Abstract
Ferriferous savannas, also known as cangas in Brazil, are nutrient-impoverished ecosystems adapted to seasonal droughts. These ecosystems support distinctive vegetation physiognomies and high plant diversity, although little is known about how nutrient and water availability shape these ecosystems. Our study was carried out in the cangas from Carajás, eastern Amazonia, Brazil. To investigate the N cycling and drought adaptations of different canga physiognomies and compare the findings with those from other ecosystems, we analyzed nutrient concentrations and isotope ratios (δ13C and δ15N) of plants, litter, and soils from 36 plots distributed in three physiognomies: typical scrubland (SB), Vellozia scrubland (VL), and woodland (WD). Foliar δ15N values in cangas were higher than those in savannas but lower than those in tropical forests, indicating more conservative N cycles in Amazonian cangas than in forests. The lower δ15N in savanna formations may be due to a higher importance of mycorrhizal species in savanna vegetation than in canga vegetation. Elevated δ13C values indicate higher water shortage in canga ecosystems than in forests. Foliar and litter nutrient concentrations vary among canga physiognomies, indicating differences in nutrient dynamics. Lower nutrient availability, higher C:N ratios, and lower δ15N values characterize VL, whereas WD is delineated by lower δ13C values and higher soil P. These results suggest lower water restriction and lower P limitation in WD, whereas VL shows more conserved N cycles due to lower nutrient availability. Differences in nutrient and water dynamics among physiognomies indicate different ecological processes; thus, the conservation of all physiognomies is required to ensure the maintenance of functional diversity in this unique ecosystem.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Metalliferous savannas, also known as cangas in Brazil, are considered hotspots of plant diversity around the world (e.g., Gibson et al. 2012; Viana et al. 2016). Cangas are characterized by severe environmental conditions, such as high daily temperatures and radiation exposure; nutrient-impoverished, shallow, acidic soils with high metal contents (especially iron); and a low water-retention capacity (Jacobi and Carmo 2008a, b; Poot and Lambers 2008). Distinct vegetation physiognomies (Mota et al. 2015) are expected to emerge along environmental gradients of nutrient and water availability (Schaefer et al. 2016). Despite their economic importance associated with their covering of the world’s largest iron ore deposits (Skirycz et al. 2014), little is known about how nutrient availability and low water retention shape these unique ecosystems (Carmo et al. 2012; Rezende et al. 2013).
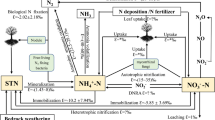
Nitrogen (N) and phosphorus (P) are the key elements that limit plant productivity in most terrestrial environments (Elser et al. 2007; Thomas et al. 2015). Thus, N cycling, which involves multiple transformations and interactions with other chemical elements, affects nearly every aspect of ecosystem functioning (Vitousek et al. 1997). For instance, increased N input and availability affect ecosystem productivity and biological diversity (Bobbink et al. 2010; Bustamante et al. 2012) as well as the rate of N loss and pathways of N cycling (Kozovits et al. 2007; Craine et al. 2007; Janssens et al. 2010). N-rich ecosystems are typically highly productive, with N circulating at high rates and concentrations (Martinelli et al. 1999; Matson and Vitousek 1987; Nardoto et al. 2008), which enable these systems to lose substantial amounts of N (Houlton et al. 2006; Hedin et al. 2009). In contrast, plants in nutrient-poor ecosystems have developed various strategies for more effective nutrient acquisition (Zemunik et al. 2015), such as the exploitation of different forms of available nutrients, atmospheric N fixation by rhizobia and colonization by mycorrhizae to facilitate soil N uptake, especially the uptake of N–NH4 (Bukovská et al. 2018; Camenzind et al. 2018). These strategies result in more conservative N cycling than observed in N-rich ecosystems, with low N leaching and volatilization in dystrophic environments (Pardo and Nadelhoffer 2010; Galloway et al. 2004). Understanding N cycling, especially in ecosystems with low nutrient availability, such as Amazonian cangas, is thus an important issue (Canfield et al. 2010; Bernhardt and Schlesinger 2013).
Because many nitrogen-transforming (bio-)chemical processes, such as nitrification or denitrification, fractionate against the heavier 15N isotope, soil and especially vegetation δ15N values reflect variation in N cycling at short time scales, making soil and vegetation δ15N valuable tools to investigate terrestrial N cycling (Robinson 2001; Bustamante et al. 2004) because fractionation is lower in dystrophic ecosystems than in nutrient-rich ecosystems (Houlton et al. 2006; Mardegan et al. 2009). Higher δ15N values in both soil and vegetation generally indicate higher availability and/or higher inputs of N in nutrient-rich ecosystems, generally accompanied by higher N losses, and higher rates of microbial processes that discriminate against 15N in the soil organic pool (Högberg et al. 2011). In contrast, more conserved N cycles, characterized by lower N losses and therefore lower δ15N values, occur in ecosystems of low nutrient availability. Therefore, lower δ15N values are expected in cangas, where nutrient availability is lower than it is in other tropical ecosystems. Furthermore, canga physiognomies with lower nutrient availability are expected to be characterized by lower δ15N values than are emergent physiognomies in soils with greater nutrient availability. Nevertheless, the interpretation of δ15N values is not straightforward, because several factors influence the δ15N values in plant tissues (Craine et al. 2015), such as the forms of absorbed N, soil depth (Asada et al. 2005), plant associations with mutualists such as N-fixing rhizobia or mycorrhizae (He et al. 2014; Nardoto et al. 2008; Hobbie and Hobbie 2008; Meira-Neto et al. 2018) and other processes related to N assimilation, resorption, and retranslocation (Evans 2001).
Foliar δ13C values reflect the biophysical processes related to diffusional limitations during the photosynthetic process (Escudero et al. 2008; Scarano et al. 2001), which is affected by changes in metabolism and environmental conditions (Heaton et al. 1997; Lloyd et al. 2009), including light intensity and water constraints. The principal differences in foliar δ13C values are caused by differences in photosynthetic pathways; C4 plants are characterized by significantly higher δ13C values than are C3 plants. Furthermore, variation in foliar δ13C values within C3 plants occurs due to variation in water availability (Mârtensson et al. 2017), as C3 plants suffering from drought events reduce their extent of water loss by stomatal closure to achieve higher water-use efficiency (e.g., Werner and Máguas 2010), resulting in increased foliar δ13C values (Alcântara et al. 2015; Silveira et al. 2016). Thus, differences among foliar δ13C values allow the water shortage in vegetation to be inferred (Martinelli et al. 2007). Due to adaptations to periodic droughts, canga vegetation is expected to present higher δ13C values than are evergreen ecosystems. Among physiognomies, formations characterized by low water shortage are expected to show lower δ13C values (Steward et al. 1995).
In this study, we analyzed the nutrient contents and isotope ratios (N and C) of soils, leaves, and litter from three distinct canga physiognomies in Carajás Mineral Province, eastern Amazonia, Brazil, to obtain insights into nutrient availability and water dynamics in these canga physiognomies. We aimed to understand (i) whether there are differences in soil properties and soil nutrient availability among the three canga physiognomies, (ii) whether nutrient concentrations in vegetation and litter differ among physiognomies and (iii) whether leaf or litter nutrient concentrations depend on nutrient availability in canga soils. We furthermore investigated whether nitrogen and carbon isotope ratios from canga ecosystems differ (iv) from those from nearby ecosystems, (v) among physiognomies or (vi) among ecosystemic compartments such as soil, litter, or vegetation, and we investigated (vi) whether nitrogen isotopic ratios in canga vegetation depend on soil nutrient availability or leaf nutrient concentrations.
Materials and methods
Study area
This study was carried out in two canga plateaus in Carajás Mineral Province, Canaã dos Carajás, Pará, Brazil (Figure S1 in Electronic Supplementary Material). Canga plateaus are restricted to higher regions of the landscape (700–900 m a.s.l.) and are surrounded by evergreen dense or evergreen open Amazonian forest. The typical climate of this region is tropical, with dry winters and rainy summers (Aw in Köppen’s climate classification, Alvares et al. 2013). The accumulated annual rainfall ranges from 1800 to 2300 mm, and the rainy season comprises approximately 80% of the annual precipitation. The average monthly temperature varies between 19 and 31 °C.
The physiognomies of canga vegetation include grasslands, scrublands, wetlands, and forest formations (Mota et al. 2015). Three canga physiognomies were sampled in this study (Figure S2). Scrubland physiognomies occur above continuous canga crusts and lack a continuous soil layer. In typical scrubland (SB), which contain species such as Mimosa acutistipula var. ferrea (Fabaceae), Bauhinia pulchella (Fabaceae), Byrsonima spicata (Malpighiaceae), Pouteria ramiflora (Sapotaceae), and Guapira opposita (Nyctaginaceae), plant occurrence is associated with the accumulation of soil and organic matter along fractures and within pockets of the parent rock. In contrast, Vellozia scrubland (VL), dominated by Vellozia glochidea (Velloziaceae), Norantea guianensis (Marcgraviaceae), Cereus hexagonus (Cactaceae), and Anemopaegma scabriusculum (Bignoniaceae), grows on bare parent rock material. In woodland (WD), large (up to 15 m tall) phanerophytes of different species form a closed canopy. WDs are small, isolated patches inserted into a matrix composed of shrubby or gramineous canga formations, and they are characterized by deeper soil (30–100 cm, Schaefer et al. 2016) than in SB and VL.
Sample collection and analysis
Nine 300 m transects were delimited; along the transects, 2 × 4 m plots were established every 100 m, resulting in four plots per transect. In each plot, soil, litter, and leaves were sampled. Composite samples of deformed soil (0–5 cm) were collected. Accumulated leaf litter was sampled using a 50 × 50 cm template, and the leaves of all plants taller than 1 m were collected.
The soil samples were air-dried and sieved using 2 mm mesh. The pH was determined in a 1:2.5 soil-to-water ratio, and organic matter (OM) was measured by the potassium dichromate (K2Cr2O7) method. Available P, K, B, Zn, Fe, Mn, and Cu were determined by the Mehlich−1 method (0.05 mol L−1 HCl, 0.0125 mol L−1 H2SO4). Exchangeable Ca, Mg, and Al were measured by AAS with 1 M KCl extracts added to lanthanum oxide (Embrapa 2009).
The litter and leaf samples were oven-dried at 60 °C to constant weight before being crushed in a Willye mill to obtain a fine powder, which was then submitted to acidic digestion in a solution of 4.0 mL of concentrated HNO3 and 2.0 mL of concentrated HClO4, first at 120 °C for 1 h and then at 220 °C until HClO4 fumes were observed. Then, the K, Ca, Mg, S, B, Cu, Fe, Mn, and Zn concentrations were determined with an atomic absorption spectrophotometer, and the total P concentration was determined by using a spectrophotometer to measure the phosphomolybdenum complex colorimetrically at 680 nm (Embrapa 2009).
To determine the isotope ratios (δ13C and δ15N), 1–2 mg subsamples of homogenized plant material (litter and leaves) and 150 mg of soil were sealed in a tin capsule. The C and N concentrations and their isotope ratios were measured by using dry combustion in a Finnigan Delta Plus isotope ratio mass spectrometer (IRMS Delta Plus, Finnigan Mat, San Jose, California, USA) linked to a Carlo Erba elemental analyzer (CHN-1110) at the Isotopic Ecology Laboratory at the Centro de Energia Nuclear na Agricultura (CENA, São Paulo, Brazil). The C and N concentrations were expressed as percentages (%) with analytical errors of 0.3 and 0.1%, respectively. For the isotope ratios, we used delta notation (δ13C and δ15N) on a parts-per-thousand basis (‰); the analytical error for each ratio did not surpass 0.3‰. We used Pee Dee Belemnite (PDB) limestone as the standard for δ13C and atmospheric air as the standard for δ15N. Sugarcane leaves (N = 0.88%, C = 43.82%, δ15N = 5.4‰, δ13C = 12.7‰) were used as the internal standard reference.
The mean foliar δ15N and δ13C values found in this study were compared with values available in the literature from similar studies carried out in other Brazilian ecosystems. To identify relevant literature, we searched the Google Scholar, SciELO, and Scopus databases for scientific articles using different combinations of the keywords ‘nitrogen,’ ‘carbon,’ ‘stable isotope,’ ‘nutrient cycling,’ ‘water stress,’ and ‘tropical forest.’ The location of the study sites (Brazil) was used as a refinement criterion for this search.
Statistical analysis
All analyses were performed in the R environment, version R 3.2.4 (R Development Core Team 2018). To determine the differences in soil, litter, and leaf concentrations among the different physiognomies, we carried out principal component analysis (PCA) followed by PERMANOVA. For these analyses, the original data were scaled.
To detect differences in nutrient concentrations and isotope ratios in soils and litter among the different physiognomies, we used ANOVA after evaluating the data for normality and homoscedasticity. Mixed effect models were used to detect differences in leaf nutrient concentrations and isotope ratios among the different physiognomies. In these models, plot was included as a random factor because leaf nutrient concentrations and isotope ratios from different plants growing in the same plot may not be independent (i.e., may be pseudoreplicates). Maximum likelihood was considered for construction of the mixed effect models based on the distributions of the data and analysis of the residuals. Similarly, mixed effect models were used to test for relationships between soil and leaf nutrient concentrations. Differences in leaf isotopic ratios between canga and other neotropical ecosystems were evaluated by the analysis of 95% confidence intervals.
Results
Nutrient availability in the canga physiognomies
In general, the canga soils were very acidic (soil pH < 4.5), with high organic matter contents, low nutrient availabilities (except K), and elevated concentrations of available Al and Fe (Table 1). The first two axes of the PCA of all soil variables explained 51.7% of the total variance in soil properties and showed a clear separation of VL from the other physiognomies (F = 3.40, df = 2, p = 0.001) (Figure S3A). In general, the VL physiognomy was associated with nutrient-poor soils (lowest contents of Ca, Mg, Al, B, Zn, and Mn), whereas WD showed the highest P and Al contents and the lowest Fe content (Table 1).
Nutrient contents and the C:N and N:P ratios in the leaves and litter differed among the canga physiognomies (Figs. 1, 2). The PCA revealed significant differences among physiognomies in leaves (F = 2.42, df = 2, p = 0.005) and litter (F = 4.36, df = 2, p = 0.001), and the first two components explained 49% of the variance in leaf nutrient concentrations and 58.5% of the variance in litter nutrient concentrations (Figure S3B and C). Both diagrams indicate that the VL leaves and litter were characterized by higher C contents (data not shown) and higher C:N and N:P ratios than were the leaves from the other physiognomies (see also Fig. 1). Furthermore, foliar N and P were lowest in VL; the highest foliar N content was detected in WD (Fig. 1).
Boxplots of nitrogen and phosphorus concentrations and elemental ratios in the litter and leaves of three different canga physiognomies from Carajás Mineral Province, Canaã dos Carajás, Pará, Brazil. Different letters indicate significant differences among physiognomies and compartments (soil, litter and leaves) according to mixed effect models. SB typical scrubland, VL Vellozia scrubland, and WD woodland
Boxplots of macro- and micro-nutrient contents in litter and leaves from three different canga physiognomies from Carajás Mineral Province, Canaã dos Carajás, Pará, Brazil. Different letters indicate significant differences among physiognomies according to mixed effect models. SB typical scrubland, VL Vellozia scrubland, and WD woodland
K and Ca concentrations are higher in the leaves from WD than in the leaves from the other physiognomies, but the Mg values found in WD leaves are lower than those from SB leaves (Fig. 2). Furthermore, Zn and Fe values from WD leaves were lower than those from VL leaves. The concentrations of the remaining leaf nutrients show no differences among the physiognomies. In litter, K, S, and B concentrations are lower than their corresponding leaf concentrations, whereas Zn and Fe show the opposite pattern. K, Ca, Mg, S, and B concentrations from WD litter are higher than those from the other physiognomies (Fig. 2). Soil and leaf P, Ca, and Mg concentrations correlate significantly (Table S1).
Isotopic characterization of leaves, litter and soils
The mean litter and foliar δ15N values were 0.8‰ (with values ranging from − 2.5‰ to 3.3‰) and 1.4‰ (− 5.2‰ to 7.9‰), respectively. The foliar δ15N values were significantly lower than those of tropical forests but higher than those from neotropical (sandy) savanna vegetation (Table 2). The mean soil δ15N value was 4.4‰, with values ranging from 1.5‰ to 6.0‰, and was higher than the mean δ15N values in leaves and litter in all physiognomies (Fig. 3). Furthermore, δ15N values varied significantly among the canga physiognomies. VL showed lower δ15N values than did WD or SB in all compartments (Fig. 3). Moreover, foliar δ15N values correlated positively with N foliar contents (Fig. 4).
Boxplots of δ15N-values in soil, litter and leaves of three different canga physiognomies from Carajás Mineral Province, Canaã dos Carajás, Pará, Brazil. Different letters indicate significant differences among physiognomies and compartments (soil, litter, and leaves) according to mixed effect models. SB typical scrubland, VL Vellozia scrubland, and WD woodland
The average foliar δ13C of canga plants was − 28.3‰, with values ranging from − 32.3‰ to − 23.2‰. Thus, the mean values were higher than the measures from tropical forests (Table 2). Compared with canga vegetations, neotropical savannas present similar or slightly lower δ13C values. Significant differences among canga physiognomies were detected (Fig. 5). WD presented lower foliar δ13C values than did SB and VL (Fig. 5); similar 13C depletion was observed in the soil and litter (Figure S4).
Boxplots of foliar δ13C values (upper panel) and a histogram of relative frequency of foliar δ13C values (lower panel) of canga plants from three different physiognomies from Carajás Mineral Province, Canaã dos Carajás, Pará, Brazil. Different letters indicate significant differences among physiognomies according to a mixed effect model. SB typical scrubland, VL Vellozia scrubland, and WD woodland
Discussion
Nutrient dynamics differ among canga physiognomies
Low soil nutrient availability, especially low N and P availability, shows that the cangas from Carajás Mineral Province are nutrient-poor ecosystems. This finding confirms the results of previous studies that related the oligotrophic conditions of these ecosystems to the poorly developed acidic soils, chemically poor parent material, slow soil disintegration, and high leaching in these ecosystems (e.g., Vasconcelos et al. 2016; Schaefer et al. 2016). Low nutrient availability may hamper the microbial decomposition of organic matter (Zech et al. 1997; Bastias et al. 2018), which may become more recalcitrant due to the formation of stable complexes of organic matter and Al or Fe in clay minerals (Benites et al. 2001; Simas 2002), thus explaining the high organic matter content in soils of all canga physiognomies.
In contrast to the two scrubland physiognomies, WD is associated with higher P concentrations in its soils. The deeper soils in WD physiognomy than in scrubland physiognomy (e.g., Nunes et al. 2015; Schaefer et al. 2016) contribute to higher P (and other nutrient) availability and buffer it against nutrient shortages. This higher nutrient availability in WD soils may support the development of larger trees in WD over that in the other examined physiognomies and in turn shift the belowground competition for nutrients that dominates in open formations to aboveground competition for light in WD. Higher plant stands (Nunes et al. 2015) in WD indicate the production of larger amounts of fresh organic matter, i.e., higher nutrient turnover, in WD than in the other physiognomies.
In contrast to the above findings, the mean foliar nutrient concentrations do not differ from the values commonly observed in shrub or tree species from other nutrient-poor ecosystems (Haridasan and Araújo 2005; Boeger et al. 2005; Machado et al. 2016; Carvalho et al. 2018). However, the elevated Zn and Fe concentrations detected in some leaf samples may indicate metal hyperaccumulation in some plants of canga vegetation, consistent with previous studies (Silva 1992, but see Schettini et al., 2018).
Generally, foliar nutrient concentrations tend to reflect the nutritional status of the vegetation and increase with nutrient availability in the soil (Schreeg et al. 2014). Although we expected to find significant relationships between foliar and soil concentrations for most nutrients in this study, no such relationships were found; however, these relationships may be masked by variation in soil depth, which is considerable among physiognomies (Schaefer et al. 2016). If we consider foliar nutrient concentrations as proxies for nutrient availability, elevated foliar concentrations of N, P, K, and Ca in WD may indicate higher macronutrient availability in this physiognomy, where soils are deeper, than in the other physiognomies.
Differences in foliar nutrient concentrations indicate that canga plants from different physiognomies differ in their nutritional demands. This variability may be due to inter- and intra-specific differences, as some species occur in more than one physiognomy, whereas others are restricted to a single physiognomy (Nunes et al. 2015). Knowledge of these nutritional requirements is indispensable for the restoration of these endangered habitats (Carvalho et al. 2018; Gastauer et al. 2018). Furthermore, the differences in foliar nutrient concentrations among plants indicate the presence of different strategies to overcome low nutrient availability in canga ecosystems, which contributes to functional diversity within and among physiognomies.
Furthermore, differences in leaf nutrient concentrations among plants contribute to differences in litter quality, which influences the conditions and resource availabilities to detritivores and decomposers. These differences may induce differences in the community composition and abundance of saprolithic species among canga physiognomies, affecting decomposition patterns and the release and cycling of nutrients. Thus, the differences in nutrient concentrations in leaves and litter among the different physiognomies indicate differences in nutrient dynamics among these physiognomies.
Canga ecosystems: moderately conserved N cycles and high water shortage
The foliar δ15N values observed in all canga physiognomies were lower than those in tropical forests, indicating more conservative N cycles for Amazonian cangas than for tropical forests. Due to higher N stocks in forests, turnover rates and fractionation against 15N in forests are higher than in Amazonian cangas, causing higher N losses and therefore less conserved N cycles (Bustamante et al. 2004; Mardegan et al. 2009).
However, the other neotropical savannas, all comprising similar physiognomies, had lower reported foliar δ15N values than those obtained for the Amazonian cangas. These savannas include the cerrado, a savannic biome that occupies central and southwestern Brazil (Bustamante et al. 2004); sandy ecosystems such as the mussunungas, i.e., sandy patches covered by savanna vegetation inserted in the Atlantic Forest matrix in Southeastern Brazil (Gastauer et al. 2017; Meira-Neto et al. 2018); and campinaranas, a savanna-like vegetation occurring above podzols in western Amazonia (Mardegan et al. 2009). These findings indicate higher conservation of N in these ecosystems than in Amazonian cangas, although the underlying reasons remain unclear.
As biological N fixation results in δ15N values similar to that in the atmosphere (Unkovich 2013), which, by definition, is zero, the high abundance of N-fixing Fabaceae species reported for cerrado communities may explain the close-to-zero δ15N values in Cerrado surveys (Sprent et al. 1996; Bustamante et al. 2004). However, a high abundance of N-fixing Fabaceae species such as Mimosa acutistipula var. ferrea, M. skinneri, and Bauhinia pulchella has also been observed in canga ecosystems, especially in the SB physiognomy (Silva 1992), and thus cannot satisfactorily explain the observed differences among ecosystems.
Mycorrhizal interactions, especially those of ectomycorrhiza, are able to reduce foliar δ15N values (He et al. 2014; Hobbie and Högberg 2012). Thus, the lower foliar δ15N values in sandy ecosystems than in cerrado than in canga vegetation are consistent with an increasing importance of mycorrhizal interactions from cangas to cerrado, mussunungas, and campinaranas (Meira-Neto et al. 2018). Recent evidence reveals that vegetation from quartzite outcrops in Southeastern Brazil, the so-called campos rupestres characterized by physiognomies similar to those of canga vegetation (Jacobi and Carmo 2008a, b; Vasconcelos et al. 2016), is dominated by nonmycorrhizal species (Zemunik et al. 2015). If the same pattern holds true for the vegetation examined here, the higher δ15N values may result from a lower importance of mycorrhizal interactions in canga ecosystems than in other neotropical savannas.
Litter 15N depletion, i.e., reduced δ15N values relative to leaf δ15N, was observed for all canga physiognomies. As outlined above, most biological processes fractionate against 15N. Such fractionation may occur during nutrient translocations within plants before leaf fall; i.e., lighter 14N may be preferably translocated while a higher portion of 15N remains in falling leaves, resulting in decreased δ15N values in litter compared to leaves. Additionally, decomposition and mineralization of litter may fractionate against 15N such that lighter 14N is liberated more rapidly and, consequently, incorporated more rapidly into biomass (plants, decomposers, or detritivores) or leached, while 15N remains in the litter fraction, decreasing litter δ15N values (Wang et al. 2015). Alternatively, N2 fixation by free-living bacteria associated with litter may reduce the δ15N values (Hyodo and Wardle 2009), as N fixed from the atmosphere tends to show δ15N values close to zero (Unkovich 2013).
In our study, the higher δ13C values found in Amazonian cangas than in tropical forests indicate higher water shortage in canga vegetation than surrounding forests. Cerrado vegetation shows values similar to or slightly lower than those in canga vegetation, indicating similar or slightly lower water shortage. Cerrado and canga ecosystems are both shaped by a seasonal climate, which explains the similar values found in the two ecosystems.
Canga physiognomies vary regarding N cycling and water availability
Differences in δ15N values in vegetation indicate the presence of different ecological processes, including those involving the exploitation of available N resources, uptake strategies, and interactions with rhizobia or mycorrhizae (Craine et al. 2015). Nevertheless, lower δ15N values are generally associated with more conserved N cycles. We found a positive correlation between foliar δ15N values and N concentrations in leaves. Thus, assuming a positive relationship between foliar N and P contents with nutrient availability in the soil, as outlined before, this relationship indicates more conservative N cycles in canga soils with lower nutrient availability, confirming results from other oligotrophic ecosystems (e.g., Meira-Neto et al. 2018).
The lowest δ15N values were observed in the compartments (leaves, litter and soils) of VL, indicating that the most conservative N cycles occur in this physiognomy. VL occurs on bare rocks that lack cracks or fissures that permit soil accumulation and help alleviate nutrient or water shortages. Furthermore, VL vegetation showed the lowest foliar P contents and the highest N:P ratios, indicating a lower P availability in this physiognomy than in the other examined canga physiognomies. The fact that the lowest δ15N occurred in this physiognomy confirms our expectation that the most conservative N cycles would be found in those canga environments with the lowest availabilities of nutrients.
Plants of the genus Vellozia, which dominate in VL (unpublished data), are characterized by scleromorphic leaves (Rossatto et al. 2015) and thick fiber bundles (Mello-Silva 2018), which enhance leaf hardness and recalcitrance, explaining the higher C:N ratios of leaves and litter observed in VL than in the other physiognomies. Elevated C:N ratios observed in litter and leaves suggest slower decomposition and mineralization processes in this physiognomy. Slower N-mineralization contributes to less fractionation against 15N, i.e., more conserved N cycles, which explains the lower δ15N values observed in VL than in the other physiognomies.
The lowest δ13C values were observed in the compartments of WD. As WDs are characterized by more profound soils than those of the other physiognomies, the higher δ13C values in this physiognomy are consistent with lower water deficits, as more profound soils are able to retain more water. Similar results regarding isotopic ratios are expected for ecosystems in which water stress and nutrient availability contribute to the emergence of distinct physiognomies, e.g., such as cerrado, campinaranas, and mussunungas.
Thus, our data show that Amazonian cangas differ from other neotropical ecosystems in their nutrient and water dynamics. Elevated δ13C values indicate a more pronounced water shortage in Amazonian cangas than in neotropical forests. Furthermore, the data indicate that the N cycles in canga vegetation are more conservative than those in tropical forests but less conservative than those in other savanna or sandy ecosystems. Lower nutrient availability, slower decomposition, and more conservative N cycles characterize VL, whereas WD is characterized by lower water restriction and higher P availability than are present in the other examined physiognomies, which confirms findings from other sandy ecosystems. Differences in nutrient dynamics, N cycling, and water availability among physiognomies explain the observed variations in nutrient concentrations and isotopic ratios in the vegetation, litter, and soil of Amazonian canga ecosystems. These findings indicate that the adaptations of plant species to the canga environment as well as the ecological processes that maintain canga vegetation vary among physiognomies. The different ecological processes regulating nutrient and water dynamics among canga physiognomies necessitate the conservation of the entire physiognomic gradient to ensure the maintenance of functional diversity in this unique ecosystem.
References
Alcantara S, de Mello-Silva R, Teodoro GS, Drequeceler K, Ackerly DD, Oliveira RS (2015) Carbon assimilation and habitat segregation in resurrection plants: a comparison between desiccation- and non-desiccation-tolerant species of Neotropical Velloziaceae (Pandanales). Funct Ecol 29:1499–1512
Alvares CA, Stape JL, Sentelhas PC, Moraes Gonçalves JL, De Sparovek G (2013) Köppen’s climate classification map for Brazil. Meteorol Zeitschrift 22:711–728
Asada T, Warner B, Aravena R (2005) Nitrogen isotope signature variability in plant species from open peatland. Aquat Bot 82:297–307
Bastias E, Ribot M, Romaní AM et al (2018) Responses of microbially driven leaf litter decomposition to stream nutrients depend on litter quality. Hydrobiologia 806:333–346
Benites VM, Schaefer CEGR, Mendonça ES, Martin Neto L (2001) Caracterização da matéroa orgânica e micromorfologia de solos sob Campos de Altitude no parque estadual da Serra do Brigadeiro (MG). Rev Bras Ciências do Solo 25:661–674
Bernhardt E, Schlesinger W (2013) Biogeochemistry: an analysis of global change, 3rd edn. Elsevier, New York
Bobbink R, Hicks K, Galloway J et al (2010) Global assessment of nitrogen deposition effects on terrestrial plant diversity: a synthesis. Ecol Appl 20:30–59
Boeger MRT, Wisniewski C, Reissmann CB (2005) Nutrientes foliares de espécies arbóreas de três estádios sucessionais de floresta ombrófila densa no sul do Brasil. Acta Bot Brasilica 19:167–181
Bukovská P, Bonkowski M, Konvalinková T et al (2018) Utilization of organic nitrogen by arbuscular mycorrhizal fungi is there a specific role for protists and ammonia oxidizers? Mycorrhiza 80:269–283. https://doi.org/10.1007/s00572-018-0825-0
Bustamante MMC, Martinelli LA, Silva DA et al (2004) 15 N natural abundance in wood plants and soils of Central Brazilian Savannas (Cerrado). Ecol Appl 14:200–213
Bustamante MMC, Brito DQ, Kozovits AR et al (2012) Effects of nutrient additions on plant biomass and diversity of the herbaceous-subshrub layer of a Brazilian savanna (Cerrado). Plant Ecol 213:795–808
Camenzind T, Hättenschwiler S, Treseder KK, Lehmann A, Rillig MC (2018) Nutrient limitation of soil microbial processes in tropical forests. Ecol Monogr 88:4–21
Canfield DE, Glazer AN, Falkowski PG (2010) The evolution and future of earth’s nitrogen cycle. Science 330:192–196
Carmo FF, Carmo FF, Campos IC, Jacobi CM (2012) Ilhas de ferro estratégicas para a conservação. Ciência Hoje 295:48–53
Carvalho JM, Ramos SJ, Furtini Neto AE et al (2018) Influence of nutrient management on growth and nutrient use efficiency of two plant species for mineland revegetation. Restor Ecol 26:303–310
Craine JM, Morrow C, Fierer N (2007) Microbial nitrogen limitation increases decomposition. Ecology 88:2105–2113
Craine JM, Brookshire ENJ, Cramer MD et al (2015) Ecological interpretations of nitrogen isotope ratios of terrestrial plants and soils. Plant Soil 396:1–26
da Silva MFF (1992) Distribuição de metais pesados na vegetação metalófica de Carajás. Acta Bot Brasilica 6:107–122
Della Coletta L, Nardoto GB, Latansio-Aidar SR, da Rocha HR (2009) Isotopic view of vegetation and carbon and nitrogen cycles in a cerrado ecosystem, southeastern Brazil. Sci Agric 66:467–475
Elser JJ, Bracken MES et al (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1135–1142
Escudero A, Mediavilla S, Heilmeier H (2008) Leaf longevity and drought: avoidance of the costs and risks of early leaf abscission as inferred from the leaf carbon isotopic composition. Funct Plant Biol 35:705–713
Evans RD (2001) Physiological mechanisms influencing plant nitrogen isotope composition. Trends Plant Sci 6:121–126
Galloway JN, Dentener FJ, Capone DG et al (2004) Nitrogen cycles: past, present, and future. Biogeochemistry 70:153–226
Gastauer M, Saporetti Junio AW, Valladares F, Meira-Neto JAA (2017) Phylogenetic community structure reveals differences in plant community assembly of an oligotrophic white-sand ecosystem from the Brazilian Atlantic Forest. Acta Botanica Brasilica 31(4):531
Gastauer M, Silva JR, Caldeira-Jr CF et al (2018) Mine land rehabilitation: modern ecological approaches for more sustainable mining. J Clean Prod 172:1409–1422
Gibson N, Meissner R, Markey AS, Thompson WA (2012) Patterns of plant diversity in ironstone ranges in arid south western Australia. Arid Environ 77:25–31. https://doi.org/10.1016/j.jaridenv.2011.08.021
Haridasan M, Araújo GM (2005) Perfil nutricional de espécies lenhosas de duas florestas semidecíduas em Uberlândia, MG. Rev Bras Botânica 3:295–303
He M, Dijkstra FA, Zhang K et al (2014) Leaf nitrogen and phosphorus of temperate desert plants in response to climate and soil nutrient availability. Sci Rep 4:1–7. https://doi.org/10.1038/srep06932
Heaton THE, Spiro B, Robertson SMC (1997) Potential canopy influences on the isotopic composition of nitrogen and sulphur in atmospheric deposition. Oecologia 109:600–607
Hedin LO, Brookshire ENJ, Menge DNL, Barron AR (2009) The nitrogen paradox in tropical forest ecosystems. Annu Rev Ecol Evol Syst 40:613–635
Hobbie EA, Hobbie JE (2008) Natural abundance of 15N in nitrogen-limited forests and tundra can estimate nitrogen cycling through mycorrhizal fungi: a review. Ecosystems 11:815–830
Hobbie EA, Högberg P (2012) Nitrogen isotopes link mycorrhizal fungi and plants to nitrogen dynamics. New Phytol 196:367–382
Högberg P, Johannisson C, Yarwood S et al (2011) Recovery of ectomycorrhiza after ‘nitrogen saturation’ of a conifer forest. New Phytol 189:515–525
Houlton BZ, Sigman DM, Hedin LO (2006) Isotopic evidence for large gaseous nitrogen losses from tropical rainforests. Proc Natl Acad Sci 103:8745–8750
Hyodo F, Wardle DA (2009) Effect of ecosystem retrogression on stable nitrogen and carbon isotopes of plants, soils and consumer organisms in boreal forest islands. Rapid Commun Mass Spectrom 23:1892–1898
Jacobi CM, Carmo FF (2008a) Diversidade dos campos rupestres ferruginosos no Quadrilátero Ferrífero, MG. Megadiversidade 4:25–33
Jacobi CM, Carmo FF (2008b) The contribution of ironstone outcrops to plant diversity in the iron quadrangle, a threatened Brazilian landscape. AMBIO A J Hum Environ 37:324–326
Janssens IA, Dieleman W, Luyssaert S et al (2010) Reduction of forest soil respiration in response to nitrogen deposition. Nat Geosci 3:315–322
Kozovits AR, Bustamante MMC, Garofalo CR et al (2007) Nutrient resorption and patterns of litter production and decomposition in a neotropical savanna. Funct Ecol 21:1034–1043
Lloyd J, Paiva RQ, Nardoto GB, Quesada CA (2009) Variations in leaf physiological properties within Amazon forest canopies. Biogeosci Discuss 6:4639–4692
Machado MR, Sampaio PD, Ferraz J, Camara R, Pereira MG (2016) Nutrient retranslocation in forest species in the Brazilian Amazon. Acta Sci Agron 38:93–101
Mardegan SF, Nardoto GB, Higuchi N, Moreira MZ, Martinelli LA (2009) Nitrogen availability patterns in white-sand vegetations of Central Brazilian Amazon. Trees 23:479–488
Mârtensson L-M, Carlsson G, Prade T, Kørup K, Lærke PE, Jensen ES (2017) Water use efficiency and shoot biomass production under water limitation is negatively correlated to the discrimination against 13C in the C3 grasses Dactylis glomerata, Festuca arundinacea and Phalaris arundinacea. Plant Physiol Biochem 113:1–5. https://doi.org/10.1016/j.plaphy.2017.01.021
Martinelli LA, Piccolo MC, Townsend AR et al (1999) Nitrogen stable isotopic composition of leaves and soil: tropical versus temperate forests. Biogeochemistry 46:45–65
Martinelli LA, Ometto JPHB, Ishida F, Domingues TF et al (2007) The use of carbon and nitrogen stable isotopes to track effects of land-use changes in the Brazilian Amazon region. In: Dawson TE, Siegwolf RTW (eds) Stable isotopes as indicators of ecological change. Elsevier, New York, pp 301–318
Matson PA, Vitousek PM (1987) Cross-system comparisons of soil nitrogen transformations and nitrous oxide flux in tropical forest ecosystems. Glob Biogeochem Cycles 1:163–170
Meira-Neto JAA, da Silva MCNA, Tolentino GS et al (2018) Shading, nitrogen and soil texture rule a sandy savanna: does facilitation rule its patchy physiognomy as well? Flora 239:45–51. https://doi.org/10.1016/j.flora.2017.11.007
Mello-Silva R (2018) Flora das cangas da Serra dos Carajás, Pará, Brasil: Velloziaceae. Rodriguésia 69:259–262
Mota NF de O, Silva LVC, Martins FD, Viana PL (2015) Vegetação sobre sistemas ferruginosos da Serra dos Carajás.In Carmo FF do, Kamino LHY (eds). Geossistemas ferruGinosos do brasil Áreas prioritárias para conservação da diversidade geológica e biológica, patrimônio cultural e serviços ambientais. Belo Horizonte, pp. 289–315
Nardoto GB, Ometto JPHB, Ehleringer JR, Higuchi N, da Bustamante MM, Martinelli CA (2008) Understanding the influences of spatial patterns on N availability within the Brazilian Amazon forest. Ecosystems 11:1234–1246
Nardoto GB, Quesadab CA, Patiñob S et al (2013) Basin-wide variations in Amazon forest nitrogen- cycling characteristics as inferred from plant and soil 15 N: 14 N measurements N:N measurements. Plant Ecol Divers 7:1–15
Nunes JA, Schaefer CEGR, Júnior WGF (2015) Soil-vegetation relationships on a banded ironstone ‘island’. Carajás Plateau, Brazilian Eastern Amazonia. 87:2097–2110
Ometto JPHB, Ehleringer JR, Domingues TF et al (2006) The stable carbon and nitrogen isotopic composition of vegetation in tropical forests of the Amazon Basin, Brazil. Biogeochemistry 79:251–274
Pardo LH, Nadelhoffer KJ (2010) Using Nitrogen Isotope Ratios to Assess Terrestrial Ecosystems at Regional and Global Scales. In: West JB, Bowen GJ, Dawson TE, Tu PU (eds) Isoscapes: understanding movement, pattern, and process on earth through isotope mapping. Springer, London, pp 221–249
Poot P, Lambers H (2008) Shallow-soil endemics: adaptive advantages and constraints of a specialized root-system morphology. New Phytol 178(2):371–381
R Development Core Team (2018) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.R-project.org
Rezende LAL, Dias LE, Assis IR, Braga R (2013) Rehabilitation of irone stones outcrops degraded by mining activity in Minas Gerais state of Brazil. J Am Soc Miningand Reclam 2:151–159
Robinson D (2001) δ 15 N as an integrator of the nitrogen. Trends Ecol Evol 16:153–162
Rossatto DR, Kolb R, Franco A (2015) Leaf anatomy is associated with the type of growth form in Neotropical savanna plants. Botany 93:507–518
Scarano FR, Duarte HM, Ribeiro KT et al (2001) Four sites with contrasting environmental stress in southeastern Brazil: relations of species, life form diversity, and geographic distribution to ecophysiological parameters. Bot J Linn Soc 136:345–364
Schaefer CGRE, Lima Neto E, Corrêa GR, Simas FNB, Campos JF, De Mendonça BAF (2016) Geoambientes, solos e estoques de carbono na Serra Sul de Carajás, Pará, Brasil Geoenviroments, soils and carbon stocks at Serra Sul of Carajás, Para State, Brazil. Bol Mus Para Emílio Goeldi Cienc Nat 11:85–101
Schettini AT, Leite MGP, Messias MCTB, Gauthier A, Li H, Kozovits AR (2018) Exploring Al, Mn and Fe phytoextraction in 27 ferruginous rocky outcrops plant species. Flora 238:175–182. https://doi.org/10.1016/j.flora.2017.05.004
Schreeg LA, Santiago LS, Wright SJ, Turner BL (2014) Stem, root, and older leaf N:P ratios are more responsive indicators of soil nutrient availability than new foliage. Ecology 95:2062–2068
Silveira FAO, Negreiros D, Barbosa NPU et al (2016) Ecology and evolution of plant diversity in the endangered campo rupestre: a neglected conservation priority. Plant Soil 403:129–152
Skirycz A, Castilho A, Chaparro C, Carvalho N, Tzotzos G, Siqueira JO (2014) Canga biodiversity, a matter of mining. Front Plant Sci 5:653. https://doi.org/10.3389/fpls.2014.00653
Sprent JI, Geoghegan IE, Whitty PW, James EK (1996) Natural abundance of 15 N and 13C in nodulated legumes and other plants in the cerrado and neighbouring regions of Brazil. Oecologia 105:440–446
Steward GR, Schmidt S, Handley LL, Turnbull MH, Erskine PD, Joly CA (1995) 15 N natural abundance of vascular rainforest epiphytes: implications for nitrogen source and acquisition. Plant, Cell Environ 18:85–90
Thomas RQ, Jack BEN, Stefan G (2015) Nitrogen limitation on land: how can it occur in Earth system models? Glob Chang Biol 21:1777–1793
Unkovich M (2013) Isotope discrimination provides new insight into biological nitrogen fixation. New Phytol 198:643–646
Vasconcelos JM, da Silva Lopes, Júnior M, de Ruivo M et al (2016) Solos metalíferos: atributos químicos nas diferentes fitofisionomias da Serra Sul, Serra dos Carajás, Pará, Brasil metalliferous soils: chemical attributes in different phytophysiognomies of the Serra Sul, Serra dos Carajás, Pará, Brazil. Bol Mus Para Emílio Goeldi Cienc Nat 11:49–55
Viana PL, de Mota NF, dos Santos Bragança Gil A et al (2016) Flora das cangas da Serra dos Carajás, Pará, Brasil: história, área de estudos e metodologia. Rodriguésia 67:1107–1124
Vitousek PM, Mooney HA, Lubchenco J, Melillo JM (1997) Human domination of earth’s ecosystems. Science 277:494–499
Wang Y, Xu Z, Zheng J, Abdullah KM (2015) δ 15 N of soil nitrogen pools and their dynamics under decomposing leaf litters in a suburban native forest subject to repeated prescribed burning in southeast Queensland, Australia. J Soils Sediments 15:1063–1074
Werner C, Máguas C (2010) Carbon isotope discrimination as a tracer of functional traits in a mediterranean macchia plant community. Funct Plant Biol 37:467–477
Zech W, Senesi N, Guggenberger G et al (1997) Factors controlling humification and mineralization of soil organic matter in the tropics. Geoderma 79:117–161
Zemunik G, Turner BL, Lambers H, Laliberté E (2015) Diversity of plant nutrient-acquisition strategies increases during long-term ecosystem development. Nat Plants 1:1–4
Acknowledgements
This research was supported by Vale S. A. in cooperation with the Instituto Tecnológico Vale. We thank the Conselho Nacional de Pesquisa e Desenvolvimento (CNPq) for granting scholarships to SKM (300362/2016-1 and 300514/2017-4), SJR (305831/2016-0), AEFN (303224/2013-4), and JOS (303580/2013-5). We are grateful for the valuable comments from two anonymous reviewers.
Author information
Authors and Affiliations
Contributions
SKM, SFM, CFC, and AEFN designed this study. SKM, CFC, and SJR collected the data, which were analyzed and interpreted by SKM, SFM, and MG. SKM, SFM, and MG wrote the manuscript with important contributions from CFC, SJR, and JOS.
Corresponding author
Additional information
Communicated by Dafeng Hui.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mitre, S.K., Mardegan, S.F., Caldeira, C.F. et al. Nutrient and water dynamics of Amazonian canga vegetation differ among physiognomies and from those of other neotropical ecosystems. Plant Ecol 219, 1341–1353 (2018). https://doi.org/10.1007/s11258-018-0883-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11258-018-0883-6