Abstract
By comparing with the historic semi-natural grassland and woodland vegetation data compiled in the 1970s and 1980s, we aimed to reveal potential roles of small and linear habitat fragments remaining in the urbanized satoyama landscape (i.e., agricultural landscape) in Japan for conservation of grassland plant species. TWINSPAN cluster analysis differentiated the current vegetation from historic one. Current vegetation of Miscanthus grassland was classified into two different groups and one of the two was equivalent to current understory vegetation of fragmented woodland. The linear woodland edge vegetation along the roads was classified into one group and separated from the other current vegetation groups. The current vegetation groups were characterized by higher richness of exotic species than historic vegetation groups. Detrended correspondence analysis (DCA) revealed that there were no groups of current vegetation that is equivalent to historic woodland understory vegetation. The vegetation quality of the current group of Miscanthus grassland and secondary woodland remaining on urban public properties, and the group of linear roadside vegetation were almost equivalent to that of historic semi-natural grassland. Both of the small and linear habitat fragments might have functioned as habitats for grassland species under regular mowing management. Although small and linear habitat fragments would not be sufficient for sustaining grassland populations in the future, these habitats can serve as key reservoirs for grassland species recovery in the conservation and restoration of grassland communities in the urbanized satoyama landscape.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Conservation of urban biodiversity is a major challenge for preventing further loss of biodiversity at local, regional and global scales, and for supporting human well-being through enhancing ecosystem functioning (Alberti and Marzluff 2004; Puppim de Oliveira et al. 2011; Werner 2011). In urban landscapes, especially in developed countries, habitats with high conservation value (i.e., “hotspots”) have mostly deteriorated (e.g. Pysek 1998; Godefroid 2001; Tait et al. 2005). From a conservation point of view, however, it is important to focus not only on hotspots but also on remaining habitat fragments, because these fragments may be relatively important for maintaining and enhancing regional species diversity in highly fragmented landscapes (Cousins 2006; Cousins and Eriksson 2008; Ramalho and Hobbs 2011). Green space that is managed in urban areas to promote recreation and people’s well-being also may support local biodiversity (Bolund and Hunhammar 1999; Colding et al. 2006; McGranahan et al. 2005). Ecosystem services in urban areas have received considerable attention, and numerous studies have revealed the function of green spaces in biodiversity conservation (e.g., Muratet et al. 2008; Sandström et al. 2006; Sattler et al. 2010). Not only small fragmented habitats in green spaces but also linear landscape components such as roadsides, railway-sides, and riverbanks can have important roles for species conservation in urban landscapes and their surroundings (Cousin 2006; Hovd 2008; Noordijk et al. 2011). It is essential to focus on these small and linear habitat fragments for preventing further loss of urban biodiversity.
Satoyama landscapes, the traditional ‘socio-ecological production landscape’ of Japan, consist of a mosaic of grasslands and woodlands, rice paddy fields, crop fields, streams and ponds (Fukamachi et al. 2001; Takeuchi et al. 2003). There are various types of socio-ecological production landscapes similar to satoyama landscapes not only in Asia but also in Europe, Africa and many other areas in the world, characterized by human-influenced nature environments with high biodiversity (The Satoyama Initiative 2010). Satoyama landscapes once covered more than 40 % of the whole land area in Japan (Ministry of the Environment 2002), but have undergone dramatic changes by intensification of agricultural system and urbanization during the past 100–150 years. Negative impacts of rapid urbanization and agricultural intensification on biodiversity can be recognized many parts of the socio-ecological production landscapes in the world including satoyama landscapes (The Satoyama Initiative 2010).
Semi-natural grassland was one of the main components of satoyama landscapes. Japanese people used to maintain a vast area of semi-natural grassland to produce fodder for farm animals and organic fertilizer for crop fields and rice paddy fields (Sprague 2001; Takeuchi et al. 2003). The total area of semi-natural grassland in Japan was estimated to have decreased from more than 10 % of the whole land area in the 1880s to less than a few percent in recent years (Himiyama 1995; Shoji 2003). Many grassland species have been designated as rare or threatened because of the drastic loss of habitats for these species by rapid urbanization and agricultural intensification, especially in the urbanized satoyama landscapes surrounding metropolitan areas (Ide 2006).
Increasing recognition of the biodiversity value of semi-natural vegetation in satoyama landscapes especially since the 1990s has made it possible to retain green spaces for conserving urban biodiversity (Takeuchi 2006; Takeuchi et al. 2003). In the course of development, some of the historic semi-natural woodland and grassland areas have been left in place (e.g., public parks), and these areas may have retained their functional role as habitats for grassland species. It is urgently needed to clarify whether populations of grassland plant species that were common in the historic satoyama landscapes can be sustained in the remaining small and linear habitats in the present urbanized satoyama landscapes. The remaining populations of grassland species could be important sources for not only maintaining but also enhancing local and regional biodiversity in urbanized satoyama landscapes.
Because most monitoring efforts have been confined to relatively rare natural or semi-natural habitats (Japan Wildlife Research Center 2006), information on the historic reference vegetation of satoyama landscapes tends to be lacking. In the early 1970s, phytosociology was introduced in Japan and nationwide vegetation surveys based on the methods of Braun-Blanquet (1964) were conducted (Miyawaki 1977). The regional flora data compiled in the 1970s and 1980s are the oldest data based on the unified phytosociological methodology, and these data could be critical for identifying references of the target semi-natural grassland vegetation in satoyama landscapes.
In this study, we aimed to reveal the potential roles of small and linear habitat fragments of semi-natural vegetation in the conservation of grassland plant species in highly urbanized satoyama landscape. We clarified whether the current semi-natural vegetation remaining in the landscape is comparable to that of historic semi-natural grassland. The specific objectives were: (1) to compare the species composition in the current small and linear grassland and open secondary woodland vegetation remaining in the urbanized satoyama landscapes with those of historic reference semi-natural vegetation; (2) to reveal richness patterns of grassland species and exotic species in the current and historic vegetation; and (3) to evaluate the vegetation quality and the roles of small and linear habitats for preventing further loss of grassland species diversity in the urbanized satoyama landscape.
Materials and methods
Study area
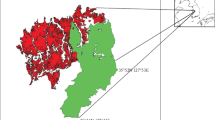
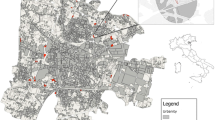
This study was conducted in southern Tsukuba City, Ibaraki Prefecture, in the Kanto region, Japan (Fig. 1). The study area was located in the Tsukuba–Inashiki upland region, where the elevation ranges from approximately 20 to 30 m above sea level. The mean annual temperature is 13.5 °C (2.3 °C in January and 25.2 °C in August), with a mean annual precipitation of 1,230 mm at Tsukuba (Tateno) meteorological observatory (Japan Meteorological Agency 2007).
Tsukuba Science City has been developed in the study area since the 1970s, and a number of research and educational institutes were constructed with both public and private housings. Semi-natural habitats for grassland species, which include both semi-natural grassland and open secondary woodlands, were widespread around the study area in the 1880s (Fig. 1). However, nearly all these habitats have disappeared particularly during the last 50 years, due to rapid urbanization accompanied with the development of Tsukuba Science City (Sakiyama and Itoga 1994). Most of the woodland areas that escaped land-use conversion outside the planning areas of Tsukuba Science City were abandoned and underwent further succession to mixed deciduous-evergreen forest with a fully developed canopy (Inui 1992). In the present highly fragmented landscape, limited numbers of semi-natural habitats for grassland species remain, mostly in areas under regular management on public properties within Tsukuba Science City. In addition to the small habitat fragments, linear roadside vegetation along the edges of remnant woodlands could be another key habitat for grassland species because the vegetation has been maintained by the regular management (mowing) to improve traffic safety.
Target semi-natural vegetation
The study area belongs to the warm-temperate, monsoonal, mixed evergreen-broadleaved forest region, with the climax dominants Castanopsis cuspidata var. sieboldii, Quercus myrsinifolia, and Machilus thunbergii. The vegetation of semi-natural grasslands maintained by the traditional agricultural management (i.e., burned in early spring and mown once in summer) found in this region belongs to the Pleioblastus chino–Miscanthus sinensis phytosociological community (Miyawaki 1986). We defined characteristic species in this community as typical grassland species in the study area. The open secondary woodland, which consists mainly of Pinus densiflora, associated with Quercus acutissima and Quercus serrata, is another type of habitat for grassland species in the study area. These woodlands were cut once every 20 to 30 years, and the understory layer was mowed to produce organic fertilizer or livestock feed. These management practices maintained high light availability which enabled many grassland species to coexist in the understory vegetation (Fujii and Zinnai 1979; Yamamoto and Itoga 1988).
Vegetation sampling
We targeted fragmented grassland vegetation dominated by M. sinensis and the understory vegetation of managed open secondary woodland remaining on urban public and private properties in the study area (i.e., in Tsukuba Science City and surrounding areas) (Fig. 1). We also targeted the linear woodland edge vegetation along the two major roads running north–south and east–west maintained by public mowing management for traffic safety. We conducted vegetation surveys at 17 grassland and 13 woodland sites, and 18 sites of roadside along the edges of woodlands during summer in 2006, 2007 and 2008. The surveys were conducted in areas that used to be semi-natural grasslands or open woodlands within the study area (Fig. 1) and where the grassland and understory of woodland vegetation plots was dominated or sub-dominated by M. sinensis.
In order to compare with historic vegetation data, the vegetation survey was based on the phytosociological method of Braun-Blanquet (Mueller-Dombois and Ellenberg 1974) which was also used in past surveys (Miyawaki and Suzuki 1974; Miyawaki et al. 1975; Miyawaki 1986; Sakurai 1982). We recorded all species using cover rank values (5: 100-75 %, 4: 75-50 %, 3: 50-25 %, 2: 25-10 %, 1: 10-1 %, +: < 1 %, r: appeared only once) at each surveyed site. Depending on the entire area of each site, the total area surveyed ranged from 15 to 400 m2 at Miscanthus grassland sites and from 20 to 75 m2 at secondary woodland sites. Each survey plot was surveyed starting from approximately the center of each fragmented habitat and survey area was increased until new species were no longer encountered. We also surveyed the woodland edge vegetation along roads within 1–2 m from the pavement edge. The surveyed lengths of roadside vegetation ranged from 10 to 40 m depending on the length of woodland edges.
Historic vegetation data
We gathered vegetation data for the P. chino–M. sinensis community and pine woodland community representing the historic grassland and secondary woodland vegetation, respectively, from the regional flora compiled in the 1970s and 1980s (Miyawaki and Suzuki 1974; Miyawaki et al. 1975; Miyawaki 1986; Sakurai 1982). We gathered the historic vegetation data conducted in Tsukuba–Inashiki and Shimousa upland regions both located in the eastern part of the Kanto plain and under the almost same geographical conditions. Historic vegetation data of Miscanthus grasslands has some variation, including semi-natural grassland maintained by the traditional management practices and grassland established on open disturbed habitats after abandonment of cultivation or land reclamation (Miyawaki 1986). By referring the information on the management of each surveyed vegetation plots, we selected vegetation data for 9 Miscanthus grassland sites representing the past semi-natural grassland community. To represent vegetation of the managed open secondary woodland, we extracted understory vegetation data of the pine woodland sites that had a sparse shrub layer with ≤40 % cover, 20 woodland sites in total.
Data analysis
Data analysis consisted of three steps. First, to identify and characterize different vegetation types, we classified historic and current vegetation data using a classification analysis called TWINSPAN (Two-way Indicator Species Analysis; Hill 1979b). Secondly, we examined differences in species richness and diversity patterns among historic and current vegetation groups. Finally, we analyzed species compositional data using an ordination method called detrended correspondence analysis (DCA; Hill 1979a) to clarify the differences in vegetation quality among the historic and current vegetation groups.
We used both historic and current vegetation data (77 sites) in the TWINSPAN classification using presence–absence data of species at each site. All species recorded in less than three sites in the historic and current vegetation data were excluded from the TWINSPAN analysis. We classified the recorded species into five species groups (grassland species, exotic species, native annuals, native perennials, and woody species), and compared the richness values of all species and each species group for both historic and current vegetation groups identified by TWINSPAN. We performed generalized linear models (GLMs) with a logarithmic link function that follows Poisson distribution to investigate the differences in richness values among the historic and current vegetation groups. We then performed pair-wise comparison of richness values between all pairs of vegetation groups using simultaneous inference methods (Hothorn et al. 2008). P-values were adjusted for multiple comparisons using the conservative Bonferroni method. Finally, we performed detrended correspondence analysis (DCA) with species compositional data of the historic and current vegetation simultaneously using log-transformed cover rank values (“+: <1 %” and “r: appeared only one time” were transformed into 0.1 and 0.01, respectively; Mueller-Dombois and Ellenberg 1974). The relationships between DCA axes1 and 2 scores and richness values of all species and different species groups were tested by Spearman’s rank correlation test. We also tested the significance of differences in the scores of DCA axes 1 and 2 among the vegetation groups identified by TWINSPAN using Tukey’s multiple comparison test. The analytical program PC-ORD for Windows version 4 (McCune and Mefford 1999) was used for TWINSPAN and DCA. All other statistical tests were performed with R version 2.12.2 (R Development Core Team 2011).
Results
A total of 285 plant species were recorded in the historic and current vegetation data. We also recorded some plants that could not be identified to the species level (13 individuals in Poaceae spp., 2 individuals in Compositae spp., 1 individual in Liliaceae spp., 17 individuals in Carex spp., 7 individuals in Rumex spp., 5 individuals in Sonchus spp., and 1 individual in Solanaceae spp.). These unidentified plant individuals were not included in the following data analyses. All the recorded typical grassland species are listed in Appendix 1. A total of 39 and 52 grassland species were recorded in the historic and current vegetation, respectively, and 35 grassland species were common to both vegetation. Four species (Patrinia villosa, Achillea alpina var. longiligulata, Bupleurum stenophyllum and Gentiana scabra var. buergeri) were recorded only in the historic vegetation data.
TWINSPAN cluster analysis classified the 77 vegetation samples into five vegetation groups, differentiating the current vegetation from the historic one (Fig. 2, Table 1). The current vegetation was classified into three groups (C-L, C-GW, and C-G). Almost half of the current Miscanthus grassland vegetation was classified into the same group with the understory vegetation of secondary woodland (C-GW) characterized by typical grassland species (Sanguisorba officinalis and Lespedeza pilosa) and also exotic species (Panicum lanuginosum). The other current Miscanthus grassland vegetation was classified into one group (C-G) characterized by annual species (Setaria faberi). Most of the current linear woodland edge vegetation along the road was classified in to one group (C-L) characterized by native perennial grass (Oplismenus undulatifolius) and broadleaved tree (Rhus javanica). The historic semi-natural grassland vegetation and the understory vegetation of pine woodland were classified into two different groups (H-G and H-W, respectively). The characteristic species of H-G were typical grassland species (Artemisia japonica) and deciduous shrub (Deutzia crenata), whereas those of H-W were ever-green shrub (Eurya japonica) and typical grassland species (Patrinia villosa).
TWINSPAN classification of the historic and current vegetation. Indicator species are shown along the branches. The typical grassland species are shown in boldface. Correspondence between the historic and current vegetation types and vegetation groups identified by TWINSPAN are also shown in Table 1
Richness values of all species were significantly higher in H-G, C-L and C-GW compared to C-G (Fig. 3a). The richness values of grassland species were also higher in H-G and C-GW compared to the other three groups. Exotic species richness was significantly higher in the current vegetation groups of C-GW and C-G compared to the historic ones (Fig. 3b). In C-G, richness of native annuals was significantly higher than the other groups, whereas richness of woody species was the lowest among the groups.
Average richness values of a all species and grassland species, and b exotic species, native annuals, native perennials and woody species of the historic and current vegetation groups classified by TWINSPAN (see Fig. 2). Differences among the groups for each of the richness value were tested for significance using pair-wise comparison between any two vegetation groups using simultaneous inference methods (Hothorn et al. 2008). P-values were adjusted for multiple comparisons using the conservative Bonferroni method. The error bars represent standard deviation within the groups. Different letters showed significant differences among the groups (P < 0.05)
The historic and current vegetation groups identified by TWINSPAN classification were roughly ordered along axis 1 of the DCA ordination (Fig. 4a). The distribution of plots based on the current and historic vegetation types are shown in Appendix 2. The scores of axis 1 (eigenvalue = 0.433) were positively correlated with the richness of exotic species (r = 0.577, P < 0.0001) and native annuals (r = 0.582, P < 0.0001), and negatively correlated with those of woody species (r = –0.599, P < 0.0001). The scores of axis 2 (eigenvalue = 0.255) were positively correlated with the richness of grassland species (r = 0.313, P = 0.006). The average scores of DCA axis 1 were significantly higher in C-G and lower in H-W among the groups (Fig. 4b). There are no significant differences in the scores between C-L and H-G and between C-GW and H-G. The average scores of DCA axis 2 were significantly lower in C-L and C-GW compared with H-G but not significantly different with H-W (Fig. 4b).
a DCA ordination showing all the vegetation plots of the historic and current vegetation groups classified by TWINSPAN (see Fig. 2). b Average scores of DCA axes 1 and 2 of the five vegetation groups. Differences among the groups for each of the richness value were tested for significance using Tukey’s multiple comparison test. Different letters show significance of differences among the groups (P < 0.05). The error bars represent standard deviation within the groups
Discussion
This study revealed that small remnant Miscanthus grassland and secondary woodland habitats on urban public and private properties could function as habitats for grassland species. The linear woodland vegetation along the roads could also serve as habitats for grassland species in the study area. Although these habitats have been invaded by exotic species, the vegetation quality (i.e. species compositions represented by DCA axis 1) showed no significant differences compared with those of the historic reference vegetation of semi-natural grassland. Among the 39 grassland species recorded in the historic vegetation data, four species were absent in the current vegetation surveyed in the study area (Appendix 1). One of the species, Bupleurum stenophyllum, and the other two species, Platycodon grandiflorus and Cynanchum paniculatum, have been listed as VU (vulnerable), VU and NT (near threatened), respectively in the national RDB (Ministry of the Environment 2000). In addition to the above species threatened at the national level, some other species that use to be common in past satoyama landscapes (e.g. Sanguisorba officinalis, Patrinia scabiosaefolia, Adenophora triphylla var. japonica) might become rare or threatened in the near future (Okubo 2002; Ide 2006).
The current vegetation group C-GW consisted both of the Miscanthus grassland and the understory of secondary woodland vegetation. This vegetation would be nearly equivalent to the historic reference semi-natural grassland vegetation (H-G) but not to the historic understory vegetation of pine woodland (H-W). Although the current vegetation of C-GW was invaded by exotic species, grassland species richness in this group was equally high compared with that of historic semi-natural grassland (H-G). Among the 19 sites classified into C-GW, 3 Miscanthus grassland sites and 10 secondary woodland sites are located on urban public properties (i.e. inside the areas of national institutes and public housing estates) and are maintained by regular management. The other Miscanthus grassland and secondary woodland sites are private and might have been managed regularly by the landowners. On the urban public and private properties, most of the remaining semi-natural vegetation has been managed regularly (i.e. mowing aboveground vegetation once or twice during the summer) to keep the open environment for esthetic preference or other purposes of people’s use. Because of the management practices, the current vegetation of these sites (not only Miscanthus grassland sites but also secondary woodland sites) has been kept at the earlier successional stage compared to historic understory vegetation of pine woodland (H-W) (Fig. 4b, represented by the score of DCA axis 1). The fact that most of these habitats were quite small (i.e. less than 100 m2) and surrounded/bordered by roads and built-up areas explain why these habitats were highly invaded by exotic species and other native ruderal annals.
Linear roadside vegetation has received increasing attention for conserving grassland plant species (e.g., Tikka et al. 2000; Cousin and Eriksson 2002; Jantunen et al. 2007). However, such vegetation has experienced the transition from traditional agrarian management (i.e. subscribed burning in early spring and mowing once in summer) to current road verge management with frequent mowing (i.e. two or three times during summer) and additional disturbances such as chemical pollution by automobile fumes and soil fertilization by adjacent land-uses (Tikka et al. 2000; Jantunen et al. 2007). This drastic change in disturbance regime has resulted in the observed changes in species compositions and grassland species richness from those of the reference semi-natural grassland (Koyanagi et al. 2012). In this study, the group of linear woodland edge vegetation was differentiated from the other vegetation types (Miscanthus grassland and understory of secondary woodland) and categorized into one group C-L. Although the current vegetation of group C-L was almost at the same successional stage as the historic semi-natural grassland (H-G) and current Miscanthus grassland and understory of woodland vegetation (C-GW), grassland species richness was significantly lower in C-L than H-G and C-GW. The linear vegetation of roadsides can serve as dispersal corridor for not only grassland species but also exotic species (Tikka et al. 2000; Thiele and Otte 2008). In addition to the exotic species invasion, linear woodland edge vegetation along the roads might have been suffering further loss of grassland species in the future (Koyanagi et al. 2009, 2012).
The current vegetation group C-G was characterized by annual species and was significantly different from historic vegetation groups of semi-natural grassland (H-G) and understory of pine woodland (H-W). All Miscanthus grassland sites classified into C-G were located on private properties, appeared to have been established by land reclamation, and were kept open for future housing developments (Koyanagi et al. 2007). Because M. sinensis is dominant, the vegetation of these grassland sites can also be classified as the P. chino–M. sinensis community to which the historic reference semi-natural grassland belonged (Miyawaki 1986), but with fewer grassland species. Most of the remaining Miscanthus grasslands classified into group C-G might be transient and established before future development of housing and other buildings by land reclamation. The vegetation was characterized by annuals and exotic species at earlier successional stage having little conservation value in the present urbanized landscape.
Although populations of grassland species still exist in the present urbanized satoyama landscape, the remaining habitats appear to be too small and fragmented for maintaining and conserving grassland species diversity in the future. The previous studies conducted in the same study area have revealed that there were significant time-lags of grassland species decline after the landscape changes occurred during the past 50 years (Koyanagi et al. 2009, 2012). This may be evidence of “extinction debt” in the landscape (Kuussaari et al. 2009). The area and quality of remaining habitats in the current landscape are not enough to sustain the grassland populations in the future unless habitats are restored. In Japanese satoyama landscapes, the earlier conservation policies have primarily considered the secondary woodlands and the species associated with these habitats (e.g. forest plant spring ephemerals). Community-based activities for management of secondary woodlands have intensified since the late 1990s, especially in the urban and rural landscapes surrounding metropolitan areas (Yamasaki et al. 2000; Okubo et al. 2005; Ide 2006). Conservation of grassland species can be possible by creating open environment such as well-managed secondary woodlands, and therefore could be part of the objectives of the existing policies and people’s activities in satoyama landscapes. Various remnants of the past semi-natural habitats with grassland plant species growing in abandoned overgrown sites can be still found in the present highly fragmented landscape. Although the existence of extinction debt has been suggested in the landscape (Koyanagi et al. 2009, 2012), there is still opportunities to prevent local extinctions of grassland species by restoring semi-natural habitats suitable for recolonization (Cousins 2009; Kuussaari et al. 2009). Resuming extensive management of the abandoned woodlands that still retain a pool of some grassland species in the roadside vegetation could be an effective conservation and restoration measures to enhance grassland species diversity in the region. Improving the management conducted both in the roadside and other small habitats in public properties can be another effective way of promoting grassland species recolonization and regeneration (Jantunen et al. 2007). Mowing management has been conducted two or three times in summer, and mown vegetation is removed by the public sector. This management regime might have negative impacts on flowering success and seed production of early and late summer-flowering grassland species, so conducting extensive mowing after their flowering and seed setting (i.e. late summer or autumn) is recommended. Although the current areas of remaining habitats are not enough for sustaining grassland species population, the small and linear habitats on urban public and private properties can serve as key reservoirs for grassland species recovery that contribute to enhance regional species diversity in the highly fragmented urbanized satoyama landscape. Utilization of historic or past vegetation data can be important tool for describing potential roles of remaining habitat fragments for species conservation and biodiversity management, especially in highly urbanized satoyama landscapes and any other ‘socio-ecological production landscapes’ around the world.
References
Alberti M, Marzluff JM (2004) Ecological resilience in urban ecosystems: Linking urban patterns to human and ecological functions. Urban Ecosyst 7:241–265
Bolund P, Hunhammar S (1999) Ecosystem services in urban areas. Ecol Econ 29:293–301
Braun-Blanquet J (1964) Pflanzensoziologie: Grundzüge der Vegetationskunde, Vienna, Austria
Colding J, Lundberg J, Folke C (2006) Incorporating green-area user groups in urban ecosystem management. Ambio 35:237–244
Cousins SAO (2006) Plant species richness in midfield islets and road verges: the effect of landscape fragmentation. Biol Conserv 127:500–509
Cousins SAO (2009) Extinction debt in fragmented grasslands: paid or not? J Veg Sci 20:3–7
Cousins SAO, Eriksson O (2002) The influence of management history and habitat on plant species richness in a rural hemiboreal landscape, Sweden. Landscape Ecol 17:517–529
Cousins SAO, Eriksson O (2008) After the hotspots are gone: Land use history and grassland plant species diversity in a strongly transformed agricultural landscape. Appl Veg Sci 11:365–374
R Development Core Team (2011) R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. <http://www.R-project.org>.
Fujii E, Zinnai I (1979) Studies on the relationship between the management of floor layers and the succession of Pinus plain forests in Kanto region. J Jap Forest Soc 61:76–82 (in Japanese with English abstract)
Fukamachi K, Oku H, Nakashizuka T (2001) The change of a satoyama landscape and its causality in Kamiseya, Kyoto Prefecture, Japan between 1970 and 1995. Landscape Ecol 16:703–717
Godefroid S (2001) Temporal analysis of the Brussels flora as indicator for changing environmental quality. Landscape Urban Plan 52:203–224
Hill MO (1979a) DECORANA - A Fortran program for detrended correspondence analysis and reciprocal averaging, Ecology and Systematics. Comell University, Ithaca
Hill MO (1979b) TWINSPAN - A Fortran program for arranging multivariate data in an ordered two-way table by classification of the individuals and attributes. Comell University, Ithaca
Himiyama Y (1995) Kokudo riyou henka no gaiyou (Outlines of national landuse changes). In: Nishikawa O (ed) Atlas - environmental changes in modern Japan. Asakura shoten, Tokyo, pp 1–16 (in Japanese)
Hothorn T, Bretz F, Westfall P (2008) Simultaneous Inference in General Parametric Models. Biometrical J 50:346–363
Hovd H (2008) Extensively managed strips in intensively cultivated grasslands as possible contributors to increased plant species richness. Acta Agr Scand B-S P 58:43–50
Ide M (2006) Relationships of rural landscape structure and biological diversity in Landscape Ecology. Landscape Ecol Mgmt 10:71–74 (in Japanese)
Inui T (1992) Geographical study on the use of the plain forests in the Kanto plain, Kokonshoin, Tokyo (in Japanese)
Jantunen J, Saarinen K, Valtonen A, Saarnio S (2007) Flowering and seed production success along roads with different mowing regimes. Appl Veg Sci 10:285–292
Japan Meteorological Agency (2007) Monthly Data in an Average Year (1971–2000) at the Tsukuba Meteorological Station. <http://www.data.jma.go.jp/jma/indexe.html>.
Japan Wildlife Research Center (2006) Monitoring Sites 1000, Biodiversity Center Japan, Fuji-Yoshida (in Japanese)
Koyanagi T, Kusumoto Y, Yamamoto S, Okuro T, Ide M, Takeuchi K (2007) Comparison of the species compositions between past and present semi-natural Miscanthus sinensis grassland in the Kanto plain. Landscape Res Jap 70:439–444 (in Japanese with English abstract)
Koyanagi T, Kusumoto Y, Yamamoto S, Okubo S, Takeuchi K (2009) Historical impacts on linear habitats: the present distribution of grassland species in forest-edge vegetation. Biol Conserv 142:1674–1684
Koyanagi T, Kusumoto Y, Yamamoto S, Okubo S, Iwasaki N, Takeuchi K (2012) Grassland plant functional groups exhibit distinct time-lags in response to historical landscape change. Plant Ecol 213:327–338
Kuussaari M, Bommarco R, Heikkinen RK, Helm A, Krauss J, Lindborg R, Ockinger E, Partel M, Pino J, Roda F, Stefanescu C, Teder T, Zobel M, Steffan-Dewenter I (2009) Extinction debt: a challenge for biodiversity conservation. Trends Ecol Evol 24:564–571
McCune B, Mefford MJ (1999) PC-ORD: multivariate analysis of ecological data. Version 4. User’s guide. MjM Software Design, Gleneden Beach
McGranahan G, Marcotullio P, Millennium Ecosystem Assessment (2005) Urban systems. In: Ecosystems and Human Well-Being: Current State and Trends. Island Press, Washington, pp 795–825
Ministry of the Environment (2000) The red data book, Japan – vascular plants –. Nature Conservation Bureau, Ministry of the Environment, Tokyo
Ministry of the Environment (2002) National Strategy for the Conservation and Sustainable Use of Biological Diversity (2002 Version). Ministry of the Environment, Government of Japan, Tokyo
Miyawaki A (1977) Flora of Japan. Gakken, Tokyo
Miyawaki A (ed.) (1986) Vegetation of Japan. Kanto, vol.7, Shibundo, Tokyo (in Japanese)
Miyawaki A, Suzuki K (1974) Flora of Chiba city. Chiba city, Chiba (in Japanese)
Miyawaki A, Okuda S, Suzuki K (1975) Report of vegetation survey in the district of Chihara-dai, in the south-eastern part of Chiba prefecture. Japanese public housing corporation, Tokyo
Mueller-Dombois D, Ellenberg H (1974) Aims and methods of Vegetation Ecology. John Wiley & Sons, Inc, New York
Muratet A, Porcher E, Devictor V, Arnal G, Moret J, Wright S, Machon N (2008) Evaluation of floristic diversity in urban areas as a basis for habitat management. Appl Veg Sci 11:451–460
Noordijk J, Schaffers AP, Heijerman T, Sýkora KV (2011) Using movement and habitat corridors to improve the connectivity for heathland carabid beetles. J Nat Conserv 19:276–284
Okubo K (2002) The present state in the study of biological diversity on semi-natural grassland in Japan. J Jap Grassl Sci 48:268–276 (in Japanese with English abstract)
Okubo S, Kamiyama A, Kitagawa Y, Yamada S, Palijon A, Takeuchi K (2005) Management and micro-scale landform determine the ground flora of secondary woodlands and their verges in the Tama Hills of Tokyo, Japan. Biodivers Conserv 14:2137–2157
Puppim de Oliveira JA, Balaban O, Doll CNH, Moreno-Peñaranda R, Gasparatos A, Iossifova D, Suwa A (2011) Cities and biodiversity: Perspectives and governance challenges for implementing the convention on biological diversity (CBD) at the city level. Biol Conserv 144:1302–1313
Pysek P (1998) Alien and native species in Central European urban floras: a quantitative comparison. J Biogeogr 25:155–164
Ramalho CE, Hobbs RJ (2011) Time for a change: dynamic urban ecology. TREE. doi:10.1016/j.tree.2011.10.008
Sakiyama N, Itoga R (1994) On the change of grasslands into Pine forests in the Inashiki upland. Ibaraki prefecture. Env Res Tsukuba 15:29–44 (in Japanese with English abstract)
Sakurai T (1982) Fundamental study on vegetation management and utilization forms of Pinus densiflora lowland woodland. Graduation thesis, University of Tsukuba, Tsukuba
Sandström UG, Angelstam P, Mikusinski G (2006) Ecological diversity of birds in relation to the structure of urban green space. Landscape Urban Plan 77:39–53
Sattler T, Duelli P, Obrist MK, Arlettaz R, Moretti M (2010) Response of arthropod species richness and functional groups to urban habitat structure and management. Landscape Ecol 25:941–954
Shoji A (2003) Landscape ecological studies for semi-natural grassland conservation. Grassland Sci 48:557–563 (in Japanese with English abstract)
Sprague DS (2001) Monitoring habitat change in Japanese agricultural systems. In: OECD (ed) Agriculture and Biodiversity – Developing Indicators for policy analysis. Publications, Paris, pp 168–179
Tait CJ, Daniels CB, Hill RS (2005) Changes in species assemblages within the Adelaide metropolitan area, Australia, 1836–2002. Ecol Appl 15:346–359
Takeuchi K (2006) Landscape Ecology. Asakura shoten, Tokyo
Takeuchi K, Brown RD, Washitani I, Tsunekawa A, Yokohari M (2003) Satoyama: The traditional rural landscape of Japan. Springer, Tokyo
The Satoyama Initiative (2010) Biodiversity and livelihoods - The satoyama Initiative concept in practice -. United Nations University Institute of Advanced Studies and Ministry of the Environment of Japan, Tokyo < http://satoyama-initiative.org/wp-content/uploads/2011/09/biodiversity_booklet_en_web.pdf>
Thiele J, Otte A (2008) Invasion patterns of Heracleum mantegazzianum in Germany on the regional and landscape scales. J Nat Conserv 16:61–71
Tikka PM, Koski PS, Kivela RA, Kuitunen MT (2000) Can grassland plant communities be preserved on road and railway verges? Appl Veg Sci 3:25–32
Werner P (2011) The ecology of urban areas and their functions for species diversity. Landscape Ecol Eng 7:231–240
Yamamoto S, Itoga R (1988) Forest forms and the distribution of Pinus densiflora plain forest in southwest part of Ibaraki prefecture. Landscape Res Jap 51:150–155 (in Japanese with English abstract)
Yamasaki H, Aoki K, Hattori T, Takeda Y (2000) Increase of species diversity due to management of vegetation in Satoyama. Landscape Res Jap 63:481–484
Acknowledgments
We thank K. Fujiwara and T. Fuji, who compiled the regional flora, for providing detailed information on the historic vegetation data. T. Nakamura kindly provided additional historic vegetation data in the study area. We also thank S. Yamada and Y. Tokuoka for helping with vegetation surveys. T. Furukawa kindly edited English and gave us informative comments to improve our manuscript. This study was financially supported by the 21st Century COE Program, Biodiversity and Ecosystem Restoration, with additional support from JSPS research fellowship to T. Koyanagi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koyanagi, T., Kusumoto, Y., Yamamoto, S. et al. Potential roles of small and linear habitat fragments in satoyama landscapes for conservation of grassland plant species. Urban Ecosyst 15, 893–909 (2012). https://doi.org/10.1007/s11252-012-0253-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-012-0253-4