Abstract
Urbanization is recognized as a major threat to biodiversity. Within an urbanized area, habitats such as forests and grasslands are often fragmented. Matrixes between fragmented habitats can influence the biodiversity because some of these can act as habitats for some species (hospitable matrixes). The importance of matrixes for biodiversity within the landscape, i.e., the fragmented habitat and matrix, can be influenced by the dispersal ability of the species within the landscape because this can affect the availability of the hospitable matrix. In this study, we evaluated the effects of both local conditions and the surrounding matrix on the species diversity of two taxa with different dispersal abilities, i.e., Carabidae, which has relatively low dispersal ability, and Heteroptera, which has relatively high dispersal ability, in the grassland of urban parks in Tokyo, the most populated region in Japan. The diversity of Carabidae was not strongly influenced by the local habitat size or the amount of surrounding hospitable matrix; however, Heteroptera was strongly influenced by both the local habitat size and amount of surrounding hospitable matrix. Thus, fragmented small habitats might have higher habitat quality depending on the surrounding hospitable matrix for species with relatively high dispersal ability. To promote biodiversity conservation in urban parks, both local habitat management and matrix management are required.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Urbanization is a major anthropogenic alteration to terrestrial ecosystems, and it constitutes a major threat to biodiversity (Beninde et al. 2015; Newbold et al. 2015; Fenoglio et al. 2020). Urbanized areas represent complex and heterogeneous systems that are characterized by fragmented, isolated, and degraded natural habitats (Grimm et al. 2008; Pickett et al. 2011). Urbanization is rapidly increasing around the world (Seto et al. 2011, 2012); thus, understanding its effects on diverse taxonomic and functional groups is essential to improving biodiversity conservation practices (Dearborn and Kark 2010).
Within an urbanized area, habitats such as forests and grasslands are often fragmented (Fischer and Lindenmayer 2007; Öckinger et al. 2009). Habitat fragmentation can cause loss of biodiversity due to increased extinction risks that can arise because of many changes including reduced population sizes, decreased gene flow, and changes to interspecific interactions (Jules and Shahani 2003; Lindenmayer and Fischer 2007). Fragmented habitats are considered analogous to islands, and the newly created area between the fragmented habitats, which is known as a “matrix,” is analogous to an ocean; the matrix is often treated as ecologically uniform and therefore unimportant to the biodiversity of fragmented habitats (Jules and Shahani 2003; Öckinger et al. 2009). The matrix, however, is typically nonuniform; i.e., it can contain several types of habitat that can influence the biodiversity of the fragmented habitats (Wiegand et al. 2005; Ewers and Didham 2006; Tscharntke et al. 2012). For example, some fragment-dwelling species could potentially compensate for habitat loss using resources in the matrix (Ewers and Didham 2006); thus, it would not be possible to strictly divide the habitat and matrix for such species (Walter 2004; Ewers and Didham 2006).
Dispersal patterns among fragmented habitats, which could potentially influence the population viability of particular species, can depend on whether the matrix is similar to the habitat of the species (i.e., a hospitable matrix) or distinct from the habitat (i.e., an inhospitable matrix) (Nowicki et al. 2014). For example, black bears can use a matrix as a corridor, but the matrix could have a different resistance value to pass that which clearly shows genetic similarity among local populations (Ohnishi et al. 2019). In addition, the population size of harvest mice is strongly influenced by several matrix types that surround their habitat and can enhance the structure of their metapopulation (Kuroe et al. 2011). In contrast, the species diversity of ants, beetles, and spiders, which have relatively low dispersal abilities, is not strongly influenced by their respective matrixes, although it is influenced by their local habitat conditions (Peng et al. 2020). Thus, the importance of the matrix for biodiversity within a landscape, i.e., the fragmented habitat and its matrix, could potentially be influenced by the dispersal ability of the specific species (Kuefler et al. 2010; Nowicki et al. 2014). For instance, if the fragmented habitat was surrounded by a hospitable matrix, the diversity of species with relatively high dispersal abilities could be high even if the habitat was small in size. Conversely, such a pattern should not be found for species with low dispersal ability as their species diversity is determined by the quality of the fragmented habitat. Understanding the importance of both the matrix and local condition is therefore essential to improving the conservation of urban biodiversity (Angold et al. 2006).
In this study, we evaluated the effects of green space in urban parks and their surrounding matrixes on the species diversity and abundance of two taxa with different dispersal abilities: Carabidae and Heteroptera. Carabids are often called ground beetles, some of which cannot fly or have limited flying ability (Shibuya et al. 2018), and considered poorer dispersers because of their “earthbound” way of life (Niemelä 2001). Heteroptera, often called stink bugs, typically have flying ability and are able to fly long distances (Pease and Zalom 2010; Osawa et al. 2018). Green space in urban parks such as secondary forest and gardens plays an important role in the biodiversity of urban areas; thus, it has long been the focus of related studies (Angold et al. 2006; Pickett et al. 2011; Soga et al. 2014; Villaseñor and Escobar 2019). Urban areas are often fragmented but are surrounded by several types of habitat other than parks, e.g., gardens and roadside verges (Angold et al. 2006; Soga et al. 2014). We hypothesized that these types of scattered habitats could act as hospitable matrixes that promote biodiversity in urban parks. If the diversity of Carabidae, which have relatively low dispersal ability, is strongly influenced by the local green space but not by the surrounding hospitable matrix, this would support our hypothesis. In contrast, if the diversity of Heteroptera, which have relatively high dispersal ability, was influenced not only by local green space but also by the surrounding hospitable matrix, our hypothesis would be supported. To test the hypothesis, we focused on the green space in urban parks and the surrounding matrix of the urbanized region.
Material and methods
Study area
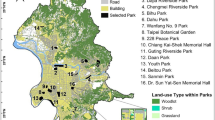
The study was conducted at 11 urban parks in the west region of Tokyo, Japan (Fig. 1). Tokyo is the most populated prefecture in Japan. This region has a wide range of residential areas with many urban parks that include broadleaf forests and grassland, which are habitat types that existed before the developed habitats, mainly semi-natural ecosystems that were fragmented. The basic attributions of the 11 urban parks are shown in Table 1. Among the 11 urban parks, Tamagawa Ryokuchi Park (G) and Kodaira Chuou Park (D) had the largest and smallest areas, respectively (Table 1). Both Tamagawa Ryokuchi Park (G) and Tamagawa Gohonmatsu Park (F) were occupied mostly by forest, whereas Naganuma Park (H) was dominated by grassland (Table 1). The land cover other than forest and grassland included “artificial land” such as open ground, open water, and buildings.
Study species and insect collection
We collected Carabidae and Heteroptera species because these two groups have different dispersal abilities and were the general insect groups found in the studied urban parks. For collection in each of the 11 parks, we placed five line transects (4 × 15 m each), each > 50 m from the other, in the grassland which covered by grass species only, and not covered by tree canopy. We focused on grassland habitat because gardens and roadside verges have similar grasslands that may serve as hospitable matrixes. The dominant plants in the transects were all grasses; Miscanthus sinensis, Setaria viridis, Pleioblastus chino, Solidago canadensis var. scabra, Conyza canadensis, Conyza sumatrensis, Sicyos angulatus, and Pueraria montana var. lobata. These species were common in all of the grassland including gardens and roadside verges in the study area.
We sampled Carabidae using pitfall traps. To ensure that the adult stage of the species was collected as much as possible, sampling was conducted from September to October 2020, which is late summer to early fall in the region. We buried transparent plastic cups (5 cm in diameter and 10 cm in depth) within line transects without attractants. Traps were not set in areas with no vegetation (bare ground). One line transect had five traps spaced 3 m apart from each other; resulting in 25 traps in total in each park. These traps were collected 3 days after they had been established. Individuals caught in the traps were fixed using 70% ethanol in the field, after which they were dried, mounted, and identified to the species level using reference material (Ueno et al. 1985) in the laboratory.
Heteroptera were collected using sweep nets (50 cm in diameter with a 0.3-mm mesh). We conducted sweeps while slowly walking the transects within grass vegetation. For each line transect, we conducted 30 sweeps; hence, 150 sweeps were performed in each park. All collected arthropods were placed into a sealable plastic bag containing 70% ethanol in the field. In the laboratory, the collected stink bugs were subsequently dried, mounted, and identified to the species level using reference material (Yasunaga et al. 1993, 2001).
Establishment of landscape data
We collected land cover data from the area to evaluate both the quality and quantity of the habitat and matrix. We used GIS data provided by the Tokyo city planning department to quantify the area of the urban parks and habitats, namely, forest and grassland. Although the GIS data differed in some ways from current land cover due to the year in which they were collected, we improved these data using air photo interpretation from the latest Geospatial information Authority of Japan map photos (taken December 21, 2016; https://maps.gsi.go.jp/). Using the updated GIS data, we calculated the total area, forest area, i.e. trees dominated area, and grassland area, i.e. grasses dominated area, in each urban park.
To evaluate the hospitable and inhospitable matrixes in the surrounding landscape, i.e., the quality of matrixes for 11 urban parks, we used the kernel Normalized Difference Vegetation Index (kNDVI), which involved using remote sensing measurements to assess the contents of the live green vegetation (Camps-Valls et al. 2021). Compared with NDVI, which is the most well-known vegetation index used to represent the vegetation conditions on terrestrial surfaces, kNDVI has greater resistance to saturation, bias, and complex phenological cycles, and it shows enhanced robustness to noise and stability across spatial and temporal scales (Camps-Valls et al. 2021). We expected the kNDVI to appropriately represent the vegetation condition and allow us to build the hospitable matrixes with minimal noise and uncertainty.
The kNDVI was analyzed from the atmospherically corrected surface reflectance observed by Landsat 8 OLI/TIRS sensors at approximately 16-day intervals with a spatial resolution of 30 m (data collected in 2020). We calculated the annual mean of the kNDVI within the study area and used this in subsequent analyses (Fig. 2a).
We also calculated the range of the kNDVI for the grassland in the studied urban parks. First, we overlayed the GIS polygon data for grassland in the urban parks with the kNDVI raster data; subsequently, we calculated both the average and standard deviation (SD) values of the kNDVI in the grassland. Using these values, we reclassed the kNDVI raster data to a binary image that did or did not match the grassland range. Specific kNDVI values that included the values of grassland in the urban parks (average ± SD) were defined as the hospitable matrix for grassland species (value = 1), whereas the other cells were defined as inhospitable matrix (value = 0) (Fig. 2b).
To quantify the hospitable matrix around each urban park, we used three buffer sizes from the outline of each park: 250, 500, and 1,000 m. We calculated the number cells with hospitable matrix for each park and for each buffer size. These values were used to represent the quality of the surrounding matrix in each urban park.
Statistical analysis
First, we conducted single regression analysis for the number of species and individuals of both Carabidae and Heteroptera in each urban park using a generalized linear model (GLM) with Poisson distribution (log link) and a Wald test, and we calculated Akaike's information criterion (AIC) to evaluate the effects of the local condition. The explanatory variables were the total areas, forest areas in the urban park, grassland areas, or total greenspace (forest plus grassland) in the urban park. All explanatory variables were log-transformed.
Second, we conducted single regression analysis for the same response variables using a GLM with Poisson distribution (log link) and a Wald test, and we calculated AIC to evaluate the matrix effects using command “glm.” In this analysis, the explanatory variables were the number of hospitable matrixes within each of the 250-, 500-, or 1,000-m buffers. For the number of hospitable matrixes, we used kNDVI-based binary values. All statistical analyses were conducted using the statistical package R version 3.6.1 (R Core Team 2019, R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria, https://cran.r-project.org/).
Results
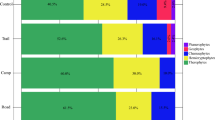
We collected 503 individuals of Carabidae of 41 species that included morphological species i.e., that could be decided different species from other but could not identify the species (32 species identified to the species level, 5 species identified to genus level). Within these 37 species, 7 species clearly had no flying ability (Appendix Table 3). Among the 11 parks, Kodaira (D) and Tamagawa Ryokuchi (G) parks has the most species (12 species), whereas Tamagawa Gohonmatsu Park (F) has the most individuals (178 individuals) (Table 1).
We collected 1,568 individuals of 58 species of Heteroptera, which included morphological species at the family level (36 species identified to the species level, 5 species identified to genus level). All species had wings. Among the 11 parks, Tamagawa Ryokuchi Park (G) has the most species (22 species), whereas Nogawa Park (J) had the most individuals (419 individuals) (Table 1).
For the number of Carabidae species, none of the explanatory variables were significantly correlated (Table 2). However, the number of Carabidae individuals was negatively correlated with both forest area and total greenspace area but positively correlated with grassland area. For the number of Carabidae species and individuals, the lowest AIC model had forest area as an explanatory variable. For the number of Heteroptera species, only grassland area was positively correlated, whereas the number of Heteroptera individuals was positively correlated with both grassland and total greenspace areas. For both the number of Heteroptera species and individuals, the lowest AIC model had grassland area as an explanatory variable.
The number of Carabidae species was not significantly correlated with the hospitable matrix (Table 3). However, the number of Carabidae individuals was negatively correlated with both the 500- and 1,000-m kNDVI. Among the three spatial variables, the lowest AIC for Carabidae was from the 1,000-m buffer in all cases. For Heteroptera, the hospitable matrixes were positively correlated with both the number of species and individuals; compared with the 1,000-m buffer, the 250-m buffer had lower AIC values for the number of species and the and 500-m had a lower AIC value for individuals. For the number of species, the AIC values were almost identical for the 250- and 500-m models.
Comparing the AIC from the within-park and hospitable matrix analyses, the AIC values for the numbers of Carabidae and Heteroptera species were similar (almost within a difference of 2; Tables 2 and 3). In contrast, the number of Carabidae individuals from the within-park analysis had a lower AIC value than that of Carabidae individuals related to the hospitable matrix, whereas the number of Heteroptera individuals from the within-park data had a higher AIC value than that of Heteroptera individuals related to the hospitable matrix (Tables 2 and 3).
Discussion
In this study we assessed the importance of local (patch effects) and landscape (matrix effects) factors for the species diversity and abundance in urban parks in Tokyo. We hypothesized that scattered green spaces around parks act as hospitable matrixes for some species diversity depending on their dispersal abilities. Results showed that the diversity of Carabidae, which has relatively low dispersal ability, was not strongly influenced by the local patch size or the amount of surrounding hospitable matrix. However, the diversity of Heteroptera, which has relatively high dispersal ability, was affected by both the local patch size and amount of surrounding hospitable matrix. Therefore, the data supported our hypothesis. Thus, the biodiversity of urban parks is apparently influenced by both local and landscape factors.
Biodiversity in the urban parks
In the 11 urban parks, we collected 503 Carabidae individuals including 41 species including morphological species (at the family level) in 275 traps; we collected 1,568 Heteroptera species including 58 species also including morphological species (at the family level) in 1,650 sweeps. On average, we collected 1.8 Carabidae in a trap and 0.94 Heteroptera in a sweep. These results indicate that urban parks have an important role to play in maintaining the regional biodiversity of both Carabidae and Heteroptera species. Previous studies have shown that urban green space is important for birds (Padilla and Rodewald 2015), mammals (Ofori et al. 2018), amphibian (Niemeier et al. 2020), insects (Angold et al. 2006; Öckinger et al. 2009; Peng et al. 2020), and plant species (Angold et al. 2006). Thus, urban green space could serve as refuges for several species (Niemeier et al. 2020). Indeed, green space in urban parks is known to be an important habitat for several taxa within urbanized areas (Angold et al. 2006; Ofori et al. 2018).
In the present study, the largest parks were 25 times larger than the smallest parks, and some parks were dominated by grassland, while some were dominated by forest. The parks F (Tamagawa Gohonmatsu park) and G (Tamagawa Ryokuchi park) were located at the riverside with long and thin shapes, which might have many ecotones, habitat edges that strongly influence for the regional biodiversity (Kotze and Samways 2001; Pryke and Samways 2012). Whatever influence these features may have had on Carabidae and Heteroptera, the effect of park and matrix size and configuration was present in the results.
The importance of local habitat sites for biodiversity
Both the number of Carabidae and Heteroptera individuals and the number of Heteroptera species were positively correlated with the grassland area in the parks. These results indicate the importance of habitat size for biodiversity, which is rooted in the island biogeography theory (Doak and Mills 1994; Jules and Shahani 2003). They also show that forest and grassland play different roles in the biodiversity of urban parks. In the present study, we collected insects from grassland in the urban parks only; thus, the size of the grassland was the most important factor for the diversity of the collected species. Although both forest and total greenspace (forest plus grassland) were negatively correlated with the number of individual Carabidae, this result is consistent with the results for the grassland area because urban parks with large forest areas tended to have relatively small grassland areas. Therefore, if we collected insects from forests, forest size might be revealed as the most important factor. Consequently, urban parks with multiple habitat types would likely have high biodiversity. Indeed, habitat heterogeneity in urban parks could contribute to the total biodiversity of the parks.
The number of Carabidae species was not influenced by the size of grassland in parks. Carabidae species are considered to have relatively low dispersal ability (Niemelä 2001); consequently, they are often used as environmental indicators (Osawa et al. 2020). In fact, some of the collected Carabidae species in this study (genus Carabus, Pterostichus, and Myas) have brachypterous hindwings and no flying ability (Appendix Table 3). These relatively large, flightless carabids are vulnerable to habitat fragmentation and disturbance (Niemelä 2001; Ribera et al. 2001; Rainio and Niemelä 2003). Conversely, many Carabid groups with macropterous hindwings and potential for flight were identified, but not all these species have sufficient flight ability, as suggested by the wing polymorphism in Chlaenius naeviger and the lack of flight muscles in some Harpalinae (Shibuya et al. 2018). Therefore, parks with high Carabidae species diversity should have suitable habitats with large sizes in theory. One possible explanation for our results is the historical effects of land cover/use because the species diversity of beetles is known to be strongly affected by land history (Cui et al. 2014). Additionally, regions with stable habitats could help conserve Carabidae communities (Osawa et al. 2014). The urban parks studied here might have had different land cover/use histories, which may have included substantial changes over time. Another possible explanation is the specific habitat quality in each urban park. A previous study suggested that the number of ground beetle species is affected by the successional stage of a habitat (Small et al. 2002), habitat shapes that influence for the edge to area ratio (Usher et al. 1993; Niemelä 2001). We did not evaluate land history or the specific quality of grassland habitats, so we may have overlooked their importance to our results. In future research, determining the effects of factors such as local habitat condition on species diversity will represent an important challenge.
The importance of the surrounding hospitable matrix for biodiversity
Similar to the size of the grassland area in parks, the surrounding hospitable matrix influenced both the number of Heteroptera species and individuals in urban parks, but it did not affect the numbers of Carabidae. These results support our hypothesis that a fragmented habitat surrounded by hospitable matrix leads to high biodiversity of species with relatively high dispersal ability but does not affect those species with low dispersal ability. Heteroptera have relatively high dispersal ability; thus, they might use the hospitable matrix as a dispersal corridor or habitat, or both. In fact, a previous study suggested that the mirid bug Stenotus rubrovittatus, which was collected in the current study, might expand on a large spatial scale along roads with small grassland areas (Osawa et al. 2018). The hospitable matrix therefore plays an important role in the biodiversity of a fragmented habitat, but this seems to apply only to species with relatively high dispersal ability.
The most effective spatial scales on which to measure the effects of the hospitable matrix on the numbers of Heteroptera species and individuals were 250 and 500 m. Within related species groups, the dispersal abilities of Heteroptera differed among species. Therefore, Heteroptera species with relatively low dispersal abilities likely depended on the hospitable matrix on a small scale, whereas species with relatively high dispersal abilities depended on the hospitable matrix on a large scale. Indeed, the collected Heteroptera included both small and large species with different dispersal abilities. For example, Cletus punctiger, which was relatively common within our collected species, can fly for approximately 2 h continuously at ~ 45 m/min on the average over 5 km in theory (Ito 1980). In contrast, Stenotus rubrovittatus, which was relatively infrequently found within our collected species, can disperse over ~ 72–122 m (Takeda et al. 2014). Our results might reveal that 250–500 m could include the several dispersal abilities of Heteroptera species.
For Carabidae, the hospitable matrix within both the 500- and 1,000-m negatively influenced the numbers of individuals. Carabidae species have relatively low dispersal ability so they might not be affected by large scale hospitable matrix directly. One possible explanation for that is natural enemy with relatively high dispersal abilities, e.g., Nyctereutes procyonoides and other mid-sized mammals. At the feeding habitat of N. procyonoides near the study area, the predators often feed on insects such as coleopterans, especially in summer (Takatsuki 2017; Takatsuki et al. 2020). Although the Carabidae themselves did not use the surrounding matrix on a large scale directly, their natural enemies that prefer grassland habitats were able to do so. The hospitable matrix around the urban parks might influence Heteroptera directly, whereas it might influence Carabidae indirectly.
In our study region, hospitable matrix is common, especially in the western areas. Importantly, it is not always true that large parks have large hospitable matrixes around them. Therefore, species diversity in each park might not be decided by the condition of the park only for both Heteroptera and Carabidae species. Our results suggested that conservation efforts invested in urban parks only and without concern for the surrounding matrix are insufficient for biodiversity conservation.
Conclusions
Both local habitats and the surrounding matrix contributed to the biodiversity of urban parks in the Tokyo, at least for Heteroptera species that have relatively high dispersal ability. Thus, species that can use the matrix effectively should be relatively more tolerant to urbanization of landscapes. In contrast, the matrix did not influence species with a low dispersal ability that could not use the matrix effectively. Moreover, the matrix might negatively contribute to the abundance of such species due to its use by natural enemies. Fragmented small habitats might be higher-quality habitats for species that can disperse relatively well depending on the surrounding hospitable matrix. Both large areas and favorable surrounding matrix are therefore required to improve biodiversity conservation because they could work synergistically (Kremen 2015). To promote biodiversity conservation in urban parks, both local habitat management and matrix management should be considered.
Our results indicated the importance of matrixes that are similar to grassland because we focused only on grassland habitats in urban parks and matrixe habitats that were similar to grassland; i.e., we did not assess forests. In the study region (the Kanto region of Japan), continuous management (e.g., mowing) is required to maintain the grassland because of climatic climax that occurs under temperate and humid conditions (Ushimaru et al. 2018). Indeed, maintaining such scattered grassland areas, including urban farmland, roadsides, and private gardens, is important for improving biodiversity in urban parks in heavily urbanized regions.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Code availability
Not applicable.
References
Angold PG, Sadler JP, Hill MO et al (2006) Biodiversity in urban habitat patches. Sci Total Environ 360:196–204. https://doi.org/10.1016/j.scitotenv.2005.08.035
Beninde J, Veith M, Hochkirch A (2015) Biodiversity in cities needs space: A meta-analysis of factors determining intra-urban biodiversity variation. Ecol Lett 18:581–592. https://doi.org/10.1111/ele.12427
Camps-Valls G, Campos-Taberner M, Moreno-Martínez Á et al (2021) A unified vegetation index for quantifying the terrestrial biosphere. Sci Adv 7:eabc7447
Cui Q-Y, Gaillard M-J, Lemdahl G et al (2014) Historical land-use and landscape change in southern Sweden and implications for present and future biodiversity. Ecol Evol 4:3555–3570
Dearborn DC, Kark S (2010) Motivaciones para conservar la biodiversidad urbana. Conserv Biol 24:432–440. https://doi.org/10.1111/j.1523-1739.2009.01328.x
Doak DF, Mills LS (1994) A useful role for theory in conservation. Ecology 75:615–626
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev 81:117–142
Fenoglio MS, Rossetti MR, Videla M (2020) Negative effects of urbanization on terrestrial arthropod communities: A meta-analysis. Glob Ecol Biogeogr 29:1412–1429. https://doi.org/10.1111/geb.13107
Fischer J, Lindenmayer DB (2007) Landscape modification and habitat fragmentation: a synthesis. Glob Ecol Biogeogr 16:265–280
Grimm NB, Faeth SH, Golubiewski NE et al (2008) Global change and the ecology of cities. Science (80-) 319:756–760. https://doi.org/10.1126/science.1150195
Ito K (1980) Seasonal change of flight ability of Cletus punctiger (Heteroptera: Coreidae). Appl Entomol Zool 15:36–44
Jules ES, Shahani P (2003) A broader ecological context to habitat fragmentation: Why matrix habitat is more important than we thought. J Veg Sci 14:459–464. https://doi.org/10.1111/j.1654-1103.2003.tb02172.x
Kotze DJ, Samways MJ (2001) No general edge effects for invertebrates at Afromontane forest/grassland ecotones. Biodivers Conserv 10:443–466
Kremen C (2015) Reframing the land-sparing/land-sharing debate for biodiversity conservation. Ann N Y Acad Sci 1355:52–76. https://doi.org/10.1111/nyas.12845
Kuefler D, Hudgens B, Haddad NM et al (2010) The conflicting role of matrix habitats as conduits and barriers for dispersal. Ecology 91:944–950
Kuroe M, Yamaguchi N, Kadoya T, Miyashita T (2011) Matrix heterogeneity affects population size of the harvest mice: Bayesian estimation of matrix resistance and model validation. Oikos 120:271–279. https://doi.org/10.1111/j.1600-0706.2010.18697.x
Lindenmayer DB, Fischer J (2007) Tackling the habitat fragmentation panchreston. Trends Ecol Evol 22:127–132. https://doi.org/10.1016/j.tree.2006.11.006
Newbold T, Hudson LN, Hill SLL et al (2015) Global effects of land use on local terrestrial biodiversity. Nature 520:45–50. https://doi.org/10.1038/nature14324
Niemeier S, Müller J, Struck U, Rödel MO (2020) Superfrogs in the city: 150 year impact of urbanization and agriculture on the European Common Frog. Glob Chang Biol 26:6729–6741. https://doi.org/10.1111/gcb.15337
Niemelä J (2001) Carabid beetles (Coleoptera: Carabidae) and habitat fragmentation: A review. Eur J Entomol 98:127–132. https://doi.org/10.14411/eje.2001.023
Nowicki P, Vrabec V, Binzenhöfer B et al (2014) Butterfly dispersal in inhospitable matrix: Rare, risky, but long-distance. Landsc Ecol 29:401–412. https://doi.org/10.1007/s10980-013-9971-0
Öckinger E, Dannestam Å, Smith HG (2009) The importance of fragmentation and habitat quality of urban grasslands for butterfly diversity. Landsc Urban Plan 93:31–37
Ofori BY, Garshong RA, Gbogbo F et al (2018) Urban green area provides refuge for native small mammal biodiversity in a rapidly expanding city in Ghana. Environ Monit Assess 190:1–11
Ohnishi N, Osawa T, Yamamoto T, Uno R (2019) Landscape heterogeneity in landform and land use provides functional resistance to gene flow in continuous Asian black bear populations. Ecol Evol 9:4958–4968
Osawa T, Watanabe K, Ikeda H, Yamamoto S (2014) New approach for evaluating habitat stability using scarce records for both historical and contemporary specimens: a case study using Carabidae specimen records. Entomol Sci 17:425–431. https://doi.org/10.1111/ens.12075
Osawa T, Yamasaki K, Tabuchi K et al (2018) Detecting crucial dispersal pathways using a virtual ecology approach: A case study of the mirid bug Stenotus rubrovittatus. Ambio 47:806–815
Osawa T, Yoshimatsu S-I, Nakatani Y (2020) Specimen-based records and geographic locations of carabid beetles (Coleoptera) collected mainly by Dr. Kazuo Tanaka. Ecol Res. https://doi.org/10.1111/1440-1703.12167
Padilla BJ, Rodewald AD (2015) Avian metapopulation dynamics in a fragmented urbanizing landscape. Urban Ecosyst 18:239–250. https://doi.org/10.1007/s11252-014-0390-z
Pease CG, Zalom FG (2010) Influence of non-crop plants on stink bug (Hemiptera: Pentatomidae) and natural enemy abundance in tomatoes. J Appl Entomol 134:626–636
Peng MH, Hung YC, Liu KL, Neoh KB (2020) Landscape configuration and habitat complexity shape arthropod assemblage in urban parks. Sci Rep 10:1–12. https://doi.org/10.1038/s41598-020-73121-0
Pickett STA, Cadenasso ML, Grove JM et al (2011) Urban ecological systems: Scientific foundations and a decade of progress. J Environ Manage 92:331–362. https://doi.org/10.1016/j.jenvman.2010.08.022
Pryke JS, Samways MJ (2012) Conservation management of complex natural forest and plantation edge effects. Landsc Ecol 27:73–85
Rainio J, Niemelä J (2003) Ground beetles (Coleoptera: Carabidae) as bioindicators. Biodivers Conserv 12:487–506
Ribera I, Dolédec S, Downie IS, Foster GN (2001) Effect of land disturbance and stress on species traits of ground beetle assemblages. Ecology 82:1112–1129
Seto KC, Fragkias M, Güneralp B, Reilly MK (2011) A meta-analysis of global urban land expansion. PLoS ONE 6:e23777
Seto KC, Güneralp B, Hutyra LR (2012) Global forecasts of urban expansion to 2030 and direct impacts on biodiversity and carbon pools. Proc Natl Acad Sci U S A 109:16083–16088. https://doi.org/10.1073/pnas.1211658109
Shibuya S, Kiritani K, Fukuda K (2018) Hind wings in ground beetles (Coleoptera: Carabidae and Brachinidae) – morphology, length, and characteristics of each subfamily. Japanese J Ecol 68:19–41 (in Japanese)
Small EC, Sadler JP, Telfer MG (2002) Carabid beetle assemblages on urban derelict sites in Birmingham, UK. J Insect Conserv 6:233–246
Soga M, Yamaura Y, Koike S, Gaston KJ (2014) Land sharing vs. land sparing: does the compact city reconcile urban development and biodiversity conservation? J Appl Ecol 51:1378–1386
Takatsuki S (2017) Food habits of the raccoon dog at the Tsuda University’s Kodaira Campus, western Tokyo. Humans Nat 28:1–10 ((in Japanese))
Takatsuki S, Yamasaki I, Shirai S (2020) Food habits of raccoon dogs in Ura-Takao, a non-urban area, west of Tokyo. Mamm Sci 60:85–93 (in Japanese)
Takeda A, Yasuda M, Tabuchi K et al (2014) Pattern of immigration of the sorghum plant bug Stenotus rubrovittatus (Hemiptera: Miridae) into paddy fields from a large source habitat. Appl Entomol Zool 49:139–147
Tscharntke T, Tylianakis JM, Rand TA et al (2012) Landscape moderation of biodiversity patterns and processes - eight hypotheses. Biol Rev 87:661–685. https://doi.org/10.1111/j.1469-185X.2011.00216.x
Ueno S, Kurosawa Y, Sato M (1985) The Coleoptera of Japan in Color, Vol. II. Hoikusha, Higashi-Osaka. 514 pp. (in Japanese)
Usher MB, Field JP, Bedford SE (1993) Biogeography and diversity of ground-dwelling arthropods in farm woodlands. Biodivers Lett 54–62
Ushimaru A, Uchida K, Suka T (2018) Grassland biodiversity in Japan: threats, management and conservation. In: Grasslands of the World. CRC press, pp 211–232
Villaseñor NR, Escobar MAH (2019) Cemeteries and biodiversity conservation in cities: how do landscape and patch-level attributes influence bird diversity in urban park cemeteries? Urban Ecosyst 22:1037–1046
Walter HS (2004) The mismeasure of islands: Implications for biogeographical theory and the conservation of nature. J Biogeogr 31:177–197. https://doi.org/10.1046/j.0305-0270.2003.00989.x
Wiegand T, Revilla E, Moloney KA (2005) Effects of habitat loss and fragmentation on population dynamics. Conserv Biol 19:108–121
Yasunaga T, Yamashita I, Takai M, Kawamura M (1993) A field guide of Japanese bugs. Zennokyo, Tokyo
Yasunaga T, Nakatani Y, Takai M, Kawasara T (2001) A field guide of Japanese bugs II. Zennokyo, Tokyo
Acknowledgements
The Tokyo local government provided the city planning GIS data. This study was partially supported by Japan Society for the Promotion of Science KAKENHI grant number 20K06096. Two anonymous reviewers and editor provided many useful suggestions.
Funding
This study was partially supported by JSPS KAKENHI grant number 20K06096.
Author information
Authors and Affiliations
Contributions
SO and TO contributed to the study conception and design. Material preparation, data collection and analysis were performed by SO and TO. SO and NS identified specimens. NT conducted establishing remote sensing products. TO led the manuscript and all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Not applicable.
Consent to participate
All patients included in this study gave written informed consent to participate in this research.
Consent for publication
All patients provided written informed consent to publish the data contained within this article.
Conflicts of interests
All authors declare they have no conflicts of interests.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ohata, S., Osawa, T., Sato, N. et al. Large, concealed islands in the urban sea: Scattered surrounding green space enhances the quality of grassland habitats in urban parks, Tokyo. Urban Ecosyst 26, 641–649 (2023). https://doi.org/10.1007/s11252-022-01311-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-022-01311-x