Abstract
This study presents the distribution of 15 major and trace elements in sediments and fish and their pericardial parasites from Lake Naivasha, Kenya. The lake is one of the few freshwater lakes in the Great Rift Valley and is under strong anthropogenic pressure mainly due to agricultural activities. Its fish provide a valuable protein source for approximately 100,000 people in the area. Fish and their parasites have been acknowledged as indicators of environmental quality due to their accumulation potential for both essential and nonessential trace elements. A total of 34 specimens of the blue spotted tilapia Oreochromis leucostictus and pooled samples of their pericardial parasite, the anisakid nematode Contracaecum multipapillatum (larvae 3), were examined. Element concentrations were determined by inductively coupled plasma-optical emission spectroscopy (ICP-OES) and graphite furnace atomic absorption spectrometry (GF-AAS). The concentrations of elements in the sediments reflected the geology of the area and did not point to pollution: none of the investigated trace elements, including Pb, Cd, Cu, and Zn, showed elevated values. In contrast, concentrations in the fish muscle were elevated for Li, Sr, Cd, and Zn, with high target hazard quotients (THQ > 0.1) indicating a potential health risk to the consumers of this fish. Fish liver showed significantly higher concentrations of the trace elements Fe, Mn, Cd, and Cu compared to the muscle and C. multipapillatum. In the parasite, Zn had the highest concentration, but the worms only minimally accumulated trace elements in relation to their fish host.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Trace elements are natural components of the aquatic environment (Ochieng et al. 2007), but their levels have significantly increased in some areas due to domestic, industrial, mining, and agricultural activities, leading to pollution. In a recent report of the world’s worst pollution problems, heavy metals are among the top threats; they jeopardize the health of tens of millions of people (McCartor and Becker 2010). The report further states that more than 100 million people worldwide are estimated to be at risk from toxic pollution at levels above the international health standards. In Kenya, the need for studies on pollutants in aquatic ecosystems and their biota was recognized more than three decades ago as a result of rapid population expansion, growing industries, and increasing urbanization (Onyari 1981). Since then, several studies have been conducted on trace elements including heavy metals in different lakes in Kenya, among them Lake Naivasha. Tarras-Wahlberg et al. (2002), Ochieng et al. (2007), Kamau et al. (2008), and others have already investigated the levels and sources of heavy metals such as Cu, Cd, Zn, Fe, Pb, and Ni in Lake Naivasha. In addition, Njogu et al. (2011) and Mutia et al. (2012) showed that the most important sources of heavy metal pollution in the Lake Naivasha basin are its major tributary, the Malewa River, and some flower farms surrounding the lake. Therefore, the studies of the latter and other authors on trace elements and heavy metals in the lake have focused on the anthropogenic sources of these metals and concentrations in the muscle of fish. No studies from Lake Naivasha have partitioned trace elements in different fish organ tissues or examined the accumulation of trace elements in fish parasites; trace elements such as Li, Mo, Sr, Rb, and others have also been neglected.
Fish parasites have been recognized for their bioindicative potential regarding pollution in the last decades. Among other properties, certain parasites have shown a high potential for metal accumulation in relation to their fish hosts. Studies have mostly involved endo-helminthes, of which acanthocephalans and cestodes appear to be the best investigated taxa. Among others, Sures (2001, 2003), Jirsa et al. (2008), and Oyoo-Okoth et al. (2010) have shown the impressive ability of those intestinal worms to accumulate heavy metals to a high degree, showing bioaccumulation factors (BAF) up to several thousand for parasites compared to fish muscle, but varying considerably from species to species.
Only a few studies are available on nematodes. In Africa, little research has been done, linking parasitism and pollution. Regarding heavy metal accumulation, Retief et al. (2006) and Oyoo-Okoth et al. (2010) have examined cestodes and their fish hosts from South African river systems.
The blue spotted tilapia Oreochromis leucostictus is a very important commercial fish in Lake Naivasha. It is benthopelagic and occupies the inshore zone, being common in lagoons. It feeds on phytoplankton and detritus. The parasite chosen for this study was an anisakid nematode, Contracaecum multipapillatum (L3), from the pericard of the fish host because it was the only helminth parasite occurring in sufficient numbers and quantity. Anisakid nematodes are widespread and abundant in many regions around the world, including Africa, Europe, Asia, Australia and North and South America (Barson and Marshall 2004). They therefore meet the basic criteria of sentinel organisms. Larval stages of Contracaecum spp. usually occur in the body cavity and mesenteries of fish, whereas the adults inhabit the intestine of piscivorous birds, especially pelicans, cormorants, herons, and darters.
The current study was designed to determine a wide spectrum of trace element concentrations in sediments and in the muscle and liver of the blue spotted tilapia and in its pericardial parasite C. multipapillatum. The aim was to study bioaccumulation in both the fish host and its parasite and therefore gain new insights into this issue. In addition, a comprehensive health risk analysis with parameters adapted to Kenyan realities was undertaken to show possible risks for human fish consumers, including some elements that have been neglected so far in this area. This is a step forward in shedding new light on potential problems arising from fish consumption from Lake Naivasha.
Materials and methods
Study site
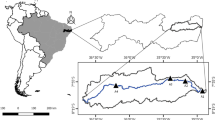
The study was conducted in Lake Naivasha, Kenya between February and August 2011. The lake is situated at 00°45′S and 36°20′E (Kamau et al. 2008) in a closed basin at an altitude of 1,890 m above sea level and covers approximately 160 km2 in the eastern Rift Valley of Kenya (Electronic supplementary material—ESM Fig. 1). It is the only freshwater lake in the Rift Valley without a surface outlet but with a substantial exchange with groundwater (Clarke et al. 1990). It is shallow (approximately 6 m mean depth), with a volume of 4.6 km3. It is bordered by papyrus Cyperus papyrus in some sections, and the overall composition of aquatic macrophytes is in a state of change (Tarras-Wahlberg et al. 2002), probably due to anthropogenic influences such as destruction of littoral vegetation, eutrophication along with plant, and animal introductions. Most of its freshwater inflow (approximately 80–90 %) comes from the Malewa River (Kamau et al. 2008) with an estimated mean annual flow of 153 million m3 and a catchment area of 1,730 km2, followed by the Gilgil River with an estimated average annual flow of 24 million m3 and a catchment area of 420 km2; an additional river, the Karati River, flows only intermittently. The basin area is generally semiarid, receiving a mean annual rainfall of 620 mm, while the mean annual evaporation is estimated at 1,735 mm. Evaporation generally exceeds precipitation throughout the year except at peak rainfall, with the rainfall trend being bimodal with a major peak in April-May and a minor peak in October-November. The water from Lake Naivasha is used extensively for agriculture (horticultural farms: approx. 77 million m3/year), geothermal power generation (approx. 1 million m3/year), domestic water supply, commercial fishing, tourism, and recreation as well as ranching and game farming. The key environmental problems facing the lake are water abstraction, leading to changes in water level, eutrophication, pollution, and invasive species as well as the decline in fish stocks and biodiversity (Harper et al. 2011).
Water and sediment sampling
In situ measurements of the pH, dissolved oxygen, conductivity, and temperature were performed 10 cm below the water surface at the point where the fishing nets were set using a portable Hach Field Case multiparameter meter (Model Multi HQ40d, USA). A total of 10 water samples (about 20 ml each) were obtained biweekly from February to August 2011 from the same depth using a syringe and were immediately filtered with 0.2-μm nylon filters into pre-cleaned (acid washed) high-density polyethylene (HDPE) bottles and acidified with 0.1 mL concentrated nitric acid (TraceSELECT®, Fluka). Similarly, 10 sediment samples were obtained biweekly from February to August 2011 using stainless Ekman grab samplers; caution was taken not to obtain the samples from sediment directly in contact with the surface of the Ekman grab sampler. The samples were placed into 50-mL polypropylene (PP) centrifuge tubes. In the laboratory, samples were first sieved through a 1-mm colander to remove pebbles, and the small fraction (<1 mm) was dried at 105 °C to a constant weight and used for further analyses.
Fish sampling and parasitological examination
In total, 34 fish were caught by fishermen using gill nets of 2.5-in. mesh size (2–4 fish obtained biweekly from February to August 2011). Fish were transported alive in aerated tanks to a laboratory at the Biological Sciences Department, Egerton University, Kenya where they were killed by cervical dislocation and dissected following standard procedures used in parasitological analyses. The fish were weighed and their total length (TL) determined. Where found, C. multipapillatum was collected from the pericardium of the fish; some were preserved in 4 % formaline, others in absolute (95 %) ethanol for further identification purposes, and still others were thoroughly rinsed with double-distilled water and dried in the oven to weight constancy at 60 °C for trace element determinations. Fish tissues were carefully removed using a ceramic knife and plastic tweezers. Approximately 1 g of dorsoventral muscle and the identical mass of liver was washed with double-distilled water and then dried in the same way as parasite samples. The water content of fish muscle was determined by weighing samples before and after drying, yielding a mean of 80 % (n = 10).
Trace elements determination
The water, dried sediment, dried fish tissues, and parasites were transported to the Institute of Inorganic Chemistry, University of Vienna. The sediment samples were first homogenized with mortar and pestle. Samples were then split, with about 1 g weighed into Teflon bombs for acid leaching, using 8 mL of HNO3 34 % (TraceSELECT® Fluka) in a microwave MARS XPRESS system (CEM Corporation), and another 1 g into crucibles for the determination of organic matter (TOC) content using the loss on ignition (LOI) method determined as percent AFDW (ash-free dry weight). After treatment in the microwave oven, samples were transferred quantitatively into 15- or 20-mL flasks and brought to volume with Millipore water. Before measurement, samples were filtered through 0.2-μm PTFE syringe filters (VWR) and, where necessary, diluted.
Fish tissues and parasites (where possible 0.2 g dry weight) were digested in 8 ml of 34 % HNO3 (TraceSELECT® Fluka). Reference samples comprising 0.2 g (dry weight) of fish protein DORM-3 and 0.2 g (dry weight) of marine sediment PACS-2 obtained from the National Research Council Canada (NRCC) were digested and diluted in the same manner as described above for fish tissues and sediments, respectively. To determine the detection limits, analytical blanks were prepared without insertion of a sample. Elements were determined using inductively coupled plasma-optical emission spectrometry (ICP-OES) using an Optima 5300DV (Perkin Elmer). When necessary, graphite furnace atomic absorption spectrometry (GF-AAS) using a PinAAcle 900Z (Pelkin Elmer) was used. The results for the reference samples showed recovery rates between 95 and 104 %, demonstrating the appropriateness of the sample preparation used. A table showing detection limits and recovery rates is given in the ESM Table 1.
Risk assessment
The target hazard quotients (THQ) were determined for the trace elements in order to assess the risk to people who consume O. leucostictus in the area. THQ is the ratio between the potential exposure to a substance and the reference dose (level at which no adverse effects are expected) (USEPA 2012). A THQ ≤ 1 means no significant health risk for fish consumers, and a THQ > 1 means a possible risk regarding the respective metals.
Furthermore, a THQ of 0.1 was later suggested for noncarcinogens to account for additive effects (USEPA 2012). Even though the standard fish equation inputs for fish consumption are given (USEPA 2012), we adapted certain variables in order to reflect the local reality because of the wide discrepancies. For example, the USEPA body weight (BWa) is given as 70 kg, whereas the average body weight in Africa is 60.7 kg (Walpole et al. 2012); the per capita fish consumption in Kenya is 5.2 kg (Lokuruka 2009) which translates to a daily consumption of 14.2 g/day, whereas USEPA (2012) set the value at 54 g/day. The equation for determining THQ according to USEPA (2012) is the following:
where the variables are defined as follows: EFr is the exposure frequency (350 days/year), EDr is the exposure duration (30 years), IRFa is the fish consumption per day (0.0142 kg/day) since the per capita is 5.2 kg/year, in Kenya (Lokuruka 2009), C is the metal concentration in the edible portion of fish (milligrams per kilogram wet weight (ww)), RfDo is the reference dose, oral (milligrams per kilogram per day, according to the updated 2012 Regional Screening Level (RSL) in the fish ingestion table (USEPA 2012), BWa is the body weight, adult 60.7 kg, for Kenya (Walpole et al. 2012), and AT is the averaging time for noncarcinogens (365 days/year). The results for metal concentrations in milligrams per kilogram dry weight were multiplied by 0.2 to refer to wet weight based on the calculated water content of 80 % (see above).
Additionally, the mean concentrations of the trace elements were also compared with FAO/WHO recommended values. In all these comparisons, an average body weight of 60.7 kg (adult) and a daily fish consumption of 14.2 g (5.2 kg per year) were adhered to as defined above.
Statistical analysis
Data were analyzed using Predictive Analytics Software (PASW statistics 18, SPSS). A nonparametric Friedman’s test was used to analyze differences for Si, Al, Fe, Cu, Zn, Mn, Sr, Pb, and Cd because data were not normally distributed. Multiple comparisons were performed using the Wilcoxon’s signed-rank test with a Bonferroni correction of the level of significance (α = 0.05). A Wilcoxon’s signed-rank test was used in analyses for Rb and Mo differences.
Descriptive statistics for Li were also determined. All graphs were drawn using SigmaPlot 10.0. The BAF were determined using the formula:
in which the variables x and y stand for matrices that are compared to each other, such as sediment, fish muscle, fish liver, and parasites.
Results
Physicochemical parameters
A summary of the physicochemical parameters monitored during the study is presented in Table 1.
Characteristics of the fish samples
The average length (TL) of the O. leucostictus sampled was 15.6 ± 0.3 cm (6.7–28.4) and weight 69.0 ± 6.9 g (5.0–418) (mean ± SE). The prevalence of C. multipapillatum was 51.8 %, with a mean intensity of 2.3 nematodes per fish. The parasite was located in the pericardium exclusively.
Trace element concentrations in sediment
The order of concentration of trace elements in sediment was Al > Fe > Mn > Zn > Rb > Sr > Li > Pb > Cu > Cd. Mo was below the detection limit. Most of the trace elements were positively correlated (p < 0.05) with the organic matter content of the sediment, with the exception of Sr (Table 2).
Trace element concentrations in O. leucostictus
The liver had the highest concentration of the trace elements Fe, Mn, Cu, and Cd compared to the muscle and C. multipapillatum (p < 0.05) (Table 3). The concentration was, in descending order, as follows: liver > C. multipapillatum > muscle for Mn, Fe, Cu, and Cd (Fig. 1). Although Al and Si concentrations were not statistically different between the muscle and C. multipapillatum (Z = −0.744, p > 0.05 and Z = −0.180, p > 0.05, respectively), they were both significantly lower than in the liver (p < 0.05). Zinc (Zn) was highest in the muscle compared to the liver and C. multipapillatum (p < 0.05) (Fig. 1). Strontium (Sr) concentration did not different significantly between muscle and liver (Z = −1.222, p > 0.05) but was higher in both than in C. multipapillatum (p < 0.05) (Table 3). Zn and Sr showed the pattern: muscle > liver > C. multipapillatum in a descending order of concentration. Mo was below the detection limit for the muscle but was higher in the liver versus C. multipapillatum (p < 0.05). Li was below the detection limit in liver and in C. multipapillatum. Rb was below the detection limit in C. multipapillatum but was not significantly different between the muscle and liver (Z = −2.009, p > 0.05). The order of trace element concentration in the muscle was Zn > Li > Si > Rb > Fe > Al > Sr > Mn > Cd > Cu > Pb, while that of the liver was Fe > Cu > Si > Zn > Mo > Mn > Rb > Al > Cd > Sr > Pb; in C. multipapillatum, the order was Zn > Fe > Si > Al > Mo > Cd > Mn > Cu > Sr > Pb.
Bioaccumulation factors for trace elements in O. leucostictus
Only a few trace elements showed BAF values >1. These included Li, Cu, Cd, and Zn for the fish tissues and Cd for C. multipapillatum when comparing element concentrations in biota samples to the respective levels in sediment (Table 4).
Target hazard quotients
The THQ values are shown in Table 5. Lithium (Li) and Zn had the highest values among the other elements.
Discussion
Physicochemical parameters of water
The values of the physicochemical parameters agreed well with earlier studies from the lake (Ochieng et al. 2007; Mutia et al. 2012) and did not fluctuate highly during the study period. In comparison to “average fresh water systems,” a particularly high K content was recorded, which implies effects in overall K metabolism and helps explain the relatively high concentrations of Rb in fish, as discussed under Rb.
Trace element concentrations in sediment
Of the analyzed trace elements, the mean concentration of Al was highest in the sediment, followed by Fe, Mn, Zn, and Rb. Table 2 shows comparisons of the mean concentrations in sediments from Lake Naivasha and other studies. Cu, Mn, and Zn levels compared well with earlier studies from the same lake, whereas Cd and Pb levels were lower than those reported by Ochieng et al. (2007) and Mutia et al. (2012). To our knowledge, this is the first report of Li, Rb, and Mo from the lake. Aluminum (Al), Li, Fe, Cu, Rb, Sr, Mo, and Pb in sediment were below the average concentration for shale of sedimentary rocks, which is considered to be the normal background level in the Earth’s crust (Turekian and Wedepohl 1961). These results reflect the lake’s geology. Manganese (Mn) and Cd were within the range of normal background levels, but Zn was elevated compared to the average shale (Turekian and Wedepohl 1961). An earlier study reported higher Cd, Fe, and Zn concentrations in the Lake Naivasha catchment and attributed this to the volcanic rocks or erosion of iron-enriched lateritic soils in the catchment (Tarras-Wahlberg et al. 2002). Cu, Cd, and Pb were below the threshold effect level (TEL) estimated by MacDonald et al. (2000), so no adverse effects are expected; Zn approximated the TEL.
Most trace elements, except Sr, showed a positive significant correlation with the organic matter content of the sediment (Table 2). These findings agree with other studies, e.g., Yang et al. (2010). As Lake Naivasha is considered eutrophic (Kitaka et al. 2002), we expect nitrates to play a role in reducing the formation of Sr-organic matter complexes, which could partly explain the insignificant correlation obtained in this study.
Variation in trace element concentrations in fish tissues
The liver of O. leucostictus had a significantly higher concentration of most trace elements than the muscle. This result reflects the general trend, but there were exceptions. For instance, whereas Zn and Sr concentration were highest in O. leucostictus muscle, Li was below the detection limit in the liver, although highly prevalent in the muscle. This is in agreement with Mohamed and Gad (2008) on Zn but contradicts findings by Uysal et al. (2009), Gül et al. (2011), and others, who found Zn highly concentrated in the liver. Zn is an essential element and fish actively regulate its concentration in the muscle (Hofer et al. 1995) but are only able to do so up to a certain concentration in the water. In contrast, Sr is a biochemical analog of Ca, and its uptake and regulation are through the Ca pump system. Since Ca plays a role in muscle function triggering contraction, the muscle concentration of Sr may be related to that of Ca, hence higher in muscle versus liver.
Each trace element concentration in the fish tissues is briefly discussed below.
Li
We detected Li in high concentration only in O. leucostictus muscle, supporting the findings by Chassard-Bouchaud et al. (1984) on marine fish species, but contrasting with the conclusion by Aral and Vecchio-Sadus (2008) that Li can occur in all organs and tissues. To the best of our knowledge, no comparable literature is available for Li concentration in freshwater fish.
The THQ for Li was 198 (Table 5), which points to a very high risk for fish consumers here. Furthermore, a provisional adult recommended daily intake of 14.3 μg/kg body weight of Li has been suggested by Aral and Vecchio-Sadus (2008). On this basis too, our study found very high concentrations in the edible part of the fish, potentially posing a threat to fish consumers here.
Al
The Al concentration was higher in the liver than the muscle. This agrees with the findings by Moiseenko and Kudryavtseva (2001) in white fish Coregonus lavaretus from the Kola region of Russia, who recorded 4.8 ± 0.6 μg/g dry weight (dw) in the muscle and 19 ± 2.9 μg/g dw in the liver; this area was considered polluted by metals due to mining and other activities. Even higher Al values have been reported by Budambula and Mwachiro (2006) for redeye labeo Labeo cylindricus from the polluted Nairobi River, Kenya (70.0 μg/g ww for the muscle and 150 μg/g ww for the liver).
The THQ for Al was 0.007, i.e., no consumer risk. Additionally, FAO/WHO (2011) reduced the provisional tolerable weekly intake value for Al from 7 to 1 mg/kg body weight/week: even on this basis, our results show no potential risk exposure.
Mn
The Mn concentration was higher in the liver than the muscle. These results generally agree with those of Budambula and Mwachiro (2006) from the Nairobi River. The Mn levels in O. leucostictus muscle compare well with literature values: 1.75 ± 0.05 μg/g dw in white fish C. lavaretus and 2.27 ± 0.5 μg/g in brown trout Salmo trutta from the metal-polluted Kola region (Moiseenko and Kudryavtseva 2001). Lower values include 1.0 ± 0.1 μg/g dw in rainbow trout Oncorhynchus mykiss from the Yesilirmak River in Tokat, Turkey (Mendil et al. 2010). Higher values than we reported were found by Silva and Shimizu (2004): 6.62 μg/g dw in Nile tilapia O. niloticus from a hydropower reservoir in Sri Lanka.
The THQ of Mn was 0.023, i.e., no risk to fish consumers here.
Cu
The Cu concentration was higher in the liver than the muscle, which generally agrees with many findings (Avenant-Oldewage and Marx 2000; Budambula and Mwachiro 2006; Jirsa et al. 2008). This reflects the livers’ important role in storing and detoxifying Cu. The muscle levels we found compare well to those reported for largemouth bass Micropterus salmoides from Lake Naivasha (0.31 ± 0.05 μg/g dw) by Njogu et al. (2011). Lower Cu values but still in the same order of magnitude as in our study have been reported from the same lake: in O. leucostictus (0.27 ± 0.08 μg/g dw) and in common carp Cyprinus carpio 0.28 ± 0.11 μg/g dw (Njogu et al. 2011).
The THQ of Cu was 0.018. FAO/WHO (2011) has set a toxicological guidance value, a provisional maximum tolerable daily intake (PMTDI) of 0.05–0.5 mg/kg body weight. These values exceed the accumulation levels we found for the muscle, demonstrating no risk due to the consumption of this fish.
Zn
In contrast to most trace elements discussed above, the Zn concentration was higher in the muscle than the liver. The reasons have been described above. The muscle levels were tenfold higher than those reported by Mendil et al. (2010) from chub Squalius cephalus (63.5 ± 6.5 μg/g dw). From Kenya, the following authors have reported lower Zn concentrations than we found: Njogu et al. (2011)—7.31 ± 0.89 μg/g ww in O. leucostictus from Lake Naivasha, Budambula and Mwachiro (2006)—70 μg/g ww in the redeye labeo L. cylindricus from Nairobi River, and Mavura and Wangila (2003)—238 μg/g dw in Tilapia grahami from Lake Nakuru.
The THQ of Zn was 2.67, demonstrating a clear risk to fish consumers here.
Rb
Rb was higher in the muscle than the liver, but not significantly different (p > 0.05). In O. leucostictus muscle, we found Rb concentrations comparable to those of Campbell et al. (2005) for several fish species from Lake Erie and two Arctic lakes (Lake Hazen and Resolute Lake). They reported 4.5 ± 0.4 μg/g ww in freshwater drum Aplodinotus grunniens as the highest concentration. Conversely, the concentrations we found are much lower than those reported by Guevara et al. (2006): mean value of 57.6(±5.7) μg/g ww for brown trout S. trutta and 45.9(±6.2) μg/g ww for creole perch Percichthys trucha in lakes of Patagonia, Argentina. Similarly, Silva and Shimizu (2004) reported a range of 20.90–70.75 μg/g dw in the flesh of nine fish species in Sri Lanka. The latter authors also reported 30.10 μg/g dw in Mozambique tilapia O. mossambicus, 21.60 μg/g dw in Nile tilapia O. niloticus, and 70.75 μg/g dw in redbreast tilapia Tilapia rendalli.
We found Rb in both muscle and liver, which agrees partly with the findings of Peters et al. (1999), showing muscle to be the main location of Rb. Note, however, that no literature is available on Rb in fish livers. Although there is some disagreement on the importance of Rb as an ultra-trace essential element to humans and other biota, even small concentrations in combination with other metals such as Pb, Mo, and As can be toxic to fish and other organisms (Yamaguchi et al. 2007). Neither safe concentrations nor the RfDo (reference dose) are known, preventing the determination of the THQ for Rb. Nonetheless, experimental toxic responses linked with elevated Rb have been observed in mammals fed low-K high-Rb diets; these responses are linked to physiological interference with K and Na (Kosla et al. 2002).
Sr
More Sr was present in the muscle than in the liver of O. leucostictus, in agreement with, for example, Moiseenko and Kudryavtseva (2001). Although those authors reported the highest concentrations in the skeleton and gills of white fish C. lavaretus and brown trout S. trutta, the values were notably higher in the muscle versus in the liver, similar to this study. Fish take up Sr and its analog Ca from water, food, and sediment. The uptake is through the Ca transport system located in the chloride cells of gills and enterocytes of the intestine. Sr is accumulated in the bone and other calcareous tissues (Chowdhury and Blust 2002) and is considered a nonessential element in fish. The THQ value for Sr is 0.590, and it could therefore pose a risk (additive effects for noncarcinogens) for fish consumers here.
Mo
In this study, Mo was below the detection limit (0.4 μg/g dw) in O. leucostictus muscle but was detected in the liver. Values for freshwater fish have not been reported before, to our knowledge, but published concentrations from marine fish species never exceed 0.5 mg/kg fresh weight in edible muscle (Eisler 2010). Since Mo was below the detection limit in the muscle, no THQ value was determined.
Cd
The Cd concentration was higher in the liver than in the muscle, which generally agrees with literature, e.g., (Jirsa et al. 2008; Uysal et al. 2009; Wang et al. 2010; Gül et al. 2011). The levels we found compare well with those (0.75 ± 0.03 μg/g dw) reported by Njogu et al. (2011) in largemouth bass M. salmoides from Lake Naivasha. Mutia et al. (2012) reported even higher values (1.59 ± 0.002 mg/kg ww) in C. carpio from Hippo point in Lake Naivasha, and much higher values were reported by Budambula and Mwachiro (2006): 52.0 μg/g in the muscle and 57.0 μg/g in the liver of L. cylindricus from Nairobi River.
Cd is a contaminant in phosphate fertilizers (Tarras-Wahlberg et al. 2002). The intense agricultural activities in the Lake Naivasha catchment area could therefore represent a significant source of easily soluble Cd compounds, which are taken up via the gills of fish and have a very long biological half life (Hofer et al. 1995) and therefore accumulate in all fish species, regardless of their diet.
The THQ value for Cd was close to 1 (0.980), which represents a risk for additive effects. The European Commission regulation set a maximum level of 0.05 mg/kg wet weight for fish consumed by humans (EU 2001). Therefore, based on the THQ value and EU regulation, Cd poses a clear risk to consumers of fish from the lake.
Pb
Our Pb results were unique and unexpected, differing greatly from earlier studies which reported very high levels in various fish species from Lake Naivasha (Njogu et al. 2011; Mutia et al. 2012). The reasons for these differences remain unknown. For example, Njogu et al. (2011) reported a range of 5.12–58.11 mg/kg ww for C. carpio muscle, while Mutia et al. (2012) reported 3.22 ± 0.15, 1.49 ± 0.1, and 1.56 ± 0.19 mg/kg ww for M. salmoides, O. leucostictus, and C. carpio, respectively. The levels reported in our study are safe and below the maximum permissible level according to the European Commission regulation (EU 2001).
Accumulation of trace elements in C. multipapillatum
The trace elements with the highest concentration in C. multipapillatum were Zn, Fe, and Si. Accordingly, C. multipapillatum strongly accumulates certain essential elements. This supports the findings by Nachev et al. (2013) on the nematode Eustrongylides sp.: it accumulates essential metals (Co, Cu, Fe, Se, Zn) better than its host fish, the barbel Barbus barbus. In our study, however, C. multipapillatum had significantly more Pb than O. leucostictus muscle. Also, C. multipapillatum seemed to accumulate more Pb than reported from another nematode, Anguillicola crassus, which infects the swim bladder of eels (Anguilla anguilla) (Sures et al. 1994). The concentrations of Mn, Fe, Cu, and Zn in C. multipapillatum were lower than those reported in the literature on other anisakid nematodes (Table 6).
Compared to the host’s muscle, the concentrations of Fe, Cd, Cu, and Pb were 2.94, 1.58, 1.96, and 7.04 times higher, respectively, in C. multipapillatum. Mo was unique because it was below the detection limit in the muscle, but was detected in the parasite. Barus et al. (2001) found Pb bioaccumulation factors between C. rudolphii tissue and the cormorant Phalacrocorax carbo muscle of 2.35 in male nematodes and 1.81 in females, whereas the Cd values were 0.52 and 0.22 in males and females, respectively—lower than those found in the present study. Overall, these bioaccumulation capacities of C. multipapillatum are much lower compared to acanthocephalans and cestodes (Sures 2003). This points to an inefficient accumulation indicator, supporting the conclusion of Sures (2001) and Barus et al. (2001) that nematodes in general are not useful as accumulation indicators. Exceptions to this have been shown by Azmat et al. (2008), Dural et al. (2011), Morsy et al. (2012), and Nachev et al. (2013). For example, Dural et al. (2011) found that Hysterothylacium aduncum accumulates more Cd, Cu, and Pb than the muscle of its final host, the sea bream Sparus aurata. Interestingly, their findings of no significant differences in the mean Cd and Cu concentrations in H. aduncum and the liver of sea bream corroborate our results.
Three probable factors influence the trace element concentrations of in C. multipapillatum: (1) the parasite’s development stage, (2) parasite location within the host, and (3) parasite feeding and excretion. The parasites obtained from O. leucostictus in our study were larval-stage (L3) nematodes, potentially contributing to the low trace element concentrations. Barus et al. (2001) however, who examined very closely related adult nematodes and their definitive hosts, also found poor bioaccumulation factors. This suggests that other complex factors play a role in influencing trace element uptake by nematodes. We hypothesize that, because C. multipapillatum inhabits the pericardial cavity where it primarily gets nutrition from blood and other host body fluids, the trace element concentrations could be determined by the levels in the immediate muscle tissues. For example, Si and Al levels were not significantly different between the muscle of O. leucostictus and C. multipapillatum. Accordingly, the regulation of these trace elements in C. multipapillatum is probably tightly linked to their concentration in the host fish muscle. Finally, C. multipapillatum has a complete digestive system, setting it apart from cestodes and acanthocephalans, which absorb nutrients through their tegument. Therefore, the uptake, deposition, and excretion of trace elements are different, although, like cestodes and acanthocephalans, nematodes cannot synthesize their own steroids and fatty acids and must ingest sterol precursors from their hosts.
Conclusions
The mean concentration of trace elements in the sediment reflected normal background levels due to the geology of the basin; Zn is the exception. The liver accumulated the highest concentration of most trace elements except Zn, Sr, Rb, and Li. Our findings therefore support most studies that recommend considering the liver of fish when conducting studies on trace elements: it proved to be a good biomarker of most trace elements examined. Although C. multipapillatum showed potential to accumulate trace elements, its values were far lower than in the O. leucostictus liver and comparable to muscle levels. The parasite is therefore not an efficient accumulation bioindicator. Li, Sr, Cd, and Zn had high THQ values and could represent a health risk to the local community that depends on fish for regular food.
References
Aral H, Vecchio-Sadus A (2008) Toxicity of lithium to humans and the environment—a literature review. Ecotoxicol Environ Saf 70(3):349–356
Avenant-Oldewage A, Marx HM (2000) Bioaccumulation of chromium, copper and iron in the organs and tissues of Clarias gariepinus in the Olifants River, Kruger National Park. Water ZA 26:569–582
Azmat R, Fayyaz S, Kazi N, Juanid Mahmood S, Uddin F (2008) Natural bioremediation of heavy metals through nematode parasite of fish. Biotechnology 7(1):139–143
Barson M, Marshall BE (2004) First record of Contracaecum spp. (Nematoda: Anisakidae) in fish-eating birds from Zimbabwe. J S Afr Vet Assoc 75(2):74–78
Barus V, Tenora F, Kracmar S, Prokes M (2001) Cadmium and lead concentrations in Contracaecum rudolphii (Nematoda) and its host, the cormorant Phalacrocorax carbo (Aves). Folia Parasitol (Praha) 48(1):77–78
Budambula NLM, Mwachiro EC (2006) Metal status of Nairobi River waters and their bioaccumulation in Labeo cylindricus. Water Air Soil Pollut 169(1–4):275–291
Campbell LM, Fisk AT, Wang X, Köck G, Muir DCG (2005) Evidence for biomagnification of rubidium in freshwater and marine food webs. Can J Fish Aquat Sci 62(5):1161–1167
Chassard-Bouchaud C, Galle P, Escaig F, Miyawaki M (1984) Bioaccumulation of lithium by marine organisms in European, American, and Asian coastal zones: microanalytic study using secondary ion emission. C R Acad Sci III 299(18):719–724
Chowdhury MJ, Blust R (2002) Bioavailability of waterborne strontium to the common carp, Cyprinus carpio, in complexing environments. Aquat Toxicol 58(3–4):215–227
Clarke MCG, Woodhall DG, Allen D, Darling G (eds) (1990) Geological, volcanic and hydrological controls on the occurrence of geothermal activity in the area surrounding L. Naivasha, Kenya. Ministry of Energy, Nairobi
Dural M, Genc E, Sangun MK, Güner Ö (2011) Accumulation of some heavy metals in Hysterothylacium aduncum (Nematoda) and its host sea bream, Sparus aurata (Sparidae) from North-Eastern Mediterranean Sea (Iskenderun Bay). Environ Monit Assess 174(1–4):147–155. doi:10.1007/s10661-010-1445-0
Eisler R (ed) (2010) Compendium of trace metals and marine biota vol 2. Elsevier, Amsterdam; Boston; Oxford
EU (2001) COMMISSION REGULATION (EC) No 466/2001:setting maximum levels for certain contaminants in foodstuffs
FAO/WHO (2011) Report of the fifth session of the Codex Committee on contaminants in foods. Codex Alimentarius Commission, Rome
Guevara SR, Bubach D, Macchi PJ, Vigliano P, Arribére M, Colombo JC (2006) Rb-Cs ratio as an indicator of fish diet in lakes of the Patagonia, Argentina. Biol Trace Elem Res 111(1–3):97–119
Gül A, Yılmaz M, Benzer S, Taşdemir L (2011) Investigation of zinc, copper, lead and cadmium accumulation in the tissues of Sander lucioperca (L., 1758) living in Hirfanlı Dam Lake, Turkey. Bull Environ Contam Toxicol 87(3):264–266
Harper DM, Morrsison EHJ, Macharia MM, Mavuthi KM (2011) Lake Naivasha, Kenya: ecology, society and future. Freshw Rev 4:89–114
Hofer R, Lackner R, Jeney G, Jeney Z (1995) Fischtoxikologie: Theorie und Praxis. G. Fischer
Jirsa F, Leodolter-Dvorak M, Krachler R, Frank C (2008) Heavy metals in the nase, Chondrostoma nasus (L. 1758), and its intestinal parasite Caryophyllaeus laticeps (Pallas 1781) from Austrian rivers: bioindicative aspects. Arch Environ Contam Toxicol 55(4):619–626
Jirsa F, Gruber M, Stojanovic A, Omondi SO, Mader D, Körner W, Schagerl M (2013) Major and trace element geochemistry of Lake Bogoria and Lake Nakuru, Kenya, during extreme draught. Chem Erde - Geochem 73(275–282)
Kamau JN, Gachanja A, Ngila C, Kazungu JM, Zhai M (2008) Anthropogenic and seasonal influences on the dynamics of selected heavy metals in Lake Naivasha, Kenya. Lakes Reserv: Res Manag 13(2):145–154
Kitaka N, Harper D, Mavuti K (2002) Phosphorus inputs to Lake Naivasha, Kenya, from its catchment and the trophic state of the lake. Hydrobiologia 488(1–3):73–80
Kosla T, Skibniewska E, Debski B, Urbanska-Slomka G (2002) Rubidium in the trophic chain soil-plants-animals. Trace Elem Electrolytes 19(4):171–176
Lokuruka M (2009) Scombrotoxicosis in African fisheries—its implications for international fish trade: an overview. Afr J Food Agric Nutr Dev 9(7):1617–1634
MacDonald DD, Ingersoll CG, Berger TA (2000) Development and evaluation of consensus-based sediment quality guidelines for freshwater ecosystems. Arch Environ Contam Toxicol 39(1):20–31
Mavura WJ, Wangila PT (2003) The pollution status of Lake Nakuru, Kenya: heavy metals and pesticide residues, 1999/2000. Afr J Aquat Sci 28(1):13–18
McCartor A, Becker D (2010) Blacksmith Institute’s world worst pollution problems report 2010: top six toxic threats. Blacksmith Institute. http://www.worstpolluted.org. Accessed 27.07.2013
Mendil D, Ünal ÖF, Tüzen M, Soylak M (2010) Determination of trace metals in different fish species and sediments from the River Yeşilırmak in Tokat, Turkey. Food Chem Toxicol 48(5):1383–1392
Mohamed FAS, Gad NS (2008) Environmental pollution-induced biochemical changes in tissues of Tilpia zilii, Solea vulgaris and Mugil capito from Lake Quarum, Egypt. Global Vet 2(6):327–336
Moiseenko TI, Kudryavtseva LP (2001) Trace metal accumulation and fish pathologies in areas affected by mining and metallurgical enterprises in the Kola Region, Russia. Environ Pollut 114(2):285–297
Morsy K, Bashtar A-R, Abdel-Ghaffar F, Mehlhorn H, Quraishy S, El-Mahdi M, Al-Ghamdi A, Mostafa N (2012) First record of anisakid juveniles (Nematoda) in the European seabass Dicentrarchus labrax (family: Moronidae), and their role as bio-indicators of heavy metal pollution. Parasitol Res 110(3):1131–1138
Mutia TM, Virani MZ, Moturi WN, Muyela B, Mavura WJ, Lalah JO (2012) Copper, lead and cadmium concentrations in surface water, sediment and fish, C. Carpio, samples from Lake Naivasha: effect of recent anthropogenic activities. Environ Earth Sci 67(4):1121–1130
Nachev M, Schertzinger G, Sures B (2013) Comparison of the metal accumulation capacity between the acanthocephalan Pomphorhynchus laevis and larval nematodes of the genus Eustrongylides sp. infecting barbel (Barbus barbus). Parasitol Vectors 6:21
Njogu PM, Keriko JM, Wanjau RN, Kitetu JJ (2011) Distributaion of heavy metals in various lake matrices; water, soil, fish and seminets: a case study of the Lake Naivasha Basin, Kenya. J Agr Sci Technol 13(1)
Ochieng EZ, Lalah JO, Wandiga SO (2007) Analysis of heavy metals in water and surface sediment in five Rift Valley lakes in Kenya for assessment of recent increase in anthropogenic activities. Bull Environ Contam Toxicol 79(5):570–576
Onyari JM The need for aquatic pollutions studies in Kenyan inland waters. In: Workshop of KMFRI on Aquatic Resources of Kenya, Kisumu, Kenya, 1981
Oyoo-Okoth E, Wim A, Osano O, Kraak MHS, Ngure V, Makwali J, Orina PS (2010) Use of the fish endoparasite Ligula intestinalis (L., 1758) in an intermediate cyprinid host (Rastreneobola argentea) for biomonitoring heavy metal contamination in Lake Victoria, Kenya. Lakes Reserv: Res Manag 15(1):63–73
Peters E, Schultz I, Newman M (1999) Rubidium and cesium kinetics and tissue distributions in channel catfish (Ictalurus punctatus). Ecotoxicology 8(4):287–300
Retief NR, Avenant-Oldewage A, du Preez H (2006) The use of cestode parasites from the largemouth yellowfish, Labeobarbus kimberleyensis (Gilchrist and Thompson, 1913) in the Vaal Dam, South Africa as indicators of heavy metal bioaccumulation. Phys Chem Earth, Parts A/B/C 31(15–16):840–847
Silva EIL, Shimizu A (2004) Concentrations of trace metals in the flesh of nine fish species found in a hydropower reservoir in Sri Lanka. Asian Fish Sci 17(4):377–384
Sures B (2001) The use of fish parasites as bioindicators of heavy metals in aquatic ecosystems: a review. Aquat Ecol 35(2):245–255
Sures B (2003) Accumulation of heavy metals by intestinal helminths in fish: an overview and perspective. Parasitology 126(07):53–60
Sures B, Taraschewski H, Jackwerth E (1994) Lead content of Paratenuisentis ambiguus (Acanthocephala), Anguillicola crassus (Nematodes) and their host Anguilla anguilla. Dis Aquat Organ 19(2):105–107
Tarras-Wahlberg H, Everard M, Harper D (2002) Geochemical and physical characteristics of river and lake [2 pt] sediments at Naivasha, Kenya. Hydrobiologia 488(1–3):27–41
Turekian KK, Wedepohl KH (1961) Distribution of the elements in some major units of the Earth’s crust. Geol Soc Am Bull 72(2):175–192
USEPA (2012) Mid-Atlantic Risk Assessment. United States Environmental Protection Agency.http://www.epa.gov/reg3hwmd/risk/human/rb-concentration_table/usersguide.htm. Accessed 07.07.2012
Uysal K, Köse E, Bülbül M, Dönmez M, Erdoğan Y, Koyun M, Ömeroğlu Ç, Özmal F (2009) The comparison of heavy metal accumulation ratios of some fish species in Enne Dame Lake (Kütahya/Turkey). Environ Monit Assess 157(1–4):355–362
Walpole SC, Prieto-Merino D, Edwards P, Cleland J, Stevens G, Roberts I (2012) The weight of nations: an estimation of adult human biomass. BMC Public Health 12:439
Wang Y, Chen P, Cui R, Si W, Zhang Y, Ji W (2010) Heavy metal concentrations in water, sediment, and tissues of two fish species (Triplohysa pappenheimi, Gobio hwanghensis) from the Lanzhou section of the Yellow River, China. Environ Monit Assess 165(1–4):97–102
Yamaguchi S, Miura C, Ito A, Agusa T, Iwata H, Tanabe S, Tuyen BC, Miura T (2007) Effects of lead, molybdenum, rubidium, arsenic and organochlorines on spermatogenesis in fish: monitoring at Mekong Delta area and in vitro experiment. Aquat Toxicol 83(1):43–51
Yang X, Xiong B, Yang M (2010) Relationships among heavy metals and organic matter in sediment cores from Lake Nanhu, an urban lake in Wuhan, China. J Freshw Ecol 25(2):243–249
Acknowledgments
We thank the Austrian Agency for International Cooperation in Education and Research (OeAD-GmbH), Centre for International Cooperation and Mobility (ICM), for funding this study through a scholarship/grant award within the Austrian Partnership Programme in Higher Education and Research for development—APPEAR. We sincerely thank Prof. Simonetta Mattiucci (University of Rome La Sapienza, Italy) for confirming the identification of Contracaecum multipapillatum. We also thank Egerton University for providing laboratory space, facilities, and technical assistance, and The National Council of Science and Technology, Kenya for the issuance of research permit number NCST/RR1/12/1/BS011/46.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Stuart Simpson
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Table 1
(DOCX 18 kb)
ESM Fig 1
(DOCX 272 kb)
Rights and permissions
About this article
Cite this article
Otachi, E.O., Körner, W., Avenant-Oldewage, A. et al. Trace elements in sediments, blue spotted tilapia Oreochromis leucostictus (Trewavas, 1933) and its parasite Contracaecum multipapillatum from Lake Naivasha, Kenya, including a comprehensive health risk analysis. Environ Sci Pollut Res 21, 7339–7349 (2014). https://doi.org/10.1007/s11356-014-2602-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-014-2602-8