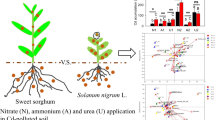
Abstract
Purpose
Rapid nitrification leads to loss of nitrogen (N) fertilizer in agricultural systems. Plant produced/derived biological nitrification inhibitors (BNIs) are an effective eco-strategy to rein-in soil nitrification to improve crop-N uptake and nitrogen use efficiency (NUE) in production systems. Sorgoleone is the major component of hydrophobic-BNI-activity in sorghum roots. However, the role of genetic differences in sorgoleone production in reducing soil nitrification and N2O emissions are not established.
Methods
Two genetic-stocks of sorghum with high-sorgoleone (HS), and two with low-sorgoleone (LS) production from roots were grown using hydroponics in a plant-growth chamber, in soil in pots in a glasshouse, and in a field experiment. Release of hydrophilic-BNI activity from roots of HS and LS genetic stocks, sorgoleone levels in rhizosphere soils, soil nitrification rates, soil-nitrifier activity and N2O emissions were measured to understand the interplay involving sorgoleone release, hydrophilic-BNI release from roots, soil nitrification, plant growth and N uptake.
Results
HS-producing genetic-stocks showed higher hydrophilic-BNI-capacity compared to LS- producing genetic-stocks. Biomass production and N uptake were significantly higher in HS than in LS genetic-stocks. Glasshouse and field studies suggest that HS genetic stocks had stronger suppressive impact on soil-nitrifier-populations (ammonia-oxidizing archaea and ammonia-oxidizing bacteria), soil-nitrification, and soil-N2O emissions than in LS genetic-stocks.
Conclusion
These results demonstrate that HS sorghum genetic-stocks suppress soil nitrifier activity and can potentially reduce N losses from NO3 − leaching and N2O emissions more effectively than LS genetic-stocks.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Biological nitrification inhibition (BNI) is proposed as a low-cost eco-strategy to limit nitrogen (N) losses and improve nitrogen use efficiency (NUE) in farmlands (Subbarao et al. 2007a; 2017; 2021; Coskun et al. 2017; Villegas et al. 2020; Lu et al. 2021; Zhang et al. 2021). Currently reported crop species with BNI-capacity include sorghum [Sorghum bicolor (L.) Moench], rice [Oryza sativa (L.)], wheat [Triticum aestivum (L.)], maize [Zea mays (L.)] and Brachiaria [Brachiaria humidicola (L.)] pasture (Zakir et al. 2008; Subbarao et al. 2009; 2021; O’Sullivan et al. 2016; Sun et al. 2016; Byrnes et al. 2017; Nakamura et al. 2020; Otaka et al. 2021). Their main function is to maintain a relatively high amounts of soil-NH4+, while reducing generation of highly mobile soil-NO3− and subsequent N2O emissions, by inhibiting the activity of soil-nitrifiers (Zakir et al. 2008; Subbarao et al. 2009; 2021; Byrnes et al. 2017; Sarr et al. 2020; Li et al. 2021a). Sorghum is the fifth most widely cultivated cereal grain globally, especially grown in the semi-arid regions of Asia, Africa, and Latin Americas, and is reported to have high BNI-capacity in root systems (Subbarao et al. 2013; Tesfamariam et al. 2014; Sarr et al. 2020). Two categories of BNIs released from sorghum roots, hydrophobic-BNIs and hydrophilic-BNIs and together they determine the BNI-capacity of root systems (Subbarao et al. 2013). Sorgoleone, a major hydrophobic-BNI-activity component released from sorghum roots has been reported to suppress Nitrosomonas by blocking AMO and HAO enzymatic pathways (Dayan et al. 2010; Subbarao et al. 2013; Sarr et al. 2020; 2021). However, sorgoleone release explains only a part of BNI-capacity (about 40%) of root systems and the remaining 60% is due to hydrophilic-BNI release (Subbarao et al. 2013).
Nitrification, driven primarily by nitrifying microorganisms, including ammonia-oxidizing bacteria (AOB) and ammonia-oxidizing archaea (AOA), leads to loss of N applied as fertilizer to farmlands (Ishikawa et al. 2003; Prosser and Nicol 2012; Gubry-Rangin et al. 2020; Prosser et al. 2020; Kaur-Bhambra et al. 2021). Soil NO3−-N, an important product of nitrification, is prone to leaching and runoff, and this has consequences for eutrophication, contamination of underground water, and the production of nitrous oxide (N2O), a powerful greenhouse gas (Davidson 2009; Subbarao et al. 2015; Stevens 2019; Maaz et al. 2021). Although synthetic nitrification inhibitors (SNIs) can effectively inhibit nitrification and reduce nitrogen losses, they are not widely adopted in production agriculture due to high costs, low effectiveness in the tropics and pollution of the environment (Coskun et al. 2017; Subbarao et al. 2017; Fu et al. 2020; Subbarao and Searchinger 2021). We have earlier reported that sorgoleone-amended soils showed reduced soil nitrification and N2O emissions in laboratory incubation studies (Subbarao et al. 2013; Tesfamariam et al. 2014). This study was initiated to test the hypothesis that sorghum genetic-stocks with high levels of sorgoleone release may have lower nitrification rates and N2O emissions compared to low sorgoleone producing genetic stocks.
Two high-sorgoleone (HS) and two low-sorgoleone (LS) producing genetic-stocks were selected for this study to test the following hypothesis, (1) HS sorghum genetic-stocks may also release higher amounts of hydrophilic-BNIs compared to LS sorghum genetic-stocks; (2) HS genetic-stocks may show better growth and N uptake than LS genetic-stocks; (3) HS genetic-stocks likely show lower soil nitrification and N2O emissions compared to LS genetic-stocks both in soil pot and field trials.
Materials and methods
Preliminary evaluation of sorghum germplasm for hydrophobic-BNI (sorgoleone) production from roots
Four sorghum genetic-stocks that have contrasting abilities for sorgoleone production from roots were selected after screening 250 germplasm lines (Subbarao and Santosh Deshpande, JIRCAS & ICRISAT, unpublished results). Two genetic-stocks were characterized as high-sorgoleone (HS) and two as low-sorgoleone (LS) producing (Table 1). These characteristics were determined by growing plants in a plant growth chamber (25◦C, 14/10 h light/dark period, average photosynthetic flux of 300 mmol m−2 s−1) for ten days. Seeds were soaked in aerated germination solution (200 μM CaSO4) for 24 h before sowing them into folded filter paper (MN 710, MACHEREY–NAGEL GmbH & Co. KG, Germany) supported by hard plates with one end touching the bottom of the seedling growth box supplied with the germination solution. This system allowed the seedlings to be continuously supplied with nutrient solutions via capillary movement (see Tesfamariam et al. 2014 for more details on methodology). At ten days after sowing (DAS), 40 seedlings were selected for each genetic-stock and divided into two batches of 20 seedlings each and considered as two replications; roots of 20 seedlings were excised and dipped in 40 mL acidified dichloromethane (DCM) (1% v/v DCM:acetic acid) for 1 min, and this is considered as root-DCM wash. Root-DCM was filtered and processed for determining hydrophobic-BNI (sorgoleone) content as described earlier (Subbarao et al. 2013; Tesfamariam et al. 2014).
Experiment 1
Characterization of HS and LS sorghum genetic-stocks for hydrophilic-BNI capacity
Four genotypes of sorghum were grown hydroponically in a plant growth chamber with day/night temperatures at 30/28 °C, photoperiod at 14/10 h with a light intensity of 300 μmol m−2 s−1, and relative humidity at 80% for 45 days before using them for collecting root exudates. Plants were grown using 1/2 strength modified Hoagland nutrient solution (Subbarao et al. 2013; Gao et al. 2014) with an optimized ratio for sorghum growth of 20% NH4+-N with 80% NO3−-N (Subbarao and Searchinger 2021), with a total N concentration maintained at 1.0 mM in nutrient solutions. Sorghum seeds were sown in seedling grow-boxes containing distilled water with 0.2 mM CaSO4 solution and seedling were grown for up to 10 days stage, then the seedlings were transferred to aerated nutrient solution in tanks with a capacity of 70 L of nutrient solution, with three sorghum plants were planted per hole and considered as one replication; three holes per styrofoam sheet, which was placed on the top of a tank i.e., giving in total nine plants per tank, representing 3 replications. Each treatment was replicated three times. The pH of the solution was allowed to change according to the treatment, and nutrient solutions in tanks were replaced once every two weeks.
On 45th day, root exudates were collected for determination of hydrophilic-BNI-activity. For that, three intact plants with the roots systems were transferred in 1.8 L aerated 1 mM NH4Cl + 0.2 mM CaCl2 solutions for 24 h. These root exudate solutions were stored at 5 °C for 5 days until completion of processing and determination of hydrophilic-BNI-activity. After root exudate collection, roots and shoots were separated, dried at 70 °C for 96 h in a forced air-circulating oven before determining dry weights and plant N content. Leaf-NO3− concentrations were determined by sampling fresh, the sorghum fully expanded blade. The hydrophilic-BNI-activity in root exudates were determined using a luminescent recombinant N. europaea assay (Subbarao et al. 2006a).
Experiment 2
Assessing the impact of HS and LS genetic-stocks on soil nitrification, soil nitrifier activity and N2O emissions in glasshouse
This glasshouse experiment was conducted during May to July 2020. Pots were filled with 2 kg (5 mm sieve) of volcanic ash soil (Typic Hapludands) from Japan International Research Center for Agricultural Sciences (JIRCAS) experimental station. Soil conditions were: soil pH, 5.5; NH4+ -N, 1.6 mg kg−1; NO3− -N, 4.3 mg kg−1; total soil C, 31.2 mg kg−1; total soil N, 2.63 g kg−1. The glasshouse conditions were day/night temperature regime of 32/25 °C, the photosynthetic photon follows to the natural sunlight and photoperiod in the summer season. The experimental design was factorial, consisted of two N levels [High-N (HN): 250 mg N kg−1 and Low-N (LN), 50 mg N kg−1, (equivalent to 1.20 g and 0.24 g of N as (NH4)2SO4 per kg of soil, respectively)] and four sorghum genotypes contrasting in sorgoleone release ability ((two HS, EC670311 and IS31861 and two LS, EC670350 and EC670402). The experiment was setup in a randomized block design with three replications. Ten seeds were planted per pot and thinned to six seedlings after 7 days. Both potassium (K) and phosphorus(P) were applied at 100 kg K2O ha−1 and 100 kg P2O5 ha−1 as basal fertilization using KCl and TSP, respectively. N fertilization was administered through (NH4)2SO4 solution in four equal splits during the plant growing period. The soil water content was maintained at 60% field capacity with deionized water. Soil was sampled on 0, 6 and 12 days after 4th application of N fertilizer (performed at 38 days after sowing) for determination of NH4+ -N and NO3− -N content. The shoot dry weight, plant N content were determined at harvest (50 DAS). Rhizosphere soils were collected, by shaking intact plants of sorghum to remove loose soil from the root system, and consider the soil still adhering to the root surface as rhizosphere (soil that were in close proximity to roots), and used for determining potential nitrification (PN), abundance of AOA and AOB amoA genes, N2O emission from laboratory incubations.
Experiment 3
Field validation of HS and LS genetic-stocks on soil nitrification and N2O emissions
A field trial was conducted (from June to August 2021) at JIRCAS experimental station in Tsukuba, Japan (36.05 N 140.08E). Soils are of volacanic ash type Typic Hapludands [pH (H2O) 5.5, clay 54.8%, silt 26.3%, sand 18.9%, total carbon 29.2 mg C g−1 soil; NH4+-N, 3.6 mg kg−1; NO3−-N, 5.7 mg kg−1]. The experimental design was consisted of two sorghum genetic-stocks with high and low sorgoleone-releasing capacity [HS (EC670311) and LS (EC670350)] and replicated three times. Four seedbeds of 1.5 m length and 3 m width in one plot. Two rows of sorghum were planted in each seedbed with a row spacing of 10 cm and a plant spacing of 20 cm, planted one sorghum seed in each hole. Both potassium (K) and phosphorus(P) were applied at 100 kg K2O ha−1 and 100 kg P2O5 ha−1 as basal fertilization using KCl and TSP, respectively. N fertilization was administered through (NH4)2SO4 solution with 50 kg N ha−1 in two equal splits during the plant growing period (addition at 30 and 50 days after sowing). Four plants and corresponding rhizosphere soils were sampled from each experimental plot after 2nd application of N fertilizer (65 DAS). Biomass and plant N content were determined. Rhizosphere soil was used for determination of potential nitrification (PN), nitrification rate (NR), abundance of AOA and AOB amoA genes, N2O emissions from laboratory incubation experiments. The collection method of rhizosphere soils was the same as that of experiment 2. Most of the rhizosphere soils were taken at a depth of 0–20 cm from the sorghum root system.
Analysis of soil and plant samples
Shoot dry weights and inorganic-N analysis
The sorghum shoots at harvest were placed at 70 °C oven until completely dried, then weighed to calculate biomass, and plant samples were grounded to a fine powder and pass through a 1 mm mesh for the determination of plant N concentration. The N concentration was determined by using a mass spectrometer IRMS (Thermo Scientific, Bremen, Germany). Total plant N content (mg plant−1) = shoot dry weight × N concentration. For leaf NO3− concentrations, one g of fresh leaf tissue was ground with 50 ml deionized water using a blender, the mixture was centrifuged, and the supernatant used for nitrate analysis in a continuous flow auto analyzer (BL-TEC K.K., Tokyo, Japan).
Determination of soil inorganic-N levels
For soil NH4+ -N and NO3− -N determinations, air-dry samples were sieved to 2 mm and 3 g soils were weighted and extracted with 30 mL of 2 mol KCl for 60 min using a shaker and mixture was filtered, then analyzed by using an autoanalyzer (BL-TEC K.K., Tokyo, Japan).
Determination of hydrophilic-BNI-activity
For collecting root exudate for determination of hydrophilic-BNI-activity, three plants were removed from nutrient solution tanks, sequentially rinsed with deionized and distilled water, and immersed in 1.8 L aerated 1 mM NH4Cl + 0.2 mM CaCl2 solutions for 24 h period. Root exudates were evaporated to dryness, extracted with 30 mL methanol, condensed to 1.5 mL methanol, then evaporate to dryness, extracted with 10 μL DMSO, which was then used for determination of hydrophilic-BNI- activity using recombinant luminescent Nitrosomonas assay described earlier (Subbarao et al. 2006a).
Potential nitrification and nitrification rates in rhizosphere soil samples collected from glasshouse and field studies
Potential soil nitrification was determined using a modified soil slurry protocol by Vazquez et al. (2020). The brief protocol was as follows: 10 g of air-dried rhizosphere soil was taken into a 250 ml Erlenmeyer flask and 100 ml of N-potential solution (1 mM KH2PO4, 1 mM (NH4)2SO4, 10 mM NaClO3, and pH adjusted to 7.0) was added. The flask was covered with a breathable sponge-cap and loaded into a temperature-controlled roto-mixer at 250 rpm and incubated at 20 °C. A volume of 10 mL of the soil slurry from each flask was sampled at 24 h interval for 10 days. The soil slurry was subjected to centrifuge and the supernatant was analyzed for NO3− -N using an autoanalyzer (BL-TEC K.K., Tokyo, Japan). The soil nitrification rate was determined after incubation period of 10 days using a temperature with 20 °C, and humidity-controlled at 80%, the soil water content was maintained at 60% WFPS incubator. 2 g of soil taken in a 10 ml glass bottle and 200 mg N kg−1 soil as (NH4)2SO4 was added for incubation experiment. Details of soil incubation study were earlier described in Subbarao et al. (2006a).
Determination of soil nitrifier activity
Soil DNA was extracted from 0.4 g of rhizosphere soil using Fast DNA Spin Kit for soil (Mo-Bio, Carlsbad, CA) according to manufacturer’s instructions with slight modifications. The obtained pure DNA was quantified by Qubit Quantification Platform dsDNA HS Assay kit (Invitrogen, Carlsbad, CA). Quantitative PCR (qPCR) was performed to assess the abundance of the amoA genes of both ammonia-oxidizing bacteria (AOB) and ammonia-oxidizing archaea (AOA). amoA-AOA, GenAOAF: ATAGAGCCTCAAGTAGGAAAGTTCTA, GenAOAR: CCAAGCGGCCATCCAGCTGTATGTCC. amoA-AOB, amoA-1Fomd: CTGGGGTTCTACTGGTGGTC, GenAOBR: GCAGTGATCATCCAGTTGCG. The detailed protocol was described earlier by Sarr et al. (2020).
Determination of N2O emissions in laboratory incubation studies
N2O emissions were assessed using air-dried rhizosphere soils following Hink et al. (2018). Briefly, 5 g soil was transferred to 100 mL glass vial (height 12.0 cm; diameter 3.5 cm) and incubated with 250 mg N kg−1 soil as (NH4)2SO4. The soil moisture levels were maintained at 70% water-holding capacity during the 21d incubation period. Glass vials with treatment soils were incubated in the dark at 20 °C with 80% RH; glass vials were covered with parafilm during the incubation period. Before 24 h of gas sampling, parafilm was removed from 100 mL glass vial followed by sealing of those vials with butyl rubber stopper and melamine white screw cap. After 24 h of closure, 30 mL of gas sample was collected from each vial using a syringe (50 mL vol) at different sampling intervals, 0–5, 7–11, 14, 17, 21 days incubation, and transferred to 10 mL pre-vacuum vial and N2O analysis was done using gas chromatograph (GC-14B, Shimadzu, Japan). After gas collections, rubber stopper was removed and air inside the glass vial was ventilated by aeration pump. The cumulative N2O emissions were obtained by summing the N2O data at each sampling days.
Statistical analysis
Statistical analysis for one-way and two-way ANOVA was done using SAS 9.1 software (SAS Inc., Cary, NC, USA).
Results
Plant growth and hydrophilic-BNI capacity in hydroponics
High-sorgoleone (HS) genetic-stocks showed significantly higher hydrophilic-BNI-activity from entire roots compared to LS (Fig. 1a). However, leaf NO3− concentrations of HS was significantly lower than in LS genotypes (Fig. 1b). In addition, shoot dry weight was positively correlated with shoot N content (Fig. S1). HS genetic-stocks had higher shoot dry weight (271% higher) and higher N content (249% higher) than in LS genotypes in hydroponic culture (Fig. S1).
Hydrophilic-BNI-activity and leaf NO3−-N concentration of contrasting BNI genetic-stocks of sorghum in hydroponic culture (a) Hydrophilic-BNI-activity (b) leaf NO3−-N concentration. LS genetic-stocks EC670350 and EC670402; HS genetic-stocks EC670311 and IS31861. ATU, allylthiourea units. Each bar represented the mean of three replicates with standard error, the different letter stood the significant difference under same N level analyzed by ANOVA at 0.05 (p < 0.05)
Shoot dry weight and N recovery in soil culture and field experiments
Consistent with the hydroponic trial, HS genetic-stocks showed higher (78%) shoot dry matter production and higher (52%) shoot nitrogen recovery compared to LS under high-N treatments (Fig. 2). However, under low-N treatments, only shoot dry weights were significantly higher (65%) in HS compared to LS genetic-stocks in glasshouse grown plants. Under field conditions, HS produced higher biomass (36%) and recovered higher amounts of shoot nitrogen (16%) compared to LS genetic stocks (Table 3).
Plant growth of contrasting BNI genetic-stocks of sorghum (LS and HS) in soil culture with low-N (50 mg N kg−1) and high-N treatment (250 mg N kg−1) in glasshouse experiment. (a) Shoot dry weight. (b) Shoot N content. 2 LS genetic-stocks EC670350 and EC670402; 2 HS genetic-stocks EC670311 and IS31861. Each bar represented the mean of three replicates with standard error, the different letter stood the significant difference under same N level analyzed by ANOVA at 0.05 (p < 0.05). There is an interaction between genetic-stocks and N level, F value = 5.01, p < 0.05 (Shoot dry weight); F value = 41.4, p < 0.01 (Shoot N content)
Soil NO3 −-N and NH4 +-N in pot soil culture
Sorgoleone production from roots had a major impact on soil N forms during different sampling days after the application of N fertilizer (Table 2). HS genotypes had significantly lower soil-NO3− levelscompared to LS genetic-stocks; these trends were more apparent under high-N fertilization compared to low-N treatments. For example, on the 6th day after the application of N fertilizer (DAF-6), soil-NO3− levels in two HS genotypes were 163 mg per kg and 83 mg per kg under high-N treatments, which were significantly lower than the 227 mg per kg and 304 mg per kg of two LS genotypes. Soil-NH4+ levels were consistently lower in HS genotypes compared to LS genotypes under high-N application, but not under low-N applications (Table 2). Both soil-NO3− and soil-NH4+ levels were significantly lower in HS compared to LS genotypes; this was largely due to substantially higher growth rates and dry matter production; this may have improved N uptake in HS genetic-stocks compared to LS genetic-stocks under high-N treatments (Table 2).
Potential Nitrification and Nitrification Rates
Potential nitrification in soils was significantly lower in HS compared to LS genotypes under high-N, but not under low-N treatments (Fig. 3) (based on glasshouse experiment). This suggests that HS genotypes can inhibit soil-nitrification (due to higher levels of sorgoleone production and hydrophilic-BNI release from roots) more effectively than LS genotypes. Potential nitrification was 68% lower in HS genotypes compared to LS under high-N conditions, but not so different under low-N conditions (Fig. 3). Field trials had confirmed this further that HS genetic-stocks had lower soil nitrification compared to LS genetic-stocks (Table 3).
PN of contrasting BNI genetic-stocks of sorghum (LS and HS) in soil culture with low-N (50 mg N kg−1) and high-N treatment (250 mg N kg−1) in glasshouse experiment. 2 LS genetic-stocks EC670350 and EC670402; and 2 HS genetic-stocks EC670311 and IS31861. Each bar represented the mean of three replicates with standard error, the different letter stood the significant difference under same N level analyzed by ANOVA at 0.05 (p < 0.05). There is an interaction between genetic-stocks and N level, F value = 41.2, p < 0.01
AOA and AOB abundance
Soil nitrifier populations (AOA and AOB) were significantly influenced by sorgoleone production capacity of genetic-stocks in glasshouse experiment ((Fig. 4). HS genotypes suppressed soil-AOA populations more effectively than LS genotypes regardless of N treatment (Fig. 4a). For example, under low-N conditions, AOA gene copies were 43% lower in HS compared to LS genotypes; under high-N conditions, AOA gene copies were 68% lower in HS than in LS genotypes. Soil-AOB populations were significantly lower in HS genetic-stocks compared to LS genetic-stocks in high-N treatments, but not in LN treatments (Fig. 4b). Compared to LS genetic-stocks, the AOB copies in HS genetic-stocks were reduced by 37% under high-N treatments. Field results also confirmed these trends, i.e. HS genotypes had higher inhibitory effect on AOA and AOB populations compared to LS genotypes (Table 3).
Abundance of AOA and AOB amoA genes of contrasting BNI genetic-stocks of sorghum (LS and HS) in soil culture with low-N (50 mg N kg−1) and high-N treatment (250 mg N kg−1) in glasshouse experiment. (a) AOA. (b) AOB. 2 LS genetic-stocks EC670350 and EC670402; and 2 HS genetic-stocks EC670311 and IS31861. Each bar represented the mean of three replicates with standard error, the different letter stood the significant difference under same N level analyzed by ANOVA at 0.05 (p < 0.05). There is an interaction between genetic-stocks and N level, F value = 10.51, p < 0.01 (AOA); F value = 9.03, p < 0.01 (AOB)
N2O emissions
Nitrogen applications had significantly increased soil-N2O emissions in pot experiment (Fig. 5a). N2O emissions were higher in high-N compared to low-N treatments. There was a 217% increase in N2O emissions in LS genetic stocks compared to an increase of only 32% in HS genetic stocks. The differences in N2O emissions were significant among sorghum genotypes only under high-N treatments, but not under low-N treatments (Fig. 5a). Soil-N2O emissions were 58% lower in HS genetic-stocks compared to LS genetic-stocks in HN treatments. The field results also confirm that HS genetic-stocks can clearly reduce soil-N2O emissions in root-zone soils, which were 19% lower than in LS genetic-stocks (Fig. 5b).
Cumulative N2O emissions of contrasting BNI genetic-stocks of sorghum (LS and HS) in soil culture and field experiment. (a) 2 LS genetic-stocks EC670350 and EC670402; and 2 HS genetic-stocks EC670311 and IS31861 with low-N (50 mg N kg−1) and high-N treatment (250 mg N kg−1) in glasshouse experiment; (b) LS-EC670350 and HS-EC670311 with N treatment in field experiment. Each bar represented the mean of three replicates with standard error, the different letter stood the significant difference under same N level analyzed by ANOVA at 0.05 (p < 0.05). There is an interaction between genetic-stocks and N level in soil culture, F value = 338.4, p < 0.01
Discussion
Biological nitrification inhibition (BNI) is a plant-mediated phenomenon that restrict nitrification process by releasing BNIs from plant roots (Subbarao et al. 2007a; 2012; 2017; 2021; Nardi et al. 2020; Ghatak et al. 2021; Zhang et al. 2021). Sorghum roots release sorgoleone, the major hydrophobic-BNI component, which determines to some extent the BNI-capacity of its root systems (Subbarao et al. 2013). This is further evident from the results during this study that suggest that high-sorgoleone producing genetic-stocks (HS) also have higher hydrophilic-BNI-capacity (Fig. 1a). The results demonstrate that HS genetic-stocks suppressed nitrifying bacteria and N2O emissions from soils compared to LS genetic-stocks based on soil-grown plants in glasshouse and from field studies (Figs. 4 and 5; Table 3). Also, HS genetic-stocks showed higher growth rates and biomass production in both hydroponic systems and in soil-grown plants compared to LS genetic-stocks (Figs. 2 and S1; Table 3). This is in conformity with earlier studies that suggested the suppressive effect of sorgoleone on soil nitrifier activity and soil-nitrate formation (Subbarao et al. 2013; Tesfamariam et al. 2014; Sarr et al. 2020). In addition, the higher biomass production of HS genetic-stocks appears to benefit from greater BNI-activity, with larger root size resulting in more hydrophilic-BNI in hydroponics and possibly in soil potted plants; this is potentially beneficial for inhibiting soil nitrification, achieving reciprocity in root biomass and released BNIs. Even so, field experiments also confirmed that higher released sorgoleone in rhizosphere soils (hydrophobic-BNI) can significantly inhibit soil nitrifying bacteria and N2O emissions, consequence in inhibiting soil nitrification is to promote the biomass of sorghum. Simply put, the function of BNI is to inhibit the nitrification of the soil, and the whole root system can uptake more ammonium, which is conducive to the formation of biomass (Subbarao and Searschinger 2021; Subbarao et al. 2021).
The hydrophilic-BNI activity in HS genetic-stocks was nearly 50% higher compared to LS genetic-stocks (based on hydroponically grown plants) (Fig. 1). Coupled with high sorgoleone release observed in HS genetic stocks, could possibly the reason for the observed lower nitrifier populations (AOA, AOB) in rhizosphere soils of greenhouse grown plants (Figs. 3 and 4; Table 3). Also, HS may facilitate the availability of more soil-NH4+ in root-zone, that can stimulate growth and possibly improve nitrogen uptake and can have multiplier impact on productivity. Replacing 20% of NO3− with NH4+ in nutrient solutions led to stimulation of plant growth in wheat and sorghum (Subbarao and Searchinger 2021). In addition, the presence of soil-NH4+ is known to stimulate BNIs production and release from sorghum roots (Zhu et al. 2012; Subbarao et al. 2013; Zeng et al. 2016; Di et al. 2018). Thus, high-sorgoleone production, in conjunction with high-levels of hydrophilic-BNI activity from roots can suppress soil-nitrifier activity more effectively in HS genetic-stocks compared to LS genetic-stocks. As NH4+ uptake and its assimilation results in rhizosphere acidification, which further activate proton pumping activity in the root plasma membrane (Subbarao et al. 2007b; Di et al. 2018; Zhang et al. 2021), that sustain hydrophilic-BNI release and acts as auto-feedback loop; Further, rhizosphere acidification associated with NH4+ uptake will also have other benefits, such as enhanced micro-nutrient availability (Subbarao et al. 2007a; 2009; 2021; Afzal et al. 2020), which can improve growth and productivity (Figs. S1 and 2). In addition, the interplay involving sorgoleone production, hydrophilic-BNI-activity of root systems, reduced nitrifier populations, lower soil-NO3− formation and lower soil-N2O emissions, all may have led to improved nitrogen uptake and better plant growth rates as evident from HS genetic-stocks compared to LS genetic-stocks.
Nevertheless, there could be many caveats to the above interpretation. During this study, the two HS genotypes are very similar in sorgoleone production (20.4 vs 20.7 μg g−1 rtwt, Table 1), yet their responses on AOA, AOB, PN, N2O emissions and biomass production were significantly different, suggesting that these observed responses could be influenced also from other factors. It needs to be emphasized that sorgoleone release from roots explains only part of BNI-capacity and significant BNI-capacity comes from hydrophilic-BNI activity release from roots. In addition, BNI-trait expression is a highly regulated function which can be influenced by environmental and growing conditions. Thus, unless we develop isogenic-lines for sorgoleone production, it would be extremely difficult to establish trait-value to overall plant growth in soil-based systems and in the field. The importance of sorgoleone in suppressing soil nitrifier activity was earlier established using sorgoleone amended soils (Subbarao et al. 2013; Tesfamariam et al. 2014).
Rapid nitrification has a major impact on soil-fertility as uncontrolled soil-nitrifier activity results in loss of soil-nitrogen from farmlands (due to nitrate leaching and denitrification), thus negatively affecting crop productivity and sustainability of production systems (Subbarao et al. 2006b; 2015; Karwat et al. 2018; 2019; Vazquez et al. 2020; Leon et al. 2021). Additions of N fertilizers led to accelerating soil nitrification (Fig. 3). The BNI function is better expressed under high-N applications compared to low-N treatments in both HS and LS genetic-stocks. HS genetic-stocks suppressed soil-nitrifier activity, lowered soil nitrification rates better than LS genetic-stocks (Figs. 3 and 4; Table 3). Soil-NO3− levels were lower in the soil-root-zones of HS genetic-stocks compared to LS genetic-stocks (Table 2). This was also reflected in leaf-NO3− levels, where HS genetic-stocks showed significantly lower leaf-NO3− levels than LS genetic-stocks (Fig. 1b). High-BNI genetic-stocks of Brachiaria pasture grasses also showed lower leaf-NO3− levels compared to low-BNI genetic-stocks (Karwat et al. 2019). Similarly, high BNI-capacity wheat genetic-stocks of BNI-Munal had nearly 30% lower leaf-NO3− compared to Munal-control (Subbarao et al. 2021).
Supplemental additions of NH4+ to nutrient solutions that rely entirely on NO3− -N led to stimulation of growth in several crops including sorghum and wheat (Britto and Kronzucker 2002; Subbarao and Searchinger 2021). The higher growth rates observed in HS genetic-stocks during this study, could be partly due to improved soil-NH4+ levels, that contribute to availability of dual nitrogen forms (NH4+ and NO3−) in root-zone, which is linked with high BNI-capacity of root systems, together suppress soil nitrifier activity better than in LS genetic-stocks; it is likely that most of the soil inorganic-N is in NO3− form in the root-zone of LS genetic-stocks.
Nitrifier populations of AOA and AOB dominate in most natural and agricultural ecosystems (Prosser and Nicol 2012; Hu et al. 2014; Kaur-Bhambra et al. 2021). In general, natural grasslands and ecosystems are dominated by AOA where AOB play relatively minor role and generally these are of low-N ecosystems (Subbarao et al. 2009; Prosser and Nicol 2012; Zhang et al. 2012; Hu et al. 2014; Byrnes et al. 2017). In contrast, agro-ecosystems receive large amounts of N additions repeatedly and AOBs tend to dominate in these high-N environments (Wang et al. 2017; Li et al. 2021b). During this study HS genetic-stocks have significantly stronger suppressive effect on soil-nitrifier populations than LS genetic-stocks (Fig. 4; Table 3). Furthermore, HS genetic-stocks appears to suppress AOAs more strongly than AOBs, which confirms earlier reports on sorghum (Sarr et al. 2020). In addition, N2O emissions were suppressed more strongly in HS genetic-stocks than in LS genetic-stocks in both soil grown plants in glasshouse and in and field experiment (Fig. 5). Generally, soil N2O are triggered from N inputs (Snider et al. 2015; Hink et al. 2018; Yao et al. 2020); our results demonstrate that maintaining low nitrifier-activity can slow down nitrification and N2O emissions (Tables 2, 3 and Fig. 5). These results clearly demonstrate that high-sorgoleone producing sorghum genetic-stocks suppress soil nitrifier activity, reduce soil-nitrate formation and lowered N2O emissions from soils compared to low-sorgoleone producing sorghum genetic-stocks.
Conclusions
Our results suggest substantial genotypic differences in sorgoleone production from sorghum roots. High sorgoleone producing (HS) genetic-stocks showed higher hydrophilic-BNI release from roots and more effectively suppressed soil nitrifier activity, reduced soil-nitrification, soil-N2O emissions, reduced leaf-nitrate levels have led to better growth and biomass production compared to LS genetic-stocks. The BNI-function appears to express better under high-N environments compared to low-N environments in both LS and HS genetic-stocks. These results support the hypothesis that HS genetic-stocks likely suppress soil nitrate formation and lowered N2O emissions compared to LS genetic-stocks.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request.
References
Afzal MR, Zhang M, Jin H, Wang G, Zhang M, Ding M, Raza S, Hu J, Zeng H, Gao X, Subbarao GV, Zhu Y (2020) Post-translation regulation of plasma membrane H+-ATPase is involved in the release of biological nitrification inhibitors from sorghum roots. Plant Soil 450:357–372. https://doi.org/10.1007/s11104-020-04511-6
Britto D, Kronzucker H (2002) NH4+ toxicity in higher plants: a critical review. J Plant Physiol 159:567–584. https://doi.org/10.1078/0176-1617-0774
Byrnes RC, Nuñez J, Arenas L, Rao I, Trujillo C, Alvarez C, Arango J, Rasche F, Chirinda N (2017) Biological nitrification inhibition by Brachiaria grasses mitigated soil nitrous emissions from bovine urine patches. Soil Biol Biochem 177:156–163. https://doi.org/10.1016/j.soilbio.2016.12.029
Coskun D, Britto DT, Shi W, Kronzucker HJ (2017) Nitrogen transformations in modern agriculture and role of biological nitrification inhibition. Nat Plants 3:17074. https://doi.org/10.1038/nplants.2017.74
Davidson EA (2009) The contribution of manure and fertilizer nitrogen to atmospheric nitrous oxide since 1860. Nat Geosci 2:659–662. https://doi.org/10.1038/ngeo608
Dayan FE, Rimando AM, Pan Z, Baerson SR, Gimsing AL, Duke SO (2010) Sorgoleone. Phytochem 10:1032–1039. https://doi.org/10.1016/j.phytochem.2010.03.011
Di T, Afzal MR, Yoshihashi T, Deshpande S, Zhu Y, Subbarao GV (2018) Further insights into underlying mechanisms for the release of biological nitrification inhibitors from sorghum roots. Plant Soil 423:99–110. https://doi.org/10.1007/s11104-017-3505-5
Fu Q, Abadie M, Blaud A, Carswell A, Misselbrook T, Clark I, Hirsch P (2020) Effects of urease and nitrification inhibitors on soil N, nitrifier abundance and activity in a sandy loam soil. Biol Fertil Soils 25:185–194. https://doi.org/10.1007/s00374-019-01411-5
Gao X, Wu M, Xu R, Pan R, Kim H, Wang X, Liao H (2014) Root interactions in a maize/soybean intercropping system control soybean soil-borne disease, red crown rot. PLoS ONE 9:e95031. https://doi.org/10.1371/journal.pone.0095031
Gubry-Rangin C, Nicol G, Prosser JI (2020) Archaea rather that bacteria control nitrification in two agricultural acidic soils. FEMS Microbiol Ecol 74:566–574. https://doi.org/10.1111/j.1574-6941.2010.00971.x
Hink L, Gubry-Rangin NGW, Prosser JI (2018) The consequences of niche and physiological differentiation of archaeal and bacterial ammonia oxidisers for nitrous oxide emissions. ISME J 12:1084–1093. https://doi.org/10.1038/s41396-017-0025-5
Hu HW, Xu ZH, He JZ (2014) Ammonia-oxidizing archaea play a predominant role in acid soil nitrification. Adv Agron 125:261–302. https://doi.org/10.1016/B978-0-12-800137-0.00006-6
Ishikawa T, Subbarao GV, Ito O, Okada K (2003) Suppression of nitrification and nitrous oxide emission by tropical grass Brachiaria humidicola. Plant Soil 255:413–419. https://doi.org/10.1007/978-94-017-2923-9_40
Karwat H, Egenolf K, Nuñez J, Rao I, Rasche F, Arango J, Moreta D, Arevalo A, Cadisch G (2018) Low 15N natural abundance in shoot tissue of Brachiaria humidicola is and indicator of reduced N losses due to biological nitrification inhibition (BNI). Front Microbiol 9:2383. https://doi.org/10.3389/fmicb.2018.02383
Karwat H, Sparke M, Fasche F, Arango J, Nuñez J, Rao I, Moreta D, Cadisch G (2019) Nitrate reductase activity in leaves as a plant physiological indicator of in vivo biological nitrification inhibition by Brachiaria humidicola. Plant Physiol Biochem 137:113–120. https://doi.org/10.1016/j.plaphy.2019.02.002
Kaur-Bhambra J, Wardak D, Prosser J, Gubry-Rangin C (2021) Revisiting plant biological nitrification inhibition efficiency using multiple archaeal and bacterial ammonia-oxidising cultures. Biol Fertil Soils. https://doi.org/10.1007/s00374-020-01533-1
Leon A, Subbarao GV, Kishii M, Matsumoto N, Kruseman G (2021) An ex ante life cycle assessment of wheat with high biological nitrification inhibition capacity. Environ Sci Pollut Res. https://doi.org/10.1007/s11356-021-16132-2
Li W, Ma J, Bowatte S, Hoogendoorn C, Hou F (2021a) Evidence of differences in nitrous oxide emissions and biological nitrification inhibition among Elymus grass species. Biol Fertil Soils. https://doi.org/10.1007/s00374-021-01592-y
Li Y, Zhang Y, Chapman SJ, Yao H (2021b) Biological nitrification inhibition by sorghum root exudates impacts ammonia-oxidizing bacteria but not ammonia-oxidizing archaea. Biol Fertil Soils 57:399–407. https://doi.org/10.1007/s00374-020-01538-w
Lu Y, Zhang X, Ma M, Zu W, Kronzucker HJ, Shi W (2021) Syringic acid from rice as a biological nitrification and urease inhibitor and its synergism with 1,9-decanediol. Biol Fertil Soils. https://doi.org/10.1007/s00374-021-01584-y
Maaz TM, Sapkota TB, Eagle AJ, Kantar MB, Bruulsema TW, Majumdar K (2021) Meta-analysis of yield and nitrous oxide outcomes for nitrogen management in agriculture. Glob Chang Biol 27:2342–2360. https://doi.org/10.1111/gcb.15588
Nakamura S, Sarr PS, Takahashi M, Ando Y, Subbarao GV (2020) The contribution of root turnover on biological nitrification inhibition and its impact on the ammonia-oxidizing archaea under brachiaria cultivations. Agronomy 10:1003. https://doi.org/10.3390/agronomy10071003
Nardi P, Laanbroek HJ, Nicol GW, Renella G, Cardinale M, Pietramellara G, Weckwerth W, Trinchera A, Ghatak A, Nannipieri P (2020) Biological nitrification inhibition in the rhizosphere: determining interactions and impact on microbially mediated processes and potential applications. FEMS Microbiol Rev 44:874–908. https://doi.org/10.1093/femsre/fuaa037
O’Sullivan CA, Fillery IRP, Poper MM, Richards RA (2016) Identification of several wheat land races with biological nitrification inhibition capacity. Plant Soil 404:61–74. https://doi.org/10.1007/s11104-016-2822-4
Otaka J, Subbarao GV, Ono H, Yoshihashi T (2021) Biological nitrification inhibition in maize-isolation and identification of hydrophobic inhibitors from root exudates. Biol Fertil Soils. https://doi.org/10.1007/s00374-021-01577-x
Prosser JI, Nicol GW (2012) Archaeal and bacterial ammonia-oxidisers in soil: the quest for niche specialization and differentiation. Trends Microbial 20:523–531. https://doi.org/10.1016/j.tim.2012.08.001
Prosser JI, Hink L, Gubry-Rangin C, Nicol GW (2020) Nitrous oxide production by ammonia oxidizers: Physiological diversity, niche differentiation and potential mitigation strategies. Glob Chang Biol 26:103–118. https://doi.org/10.1111/gcb.14877
Sarr PS, Ando Y, Nakamura S, Deshpande S, Subbarao GV (2020) Sorgoleone release from sorghum roots shapes the composition of nitrifying populations, total bacteria, and archaea and determines the level of nitrification. Biol Fertil Soils 56:145–166. https://doi.org/10.1007/s00374-019-01405-3
Sarr PS, Nakamura S, Ando Y, Iwasaki S, Subbarao GV (2021) Sorgoleone production enhances mycorrhizal association and reduces soil nitrification in sorghum. Rhizosphere 17:100283. https://doi.org/10.1016/j.rhisph.2020.100283
Snider D, Thompson K, Wagner-Riddle C, Spoelstra J, Dunfield K (2015) Molecular techniques and stable isotope ratios at natural abundance give complementary inferences about N2O production pathway in an agricultural soil following a rainfall event. Soil Biol Biochem 88:197–213. https://doi.org/10.1016/j.soilbio.2015.05.021
Stevens CJ (2019) Nitrogen in the environment. Science 363:578–580. https://doi.org/10.1126/science.aav8215
Subbarao GV, Searchinger TD (2021) Opinion: A “more ammonium solution” to mitigate nitrogen pollution and boost crop yields. Proc Natl Acad Sci (PNAS) (USA) 118:e2107576118. https://doi.org/10.1073/pnas.2107576118
Subbarao GV, Ishikawa T, Ito O, Nakahara K, Wang HY, Berry WL (2006a) A bioluminescence assay to detect nitrification inhibitors released from plant roots: a case study with Brachiaria humidicola. Plant Soil 288:101–112. https://doi.org/10.1007/s11104-006-9094-3
Subbarao GV, Ito O, Sahrawat KL, Ishikawa T, Berry WL, Nakahara K, Ishikawa T, Watanabe T, Suenaga K, Rondon M, Rao IM (2006b) Scope and strategies for regulation of nitrification in agricultural systems-challenges and opportunities. Crit Rev Plant Sci 25:303–335. https://doi.org/10.1080/07352680600794232
Subbarao GV, Rondon M, Ito O, Ishikawa T, Rao IM, Nakahara K, Lascano C, Berry WL (2007a) Biological nitrification inhibition (BNI) – is it a widespread phenomenon? Plant Soil 294:5–18. https://doi.org/10.1270/jsbbs.59.529
Subbarao GV, Wang HY, Ito O, Nakahara K, Berry WL (2007b) NH4+ triggers the synthesis and release of biological nitrification inhibition compounds in Brachiara humidicola roots. Plant Soil 290:245–257. https://doi.org/10.1007/s11104-006-9156-6
Subbarao GV, Nakahara K, Hurtodo MP, Ono H, Moreta DE, Salcedo AF, Yoshihashi T, Ishikawa T, Ishitani M, Ohnishi KM, Yoshida M, Rondon M, Rao IM, Lascano CE, Berry WL, Ito O (2009) Evidence for biological nitrification inhibition in Brachiaria pastures. Proc Natl Acad Sci (PNAS) (USA) 106:17302–17307. https://doi.org/10.1073/pnas.0903694106
Subbarao GV, Sahrawat KL, Nakahara K, Ishikawa T, Kudo N, Kishii M, Rao IM, Hash CT, George TS, Srinivasa RP, Nardi P, Bonnett D, Berry WL, Suenage K, Ito O, Lata JC (2012) Biological nitrification inhibition (BNI) – a noval strategy to regulate nitrification in agricultural systems. Adv Agron 114:249–302. https://doi.org/10.1016/B978-0-12-394275-3.00001-8
Subbarao GV, Nakahara K, Ishikawa T, Ono H, Yoshida M, Yoshihashi T, Zhu Y, Zakir H, Deshpande SP, Hash CT, Sahrawat KL (2013) Biological nitrification inhibition (BNI) activity in sorghum and its characterization. Plant Soil 366:243–259. https://doi.org/10.1007/s11104-012-1419-9
Subbarao GV, Yoshihashi T, Worthington M, Nakahara K, Ando Y, Sahrawat KL, Rao IM, Lata JC, Kishii M, Braun HJ (2015) Suppression of soil nitrification by plants. Plant Sci 233:155–164. https://doi.org/10.1016/j.plantsci.2015.01.012
Subbarao GV, Arango J, Masahiro K, Hooper AM, Yoshihashi T, Ando Y, Nakahara K, Deshpande S, Ortiz-Monasterio I, Ishitani M, Peters M, Chirinda N, Wollenberg L, Lata JC, Gerard B, Tobita S, Rao IM, Braum HJ, Kommerell V, Tohme J, Iwanaga M (2017) Genetic mitigation strategies to tackle agricultural GHG emissions: The case for biological nitrification inhibition technology. Plant Sci 262:165–168. https://doi.org/10.1016/j.plantsci.2017.05.004
Subbarao GV, Kishii M, Bozal-Leorri A, Ortiz-Monasterio I, Gao X, Ibba MI, Karwat H, Gonzalez-Moro MB, Gonzalez-Murua C, Yoshihashi T, Tobita S, Kommerell V, Braun HJ, Iwanaga M (2021) Enlisting wild-grass-genes to combat nitrification in wheat farming: a nature-based solution. Proc Natl Acad Sci (PNAS) (USA) 118:e2106595118. https://doi.org/10.1073/pnas.2106595118
Sun L, Lu YF, Yu FW, Kronzucker HJ, Shi WM (2016) Biological nitrification inhibition by rice root exudates and its relationship with nitrogen-use efficiency. New Phytol 212:646–656. https://doi.org/10.1111/nph.14057
Tesfamariam T, Yoshinaga H, Deshpande SP, Rao PS, Sahrawat KL, Ando Y, Nakahara K, Hash CT, Subbarao GV (2014) Biological nitrification inhibition in sorghum: the role of sorgoleone production. Plant Soil 379:325–335. https://doi.org/10.1007/s11104-014-2075-z
Varshney RR, Subbarao GV, Chaturvedi P, Weckwerth (2021) Root exudation of contrasting drought-stressed pearl millet genotypes conveys varying biological nitrification inhibition (BNI) activity. Biol Fertil Soils. https://doi.org/10.1007/s00374-021-01578-w
Vazquez E, Teutscherova N, Dannenmann M, Tochterle P, Butterbach-Bahl K, Pulleman M, Arango J (2020) Gross nitrogen transformations in tropical pasture soils as affected by Urochloa genotypes differing in biological nitrification inhibition (BNI) capacity. Soil Biol Biochem 151:108058. https://doi.org/10.1016/j.soilbio.2020.108058
Villegas D, Arevalo A, Nuñez J, Mazabel J, Subbarao G, Rao I, Vega J, Arango J (2020) Biological nitrification inhibition (BNI): phenotyping of a core germplasm collection of the tropical forage grass Megathyrsus maximus under greenhouse conditions. Front Plant Sci 11:820. https://doi.org/10.3389/fpls.2020.00820
Wang Q, Liu YR, Zhang CJ, Zhang LM, Han LL, Shen JP, He JZ (2017) Responses of soil nitrous oxide production and abundance and composition of associated microbial communities to nitrogen and water amendment. Biol Fertil Soils 53:601–611. https://doi.org/10.1007/s00374-017-1203-3
Yao Z, Pelster D, Liu C, Zheng X, Butterbach-Bahl K (2020) Soil N intensity as a measure to estimate annual N2O and NO fluxes from natural and managed ecosystems. Curr Opin Environ Sustain 47:1–6. https://doi.org/10.1016/j.cosust.2020.03.008
Zakir HAKM, Subbarao GV, Pearse SJ, Gopalakrishnan S, Ito O, Ishikawa T, Kawano N, Nakahara K, Yoshihashi T, Ono H, Yoshida M (2008) Detection, isolation and characterization of a root-exuded compound, methyl 3-(4-hydroxyphenyl) propionate, responsible for biological nitrification inhibition by sorghum (Sorghum Bicolor). New Phytol 180:442–451. https://doi.org/10.1111/j.1469-8137.2008.02576.x
Zeng H, Di T, Zhu Y, Subbarao GV (2016) Transcriptional response of plasma membrane H+ ATPase genes to ammonium nutrition and its functional link to the release of biological nitrification inhibitors from sorghum roots. Plant Soil 398:301–312. https://doi.org/10.1007/s11104-015-2675-2
Zhang LM, Hu HW, Shen JP, He JZ (2012) Ammonia oxidizing archaea have more important role than ammonia-oxidizing bacteria in ammonia oxidation of strongly acid soils. ISME J 6:1032–1045. https://doi.org/10.1038/ismej.2011.168
Zhang M, Zeng H, Afzal MR, Gao X, Li Y, Subbarao GV, Zhu Y (2021) BNI-release mechanisms in plant root system: current status of understanding. Biol Fertil Soils. https://doi.org/10.1007/s00374-021-01568-y
Zhu Y, Zeng HQ, Shen QR, Ishikawa T, Subbarao GV (2012) Interplay among NH4+ uptake, rhizosphere pH and plasma membrane H+-ATPase determine the release of BNIs in sorghum roots – possible mechanisms and underlying hypothesis. Plant Soil 358:131–141. https://doi.org/10.1007/s11104-012-1151-5
Acknowledgements
We would like to appreciate the JIRCAS Visiting Research Fellowship Program, and thank Makoto Yamamoto, Yukiko Ishikawa, Hiroko Aoki, Yoko Koizumi, Raphael Obias Mubanga, Sanae Suzuki, Masami Aoyama for their help with the laboratory and field experiments.
Funding
This work was supported by Japan International Research Center for Agricultural Sciences (JIRCAS) Visiting Research Fellowship Program.
Author information
Authors and Affiliations
Contributions
XG, GVS and TY designed the experiments. XG conducted the experiments. GVS performed biological studies. TY responsible for sorgoleone analysis. PSS conducted amoA gene measurements. KU performed N2O emission measurements. XG and GVS wrote the manuscript. All authors have contributed to the writing of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Responsible Editor: Richard J. Simpson.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
11104_2022_5474_MOESM1_ESM.pptx
Fig. S1 The correlation analysis of contrasting BNI genetic-stocks of sorghum between shoot N content and shoot dry weight in hydroponic culture, n = 12. LS genetic-stocks EC670350 and EC670402; HS genetic-stocks EC670311 and IS31861. For correlation analysis: R2 value on asterisk * and ** denoted with p < 0.05 and p < 0.01, respectively (PPTX 125 KB)
Rights and permissions
About this article
Cite this article
Gao, X., Uno, K., Sarr, P.S. et al. High-sorgoleone producing sorghum genetic stocks suppress soil nitrification and N2O emissions better than low-sorgoleone producing genetic stocks. Plant Soil 477, 793–805 (2022). https://doi.org/10.1007/s11104-022-05474-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-022-05474-6