Abstract
The aquatic environment represents the final repository for many human-generated pollutants associated with anthropogenic activities. The quality of natural freshwater systems is easily disrupted by the introduction of pollutants from urban, industrial and agricultural processes. To assess the extent of chemical perturbation and associated environmental degradation, physico-chemical parameters have been monitored in conjunction with biota in numerous biological monitoring protocols. Most studies incorporating organisms into such approaches have focussed on fish and macroinvertebrates. More recently, interest in the ecology of parasites in relation to environmental monitoring has indicated that these organisms are sensitive towards the quality of the macroenvironment. Variable responses towards exposure to pollution have been identified at the population and component community level of a number of parasites. Furthermore, such responses have been found to differ with the type of pollutant and the lifestyle of the parasite. Generally, endoparasite infection levels have been shown to become elevated in relation to poorer water quality conditions, while ectoparasites are more sensitive, and exposure to contaminated environments resulted in a decline in ectoparasite infections. Furthermore, endoparasites have been found to be suitable accumulation indicators for monitoring levels of several trace elements and metals in the environment. The ability of these organisms to accumulate metals has further been observed to be of benefit to the host, resulting in decreased somatic metal levels in infected hosts. These trends have similarly been found for host–parasite models in African freshwater environments, but such analyses are comparatively sparse compared to other countries. Recently, studies on diplozoids from two freshwater systems have indicated that exposure to poorer water quality resulted in decreased infections. In the Vaal River, the poor water quality resulted in the extinction of the parasite from a site below the Vaal River Barrage. Laboratory exposures have further indicated that oncomiracidia of Paradiplozoon ichthyoxanthon are sensitive to exposure to dissolved aluminium. Overall, parasites from African freshwater and marine ecosystems have merit as effect and accumulation indicators; however, more research is required to detail the effects of exposure on sensitive biological processes within these organisms.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Assessment of environmental health with biota has become increasingly more attractive, and the number of identified indicator species has similarly increased. One of the most well-known examples of an organism’s usefulness as an indicator of the quality of an environment is that of canaries in coal mines. Historically, canaries accompanied miners into coal mines where the sensitivity of the birds to trace amounts of noxious gases, methane and carbon monoxide (Smith and Baker 2003; Holt and Miller 2010) was used as an early warning of toxic conditions to miners. As a result of their acute sensitivity towards the gases, exposure to low concentrations resulted in death of the birds which served as an unequivocal signal for miners to evacuate mining shafts (Smith and Baker 2003). Within aquatic ecosystems, a similar principle exists for determining the suitability of aquatic ecosystems in meeting a number of different services from environmental functionality to human uses, and with that, a number of organisms have been suggested as suitable indicators in this regard. Some of these include fish (e.g. Zhou et al. 2008; Squadrone et al. 2013), macroinvertebrates (e.g. Goodyear and McNeill 1999; Wah Chu and Chow 2002; Bird et al. 2011; Martins et al. 2011; Leonard and Wood 2013), bivalves (e.g. Kádár et al. 2006; Schintu et al. 2008; Minguez et al. 2011; Le et al. 2014) and more recently parasites (Sures 2001; Retief et al. 2006, 2009; Bayoumy et al. 2008; Sures et al. 2017).
Over the past 25 years, there has been growing interest into the interactions between parasites and the macroenvironment and with that the potential application of parasites as sentinel organisms for monitoring habitat quality and ecosystem health in relation to anthropogenic impacts (e.g. Khan and Thulin 1991; Šebelová et al. 2002; Sures 2004; Pečínková et al. 2005; Retief et al. 2006, 2009; Vidal-Martínez et al. 2010). Williams and MacKenzie (2003) suggested that the suitability of parasites as indicators stems from the variety of life stages in the life cycle of parasites and the variety of hosts involved in life cycles, some of which have been shown to be suitable indicators. Interest in the inter-relationship between parasites and the environment has appeared in a number of publications since the early 1980s (Sures 2001; Vidal-Martínez et al. 2010) where Khan and Thulin (1991) reviewed the interactions and effects of different pollutants on a number of different parasites. Later, Lafferty (1997) provided a detailed review and summary of literature pertaining to faunal differences of parasites towards pollution and the relationship between parasites and pollution. There are a variety of ways anthropogenic impacts result in environmental alterations which directly or indirectly affect parasites (Lafferty 1997; Galli et al. 2001). Responses of parasites towards environmental alterations have been analysed at species, population, behavioural and biochemical levels, and from this, such findings have deepened the suggestion of their suitability as bioindicators for monitoring the state of the macroenvironment (Sures 2001). Based on the responses displayed by parasites, they can be divided into two main indicator groups, namely effect indicators and accumulation indicators (Sures 2001, 2004). These aspects have been summarised in a number of reviews which deal directly with the usefulness of parasites as effect and bioaccumulation indicators (Lafferty 1997; MacKenzie 1999; Sures 2001, 2003, 2004 2005, 2008; Williams and MacKenzie 2003; Marcogliese 2004, 2005; Blanar et al. 2009; Vidal-Martínez et al. 2010; MacLeod and Poulin 2012; Le et al. 2014; Sures et al. 2017). Collectively, the assessment of environmental pollution using parasites has been termed “environmental parasitology” (Sures et al. 2017). In the most recent review on the subject by Sures et al. (2017), a summary of work from the past 20 years dealing with parasites as pollution indicators was presented and three main facets in the field were identified: “parasites as bioindicators”, “parasites as effect indicators” and “parasites interacting with established bioindicators”, and provides a holistic view of the advances made in the field at multiple levels of biological organisation relating to parasites (Sures et al. 2017).
Infrapopulation differences in parasite communities have been linked to changes in host population size and composition (Poulin 1995; Crafford and Avenant-Oldewage 2009; Watson et al. 2012; Saliu et al. 2014; Otachi et al. 2015), seasonal variances (Hakalahti and Valtonen 2003; Mbokane et al. 2015) and water quality (Khan and Thulin 1991; Broeg et al. 1999; Brito et al. 2014). Most studies conducted are related to effect indication by parasites, where changes in the infection biology and component community of a number of parasites in relation to differences in the chemical and physical constituents of aquatic environments have been assessed in relation to different types of pollution, whereas most bioaccumulation studies relating to parasites have focussed on the uptake of metals by these organisms with few investigations into organic pollutant accumulation.
The interest into metals could be related to their persistence in aquatic environments, their ability to accumulate within organisms and the array of deleterious effects associated with their presence in ecosystems (e.g. Avenant-Oldewage and Marx 2000; Le et al. 2014). Metals are naturally occurring and ubiquitous components of aquatic ecosystems, usually present in low concentrations (Förstner and Prosi 1979; Biney et al. 1994). The ecotoxicology of metals in aquatic ecosystems is complex (Wang 1987) and toxicity can be affected by changes in physical water quality variables, such as pH, alkalinity and hardness, and chemical variables such as carbon dioxide levels, total dissolved salts and chelating agents (Förstner and Prosi 1979; Biney et al. 1994). Furthermore, the variability in toxicity of metals in natural systems is linked to changes in the speciation of metals in the environment as a result of interactions with physico-chemical variables, some of which have been identified above (Hodson 1988). Furthermore, synergistic and antagonistic interactions between the metals present in aquatic systems can further affect their toxicity towards biota (Siegel et al. 1991; Tao et al. 1999; Zargar et al. 2012a, b).
In Africa as a whole and specifically South Africa, a large volume of studies have been produced which detail the accumulation of metals by aquatic organisms and the effects on the community structures; however, comparatively fewer studies have been conducted to investigate the usefulness of parasites as indicators and the effects of pollution exposure on population, organismal and physiological processes. This has, therefore, left a large gap in knowledge for the continent and country compared to the world. The aims of this review are to highlight the aspects of pollution identification using parasites in general and provide detail on the effects of pollution exposure at the component community level and the organismal level from studies across the globe and then comparing to the subject in context to Africa and South African examples.
Physico-chemical water quality variables
In order to properly ascertain the effectiveness of parasites as indicators, adequate information of natural fluctuations within component communities is required. The influence of seasonal fluctuations in abiotic factors (e.g. temperature) influences parasite populations and plays an important role in governing helminth infections in fishes (Khan and Rasheed 1993). Because of the complexity of biological systems and the host–parasite relationship, it is almost impossible to accurately determine or predict what consequence alterations in abiotic factors may have on parasite infections (Marcogliese 2016). The relationship between abiotic environmental factors and the occurrence of parasites in aquatic ecosystems has been addressed in a number of reviews (see Harvell et al. 1999, 2002; Hayes et al. 2001; Marcogliese 2001, 2008, 2016; Marcos-López et al. 2010; Lõhmus and Björklund 2015) under the umbrella term “climate change”. Overall, the interactions between parasites and the conditions of the macroenvironment are highly complex and relate to the life cycle characteristics of the parasite (see Anderson and Sukhdeo 2010). The interplay between biotic and abiotic factors influencing parasites has been demonstrated by Anderson and Sukhdeo (2010) who found that influences by abiotic factors operated secondarily to biotic influences on parasite populations. This finding was related to the effect that abiotic factors have on intermediate host occurrence, which had a greater effect on parasitic infections and therefore indirectly affected the dynamics of parasite populations (Zander and Reimer 2002; Marcogliese 2005; Anderson and Sukhdeo 2010). For the purposes of this review, the focus will be directed towards the effects of abiotic factors on parasites.
Of the abiotic factors analysed in studies and the relationship with helminth infections, temperature has been suggested to be the principal factor mediating infrapopulation differences of parasites either directly by affecting the parasite or indirectly affecting host abundance and distribution (Marcogliese 2001, 2016). Examples of direct and indirect effects of temperature on parasite life cycles have been presented in numerous reviews and provide details on effects towards development of life stages and host susceptibility towards infection (Harvell et al. 1999; Hayes et al. 2001; Marcogliese 2001, 2008, 2016; Harvell et al. 2002; Marcos-López et al. 2010; Lõhmus and Björklund 2015). Such temperature dependence and influence has been related to parasite incubation period which is temperature dependent (Paperna 1996). Seasonal fluctuations in temperature of aquatic ecosystems have been suggested to weaken the immune response of the host, making the host more susceptible towards infection (Altizer et al. 2006; Lamková et al. 2007). This trend has specifically been related to increases in infections by endoparasites such as cestodes and acanthocephalans. For example, infections by the Asian tapeworm (Granath and Esch 1983; Retief et al. 2007), Schyzocotyle acheilognathi [(formally Bothriocephalus acheilognathi) (Brabec et al. 2015)], have been shown to increase as water temperature decreases. Similarly, gyrodactylid infections have been found to peak when water temperatures are cooler (Gelnar 1991; Blažek et al. 2008), whereas the opposite has been observed in seasonal fluctuations of dactylogyrids, with infections peaking when temperatures increase (Šimková et al. 2001). In the acanthocephalan, Pomphorhynchus laevis, Nachev and Sures (2016) indicated that the mean weight of parasite infrapopulations differed seasonally, with significantly higher in summer compared to autumn and spring. Furthermore, they found that seasonal infrapopulation differences were related to different stages of maturation of adult worms, with young preadults being most prevalent in autumn and gravid adults in summer (Nachev and Sures 2016). Such seasonal change would not relate to alterations in the immune system of the host but rather the abundance of intermediate hosts in this case during the summer period (Rohde 1984). Thermophilicity has similarly been observed for diplozoid monogeneans (Höglund and Thulin 1989; Koskivaara et al. 1991; Koskivaara and Valtonen 1991) and has been suggested to result from the influence on reproductive success (Höglund and Thulin 1989). However, in a study on Paradiplozoon homoion, MacDonald and Jones (1978) found that egg laying in this parasite was not affected by temperature but instead correlated with photoperiod. Gravidity in Paradiplozoon ichthyoxanthon was further observed to increase in colder temperatures compared to warmer ones in the Vaal Dam (Gilbert and Avenant-Oldewage 2016a).
With regard to dactylogyrids, Koskivaara et al. (1992) suggested that temperature influenced this group in two ways: firstly, by directly affecting reproduction and development; and secondly, indirectly through changes in the immunological state of the host. Temperature effects on host immunology in relation to monogenean infections were analysed by Lamková et al. (2007) in the chub (Leuciscus cephalus), and they indicate that given the diversity of monogeneans, responses differ between the different groups. However, an adequate sample size is necessary to properly elucidate seasonal changes in parasite populations as le Roux et al. (2011) demonstrated for Cichlidogyrus philander; sample size greatly influenced infection statistics. Similarly, Crafford et al. (2014) and Mbokane et al. (2015) found that the prevalence of a number of Dactylogyrus sp. from fishes inhabiting the Vaal River and Limpopo River systems, respectively, fluctuated with season, but in all cases, the prevalence increased as the temperature increased with the season. In their studies, the authors found that the highest prevalence was present in either summer or spring seasons. Tsotetsi et al. (2004) and Austin and Avenant-Oldewage (2009) indicated that the variances in seasonality of Lamproglena clariae and Lamproglena hoi from the Vaal River and the Inkomati River and Pongola River systems, respectively, were similar to trends outlined for monogeneans, with prevalence of these parasites increasing with water temperature. For the monogenean, Afrodiplozoon polycotyleus, Chapman et al. (2000) and Raymond et al. (2006) indicated that temperature fluctuations, which related to rainfall and host habitat size, were factors influencing prevalence of A. polycotyleus with higher prevalence when temperatures were high.
However, changes in parasitic infections and population structures are not solely due to changes in temperature (Marcogliese 2008), and a number of factors have been suggested to operate in synergy with temperature to result in alterations. Such additional factors include changes in acidification, water level, eutrophication and stratification (Marcogliese 2001), together with anthropogenic intrusion (Marcogliese 2008) which will ultimately influence parasites (Marcogliese 2016). Salinity has been identified as another principal abiotic factor which affects parasite population structures. Several studies in both aquatic and marine ecosystems have indicated that changes in salinity affect infection biology of parasites (Harvell et al. 1999, 2002; Hayes et al. 2001; Marcogliese 2008, 2016). Generally, increases in salinity have been shown to negatively correlate with the population density and abundance of parasites (Soleng and Bakke 1997; Schmidt et al. 2003). Zander and Reimer (2002) found that parasite infections in the Baltic Sea were variable and correlated with the salinity gradient in this region. Salinity in the Baltic Sea decreases from west to east, and parasite distribution has been found to be limited along the salinity gradient (Zander and Reimer 2002). Although freshwater, marine and brackish water parasite taxa are located in this region, the distribution is spatially variable with few freshwater species being found in the western parts where salinity levels are high (Zander and Reimer 2002). Later, Jakob et al. (2008) observed that salinity dependence of 27 parasite taxa, infecting the European eel (Anguilla anguilla), reflected the habitat preference of the host for freshwater, marine and brackish water habitats with parasites clustering according to the habitat type and differences not being related to a salinity gradient between habitats. In their study, brackish sites investigated were similarly located in the Baltic Sea where both freshwater and marine parasites were found infecting eels and differences were not related to a salinity gradient at the site (Jakob et al. 2008). Sensitivity of freshwater parasites towards variances in salinity has been experimentally demonstrated for Gyrodactylus salaris infecting Atlantic salmon (Salmo salar) (Soleng and Bakke 1997). Soleng and Bakke (1997) observed that elevated salinity negatively correlated with survival of G. salaris, and therefore, the occurrence of the parasite was affected as salinity increased. In the German Bight, Schmidt et al. (2003) found that occurrence of copepod parasite Lepeophtheirus pectoralis corresponded to seasonal changes in salinity rather than temperature. Similarly, Blanar et al. (2011) indicated that the effect of salinity on occurrence of parasites on mummichog (Fundulus heteroclitus) in the Miramichi River was determined by a salinity gradient, with greater parasite infections being recorded for freshwater sites and lower intensities being recorded in areas with higher salinity. Poulin et al. (2011) also indicated in a comparison of the community ecology of metazoan parasites infecting stickleback (Gasterosteus aculeatus) from Eurasia and North America that structural differences in parasite communities were related to salinity and geographical distance.
Effects of temperature and salinity therefore correspond to the type of environment, where temperature fluctuations correspond to changes in the population ecology of freshwater parasites, whereas salinity differences are an important abiotic variable influencing the occurrence of parasites in coastal waters (Schmidt et al. 2003).
Effect indicators
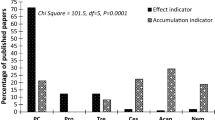
Effect indicators are those taxa which provide information about their environment based on their presence or absence (Sures 2001; Williams and Mackenzie 2003). With regard to water quality, variations in parasite communities have been found to vary with pollution levels in aquatic systems (Khan and Thulin 1991; Lafferty 1997; Sures 2005; Blanar et al. 2009). Under field conditions, the exact variable resulting in changes is often difficult to identify given the large number of factors that need to be considered, as well as any synergistic and antagonistic relationships between variables. The effects observed are therefore likely the result of a number of responses of parasites towards pollution and natural environmental influences. Studies detailing the effect of pollution at the component community level of parasites from freshwater, marine and terrestrial environments from across the globe have been summarised in Table 1. From the table, it is clear that such analyses have been conducted on a diverse group of parasites, but most studies have focussed on differences in component communities of particular parasite groups, such as Monogenea (number of studies 33), Trematoda (number of studies 21), Nematoda (number of studies 16) and Protozoa (number of studies 15). Overall, under particular conditions, parasites are more sensitive towards exposure to pollution than their hosts. Comparatively greater research interest has been devoted to freshwater and marine environments, while very few examples of terrestrial host–parasite models have been analysed.
Changes in the macroenvironment can either indirectly affect parasites through the hosts or directly where the parasites are affected by changes in the environment (Rohde 1984). Direct effects of pollution on the host have been linked to increases in parasitism, specifically towards the effect on immune mechanisms of the host (Bagge and Valtonen 1996), as well as increases in population density of definitive and intermediate hosts (Rohde 1984). Differences in digenean infections, for instance, as a result of pollution in the Kymijoki water system have been linked to decreased population density of clam intermediate hosts due to intolerably high levels of pollution in reaches of this water system (Valtonen et al. 1997; Karvonen et al. 2005). Nachev and Sures (2009) also suggested that greater endohelminth diversity in barbel (Barbus barbus) at locations impacted by eutrophication could occur due to higher occurrence of tolerant intermediate hosts. Shah et al. (2013) also attributed differences in helminth infections to densities of intermediate hosts between study sites. However, exposure of parasites and their hosts to particular pollutants has been conducted in an attempt to elucidate the effects that chemicals have on the population structure and infection variables of parasites. Siddall et al. (1997) exposed Rutilus rutilus (roach) infected with dactylogyrids to effluent collected from Lake Vatia and observed that in all cases the abundance and mean intensity of the infections decreased as the effluent concentration increased. Similarly, brown trout exposed to heavy metals were found to harbour more cysts of Eustrongyloides than unexposed fish (Brotheridge et al. 1998). From a population perspective, ectoparasitic monogeneans are ideal organisms to study changes in infection dynamics in relation to water quality as is evident from the large number of studies involving this group of parasites (Koskivaara et al. 1992; Bagge and Valtonen 1996; Dušek et al. 1998; Zargar et al. 2012a). Such organisms often require a single host for the completion of their life cycle (in the instance of crustaceans, only the adult is parasitic), and all life stages are in constant contact with the macroenvironment to which they have had to adapt. As a result of their ectoparasitic nature, they are exposed to both the host and surrounding environment (Sures 2004). Alterations in the community structure of ectoparasites have been identified in relation to a variety of different types of pollution and include eutrophication (Koskivaara and Valtonen 1991; Valtonen et al. 1997), pulp and paper mill effluent (Khan et al. 1994; Siddall et al. 1997) as well as changes in physical water quality variables such as temperature (Höglund and Thulin 1989), pH (Baker and Cone 2000; Halmetoja et al. 2000; MacLeod and Poulin 2012) and salinity (Soleng and Bakke 1997; Kua et al. 2013). In a meta-analysis of the effect size of pollution on parasite infrapopulations, Blanar et al. (2009) found that the largest effect sizes were associated with exposure to pulp and paper mill effluent, metals, pesticides and acidification. Of the groups of ectoparasite taxa studied, they further identified that Monogenea were the most affected by exposure to the abovementioned pollutants, whereas members of Arthropoda did not show any significant negative effect (Blanar et al. 2009). Meanwhile, for endoparasite taxa, such as the acanthocephalans, cestodes and nematodes, Blanar et al. (2009) found that there was no significant relationship between exposure to pollution and the presence or absence of these taxa. Digenea were the only endoparasitic taxa to be found to exhibit negative effects in relation to PCBs, pulp mill effluent, metals and eutrophication. In monogenean communities, alterations at the population level occur in a concentration-dependent manner and are governed by the type of pollutant exposed to. Sensitivity of some monogeneans towards metals is supported by laboratory studies where gyrodactylids have been exposed to a number of different metals (see Poléo et al. 2004; Soleng et al. 2005; Gheorghiu et al. 2007). However, pollution sensitivity of monogeneans also appears to be related to the concentration of toxicants within aquatic ecosystems. Skinner (1982) indicated that infection of fish by monogeneans increased as the concentrations of nutrients increased. Khan and Kiceniuk (1988) found that gyrodactylid infrapopulations did not significantly differ between treated and control groups, but results of their study indicated that mean intensity of infections increased as crude oil concentrations increased. Exposure to low concentrations of paper mill effluent resulted in a proliferation in the infrapopulation of Dactylogyrus (Bagge and Valtonen 1996) due to exposure compromising the immune system of the host, as well as stimulating the production of large amounts of mucous creating a favourable environment for the parasites. Valtonen et al. (1997) found that the lower prevalence of parasites in Lake Vatia compared to other sites was due to extensive pollution by pulp and paper mill effluent. Hypoxia, eutrophication and humus content have also been shown to affect gyrodactylid infrapopulations (Koskivaara et al. 1991).
Tuuha et al. (1992) found that of the five ergaslid species infecting fish from an interconnected lake system in Central Finland, Ergasilus sieboldi infections were related to the condition of the lakes, which were highest in an oligotrophic section of the system compared to other sections. Infections by other ergaslids from the same system were not related to water quality or pollution but instead were dependent on seasonal fluctuations. In the same lake system, Valtonen et al. (1997) demonstrated a similar trend for ergaslids from the same fish species, with little deviation in parasite prevalence between the different sites. Recovery in parasite infrapopulations has also been found to correlate to improvements in water quality as pollution levels decrease (Valtonen et al. 2003). Siddall et al. (1997) found that experimental exposure of several species of Dactylogyrus to a concentration of 10% pulp and paper mill effluent was sufficient to reduce the abundance and mean intensity of the parasites. In the Kymijoki River system in the Gulf of Finland, Koskivaara and Valtonen (1991) found differential effects of pulp and paper mill effluent and eutrophication on the mean abundance of monogeneans, protozoan and copepod parasites infecting R. rutilus (roach). For the monogenean, P. homoion, they identified that exposure to paper mill effluent reduced the mean abundance, whereas exposure to eutrophication led to an increase in the mean abundance when compared to the infection dynamics in an oligotrophic lake within the same river system. However, for dactylogyrid and gyrodactylid monogeneans, and the protozoan, Ichthyophthirius multifiliis, exposure to both pollution and eutrophication resulted in higher mean abundances compared to the mean abundance of the parasite at the oligotrophic site, and for the copepod, Ergasilus briani, the mean abundance at the oligotrophic site was higher than at the polluted and eutrophic sites (Koskivaara and Valtonen 1991).
Kuperman (1992) found that exposure to metals, phenol, naphthalene and oil compounds resulted in a reduction in mean abundance and intensity of Diplozoon paradoxum. Dušek et al. (1998) further indicated that the responses of specialist and generalist monogeneans differ with exposure to pollution. Specialist monogenean assemblages were found to be significantly reduced with exposure to eutrophication, whereas the opposite was identified for generalist parasites, which were found to parasitise a range of different hosts (Dušek et al. 1998).
Metal sensitivity has been demonstrated as a possible treatment for a wide range of parasite infections in aquaculture and under laboratory conditions. The sensitivity of gyrodactylids has been shown for a number of different metals under laboratory conditions (Poléo et al. 2004; Soleng et al. 2005; Pettersen et al. 2006; Gheorghiu et al. 2007). Poléo et al. (2004) found that of the metals analysed, only Al and Zn significantly reduced the infrapopulation size of G. salaris in a concentration-dependent manner, whereas exposure to Fe, Mn and Cu did not affect the parasites. The effect of Al on the survival of G. salaris was confirmed by Soleng et al. (2005). Pettersen et al. (2006) tested the sensitivity of Gyrodactylus derjavini and Gyrodactylus macronychus and similarly indicated complete elimination of the parasite from host fishes exposed to elevated metal concentrations. Gheorghiu et al. (2007) suggested that a reduction in the community size and sensitivity of Gyrodactylus turnbulli towards elevated Zn concentrations could be related to the direct effect of the metal on the in utero daughter parasites as well as stimulating the production of mucous by the host which later prevented reestablishment of the parasite. Hagen et al. (2014) found that reduced survival of G. salaris exposed to sodium hypochlorite resulted from toxicity of Cl− ions towards the parasite.
Zharikova (1993) suggested that a reduction in the abundance of D. paradoxum exposed to Cu under environmental conditions would result from the effects of the metal on the survival of oncomiracidium larvae. Reduced survival in other larval parasites exposed to metals has been studied extensively in digeneans (Cross et al. 2001; Morley et al. 2001, 2003, 2004). A reduction in the survival and swimming performance of larvae of Cryptocotyle lingua exposed to Zn and Mn was indicated by Cross et al. (2001), which ultimately led to a reduction in the infection of definitive hosts. Exposure of adult Fasciola hepatica to Pb reduced survival up to 70% when exposed to 200 mg Pb/L. In addition, exposure of snail intermediate hosts to Cd has been found to result in transference of toxicity towards digenean larval stages through reduced encystment rate within snails (Morley et al. 2004; Pietrock et al. 2008). The effect of metal exposure on the emergence rate of cercariae of a number of digeneans from snail intermediate hosts has been studied under laboratory conditions (e.g. Morley et al. 2001, 2003, 2004, 2005a, b). Exposure to Cd was further found to affect the emergence rate of cercariae of Diplostomum spathaceum from snail intermediate hosts compared to Echinoparyphium recurvatum which did not display any difference in emergence rates (Morley et al. 2003). Morley et al. (2004) then indicated that cercariae of E. recurvatum prematurely encysted within snail intermediate hosts due to unfavourable conditions of Cd-exposed snails.
Following on from this, Khalil et al. (2009) observed that S. acheilognathi coracidia would only succumb to the toxicity of Cd once hatched from the egg. Even when exposed to concentrations as high as 10 mg/L, normal coracidial development within the egg was observed. This is contrary to the findings by Morley et al. (2001) who observed reduced hatchability of the eggs of the trematode Schistosoma mansoni when exposed to 10 mg Cd/L. The authors suggested that hatching inhibition as a result of metal exposure resulted from the effect of Cd on aminopeptidase activity which facilitates the hatching process (Morley et al. 2001). In light of the similarities between monogenean and digenean eggs, it is likely that a similar trend would be observed for monogeneans exposed to metals under laboratory conditions. However, in a study by Thoney (1990), hatching of the eggs of the monogenean Benedeniella posterocolpa exposed to Cu was unaffected and was similarly shown by Khalil et al. (2009) for eggs of S. acheilognathi which were unaffected until the eggs hatched. Similarly, Gilbert and Avenant-Oldewage (2016c) showed that exposure to elevated levels of aluminium negatively affected the survival of oncomiracidia of P. ichthyoxanthon only once the larvae had hatched from the egg and became exposed to the metal. Both studies therefore indicate that the egg shell is a very effective barrier which provides a high degree of protection towards the developing larvae.
Accumulation indicators
Comparatively fewer studies have analysed pollutant levels within tissues of parasites compared to other accumulation indicators. Furthermore, as indicated above, there has been extensive work conducted on effect indication by parasites for monitoring the quality of aquatic environments with fewer studies addressing toxicant accumulation in parasites. Most of the studies addressing this aspect in parasites have analysed the levels of a number of metals within the tissues of endoparasites, particularly acanthocephalans and cestodes (summarised in Table 2). The majority of studies have focussed on endoparasites, with few ectoparasite examples being analysed in this regard. From the volume of work conducted on this aspect, it is clear based on the ability of accumulate metals that acnahtocephalans and cestodes, followed by nemtodes and lastly digeneans are of particular interest. Acanthocephalans are particularly effective accumulation indicators given their ability to bioconcentrate metals at levels orders of magnitude higher than those in their hosts (e.g. Siddall and Sures 1998; Nachev et al. 2010), and to levels which are comparatively greater than those demonstrated in other intestinal helminths, therefore showing the highest accumulation potential of the parasites studied thus far.
Further to this, parasites have been shown to affect the concentrations of metals in their hosts, where comparison between infected and uninfected hosts has identified lower somatic metal levels in the infected hosts compared to uninfected ones (Sures and Siddall 1999). This is believed to occur as both cestodes and acanthocephalans interrupt the hepatic–intestinal reabsorption of metal bound bile salts by the intestinal tract of the definitive hosts (Sures and Siddall 1999). Studies which have analysed the levels of metals in intermediate hosts compared to larval stages of the parasite found the opposite trend, where larval stages did not accumulate metals to higher levels than the invertebrate intermediate host and, in fact, the host had higher metal concentrations than the parasite (Sures 2006; Tekin-Özan and Barlas 2008; Frank et al. 2013). More recently, Le et al. (2016) further provided a model for metal uptake in host–parasite systems. The authors based this on the chub–acanthocephalan model (Squalius cephalus–P. laevis) exposed to Pb over a 36-day period. Nachev and Sures (2016) further indicated that the accumulation of trace elements by P. laevis differed between seasons. When temperatures were highest, accumulation of metals by this parasite was highest compared to lower temperatures. They related this finding to accumulation differing between different life stages of the parasite. Younger life stages, which were more abundant when water temperatures were cooler (autumn–winter), accumulated lower metal concentrations than older adults which were more abundant when water temperatures were higher (spring–summer).
Metal accumulation in cestodes and acanthocephalans has been found to be variable between segments of the strobila and male and female worms, respectively (Riggs et al. 1987; Sures et al. 1997a, b; Scheef et al. 2000). In both cases, it has been suggested that metals binding to the eggs of these parasites represent a regulatory function through which the parasites are able to detoxify themselves (Scheef et al. 2000; Degger et al. 2009; Khalil et al. 2009). Studies by Riggs et al. (1987) and Sures et al. (1997a) have indicated differential metal accumulation between body segments of the cestodes S. acheilognathi and Bothriocephalus scorpii, respectively. In both studies, metals were more concentrated in the posterior body segments compared to anterior segments. In the acanthocephalan, Moniliformis moniliformis, Scheef et al. (2000) identified higher concentrations of Cd in female worms compared to males and attributed this to the high number of eggs within females. This was similarly observed in two separate studies by Degger et al. (2009) and Khalil et al. (2009) for the cestode, S. acheilognathi. In the former study, positive fluorescence for metals was observed for the egg shells, whereas the latter detected Cd in the egg shells with the use of X-ray microanalysis (EDXMA). The sequestration of metals in the egg shells of these parasites is believed to occur through the enzymatic hardening or tanning of the egg shells whereby metals become incorporated into the shell matrix. This finding corroborated with studies where incorporation of metals into the egg cases of skates and dogfish (Koob 1991; Knight et al. 1996; Jeffree et al. 2008) was observed and suggested to occur during hardening of the case via enzymatic incorporation of catechol and oxidative cross linking with phenolic compounds (Degger et al. 2009). The involvement of the egg shell in removing metals from the bodies of adult organisms has also been indicated for a number of other invertebrates (Hook and Fisher 2001; Craig and Overnell 2003; Rosa et al. 2015) and vertebrates (Richards 1997; Blanco et al. 2003; Riggio et al. 2003; Brasfield et al. 2004).
In the case of ectoparasites, few studies have investigated metal accumulation in this group. One study (Gao and Nie 2000) attempted to analyse metal accumulation in the monogenean, Ancryocephalus mogurndae, which accumulated Pb at higher levels than the host. This indicates that like their endoparasite counterparts, monogeneans are able to accumulate metals at levels which surpass those in their hosts; however, further studies incorporating other members of this group are needed to confirm this suggestion.
In ectoparasites, studies on the elemental composition of the hard structures of the attachment organ have shown that some metals are incorporated into the structural components of these organs (Shinn et al. 1995). Shinn et al. (1995) indicated that elements such as V, Si, Na, Cl and P were present in different proportions within the sclerotised marginal hooklets, anchors and ventral bar of the haptor of Gyrodactylus. However, similar to the documentation of metal accumulation in ectoparasites, little is known about the sequestration mechanisms in this group. In invertebrates, a number of studies have indicated that metals are incorporated into sclerotised or hardened structures as a means of providing functional support to these organs.
Fewer studies documenting the accumulation of organic pollutants by parasites have been conducted and, therefore, with much research being devoted to the accumulation of metals in these organisms (e.g. Le et al. 2014). Le et al. (2014) reviewed the accumulation of persistent organic pollutants in parasites and indicated that lower accumulation potential for these compounds could be attributed to lower lipid content in the parasites compared to their hosts. Differences in organic pollutant content between infected and uninfected hosts have been related to the effect of the parasite on the physiology of the host (Sures 2006; Marcogliese and Pietrock 2011), which then affects the tolerance of the host towards exposure to organic pollutants (Heinonen et al. 2000; Le et al. 2014). Le et al. (2014) further suggested that differences in organic pollutant content between hosts and parasites could relate to the feeding mechanisms of the parasites and thus the trophic level of the parasite. Ectoparasites and endoparasites feeding on host tissue are therefore expected to have higher organic pollutant levels than endoparasites which feed by absorbing toxicants bound to compounds such as bile.
Biomarker responses in parasites
The toxic effects of pollutants have been studied for a number of aquatic organisms. Most studies involving the analysis of physiological responses of parasites towards pollution have dealt with metals. The degree of damage as a result of exposure differs with the nature and level of metals (Madoni and Romeo 2006; Wei and Yang 2015). Studies have associated metal exposure in organisms with the production of reactive oxygen species or intermediates (ROS/ROI), which result in oxidative stress (Hu 2000). A limited number of studies conducted on protozoan parasites and free living ciliates have documented the production of ROS in response to exposure to toxicants (Madoni and Romeo 2006; Mehta and Shaha 2006; Shukla et al. 2012). Mehta and Shaha (2006) observed that exposure of Leishmania donovani to metalloids, AsIII and SbIII, affected cells differently, and therefore, although the two metalloids are similar, resultant toxic effects manifest via two different mechanisms.
Other biomarker responses in helminths have been indicated for larval acanthocephalans (Sures and Radszuweit 2007) and nematodes (Chen et al. 2014; Keppel et al. 2014). In relation to metals, only Sures and Radszuweit (2007) have indicated a response in the production of heat shock protein 70 (Hsp-70) due to metal exposure. In this study, the authors observed that cysticanths exposed to metals produced higher levels of Hsp-70 than unexposed larvae, and further to this, Hsp-70 production in hosts infected with cysticanths was lower than uninfected exposed hosts. The production of Hsp-70 in nematodes, Anisakis spp. and Anguillicola crassus and Anguillicola novaezelandiae, respectively, has been observed in relation to increased temperature (Chen et al. 2014) and to the immune response of the host (Keppel et al. 2016), respectively.
Water quality, ecoparasitology and ecotoxicology in southern Africa
Water quality monitoring in South Africa has occurred since the 1950s (Roux et al. 1993; Claassens et al. 2016). Initial surveys were focussed on water supply for population growth and infrastructural expansion, but more recently, the focus has shifted to management and development following the inception of the National Water Quality Guidelines to assist in the determination of the suitability of water for all users (Department of Water Affairs and Forestry 1996). This was followed by the publication of the National Water Act in 1998 which relies on the integrated water resources management (IWRM) approach (Nomquphu et al. 2007).
Water resources in Africa are under threat due to continuing impact from anthropogenic pressures such as mining, agriculture, industrial activities and urbanisation (Wepener et al. 2011). Water quality monitoring methods have therefore evolved from traditional chemical analysis of water and sediment samples to biomonitoring (Roux et al. 1993; Crafford and Avenant-Oldewage 2010, 2011; Watson et al. 2012; Claassens et al. 2016), providing a time integrated analysis of the quality of aquatic ecosystems (Crafford and Avenant-Oldewage 2009; Watson et al. 2012). Such biomonitoring approaches have similarly incorporated a number of aquatic and marine organisms, such as fish (e.g. Mhlanga 2000; Wepener et al. 2011; Otachi et al. 2014), molluscs (e.g. Wepener et al. 2005; Cannicci et al. 2009; Mahmoud and Abu Taleb 2013), mussels (e.g. Degger et al. 2011a, b; Greenfield et al. 2011, 2014; Dahms et al. 2017), macroinvertebrates (e.g. Dickens and Graham 2002; Tate and Husted 2015), birds (e.g. Baker et al. 2016; van der Schyff et al. 2016), diatoms (e.g. Taylor et al. 2007; Harding and Taylor 2014) and in a few instances artificial mussels (Degger et al. 2011b; Greenfield et al. 2014; Claassens et al. 2016). More recently, attention has been devoted to exploring the usefulness of parasites from aquatic ecosystems (e.g. Retief et al. 2006, 2007; Oyoo-Okoth et al. 2010; Otachi et al. 2014; Torres et al. 2014; Gilbert and Avenant-Oldewage 2016b, c).
Incorporation of parasites as indicators in South African environments has been conducted at both population and individual levels. Populations of parasites have been incorporated into both pollution biomonitoring in freshwater and marine ecosystems (e.g. Crafford and Avenant-Oldewage 2009; Retief et al. 2006, 2007; Madanire-Moyo et al. 2012; Gilbert and Avenant-Oldewage 2016b, c; Morris et al. 2016), as well as indicators for monitoring sardine stocks, off the southern and western coasts of the country (e.g. Reed et al. 2012; Weston et al. 2015).
Overall, parasitological surveys in Africa have mostly focussed on parasite diversity while fewer studies have addressed the effects of pollution on these organisms in freshwater ecosystems (Retief et al. 2006; Olivier et al. 2009; Madanire-Moyo and Barson 2010; Madanire-Moyo et al. 2012; Gilbert and Avenant-Oldewage 2016b) compared to studies conducted in European rivers. Such studies are, however, sparse and predominantly localised to specific aquatic systems, namely the Vaal River and Limpopo River systems in southern Africa (e.g. Retief et al. 2006, 2009; Crafford and Avenant-Oldewage 2009; Madanire-Moyo et al. 2012; Gilbert and Avenant-Oldewage 2016b, c); terrestrial, aquatic and marine environments in northern Africa (e.g. Sures et al. 2003a, b; Torres et al. 2014; Abdel-Ghaffar et al. 2015; Abdel-Gaber et al. 2016); and aquatic ecosystems in Kenya (e.g. Otachi et al. 2014).
Following the development and implementation of the fish health assessment index (HAI), the parasite index (PI) was developed as an associated index to account for parasites associated with popular fish indicator species (Crafford and Avenant-Oldewage 2009). This index has been applied in riverine systems such as the Olifants River (Marx 1996; Watson et al. 2012) and the Vaal River (Crafford and Avenant-Oldewage 2009). Crafford and Avenant-Oldewage (2009) found that the prevalence of endoparasites and ectoparasites infecting the sharptooth catfish (Clarias gariepinus) was variable between two sites in the Vaal River. Endoparasites, overall, demonstrated higher prevalence at polluted sites compared to ectoparasites, which were higher in an unpolluted site. Crafford and Avenant-Oldewage (2009) therefore concluded that from such an analysis, it was possible to discriminate between study sites of differing water quality based on the presence or absence of parasites. The sensitivity of ectoparasites towards pollution levels within the Vaal River was later confirmed for a monogenean–host model, P. ichthyoxanthon infecting smallmouth yellowfish (Labeobarbus aeneus) where a local extinction of the parasites appeared to occur at a site below the Vaal River Barrage (Gilbert and Avenant-Oldewage 2016b).
Regarding accumulation of metals in parasites infecting South African fishes, a series of papers by Retief et al. (2006, 2009) detailed the usefulness of the cestode S. acheilognathi as an accumulation indicator for an array of trace elements in the Vaal River. Following this, Degger et al. (2009) determined with the use of fluorescence microscopy that the egg shells of the S. acheilognathi were sites where metals became sequestered and possibly indicated a mechanism for regulating the levels of metals in the tapeworms. Following from these studies, Morris et al. (2016) found that two endoparasites collected from sharks off the South African coast similarly accumulated trace elements 2–6 times higher than in the tissues of their hosts. For ectoparasites, the accumulation capacity of representatives of this group has yet to be examined. However, a study by Gilbert and Avenant-Oldewage (2017) indicated that metals accumulated by P. ichthyoxanthon collected from the Vaal Dam become sequestered in the vitellaria and sclerites of this parasite.
In other African countries, trace element and metal accumulation in numerous aquatic, marine and terrestrial host–parasite models have similarly been investigated and are mostly localised within North Africa and Central Africa. For terrestrial environments, a single study by Sures et al. (2003a, b) showed that the cestode, Hymenolepis diminuta, accumulated Pb to levels which were 97–36-fold greater than in the organs of the host, Rattus norvegicus. For the marine environment, Torres et al. (2014) studied the accumulation of Se and Hg in two fish hosts (Sarpa salpa and Balistes capriscus) and their respective digenean parasites (Robphildollfusium fractum and Neoapocreadium chabaudi) and indicated that levels of Se and Hg were higher in parasites than their hosts. Abdel-Ghaffar et al. (2015) similarly indicated that Fe, Cu, Cd and Ni were accumulated at higher concentrations by the nematode, Hysterothylacium aduncum, than the host fish sole (Solea solea) collected along the coast of Alexandria City, Egypt. In aquatic ecosystems, Otachi et al. (2014) found that only Zn was accumulated to higher levels in Contracaecum multipapillatum (L3) compared to the host and other trace elements were higher in host tissues than the parasites. Abdel-Gaber et al. (2016) similarly indicated that along with Pb, Zn, Cu, Mn, Cd and Ni were accumulated in P. clarias to greater concentrations than in the host fish, C. gariepinus, collected from Lake Manzala in the Nile Delta.
Marx (1996) indicated that the incorporation of parasites into the HAI accurately represented the condition of the environment and that parasite infections were lower when water quality deteriorated and this overall correlated with a higher HAI value for fish from the polluted sites along the Olifants River. In the Manyame River catchment in Zimbabwe, Madanire-Moyo and Barson (2010) found that parasite infections in C. gariepinus were related to water quality and exposure to sewage effluent. The levels of infection for all parasites were found to generally decrease as organic contaminant levels increased from the unpolluted site towards the polluted site. Variability was further identified between different parasites with pollution-tolerant species being dominant in polluted sites. The cestode Proteocephalus sp. was the only parasite present in the polluted site, and this could possibly relate to the eutrophic conditions favouring increased population density of intermediate hosts. Tylodephys sp. and Macrogyrodactylus sp. were pollution sensitive, and exposure to altered environmental conditions resulted in decreased infections along the length of the river in relation to levels of organic pollution content. Other parasites, such as Diplostomum sp., Caryophyllaeus sp. and Polyonchobothrium clarias, had highest prevalence in areas moderately affected by pollution, but as the levels increased, so the prevalence decreased similarly to those which are sensitive towards pollution (Madanire-Moyo and Barson 2010).
In the Limpopo River system, Madanire-Moyo et al. (2012) found that parasite infections in Oreochromis mossambicus were variably affected by water quality and exposure to sewage effluent at three dams in the Limpopo River and Olifants River systems. All parasites were found more prevalent on fish in the reference site compared to the polluted site, with the exception of gryporynchid larvae, which were more numerous at the polluted site (Madanire-Moyo et al. 2012). In the Olifants River, Watson et al. (2012) similarly indicated through the use of the HAI-PI that variations between infections by endoparasites and ectoparasites were related to water quality, with more endoparasites in impacted regions of the river and fewer ectoparasites. More recently, Dos Santos and Avenant-Oldewage (2016) indicated that the infection biology of a newly described diplozoid monogenean, from the two Labeo species inhabiting the Olifants and Selati rivers, Paradiplozoon krugerense, was negatively affected by decreased water quality and trace elements. No studies as of yet have investigated metal accumulation in parasites from fishes in this river system.
From the general trends outlined above for studies detailing changes in the infection biology of parasites in relation to water quality, it is evident that recent work has focussed on monogenean parasites in particular. In an experimental study, Gilbert and Avenant-Oldewage (2016c) indicated that exposure to aluminium reduced the survival of oncomiracidia of P. ichthyoxanthon, but only after the eggs had hatched and the larvae became completely exposed to the metal. This further suggested that the egg shell affords a high level of protection to the developing larvae and effectively prevents passage of foreign particles across the egg shell and into the egg. These results corroborate with the suggestion by Zharikova (1993) that changes in the abundance of monogeneans can be related to the sensitivity of the larval stages towards environmental contamination.
Conclusion
Recent studies have shown that parasites are affected by the condition of the macroenvironment, with effects between endoparasites and ectoparasites being variable. For this reason, it has been suggested in a number of reviews that parasites are suitable biological indicators, often responding to lower levels of perturbation which the host is not susceptible toward. Many studies have been conducted on a number of parasites from aquatic and marine ecosystems which have detailed the effects of pollution exposure at the component community level, while fewer studies have detailed the suitability of parasites as viable bioaccumulation indicators for monitoring levels of metals in the environment. In their meta-analysis of the effects of different types of pollution, Blanar et al. (2009) not only highlighted the sensitivity of different parasite taxa through variations in component communities towards pollution but also provided an account of the extent of the severity of exposure to numerous pollutants. Of the taxa assessed, they indicated that monogeneans and digeneans were the most sensitive. For both groups, sensitivity towards heavy metals have been tested and confirmed under laboratory conditions. Most studies have reported on the negative effects of pollution on parasite populations and decreases in infection variables in relation to water quality, and reports have also indicated that improvements in water quality can have a positive effect on parasite component communities. In such cases, decreases in pollution levels and improvement in water quality have been linked with increases in the infections by some parasites, whereas a decrease in other parasites associated with an improvement in the host immune response with decreased pollution levels was also indicative of improved water quality (Valtonen et al. 2003). From an accumulation perspective, parasites have been shown to be able to accumulate metals at very low exposure levels, which further supports their viability as accumulation indicators. The majority of these studies have been conducted on endoparasites, such as acanthocephalans, cestodes, nematodes and digeneans. Accumulation of metals by endoparasites have further been shown to be of benefit to the host, in that the parasites receive metals from their hosts, and in this way, infections diminish the somatic metal levels in the host tissues. In cestodes and acanthocephalans, it was further indicated that these organisms are able to regulate the levels of metals in their bodies through sequestration to the egg shells. It has been suggested that this process occurs during hardening of the egg shells during their formation. In cestodes, this has been shown to result in variable metal levels between the immature and gravid segments of the strobila and, therefore, further indicates that segments of the worms may be collected when they are released from the host and analysed for metals. In this way, the need to sacrifice the host would be avoided. Further study into the viability of this method of monitoring the levels of metals in the environment is however in need of validation.
Compared to Africa and South Africa, comparatively fewer investigations have addressed the effects of water quality on parasites. However, those that have, have indicated similar trends to those outlines in other studies. Furthermore, similarly to studies in host–parasite models in European river systems, parasites infecting African fishes are suitable indicators for monitoring the condition of the environment. The distribution of the studies conducted is sporadic and sparse; in South Africa for instance, these have only been conducted in two of the country’s major river systems. Furthermore, the focus of these investigations has mostly been at the population level, with three studies documenting the accumulation of metals in endoparasites from marine and freshwater ecosystems. However, despite the poverty in studies from the African continent detailing the effects of pollution on parasites, the general trends identified are similar to other studies from across the world. With that said, more intense investigation into the effects of pollution on parasites is required to adequately understand the processes occurring in these organisms in relation to the condition of the macroenvironment.
References
Abdel-Gaber R, Abdel-Ghaffar F, Bashtar A-R, Morsy K, Saleh R (2016) Interactions between the intestinal cestode Polyonchobothrium clarias (Pseudophyllidea: Ptychobothriidae) from the African sharptooth catfish Clarias gariepinus and heavy metal pollutants in an aquatic environment in Egypt. J Helminthol 90:742–752. doi:10.1017/S0022149X15001054
Abdel-Ghaffar F, Abdel-Gaber R, Bashtar A-R, Morsy K, Mehlhorn H, Al Quraishy S, Saleh R (2015) Hysterothylacium aduncum (Nematoda, Anisakidae) with a new host record from the common sole Solea solea (Soleidae) and its role as a biological indicator of pollution. Parasitol Res 114:513–522. doi:10.1007/s00436-014-4213-1
Altizer S, Dobson A, Hosseini P, Hudson P, Pascual M, Rohani P (2006) Seasonality and the dynamics of infectious diseases. Ecol Lett 9:467–484. doi:10.1111/j.1461-0248.2005.00879.x
Anderson TK, Sukhdeo MVK (2010) Abiotic versus biotic hierarchies in the assembly of parasite populations. Parasitology 137:743–754. doi:10.1017/S0031182009991430
Austin A, Avenant-Oldewage A (2009) Ecological parameters of Lamproglena hoi (Copepoda: Lernaeidae) infection on the Bushveld smallscale yellowfish, Labeobarbus polylepis (Boulenger, 1907). Onderstepoort J Vet Res 76:227–234. doi:10.4102/ojvr.v76i2.47
Avenant-Oldewage A, Marx HM (2000) Bioaccumulation of chromium, copper and iron in the organs and tissues of Clarias gariepinus in the Olifants River, Kruger National Park. Water SA 26:569–582
Bagge AM, Valtonen ET (1996) Experimental study on the influence of paper and pulp mill effluent on the gill parasite communities of roach (Rutilus rutilus). Parasitology 112:499–508. doi:10.1017/S0031182000076964
Baker DE, Cone DK (2000) Occurrence of Ergasilus celestis (Copepoda) and Pseudodactylogyrus anguillae (Monogenea) among wild eels (Anguilla anguilla) in relation to stream flow, pH and temperature and recommendations for controlling their transmission among captive eels. Aquaculture 187:261–274
Baker N, Maina J, Greenfield R (2016) Zinc and cadmium concentrations in the house sparrow (Passer domesticus), Thohoyandou, Limpopo, South Africa. In: 7th International Toxicology Symposium in Africa
Baruš V, Jarkovský J, Prokeš M (2007) Philometra ovata (Nematoda: Philometroidea): a potential sentinel species of heavy metal accumulation. Parasitol Res 100:929–933. doi:10.1007/s00436-006-0384-8
Bayoumy EM, Osman HAM, El-Bana LF, Hassanain MA (2008) Monogenean parasites as bioindicators for heavy metals status in some Egyptian Red Sea fishes. Glob Vet 2:117–122
Bergey L, Weis JS, Weis P (2002) Mercury uptake by the estuarine species Palaemonetes pugio and Fundulus heteroclitus compared with their parasites, Probopyrus pandalicola and Eustrongylides sp. Mar Pollut Bull 44:1046–1050. doi:10.1016/S0025-326X(02)00154-6
Billiard SM, Khan RA (2003) Chronic stress in cunner, Tautogolabrus adspersus, exposed to municipal and industrial effluents. Ecotoxicol Environ Saf 55:9–18. doi:10.1016/S0147-6513(02)00090-8
Biney C, Amuzu AT, Calamari D, Kaba N, Mbome IL, Naeve H, Ochumba PBO, Osibanjo O, Radegonde V, Saad MAH (1994) Review of heavy metals in the African aquatic environment. Ecotoxicol Environ Saf 28:134–159
Bird DJ, Duquesne S, Hoeksema SD, Langston WJ, Potter IC (2011) Complexity of spatial and temporal trends in metal concentrations in macroinvertebrate biomonitor species in the Severn Estuary and Bristol Channel. J Mar Biol Assoc U K 91:139–153. doi:10.1017/S0025315410001918
Blanar CA, Munkittrick KR, Houlahan J, MacLatchy DL, Marcogliese DJ (2009) Pollution and parasitism in aquatic animals: a meta-analysis of effect size. Aquat Toxicol 93:18–28. doi:10.1016/j.aquatox.2009.03.002
Blanar CA, Marcogliese DJ, Couillard C (2011) Natural and anthropogenic factors shape metazoan parasite community structure in mummichog (Fundulus heteroclitus) from two estuaries in New Brunswick, Canada. Folia Parasitol (Praha) 58:240–248. doi:10.14411/fp.2011.023
Blanco G, Frías O, Jiménez B, Gómez G (2003) Factors influencing variability and potential uptake routes of heavy metals in black kites exposed to emissions from a solid-waste incinerator. Environ Toxicol Chem 22:2711–2718. doi:10.1897/02-519
Blažek R, Jarkovský J, Koubková B, Gelnar M (2008) Seasonal variation in parasite occurrence and microhabitat distribution of monogenean parasites of gudgeon Gobio gobio (L.) Helminthologia 45:185–191. doi:10.2478/s11687-008-0037-9
Boggs JF, McMurry ST, Leslie DM, Engle DM, Lochmiller RL (1991) Influence of habitat modification on the community of gastrointestinal helminths of cotton rats. J Wildl Dis 27:584–593. doi:10.7589/0090-3558-27.4.584
Brabec J, Waeschenbach A, Scholz T, Littlewood DTJ, Kuchta R (2015) Molecular phylogeny of the Bothriocephalidea (Cestoda): molecular data challenge morphological classification. Int J Parasitol 45:761–771. doi:10.1016/j.ijpara.2015.05.006
Brasfield SM, Bradham K, Wells JB, Talent LG, Lanno RP, Janz DM (2004) Development of a terrestrial vertebrate model for assessing bioavailability of cadmium in the fence lizard (Sceloporus undulatus) and in ovo effects on hatchling size and thyroid function. Chemosphere 54:1643–1651. doi:10.1016/j.chemosphere.2003.09.030
Brázová T, Torres J, Eira C, Hanzelová V, Miklisová D, Šalamún P (2012) Perch and its parasites as heavy metal biomonitors in a freshwater environment: the case study of the Ružín Water Reservoir, Slovakia. Sensors 12:3068–3081. doi:10.3390/s120303068
Brázová T, Hanzelová V, Miklisová D, Šalamún P, Vidal-Martínez VM (2015) Host-parasite relationships as determinants of heavy metal concentrations in perch (Perca fluviatilis) and its intestinal parasite infection. Ecotoxicol Environ Saf 122:551–556. doi:10.1016/j.ecoenv.2015.09.032
Brito SV, Ferreira FS, Ribeiro SC, Anjos LA, Almeida WO, Mesquita DO, Vasconcellos A (2014) Spatial-temporal variation of parasites in Cnemidophorus ocellifer (Teiidae) and Tropidurus hispidus and Tropidurus semitaeniatus (Tropiduridae) from Caatinga areas in northeastern Brazil. Parasitol Res 113:1163–1169. doi:10.1007/s00436-014-3754-7
Broeg K, Zander S, Diamant A, Körting W, Krüner G, Paperna I, Westernhagen HV (1999) The use of fish metabolic, pathological and parasitological indices in pollution monitoring. I. North Sea. Helgol Mar Res 53:171–194. doi:10.1007/s101520050024
Brotheridge RM, Newton KE, Evans SW (1998) Presence of a parasitic nematode (Eustrongyloides sp.) in brown trout (Salmo trutta) from a heavy metal contaminated aquatic ecosystem. Chemosphere 37:2921–2934
Brown AF, Pascoe D (1989) Parasitism and host sensitivity to cadmium: an acanthocephalan infection of the freshwater amphipod Gammarus pulex. J Appl Ecol 26:473–487
Čadková Z, Miholová D, Száková J, Válek P, Jankovská I, Langrová I (2014) Is the tapeworm able to affect tissue Pb-concentrations in white rat? Parasitology 141:826–836. doi:10.1017/S0031182013002242
Cannicci S, Bartolini F, Dahdouh-Guebas F, Fratini S, Litulo C, Macia A, Mrabu EJ, Penha-Lopes G, Paula J (2009) Effects of urban wastewater on crab and mollusc assemblages in equatorial and subtropical mangroves of East Africa. Estuar Coast Shelf Sci 84:305–317. doi:10.1016/j.ecss.2009.04.021
Chang ACG, Flores MJC (2015) Morphology and viability of adult Fasciola gigantica (giant liver flukes) from Philippine carabaos (Bubalus bubalis) upon in vitro exposure to lead. Asian Pac J Trop Biomed 5:493–496. doi:10.1016/j.apjtb.2015.03.008
Chapman LJ, Lanciani CA, Chapman CA (2000) Ecology of a diplozoon parasite on the gills of the African cyprinid Barbus neumayeri. Afr J Ecol 38:312–320. doi:10.1046/j.1365-2028.2000.00252.x
Chen HY, Cheng YS, Grabner DS, Chang SH, Shih HH (2014) Effect of different temperatures on the expression of the newly characterized heat shock protein 90 (Hsp90) in L3 of Anisakis spp. isolated from Scomber australasicus. Vet Parasitol 205:540–550. doi:10.1016/j.vetpar.2014.09.013
Chibani M, Ziólkowska M, Kijewska A, Rokicki J (2001) Pomphorhynchus laevis parasite of flounder Platichthys flesus as a biological indicator of pollution in the Baltic Sea. J Mar Biol Assoc U K 81:165–166. doi:10.1017/S0025315401003514
Claassens L, Dahms S, van Vuren JHJ, Greenfield R (2016) Artificial mussels as indicators of metal pollution in freshwater systems: a field evaluation in the Koekemoer Spruit, South Africa. Ecol Indic 60:940–946. doi:10.1016/j.ecolind.2015.08.047
Crafford D, Avenant-Oldewage A (2009) Application of a fish health assessment index and associated parasite index to Clarias gariepinus (Teleostei: Clariidae) in the Vaal River system, South Africa. Afr J Aquat Sci 34:261–272. doi:10.2989/AJAS.2009.34.3.8.984
Crafford D, Avenant-Oldewage A (2010) Bioaccumulation of non-essential trace metals in tissues and organs of Clarias gariepinus (sharptooth catfish) from the Vaal River system—strontium, aluminium, lead and nickel. Water SA 36:621–640. doi:10.4314/wsa.v36i5.61996
Crafford D, Avenant-Oldewage A (2011) Uptake of selected metals in tissues and organs of Clarias gariepinus (sharptooth catfish) from the Vaal River system—chromium, copper, iron, manganese and zinc. Water SA 37:181–200. doi:10.4314/wsa.v37i2.65864
Crafford D, Luus-Powell W, Avenant-Oldewage A (2014) Monogenean parasites from fishes of the Vaal Dam, Gauteng Province, South Africa I. Winter survey versus summer survey comparison from Labeo capensis (Smith 1841) and Labeo umbratus (Smith, 1841) hosts. Acta Parasitol 59:17–24. doi:10.2478/s11686-014-0205-7-x
Craig S, Overnell J (2003) Metals in squid, Loligo forbesi, adults, eggs and hatchlings. No evidence for a role for Cu- or Zn-metallothionein. Comp Biochem Physiol - C Toxicol Pharmacol 134:311–317. doi:10.1016/S1532-0456(02)00274-0
Cross MA, Irwin SW, Fitzpatrick SM (2001) Effects of heavy metal pollution on swimming and longevity in cercariae of Cryptocotyle lingua (Digenea: Heterophyidae). Parasitology 123:499–507. doi:10.1017/S0031182001008708
Dahms S, Baker NJ, Greenfield R (2017) Ecological risk assessment of trace elements in sediment: a case study from Limpopo, South Africa. Ecotoxicol Environ Saf 135:106–114. doi:10.1016/j.ecoenv.2016.09.036
de Buron I, James E, Riggs-Gelasco P, Ringwood AH, Rolando E, Richardson D (2009) Overview of the status of heavy metal accumulation by helminths with a note on the use of in vitro culture of adult acanthocephalans to study the mechanisms of bioaccumulation. Neotrop Helminthol 3:101–110
Degger N, Avenant-Oldewage A, Greenfield R (2009) Innovative fluorescence detection technique for metals in cestode egg-shells. Afr Zool 44:204–207. doi:10.3377/004.044.0208
Degger N, Wepener V, Richardson BJ, Wu RSS (2011a) Brown mussels (Perna perna) and semi-permeable membrane devices (SPMDs) as indicators of organic pollutants in the South African marine environment. Mar Pollut Bull 63:91–97. doi:10.1016/j.marpolbul.2011.04.024
Degger N, Wepener V, Richardson BJ, Wu RSS (2011b) Application of artificial mussels (AMs) under South African marine conditions: a validation study. Mar Pollut Bull 63:108–118. doi:10.1016/j.marpolbul.2011.04.040
Department of Water Affairs and Forestry (1996) South African Water Quality Guidelines. Volume 7: aquatic ecosystems
Dickens CWS, Graham PM (2002) The South African Scoring System (SASS) version 5 rapid bioassessment method for rivers. Afr J Aquat Sci 27:1–10. doi:10.2989/16085914.2002.9626569
Dos Santos QM, Avenant-Oldewage A (2016) The description of a new diplozoid species, Paradiplozoon krugerense n. sp., from Labeo rosae Steindachner, 1894 and Labeo congoro Peters, 1852 in the Kruger National Park, South Africa with notes on the effect of water quality on its infection variables. Hydrobiologia 777:225–241. doi:10.1007/s10750-016-2776-9
Dušek L, Gelnar M, Šebelová Š (1998) Biodiversity of parasites in a freshwater environment with respect to pollution: metazoan parasites of chub (Leuciscus cephalus L.) as a model for statistical evaluation. Int J Parasitol 28:1555–1571. doi:10.1016/S0020-7519(98)00130-1
Dzika E, Kuształa A, Kuształa M (2007) Parasites of carp bream, Abramis brama, from Lake Jamno, Poland. Helminthologia 44:222–225
Dzikowski R, Paperna I, Diamant A (2003) Use of fish parasite species richness indices in analyzing anthropogenically impacted coastal marine ecosystems. Helgol Mar Res 57:220–227. doi:10.1007/s10152-003-0138-2
Eira C, Torres J, Vingada J, Miquel J (2005) Concentration of some toxic elements in Oryctolagus cuniculus and in its intestinal cestode Mosgovoyia ctenoides, in Dunas de Mira (Portugal). Sci Total Environ 346:81–86. doi:10.1016/j.scitotenv.2004.11.014
Eira C, Torres J, Miquel J, Vaqueiro J, Soares AMVM, Vingada J (2009) Trace element concentrations in Proteocephalus macrocephalus (Cestoda) and Anguillicola crassus (Nematoda) in comparison to their fish host, Anguilla anguilla in Ria de Aveiro, Portugal. Sci Total Environ 407:991–998. doi:10.1016/j.scitotenv.2008.10.040
Faulkner BC, Lochmiller RL (2000) Ecotoxicity revealed in parasite communities of Sigmodon hispidus in terrestrial environments contaminated with petrochemicals. Environ Pollut 110:135–145. doi:10.1016/S0269-7491(99)00276-6
Förstner U, Prosi F (1979) Heavy metal pollution in freshwater ecosystems. In: Ravera O (ed) Biological aspects of freshwater pollution. Pergamon, Oxford, pp 129–214
Frank SN, Godehardt S, Nachev M, Trubiroha A, Kloas W, Sures B (2013) Influence of the cestode Ligula intestinalis and the acanthocephalan Polymorphus minutus on levels of heat shock proteins (HSP70) and metallothioneins in their fish and crustacean intermediate hosts. Environ Pollut 180:173–179. doi:10.1016/j.envpol.2013.05.014
Galli P, Crosa G, Mariniello L, Ortis M, D’Amelio S (2001) Water quality as a determinant of fish parasite communities. Hydrobiologia 452:173–179
Gao Q, Nie P (2000) Lead content in the monogenean Ancryocephalus mogurndae and in different organs of its host, the mandarin fish, Siniperca chuatsi. China Environ Sci 20:233–236
Gelnar M (1991) Experimental verification of the effect of constant and changing water temperature on the micropopulation growth in Gyrodactylus gobiensis Gläser, 1974 (Monogenea) parasitizing Gudgeon (Gobio gobio L.) Folia Parasitol (Praha) 38:123–131
Gheorghiu C, Cable J, Marcogliese DJ, Scott ME (2007) Effects of waterborne zinc on reproduction, survival and morphometrics of Gyrodactylus turnbulli (Monogenea) on guppies (Poecilia reticulata). Int J Parasitol 37:375–381. doi:10.1016/j.ijpara.2006.09.004
Gheorgiu C, Marcogliese DJ, Scott M (2006) Concentration-dependent effects of waterborne zinc on population dynamics of Gyrodactylus turnbulli (Monogenea) on isolated guppies (Poecilia reticulata). Parasitology 132:225–232. doi:10.1017/S003118200500898X
Gilbert BM, Avenant-Oldewage A (2016a) Seasonal occurrence and microhabitat specificity of Paradiplozoon ichthyoxanthon Avenant-Oldewage in Avenant-Oldewage et al., 2014 (Monogenea: Diplozoidae) infecting Labeobarbus aeneus (Burchell) (Teleostei: Cyprinidae) from the Vaal Dam, South Africa: water quality and host size as determining factors? Folia Parasitol (Praha) 63:4. doi:10.14411/fp.2016.004
Gilbert BM, Avenant-Oldewage A (2016b) Effects of altered water quality and trace elements on the infection variables of Paradiplozoon ichthyoxanthon (Monogenea: Diplozoidae) from two sites in the Vaal River system, South Africa. Acta Parasitol 61:52–62. doi:10.1515/ap-2016-0005
Gilbert BM, Avenant-Oldewage A (2016c) Hatchability and survival of oncomiracidia of Paradiplozoon ichthyoxanthon (Monogenea: Diplozoidae) exposed to aqueous aluminium. Parasit Vectors 9:420
Gilbert BM, Avenant-Oldewage A (2017) Metal sequestration in vitellaria and sclerites, and reactive oxygen intermediates in a freshwater monogenean, Paradiplozoon ichthyoxanthon. PLoS One 12:e0177558. doi:10.1371/journal.pone.0177558
Goodyear KL, McNeill S (1999) Bioaccumulation of heavy metals by aquatic macro-invertebrates of different feeding guilds: a review. Sci Total Environ 229:1–19. doi:10.1016/S0048-9697(99)00051-0
Granath W, Esch GW (1983) Temperature and other factors that regulate the composition and infrapopulation densities of Bothriocephalus acheilognathi (Cestoda) in Gambusia affinis (Pisces). J Parasitol 69:1116–1124
Greenfield R, Wepener V, Degger N, Brink K (2011) Richards Bay Harbour: metal exposure monitoring over the last 34 years. Mar Pollut Bull 62:1926–1931. doi:10.1016/j.marpolbul.2011.04.026
Greenfield R, Brink K, Degger N, Wepener V (2014) The usefulness of transplantation studies in monitoring of metals in the marine environment: South African experience. Mar Pollut Bull 85:566–573. doi:10.1016/j.marpolbul.2014.03.032
Hagen AG, Hytterød S, Olstad K (2014) Low concentrations of sodium hypochlorite affect population dynamics in Gyrodactylus salaris (Malmberg, 1957): practical guidelines for the treatment of the Atlantic salmon, Salmo salar L. parasite. J Fish Dis 37:1003–1011. doi:10.1111/jfd.12218
Hakalahti T, Valtonen ET (2003) Population structure and recruitment of the ectoparasite Argulus coregoni Thorell (Crustacea: Branchiura) on a fish farm. Parasitology 127:79–85. doi:10.1017/S0031182003003196
Halmetoja A, Valtonen ET, Koskenniemi E (2000) Perch (Perca fluviatilis L.) parasites reflect ecosystem conditions: a comparison of a natural lake and two acidic reservoirs in Finland. Int J Parasitol 30:1437–1444. doi:10.1016/S0020-7519(00)00115-6
Hanzelova V (1992) Proteocephalus neglectus as a possible indicator of changes in the ecological balance of aquatic environments. J Helminthol 66:17–24. doi:10.1017/S0022149X00012517
Harding WR, Taylor JC (2014) Diatoms as indicators of historical water quality: a comparison of samples taken in the Wemmershoek catchment (Western Province, South Africa) in 1960 and 2008. Water SA 40:601–606. doi:10.4314/wsa.v40i4.4
Harvell CD, Kim K, Burkholder JM, Colwell RR, Epstein PR, Grimes DJ, Hofmann EE, Lipp EK, Osterhaus ADME, Overstreet RM, Porter JW, Smith GW, Vasta GR (1999) Emerging marine diseases: climate links and anthropogenic factors. Science 285:1505–1510. doi:10.1126/science.285.5433.1505
Harvell CD, Mitchell CE, Ward JR, Altizer S, Dobson AP, Ostfeld RS, Samuel MD (2002) Climate warming and disease risks for terrestrial and marine biota. Science 296:2158–2162. doi:10.1126/science.1063699
Hayes ML, Bonaventura J, Mitchell TP, Prospero JM, Shinn EA, Van Dolah F, Barber RT (2001) How are climate and marine biological outbreaks functionally linked? Hydrobiologia 460:213–220
Heinonen J, Kukkonen JVK, Holopainen IJ (2000) Toxicokinetics of 2,4,5-trichlorophenol and benzo(a)pyrene in the clam Pisidium amnicum: effects of seasonal temperatures and trematode parasites. Arch Environ Contam Toxicol 39:352–359. doi:10.1007/s002440010115
Hodson PV (1988) The effect of metal metabolism on uptake, disposition and toxicity in fish. Aquat Toxicol 11:3–18
Hofmann H, Blasco-Costa I, Knudsen R, Matthaei CD, Valois A, Lange K (2016) Parasite prevalence in an intermediate snail host is subject to multiple anthropogenic stressors in a New Zealand river system. Ecol Indic 60:84–852. doi:10.1016/j.ecolind.2015.08.022
Höglund J, Thulin J (1989) Thermal effects on the seasonal dynamics of Paradiplozoon homoion (Bychowsky & Nagibina, 1959) parasitizing roach, Rutilus rutilus (L.) J Helminthol 63:93. doi:10.1017/S0022149X0000883X
Holt EA, Miller SW (2010) Bioindicators: using organisms to measure environmental impacts. Nat Educ Knowl 3:8. doi:10.5962/bhl.title.62081
Hook SE, Fisher NS (2001) Reproductive toxicity of metals in calanoid copepods. Mar Biol 138:1131–1140. doi:10.1007/s002270000533
Hu H (2000) Exposure to metals. Prim Care - Clin Off Pract 27:983–996. doi:10.1016/S0095-4543(05)70185-8
Jakob E, Hanel R, Klimpel S, Zumholz K (2008) Salinity dependence of parasite infestation in the European eel Anguilla anguilla in northern Germany. ICES J Mar Sci 66:358–366. doi:10.1093/icesjms/fsn160
Jankovská I, Vadlejch J, Száková J, Miholová D, Kunc P, Knížková I, Čadková Z, Langrová I (2010) Experimental studies on the cadmium accumulation in the cestode Moniezia expansa (Cestoda: Anoplocephalidae) and its final host (Ovis aries). Exp Parasitol 126:130–134. doi:10.1016/j.exppara.2010.04.010
Jeffree RA, Oberhansli F, Teyssie J-L (2008) The accumulation of lead and mercury from seawater and their depuration by eggs of the spotted dogfish Scyliorhinus canicula (Chondrichthys). Arch Environ Contam Toxicol 55:451–461
Jeney Z, Valtonen ET, Jeney G, Jokinen EI (2002) Effect of pulp and paper mill effluent (BKME) on physiological parameters of roach (Rutilus rutilus) infected by the digenean Rhipidocotyle fennica. Folia Parasitol (Praha) 49:103–108
Jirsa F, Leodolter-Dvorak M, Krachler R, Frank C (2008) Heavy metals in the nase, Chondrostoma nasus (L. 1758), and its intestinal parasite Caryophyllaeus laticeps (Pallas 1781) from Austrian rivers: bioindicative aspects. Arch Environ Contam Toxicol 55:619–626. doi:10.1007/s00244-008-9154-1
Jirsa F, Konecny R, Frank C, Sures B (2011) The parasite community of the nase Chondrostoma nasus (L. 1758) from Austrian rivers. J Helminthol 85:255–262. doi:10.1017/S0022149X10000490
Kádár E, Costa V, Santos RS, Powell JJ (2006) Tissue partitioning of micro-essential metals in the vent bivalve Bathymodiolus azoricus and associated organisms (endosymbiont bacteria and a parasite polychaete) from geochemically distinct vents of the Mid-Atlantic Ridge. J Sea Res 56:45–52. doi:10.1016/j.seares.2006.01.002
Karvonen A, Bagge AM, Valtonen ET (2005) Parasite assemblages of crucian carp (Carassius carassius)—is depauperate composition explained by lack of parasite exchange, extreme environmental conditions or host unsuitability? Parasitology 131:273–278. doi:10.1017/S0031182005007572
Keppel M, Dangel KC, Sures B (2014) Comparison of infection success, development and swim bladder pathogenicity of two congeneric Anguillicola species in experimentally infected Anguilla anguilla and A. japonica. Parasitol Res 113:3727–3735. doi:10.1007/s00436-014-4038-y
Keppel M, Dangel KC, Sures B (2016) The Hsp70 response of Anguillicola species to host-specific stressors. Parasitol Res 115:2149–2154. doi:10.1007/s00436-016-4956-y
Khalil M, Furness D, Polwart A, Hoole D (2009) X-ray microanalysis (EDXMA) of cadmium-exposed eggs of Bothriocephalus acheilognathi (Cestoda: Bothriocephalidea) and the influence of this heavy metal on coracidial hatching and activity. Int J Parasitol 39:1093–1098. doi:10.1016/j.ijpara.2009.02.023
Khan RA, Kiceniuk J (1983) Effects of crude oils on the gastrointestinal parasites of two species of marine fish. J Wildl Dis 19:253–258
Khan RA, Kiceniuk JW (1988) Effect of petroleum aromatic hydrocarbons on monogeneids parasitizing Atlantic cod, Gadus morhua L. Bull Environ Contam Toxicol 41:94–100
Khan AR, Rasheed M (1993) Influence of season on the intensity of helminth infection in Schizothorax esocinus heckel from the Wular Lake. Indian J Helminthol 45:64–73
Khan RA, Thulin J (1991) Influence of pollution on parasites of aquatic animals. Adv Parasitol 30:201–239
Khan RA, Barker DE, Williams-Ryan K, Hooper RG (1994) Influence of crude oil and pulp and paper mill effluent on mixed infections of Trichodina cottidarium and T. saintjohnsi (Ciliophora) parasitizing Myoxocephalus octodecemspinosus and M. scorpius. Can J Zool 72:247–251
Knight DP, Feng D, Stewart M (1996) Structure and function of the selachian egg case. Biol Rev 71:81–91
Koob TJ (1991) Deposition and binding of calcium and magnesium in egg capsules of Raja erinacea Mitchill during formation and tanning in utero. Copeia 1991:339–347
Koskivaara M, Valtonen ET (1991) Paradiplozoon homoion (Monogenea) and some other gill parasites on roach Rutilus rutilus in Finland. Aqua Fenn 21:137–143
Koskivaara M, Valtonen ET, Prost M (1991) Seasonal occurrence of gyrodactylid monogeneans on the roach (Rutilus rutilus) and variations between four lakes of differing water quality in Finland. Aqua Fenn 21:47–55. doi:10.1016/0020-7519(91)90061-B
Koskivaara M, Valtonen ET, Vuori KM (1992) Microhabitat distribution and coexistence of Dactylogyrus species (Monogenea) on the gills of roach. Parasitology 104:273–281. doi:10.1017/S0031182000061710
Kua BC, Choong FC, Leaw YY (2013) Effect of salinity and temperature on marine leech, Zeylanicobdella arugamensis (De Silva) under laboratory conditions. J Fish Dis 37:201–207. doi:10.1111/jfd.12087
Kuperman BI (1992) Fish parasites as biological indicators of pollution of water bodies. Parazitologiya 26:479–482
Lafferty KD (1997) Reviews environmental parasitology: what can parasites tell us about human impacts on the environment? Parasitol Today 13:251–255
Lamková K, Šimková A, Palíková M, Jurajda P, Lojek A (2007) Seasonal changes of immunocompetence and parasitism in chub (Leuciscus cephalus), a freshwater cyprinid fish. Parasitol Res 101:775–789. doi:10.1007/s00436-007-0546-3
le Roux LE, Avenant-Oldewage A, van der Walt FC (2011) Aspects of the ecology of Cichlidogyrus philander collected from Pseudocrenilabrus philander philander from the Padda Dam, Gauteng, South Africa. Afr Zool 46:103–116. doi:10.1080/15627020.2011.11407484
Le TTY, Rijsdijk L, Sures B, Jan Hendriks A (2014) Accumulation of persistent organic pollutants in parasites. Chemosphere 108:145–151. doi:10.1016/j.chemosphere.2014.01.036
Le TTY, Nachev M, Grabner D, Hendriks AJ, Sures B (2016) Development and validation of a biodynamic model for mechanistically predicting metal accumulation in fish-parasite systems. PLoS ONE 11(8):e01610901. doi:10.1371/journal.pone.0161091
Lefcort H, Aguon MQ, Bond KA, Chapman KR, Chaquette R, Clark J, Kornachuk P, Lang BZ, Martin JC (2002) Indirect effects of heavy metals on parasites may cause shifts in snail species compositions. Arch Environ Contam Toxicol 43:34–41. doi:10.1007/s00244-002-1173-8
Leonard EM, Wood CM (2013) Acute toxicity, critical body residues, Michaelis-Menten analysis of bioaccumulation, and ionoregulatory disturbance in response to waterborne nickel in four invertebrates: Chironomus riparius, Lymnaea stagnalis, Lumbriculus variegatus and Daphnia pulex. Comp Biochem Physiol - C Toxicol Pharmacol 158:10–21. doi:10.1016/j.cbpc.2013.03.008
Lins DC, Meirelles ME, Malm O, Lima NRW (2008) Mercury concentration in the freshwater bonefish Cyphocharax gilbert (Curimatidae) and its parasite the crustacean Riggia paranensis (Cymothoidae). Neotrop Ichthyol 6:283–288. doi:10.1590/S1679-62252008000200017
Lõhmus M, Björklund M (2015) Climate change: what will it do to fish–parasite interactions? Biol J Linn Soc 116:397–411. doi:10.1111/bij.1284
MacDonald S, Jones A (1978) Egg-laying and hatching rhythms in the monogenean Diplozoon homoion gracile from the southern barbel (Barbus meridionalis). J Helminthol 52:23–28
MacKenzie K (1999) Parasites as pollution indicators in marine ecosystems: a proposed early warning system. Mar Pollut Bull 38:955–959. doi:10.1016/S0025-326X(99)00100-9
MacLeod CD, Poulin R (2012) Host-parasite interactions: a litmus test for ocean acidification? Trends Parasitol 28:365–369. doi:10.1016/j.pt.2012.06.007
Madanire-Moyo GN, Barson M (2010) Diversity of metazoan parasites of the African catfish Clarias gariepinus (Burchell, 1822) as indicators of pollution in a subtropical African river system. J Helminthol 84:216–227. doi:10.1017/S0022149X09990563
Madanire-Moyo GN, Luus-Powell WJ, Olivier PAS (2012) Diversity of metazoan parasites of the Mozambique tilapia, Oreochromis mossambicus (Peters, 1852), as indicators of pollution in the Limpopo and Olifants River systems. Onderstepoort J Vet Res 79:1–9. doi:10.4102/ojvr.v79i1.362
Madoni P, Romeo MG (2006) Acute toxicity of heavy metals towards freshwater ciliated protists. Environ Pollut 141:1–7. doi:10.1016/j.envpol.2005.08.025
Mahmoud KMA, Abu Taleb HMA (2013) Fresh water snails as bioindicator for some heavy metals in the aquatic environment. Afr J Ecol 51:193–198. doi:10.1111/aje.12019
Malek M, Haseli M, Mobedi I, Ganjali MR, MacKenzie K (2007) Parasites as heavy metal bioindicators in the shark Carcharhinus dussumieri from the Persian Gulf. Parasitology 134:1053–1056. doi:10.1017/S0031182007002508
Marcogliese DJ (2001) Implications of climate change for parasitism of animals in the aquatic environment. Can J Zool 79:1331–1352. doi:10.1139/z01-067
Marcogliese DJ (2004) Parasites: small players with crucial roles in the ecological theater. EcoHealth 1:151–164. doi:10.1007/s10393-004-0028-3
Marcogliese DJ (2005) Parasites of the superorganism: are they indicators of ecosystem health? Int J Parasitol 35:705–716. doi:10.1016/j.ijpara.2005.01.015
Marcogliese DJ (2008) The impact of climate change on the parasites and infectious diseases of aquatic animals. Rev Sci Tech Int Off Epizoot 27:467–484
Marcogliese DJ (2016) The distribution and abundance of parasites in aquatic ecosystems in a changing climate: more than just temperature. Integr Comp Biol 56:611–619. doi:10.1093/icb/icw036
Marcogliese DJ, Pietrock M (2011) Combined effects of parasites and contaminants on animal health: parasites do matter. Trends Parasitol 27:123–130. doi:10.1016/j.pt.2010.11.002
Marcogliese DJ, Nagler JJ, Cyr DG (1998) Effects of exposure to contaminated sediments on the parasite fauna of American plaice (Hippoglossoides platessoides). Bull Environ Contam Toxicol 61:88–95. doi:10.1007/s001289900733
Marcos-López M, Gale P, Oidtmann BC, Peeler EJ (2010) Assessing the impact of climate change on disease emergence in freshwater fish in the United Kingdom. Transbound Emerg Dis 57:293–304. doi:10.1111/j.1865-1682.2010.01150.x
Martins CDMG, Barcarolli IF, de Menezes EJ, Giacomin MM, Wood CM, Bianchini A (2011) Acute toxicity, accumulation and tissue distribution of copper in the blue crab Callinectes sapidus acclimated to different salinities: in vivo and in vitro studies. Aquat Toxicol 101:88–99. doi:10.1016/j.aquatox.2010.09.005
Marx HM (1996) Evaluation of a health assessment index with reference to metal bioaccumulation in Clarias gariepinus and aspects of the biology of the parasite Lamproglena clariae. University of Johannesburg, Johannesburg
Mazhar R, Shazili NA, Harrison FS (2014) Comparative study of the metal accumulation in Hysterothalycium reliquens (nematode) and Paraphilometroides nemipteri (nematode) as compared with their doubly infected host, Nemipterus peronii (notched threadfin bream). Parasitol Res 113:3737–3743. doi:10.1007/s00436-014-4039-x
Mbokane EM, Matla MM, Theron J, Luus-Powell WJ (2015) Seasonal dynamics and occurrences of three Dactylogyrus species on the gills of three cyprinids at Nwanedi – Luphephe dams in Limpopo province, South Africa. Afr Zool 50:119–125. doi:10.1080/15627020.2015.1021175
Mehta A, Shaha C (2006) Mechanism of metalloid-induced death in Leishmania spp.: role of iron, reactive oxygen species, Ca2+, and glutathione. Free Radic Biol Med 40:1857–1868. doi:10.1016/j.freeradbiomed.2006.01.024
Mhlanga W (2000) Mercury in tigerfsh (Hydrocynus vittatus, Castelnau), green happy (Sargochromis codringtonii, Boulenger) and kapenta (Limnothrissa miodon, Boulenger) from Lake Kariba, Zimbabwe. Afr J Ecol 38:224–229
Minguez L, Molloy DP, Guérold F, Giambérini L (2011) Zebra mussel (Dreissena polymorpha) parasites: potentially useful bioindicators of freshwater quality? Water Res 45:665–673. doi:10.1016/j.watres.2010.08.028
Morley NJ, Crane M, Lewis JW (2001) Toxicity of cadmium and zinc to miracidia of Schistosoma mansoni. Parasitology 122(Pt 1):81–85. doi:10.1017/S0031182000007083
Morley NJ, Crane M, Lewis JW (2003) Cadmium toxicity and snail-digenean interactions in a population of Lymnaea spp. J Helminthol 77:49–55. doi:10.1079/JOH2002148
Morley NJ, Crane M, Lewis JW (2004) Influence of cadmium exposure on the incidence of first intermediate host encystment by Echinoparyphium recurvatum cercariae in Lymnaea peregra. J Helminthol 78:329–332. doi:10.1079/JOH2004267
Morley NJ, Crane M, Lewis JW (2005a) Toxicity of cadmium and zinc mixtures to cercarial tail loss in Diplostomum spathaceum (Trematoda:Diplostomidae). Ecotoxicol Environ Saf 60:53–60. doi:10.1016/j.ecoenv.2003.12.018
Morley NJ, Crane M, Lewis JW (2005b) Changes in survival characteristics of Diplostomum spathaceum cercariae emerged from cadmium-exposed Lymnaea stagnalis. J Helminthol 79:55–59. doi:10.1079/JOH2004271
Morley NJ, Costa HH, Lewis JW (2010) Effects of a chemically polluted discharge on the relationship between fecundity and parasitic infections in the chub (Leuciscus cephalus) from a river in southern England. Arch Environ Contam Toxicol 58:783–792. doi:10.1007/s00244-009-9386-8
Morris T, Avenant-Oldewage A, Lamberth S, Reed C (2016) Shark parasites as bio-indicators of metals in two South African embayments. Mar Pollut Bull. doi:10.1016/j.marpolbul.2016.01.027
Nachev M, Sures B (2009) The endohelminth fauna of barbel (Barbus barbus) correlates with water quality of the Danube River in Bulgaria. Parasitology 136:545–552. doi:10.1017/S003118200900571X
Nachev M, Sures B (2016) Seasonal profile of metal accumulation in the acanthocephalan Pomphorhynchus laevis: a valuable tool to study infection dynamics and implications for metal monitoring. Parasit Vectors 9:300. doi:10.1186/s13071-016-1576-4
Nachev M, Zimmermann S, Rigaud T, Sures B (2010) Is metal accumulation in Pomphorhynchus laevis dependent on parasite sex or infrapopulation size? Parasitology 137:1239–1248. doi:10.1017/S0031182010000065
Nomquphu W, Braune E, Mitchell S (2007) The changing water resources monitoring environment in South Africa. S Afr J Sci 103:306–310
Ogut H, Palm HW (2005) Seasonal dynamics of Trichodina spp. on whiting (Merlangius merlangus) in relation to organic pollution on the eastern Black Sea coast of Turkey. Parasitol Res 96:149–153. doi:10.1007/s00436-005-1346-2
Olivier PAS, Luus-Powell WJ, Saayman JE (2009) Report on some monogenean and clinostomid infestations of freshwater fish and waterbird hosts in Middle Letaba Dam, Limpopo Province, South Africa. Onderstepoort J Vet Res 76:187–199
Otachi EO, Körner W, Avenant-Oldewage A, Fellner-Frank C, Jirsa F (2014) Trace elements in sediments, blue spotted tilapia Oreochromis leucostictus (Trewavas, 1933) and its parasite Contracaecum multipapillatum from Lake Naivasha, Kenya, including a comprehensive health risk analysis. Environ Sci Pollut Res 21:7339–7349. doi:10.1007/s11356-014-2602-8
Otachi EO, Szostakowska B, Jirsa F, Fellner-Frank C (2015) Parasite communities of the elongate tigerfish Hydrocynus forskahlii (Cuvier 1819) and redbelly tilapia Tilapia zillii (Gervais 1848) from Lake Turkana, Kenya: influence of host sex and size. Acta Parasitol 60:9–20. doi:10.1515/ap-2015-0002
Oyoo-Okoth E, Admiraal W, Osano O, Hoitinga L, Kraak MHS (2010) Metal specific partitioning in a parasite-host assemblage of the cestode Ligula intestinalis and the cyprinid fish Rastrineobola argentea. Sci Total Environ 408:1557–1562. doi:10.1016/j.scitotenv.2009.11.054
Palm HW, Dobberstein RC (1999) Occurrence of trichodinid ciliates (Peritricha: Urceolariidae) in the Kiel Fjord, Baltic Sea, and its possible use as a biological indicator. Parasitol Res 85:726–732. doi:10.1007/s004360050622
Paperna I (1996) Parasites, infections and diseases of fishes in Africa—an update. CIF Technical Paper. No. 31. Rome
Paul-Pont I, De Montaudouin X, Gonzalez P, Jude F, Raymond N, Paillard C, Baudrimont M (2010a) Interactive effects of metal contamination and pathogenic organisms on the introduced marine bivalve Ruditapes philippinarum in European populations. Environ Pollut 158:3401–3410. doi:10.1016/j.envpol.2010.07.028
Paul-Pont I, Gonzalez P, Baudrimont M, Jude F, Raymond N, Bourrasseau L, Le Goïc N, Haynes F, Legeay A, Paillard C, de Montaudouin X (2010b) Interactive effects of metal contamination and pathogenic organisms on the marine bivalve Cerastoderma edule. Mar Pollut Bull 60:515–525. doi:10.1016/j.marpolbul.2009.11.013
Pech D, Vidal-Martínez VM, Aguirre-Macedo ML, Gold-Bouchot G, Herrera-Silveira J, Zapata-Pérez O, Marcogliese DJ (2009) The checkered puffer (Spheroides testudineus) and its helminths as bioindicators of chemical pollution in Yucatan coastal lagoons. Sci Total Environ 407:2315–2324. doi:10.1016/j.scitotenv.2008.11.054
Pečínková M, Matějusová I, Koubková B, Gelnar M (2005) Classification and occurrence of abnormally developed Paradiplozoon homoion (Monogenea, Diplozoinae) parasitising gudgeon Gobio gobio. Dis Aquat Org 64:63–68. doi:10.3354/dao064063
Pettersen RA, Vøllestad LA, Flodmark LEW, Poléo ABS (2006) Effects of aqueous aluminium on four fish ectoparasites. Sci Total Environ 369:129–138. doi:10.1016/j.scitotenv.2006.05.024
Pietrock M, Meinelt T, Marcogliese DJ (2008) Effects of cadmium exposure on embryogenesis of Stagnicola elodes (Mollusca, Gastropoda): potential consequences for parasite transmission. Arch Environ Contam Toxicol 55:43–48. doi:10.1007/s00244-007-9083-4
Pilecka-Rapacz M, Piasecki W, Czerniawski R, Sługocki Ł, Krepski T, Domagała J (2015) The effect of warm discharge waters of a power plant on the occurrence of parasitic Metazoa in freshwater bream, Abramis brama (L.) Bull Eur Assoc Fish Pathol 35:94–103
Poléo ABS, Schjolden J, Hansen H, Bakke TA, Mo TA, Rosseland BO, Lydersen E (2004) The effect of various metals on Gyrodactylus salaris (Platyhelminthes, Monogenea) infections in Atlantic salmon (Salmo salar). Parasitology 128:169–177. doi:10.1017/S0031182003004396
Poulin R (1995) Phylogeny, ecology, and the richness of parasite communities in vertebrates. Ecol Monogr 65:283–302
Poulin R, Blanar CA, Thieltges DW, Marcogliese DJ (2011) The biogeography of parasitism in sticklebacks: distance, habitat differences and the similarity in parasite occurrence and abundance. Ecography (Cop) 34:540–551. doi:10.1111/j.1600-0587.2010.06826.x
Raymond KMN, Chapman L, Lanciani CA (2006) Host, macrohabitat, and microhabitat specificity in the gill parasite Afrodiplozoon polycotyleus (Monogenea). J Parasitol 92:1211–1217. doi:10.1645/GE-621R.1
Reed C, MacKenzie K, Van der Lingen CD (2012) Parasites of South African sardines, Sardinops sagax, and an assessment of their potential as biological tags. Bull Eur Assoc Fish Pathol 32:41–48
Retief NR, Avenant-Oldewage A, Du Preez H (2006) The use of cestode parasites from the largemouth yellowfish, Labeobarbus kimberleyensis (Gilchrist and Thompson, 1913) in the Vaal Dam, South Africa as indicators of heavy metal bioaccumulation. Phys Chem Earth 31:840–847. doi:10.1016/j.pce.2006.08.004
Retief NR, Avenant-Oldewage A, Du Preez HH (2007) Ecological aspects of the occurrence of Asian tapeworm, Bothriocephalus acheilognathi Yamaguti, 1934 infection in the largemouth yellowfish, Labeobarbus kimberleyensis (Gilchrist and Thompson, 1913) in the Vaal Dam, South Africa. Phys Chem Earth 32:1384–1390. doi:10.1016/j.pce.2007.07.044
Retief NR, Avenant-Oldewage A, Du Preez HH (2009) Seasonal study on Bothriocephalus as indicator of metal pollution in yellowfish, South Africa. Water SA 35:315–322
Richards MP (1997) Trace mineral metabolism in the avian embryo. Poult Sci 76:152–164. doi:10.1093/ps/76.1.152
Riggio M, Trinchella F, Filosa S, Parisi E, Scudiero R (2003) Accumulation of zinc, copper, and metallothionein mRNA in lizard ovary proceeds without a concomitant increase in metallothionein content. Mol Reprod Dev 66:374–382. doi:10.1002/mrd.10365
Riggs MR, Lemly AD, Esch GW (1987) The growth, biomass, and fecundity of Bothriocephalus acheilognathi in a North Carolina cooling reservoir. J Parasitol 73:893–900
Robinson SA, Forbes MR, Hebert CE (2010) Mercury in parasitic nematodes and trematodes and their double-crested cormorant hosts: bioaccumulation in the face of sequestration by nematodes. Sci Total Environ 408:5439–5444. doi:10.1016/j.scitotenv.2010.07.071
Rohde K (1984) Ecology of marine parasites. Helgoländer Meeresun 37:5–33
Rosa IC, Raimundo J, Lopes VM, Brandão C, Couto A, Santos C, Cabecinhas AS, Cereja R, Calado R, Caetano M, Rosa R (2015) Cuttlefish capsule: an effective shield against contaminants in the wild. Chemosphere 135:7–13. doi:10.1016/j.chemosphere.2015.03.050
Roux DJ, van Vliet HR, van Veelen M (1993) Towards integrated water quality monitoring: assessment of ecosystem health. Water SA 19:275–280
Sabo R, Sabová L, Legáth J (2009) The use of parasites as bioindicators of pesticide exposure. Interdiscip Toxicol 2:187–189. doi:10.2478/v10102-009-0015-1
Saliu JK, Akinsanya B, Ukwa UD, Odeozie J, Ganiu Y (2014) Host condition, parasite interaction and metal accumulation in Tilapia guineensis from Iddo area of Lagos lagoon, Nigeria. Iran J Ichthyol 1:289–297
Scheef G, Sures B, Taraschewski H (2000) Cadmium accumulation in Moniliformis moniliformis (Acanthocephala) from experimentally infected rats. Parasitol Res 86:688–691. doi:10.1007/PL00008553
Schintu M, Durante L, Maccioni A, Meloni P, Degetto S, Contu A (2008) Measurement of environmental trace-metal levels in Mediterranean coastal areas with transplanted mussels and DGT techniques. Mar Pollut Bull 57:832–837. doi:10.1016/j.marpolbul.2008.02.038
Schludermann C, Konecny R, Laimgruber S, Lewis JW, Schiemer F, Chovanec A, Sures B (2003) Fish macroparasites as indicators of heavy metal pollution in river sites in Austria. Parasitology 126(Suppl):S61–S69. doi:10.1017/S0031182003003743
Schmidt V, Zander S, Körting W, Steinhagen D (2003) Parasites of the flounder Platichthys flesus (L.) from the German Bight, North Sea, and their potential use in ecosystem monitoring: A. Infection characteristics of potential indicator species. Helgol Mar Res 57:236–251. doi:10.1007/s10152-003-0147-1
Šebelová Š, Kuperman B, Gelnar M (2002) Abnormalities of the attachment clamps of representatives of the family Diplozoidae. J Helminthol 76:249–259. doi:10.1079/JOH2002133
Shah HB, Yousuf AR, Chishti MZ, Ahmad F (2013) Helminth communities of fish as ecological indicators of lake health. Parasitology 140:352–360. doi:10.1017/S0031182012001679
Shinn AP, Gibson DI, Sommerville C (1995) A study of the composition of the sclerites of Gyrodactylus Nordmann, 1832 (Monogenea) using X-ray elemental analysis. Int J Parasitol 25:797–805. doi:10.1016/0020-7519(95)00008-P
Shukla AK, Patra S, Dubey V (2012) Iridoid glucosides from Nyctanthes arbortristis result in increased reactive oxygen species and cellular redox homeostasis imbalance in Leishmania parasite. Eur J Med Chem 54:49–58. doi:10.1016/j.ejmech.2012.04.034
Siddall R, Sures B (1998) Uptake of lead by Pomphorhynchus laevis cystacanths in Gammarus pulex and immature worms in chub (Leuciscus cephalus). Parasitol Res 84:573–577. doi:10.1007/s004360050451
Siddall R, Koskivaara M, Valtonen ET (1997) Dactylogyrus (Monogenea) infections on the gills of roach (Rutilus rutilus L.) experimentally exposed to pulp and paper mill effluent. Parasitology 114:439–446. doi:10.1017/S003118209600875X
Siegel BZ, Siegel SM, Correa T, Dagan C, Galvez G, LeeLoy L, Padua A, Yaeger E (1991) The protection of invertebrates, fish, and vascular plants against inorganic mercury poisoning by sulfur and selenium derivatives. Arch Environ Contam Toxicol 20:241–246. doi:10.1007/BF01055910
Šimková A, Sasal P, Kadlec D, Gelnar M (2001) Water temperature influencing dactylogyrid species communities in roach, Rutilus rutilus, in the Czech Republic. J Helminthol 75:373–383. doi:10.1079/JOH200176
Skinner RH (1982) The interrelation of water quality, gill parasites, and gill pathology of some fishes from South Biscayne Bay, Florida. Fish Bull 80:269–280
Smith GL, Baker TR (2003) Lichens as bioindicators. Sci Scope 27:16–19
Soleng A, Bakke TA (1997) Salinity tolerance of Gyrodactylus salaris (Platyhelminthes, Monogenea): laboratory studies. Can J Fish Aquat Sci 54:1837–1845
Soleng A, Bakke TA, Hansen LP (1998) Potential for dispersal of Gyrodactylus salaris (Platyhelminthes, Monogenea) by sea-running stages of the Atlantic salmon (Salmo salar): field and laboratory studies. Can J Fish Aquat Sci 55:507–514. doi:10.1139/cjfas-55-2-507
Soleng A, Poléo ABS, Alstad NEW, Bakke TA (1999) Aqueous aluminium eliminates Gyrodactylus salaris (Platyhelminthes, Monogenea) infections in Atlantic salmon. Parasitology 119:19–25. doi:10.1017/S0031182099004436
Soleng A, Poléo ABS, Bakke TA (2005) Toxicity of aqueous aluminium to the ectoparasitic monogenean Gyrodactylus salaris. Aquaculture 250:616–620. doi:10.1016/j.aquaculture.2005.05.006
Squadrone S, Prearo M, Brizio P, Gavinelli S, Pellegrino M, Scanzio T, Guarise S, Benedetto A, Abete MC (2013) Heavy metals distribution in muscle, liver, kidney and gill of European catfish (Silurus glanis) from Italian rivers. Chemosphere 90:358–365. doi:10.1016/j.chemosphere.2012.07.028
Sures B (2001) The use of fish parasites as bioindicators of heavy metals in aquatic ecosystems: a review. Aquat Ecol 35:245–255. doi:10.1023/A:1011422310314
Sures B (2003) Accumulation of heavy metals by intestinal helminths in fish: an overview and perspective. Parasitology 126:53–60. doi:10.1017/S003118200300372X
Sures B (2004) Environmental parasitology: relevancy of parasites in monitoring environmental pollution. Trends Parasitol 20:170–177. doi:10.1016/j.pt.2004.01.014
Sures B (2005) Effects of pollution on parasites and use of parasites in pollution monitoring. In: Rohde K (ed) Marine parasitology. CABI Publishing, London, pp 421–425
Sures B (2006) How parasitism and pollution affect the physiological homeostasis of aquatic hosts. J Helminthol 80:151–157. doi:10.1079/JOH2006346
Sures B (2008) Host-parasite interactions in polluted environments. J Fish Biol 73:2133–2142. doi:10.1111/j.1095-8649.2008.02057.x
Sures B, Radszuweit H (2007) Pollution-induced heat shock protein expression in the amphipod Gammarus roeseli is affected by larvae of Polymorphus minutus (Acanthocephala). J Helminthol 81:191–197. doi:10.1017/S0022149X07751465
Sures B, Reimann N (2003) Analysis of trace metals in the Antarctic host-parasite system Notothenia coriiceps and Aspersentis megarhynchus (Acanthocephala) caught at King George Island, South Shetland Islands. Polar Biol 26:680–686. doi:10.1007/s00300-003-0538-4
Sures B, Siddall R (1999) Pomphorhynchus laevis: the intestinal acanthocephalan as a lead sink for its fish host, chub (Leuciscus cephalus). Exp Parasitol 93:66–72. doi:10.1006/expr.1999.4437
Sures B, Siddall R (2001) Comparison between lead accumulation of Pomphorhynchus laevis (Palaeacanthocephala) in the intestine of chub (Leuciscus cephalus) and in the body cavity of goldfish (Carassius auratus auratus). Int J Parasitol 31:669–673
Sures B, Siddall R (2003) Pomphorhynchus laevis (Palaeacanthocephala) in the intestine of chub (Leusciscus cephalus) as an indicator of metal pollution. Int J Parasitol 33:65–70. doi:10.1016/S0020-7519(02)00249-7
Sures B, Taraschewski H (1995) Cadmium concentrations in two adult acanthocephalans, Pomphorhynchus laevis and Acanthocephalus lucii, as compared with their fish hosts and cadmium and lead levels in larvae of A. lucii as compared with their crustacean host. Parasitol Res 81:494–497. doi:10.1007/BF00931792
Sures B, Taraschewski H, Jackwerth E (1994a) Lead content of Paratenuisentis ambiguus (Acanthocephala), Anguillicola crassus (Nematodes) and their host Anguilla anguilla. Dis Aquat Org 19:105–107
Sures B, Taraschewski H, Jackwerth E (1994b) Comparative study of lead accumulation in different organs of perch (Perca fluviatilis) and its intestinal parasite Acanthocephalus lucii. Bull Environ Contam Toxicol 52:269–273. doi:10.1007/BF00198498
Sures B, Taraschewski H, Jackwerth E (1994c) Lead accumulation in Pomphorhynchus laevis and its host. J Parasitol 80:355–357
Sures B, Taraschewski H, Rokicki J (1997a) Lead and cadmium content of two cestodes, Monobothrium wageneri and Bothriocephalus scorpii and their fish hosts. Parasitol Res 83:618–623. doi:10.1007/s004360050307
Sures B, Taraschewski H, Rydlo M (1997b) Intestinal fish parasites as heavy metal bioindicators: a comparison between Acanthocephalus lucii (Palaeacanthocephala) and the zebra mussel, Dreissena polymorpha. Bull Environ Contam Toxicol 59:14–21. doi:10.1007/s001289900437
Sures B, Jürges G, Taraschewski H (1998) Relative concentrations of heavy metals in the parasites Ascaris suum (Nematoda) and Fasciola hepatica (Digenea) and their respective porcine and bovine definitive hosts. Int J Parasitol 28:1173–1178. doi:10.1016/S0020-7519(98)00105-2
Sures B, Siddall R, Taraschewski H (1999) Parasites as accumulation indicators of heavy metal pollution. Parasitol Today 15:16–21. doi:10.1016/S0169-4758(98)01358-1
Sures B, Grube K, Taraschewski H (2002) Experimental studies on the lead accumulation in the cestode Hymenolepis diminuta and its final host, Rattus norvegicus. Ecotoxicology 11:365–368. doi:10.1023/A:1020561406624
Sures B, Scheible T, Bashtar AR, Taraschewski H (2003a) Lead concentrations in Hymenolepis diminuta adults and Taenia taeniaeformis larvae compared to their rat hosts (Rattus norvegicus) sampled from the city of Cairo, Egypt. Parasitology 127:483–487. doi:10.1017/S0031182003003901
Sures B, Dezfuli BS, Krug HF (2003b) The intestinal parasite Pomphorhynchus laevis (Acanthocephala) interferes with the uptake and accumulation of lead (210Pb) in its fish host chub (Leuciscus cephalus). Int J Parasitol 33:1617–1622. doi:10.1016/S0020-7519(03)00251-0
Sures B, Thielen F, Baska F, Messerschmidt J, von Bohlen A (2005) The intestinal parasite Pomphorhynchus laevis as a sensitive accumulation indicator for the platinum group metals Pt, Pd, and Rh. Environ Res 98:83–88. doi:10.1016/j.envres.2004.05.010
Sures B, Nachev M, Selbach C, Marcogliese DJ (2017) Parasite responses to pollution: what we know and where we go in “environmental parasitology”. Parasit Vectors 10:65. doi:10.1186/s13071-017-2001-3
Szefer P, Rokicki J, Frelek K, Skóra K, Malinga M (1998) Bioaccumulation of selected trace elements in lung nematodes, Pseudalius inflexus, of harbor porpoise (Phocoena phocoena) in a Polish zone of the Baltic Sea. Sci Total Environ 220:19–24
Tao S, Liang T, Cao J, Dawson RW, Liu C (1999) Synergistic effect of copper and lead uptake by fish. Ecotoxicol Environ Saf 44:190–195. doi:10.1006/eesa.1999.1822
Tate RB, Husted A (2015) Aquatic macroinvertebrate responses to pollution of the Boesmanspruit River system above Carolina, South Africa. Afr J Aquat Sci 40:153–163. doi:10.2989/16085914.2015.1037237
Taylor JC, Prygiel J, Vosloo A, de la Rey PA, van Rensberg L (2007) Can diatom-based pollution indices be used for biomonitoring in South Africa? A case study of the Crocodile West and Marico water management area. Hydrobiologia 592:455–464. doi:10.1007/s10750-007-0788-1
Tekin-Özan S, Barlas M (2008) Concentrations of selected heavy metals in Ligula intestinalis L., 1758 plerocercoids (Cestoda) compared to it host’s (Tinca tinca L., 1758) organs from Beyşehir Lake (Turkey). Helminthologia 45:76–80. doi:10.2478/s11687-008-0014-3
Tekin-Özan S, Kir I (2005) Comparative study on the accumulation of heavy metals in different organs of tench (Tinca tinca L. 1758) and plerocercoids of its endoparasite Ligula intestinalis. Parasitol Res 97:156–159. doi:10.1007/s00436-005-1412-9
Tellez M, Merchant M (2015) Biomonitoring heavy metal pollution using an aquatic apex predator, the American alligator, and its parasites. PLoS One 10:1–18. doi:10.1371/journal.pone.0142522
Thielen F, Zimmermann S, Baska F, Taraschewski H, Sures B (2004) The intestinal parasite Pomphorhynchus laevis (Acanthocephala) from barbel as a bioindicator for metal pollution in the Danube River near Budapest, Hungary. Environ Pollut 129:421–429. doi:10.1016/j.envpol.2003.11.011
Thoney DA (1990) The effects of trichlorfon, praziquantel and copper sulphate on various stages of the monogenean Benedeniella posterocolpa, a skin parasite of the cownose ray, Rhinoptera bonasus (Mitchill). J Fish Dis 13:385–389. doi:10.1111/j.1365-2761.1990.tb00797.x
Tóro RM, Gessner AAF, Furtado NAJC, Ceccarelli PS, de Albuquerque S, Bastos JK (2003) Activity of the Pinus elliottii resin compounds against Lernaea cyprinacea in vitro. Vet Parasitol 118:143–149
Torres J, Peig J, Eira C, Borrás M (2006) Cadmium and lead concentrations in Skrjabinotaenia lobata (Cestoda: Catenotaeniidae) and in its host, Apodemus sylvaticus (Rodentia: Muridae) in the urban dumping site of Garraf (Spain). Environ Pollut 143:4–8. doi:10.1016/j.envpol.2005.11.012
Torres J, Kacem H, Eira C, Neifar L, Miquel J (2014) Total mercury and selenium concentrations in Sarpa salpa and Balistes capriscus and in their respective digenean endoparasites Robphildollfusium fractum and Neoapocreadium chabaudi from Tunisia. Acta Parasitol 59:580–585. doi:10.2478/s11686-014-0293-4
Tsotetsi AM, Avenant-Oldewage A, Mashego SN (2004) Aspects of the ecology of Lamproglena clariae ( Copepoda: Lernaeidae ) from the Vaal River system, South Africa. J Crustac Biol 24:529–536
Tuuha H, Valtonen ET, Taskinen J (1992) Ergasilid copepods as parasites of perch Perca fluviatilis and roach Rutilus rutilus in central Finland: seasonality, maturity and environmental influence. J Zool 228:405–422
Valtonen ET, Holmes JC, Koskivaara M (1997) Eutrophication, pollution and fragmentation: effects on parasite communities in roach (Rutilus rutilus) and perch (Perca fluviatilis) in four lakes in Central Finland. Can J Fish Aq Sci 54:572–585. doi:10.1139/cjfas-54-3-572
Valtonen ET, Holmes JC, Aronen J, Rautalahti I (2003) Parasite communities as indicators of recovery from pollution: parasites of roach (Rutilus rutilus) and perch (Perca fuviatilis) in central Finland. Parasitology 126(Suppl):S43–S52. doi:10.1017/S0031182003003494
Van der Schyff V, Pieters R, Bouwman H (2016) The heron that laid the golden egg: metals and metalloids in ibis, darter, cormorant, heron, and egret eggs from the Vaal River catchment, South Africa. Environ Monit Assess 188:372. doi:10.1007/s10661-016-5378-0
Vidal-Martínez VM, Pech D, Sures B, Purucker ST, Poulin R (2010) Can parasites really reveal environmental impact? Trends Parasitol 26:44–51. doi:10.1016/j.pt.2009.11.001
Vidal-Martínez VM, Centeno-Chalé OA, Torres-Irineo E, Sánchez-Ávila J, Gold-Bouchot G, Aguirre-Macedo ML (2014) The metazoan parasite communities of the shoal flounder (Syacium gunteri) as bioindicators of chemical contamination in the southern Gulf of Mexico. Parasit Vectors 7:541. doi:10.1186/s13071-014-0541-3
Wah Chu K, Chow KL (2002) Synergistic toxicity of multiple heavy metals is revealed by a biological assay using a nematode and its transgenic derivative. Aquat Toxicol 61:53–64. doi:10.1016/S0166-445X(02)00017-6
Wang W (1987) Factors affecting metal toxicity to (and accumulation by) aquatic organisms—overview. Environ Int 13:437–457
Watson RM, Crafford D, Avenant-Oldewage A (2012) Evaluation of the fish health assessment index in the Olifants River system, South Africa. Afr J Aquat Sci:1–17. doi:10.2989/16085914.2012.677745
Wei K, Yang J (2015) Oxidative damage induced by copper and beta-cypermethrin in gill of the freshwater crayfish Procambarus clarkii. Ecotoxicol Environ Saf 113:446–453. doi:10.1016/j.ecoenv.2014.12.032
Wepener V, van Vuren JH, Chatiza FP, Mbizi Z, Slabbert L, Masola B (2005) Active biomonitoring in freshwater environments: early warning signals from biomarkers in assessing biological effects of diffuse sources of pollutants. Phys Chem Earth 30:751–761. doi:10.1016/j.pce.2005.08.018
Wepener V, van Dyk C, Bervoets L, O’Brien G, Covaci A, Cloete Y (2011) An assessment of the influence of multiple stressors on the Vaal River, South Africa. Phys Chem Earth 36:949–962. doi:10.1016/j.pce.2011.07.075
Weston LF, Reed C, Hendricks M, Winker H, Van der Lingen CD (2015) Stock discrimination of South African sardine (Sardinops sagax) using a digenean parasite biological tag. Fish Res 164:120–129. doi:10.1016/j.fishres.2014.11.002
Williams HH, Mackenzie K (2003) Marine parasites as pollution indicators: an update. Parasitology 126:27–41. doi:10.1017/S0031182003003640
Woelfl S, Mages M, Torres P (2008) Trace metal concentrations in single specimens of the intestinal broad flatworm (Diphyllobothrium latum), compared to their fish host (Oncorhynchus mykiss) measured by total reflection X-ray fluorescence spectrometry. Spectrochim Acta - Part B At Spectrosc 63:1450–1454. doi:10.1016/j.sab.2008.10.009
Yen Nhi TT, Mohd Shazili NA, Shaharom-Harrison F (2013) Use of cestodes as indicator of heavy-metal pollution. Exp Parasitol 133:75–79. doi:10.1016/j.exppara.2012.10.014
Yeomans WE, Chubb JC, Sweeting RA (1997) Use of protozoan communities for pollution monitoring. Parassitologia 39:201–212
Zander CD, Reimer LW (2002) Parasitism at the ecosystem level in the Baltic Sea. Parasitology 124:119–135. doi:10.1017/S0031182002001567
Zargar UR, Chishti MZ, Yousuf AR, Fayaz A (2012a) Infection level of monogenean gill parasite, Diplozoon kashmirensis (Monogenea, Polyopisthocotylea) in the Crucian carp, Carassius carassius from lake ecosystems of an altered water quality: what factors do have an impact on the Diplozoon infection? Vet Parasitol 189:218–226. doi:10.1016/j.vetpar.2012.04.029
Zargar UR, Yousuf AR, Chishti MZ, Ahmed F, Bashir H, Ahmed F (2012b) Effects of water quality and trophic status on helminth infections in the cyprinid fish, Schizothorax niger Heckel, 1838 from three lakes in the Kashmir Himalayas. J Helminthol 86:70–76. doi:10.1017/S0022149X11000071
Zharikova TI (1993) Effect of water pollution on ectoparasites of bream (Abramis brama). J Ichthyol 33:50–62
Zhou Q, Zhang J, Fu J, Shi J, Jiang G (2008) Biomonitoring: an appealing tool for assessment of metal pollution in the aquatic ecosystem. Anal Chim Acta 606:135–150. doi:10.1016/j.aca.2007.11.018
Zimmermann S, Sures B, Taraschewski H (1999) Experimental studies on lead accumulation in the eel-specific endoparasites Anguillicola crassus (Nematoda) and Paratenuisentis ambiguus (Acanthocephala) as compared with their host, Anguilla anguilla. Arch Environ Contam Toxicol 37:190–195. doi:10.1007/s002449900505
Zimmermann S, von Bohlen A, Messerschmidt J, Sures B (2005) Accumulation of the precious metals platinum, palladium and rhodium from automobile catalytic converters in Paratenuisentis ambiguus as compared with its fish host, Anguilla anguilla. J Helminthol 79:85–89. doi:10.1079/JOH2004261
Acknowledgements
The authors extend thanks to the National Research Foundation and the University of Johannesburg for providing funding to AAO.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Gilbert, B.M., Avenant-Oldewage, A. Parasites and pollution: the effectiveness of tiny organisms in assessing the quality of aquatic ecosystems, with a focus on Africa. Environ Sci Pollut Res 24, 18742–18769 (2017). https://doi.org/10.1007/s11356-017-9481-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-9481-8