (ESI) and multiple reaction monitoring (MRM), with the selection of a precursor ion and two ion products to quantify and qualify each compound. The data were recorded and processed using the Analyst 1.5.2 software (ABSciex). The MRM parameters for positive and negative ion modes, limit of detection (LOD) and limit of quantification (LOQ) are shown in Table S1a. The analytical quality control data is also show in Table S1b.
Ecological risk assessment
The ecological risk assessment followed the works of Roveri et al. (2020a, 2020b). The risk quotient (RQ) for three different aquatic organisms (algae, crustaceans and fishes) was calculated following the equation RQ = MEC/PNEC, in which MEC is the maximum Measured Environmental Concentration, and PNEC the Predicted No Effect Concentration, both expressed in ng/L. The PNEC values for the acute and chronic toxicity data were obtained from peer-reviewed publications by performing searches in the Ecotoxicology Database (ECOTOX) (USEPA, 2019), as well as in other literature sources using the PubMed database (see Table 2). When ecotoxicity laboratory experimentally derived data were not available, PNEC was estimated using the Ecological Structure Activity Relationships Programme (ECOSAR, v 2.0) (USEPA, 2017). An attempt was made to compile specifically PNEC data for marine coastal species. However, due to the strong land-sea interaction in this study area and the lack of marine toxicity data, the freshwater species were also taken into consideration in the present study (Roveri et al. 2020a, 2020b). The PNEC values for the acute and chronic toxicity data were thereafter calculated by dividing each toxicological endpoint by an assessment factor (AF). For saltwater environments, an AF of 10,000 and 100 should be considered in short- and long-term data sets, respectively. For further details, see the European Chemical Bureau (ECB, 2003) and the European Chemicals Agency (ECHA, 2008) guidelines. The toxicological endpoints selected for the calculation of the PNECs are shown in Table S3. Finally, RQ was categorised into four levels: no (RQ < 0.01), low (0.01 ≤ RQ < 0.1), moderate (0.1 ≤ RQ < 1.0) and high ecological risk (RQ ≥ 1.0) to aquatic organisms (Hernando et al. 2006).
Results and discussion
First evidence of PPCPs occurrence in the channels of Santos
The screening, identification and occurrence of compounds of different therapeutic classes, including illicit drugs such as cocaine and its metabolite benzoylecgonine, in the channels of Santos, is shown in Table 1. Of the 21 PPCPs detected (>LOD), 13 compounds were detected in all channels (100.0% frequency), indicating that those pollutants were ubiquitous in the marine environment of Santos: antiepileptics (carbamazepine); stimulants (caffeine, cocaine and benzoylecgonine); analgesics/antiinflammatories (acetaminophen, diclofenac, and orphenadrine); antihypertensives (atenolol, enalapril, losartan, and valsartan); an anticholesteremic (rosuvastatin); and an antihistamine (chlorpheniramine). Antidepressant (citalopram: 85.7%), diuretic (chlortalidone: 71.4%) and antihypertensive (propranolol: 57.1%) drugs were also detected. Five other compounds were detected at a frequency below 50%: antiplatelets (clopidogrel) and statins (atorvastatin) (both at 42.9%); and anxiolytics (midazolam), antiulcerous drugs (ranitidine) and antiepileptics (diazepam) (all at 28.6%).
This occurrence profile corresponds somewhat to the Brazilian pattern of PPCPs production and consumption. Brazil is the ninth largest producer of pharmaceutical drugs in the world; moreover, the pharmacies and drugstores, namely in the southeast region, are responsible for the majority of the sales in the country (Locatelli et al. 2010; Quadra et al. 2017; Cmed, 2019). These establishments sell nonprescription drugs, which means that the consumption of acetaminophen, diclofenac and orphenadrine is not controlled (Locatelli et al. 2010; Quadra et al. 2017; Cmed, 2019). The human excretion of PPCPs and illicit drugs in their parental, metabolised or conjugated forms trough urine and faeces, explains their occurrence in different environmental matrices (e.g. urban channels and marine environment) in the coastal zone of São Paulo (Pereira et al. 2016; Fontes et al. 2019; Roveri et al. 2020a, 2020b).
The nine PPCPs with concentrations above LOQ in the urban channels of Santos were caffeine (312.4 ± 152.3 ng/L; MEC = 516.0 ng/L) > losartan (11.8 ± 7.1 ng/L; MEC = 21.8 ng/L) > atenolol (7.9 ± 6.9 ng/L; MEC = 18.2 ng/L) > acetaminophen (6.0 ± 5.3 ng/L; MEC = 13.8 ng/L) > benzoylecgonine (2.6 ± 1.6 ng/L; MEC =4.8 ng/L) > carbamazepine (2.5 ± 1.1 ng/L; MEC = 4.0 ng/L), diclofenac (2.6 ± 0.6 ng/L; MEC =3.5 ng/L) > cocaine (1.1 ± 0.4 ng/L; MEC =1.7 ng/L) and orphenadrine (0.3 ± 0.2 ng/L; MEC = 0.8 ng/L) (Table 1; Fig. 2). These seven channels pass through six crowded neighbourhoods in Santos (e.g. José Menino; Gonzaga; Boqueirão; Embaré; Aparecida, and Ponta da Praia), where several commercial establishments, including restaurants, bars, supermarkets, pharmacies, drugstores, numerous hotel establishments, and also medical clinics and hospitals could be found. Consequently, these channels receive the daily input of urban runoff waters, which are usually mixed with unregulated domestic sewage, before they even reach the beaches of Santos (Lamparelli et al. 2015; Cetesb, 2019; Gandra et al. 2020). Moreover, sample collection was performed during the dry season and no rainfall was recorded for 72 h prior to collection. This means that channel floodgates 1 to 6 were closed, so that these diffuse loads drained towards the WWTP (for further information about the floodgates system, see Fig. 1) (Ambrozevicius and Abessa, 2008; Ferraz et al. 2012; Cetesb, 2019). Because the Santos WWTP is only a primary level system, typically designed to remove suspended solids from wastewaters and therefore inefficient in removing PPCPs and illicit drugs, the final destination of the preconditioned sewage (containing these 21 compounds) was a submarine outfall that disposes the sewage daily into the Santos Bay (a semiclosed and low energy coastal system) (Pereira et al. 2016; Ortiz et al. 2016; Fontes et al. 2019). Channel 7 does not have a floodgate system, and therefore all the diffuse load containing PPCPs and illicit drugs flow directly into Santos Bay without passing by the WWTP (South Atlantic Ocean) (Ambrozevicius and Abessa, 2008; Ferraz et al. 2012; Cetesb, 2019).
Occurrence of stimulants (caffeine)
The individual PPCPs concentrations quantified were generally below 50.0 ng/L for most of the target chemicals, except for caffeine (predominant compound in this study) which was present in considerably high concentrations in all channels (143.0-516.0 ng/L) (Fig. 2). Caffeine consumption is very high in Brazil, mainly due to the national consumption of various foods containing caffeine, such as beverages, stimulants, energy drinks, tea, coffee, soft drinks and painkillers (e.g. use in the formulation of acetaminophen) (Quadra et al. 2017). A study conducted in the stormwater pipes of Montreal, Canada, showed that caffeine concentrations above 400 ng/L are strongly correlated with water samples including at least 200 colony-forming-unit (CFU) per 100 mL of faecal coliforms (Potera, 2012; Sauvé et al. 2012). Moreover, some studies conducted by the São Paulo Environmental Agency (between 2010 and 2019) detected Escherichia coli (E. coli) at high concentrations in the Santos channels ( > 600 CFU/100 mL E. coli in 87.0% of samples). This indicates poor sanitation conditions, as this bacterium is an important marker of domestic sewage (present in human faeces in percentages between 96.0% and 99.0%) (Lamparelli et al. 2015; Cetesb, 2019). In this context, the concentrations of caffeine detected in this study (range: 143.4–516.0 ng/L) (Table 1) may also indicate faulty sanitation, since it is hardly removed with primary treatment (Sui et al. 2010). The high concentration of caffeine in urban surface waters has also been found in other countries, such as Singapore (1389.0 ng/L) (Bayen et al. 2016), China (430.0 ng/L) (Yang et al. 2018) and Uruguay (1120.0 ng/L) (Griffero et al. 2019). In short, the caffeine detected in the channels of Santos is a suitable marker for sewage contamination, due to its widespread occurrence, environmental persistence and high concentration.
Occurrence of stimulants (cocaine and benzoylecgonine)
In addition to caffeine, other psychoactive stimulants, such as cocaine and its metabolite benzoylecgonine, were also found in all channels (Fig.2). South America is almost responsible for all the world’s cocaine production, and specifically in Brazil, 1.75% of the young adult population of both sexes frequently uses it (Abdalla et al. 2014; Fontes et al. 2020; UNODC, 2020). This data raises concerns, because the use of this illicit drug may have negative effects on the health and social life of the population of Santos (Pereira et al. 2016; Campestrini and Jardim, 2017; Fontes et al. 2019). Cocaine is also a threat to the aquatic ecosystem, since it can interact biologically with nontarget organisms exerting deleterious effects (Fontes et al. 2019, 2020). The combined presence of cocaine and benzoylecgonine provides a reliable indication of human consumption of cocaine in Santos city, since cocaine is readily metabolised into benzoylecgonine, and higher quantities of benzoylecgonine are expected in the water, compared to those of cocaine (Li et al. 2016; González-Mariño et al. 2019; Maasz et al. 2019). In this context, the hereby results are similar to those obtained by other studies performed in surface waters of other countries, where the levels of benzoylecgonine detected were higher than those of cocaine, such as in Belgium (8.6 ng/L and 4.1 ng/L, respectively) (Van Nuijs et al. 2009), China (1.4 ng/L and 0.7 ng/L, respectively) (Li et al. 2016), and Hungary (2.3 ng/L and1.2 ng/L, respectively) (Maasz et al. 2019). Other studies have already shown the widespread contamination of Santos Bay by cocaine and benzoylecgonine in different environmental matrices (water, marine sediments and mussels), mainly, due to discharge from the WWTP (Pereira et al. 2016; Fontes et al. 2019, 2020). However, the present study also shows that the urban channels of Santos are also a source of PPCPs and illicit drugs for the marine ecosystem.
Occurrence of analgesic/ antiinflammatory and antihypertensive drugs
Another important factor that leads to a high consumption of pharmaceuticals worldwide, is the ageing population, as the immune system is weaker and the incidence of chronic diseases normally increases (Linjakumpu et al. 2002; Tummala et al. 2010). It is estimated that the population over 65 years of age usually consumes 5–10 pills/patient/day (Lacorte et al., 2017). Atenolol and losartan, for instance, are antihypertensives commonly taken by elderly people (Pereira et al. 2016). Even diclofenac, an antiinflammatory broad spectrum drug, can be used to treat osteoarthritis and rheumatoid arthritis (Locatelli et al. 2010; Cmed, 2019). Studies conducted in Germany (Herrmann et al. 2015), France, Spain and Portugal (Lacorte et al., 2017), showed that the most commonly administered pharmaceuticals in senior residences are carbamazepine, acetaminophen, and diclofenac (Herrmann et al. 2015; Lacorte et al., 2017). The elderly people of Santos is approximately 19.0% of the population, one of the largest values in the country (Ibge, 2019). This context could explain the occurrence of these five PPCPs in Santos, which were detected in all channels in relatively high concentrations (losartan: 4.2–21.8 ng/L, atenolol: 1.1–18.2 ng/L, acetaminophen: 1.5–13.8 ng/L, carbamazepine:1.1–4.0 ng/L and diclofenac: 1.9–3.5 ng/L) (Table 1; Fig. 2).
Warning signs
Overall, the concentrations of 21 PPCPs and illicit drugs detected in the seven urban drainage channels of Santos ranged from <LOQ to 516.0 ng/L (Table 1). This study also considers as warning signs even the twelve drugs with concentrations at levels <LOQ, but >LOD (e.g. citalopram, propranolol, diazepam, rosuvastatin, atorvastatin, midazolam, ranitidine, chlortalidone, clopidogrel, chlorpheniramine, enalapril and valsartan), because: (i) six of these PPCPs (citalopram, atorvastatin, midazolam, clopidogrel, chlorpheniramine and valsartan) have high n-octanol/water partition coefficients (log Kow ≥ 3), which indicate that they could bioaccumulate and exert toxicity (see Log Kow in Table S1a) (EMA, 2006; Mendoza et al. 2015; USEPA, 2017); (ii) although the samples were collected during low season, and therefore only involved the disposal of PPCPs and illicit drugs by the resident population of Santos, tourism increases the population in Santos during the summer season, holidays and weekends, and also the consumption of pharmaceuticals and illicit drugs (Pereira et al. 2016; Fontes et al. 2019; Molnar et al., 2020); (iii) according to recent reviews (Starling et al. 2018; Peña-Guzmán et al. 2019; Valdez-Carrillo et al. 2020), and to the best of our knowledge, this study seems to be the first report on the occurrence of midazolam, ranitidine and chlorpheniramine in surface waters in Latin America, which thus, deserve more attention and further investigation.
Comparison with other studies
The concentrations of the nine PPCPs quantified in the channels in this study were within those values reported in Europe (Wilkinson et al. 2017; Mijangos et al. 2018; Maasz et al. 2019), North America (Klosterhaus et al. 2013; Lara-Martín et al. 2014; Anumol and Snyder, 2015), Latin America (Spongberg et al. 2011; Rivera-Jaimes et al. 2018; Griffero et al. 2019) and Asia (Bayen et al. 2016; Li et al. 2016; Hossain et al. 2018). For more details, see Table S2. Again, these results also reflect the pattern of consumption on the coast of São Paulo and were already expected. Of the nine PPCPs quantified (including illicit drugs), seven compounds (caffeine, cocaine, benzoylecgonine, acetaminophen, diclofenac, atenolol and losartan) were also detected in the sewage discharges from Santos Bay and Enseada of Guarujá (Pereira et al. 2016; Fontes et al. 2019; Fontes et al. 2020; Roveri et al. 2020b), and mainly in urban channels and tourist beaches from Guarujá, where the detected concentrations were much higher than Santos (Table S2) (Roveri et al. 2020a). Therefore, these PPCPs could be considered environmental tracers of wastewater on the coast of São Paulo. It is necessary to understand the risks of these PPCPs to aquatic biota, because the MEC for caffeine (530.0 ng/L), losartan (21.8 ng/L), atenolol (18.2 ng/L) and acetaminophen (13.8 ng/L) were higher than 10.0 ng/L (Table 1), which is considered the threshold for the risk evaluation of pharmaceuticals in surface waters according to the European Medicines Agency (EMA, 2006).
Risk assessment of PPCPs and illicit drugs
Using the PNEC from data available in the scientific peer-reviewed literature or estimated by the ECOSARr programme, the RQ in the channels of Santos were calculated using the maximum MEC to evaluate the worst case of environmental risk for the aquatic biota. For further details, see Table 2 (summary data with the four compounds that indicated ecological potential risks) and Table S3 (complete data for the nine detected and quantified PPCPs).
The acute and chronic RQs for most individual PPCPs (e.g. carbamazepine, cocaine, benzoylecgonine, diclofenac, orphenadrine and atenolol) were equal or below 0.01, demonstrating a low to none ecological risk. However, even to some compounds which showed no ecological risk, other environmental issues, such as a high lipophilic character, low biodegradability and/or environmental pseudo-persistence cannot be neglected. Cocaine/carbamazepine and orphenadrine/diclofenac have moderate (log Kow > 2.3) to high (log Kow > 3.0) hydrophobicity, respectively, and therefore can potentially bioaccumulate (Table S1a) (EMA, 2006; Mendoza et al. 2015; USEPA, 2017), such as demonstrated for algae (Pseudokirchneriella subcapitata) and crustacean (Thamnocephalus platyurus) (Vernouillet et al. 2010). Moreover, the tendency of a substance for biological degradation (biodegradability) can be estimated by its kinetic reaction rate (k biol), based on grammes of suspended solids (ss) and days (L/gss day). In this context, diclofenac and carbamazepine are barely degradable (k biol < 0.5 L/gss day) and atenolol is considered moderately degradable: (0.5 < k biol < 1.0 L/gss day) (Schröder et al. 2016; Arola et al. 2017). Finally, PPCPs and illicit drugs have typical environmental pseudo-persistence (e.g. carbamazepine: half-life > 100 days in seawater) (USEPA, 2017) due to continuous input through these channels, and are likely to exert long-term effects which cannot usually be detected after short-term exposure (condition observed in this study, Table S3) (Di Poi et al., 2017). It means that toxic effects may therefore still be generated after a certain concentration is long-term accumulated in vivo (Wang et al. 2019; Radwan et al., 2019; Valdez-Carrillo et al. 2020).
Another important issue is that these compounds, even at very low concentrations, can have exerted toxicity in a mixture because of their combined effects (e.g. noninteractive, additive action, and antagonism or synergism) (Di Poi et al., 2017). PPCPs and illicit drugs found in the Santos channels are in a complex mixture of various therapeutic classes which may lead to an increase of the overall ecological risk (Wang et al. 2019; Radwan et al., 2019; Valdez-Carrillo et al. 2020). In this context, some studies have already showed that: (i) the combined effects of cocaine and benzoylecgonine induced genotoxicity and apoptotic cells in zebra mussels (Dreissena polymorpha) and in zebrafish (Danio rerio) embryos (Parolini et al. 2015, 2018); (ii) the sea urchin Paracentrotus lividus experienced developmental abnormalities when exposed to environmental combined concentrations of carbamazepine and ibuprofen (Aguirre-Martínez et al. 2015); and (iii) adult zebrafish (Danio rerio) showed significantly decreased embryo production after a 6 week exposure to environmental concentrations of acetaminophen, carbamazepine and gemfibrozil (Galus et al. 2013).
The occurrence of caffeine, acetaminophen and losartan are a cause for environmental concern because the RQ were high suggesting low to moderate ecological risk (for both acute and chronic exposures) for algae, crustacean and/or fishes (Table 2). The risks of these PPCPs have already been reported in previous works. For example, the MEC of caffeine detected in different Spanish freshwater bodies (similar to that obtained in Santos), including the Jarama River (MEC=410.0 ng/L) (Fernández et al., 2010), Henares River (MEC=670.0 ng/L) (Valcárcel et al. 2011) and Guadalquivir River (MEC=230.0 ng/L) (Robles-Molina et al. 2014), indicated a moderate ecological risk for the aquatic biota. Caffeine was also identified as high risk in the priority list of pharmaceuticals in European surface waters (Zhou et al. 2019). Previous studies also identified a moderate risk from acetaminophen to Daphnia magna in surface waters of the Pego–Oliva Marshlands, Valencia, Spain (RQ=0.3: Vazquez-Roig et al. 2012), Sindian river, Taiwan (RQ=0.9: Yu-Chen Lin et al. 2010) and Lahore channel, Pakistan (RQ=0.4: Ashfaq et al. 2019). Although studies on the toxicity of losartan are poorly documented (Pereira et al. 2016; Desbiolles et al. 2018), a recent study detected cytotoxic effects on the gills and hemocytes of the mussel Perna perna exposed to environmental concentrations of up to 300.0 ng/L of losartan (Cortez et al. 2018).
Another issue to be considered in the present study is that the concentrations of PPCPs vary seasonally as result of the land occupation (low or high tourist seasons) and climate effects (e.g. rainfall, wind and temperature), which will affect the risk assessment results (Zhang et al. 2017; Wang et al. 2019; Molnar et al. 2020). The present study was however a preliminary assessment, it means that diffuse load samples were collected in a single day during the dry season and low tourist season. Therefore, in order to clearly understand the risk of PPCPs to the aquatic environment, further studies (including water collection during the rainy season) are needed. A recent study conducted in the urban drainage channels of Guarujá city, Brazil, during the rainy season and high tourist season, also found moderate to high ecological risks from caffeine, acetaminophen and losartan. However, the MEC of these three PPPCs were much higher than those recorded in the hereby study (Roveri et al. 2020a).
Conclusion
This study reported, for the first time, the occurrence of 21 PPCPs in the urban drainage channels of Santos city, Brazil, during the dry season and low tourist season in late October 2020. The results showed that although the concentrations of twelve detected PPCPs were below the LOQ, the other nine (e.g. carbamazepine, caffeine, cocaine, benzoylecgonine, acetaminophen, diclofenac, orphenadrine, atenolol and losartan) were quantified in all urban channels of Santos, indicating that those pollutants were ubiquitous in the study area. This preliminary ecological risk assessment revealed that caffeine, acetaminophen and losartan were of moderate risk, and carbamazepine of low risk to sensitive aquatic organisms in the maximum measured environmental concentrations. It is therefore of the utmost importance to the environmental quality of Santos Bay, and thus, also for all the Brazilian coastal areas, that stakeholders (e.g. civil society, public authorities and environmental agencies) took substantial efforts to: (i) educate the public about the use and correct disposal of pharmaceutical waste in the environment; (ii) review the laws on the proper disposal of pharmaceutical waste in the environment; (iii) include PPCPs and illicit drugs in Brazilian environmental legislation as priority pollutants; (iv) define more rigorous guidelines for oceanic sewage disposal along the Brazilian coastal zone, namely where there is insufficient wastewater treatment plants; (v) implement continuous environmental monitoring programmes of PPCPs and illicit drugs in Brazilian coastal zone; and finally (vi) to conduct further research about the occurrence and the potential ecological risk of PPCPs and illicit drugs along the Brazilian coastal zone.
Availability of data and materials
Not applicable.
References
Abdalla RR, Madruga CS, Ribeiro M, Pinsky I, Caetano R, Laranjeira R (2014) Prevalence of cocaine use in Brazil: data from the II Brazilian national alcohol and drugs survey (BNADS). Addict Behav 39(1):297–301. https://doi.org/10.1016/j.addbeh.2013.10.019
Aguirre-Martínez GV, Owuor MA, Garrido-Pérez C, Salamanca MJ, Del Valls TA, Martín-Díaz ML (2015) Are standard tests sensitive enough to evaluate effects of human pharmaceuticals in aquatic biota? Facing changes in research approaches when performing risk assessment of drugs. Chemosphere 120:75–85. https://doi.org/10.1016/j.chemosphere.2014.05.087
Ambrozevicius AP, Abessa DMS (2008) Acute toxicity of waters from the urban drainage channels of Santos (São Paulo, Brazil). Pan-Am J Aquat Sci 3(2):108–115
Anumol T, Snyder SA (2015) Rapid analysis of trace organic compounds in water by automated online solid-phase extraction coupled to liquid chromatography–tandem mass spectrometry. Talanta 132:77–86. https://doi.org/10.1016/j.talanta.2014.08.011
Arola K, Hatakka H, Mänttäri M, Kallioinen M (2017) Novel process concept alternatives for improved removal of micropollutants in wastewater treatment. Sep Purif Technol 186:333–341. https://doi.org/10.1016/j.seppur.2017.06.019
Ashfaq M, Li Y, Rehman MSU, Zubair M, Mustafa G, Nazar MF, Sun Q (2019) Occurrence, spatial variation and risk assessment of pharmaceuticals and personal care products in urban wastewater, canal surface water, and their sediments: a case study of Lahore, Pakistan. Sci Total Environ 688:653–663. https://doi.org/10.1016/j.scitotenv.2019.06.285
Baptistelli SC, Marcellino EB (2016) Seawater Monitoring under the Influence of Sabesp Sea Outfalls in Baixada Santista (South Coast) and North Coast—São Paulo State, Brazil. Revista DAE 64:47–56. https://doi.org/10.4322/dae.2016.012
Bayen S, Estrada ES, Juhel G, Kit LW, Kelly BC (2016) Pharmaceutically active compounds and endocrine disrupting chemicals in water, sediments and mollusks in mangrove ecosystems from Singapore. Mar Pollut Bull 109(2):716–722. https://doi.org/10.1016/j.marpolbul.2016.06.105
Blackburn S, Pelling M, Marques C, 2019. Megacities and the coast: global context and scope for transformation. Coasts Estuaries, 661–669. doi:https://doi.org/10.1016/b978-0-12-814003-1.00038-1
Blaise C, Gagné F, Eullaffroy P, Férard JF (2006) Ecotoxicity of selected pharmaceuticals of urban origin discharged to the Saint-Lawrence river (Québec, Canada): a review. Ecotoxicity of selected pharmaceuticals of urban origin discharged to the Saint-Lawrence river (Québec, Canada): a review. Braz J Aquat Sci Technol 10(2):29–51
Brain RA, Johnson DJ, Richards SM, Hanson ML, Sanderson H, Lam MW, Solomon KR (2004) Microcosm evaluation of the effects of an eight pharmaceutical mixture to the aquatic macrophytes Lemna gibba and Myriophyllum sibiricum. Aquat Toxicol 70(1):23–40. https://doi.org/10.1016/j.aquatox.2004.06.011
Campestrini I, Jardim WF (2017) Occurrence of cocaine and benzoylecgonine in drinking and source water in the São Paulo State region, Brazil. Sci Total Environ 576:374–380. https://doi.org/10.1016/j.scitotenv.2016.10.089
Capaldo A, Gay F, Laforgia V (2019) Changes in the gills of the European eel (Anguilla anguilla) after chronic exposure to environmental cocaine concentration. Ecotoxicol Environ Saf 169:112–119. https://doi.org/10.1016/j.ecoenv.2018.11.010
Cetesb - Companhia Estadual de Tecnologia e Saneamento ambiental, 2019. Relatório de qualidade das águas costeiras no estado de São Paulo 2019. Série Relatórios/Agência Ambiental do Estado de São Paulo. ISSN 0103-4103.
Chen H, Gu X, Zeng Q, Mao Z (2019) Acute and Chronic Toxicity of Carbamazepine on the Release of Chitobiase, Molting, and Reproduction in Daphnia similis. Int J Environ Res Public Health 16(2):209. https://doi.org/10.3390/ijerph16020209
Claessens M, Vanhaecke L, Wille K, Janssen CR (2013) Emerging contaminants in Belgian marine waters: single toxicant and mixture risks of pharmaceuticals. Mar Pollut Bull 71(1-2):41–50. https://doi.org/10.1016/j.marpolbul.2013.03.039
CMED - Câmara de Regulação do Mercado de Medicamentos, (2019). Anuário Estatístico do Mercado Farmacêutico. ANVISA, Brasília, Brazil. Disponível em http://portal.anvisa.gov.br/
Cortez FS, da Silva Souza L, Guimarães LL, Almeida JE, Pusceddu FH, Maranho LA, Pereira CDS (2018) Ecotoxicological effects of losartan on the brown mussel Perna perna and its occurrence in seawater from Santos Bay (Brazil). Sci Total Environ 637-638:1363–1371. https://doi.org/10.1016/j.scitotenv.2018.05.069
Dafouz R, Cáceres N, Rodríguez-Gil JL, Mastroianni N, López de Alda M, Barceló D, Valcárcel Y (2018) Does the presence of caffeine in the marine environment represent an environmental risk? A regional and global study. Sci Total Environ 615:632–642. https://doi.org/10.1016/j.scitotenv.2017.09.155
De Souza Abessa DM, De Figueredo R, Rodarte B, De Oliveira M, De Oliveira GA, Cardoso AJF (2012) Environmental effects of sewage oceanic disposal by submarine outfalls: a review. Mundo da Saude 36:643–661. https://doi.org/10.15343/0104-7809.2012364643661
Desbiolles F, Malleret L, Tiliacos C, Wong-Wah-Chung P, Laffont-Schwob I (2018) Occurrence and ecotoxicological assessment of pharmaceuticals: is there a risk for the Mediterranean aquatic environment? Sci Total Environ 639:1334–1348. https://doi.org/10.1016/j.scitotenv.2018.04.351
Di Poi C, Costil K, Bouchart V, Halm-Lemeille M-P (2017) Toxicity assessment of five emerging pollutants, alone and in binary or ternary mixtures, towards three aquatic organisms. Environ Sci Pollut Res 25(7):6122–6134. https://doi.org/10.1007/s11356-017-9306-9
ECB, 2003. Technical Guidance Document on Risk Assessment for existing substances, Part II, pp 108-110
ECHA, (2008). Guidance on information requirements and chemical safety assessment. Chapter R.10: Characterisation of dose [concentration]-response for environment, pp7-29
EMA - European Medicines Agency, Committee for Medicinal Products for Human use (CHMP). (2006). Guideline on the Environmental Risk Assessment of Medicinal Products for Human use. Doc. Ref.: EMEA/CHMP/SWP/4447/00 corr 1, London, UK.
FDA – U.S. Food and Drug Administration, (2012). Center for drug evaluation and research. Approach Package for: Aplication number 20-386/S-019 and 029. Environment Assesment/ Fonsi. p.8.
Fernández C, González-Doncel M, Pro J, Carbonell G, Tarazona JV (2010) Occurrence of pharmaceutically active compounds in surface waters of the Henares-jarama-tajo river system (Madrid, Spain) and a potential risk characterization. Sci Total Environ 408(3):543–551. https://doi.org/10.1016/j.scitotenv.2009.10.009
Ferrari B, Paxéus N, Giudice RL, Pollio A, Garric J (2003) Ecotoxicological impact of pharmaceuticals found in treated wastewaters: study of carbamazepine, clofibric acid, and diclofenac. Ecotoxicol Environ Saf 55(3):359–370. https://doi.org/10.1016/s0147-6513(02)00082-9
Ferraz AM, Choueri RB, Fiori EF, Nobre CR, César A, Pereira CDS (2012) Sediment quality assessment of Santos shoreline through toxicity assays and characterization of macrobenthic community structure. O Mundo da Saúde 36(4):625–634. https://doi.org/10.15343/0104-7809.2012364625634
Fontes MK, de Campos BG, Cortez FS, Pusceddu FH, Moreno BB, Maranho LA, Pereira CDS (2019) Seasonal monitoring of cocaine and benzoylecgonine in a subtropical coastal zone (Santos Bay, Brazil). Mar Pollut Bull 149:110545. https://doi.org/10.1016/j.marpolbul.2019.110545
Fontes MK, de Campos BG, Cortez FS, Pusceddu FH, Nobre CR, Moreno BB, Pereira CDS (2020) Mussels get higher: a study on the occurrence of cocaine and benzoylecgonine in seawater, sediment and mussels from a subtropical ecosystem (Santos Bay, Brazil). Sci Total Environ 757:143808. https://doi.org/10.1016/j.scitotenv.2020.143808
Galus M, Jeyaranjaan J, Smith E, Li H, Metcalfe C, Wilson JY (2013) Chronic effects of exposure to a pharmaceutical mixture and municipal wastewater in zebrafish. Aquat Toxicol 132-133:212–222. https://doi.org/10.1016/j.aquatox.2012.12.016
Gandra CV, Guimarães LL, Santos AR, Cortez FS, Pusceddu FH, 2020. Physical-chemical, microbiological and ecotoxicological characterization of the pluvial waters of the urban Santos drainage system (São Paulo, Brazil). Res Soc Dev, 9 (12). https://doi.org/10.33448/rsd-v9i12.10739
Godoy AA, Kummrow F (2017) What do we know about the ecotoxicology of pharmaceutical and personal care product mixtures? A critical review. Crit Rev Environ Sci Technol 47(16):1453–1496. https://doi.org/10.1080/10643389.2017.1370991
Godoy AA, Kummrow F, Pamplin PAZ (2015) Ecotoxicological evaluation of propranolol hydrochloride and losartan potassium to Lemna minor L. (1753) individually and in binary mixtures. Ecotoxicology 24(5):1112–1123. https://doi.org/10.1007/s10646-015-1455-3
González-Mariño I, Baz-Lomba JA, Alygizakis NA, Andrés-Costa MJ, Bade R, Barron LP, Bodík I (2019) Spatio-temporal assessment of illicit drug use at large scale: evidence from 7 years of international wastewater monitoring. Addiction. 115:109–120. https://doi.org/10.1111/add.14767
Griffero L, Alcántara-Durán J, Alonso C, Rodríguez-Gallego L, Moreno-González D, García-Reyes JF, Pérez-Parada A (2019) Basin-scale monitoring and risk assessment of emerging contaminants in South American Atlantic coastal lagoons. Sci Total Environ 697:134058. https://doi.org/10.1016/j.scitotenv.2019.134058
Hernando MD, Mezcua M, Fernandez-Alba AR, Barcelo D (2006) Environmental risk assessment of pharmaceutical residues in wastewater effluents, surface waters and sediments. Talanta 69(2):334–342. https://doi.org/10.1016/j.talanta.2005.09.037
Herrmann M, Olsson O, Fiehn R, Herrel M, Kümmerer K (2015) The significance of different health institutions and their respective contributions of active pharmaceutical ingredients to wastewater. Environ Int 85:61–76. https://doi.org/10.1016/j.envint.2015.07.020
Hossain A, Nakamichi S, Habibullah-Al-Mamun M, Tani K, Masunaga S, Matsuda H (2018) Occurrence and ecological risk of pharmaceuticals in river surface water of Bangladesh. Environ Res 165:258–266. https://doi.org/10.1016/j.envres.2018.04.030
Ibge – Instituto brasileiro de Geografia e Estatística, (2019). Estimativa da população brasileira. Rio de Janeiro.
Kim J-W, Ishibashi H, Yamauchi R, Ichikawa N, Takao Y, Hirano M, Arizono K (2009) Acute toxicity of pharmaceutical and personal care products on freshwater crustacean (Thamnocephalus platyurus) and fish (Oryzias latipes). J Toxicol Sci 34(2):227–232. https://doi.org/10.2131/jts.34.227
Klosterhaus SL, Grace R, Hamilton MC, Yee D (2013) Method validation and reconnaissance of pharmaceuticals, personal care products, and alkylphenols in surface waters, sediments, and mussels in an urban estuary. Environ Int 54:92–99. https://doi.org/10.1016/j.envint.2013.01.009
Lacorte S, Luis S, Gómez-Canela C, Sala-Comorera T, Courtier A, Roig B, Calas-Blanchard C (2017) Pharmaceuticals released from senior residences: occurrence and risk evaluation. Environ Sci Pollut Res 25(7):6095–6106. https://doi.org/10.1007/s11356-017-9755-1
Lamparelli CC, Pogreba-Brown K, Verhougstraete M, Sato MIZ, de Castro Bruni A, Wade TJ, Eisenberg JNS (2015) Are fecal indicator bacteria appropriate measures of recreational water risks in the tropics? A cohort study of beach goers in Brazil. Water Res 87:59–68. https://doi.org/10.1016/j.watres.2015.09.001
Lara-Martín PA, González-Mazo E, Petrovic M, Barceló D, Brownawell BJ (2014) Occurrence, distribution and partitioning of nonionic surfactants and pharmaceuticals in the urbanized Long Island Sound Estuary (NY). Mar Pollut Bull 85(2):710–719. https://doi.org/10.1016/j.marpolbul.2014.01.022
Li K, Du P, Xu Z, Gao T, Li X (2016) Occurrence of illicit drugs in surface waters in China. Environ Pollut 213:395–402. https://doi.org/10.1016/j.envpol.2016.02.036
Linjakumpu T, Hartikainen S, Klaukka T, Veijola J, Kivelä S-L, Isoaho R (2002) Use of medications and polypharmacy are increasing among the elderly. J Clin Epidemiol 55(8):809–817. https://doi.org/10.1016/s0895-4356(02)00411-0
Locatelli MAF, Sodré FF, Jardim WF (2010) Determination of antibiotics in Brazilian surface waters using liquid chromatography–electrospray tandem mass spectrometry. Arch Environ Contam Toxicol 60(3):385–393. https://doi.org/10.1007/s00244-010-9550-1
Maasz G, Mayer M, Zrinyi Z, Molnar E, Kuzma M, Fodor I, Takács P (2019) Spatiotemporal variations of pharmacologically active compounds in surface waters of a summer holiday destination. Sci Total Environ 677:545–555. https://doi.org/10.1016/j.scitotenv.2019.04.286
Mendoza A, Aceña J, Pérez S, López de Alda M, Barceló D, Gil A, Valcárcel Y (2015) Pharmaceuticals and iodinated contrast media in a hospital wastewater: a case study to analyse their presence and characterise their environmental risk and hazard. Environ Res 140:225–241
Mijangos L, Ziarrusta H, Ros O, Kortazar L, Fernández LA, Olivares M, Etxebarria N (2018) Occurrence of emerging pollutants in estuaries of the Basque Country: analysis of sources and distribution, and assessment of the environmental risk. Water Res 147:152–163. https://doi.org/10.1016/j.watres.2018.09.033
Minguez L, Pedelucq J, Farcy E, Ballandonne C, Budzinski H, Halm-Lemeille M-P (2014) Toxicities of 48 pharmaceuticals and their freshwater and marine environmental assessment in northwestern France. Environ Sci Pollut Res 23(6):4992–5001. https://doi.org/10.1007/s11356-014-3662-5
Molnar E, Maasz G, Pirger Z (2020) Environmental risk assessment of pharmaceuticals at a seasonal holiday destination in the largest freshwater shallow lake in Central Europe. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-020-09747-4
Moore MT, Greenway SL, Farris JL, Guerra B (2008) Assessing caffeine as an emerging environmental concern using conventional approaches. Arch Environ Contam Toxicol 54(1):31–35. https://doi.org/10.1007/s00244-007-9059-4
Moreira FA, Rampazo NAM, Castelanno MS (2017) Impacts of rainfall and vulnerabilities in the metropolitan region of Baixada Santista. Int J Saf Secur Eng 7(2):169–179
Ojemaye CY, Petrik LP (2018) Pharmaceuticals in the marine environment: a review. Environ Rev 27:151–165. https://doi.org/10.1139/er-2018-0054
Ortiz JP, Braulio A, Yanes JP (2016) Wastewater Marine Disposal through Outfalls on the coast of São Paulo State Brazil: an overview. Revista DAE 64:29–46. https://doi.org/10.4322/dae.2016.015
Parolini M, Magni S, Castiglioni S, Zuccato E, Binelli A (2015) Realistic mixture of illicit drugs impaired the oxidative status of the zebra mussel (Dreissena polymorpha). Chemosphere 128:96–102. https://doi.org/10.1016/j.chemosphere.2014.12.092
Parolini M, De Felice B, Ferrario C, Salgueiro-González N, Castiglioni S, Finizio A, Tremolada P (2018) Benzoylecgonine exposure induced oxidative stress and altered swimming behavior and reproduction in Daphnia magna. Environ Pollut 232:236–244. https://doi.org/10.1016/j.envpol.2017.09.038
Peña-Guzmán C, Ulloa-Sánchez S, Mora K, Helena-Bustos R, Lopez-Barrera E, Alvarez J, Rodriguez-Pinzón M (2019) Emerging pollutants in the urban water cycle in Latin America: a review of the current literature. J Environ Manag 237:408–423. https://doi.org/10.1016/j.jenvman.2019.02.100
Pereira CDS, Maranho LA, Cortez FS, Pusceddu FH, Santos AR, Ribeiro DA, Cesar A, Guimarães LL (2016) Occurrence of pharmaceuticals and cocaine in a Brazilian coastal zone. Sci Total Environ 548-549:148–154. https://doi.org/10.1016/j.scitotenv.2016.01.051
Potera C (2012) Caffeine in wastewater is a tracer for human fecal contamination. Environ Health Perspect 120(3):A108–A109. https://doi.org/10.1289/ehp.120-a108a
Quadra GR, Oliveira de Souza H, dos Santos Costa R, dos Santos Fernandez MA (2017) Do pharmaceuticals reach and affect the aquatic ecosystems in Brazil? A critical review of current studies in a developing country. Environ Sci Pollut Res 24(2):1200–1218. https://doi.org/10.1007/s11356-016-7789-4
Radwan EK, Ibrahim MBM, Adel A, Farouk M (2019) The occurrence and risk assessment of phenolic endocrine-disrupting chemicals in Egypt’s drinking and source water. Environ Sci Pollut Res 27:1776–1788. https://doi.org/10.1007/s11356-019-06887-0
Rivera-Jaimes JA, Postigo C, Melgoza-Alemán RM, Aceña J, Barceló D, López de Alda M (2018) Study of pharmaceuticals in surface and wastewater from Cuernavaca, Morelos, Mexico: occurrence and environmental risk assessment. Sci Total Environ 613-614:1263–1274. https://doi.org/10.1016/j.scitotenv.2017.09.134
Robles-Molina J, Gilbert-López B, García-Reyes JF, Molina-Díaz A (2014) Monitoring of selected priority and emerging contaminants in the Guadalquivir River and other related surface waters in the province of Jaén, South East Spain. Sci Total Environ 479-480:247–257. https://doi.org/10.1016/j.scitotenv.2014.01.121
Rocha S, Pinto RMF, Floriano AP, Teixeira LH, Bassili B, Martinez A, Caseiro MM (2011) Environmental analyses of the parasitic profile found in the sandy soil from the Santos municipality beaches, SP, Brazil. Rev Inst Med Trop Sao Paulo 53(5):277–281
Roveri V, Guimarães LL, Toma C, A. T. (2020a) Occurrence and ecological risk assessment of pharmaceuticals and cocaine in a beach area of Guarujá, São Paulo State, Brazil, under the influence of urban surface runoff. Environ Sci Pollut Res 27:45063–45075. https://doi.org/10.1007/s11356-020-10316-y
Roveri V, Guimarães LL, Toma W, Correia AT (2020b) Occurrence and risk assessment of pharmaceuticals and cocaine around the coastal submarine sewage outfall in Guarujá, São Paulo State, Brazil. Environ Sci Pollut Res 28:11384–11400. https://doi.org/10.1007/s11356-020-11320-y
Sauvé S, Aboulfadl K, Dorner S, Payment P, Deschamps G, Prévost M (2012) Fecal coliforms, caffeine and carbamazepine in stormwater collection systems in a large urban area. Chemosphere 86(2):118–123. https://doi.org/10.1016/j.chemosphere.2011.09.033
Schröder P, Helmreich B, Škrbić B, Carballa M, Papa M, Pastore C, Mascolo G (2016) Status of hormones and painkillers in wastewater effluents across several European states—considerations for the EU watch list concerning estradiols and diclofenac. Environ Sci Pollut Res 23(13):12835–12866. https://doi.org/10.1007/s11356-016-6503-x
Shihomatzu HM, (2015). Desenvolvimento e Validação de Metodologia SPE-LC-MS/MS para determinação de Fármacos e Droga de Abuso nas Águas da Represa Guarapiranga, São Paulo/SP, Brasil. IPEN/USP. https://doi.org/10.11606/T.85.2015.tde-28042015-095207
SMA/CPLA – Secretaria de Meio Ambiente do Estado de São Paulo/Coordenação de Planejamento Ambiental, (2018). Zona Costeira Paulista: Relatório de Qualidade Ambiental. Org. Organizadores Nádia Gilma Beserra de Lima e Tatiana Camolez Morales Ferreira. SMA/CPLA, São Paulo. Brasil.
SMA/CPLEA – Secretaria do Meio Ambiente do Estado de São Paulo/Coordenadoria de Planejamento e Educação Ambiental, (2016). Zoneamento Ecológico - Econômico – Baixada Santista, São Paulo, Brasil, 55 p.
Spongberg AL, Witter JD, Acuña J, Vargas J, Murillo M, Umaña G, Perez G (2011) Reconnaissance of selected PPCP compounds in Costa Rican surface waters. Water Res 45(20):6709–6717. https://doi.org/10.1016/j.watres.2011.10.004
Starling MCVM, Amorim CC, Leão MMD (2018) Occurrence, control and fate of contaminants of emerging concern in environmental compartments in Brazil. J Hazard Mater 372:17–36. https://doi.org/10.1016/j.jhazmat.2018.04.043
Sui Q, Huang J, Deng S, Yu G, Fan Q (2010) Occurrence and removal of pharmaceuticals, caffeine and DEET in wastewater treatment plants of Beijing, China. Water Res 44:417–426. https://doi.org/10.1016/j.watres.2009.07.010
Tummala MK, Taub DT, Ershler WB (2010) Clinical immunology: immune senescence and the acquired immune deficiency of aging. In: Fillit HM, Rockwood K, Woodhouse K (eds) Brocklehurst’s textbook of geriatric medicine and gerontology, 7th edn. Elsevier Saunders, Philadelphia
UNODC. United Nations Office on Drugs and Crime. (2020). World Drug Report 2020. Booklet 1. Executive Summary. Available at: https://wdr.unodc.org/wdr2020/en/exsum.html
USEPA - United States Environmental Protection Agency, (2007) Method 1694: Pharmaceuticals and Personal Care Products in Water, Soil Sediment, and Biosolids by HPLC/MS/MS. Washington.
USEPA - United States Environmental Protection Agency, (2017). Ecological Structure-Activity Relationship Model (ECOSAR) Class Program. MS-Windows Version 2.0. https://www.epa.gov/tsca732 screening-tools/ecological-structure-activity-relationships-ecosarcpredictive-model.
USEPA - United States Environmental Protection Agency, (2019). ECOTOX User Guide: Ecotoxicology Database System, Version 4.0. http://www.epa.gov/ecotox/.
Valcárcel Y, González Alonso S, Rodríguez-Gil JL, Gil A, Catalá M (2011) Detection of pharmaceutically active compounds in the rivers and tap water of the Madrid Region (Spain) and potential ecotoxicological risk. Chemosphere 84(10):1336–1348. https://doi.org/10.1016/j.chemosphere.2011.05.014
Valdez-Carrillo M, Abrell L, Ramírez-Hernández J, Reyes-López JA, Carreón-Diazconti C (2020) Pharmaceuticals as emerging contaminants in the aquatic environment of Latin America: a review. Environ Sci Pollut Res 27:44863–44891. https://doi.org/10.1007/s11356-020-10842-9
Van Nuijs ALN, Pecceu B, Theunis L, Dubois N, Charlier C, Jorens PG, Covaci A (2009) Cocaine and metabolites in waste and surface water across Belgium. Environ Pollut 157(1):123–129. https://doi.org/10.1016/j.envpol.2008.07.020
Vazquez-Roig P, Andreu V, Blasco C, Picó Y (2012) Risk assessment on the presence of pharmaceuticals in sediments, soils and waters of the Pego–Oliva Marshlands (Valencia, eastern Spain). Sci Total Environ 440:24–32. https://doi.org/10.1016/j.scitotenv.2012.08.036
Vernouillet G, Eullaffroy P, Lajeunesse A, Blaise C, Gagné F, Juneau P (2010) Toxic effects and bioaccumulation of carbamazepine evaluated by biomarkers measured in organisms of different trophic levels. Chemosphere 80(9):1062–1068. https://doi.org/10.1016/j.chemosphere.2010.05.010
Wang Y, Liu Y, Lu S, Liu X, Meng Y, Zhang G, Guo X (2019) Occurrence and ecological risk of pharmaceutical and personal care products in surface water of the Dongting Lake, China-during rainstorm period. Environ Sci Pollut Res 26:28796–28807. https://doi.org/10.1007/s11356-019-06047-4
Wilkinson JL, Hooda PS, Swinden J, Barker J, Barton S (2017) Spatial distribution of organic contaminants in three rivers of Southern England bound to suspended particulate material and dissolved in water. Sci Total Environ 593-594:487–497. https://doi.org/10.1016/j.scitotenv.2017.03.167
Yang Y-Y, Zhao J-L, Liu Y-S, Liu W-R, Zhang Q-Q, Yao L, Ying G-G (2018) Pharmaceuticals and personal care products (PPCPs) and artificial sweeteners (ASs) in surface and ground waters and their application as indication of wastewater contamination. Sci Total Environ 616-617:816–823. https://doi.org/10.1016/j.scitotenv.2017.10.241
Yu-Chen Lin A, Panchangam SC, Chen H-Y (2010) Implications of human pharmaceutical occurrence in the Sindian river of Taiwan: a strategic study of risk assessment. J Environ Monit 12(1):261–270. https://doi.org/10.1039/b903880a
Zhang Y, Zhang T, Guo C, Lv J, Hua Z, Hou S, Xu J (2017) Drugs of abuse and their metabolites in the urban rivers of Beijing, China: occurrence, distribution, and potential environmental risk. Sci Total Environ 579:305–313. https://doi.org/10.1016/j.scitotenv.2016.11.101
Zhou SB, Paolo CD, Wu XD, Shao Y, Seiler TB, Hollert H (2019) Optimization of screening-level risk assessment and priority selection of emerging pollutants – The case of pharmaceuticals in European surface waters. Environ Int 128:1–10
Acknowledgements
Authors want to acknowledge Daniel Temponi Lebre from the Centro de Espectrometria de Massa Aplicada—Instituto de Pesquisas Energéticas e Nucleares (CEMSA, IPEN, São Paulo, Brazil) for technical support regarding the LC–MS/MS analyses. Thanks also to Professor Ítalo Braga de Castro, from Instituto do Mar, Universidade Federal de São Paulo (IMAR-UNIFESP, São Paulo, Brazil) for technical support with the Manifold.
Funding
Correia AT was supported by national funds through FCT—Foundation for Science and Technology within the scope of UIDB/04423/2020 and UIDP/04423/2020.
Author information
Authors and Affiliations
Contributions
VR: Conceptualization, Data curation, Formal analysis, Methodology, Writing—original draught. LLG: Supervision, Writing—review and editing. WT: review and editing. ATC: Funding acquisition, Supervision, Writing—original draught, Writing—review and editing.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Ester Heath
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 827 kb)
Rights and permissions
About this article
Cite this article
Roveri, V., Guimarães, L.L., Toma, W. et al. Occurrence and ecological risk assessment of pharmaceuticals and cocaine in the urban drainage channels of Santos beaches (São Paulo, Brazil): a neglected, but sensitive issue. Environ Sci Pollut Res 28, 65595–65609 (2021). https://doi.org/10.1007/s11356-021-15249-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-15249-8