Abstract
Perfluorinated compounds (PFCs) have been widely used in industrial and consumer products and frequently detected in many environmental media. Potential reproductive effects of perfluorooctanesulfonate (PFOS), perfluorooctanoic acid (PFOA) and perfluorononanoic acid (PFNA) have been reported in mice, rats and water birds. PFOS and PFOA were also confirmed developing toxicants towards zebrafish embryos; however, the reported effect concentrations were contradictory. Polyfluorinated alkylated phosphate ester surfactants (including FC807) are precursor of PFOS and PFOA; however, there is no published information about the effects of FC807 and PFNA on zebrafish embryos. Therefore, this study was conducted to determine the effects of these four PFCs on zebrafish embryos. Normal fertilized zebrafish embryos were selected to be exposed to several concentrations of PFOA, PFNA, PFOS or FC807 in 24-well cell culture plates. A digital camera was used to image morphological anomalies of embryos with a stereomicroscope. Embryos were observed through matching up to 96-h post-fertilization (hpf) and rates of survival and abnormalities recorded. PFCs caused lethality in a concentration-dependent manner with potential toxicity in the order of PFOS > FC807 > PFNA > PFOA based on 72-h LC50. Forty-eight-hour post-fertilization pericardial edema and 72- or 96-hpf spine crooked malformation were all observed. PFOA, PFNA, PFOS and FC807 all caused structural abnormalities using early stages of development of zebrafish. The PFCs all retarded the development of zebrafish embryos. The toxicity of the PFCs was related to the length of the PFC chain and functional groups.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Perfluorinated compounds and their polyfluorinated precursor compounds (PFCs) have been widely produced and used as surfactants, lubricants, polishes and fire-retardants and primarily to repel both moisture and oil. These uses have resulted in global distribution of stable precursors and transformation products in humans and wildlife (Giesy and Kannan 2002), such as perfluorooctanesulfonate (PFOS) and perfluorooctanoic acid (PFOA) (Giesy and Kannan 2001; Hansen et al. 2001). At present, biomagnifications of PFCs have aroused great concern because increasing concentrations of PFCs have been detected in surface water, sediments and even in wildlife from the Arctic (Butt et al. 2010). Generally, PFOS and PFOA are the most frequently detected PFCs in environment media (Ahrens 2011). The concentrations of PFOS and PFOA are detected mostly at the level of nanograms per liter in lakes, rivers and coastal water (Saito et al. 2004; So et al. 2004; Lu et al. 2011; Zhu et al. 2011). In soils and sediments, the levels of PFOA and PFOS ranged from several nanograms per gram to a few hundred of nanograms per gram dry weight (Sepulvado et al. 2011; Wang et al. 2011). PFCs can enter bodies and adhere to proteins in the blood, liver and muscle, and mostly concentrations in the blood and liver are the highest (Giesy and Kannan 2001). PFOS and PFOA in serum of Amur tiger (Panthera tigris altaica), loggerhead sea turtles (Caretta caretta) and Kemp’s ridley sea turtles (Lepidochelys kempii), and eggs of Herring Gulls (Larus argentatus) and other water birds were in the range of nanograms per milliliter (Gebbink et al. 2009; Keller et al. 2005; Li et al. 2008; Wang et al. 2008). Concentration of PFOS in fish can be up to as great as 612 ng/g dw from estuarine and coastal areas of Korea (Naile et al. 2010) and in blood plasma of bald eagles from the midwestern USA can even be up to 2.57 mg/L (Giesy and Kannan 2001), which was approximately 100,000-fold greater than the concentrations of PFOS in coastal seawaters of Hong Kong, the Pearl River Delta, including the South China Sea, and Korea (So et al. 2004). Perfluorononanoic acid (PFNA) is the largest perfluorinated carboxylic acid surfactant. It was detected in the Pacific and Atlantic Oceans at concentrations that were intermediate between PFOS and PFOA (Yamashita et al. 2005). It has also been detected in blood and liver of wildlife at the level ranging from picograms per liter to nanograms per liter (Calafat et al. 2007; Houde et al. 2005; Moon et al. 2010; Weihe et al. 2008; Yeung et al. 2006). PFOS, PFOA and PFNA can all be detected in the serum of human even in umbilical cord blood at the level of nanograms per milliliter (Monroy et al. 2008; Lien et al. 2011). The highest concentrations of PFOS and PFOA were detected in the blood sera of employees in the fluorochemical manufacturing industry at concentrations up to 12.8 and 114 mg/mL, respectively (Bossi et al. 2005).
Polyfluorinated alkylated phosphate ester surfactants (including FC807) are used in greaseproof food contact paper products (Begley et al. 2005; Trier et al. 2011) and have been found in wastewater, and they are also detected in blood of humans (D’eon et al. 2009). Recently, polyfluorinated dialkylated phosphate ester surfactants (diPAPS) have been found to degrade to form smaller perfluorinated carboxylic acids (PFCAs), including PFOA and PFNA (D’eon and Mabury 2007, 2011). Precursors of PFOS and PFOA have also been detected in waste water (Huset et al. 2008). Thus, FC807 with the similar structure as the other diPAPS is guessed the potential sources of PFNA, PFOA and PFOS.
Because of the persistence, potential to bioaccumulation and global distribution of PFOS and PFOA, there has been interest in their sources and potential to cause toxicity. When exposed to mice, PFOS or PFOA alters the number of circulating neutrophils and enhances the inflammatory responses of macrophages to lipopolysaccharide (LPS) (Qazi et al. 2009). PFOS and PFOA can alter behavior in mice, which is manifested as reduced and/or lack of habituation and hyperactivity (Johansson et al. 2008). The hypothalamus–pituitary–thyroid axis could be disturbed when zebrafish embryos are exposed to PFOS (Shi et al. 2009). PFOA can alter expression of peroxisomal enzymes resulting in greater formation of 8-hydroxydeoxyguanosine in rat liver (Abdellatif et al. 2003). PFOS or PFOA can also induce calcium release from storage sites, which is associated with cytokinesis in the zebrafish embryo (Liu et al. 2011). PFOA can affect mice’s reproduction (Yahia et al. 2010) and causes lesser masses of both genders of Drosophila melanogaster W1118 stock and shorter life span of adult males (Wang et al. 2010). Besides, cell apoptosis in rates can be caused by PFNA (Fang et al. 2010). Survival rate and development were also decreased when prenatal mice were exposed to PFNA (Wolf et al. 2010). These results indicated that PFOS, PFOA and PFNA could cause a number of toxic effects in liver, the nervous system, especially on development and on reproduction. Among PFCs, PFOA and PFOS often had the greatest concentration in the eggs of fish (Gebbink et al. 2009) and in the serum of humans (Bossi et al. 2005) even in umbilical cord blood (Monroy et al. 2008; Lien et al. 2011). Superadded with the potential developmental disrupting potency of PFCs, more attention should be paid to the toxicities of PFCs to fish embryos. Huang et al. reported that PFOS induced cell death at 24 hpf in the brain, eye and tail region of zebrafish embryos and lesions in the muscle fibers with histological examination (Huang et al. 2010). Effects of PFOS and PFOA to zebrafish embryos have been studied by several researchers, but the results were not consistent. The LC50 (96 h) values reported for larvae of zebrafish are lower than 500 mg/L and 71 mg/L for PFOA and PFOS (Ye et al. 2007), but Hagenaars et al. reported that the LC50 (96 h) values were significantly greater than 500 mg/L for PFOA and 58.47 mg/L for PFOS (Hagenaars et al. 2011). There is some information on the toxicity of PFNA to mammals (rats and mice) (Fang et al. 2010; Feng et al. 2010; Wolf et al. 2010), but there was no published available information on toxicity of PFNA and FC807 to aquatic organisms. As an excellent model to study teratogenesis, the zebrafish embryo was used in in ovo study to explore the developmental toxicity of PFOA, PFNA, PFOS and FC807.
Materials and methods
Test species
Adult wild-type zebrafish (Danio rerio) were obtained from Model Animal Research Center of Nanjing University and kept in a semiautomatic rearing system, with five females and ten males in each 10-L tank at 28 ± 1°C. Tap water was treated to remove residual ammonia, chlorine and chloramines, filtered and then treated with UV light to kill microbial pathogens. Water was exchanged at a rate of 1/3 daily. The lighting was 14/10 h light/dark and 1000 lux. Zebrafish were fed frozen blood worms and dry food twice a day. Nylon nets were used at the bottom of each tank to allow eggs to settle and kept from being eaten by the adult fish. Spawning and fertilization took place within 30 min after the lights were turned on in the morning. Eggs were transferred to a Petri dish. Clean embryos were cultured with aerated embryonic rearing water, with the following characteristics: 24.65 mg/L MgSO4·7H2O, 58.8 mg/L CaCl2·2H2O, 1.15 mg/L KCl, 12.5 mg/L NaHCO3 with pH of 8.3 ± 0.2, dissolved oxygen concentration of 6.07 ± 0.24 mg/L (Thermo Scientific Orion 5-Star Plus) at the beginning and end of experiments. The test conformed to the guidelines developed by the Organization for Economic Cooperation and Development (OECD 1996).
Test chemicals
PFOA (C8HF15O2, CAS 335-67-1, 95 %) was provided by TCI (Japan). PFOS (C8HF17O3S, CAS 1763-23-1, 98 %) and PFNA (C9HF17O2, CAS 375-95-1, 97 %) were purchased from Alfa (USA). FC807 (C27H25F34N2O8PS2, molecular weight: 1,246 g/mol, chemical name: Perfluoro alkyl phosphate, trade name: FC807, 80 %) is a commercial product, one of polyfluorinated alkylated phosphate ester surfactants, and was obtained from Hubei Hengxin (China). For FC807, a stock solution was prepared by dissolving the crystals in dimethyl sulfoxide (DMSO) and stored at 4°C. No solvent was used for the other PFCs because of their high water solubility (PFOA, 9.5 g/L; PFOS, 500 mg/L; PFNA, 9.5 g/L). Exposure solutions were diluted from the stock solutions with embryonic water. Concentrations of DMSO in the greatest concentration of exposure solution were less than 0.05 % (v/v).
Experimental design
Embryos were transferred to exposure solutions immediately after fertilization and examined under a stereomicroscope. Damaged or unfertilized embryos were discarded. Zebrafish embryos were exposed in 24-well cell culture plates with 2-mL solution per well. Twenty normally shaped fertilized embryos were assigned to each treatment or control group. In each plate, the remaining four wells were filled with control solution and control eggs. For PFOS, PFOA and PFNA, the control group was embryonic culture water while for FC807, 0.05 % DMSO was included in embryonic water. Before experiment, three to four times range-finding preliminary studies were conducted to determine the effect concentrations of PFOA (0, 150, 200, 212, 225, 240, 255, 270 mg/L), PFNA (0, 6.25, 12.5, 25, 50, 100, 200 mg/L), PFOS (0, 6.25, 12.5, 25, 50, 100, 200 mg/L) and FC807 (0, 25, 44, 50, 100, 132, 200, 400 mg/L). All concentrations were repeated in triple at different days with different batches of eggs. Embryos were cultured in an incubator at 28.5°C after exposure.
Toxicological endpoints included time until hatching, whether eggs were clear or opaque at 4, 8, 12, 24, 48 and 72 hpf, edema at 48 hpf and structural malformations at 72 or 96 hpf (Table 1). Malformations of the spine crooked were defined as scoliosis and curvature of the tail.
Statistical analyses
The proportion of normal embryos in the control group was >80 %. The normality of each sample set was assessed with the Kolomogrov–Smirnov one-sample test before parametric analysis. Then Duncan’s multiple comparisons test was used if appropriate. A Student t-test was used to test the null hypothesis that there was no significant difference between the parallelisms of each treatment. One-way ANOVA was used to test the null hypothesis that there was no significant difference between the mean of each parameter measured in the treated group and the control group. Differences were considered significant if p < 0.05. Probit model or logistic model was used to calculate the EC50/LC50 of the endpoints that appeared in the development stages of zebrafish embryo. The software of Sigma plot 11.0 was used to draw figures.
Results
Developmental toxicity of PFOA
PFOA was acutely toxic to zebrafish embryos, with LC50 values of 262 mg/L PFOA, at 8 and 72 hpf, respectively. The number of opaque embryos did not increase between 8 and 72 hpf. PFOA was more toxic during early development of zebrafish embryos and caused a non-specific lethality. At 8 hpf, the proportions of opaque and deformed embryos were approximately equal, which indicated that the lethality was the primary effect. PFOA can also bring on 48 hpf edema and series malformations after hatching. The 96-hpf EC50 value of spine crooked malformations was 198 mg/L.
A small amount of edema occurred at 48 hpf, and hatching delays and spine crooked malformations occurred at 72 hpf, but these effects occurred only at 200 mg/L or at greater concentrations (Fig. 1). Almost 100 % mortality occurred when exposed to 270 mg/L PFOA. The LOEC based on edema observed at 48 hpf, hatching delay and spine crooked malformations at 72 hpf were 225, 212 and 212 mg/L, respectively. At concentrations greater than these, PFOA caused significant effects relative to the controls. Spine crooked malformation was the most sensitive toxicological endpoint. At 72 hpf, the rate of spine crooked malformations increased until 96 hpf; the rate of spine crooked malformations was significantly different from that of the controls and exhibited a concentration-dependent relationship.
Developmental toxicity of PFNA
Exposure to PFNA caused toxicity at several time points (Table 2). The proportion of opaque embryos was proportional to concentration with an LC50 of 84 mg/L at 72 hpf. PFNA also delayed hatching. The EC50 based on hatching rate at 72 hpf was 214 mg/L. At 8 hpf, some embryos were disintegrating that embryos stopped cleavage and became opaque. There were no significant differences in rates of edema malformations at hatching between the control and treatments.
Developmental toxicity of PFOS
LC50 values at 24, 48 and 72 hpf for PFOS were 69, 68 and 68 mg/L, respectively (Table 3). At this range of concentrations, embryos were disintegrated by 4 hpf. The most sensitive endpoint was spine crooked malformations observed at 72 hpf, with an EC50 of 37 mg/L. The LOEC based on malformations was 12.5 mg/L. PFOS also caused edemas and delayed hatching but no significant difference (Fig. 2). The LOEC based on incidence of edemas at 48 hpf was 50 mg/L, and the LOEC based on hatching delay at 72 hpf was 6.5 mg/L.
Developmental toxicity of FC807
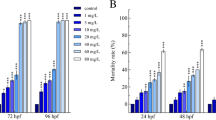
Embryos exposed to FC807 displayed no visible abnormalities from 0 to 48 hpf. The most sensitive endpoint was edema observed at 48 hpf (Table 4). At 48 hpf, 25 mg/L FC807 resulted in a significant difference of 19 % of embryos with pericardial-sac or yolk-sac edema (Fig. 3), relative to the controls. In the group exposed to 44 mg/L FC807, 61 % of embryos were abnormal. The NOEC based on edema at 48 hpf was less than 25 mg/L. The severity of edema was greater with concentrations increasing, and edema ultimately resulted in lethality. The LC50 based on opaqueness at 72 hpf was 211 mg/L.
Discussion
Although the concentrations of PFCs in surface water mostly ranged from several nanograms per liter to several hundreds of nanograms per liter, the level of PFCs in the area of manufacture can be very high. The total PFCs concentrations in surface water samples of discharge following perfluorinated material ranged from <10 to 17,000 μg/L (Moody et al. 2002). A bioaccumulation factor (BAF) range of 6300–125000 was calculated for PFOS based on concentrations in fish liver and surface water (Moody et al. 2002). The concentrations in human blood can even get up to hundreds of milligrams per milliliter (Bossi et al. 2005). So it was significative to study PFCs in milligrams per milliliter range. Zebrafish is considered as a model organism for the study of teratogens in vertebrates (Nagel 2002; Yang et al. 2009). Thus, for comparative purposes the concentrations studied in the study with zebrafish were appropriate.
All the PFCs caused significant effects of development in in ovo experiments, upon which we report here. The physical and chemical properties of PFCs determined that they can be biomagnified (Loi et al. 2011). Assuming that concentrations in our in ovo experiments can be compared with maximal estimated blood concentrations in humans (Bossi et al. 2005), in ovo developmental effects by PFCs would not be expected at current concentrations in the environment.
Generally, the order of potency based on the LC50 value at 72 hpf was PFOS > FC807 > PFNA > PFOA. The length of the perfluorinated tail of PFC molecules has been identified as an important factor in determining toxicity. Based on LC50, EC50, NOEC and LOEC values, PFCs with eight carbons in the PFC tail tend to be more toxic than those with four carbons (Hagenaars et al. 2011; Qin et al. 2010). Hydrophobic properties of PFCs may illustrate this phenomenon well because molecules with longer carbon chains tend to be more hydrophobic than the shorter one. In this study, PFNA, which has one more carbon than PFOA, was more toxic than PFOA.
PFOS is generally more toxic than PFOA (Ye et al. 2007; Hagenaars et al. 2011). In those studies, PFOS was more potent than PFOA and even PFCs with nine carbons in the perfluorinated tail. FC807 with two sulfonate groups was more toxic than PFOA or PFNA. Hagenaars et al. reported that PFCs with a sulfonate group have a larger toxic potential than those with a carboxyl group (Hagenaars et al. 2011). Therefore, the results of this study were consistent with those of the previously conducted studies.
FC807, which contains two PFOS precursor molecules and a phosphoric acid molecule, has been confirmed as a precursor of PFOS. There were no significant differences in the amount of edema between treated and control groups within 48 hpf, but the rates increased significantly from 48 h and embryos became opaque at 72 hpf. Although both PFOS and FC807 have sulfur element, they seemed to have different mechanisms of action. Lethality was the main toxicity of PFOS while pericardial edema and yolk-sac edema were the most severe malformation caused by FC807 (Fig. 3). It was reported that many biochemical and molecular mechanisms occur among cell, tissues and organs during embryogenesis, and a great number of pollutants could specifically influence these mechanisms (Fraysse et al. 2006). Pericardial edema was often considered as the result of heart failure or circulatory failure (Fraysse et al. 2006; Merrill 1946), so FC807 seemed to hurt heart functions significantly.
PFCs especially FC807 can also result in yolk-sac edema at 48 hpf (Fig. 3). Edema rates of PFOS, PFOA and PFNA did not follow a concentration–response relationship, but there was a standard concentration–response relationship in the effect of FC807 (data not shown). In freshwater fishes, there is a water barrier around the membrane to maintain the intracellular hyperosmotic fluids compared to the surrounding water, so a barrier must be maintained in order to minimize water entry and excrete excess water (Hill et al. 2004). FC807 might have the similar effects as TCDD to produce defects in kidney development and/or function and disturb this water permeability barrier and let water in to cells to cause yolk-sac and pericardial edemas (Hill et al. 2004). In this study, FC807 tended to cause more severe edemas than the smaller PFCs. PFOS, PFOA and FC807 can result in spine crooked malformation in hatched larva in a concentration-dependent manner (Fig. 4). Embryos exposed to PFOS showed the most severe acute toxicity that was in accordance with other reports (Hagenaars et al. 2011; Shi et al. 2008).
Conclusions
PFOA, PFNA, PFOS and FC807 were all toxic to zebrafish embryo. All of lethality, 48-hpf edemas and 72-hpf spine crooked malformations occurred throughout the duration of the study. Based on the LC50 value, PFOS was the most potent of the four PFCs, and PFNA was more potent than PFOA. Although all the PFCs tested caused malformations, FC807, with the larger ester molecule, caused more yolk-sac and pericardial-sac edemas than other PFCs. FC807 might more easily disturb the water barrier around the embryos and disturb heart functions to cause edemas.
References
Abdellatif A, Al-Tonsy AH, Awad ME, Roberfroid M, Khan MNU (2003) Peroxisomal enzymes and 8-hydroxydeoxyguanosine in rat liver treated with perfluorooctanoic acid. Dis Markers 19(1):19–25
Ahrens L (2011) Polyfluoroalkyl compounds in the aquatic environment: a review of their occurrence and fate. J Environ Monit 13(1):20–31. doi:i 10.1039/C0em00373e
Begley TH, White K, Honigfort P, Twaroski ML, Neches R, Walker RA (2005) Perfluorochemicals: potential sources of and migration from food packaging. Food Addit Contam 22(10):1023–1031. doi:10.1080/02652030500183474
Bossi R, Riget FF, Dietz R, Sonne C, Fauser P, Dam M, Vorkamp K (2005) Preliminary screening of perfluorooctane sulfonate (PFOS) and other fluorochemicals in fish, birds and marine mammals from Greenland and the Faroe Islands. Environ Pollut 136(2):323–329. doi:10.1016/j.envpol.2004.12.020
Butt CM, Berger U, Bossi R, Tomy GT (2010) Levels and trends of poly- and perfluorinated compounds in the arctic environment. Sci Total Environ 408(15):2936–2965. doi:10.1016/j.scitotenv.2010.03.015
Calafat AM, Wong LY, Kuklenyik Z, Reidy JA, Needham LL (2007) Polyfluoroalkyl chemicals in the US population: data from the National Health and Nutrition Examination Survey (NHANES) 2003–2004 and comparisons with NHANES 1999–2000. Environ Health Perspect 115(11):1596–1602. doi:10.1289/ehp.10598
D’eon JC, Mabury SA (2007) Production of perfluorinated carboxylic acids (PFCAs) from the biotransformation of polyfluoroalkyl phosphate surfictants (PAPS): exploring routes of human contamination. Environ Sci Technol 41(13):4799–4805. doi:10.1021/Es070126x
D’eon JC, Mabury SA (2011) Exploring indirect sources of human exposure to perfluoroalkyl carboxylates (PFCAs): evaluating uptake, elimination, and biotransformation of polyfluoroalkyl phosphate esters (PAPs) in the rat. Environ Health Perspect 119(3):344–350. doi:10.1289/Ehp.1002409
D’eon JC, Crozier PW, Furdui VI, Reiner EJ, Libelo EL, Mabury SA (2009) Observation of a commercial fluorinated material, the polyfluoroalkyl phosphoric acid diesters, in human sera, wastewater treatment plant sludge, and paper fibers. Environ Sci Technol 43(12):4589–4594. doi:10.1021/Es900100d
Fang XM, Feng YX, Wang JS, Dai JY (2010) Perfluorononanoic acid-induced apoptosis in rat spleen involves oxidative stress and the activation of caspase-independent death pathway. Toxicology 267(1–3):54–59. doi:10.1016/j.tox.2009.10.020
Feng YX, Fang XM, Shi ZM, Xu MQ, Dai JY (2010) Effects of PFNA exposure on expression of junction-associated molecules and secretory function in rat Sertoli cells. Reprod Toxicol 30(3):429–437. doi:10.1016/j.reprotox.2010.05.010
Fraysse B, Mons R, Garric J (2006) Development of a zebrafish 4-day embryo-larval bioassay to assess toxicity of chemicals. Ecotoxicol Environ Saf 63(2):253–267. doi:10.1016/j.ecoenv.2004.10.015
Gebbink WA, Hebert CE, Letcher RJ (2009) Perfluorinated carboxylates and sulfonates and precursor compounds in herring gull eggs from colonies spanning the Laurentian Great Lakes of North America. Environ Sci Technol 43(19):7443–7449. doi:10.1021/Es901755q
Giesy JP, Kannan K (2001) Global distribution of perfluorooctane sulfonate in wildlife. Environ Sci Technol 35(7):1339–1342. doi:10.1007/s11356-009-0178-5
Giesy JP, Kannan K (2002) Perfluorochemical surfactants in the environment. Environ Sci Technol 36:146a–152a. doi:10.1021/es022253t
Hagenaars A, Vergauwen L, De Coen W, Knapen D (2011) Structure–activity relationship assessment of four perfluorinated chemicals using a prolonged zebrafish early life stage test. Chemosphere 82(5):764–772. doi:10.1016/j.chemosphere.2010.10.076
Hansen KJ, Clemen LA, Ellefson ME, Johnson HO (2001) Compound-specific, quantitative characterization of organic: fluorochemicals in biological matrices. Environ Sci Technol 35(4):766–770. doi:10.1021/es001489z
Hill AJ, Bello SM, Prasch AL, Peterson RE, Heideman W (2004) Water permeability and TCDD-induced edema in zebrafish early-life stages. Toxicol Sci 78(1):78–87. doi:10.1093/toxsci/kfh056
Houde M, Wells RS, Fair PA, Bossart GD, Hohn AA, Rowles TK, Sweeney JC, Solomon KR, Muir DCG (2005) Polyfluoroalkyl compounds in free-ranging bottlenose dolphins (Tursiops truncatus) from the Gulf of Mexico and the Atlantic Ocean. Environ Sci Technol 39(17):6591–6598. doi:10.1021/es0506556
Huang HH, Huang CJ, Wang LJ, Ye XW, Bai CL, Simonich MT, Tanguay RL, Dong QX (2010) Toxicity, uptake kinetics and behavior assessment in zebrafish embryos following exposure to perfluorooctanesulphonicacid (PFOS). Aquat Toxicol 98(2):139–147. doi:10.1016/j.aquatox.2010.02.003
Huset CA, Chiaia AC, Barofsky DF, Jonkers N, Kohler HPE, Ort C, Giger W, Field JA (2008) Occurrence and mass flows of fluorochemicals in the Glatt Valley watershed, Switzerland. Environ Sci Technol 42(17):6369–6377. doi:10.1021/Es703062f
Johansson N, Fredriksson A, Eriksson P (2008) Neonatal exposure to perfluorooctane sulfonate (PFOS) and perfluorooctanoic acid (PFOA) causes neurobehavioural defects in adult mice. Neurotoxicology 29(1):160–169. doi:10.1016/j.neuro.2007.10.008
Keller JM, Kannan K, Taniyasu S, Yamashita N, Day RD, Arendt MD, Segars AL, Kucklick JR (2005) Perfluorinated compounds in the plasma of loggerhead and Kemp’s ridley sea turtles from the southeastern coast of the United States. Environ Sci Technol 39(23):9101–9108. doi:10.1021/Es050690c
Li XM, Yeung LWY, Taniyasu S, Li M, Zhang HX, Liu D, Lam PKS, Yamashita N, Dai JY (2008) Perfluorooctanesulfonate and related fluorochemicals in the Amur tiger (Panthera tigris altaica) from China. Environ Sci Technol 42(19):7078–7083. doi:10.1021/Es801453d
Lien GW, Wen TW, Hsieh WS, Wu KY, Chen CY, Chen PC (2011) Analysis of perfluorinated chemicals in umbilical cord blood by ultra-high performance liquid chromatography/tandem mass spectrometry. J Chromatogr B 879(9–10):641–646. doi:10.1016/j.jchromb.2011.01.037
Loi EIH, Yeung LWY, Taniyasu S, Lam PKS, Kannan K, Yamashita N (2011) Trophic magnification of poly- and perfluorinated compounds in a subtropical food web. Environ Sci Technol 45(13):5506–5513. doi:10.1021/Es200432n
Lu GH, Yang YL, Taniyasu S, Yeung LWY, Pan J, Zhou BS, Lam PKS, Yamashita N (2011) Potential exposure of perfluorinated compounds to Chinese in Shenyang and Yangtze River Delta areas. Environ Chem 8(4):407–418. doi:10.1071/En10139
Merrill AJ (1946) Edema and decreased renal blood flow in patients with chronic congestive heart failure: evidence of “forward failure” as the primary cause of edema. J Clin Invest 25(3):389–400. doi:10.1172/JCI101720
Monroy R, Morrison K, Teo K, Atkinson S, Kubwabo C, Stewart B, Foster WG (2008) Serum levels of perfluoroalkyl compounds in human maternal and umbilical cord blood samples. Environ Res 108(1):56–62. doi:10.1016/j.envres.2008.06.001
Moody CA, Martin JW, Kwan WC, Muir DCG, Mabury SC (2002) Monitoring perfluorinated surfactants in biota and surface water samples following an accidental release of fire-fighting foam into Etobicoke Creek. Environ Sci Technol 36(4):545–551. doi:10.1021/es011001+
Moon HB, Kannan K, Yun S, An YR, Choi SG, Park JY, Kim ZG, Moon DY, Choi HG (2010) Perfluorinated compounds in minke whales (Balaenoptera acutorostrata) and long-beaked common dolphins (Delphinus capensis) from Korean coastal waters. Mar Pollut Bull 60(7):1130–1135. doi:10.1016/j.marpolbul.2010.04.007
Nagel R (2002) DarT: the embryo test with the zebrafish Danio rerio—a general model in ecotoxicology and toxicology. Altex-Altern Tierexp 19:38–48
Naile JE, Khim JS, Wang TY, Chen CL, Luo W, Kwon BO, Park J, Koh CH, Jones PD, Lu YL, Giesy JP (2010) Perfluorinated compounds in water, sediment, soil and biota from estuarine and coastal areas of Korea. Environ Pollut 158(5):1237–1244. doi:10.1016/j.envpol.2010.01.023
OECD (1996) OECD guidelines for testing chemicals: fish, embryo toxicity test with the zebrafish (B. Rerio). pp 62–76
Qazi MR, Bogdanska J, Butenhoff JL, Nelson BD, DePierre JW, Abedi-Valugerdi M (2009) High-dose, short-term exposure of mice to perfluorooctanesulfonate (PFOS) or perfluorooctanoate (PFOA) affects the number of circulating neutrophils differently, but enhances the inflammatory responses of macrophages to lipopolysaccharide (LPS) in a similar fashion. Toxicology 262(3):207–214. doi:10.1016/j.tox.2009.06.010
Qin PF, Liu RT, Pan XR, Fang XY, Mou Y (2010) Impact of carbon chain length on binding of perfluoroalkyl acids to bovine serum albumin determined by spectroscopic methods. J Agr Food Chem 58(9):5561–5567. doi:10.1021/jf100412q
Saito N, Harada K, Inoue K, Sasaki K, Yoshinaga T, Koizumi A (2004) Perfluorooctanoate and perfluorooctane sulfonate concentrations in surface water in Japan. J Occup Health 46(1):49–59. doi:10.1539/joh.46.49
Sepulvado JG, Blaine AC, Hundal LS, Higgins CP (2011) Occurrence and fate of perfluorochemicals in soil following the land application of municipal biosolids. Environ Sci Technol 45(19):8106–8112. doi:10.1021/Es103903d
Shi XJ, Du YB, Lam PKS, Wu RSS, Zhou BS (2008) Developmental toxicity and alteration of gene expression in zebrafish embryos exposed to PFOS. Toxicol Appl Pharm 230(1):23–32
Shi X, Liu C, Wu G, Zhou B (2009) Waterborne exposure to PFOS causes disruption of the hypothalamus–pituitary–thyroid axis in zebrafish larvae. Chemosphere 77(7):1010–1018. doi:10.1016/j.chemosphere.2009.07.074
So MK, Taniyasu S, Yamashita N, Giesy JP, Zheng J, Fang Z, Im SH, Lam PKS (2004) Perfluorinated compounds in coastal waters of Hong Kong, South China, and Korea. Environ Sci Technol 38(15):4056–4063. doi:10.1021/Es049441z
Trier X, Granby K, Christensen JH (2011) Polyfluorinated surfactants (PFS) in paper and board coatings for food packaging. Environ Sci Pollut Res 18(7):1108–1120. doi:10.1007/s11356-010-0439-3
Wang Y, Yeung LWY, Taniyasu S, Yamashita N, Lam JCW, Lam PKS (2008) Perfluorooctane sulfonate and other fluorochemicals in waterbird eggs from south China. Environ Sci Technol 42(21):8146–8151. doi:10.1021/Es8006386
Wang JS, Li Y, Liu Y, Zhang HX, Dai JY (2010) Disturbance of perfluorooctanoic acid on development and behavior in Drosophila larvae. Environ Toxicol Chem 29(9):2117–2122. doi:10.1002/Etc.237
Wang TY, Chen CL, Naile JE, Khim JS, Giesy JP, Lu YL (2011) Perfluorinated compounds in water, sediment and soil from guanting reservoir, China. B Environ Contam Tox 87(1):74–79. doi:10.1007/s00128-011-0307-y
Weihe P, Kato K, Calafat AM, Nielsen F, Wanigatunga AA, Needham LL, Grandjean P (2008) Serum concentrations of polyfluoroalkyl compounds in Faroese whale meat consumers. Environ Sci Technol 42(16):6291–6295. doi:10.1021/es800695m
Wolf CJ, Zehr RD, Schmid JE, Lau C, Abbott BD (2010) Developmental effects of perfluorononanoic acid in the mouse are dependent on peroxisome proliferator-activated receptor-alpha. PPAR Res. doi:10.1155/2010/282896
Yahia D, Abd El-Nasser M, Abedel-Latif M, Tsukuba C, Yoshida M, Sato I, Tsuda S (2010) Effects of perfluorooctanoic acid (PFOA) exposure to pregnant mice on reproduction. J Toxicol Sci 35(4):527–533
Yamashita N, Kannan K, Taniyasu S, Horii Y, Petrick G, Gamo T (2005) A global survey of perfluorinated acids in oceans. Mar Pollut Bull 51(8–12):658–668. doi:10.1016/j.marpolbul.2005.04.026
Yang LX, Ho NY, Alshut R, Legradi J, Weiss C, Reischl M, Mikut R, Liebel U, Muller F, Strahle U (2009) Zebrafish embryos as models for embryotoxic and teratological effects of chemicals. Reprod Toxicol 28(2):245–253. doi:10.1016/j.reprotox.2009.04.013
Ye L, Wu LL, Zhang CJ, Chen L (2007) Aquatic toxicity of perfluorooctane acid and perfluorooctyl sulfonates to zebrafish embryos. Prog Environ Sci Technol I:134–137
Yeung LWY, So MK, Jiang GB, Taniyasu S, Yamashita N, Song MY, Wu YN, Li JG, Giesy JP, Guruge KS, Lam PKS (2006) Perfluorooctanesulfonate and related fluorochemicals in human blood samples from China. Environ Sci Technol 40(3):715–720. doi:10.1021/es052067y
Zhu LY, Yang LP, Liu ZT (2011) Occurrence and partition of perfluorinated compounds in water and sediment from Liao River and Taihu Lake, China. Chemosphere 83(6):806–814. doi:10.1016/j.chemosphere.2011.02.075
Acknowledgements
This work was jointly funded by the National Natural Science Foundation of China (No. 20737001, 20977047, 21007022) and the Environmental Monitoring Research Foundation of Jiangsu Province (No. 1114). The research was supported by a Discovery Grant from the Natural Science and Engineering Research Council of Canada (Project # 326415-07). The authors wish to acknowledge the support of an instrumentation grant from the Canada Foundation for Infrastructure. Prof. Giesy was supported by the Canada Research Chair program, The Department of Biology and Chemistry and State Key Laboratory in Marine Pollution, City University of Hong Kong, The Einstein Professor Program of the Chinese Academy of Sciences and the Visiting Professor Program of King Saud University.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible editor: Henner Hollert
Rights and permissions
About this article
Cite this article
Zheng, XM., Liu, HL., Shi, W. et al. Effects of perfluorinated compounds on development of zebrafish embryos. Environ Sci Pollut Res 19, 2498–2505 (2012). https://doi.org/10.1007/s11356-012-0977-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-012-0977-y