Abstract
Previous studies have tested the “vernal dam” hypothesis of spring ephemeral herbs in hardwood forests. The desert annual is a component of the desert ecosystem that takes advantage of water resources and temperature conditions during the rainy season to rapidly complete its life cycle within several months. To understand the role desert annual/ephemeral plants play in nutrient flow, we studied vegetation cover, nitrogen content and litter production of annual plants and litter decomposition rate in plant communities dominated by four shrubs (Haloxlon ammodendron, Hedysarum scoparium, Calligonum mongolicum, and Nitraria tangutorum) and two dominant annuals (Agriophyllum squarrosum and Halogeton arachnoideus Moq) in Minqin, northwestern China. Results indicate that over half of the total vegetation cover was provided by annuals. Annuals also took up a large amount of nitrogen (0.46–3.78 g N m−2) along the oasis–desert ecotone. Litter production and nutrient content were higher in areas dominated by annual plants than in areas dominated by shrubs. Furthermore, the litter decomposition rate of the annuals was higher than that of the shrubs, except for the shrub H. ammodendron, although almost all of the litter’s carbon (C) and nitrogen (N) remained after 6 months of decomposition. Without the annuals, more nutrients and rainwater might be lost through leaching or dust transfer caused by the wind erosion. In addition, green twigs of the annuals are the food for some animals, we found some green twigs and litter from annuals left in front of gerbil and rabbit burrows, sometimes even blocking these burrows. Thus, desert summer annuals, like nutrient reservoirs and providers, take up nutrients during the rainy season, providing some animals and microbes with food, and finally release these nutrients after death.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Muller and Bormann (1976) reported that in a hardwood forest ecosystem, the spring ephemeral/annual herb Erythronium americanum could take up nitrogen (N) and potassium (K), and grow rapidly during the period between snowmelt and the leafing-out of the deciduous canopy. The amount of N and K that was taken up was similar to that of the total system loss during that period. Once the canopy closed, the ephemerals senesced and released N and K while trees were clearly active in growth and nutrient uptake. Therefore, the ephemeral herb could act as a short-term sink or a “vernal dam,” leading to a reduction in nutrient loss through its nutrient uptake during an appropriate period and the preservation of the nutrient capital of the ecosystem. Many studies have tested this “vernal dam” hypothesis (Blank et al. 1980; Zak et al. 1990; Rothstein 2000).
Annual plants survive and finish their life cycle during short periods that are favorable for germination and growth. Generally, the phenological characteristics of annuals are strongly related to the climate (temperature and moisture) of desert ecosystem (Beatley 1974; Pan and Huang 1995; Weltzin and Tissue 2003). Desert annual plants are classified as “summer” annuals if they germinate after summer or early autumn rains and as “winter” annuals if they germinate during autumn or early winter rains (Beatley 1974). The initial “vernal dam” studies of annuals were conducted in hardwood forests, rather than desert. Desert ecosystems, with lower nutrient levels and quantity of water, are quite different from hardwood forests. Our previous study showed that desert ephemeral or annuals played an important role in improving soil nutrient content (Chen et al. 2007). We hypothesized that annuals in desert may play a more important role in nutrient conservation than those in temperate hardwood forests.
Most studies of desert annuals were limited to the Chihuahuan Desert (Gutierrez and Whitford 1987; Guo and Brown 1997; Fernández and Reynolds 2000), Mojave Desert (Beatley 1974; Brooks 2000; DeFalco et al. 2001), Sonoran Desert (Halvorson and Patten 1975; Werk et al. 1983; Forseth et al. 1984; Adondakis and Venable 2004), and Negev Desert in Israel (Gutterman 2000), where winter rainfall plays an important role in ephemeral/annual growth, and most of the annuals are spring or winter plants. Moreover, few studies have focused on the role of desert annuals (including both spring/winter annuals and summer annuals) in nutrient conservation (Parker et al. 1984), though annuals play a vital role in soil rehabilitation and production in desert ecosystems due to stabilization of dune surface, prevention of wind erosion, and stability maintenance of desert ecosystems (Wang et al. 2003; Li et al. 2004).
The annuals in the Minqin Desert ecosystem are referred to as summer annuals, since rainfall mainly occurs during the summer and autumn seasons with little or no rainfall during the winter and spring. When the temperature remains extremely high, summer annuals finish their entire life cycles during summer and autumn, which is quite different from the spring/winter annuals (Freas and Kemp 1983). In order to understand the role of summer annuals in nutrient conservation, we investigated vegetation cover, N uptake, litter production and decomposition of four dominant shrubs and two dominant annuals in Minqin, located on the west edge of the Tengger Desert in northwestern China.
Materials and methods
Study site
The study was conducted in Minqin County (38°05′–39°06′N, 103°02′–104°02′E) of Gansu Province, northwestern China, where two large deserts—the Tengger and Badain Jaran—are joined. The county is surrounded by deserts to the east, west and north, and is the location of the Hongyashan Reservoir, which is the largest desert reservoir in Asia. Minqin is downstream of the Shiyang River in the Hexi Corridor, and is located in an inland river basin of China’s arid zone. This corridor experiences sporadic, light precipitation with an annual rainfall of 113 mm, which mainly occurs between May and October. The annual mean temperature is 7.6°C, and the annual evaporation is about 2603.4 mm (Han and Wang 2002). Figure 1 shows the precipitation distribution and monthly temperature in 2002. The soil was typical sand alkaline soil.
The sampling transect and quadrats are located between the Hongyashan Reservoir and the Tengger Desert (Fig. 2). Twenty-one quadrats (10 × 10 m) were established along the transect (from oasis to desert) at 50-m intervals. Quadrat No. 1 was closest to the Reservoir and quadrat No. 21 was closest to the desert.
Vegetation description
In order to protect the Hongyashan Reservoir, a man-made community of Haloxylon ammodendron was established to the east of the reservoir in 1960s. The area close to the reservoir was dominated by Haloxylon ammodendron with the inclusion of less abundant shrubs Hedysarum scoparium and Calligonum mongolicum and some annuals (Agriophyllum squarrosum and Halogeton arachnoideus Moq) interspersed among the shrubs instead of under the shrub canopies. As the distance increased from oasis, more annuals encroached upon the shrubs. The area close to the desert was mainly dominated by the strong xerophil—Nitraria tangutorum (a spiny shrub) which lives in the sand dunes. The annuals Agriophyllum squarrosum and Halogeton arachnoideus Moq were interspersed among the dunes. Airborn sand and dust were intercepted by N. tangutorum and gathered around their stems. Roots emerged from the stems buried by sand, causing N. tangutorum sand dunes to form and develop year by year. Chen et al. (2007) provides a description of the vegetation along the oasis–desert transitional zone in northwestern China.
Vegetation cover and N taken up by annuals
In each quadrat along the sampling transect, vegetation cover was recorded by percentage for all species for July, August and September 2002 separately. Simultaneously, the number of each plant species was counted, and 6–10 individuals of each annual were randomly harvested to determine the mean biomass per plant of the two annuals. They were dried at 60°C to determine dry biomass and subsamples were ground into powder to measure total N concentration. Total nitrogen was measured using a Kjeltec auto 1030 analyser (Tecator, Höganäs, Sweden). Biomass was estimated by multiplying the mean biomass per plant. Then, the uptake of N by the annuals/ephemerals was estimated by multiplying the dry biomass by the concentration of N.
Plant litter
In each quadrat the litter of the six dominant species was collected from randomly selected plants (five plants for each shrub species and 10 for each annual species) in the middle of the transect (quadrats No. 7, 9, 11, and 13) from October to November. Then they were mixed and dried at 60°C to determine the average litter production per plant and N concentration. Most of the annual’s litter was standing, while that of shrubs fell on the ground. The litter from the annuals was collected by hand. Shrub litter was collected using litter traps and plastic placed on the forest floor under the selected shrubs once a week when litter was actually falling. This study only investigated the aboveground litter. The input of nitrogen into soil by litter was calculated by multiplying the total N of the litter and the litter production of each plant. The total N input was the sum of all the dominant plants in each quadrat. Total N was measured with a Kjeltec auto 1030 analyser (Tecator, Höganäs, Sweden). Litter C content was determined using the K2Cr2O7 method (Allen 1989).
The litterbag technique (Bocock and Gilbert 1957) was used to quantify the litter decomposition rate. Nylon-net bags (10 × 10 cm, 1-mm mesh) each containing 5 g of dried litter were randomly buried in interspace soil (at a depth of about 5 cm) in December. The mesh size (1 mm) was large enough to permit aerobic microbial activity and to allow free entry of small soil animals. After 6 months of decomposition, six replicated litterbags of each species were recovered. Litter decomposition rate was expressed by the percentage of mass lost.
Nutrient contents of the litter after decomposition were calculated as follows:
where Clb is the initial C content of the litter, Cla is the C content of the litter after decomposition, Nlb represents the initial N content of the litter, Nla is the N content of the litter after decomposition, M lb is the mass of the litterbags before decomposition, and M la is the litter mass remaining after decomposition.
Statistics
Statistical analyses were conducted using SPSS 11.0 for Windows. Correlation coefficients of litter quality with litter decomposition rate were analyzed. One-Way ANOVA was used to analyze the differences in plant litter decomposition rates and litter C and N concentrations between plants.
Results
Vegetation cover
During the rainy season, the annual cover was much higher than the shrub along the oasis–desert transitional zone in northwest China (Fig. 3). The annual plant cover ranged from 7.5 to 44.5% and accounted for 65.3% of the total vegetation cover along the transect on average. The shrub cover ranged from 1.7 to 23.4%. The total vegetation cover ranged from 9.2 to 48.7%, and showed no discernible trend in large variations along the transect (from quadrat No. 1 to 21). Shrubs dominated the areas close to the oasis, while annuals dominated the areas close to the desert. An analysis of the vegetation cover indicates that the annuals took up the largest proportion along the zone in Minqin.
Nitrogen taken up by annuals
In general, desert annuals complete their life cycle during a rainy season when temperature and moisture are favorable for growth (Mulroy and Rundel 1977). During their life cycles, they have a higher potential to absorb the nutrients needed to complete their lives (Eickmeier and Schussler 1993). Along the transect, the quantity of N taken up by annuals ranged from 0.46 to 3.78 g N m−2, and N uptake was higher in the areas dominated by annuals than in the areas dominated by shrubs (Fig. 4). The highest N uptake 3.78 (g N m−2) appeared in quadrat No. 15, the lowest 0.46 (g N m−2) was found in quadrat No. 12. Additionally, N uptake was tightly related to the biomass of the plant.
Litter production and N return through litter
The litter production of the annuals was much higher than that of the shrubs (Fig. 5a). It was two to threefold greater in the quadrats dominated by the annuals (No. 13–18) than other quadrats. The highest litter dry mass reached 170.7 g m−2 (quadrat No. 15), the lowest was merely 35 g m−2 (quadrat No. 7) (Fig. 5a). The quantity of N (litter total N × litter mass) returned through litter showed similar trends to litter production (Fig. 5b).
Litter decomposition
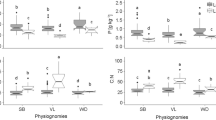
Litter decomposition largely reflects litter nutrient release and the nutrient cycle between plants and soil. Results showed significant differences in litter decomposition and C and N concentration among the six plants (Table 1). After decomposing for 6 months, the highest levels of remaining C and N were found in C. alaschanicum. The lowest levels of remaining C were found in A. squarrosum and the lowest remaining N was found in N. tangutorumr (Table 2). Nitrogen remains were much higher than C remains in the litter, which was likely due to the higher N concentration in litter after decomposition compared to levels before decomposition (Tables 1 and 2). This finding indicates that most nutrients remained in the litter after 6 months’ decomposition (from December to May), which might be favorable for nutrient conservation in desert ecosystems.
Discussion
Generally speaking, desert soils are poorly developed and low in nutrients compared with soils of other ecosystems. Nitrogen is considered the second most limiting factor after water for arid land productivity (Ettershank et al. 1978; Fisher et al. 1988). Additionally, the canopy is unclosed in desert ecosystems. In the deserts of northwestern China, annuals germinate during summer and autumn since the constant and frequent rainfall provides them with a prerequisite to live, and a part will survive and complete their reproduction in autumn. Most summer annuals have a high-photosynthetic efficiency at increased temperatures when water is available (Mulroy and Rundel 1977).
In this study, the annuals uptake of N ranged from 0.46 to 3.78 g N m−2, which is much higher than the net N uptake by the spring ephemeral/annual herb Erythronium americanum (0.1 g m−2, equal to 1.0 kg ha−1) in hardwood forests. This finding suggests that desert annuals play a more important role in retaining nutrients than those in hardwood forests. Rothstein (2000) tested the “venal dam” hypothesis and found that in hardwood forests soil microorganisms, instead of annuals, played a key role in N cycling. In hardwood forests, summer-green tree species accounted for 94% of the aboveground net production, while the annuals accounted for less than 5% of the aboveground production (Muller and Bormann 1976). In the present study, the summer desert annuals accounted for over half of the vegetation cover along the transect (Fig. 3). It was reported that the mean coverage of ephemeral plants reached 13.9% during seasons that experience low rainfall (Spring), and 40.2% during rainy seasons (Summer: May to June) at Gurbantunggut Desert in Xinjiang. At this location, ephemeral plants acted as the major contributor to dune surface stabilization by reducing nutrient loss through wind erosion in the desert of northwestern China (Wang et al. 2003). Thus, it can be seen that desert annuals play a vital role in N cycling and conservation, mainly due to the mass of annuals per square meter rather than their potential nutrient uptake.
At our experimental site, it was also observed that germination and death of the annuals occurred frequently, suggesting that incessant events of annual germination and death also changed the microhabitat. Our previous study showed that soil nutrients beneath the annual plants were higher than in the adjacent bare soil, suggesting that annuals have the potential to improve the nutrient level of the soil (Chen et al. 2007). Thus, without annuals, more bare soil might appear in the region, which may lead to higher nutrient loss through wind erosion (Prospero and Lamb 2003), and NH3 volatilization, nitrification and denitrification (Peterjohn and Schlesinger 1990; Evans and Ehleringer 1993; Zaady et al. 1996; Evans and Belnap 1999; Schaeffer et al. 2003).
In our experimental site, several summer annuals were scattered among shrubs and large covered areas. It should be noted that the timing of the annuals’ growth season was in the range of the shrubs’ growth season. In general, shrubs leafed out in May, and senesced in October, while the annuals germinated between May and June and senesced in September. Nutrient competition might occur between them, although it would not be severe since most of the annuals grow among the shrubs rather than under them. Regardless of whether the shrubs competed for the nutrients against the summer annuals or not, the existing and living annuals in the desert per se fixed a part of the soil nutrient (especially N) and atmospheric C and N (Figs. 3 and 4), though the annuals might not be the dominant sink during that season. The annuals act as nutrient conservers or resource sinks and play an important role in desert ecosystems.
Water, a limiting resource for plant growth in the desert, mainly comes from rain. The annual plant community is, therefore, dependent on rainwater (Went 1949; Gutterman 2000). The omnipresent annuals will absorb rainwater and grow after an appropriate rainfall, even if some of them die before completing their life cycles. During summer and autumn, which have large and frequent rainfall pulses (Fig. 1b), rainwater is not completely taken up within the short-time period, leading to water and nutrient loss caused by run off.
By studying American deserts, Walvoord et al. (2003) demonstrated that substantial quantities of N, in the form of nitrate, were leached and accumulated beneath the soil zone over millennial periods, indicating that nitrate leaching occurred in deserts. However, the existence of annuals partly reduces such nutrient leaching loss. Moreover, annuals undoubtedly enhance the efficiency of rainwater use in desert ecosystems via reduction of evaporation and, consequently, reduction of nitrogen loss with rainwater since we observed nitrate and ammonium levels of 0.78 and 0.63 mg l−1, respectively, in the rainwater at our study site. Therefore, the summer annuals could be considered as a “resource-reserve” coupled with water-pulse, acting as a nutrient container and a carbon assimilator. Meanwhile, they partly reduce nutrient loss and water flow in desert ecosystems.
Litter also played an important role as a nutrient regulator in the desert. When the annuals died, nutrients (C, N, etc.) retained in their stand litter and dead roots (Table 1) were transformed or released into the soil during decomposition by microbes and animals. Results showed that the rate of annual plant litter decomposition was higher than that of the shrubs, except H. ammodendron, due to the narrow C–N ratio in litter (Table 1). This result indicates that plants in a desert ecosystem have a high nutrient input because of the higher litter chemical quality, expressed as narrow C–N nutrient ratios (Parker et al. 1984). Furthermore, the lower C–N ratio in litter might contribute to the nutrient release from litter (Singh et al. 1999). However, our results indicated that after decomposing for 6 months most C and N remained in the litter (Table 2), and the remains might be used in the upcoming growth season.
In addition, desert annuals provide some small animals with food. We observed that some green twigs and annual litter was left in front of some gerbil and rabbit burrows, even blocking the burrows in some cases. The rapid growth of desert summer annuals represents an important pulse in soil nutrient supply (Mulroy and Rundel 1977; Parker et al. 1984), and plays an important role in desert productivity (Halvorson and Patten 1975). Hence, summer annuals provide some microbes and animals with high-quality food in desert ecosystems. Simultaneously, the released nutrients could be used for plant growth in the next season.
Without annuals, more nutrients and rainwater might be lost through leaching or dust transfer caused by the wind erosion. In this case, the desert ecosystem would be an unimaginable landscape, resulting in waste of rainwater, loss of soil nutrients, and finally aggravated desertification (Martínez-mena et al. 2002). Desert annuals, like a nutrient dam and protector, take up nutrients during the rainy seasons, reduce nutrient loss caused by leaching and wind erosion, and provide some animals and microbes with food. In the future, more attention should be paid to the community function of desert annual plants.
References
Adondakis S, Venable DL (2004) Dormancy and germination in a guild of Sonoran Desert annuals. Ecology 85:2582–2590. doi:10.1890/03-0587
Allen SE (1989) Chemical analysis of ecological materials. Blackwell, Oxford
Beatley JC (1974) Phenological events and their environmental triggers in Mojave Desert ecosystems. Ecology 55:856–863. doi:10.2307/1934421
Blank JL, Olson RK, Vitousek PM (1980) Nutrient uptake by a diverse spring annual community. Oecologia 47:96–98. doi:10.1007/BF00541781
Bocock KL, Gilbert O (1957) The disappearance of leaf litter under different woodland conditions. Plant Soil 9:179–185. doi:10.1007/BF01398924
Brooks ML (2000) Competition between alien annual grasses and native annual plants in the Mojave Desert. Am Midl Nat 144:92–108. doi:10.1674/0003-0031(2000)144[0092:CBAAGA]2.0.CO;2
Chen BM, Wang GX, Cheng DL, Deng JM, Peng SL (2007) Vegetation change and soil nutrient distribution along an oasis–desert transitional zone in northwestern China. J Integr Plant Biol 49(11):1537–1547. doi:10.1111/j.1774-7909.2007.00531.x
DeFalco LA, Detling JK, Tracy CR, Warren SD (2001) Physiological variation among native and exotic winter annual plants associated with microbiotic crusts in the Mojave Desert. Plant Soil 234:1–14. doi:10.1023/A:1010323001006
Eickmeier WG, Schussler EE (1993) Responses of the spring ephemeral Claytonia virginica L. to light and nutrient manipulations and implications for the “vernal-dam” hypothesis. Bull Torrey Bot Club 120(2):157–165. doi:10.2307/2996945
Ettershank G, Ettershank J, Bryant M, Whitford WG (1978) Effects of nitrogen fertilization on primary production in a Chihuahuan Desert ecosystem. J Arid Environ 1:135–139
Evans RD, Belnap J (1999) Long-term consequences of disturbance on nitrogen dynamics in an arid ecosystem. Ecology 80:150–160
Evans RD, Ehleringer JR (1993) A break in the nitrogen cycle in arid lands? Evidence from δ15N of soils. Oecologia 94:314–317. doi:10.1007/BF00317104
Fernández RJ, Reynolds JF (2000) Potential growth and drought tolerance of eight desert grasses: lack of a trade-off? Oecologia 123:90–98. doi:10.1007/s004420050993
Fisher FM, Zak JC, Cunningham GL, Whitford WG (1988) Water and nitrogen effects on growth allocation patterns of creosotebush in the northern Chihuahuan Desert. J Range Manage 41:387–391. doi:10.2307/3899572
Forseth IN, Ehleringer JR, Werk KS, Cook CS (1984) Field water relations of Sonoran Desert annuals. Ecology 65:1436–1444. doi:10.2307/1939124
Freas KE, Kemp PR (1983) Some relationships between environmental reliability and seed dormancy in desert annual plants. J Ecol 71:211–217. doi:10.2307/2259973
Guo Q, Brown JH (1997) Interactions between winter and summer annuals in the Chihuahuan Desert. Oecologia 111:123–128. doi:10.1007/s004420050215
Gutierrez JR, Whitford WG (1987) Chihuahuan desert annuals: importance of water and nitrogen. Ecology 68:2032–2045. doi:10.2307/1939894
Gutterman Y (2000) Environment factors and survival strategies of annual plant species in the Negev Desert, Israel. Plant Species Biol 15:113–125. doi:10.1046/j.1442-1984.2000.00032.x
Halvorson WL, Patten DT (1975) Productivity and flowering of winter ephemerals in relation to Sonoran Desert shrubs. Am Midl Nat 93:311–319. doi:10.2307/2424164
Han FG, Wang J (2002) The ecological climate resources and its exploitation along the edge of Minqin oasis. J Arid Land Resour Environ 16(3):51–56
Li XR, Xiao HL, Zhang JG, Wang XP (2004) Long-term ecosystem effects of sand-binding vegetation in Shapotou region of Tengger Desert, Northern China. Restor Ecol 12:376–390. doi:10.1111/j.1061-2971.2004.00313.x
Martínez-mena M, Rogel JA, Castillo V, Albaladejo J (2002) Organic carbon and nitrogen losses influenced by vegetation removal in a semiarid mediterranean soil. Biogeochemistry 61:309–321. doi:10.1023/A:1020257208048
Muller RN, Bormann FH (1976) Role of Erythronium americanum Kerin energy flow and nutrient dynamics of a northern hardwood forest ecosystem. Science 193:1126–1128. doi:10.1126/science.193.4258.1126
Mulroy TW, Rundel PW (1977) Annual plants: adaptations to desert environments. Bioscience 27:109–114. doi:10.2307/1297607
Pan WB, Huang PY (1995) The ecology of four ephemeral plants. Acta Phytoecol Sin 19:85–91
Parker LW, Santos PF, Phillips J, Whitford WG (1984) Carbon and nitrogen dynamics during the decomposition of litter and roots of a Chihuahuan Desert annual Lepidium lasiocarpum. Ecol Monogr 54:339–360. doi:10.2307/1942501
Peterjohn WT, Schlesinger WH (1990) Nitrogen loss form deserts in the southwestern United States. Biogeochemistry 10:67–79. doi:10.1007/BF00000893
Prospero JM, Lamb PJ (2003) African droughts and dust transport to the Caribbean: climate change implications. Science 302:1024–1027. doi:10.1126/science.1089915
Rothstein DE (2000) Spring ephemeral herbs and nitrogen cycling in a northern hardwood forest: an experimental test of the vernal dam hypothesis. Oecologia 124:446–453. doi:10.1007/PL00008870
Schaeffer SM, Billings SA, Evans RD (2003) Responses of soil nitrogen dynamics in a Mojave Desert ecosystem to manipulations in soil carbon and nitrogen availability. Oecologia 134:547–553
Singh KP, Singh PK, Tripathi SK (1999) Litterfall, litter decomposition and nutrient release patterns in four native tree species raised on coal mine spoil at Singrauli, India. Biol Fertil Soils 29:371–378. doi:10.1007/s003740050567
Walvoord MA, Phillips FM, Stonestrom DA, Evans RD, Hartsough PC, Newman BD, Striegl RG (2003) A reservoir of nitrate beneath desert soils. Science 302:1021–1024. doi:10.1126/science.1086435
Wang X, Jiang J, Lei J, Zhang W, Qian Y (2003) Distribution of ephemeral plants and their significance in dune stabilization in Gurbantunggut Desert. J Geogr Sci 13(3):323–330. doi:10.1007/BF02837507
Weltzin JF, Tissue DT (2003) Resource pulses in arid environments—patterns of rain, patterns of life. New Phytol 157:171–173. doi:10.1046/j.1469-8137.2003.00672.x
Went FW (1949) Ecology of desert plants II. The effects of rain and temperature on germination and growth. Ecology 30:1–13. doi:10.2307/1932273
Werk KS, Ehleringer JR, Forseth IN, Cook CS (1983) Photosynthetic characteristics of Sonoran Desert winter annuals. Oecologia 59:101–105. doi:10.1007/BF00388081
Zaady E, Groffman PM, Shachak M (1996) Litter as a regulator of N and C dynamics in macrophytic patches in Negev Desert soils. Soil Biol Biochem 28:39–46. doi:10.1016/0038-0717(95)00097-6
Zak DR, Groffman PM, Pregitzer KS, Christensen S, Tiedje JM (1990) The vernal dam: plant-microbe competition for nitrogen in northern hardwood forests. Ecology 71:651–656. doi:10.2307/1940319
Acknowledgments
The study was supported by the Open Fund of State Key Laboratory of Biocontrol (SKLBC08K02) of Sun Yat-Sen University and National Natural Science Foundation of China (30730020, U0633002). We are grateful to the colleagues of Lanzhou University and the colleagues of Minqin Desert and Experiment Station of Gansu Desert Control Research Institute who contributed to the completion of this research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Bao-Ming Chen and Gen-Xuan Wang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Chen, BM., Wang, GX. & Peng, SL. Role of desert annuals in nutrient flow in arid area of Northwestern China: a nutrient reservoir and provider. Plant Ecol 201, 401–409 (2009). https://doi.org/10.1007/s11258-008-9526-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-008-9526-7