Abstract
Woodland fragments, in small historical cities, are commonly regarded as temporary voids in an urban matrix, yet to be allocated a land-use, under city planning regulations. However, they could display relevant plant diversity, and contribute to urban ecosystem services. This study combined surveys at 100 m2, and at patch level, with the aim to investigate how patch size, stand and urbanization, affected the structure of plant communities in thirty woodland fragments (0.1–2 ha), spontaneously developing in the small, historical city of Padova (Northern Italy). Trees, shrubs and other perennial species dominated the plant communities in these patches. Alien species were common, in both the understory (freq. = 97 %, mean richness = 4.33) and tree layer (freq. = 90 %, mean richness = 1.50). Species typical of native communities also occurred. Understory communities were associated with ancient forest, nitrophilous, and ruderal species; highlighting an overall heterogeneity. Road and railway density was moderately correlated with total species richness in the understory, whereas, urbanity (i.e. the concentration of built environment excluding road and railway density), and tree density were not. Furthermore, alien tree dominance negatively influenced total and native tree layer species richness and, moderately positively, native understory species richness. These results highlight that spontaneous novel woodland patches, even if they are minor fragments in small historical cities, maintain diverse green infrastructures that may supply an array of urban ecosystem services, when adequately recognised by city plans.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Understanding of urban woodlands is increasing (Croci et al. 2008; Vallet et al. 2010; Trentanovi et al. 2013), this follows significant developments in the field of urban ecology, having understood the services that can be provided by these ecosystems at local, regional, national and global levels (Breuste et al. 2013). Urban woodlands are a product of the uniqueness of the urban environment; there are complex interactions of abiotic and biotic processes (Vallet et al. 2010; Werner 2011) creating ubiquitous ecological conditions (Rebele 1994). Woods are often fragmented in urban areas (Werner 2011); forming patches within the urban matrix, they are one of the most common natural habitats in European towns (Croci et al. 2008). There are different types of canopy, vegetation and plant establishment processes, on abandoned or neglected land and sites disturbed by humans (Kowarik 2005; Mathey and Rink 2010).
In urban landscapes, vegetation is influenced by features of urbanisation, such as, the neighbouring built-up area, surrounding household density (e.g., Kühn and Klotz 2006; McDonnell and Hahs 2008), road and railway density (e.g., von der Lippe and Kowarik 2008; Penone et al. 2012). In turn, these factors are intrinsically shaped by city planning (Sukopp et al. 1995), they mainly regard the development of residential, and industrial areas, in greenfield and brownfield sites, and the related construction of roads. However, in the case of urban forests, other stand-level factors may play an important role in determining the vegetation communities (e.g., Gilliam 2007; Barbier et al. 2008), together with the size of the patch (e.g., Hobbs 1988; Iida and Nakashizuka 1995).
The majority of historical cities are from the Mediterranean basin and Asia; areas subjected to human influence for millennia. Therefore, results from outside these regions may contrast with those observed within. Authors have outlined that hot spots of species richness are found in European cities (e.g., Zerbe et al. 2003; Kowarik 2011), there is a positive relationship between settlements and/or buildings age and plant species composition (Celesti-Grapow et al. 2006).
Spontaneous vegetation can form a part of urban woodlands (Kowarik 2005; Celesti-Grapow et al. 2006) and suburban areas (Barbati et al. 2013); the ecosystem services that can potentially be provided by spontaneous vegetation are considerable, such as habitat provision, wildlife dispersal, climate regulation and carbon sequestration (Robinson and Lundholm 2012; Barbati et al. 2013). The influx of alien species into European cities has been widely documented (e.g., Pyšek 1998; Wittig 2004), an overstory dominated by alien tree species, may have an influence on understory communities (e.g., Richardson et al. 1989; Sitzia et al. 2012). Species which are favoured by human disturbance, and fragmentation, tend to prevail in spontaneous woodland patches (Kowarik 2005; Del Tredici 2010; Kowarik et al. 2013; Trentanovi et al. 2013). Newly established plant assemblages, formed by alien species can be addressed as ‘novel ecosystems’ (Hobbs et al. 2006; Kowarik 2011).
Current research in Europe on urban woodlands focuses on large cities - for example; Rome (Celesti-Grapow et al. 2006) and Berlin (Trentanovi et al. 2013), or on mixed habitats (e.g., Celesti-Grapow and Blasi 1998; Chocholoušková and Pyšek 2003), woodlands which range from ‘wild’ to managed (Croci et al. 2008), or relatively old and/or studying the effects of rural to urban gradients (Lehvävirta and Rita 2002; Vallet et al. 2010). Species richness in small patches of spontaneous urban woodlands in small cities is relatively unknown, as well as the existence of these novel patches within the urban planning context, and for the people living in and around them.
The purpose of this paper is to develop knowledge on small novel transient woodland patches in a small historical city in Mediterranean Europe, firstly, to understand the composition of plant species assemblages, and secondly, to understand what factors are shaping plant species richness in these patches. Patches were selected randomly, and their boundaries defined. Their main vegetation characteristics were studied, native and alien species were recorded in the understory and tree layers at the sampling plot level (100 m2), and for woody species at the patch level. Then, we assessed the effect of adjacent land-uses, which are commonly allocated in urban plans (i.e., built-up area and road and railway density), and factors important at the stand level (i.e., tree density and alien tree dominance), on the species richness.
Methods
Study area
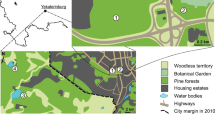
The small sized historical city of Padova (English: Padua), founded around X-IX sec. BC, is a municipality of 92.85 km2 (210,000 residents) with an ancient town centre of 4.54 km2 (Comune di Padova 2012); it is located in the Veneto region, Northeast of Italy (45°23′N, 11° 52′ E). The climate is sub-Mediterranean; the annual precipitation is 846 mm and the mean annual temperature is 12.9°C. Land use is mainly built up urban residential settlements (~55.5 %) with areas mixed with agricultural uses (~ 41.5 %); most of the territory is modelled artificially. Woody vegetation is very limited and mostly confined to the margins of rivers. According to ARPAV (2013), spontaneous wooded areas in Padova are almost non-existent. Road and railway networks extend for 982 and 31.5 km within the territory, respectively (Comune di Padova 2012).
Data collection
Sampling was performed from the start of June, to the first weekend of August 2013, with most of the work being performed in July. Within the municipality boundary, we searched woodland patches ≥ 1000 m2 to avoid those that would have been dominated by edge effects (Matlack 1994). We excluded patches that showed strong active management, or no spontaneous vegetation, or no trees ≥3 cm diameter breast height (DBH), and height ≥5 m. A sample of thirty of these patches were randomly selected (Fig. 1). From observing signs of spontaneous vegetation in historical images, the age of these patches ranged between 10 and 30 years. Only sporadic cuts were observed in certain patches and their boundaries were often limited by human elements or activities, like roads, buildings and cultivation. The interior of these patches was frequently used by people, where shelters and tracks were frequently observed (Fig. 2). A working protocol was adopted to collect information on these patches and to easily enable future replications. In all the patches, all woody species, including seedlings, were identified. Within these patches, a 100 m2 plot was surveyed, to record the plant species of the understory and tree layer vegetation. The cover-abundance scale of Braun-Blanquet and Pavillard (1928) (r = solitary small individual; + = few individuals; 1 = <5 %; 2 = 5–25 %; 3 = 25–50 %; 4 = 50–75 %; 5= > 75 %) was used to estimate understory species cover. The basal area was calculated from all trees with DBH ≥3 cm.
Data analysis
Cover-abundance values of recorded species were converted into Tüxen and Ellenberg (1937) percentage values (r = 0.02; + = 0.1; 1 = 2.5; 2 = 15; 3 = 37.5; 4 = 62.5; 5 = 87.5).
First we identified life-form; life-span and chorological type, using Pignatti (1982) and other databases, for each species recorded in the understory, and then the overall proportion of these life traits was calculated. Secondly, we ascertained that there was not any spatial autocorrelation present in the species richness values running the command Moran.I of the package “ape” in R (Paradis et al. 2004), using the inverse Euclidean distance matrix among patches’ centroids. Then, to investigate drivers of species richness, richness values were calculated at patch, and plot level, for total, native and alien species. In our study, alien species were identified according to Celesti-Grapow et al. (2009) and Masin and Scortegagna (2012) and considering also those species intentionally planted and unlikely to occur spontaneously.
The Bray-Curtis similarity distance matrix, from the understory species cover values at each site, were subjected to agglomerative hierarchical clustering analyses by using Ward’s clustering method. We then identified three groups of sites that adequately represented the observed flora. To investigate which plant species defined these groups, we performed Indicator Species Analysis (ISA) (Dufrêne and Legendre 1997). “Multipatt” function of the “indicspecies” package in R software (De Caceres et al. 2010) was used as the indicator value method (IndVal) and the statistical significance was tested with the Monte Carlo test, based on 9999 randomisations. This method combines data on both abundance and frequency. The resulting association index, IndVal.g, is the square root of the IndVal index in Dufrêne and Legendre (1997), it is at maximum (=1) when all occurrences of a species are found in a single group of sites, and when the species occurs in all sites of that group. To understand what factors affect the response of understory species richness, two groups of predictor variables, land use (urbanity, road and rail density) and stand (mean DBH, basal area and alien dominance) characteristics, were used. To calculate the land use variables, Quantum GIS (Quantum GIS Development Team 2012) was used. A 500m buffer was established for each patch, this buffer size has been shown to yield the best predictor set, compared to 100m and 200m buffers (e.g., Knapp et al. 2009; Westermann et al. 2011). Urbanity, as described by Trentanovi et al. (2013), was derived by subtracting rail and road density from urban land-use, based on Corine Land Cover classification (Bossard et al. 2000), calculating its proportion in the buffer. For road and rail density, the total length within the buffer was divided by the total buffer area. Basal area was used because it is a common parameter of tree density and canopy cover, particularly in reference to overstory-understory relationships (Mitchell and Popovich 1997), it is understood that, with increasing basal area, there is a decrease in light transmittance to the forest floor (Korhonen et al. 2006). The alien tree dominance was computed as the proportion of alien species basal area. The nature and strength of covariation between richness values and urbanity, road and rail density, basal area and alien tree dominance was tested through the Pearson product-moment correlation coefficient, after removal of outliers, if any were identified with the diagnostics method performed by the function “influence.measures” in R software (Belsley et al. 1980) or log-transformation of variables with skewed distributions. Where variables exhibited strong departures from normal distribution, we used Spearman rank correlation test. We also investigated the influence of patch size on the total richness of woody species in the patch. Linear regression analysis was used to find the best-fit line relating patch size to woody species richness. R software (Version 3.0.1) (R Development Core Team 2013) was used for all statistical analyses.
Results
The thirty surveyed patches had a mean size of 0.645 (SD: 0.50; min: 0.12; max: 2.32) ha, a mean tree basal area of 42 (SD: 30.1; min: 6; max: 157) m2/ha. Yet the highest basal area values were due to the presence of sizeable, old trees, pre-existent to the land use abandonment.
A total of 106 species were identified, of which 38 were alien (Suppl. Material). Table 1 shows the most frequent species recorded at the plot level (total: 92, aliens: 34). Species richness in the understory and tree layer (Table 2), and for woody species at the patch level (Table 3) underlined the importance of the alien component.
Phanerophytes and perennial species were dominant in the understory. The chorological spectrum highlighted the relevancy of species with Asiatic, additionally to those with European, origin (Fig. 3).
Life traits for the recorded understory species in thirty woodland patches of Padova (Northern Italy). The life form (phanerophytes: P; geophytes: G; hemicryptophytes: H; nano-phanerophytes: NP; therophytes: T), life span (annual: A; biannual: B; perennial: P) and chorological type (AS asiatic, EU European/Caucasic, EURAS eurasiatic, D dubious origin, M Mediterranean, W widely-distributed, B circumboreal, A Atlantic) are reported
Understory assemblage
The IndVal analysis, conducted for the three groups derived from the cluster analysis, produced a total of eight indicator species (Table 4). Group A was associated with Hedera helix and Bryonia dioica, species common in most types of woodlands and sheltered sites, with preference for heavy, fertile soils (Harding and Hilton 1992; Metcalfe 2005). When occurring in woodland, the Hedera helix is frequently dominant in the field layer (see review by Metcalfe 2005), which was also verified here.
Ulmus minor subsp. minor was significantly associated with group B, it is a fast growing tree species able to colonise abandoned land, a species which is now re-establishing after gaining resistance to Dutch elm disease (Solla et al. 2005; Sitzia et al. 2012).
Group C was characterised by two ruderal and nitrophilous species: Rubus fruticosus agg. and Parietaria officinalis, also present was Brachypodium sylvaticum; a species regarded by some authors as an ancient forest species (Hermy et al. 1999).
Groups A and C shared a common indicator species, Sambucus nigra, a species typical of disturbed, highly eutrophic soils, subjected to disturbance, either naturally, on floodplain terraces and woodland margins, or anthropogenically, in derelict gardens, farmyards and post-industrial wasteland (Atkinson and Atkinson 2002).
Richness and correlated factors
Surprisingly, no relation between species richness, and both urbanity and basal area was found. Road and railway density moderately correlated to understory total (r = 0.41, p = 0.023) species richness, but not native or alien species richness. Understory native species richness seemed also to be moderately positively correlated with alien dominance (r = 0.37, p = 0.042). As expected, alien tree dominance negatively correlated to species richness, at a limited extent, tree layer total (ρ = -0.39, p = 0.035) and, strongly, native (ρ = -0.71, p < 0.001) species richness.
The diversity of woody species on patches was related to the size of the patch, and the relationship, after removal of two outliers, conformed to: woody species richness = 7.179 (patch size in hectares) + 13.188 (R2 adj =0.43, F = 21.515, p < 0.001).
Discussion
Prior work has acknowledged the occurrence, and relevance, of the spontaneous development of vegetation communities in urban areas (Millard 2000, 2004). Nevertheless, studies of biodiversity in the urban environment do not normally use forests as the main foci, or they focus mainly on forest remnants (e.g., Guntenspergen and Levenson 1997; Godefroid and Koedam 2003; Doody et al. 2010). The importance of secondary woodlands spontaneously growing on abandoned lands in urban areas has been recognised (see Kowarik 2005), but studies in southern Europe are lacking and, to date, research has neglected spontaneous forests within small historical urban cities. In this study we have partially filled the research gap, by analysing vegetation assemblages within relatively young, spontaneously developing woodland patches, on neglected land, within a small historical city. We found that, in particular in the tree layer, alien species have an important role, and that the understory shows a relatively heterogeneous composition and richness.
A high proportion, and dominance of alien species, is generally observed in urban plant assemblages (Pyšek 1998; Chocholoušková and Pyšek 2003) and in particular also in wild urban woods (Kowarik 2005). In our context, their high number and relevancy may also be explained by a number of factors: the small patch area, because edge proximity favours alien species both in the understory, and overstory (LaPaix et al. 2012); by the long historical influence of the city, and by the moderate levels of urbanization (McKinney 2008).
As suggested by other studies (e.g., Prach and Pyšek 2001; Celesti-Grapow and Blasi 2003), ours highlights that woody species are important invaders of abandoned urban land. Our study emphasises the dominance of perennial species in the understory. Analysis of the understory composition highlighted the presence of groups of patches characterised by ruderal, and forest-related species. These findings, in accordance with Kowarik (2011), underline the presence of transient novel forest assemblages, in the studied urban context, a product of inadvertent action by humans, which feature a pronounced alien component. Some patches were also dominated by native trees known to be colonising species, or species typical of the Po plain forests, such as Ulmus minor, Quercus robur, Carpinus betulus and Salix alba. This heterogeneity of the composition, both in the tree layer and understory, indicates the high value of spontaneous woodland patches for biodiversity in the urban matrix.
As highlighted for parks and woodlots in general (Alvey 2006), even in small spontaneous patches, woody species richness tends to increase with patch size. The limited relationship between explanatory variables and the understory and tree layer plant richness strengthens the discourse that the city is an integrated ecosystem (Rebele 1994); where it is not possible to define one variable in particular that is affecting species richness. There is a degree of chaos theory involved, promoting heterogeneity. Richness is a result of complex interactions of abiotic and biotic processes at different scales (Werner 2011).
The fact that we did not consider forest edge and forest interior as two distinct habitats hinders any quantitative assessment of edge effect which are probable in small woodlands (Gonzalez et al. 2010). However, in addition to road and railway density, and alien dominance, as highlighted by our results, it is possible that other factors may play a role, even obscuring, or filtering, those that we analysed, such as, habitat history and configuration (De Sanctis et al. 2010), which, in turn, may influence edge effects and seed source availability from neighbouring roadside and river vegetation (von der Lippe et al. 2008; Säumel and Kowarik 2010), or residential gardens. Edge effects are related to the adjacent urban matrix and forest trails and paths (LaPaix et al. 2012), a higher number of alien species have been found in forest edges adjacent to urban areas compared to agricultural areas (Gonzalez-Moreno et al. 2013). Edges and their aspect effects light radiation to the forest floor, influencing species composition and richness. North-facing forest margins may not show edge effects on plant composition, or, its spatial penetration within forests may be reduced, while edge effects may be pronounced in south-facing aspect margins (Hamberg et al. 2010). Recreational pressure, presence of paths, related trampling, concentration of nutrients and pollutants, and other kinds of microhabitat features might also explain a portion of the observed species richness variability (Malmivaara-Lämäs et al. 2008; Hamberg et al. 2010). Furthermore, as colonisation is an on-going process, transient communities, and the factors shaping these communities, may be better explained by a study with a longer temporal scale (Lososová et al. 2012).
This study reveals that in small historical cities, small woodland patches that spontaneously develop within an urban landscape can play an important role for biodiversity, by forming both new communities of species, which did not exist in the past, and native habitats that had previously disappeared. They may have the opportunity to convey a set of services; the spatial concentration of people in cities increases the demand for ecosystem services (McDonald 2015). Urban trees and woodlands have a wide range of benefits and uses (e.g., Konijnendijk 2008; Jim and Chen 2009; Escobedo et al. 2011), such as carbon sequestration and the positive effects on humans’ wellbeing. Small spontaneous forest patches may act as an added value in respect to the existing and recognised green infrastructures of small urban parks (Nordh and Østby 2013) and roadside wild vegetation (Weber et al. 2014), enhancing ecosystem services in small historical cities, in particular those related to recreation. Given the small size of woodlands, it is feasible to plan recreational trails at fine-scale resolutions, and carefully assess their environmental impact (Sitzia et al. 2014). The acknowledgement of spontaneous woodland patches is important for the planning and development in small sized cities. The opportunities provided by spontaneous, “unofficial” vegetation; described by Mabey (1973) or “unintentional”; as by Kühn (2006), vegetation for urban landscape design is now being understood, with novel perspectives and innovative approaches being proposed and applied (Millard 2000; Prach and Pyšek 2001; Kowarik and Langer 2005; Kühn 2006; Ignatieva et al. 2010). A key problem is the anomaly of these patches in town planning; they can be subjected to land use change before their value is fully comprehended. Temporary measures, suitable for these habitats, could be implemented in greenfield and brownfield sites, potentially on a medium term basis, until the foreseen building development is realised (see Kattwinkel et al. 2011).
Our study underlines the great potential for urban planning, and the importance of plant communities in small historical cities, further research is needed on vegetation composition and dynamics. Specifically, future studies will help to increase understanding of how these novel transient woodlands will develop, positing what potential vegetation communities can be hosted by these unmanaged woodlands. Furthermore, as forest succession on abandoned urban land is common and brownfields rehabilitation is an option in many small cities in Europe (Acosta et al. 2005; Lafortezza et al. 2008), in North America (Greenberg et al. 2001), and also likely in other regions of the world where forests are spontaneously expanding (see review by Sitzia et al. 2010), it is important to trigger the awareness of planners and designers of the opportunities that these areas can give. A call, for new planning approaches, for transient novel woodlands in small historical cities, is required.
References
Acosta A, Carranza LM, Giancola M (2005) Landscape change and ecosystem classification in a Municipal district of small city (Isernia, Central Italy). Environ Monit Assess 108:323–335
Alvey AA (2006) Promoting and preserving biodiversity in the urban forest. Urban For Urban Green 5:195–201
ARPAV (2013) Pollini e spore fungine allergenici nel Veneto—Rapporto anno 2012. http://www.arpa.veneto.it/temi-ambientali/pollini/file-e-allegati/rapporto-2012/RAPPORTO_Pollini_2012.pdf. Accessed 30 January 2014
Atkinson MD, Atkinson E (2002) Sambucus nigra L. J Ecol 90:895–923
Barbati A, Corona P, Salvati L, Gasparella L (2013) Natural forest expansion into suburban countryside: gained ground for a green infrastructure? Urban For Urban Green 12:36–43
Barbier S, Gosselin F, Balandier P (2008) Influence of tree species on understory vegetation diversity and mechanisms involved – a critical review for temperate and boreal forests. For Ecol Manag 254:1–15
Belsley DA, Kuh E, Welsch RE (1980) Regression diagnostics. Wiley, New York
Bossard M, Feranec J, Otahel J (2000) CORINE land cover technical guide—Addendum 2000. European Environmental Agency, Technical Report No. 40, Copenhagen, Denmark. http://www.eea.eu.int. Accessed 12 Dicember 2013
Braun-Blanquet J, Pavillard J (1928) Vocabulaire de sociologie végétale. Imprimerie Roumégous & Déhan, Montpellier
Breuste J, Haase D, Elmqvist T (2013) Urban landscapes and ecosystem services. In: Wratten S, Sandhu H, Cullen R, Costanza R (eds) Ecosystem services in agricultural and urban landscapes. Wiley, Chichester, pp 83–104
Celesti-Grapow L, Blasi C (1998) A comparison of the urban flora of different phytoclimatic regions in Italy. Glob Ecol Biogeogr 7:367–378
Celesti-Grapow L, Blasi C (2003) Archaeological sites as areas for biodiversity conservation in cities: the spontaneous vascular flora of the Caracalla Baths in Rome. Webbia 58:77–102
Celesti-Grapow L, Pyšek P, Jarošík V, Blasi C (2006) Determinants of native and alien species richness in the urban flora of Rome. Divers Distrib 12:490–501
Celesti-Grapow L, Alessandrini A, Arrigoni PV, Banfi E, Bernardo L, Bovio M, Brundu G, Cagiotti MR, Camarda I, Carli E, Conti F, Fascetti S, Galasso G, Gubellini L, La Valva V, Lucchese F, Marchiori S, Mazzola P, Peccenini S, Poldini L, Pretto F, Prosser F, Siniscalco C, Villani MC, Viegi L, Wilhalm T, Blasi C (2009) Inventory of the non‐native flora of Italy. Plant Biosyst 143:386–430
Chocholoušková Z, Pyšek P (2003) Changes in composition and structure of urban flora over 120 years: a case study of the city of Plzeň. Flora 198:366–376
Croci S, Butet A, Georges A, Aguejdad R, Clergeau P (2008) Small urban woodlands as biodiversity conservation hot-spot: a multi-taxon approach. Landsc Ecol 23:1171–1186
De Caceres M, Legendre P, Moretti M (2010) Improving indicator species analysis by combining groups of sites. Oikos 119:1674–1684
De Sanctis M, Alfò M, Attore F, Francesconi F, Bruno F (2010) Effect of habitat configuration and quality on species richness and distribution in fragmented forest patches near Rome. J Veg Sci 21:55–65
Del Tredici PD (2010) Spontaneous urban vegetation: reflections of change in a globalized world. Nat Cult 5:299–315
Development Core Team R (2013) R: A language and environment for statistical computing. R foundation for statistical computing, Vienna
Comune di Padova (2012) Annuario statistico comunale 2012. www.padovanet.it. Accessed 12 December 2013
Doody BJ, Sullivan JJ, Meurk CD, Stewart GH, Perkins HC (2010) Urban realities: the contribution of residential gardens to the conservation of urban forest remnants. Biodivers Conserv 19:1385–1400
Dufrêne M, Legendre P (1997) Species assemblages and indicator species: the need for flexible assymetrical approach. Ecol Monog 67:345–366
Escobedo FJ, Kroeger T, Wagner JE (2011) Urban forests and pollution mitigation: analyzing ecosystem services and disservices. Environ Pollut 159:2078–2087
Gilliam FS (2007) The ecological significance of the herbaceous layer in temperate forest ecosystems. Bioscience 57:845–858
Godefroid S, Koedam S (2003) How important are large vs. small forest remnants for the conservation of the woodland flora in an urban context? Glob Ecol Biogeogr 12:287–298
Gonzalez M, Ladet S, Deconchat M, Cabanettes A, Alard D, Balent G (2010) Relative contribution of edge and interior zones to patch size effect on species richness: an example for woody plants. For Ecol Manag 259:266–274
Gonzalez-Moreno P, Pino J, Gassó N, Vilá M (2013) Landscape context modulates alien plant invasion in Mediterranean forest edges. Biol Inv 15:547–557
Greenberg M, Lowrie K, Mayer H, Miller KT, Solitare L (2001) Brownfield redevelopment as a smart growth option in the United States. Environmentalist 21:129–143
Guntenspergen GR, Levenson JB (1997) Understory plant species composition in remnant stands along an urban-to-rural lan-use gradient. Urban Ecosyst 1:155–169
Hamberg L, Fedrowitz K, Lehvävirta S, Kotze DJ (2010) Vegetation changes at sub-xeric urban forest edges in Finland—the effects of edge aspect and trampling. Urban Ecosyst 13:583–603
Harding DJ, Hilton GM (1992) Functional ecology of woodlands and forests. Springer, Berlin
Hermy M, Honnay O, Firbank L, Grashof-Bokdam C, Lawesson JE (1999) An ecological comparison between ancient and other forest plant species of Europe, and the implications for forest conservation. Biol Conserv 91:9–22
Hobbs ER (1988) Species richness in urban forest patches and implications for urban landscape diversity. Landsc Ecol 1:141–152
Hobbs RJ, Arico S, Aronson J, Baron JS, Bridgewater P, Cramer VA, Epstein PR, Ewel JJ, Klink CA, Lugo AE, Norton D, Ojima D, Richardson DM, Sanderson EW, Valladares F, Vilà M, Zamora R, Zobel M (2006) Novel ecosystems:theoreticla and management aspects of the new ecological world order. Glob Ecol Biogeogr 15:1–7
Ignatieva M, Stewart GH, Meurk C (2010) Planning and design of ecological networks in urban areas. Landsc Ecol Eng 7:17–25
Iida S, Nakashizuka T (1995) Forest fragmentation and its effect on species diversity in suburban coppice forests in Japan. For Ecol Manag 73:197–210
Jim CY, Chen WY (2009) Ecosystem services and valuation of urban forests in China. Cities 26:187–194
Kattwinkel M, Bierdermann R, Kleyer M (2011) Temporary conservation for urban biodiversity. Biol Conserv 144:2335–2343
Knapp S, Kühn I, Bakker JP, Kleyer M, Klotz S, Ozinga WA, Poschlod P, Thompson K, Thuiller W, Römermann C (2009) How species traits and affinity to urban land use control large-scale species frequency. Divers Distrib 15:533–546
Konijnendijk CC (2008) The forest and the city: the cultural landscape of urban woodland. Springer, Berlin
Korhonen L, Korhonen KT, Rautiainen M, Stenberg P (2006) Estimation of forest canopy cover: a comparison of field measurement techniques. Silva Fenn 40:577–588
Kowarik I (2005) Wild urban woodlands: towards a conceptual framework. In: Kowarik I, Körner S (eds) Wild urban woodlands. new perspectives for urban forestry. Springer, Berlin, pp 1–32
Kowarik I (2011) Novel urban ecosystems, biodiversity, and conservation. Environ Pollut 159:1974–1983
Kowarik I, Langer A (2005) Natur-park Südgelände: linking conservation and recreating in an abandoned rail yard in Berlin. In: Kowarik I, Körner S (eds) Wild urban woodlands. new perspectives for urban forestry. Springer, Berlin, pp 287–299
Kowarik I, von der Lippe M, Cierjacks A (2013) Prevalence of alien versus native species of woody plants in Berlin differs between habitats and at different scales. Preslia 85:113–132
Kühn N (2006) Intentions for the unintentional: spontaneous vegetation as the basis for innovative planting design in urban areas. J Landsc Archit 1:46–53
Kühn I, Klotz S (2006) Urbanization and homogenization—comparing the floras of urban and rural areas in Germany. Biol Conserv 127:292–300
Lafortezza R, Corry RC, Sanesi G, Brown RD (2008) Visual preference and ecological assessments for designed alternative brownfield rehabilitations. J Environ Manag 89:257–269
LaPaix R, Harper K, Freedman B (2012) Patterns of exotic plants in relation to anthropogenic edges within urban forest remnants. Appl Veg Sci 15:525–535
Lehvävirta S, Rita H (2002) Natural regeneration of trees in urban woodlands. J Veg Sci 13:57–66
Lososová Z, Chytrý M, Tichý L, Danihelka J, Fajmon K, Hájek O, Kintrová K, Láníková D, Otýpková Z, Řehořek V (2012) Biotic homogenization of Central European urban floras depends on residence time of alien species and habitat types. Biol Conserv 145:179–184
Mabey R (1973) The unofficial countryside. William Collins Sons and Co, Glasgow
Malmivaara-Lämäs M, Löfström HL, Vanha-Majamaa I, Niemelä J (2008) Trampling tolerance of understorey vegetation in different hemiboreal urban forest site types in Finland. Urban Ecosyst 11:1–16
Masin Rizzieri R, Scortegagna S (2012) Flora alloctona del Veneto centro-meridionale (Province di Padova, Rovigo, Venezia e Vicenza—Veneto - NE Italia). Nat Vicentina 15:5–54
Mathey J, Rink D (2010) Urban Wastelands—a change for biodiversity in cities? Ecological aspects, social perceptions and acceptance of wilderness by residents. In: Müller N, Werner P, Kelcey JG (eds) Urban biodiversity and design. Wiley-Blackwell, Hoboken, New Jersey, pp 406–424
Matlack GR (1994) Vegetation dynamics of forest edge—trends in space and successional time. J Ecol 82:113–123
McDonald RI (2015) The effectiveness of conservation interventions to overcome the urban-environmental paradox. Ann N Y Acad Sci. doi:10.1111/nyas.12752
McDonnell M, Hahs A (2008) The use of gradient analysis studies in advancing our understanding of the ecology of urbanizing landscapes: current status and future directions. Landsc Ecol 23:1143–1155
McKinney ML (2008) Effects of urbanization on species richness: a review of plants and animals. Urban Ecosyst 11:161–176
Metcalfe DJ (2005) Hedera helix L. J Ecol 93:632–648
Millard A (2000) The potential role of natural colonisation as a design tool for urban forestry—a pilot study. Landsc Urban Plan 52:173–180
Millard A (2004) Indigenous and spontaneous vegetation: their relationship to urban development in the city of Leeds, UK. Urban For Urban Green 3:39–47
Mitchell JE, Popovich SJ (1997) Effectiveness of basal area for estimating canopy cover of ponderosa pine. For Ecol Manag 95:45–51
Nordh H, Østby K (2013) Pocket parks for people—a study of park design and use. Urban For Urban Green 12:12–17
Paradis E, Claude J, Strimmer K (2004) APE: analyses of phylogenetics and evolution in R language. Bioinformation 20:289–290
Penone C, Machon N, Julliard R, Le Viol I (2012) Do railway edges provide functional connectivity for plant communities in an urban context? Biol Conserv 148:126–133
Pignatti S (1982) Flora d’Italia. Edagricole, Bologna
Prach K, Pyšek P (2001) Using spontaneous succession for restoration of human-disturbed habitats: experience from Central Europe. Ecol Eng 17:55–62
Pyšek P (1998) Alien and native species in Central European urban floras: a quantitative comparison. J Biogeogr 25:155–163
Quantum GIS Development Team (2012) Quantum GIS geographic information systen. open source geospatial foundation project. http://qgis.osgeo.org
Rebele F (1994) Urban ecology and special features of urban ecosystems. Glob Ecol Biogeogr Lett 4:173–187
Richardson DM, MacDonald IAW, Forsyth GG (1989) Reduction in plant species richness under stands of alien trees and shrubs in the Fynbos Biome. South Afr For J 149:1–8
Robinson S, Lundholm J (2012) Ecosystem services provided by urban spontaneous vegetation. Urban Ecosyst 15:545–557
Säumel I, Kowarik I (2010) Urban rivers as dispersal corridors for primarily wind-dispersed invasive tree species. Landsc Urban Plan 94:244–249
Sitzia T, Semenzato P, Trentanovi G (2010) Natural reforestation is changing spatial patterns of rural mountain and hill landscapes: a global overview. For Ecol Manag 259:1354–1362
Sitzia T, Campagnaro T, Dainese M, Cierjacks A (2012) Plant species diversity in alien black locust forests: a paired comparison with native stands across a north-Mediterranean range expansion. For Ecol Manag 285:85–91
Sitzia T, Rizzi A, Cattaneo D, Semenzato P (2014) Designing recreational trails in a forest dune habitat using least-cost path analysis at the resolution of visitor sight distance. Urban For Urban Green 13:861–868
Solla A, Bohnens J, Collin E, Diamandis S, Franke A, Gil L, Burón M, Santini A, Mittempergher L, Pinon J, Broeck AV (2005) Screening European elms for resistance to Ophiostoma novo-ulmi. For Sci 51:134–141
Sukopp H, Numata M, Huber A (1995) Urban ecology as the basis of urban planning. SPB Academic Publishing, Amsterdam
Trentanovi G, von der Lippe M, Sitzia T, Ziechmann U, Kowarik I, Cierjacks A (2013) Biotic homogenization at the community scale: disentangling the roles of urbanization and plant invasion. Divers Distrib 19:738–748
Tüxen R, Ellenberg H (1937) Der systematische und ökologische gruppenwert. ein beitrag zur begriffsbildung und methodik der pflanzensoziologie. Mitt Florist-Soziol Arbgem 3:171–184
Vallet J, Daniel H, Beaujouan V, Rozé F, Pavoine S (2010) Using biological traits to assess how urbanization filters plant species of small woodlands. Appl Veg Sci 13:412–424
von der Lippe M, Kowarik I (2008) Do cities export biodiversity? traffic as dispersal vector across urban-rural gradients. Divers Distrib 14:18–25
Weber F, Kowarik I, Säumel I (2014) A walk on the wild side: Perceptions of roadside vegetation beyond trees. Urban For Urban Green 13:205–212
Werner P (2011) The ecology of urban areas and their functions for species diversity. Landsc Ecol Eng 7:231–240
Westermann JR, vor der Lippe M, Kowarik I (2011) Seed traits, landscape and environmental parameters as predictors of species occurrence in fragmented urban railway habitats. Basic Appl Ecol 12:29–37
Wittig R (2004) The origin and development of the urban flora of Central Europe. Urban Ecosyst 7:323–329
Zerbe S, Maurer U, Schmitz S, Sukopp H (2003) Biodiversity in Berlin and its potential for nature conservation. Landsc Urban Plan 62:139–148
Acknowledgments
We thank Ingo Kowarik for his helpful review of an earlier version of this manuscript. This work was conducted as part of a M.S. thesis by R. Weir and of a PhD thesis by T. Campagnaro, at the University of Padova, Italy, under the supervision of T. Sitzia. The work has been supported by the University of Padova (ex 60 % research project 60A08-1078/13 to T. Sitzia and PhD scholarship to T. Campagnaro).
Conflict of interest
The authors have no conflict of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 51 kb)
Rights and permissions
About this article
Cite this article
Sitzia, T., Campagnaro, T. & Weir, R.G. Novel woodland patches in a small historical Mediterranean city: Padova, Northern Italy. Urban Ecosyst 19, 475–487 (2016). https://doi.org/10.1007/s11252-015-0475-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11252-015-0475-3