Abstract
Aims
The combined effects of nitrogen (N) deposition and management practices on fine root decomposition remain unknown. The objective of this study was to investigate the effects of the two factors on fine root decay in Moso bamboo plantations.
Methods
This study was performed over a three-year period and included three nitrogen treatments (30, 60, and 90 kg N ha−1 yr.−1) and two management practices (conventional and intensive).
Results
Fine root decomposition was significantly affected by N inputs and management practices both separately and in combination (P < 0.01). N inputs had a stronger effect than management practices. The low-N input (30 kg N ha−1 yr.−1) accelerated fine root decomposition and nutrient release, whereas high-N inputs (≥ 60 kg N ha−1 yr.−1) inhibited decomposition and nutrient release. Moreover, intensive management practices strengthened the inhibitory effects of the high-N inputs.
Conclusions
Moderate N deposition (< 60 kg N ha−1 yr.−1) may decrease soil carbon storage but increase Moso bamboo productivity, while excessive N deposition (≥ 90 kg N ha−1 yr.−1) may have opposing effects. The combined effects of management practices and nitrogen amendment should be considered when estimating the effects of increasing atmospheric N deposition on plantation ecosystems.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Fine roots (≤2 mm in diameter) account for 33 % of the annual terrestrial net primary production (NPP) (Jackson et al. 1997). Fine root decomposition is the key process by which root nutrients are mineralized and carbon (C) is lost. Carbon and nitrogen (N) fluxes through fine root turnover may exceed those for aboveground C and N fluxes (Nadelhoffer and Raich 1992). Fine root decomposition across different forest ecosystems has been previously studied (Ostertag and Hobbie 1999; Silver and Miya 2001; Yang et al. 2004; Lin et al. 2011; Sun et al. 2013). Root chemistry, including the C:N ratio and the concentrations of N and lignin, plays a greater role than climatic factors in controlling decomposition rates on a global scale (Silver and Miya 2001; Zhang et al. 2008).
Atmospheric N input to terrestrial ecosystems has sharply increased in the last three decades (Galloway et al. 2008; Reay et al. 2008). The largest increases occurred in East Asia and South Asia and are predicted to continue (Reay et al. 2008; Tian et al. 2015), particularly in subtropical China (Liu et al. 2013). Nitrogen deposition can have positive, negative, or no effects on surface leaf litter decomposition by changing soil N availability, and the diversity and activity of soil microbes (Knorr et al. 2005; Fang et al. 2007; Hobbie 2008; Keeler et al. 2009; Song et al. 2014, 2015). However, little information is available on the effects of N deposition on underground fine root decomposition (Tu et al. 2015).
Moso bamboo (Phyllostachys edulis) is widely distributed in southern China and neighboring countries (Li et al. 2000). The Moso bamboo forest in China has an area of 3.87 million ha, which comprises 70 % of the total bamboo forest area in China and 80 % of the Moso bamboo forest area worldwide (Li and Lei 2010; Song et al. 2011). In recent decades, farmers have increasingly adopted intensive management (IM) practices such as fertilization, plowing, and cleaning understory vegetation, none of which was practiced in the conventional management (CM) of Moso bamboo forest (Zhou et al. 2010; Song et al. 2011, 2015). IM practices have increased bamboo production and soil organic carbon (SOC) mineralization (Zhou et al. 2006; Jiang et al. 2009) but have also decreased soil microbe diversity (Xu et al. 2008; Li et al. 2016).
In subtropical China, the Moso bamboo plantation region receives 30–37 kg N ha−1 yr.−1 from the atmosphere (Xie et al. 2008; Cui et al. 2014; Jia et al. 2014). The combined effects of increasing N deposition and anthropogenic management practices on Moso bamboo plantation ecosystems have been receiving much attention (Song et al. 2015, 2016). Our previous study showed that simulated low-N addition (30 kg ha−1 yr.−1) had significant positive effects on leaf litter decomposition in a Moso bamboo plantation, while excessive N addition (90 kg ha−1 yr.−1) had the opposite effect and was also significant (P < 0.05). IM alone did not significantly affect leaf litter decomposition but it amplified both the positive effects oflow-N addition and the negative effects of high-N addition (Song et al. 2015). Moso bamboo roots account for 19.2 % of the biomass and 19.8 % of the carbon storage within the whole plant. These proportions are much higher than those of the leaves (5.3 % and 4.9 %, respectively) (Zhou and Jiang 2004). The roots play important roles in carbon- and nutrient inputs to the soil. Nevertheless, the combined effects of N deposition and management practices on Moso bamboo fine root decomposition remains unclear. Consequently, our understanding of the complex response of the Moso bamboo forest to atmospheric N depositions is limited.
In the present study, fine root decomposition in a Moso bamboo forest was investigated for 36 months. A 24-month leaf litter decomposition experiment had just been completed there (Song et al. 2015). The objectives of the study were to (1) determine how fine root decomposition responds to management practices; (2) how fine root decomposition responds to increasing atmospheric N deposition; and (3) whether management practices influence the effects of N deposition on fine root decomposition.
Materials and methods
Study site
The details of study site can be found in Song et al. (2015). Briefly, this study site receives a mean annual precipitation of 1420 mm and has a mean annual temperature of 15.6 °C. CM and IM Moso bamboo plantations were established in the late 1970s and in 2001, respectively.
Experimental design and measurement
In November 2012, we established 12 CM plots and 12 IM plots of 20 m × 20 m and each one was surrounded by a 20-m buffer zone. The initial characteristics of both the CM and IM are summarized in the literature (Song et al. 2015). The local background atmospheric N deposition rate is 30–37 kg N ha−1 yr.−1 (Xie et al. 2008; Cui et al. 2014; Jia et al. 2014). Liu et al. (2016) reported that NH4 + and NO3 − accounted for 56.1 % and 43.9 % of the wet N deposition in China, respectively. The average NH4 +:NO3 − was 1.28. Based on atmospheric N deposition and simulation methods used in similar studies (Mo et al. 2007; Fang et al. 2007), we used NH4NO3 as the nitrogen source for a low-N (30 kg ha−1 yr.−1) treatment (N30), a medium-N (60 kg ha−1 yr.−1) treatment (N60), and a high-N (90 kg ha−1 yr.−1) treatment (N90). Three replicate plots per treatment and the control (N-free addition) were randomized in each management type.
Beginning in January 2013 and each month thereafter, NH4NO3 was quantitatively dissolved in 10 L water and sprayed evenly onto the forest floor of each plot. Each control plot received 10 L of N-free water to control for the effects of water added in the form of 0.3 mm rainfall/year (Song et al. 2015).
Fine root litter collection, decomposition and chemical analysis
It has been reported that >80 % of the Moso bamboo roots are localized in the top 30 cm of soil (Zhou and Fu 2004). Fine roots in the top 40 cm of soil were collected outside the treatment plots in July 2012. At that time, the fine root biomass was at its annual maximum (Zhou and Fu 2008). In the laboratory, the roots were cleaned, air-dried to a constant weight, then cut into approximately 5-cm-long pieces. Subsamples were oven dried at 65 °C to determine the water, carbon, N, phosphorus (P), and lignin content of the air-dried samples (Table 1).
The fine root decomposition rate was determined using the litterbag method (Bloomfield et al. 1993; Lin et al. 2011). Litterbags were made of polypropylene 10 cm × 10 cm in size and with a 0.1 mm mesh. Five grams air-dried fine roots were placed in each litterbag. In December 2012, eighteen litterbags were spaced equally in three blocks per plot. In each block (0.5 m × 1.5 m), the soil was sliced at a 45° angle down to the mineral horizon at a depth of 15 cm. Six connected litterbags were inserted at 20-cm intervals into each incision. There were 432 litterbags used in the 24 treatment plots.
Three replicate litterbags were retrieved from each plot (one from each block) after 4, 8, 12, 18, 24, and 36 months. The litterbags were air dried. Soil and other materials adhering to them were removed from them with a fine brush and tongs. Fine roots were oven-dried at 65 °C to a constant weight. Standard methods were used to determine total carbon, N, P, and lignin concentrations. Details can be found in Song et al. (2015).
Data and statistical analysis
The remaining fine root mass in each sample was expressed as a percentage of the initial fine root dry weight. We applied first-order exponential decay model Xt/X0 = e -kt to fit the decomposition data (Olson 1963). Xt is the net oven-dried weight remaining at time t; X0 is the initial oven-dried weight; and k is the annual decomposition rate constant (yr−1).
The amounts of nutrients released from decomposing fine roots were expressed as percentages of the initial nutrient content, and were calculated by the eq. E = [(Mt × Ct)/(M0 × C0)] × 100, where E is the amount of nutrients released (%); M0 is the initial oven-dried mass (g); C0 is the initial nutrient concentration (mg g−1); Mt. is the oven-dried mass at time t; and Ct is the nutrient concentration at time t. E > 100 indicates nutrient immobilization; and E < 100 indicates nutrient release (Brandt et al. 2010; Song et al. 2014).
A one-way analysis of variance (ANOVA) and posthoc multiple comparisons were performed using the Bonferroni method to determine the statistical significance of the differences among the annual decomposition rates. A two-way ANOVA was applied to determine the effect of combining N deposition and management practices on the decomposition rates. The data satisfied the assumption of homogeneity of variance. Statistical analyses were conducted using SPSS (Statistical Package for the Social Sciences) 18.0 for Windows (SPSS Inc., Chicago, Illinois).
Results
Fine root mass loss and annual decomposition rate
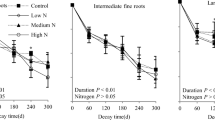
In the CM plots (Fig. 1, Table 2), fine root mass was rapidly lost (48.4–61.8 %) in the first twelve months. The rate of mass loss then slowed down (eventually to zero) within the final twelve months. Maximum fine root mass loss and annual decomposition rate occurred in the N30 treatment. The decay rate was 10.2 % greater than that in the control (P < 0.05). The minimum decay rate occurred in the N90 treatment and was 18.4 % less than that in the control (P < 0.05). No significant differences were found between the N60 treatment and the control or the N30 treatment (P > 0.05). In the IM plots, the fine root mass loss pattern resembled that of the CM plots. The highest decomposition rate occurred in the N30 treatment and the lowest in the N90 treatment (Fig. 1, Table 2).
Mean and standard deviation (n = 3) of the remaining fine root mass as a percentage of the initial mass over the 36-monthfine root decomposition analysis of Phyllostachys edulis in a Moso bamboo forest under conventional management (CM) and intensive management (IM) and four treatments: low-N treatment (N30, 30 kg ha−1 yr.−1), medium-N treatment (N60, 60 kg ha−1 yr.−1), high-N treatment (N90, 90 kg ha−1 yr.−1), and the control treatment
No significant differences in the fine root decomposition rates were found between the CM and IM plots for the control and N30 treatments (P > 0.05). The rates were significantly higher in the CM plots than the IM plots for the N60 and N90 treatments (P < 0.05) (Table 2). A two-way ANOVA showed that the N addition treatments and management practices had significant effects both separately and in combination on the fine root decomposition rates (P < 0.01) (Table 3).
N and P loss dynamics
In the CM plots (Fig. 2), for all four treatments, N was immobilized within the first eight months of decomposition. The maximum immobilization rate was 143.7 % and occurred in the N90 treatment. The net N loss was rapid for twenty-four months, then the rate decreased thereafter. The fastest fine root N losses occurred under the N30 and N60 treatments. In the IM plots, N was immobilized mainly within the first four months. The maximum immobilization rate was 171.2 % and occurred in the N90 treatment. The net N loss was rapid for eighteen months, then the rate decreased thereafter. N loss was fastest for the N30 treatment and the control.
Mean root nitrogen (N, % of initial) and standard deviation over time in the fine root decomposition of Phyllostachys edulis in a Moso bamboo forest under conventional management (CM) and intensive management (IM) and four treatments: low-N treatment (N30, 30 kg ha−1 yr.−1), medium-N treatment (N60, 60 kg ha−1 yr.−1), high-N treatment (N90, 90 kg ha−1 yr.−1), and the control treatment
The immobilization rate of P resembled that of N in the first four months, in both the CM and IM plots, and under all four treatments. Maximum immobilization rates of 138.3 % and 173.3 % were found in the CM and IM plots, respectively (Fig. 3). After the first four months, the net loss of P was rapid but tended to slow after eighteen months. In both the CM and IM plots, P loss was fastest under the N30 treatment and slowest under the N90 treatment.
Mean root phosphorus (P, % of initial) and standard deviation over time in the fine root decomposition of Phyllostachys edulis in a Moso bamboo forest under conventional management (CM) and intensive management (IM) and four treatments: low-N treatment (N30, 30 kg ha−1 yr.−1), medium-N treatment (N60, 60 kg ha−1 yr.−1), high-N treatment (N90, 90 kg ha−1 yr.−1), and the control treatment
Discussion
Fine root decomposition
Although the fine root decay rate was 8.2 % higher in IM than CM for the N-free treatment, this difference was not significant (Table 2). Similar effects on leaf litter decomposition were observed at the same site (Song et al. 2015). IM practices can increase soil microbe and enzyme activity, thereby facilitating SOC mineralization (Balesdent et al. 1998; Xu et al. 1998). Our results indicate that these beneficial effects did not significantly contribute to differences in fine root degradation.
The low-N treatment (30 kg N ha−1 yr.−1) increased the fine root decomposition rate, particularly in the CM plots (Table 2). This effect may be attributed to the N inputs, which improved soil enzyme activity and N availability (Knorr et al. 2005; Manning et al. 2008). Nevertheless, high-N inputs (90 kg N ha−1 yr.−1) significantly decreased fine root decomposition rates (Table 2). This result is consistent with the inhibitory effect of high-N treatment on aboveground leaf litter decomposition (Song et al. 2015). It was caused mainly by the suppression of lignin degradation. Excessive N inputs inhibit ligninolytic enzyme activity (Fog 1988; Carreiro et al. 2000; Thirukkumaran and Parkinson 2000; Hobbie et al. 2012). Inorganic N reacts with lignin and phenolic substances during decomposition and forms new polymers that are resistant to degradation (Axelsson and Berg 1988; Berg et al. 2001; Hobbie et al. 2012). In the early stages of decomposition, soluble- and low molecular weight compounds are rapidly lost. Thereafter, lignin degradation predominates in the litter decomposition process (Berg and McClaugherty 2008; Austin and Ballaré 2010; Manzoni et al. 2010). Thus, the inhibition of lignin degradation by excessive N significantly contributes to the negative effects of high-N inputs on fine root decomposition (Fig. 1). In fact, the fine roots of Moso bamboo have a high lignin content (Table 1). Tu et al. (2015) demonstrated the same decomposition mechanism in Pleioblastus amarus (Amarus bamboo) fine root.
Two-way ANOVA showed that, individually, N inputs and management practices separately altered fine root decomposition rates significantly. The combination of the two factors had even stronger effects (Table 3). The medium-N treatment (60 kg ha−1 yr.−1) significantly decreased the fine root decomposition rate in the IM plots but not in the CM plots. One possible explanation is that the IM plot received high N inputs (67.5 kg ha−1 yr.−1) from annual fertilization. This extra N, combined with the medium-N treatment and background atmospheric N deposition (30–37 kg ha−1 yr.−1) resulted in an initial excessive N input (> 150 kg ha−1 yr.−1) and greater inhibition of fine root decomposition in the IM plots than the CM plots. The inhibitory effect in the CM plot only occurred under high-N treatment (90 kg ha−1 yr.−1). Tu et al. (2015) reported that simulated N addition (≥ 50 kg ha−1 yr.−1) combined with high background atmospheric N deposition (95 kg ha−1 yr.−1) significantly decreased fine root decay in Pleioblastus amarus.
Excessive N inputs can greatly increase soil TN, losses of NO3 − and cations from the soil solution, and soil H+ via the NH4 + nitration reaction (Matson et al. 1999; Xu 2013). These changes resulted in significant reduction in soil pH in Moso bamboo plantations (Song et al. 2016), ligninolytic activity (Sinsabaugh 2010; Tu et al. 2015), soil C availability for microbial decomposers (Thirukkumaran and Parkinson 2000), and fine root decomposition rates.
Fine root nutrient losses
The immobilization or mineralization of N and P derived from decomposing litter generally occur simultaneously and are determined primarily by the litter substrate quality and the microbial decomposer stoichiometric requirements (Manzoni et al. 2008, 2010; Xu and Hirata 2005). A net N or P loss occurs when the decomposing litter C:N ratio falls below the critical threshold of 5–15 (Manzoni et al. 2008; Parton et al. 2007) or the C:P falls below 200–480 (Gosz et al. 1973; Manzoni et al. 2010). When N and P are insufficient in decomposing litter, microbial decomposers usually immobilized them from the environment to maintain stoichiometric homeostasis (Manzoni et al. 2010; Mary et al. 1996).
In the present study, both the fine root C:N ratio (110.7) and C:P ratio (1568.2) (Table 1) far exceeded the critical thresholds, so both N and P were mineralized in the initial stage of root decomposition. Moreover, in the IM plots, annual fertilization provided large amounts of N and P, increasing the availability of immobilized N and P to microbial decomposers and resulting in greater N and P mineralization than in the CM plots. The extra inputs from the high-N treatment also resulted in stronger immobilization than that observed with the low-N treatment (Figs. 2, 3). With the immobilization of N and P and the loss of C in the decomposing roots, the ratios of C:N and C:P declined and met the stoichiometric requirements of the microbial decomposers. Consequently, a net loss of nutrients occurred after several months. In the later stages of decomposition, cumulative N inputs may decrease the availability of C to microbial decomposers (Andersson et al. 2004; Thirukkumaran and Parkinson 2000). Moreover, the polymers generated from lignin degradation during excessive N input (Axelsson and Berg 1988; Berg et al. 2001; Hobbie et al. 2012) also limit N and P losses.
Fine roots and leaf litter are the main plant residue inputs to forest soil (Van Groenigen et al. 2006). In Moso bamboo plantations, the roots have much greater biomass and store far more carbon than do the leaves (Zhou and Jiang 2004). Therefore, the roots contribute more carbon and nutrient to the soil than the leaves. Our previous study in the same site found that Moso bamboo plantations were still deficient in N and P (Song et al. 2016). Our results suggest that moderate atmospheric N deposition (<60 kg ha−1 yr.−1, of which <30 kg ha−1 yr.−1 is simulated N addition and <30 kg ha−1 yr.−1 is atmospheric N deposition) may decrease soil C storage but increase Moso bamboo productivity by accelerating fine root and leaf litter decomposition. Excessive atmospheric N deposition (≥90 kg ha−1 yr.−1, of which 60 kg ha−1 yr.−1 is simulated N addition and 30 kg ha−1 yr.−1 is atmospheric N deposition) may increase soil C storage but decrease Moso bamboo productivity by inhibiting decomposition. Moreover, current IM practices will enhance these effects. Therefore, it is necessary to estimate the impact of increasing atmospheric N deposition on C sequestration in Moso bamboo plantations. The combined effects of anthropogenic management practices and N inputs should also be considered.
Fine roots were collected only in July when their annual biomass was the maximum. It is not known exactly when fine roots start to senesce in Moso bamboo plantations. The chemistry of the sampled roots may differ from that of naturally shed roots. To some degree, this difference might have affected the results of the present study.
References
Andersson M, Kjøller A, Struwe S (2004) Microbial enzyme activities in leaf litter, humus and mineral soil layers of European forests. Soil Biol Biochem 36:1527–1537
Austin AT, Ballaré CL (2010) Dual role of lignin in plant litter decomposition in terrestrial ecosystems. Proc Natl AcadSci USA 107:4618–4622
Axelsson G, Berg B (1988) Fixation of ammonia (15N) to scots pine needle litter in different stages of decomposition. Scand J For Res 3:273–279
Balesdent J, Besnard E, Arrouays D, Chenu C (1998) The dynamics of carbon in particle-size fractions of soil in a forest-cultivation sequence. Plant Soil 201:49–57
Berg B, McClaugherty C (2008) Plant litter: Decomposition, humus formation, carbon sequestration, 2nd edn. Springer, Heidelberg
Berg B, McClaugherty C, Santo AVD, Johnson D (2001) Humusbuildup in boreal forests: effects of litter fall and its Nconcentration. Can JFor Res 31:988–998
Bloomfield J, Vogt KA, Vogt DJ (1993) Decay rate and substrate quality of fine roots and foliage of two tropical tree species in the Luquillo experimental forest, Puerto Rico. Plant Soil 150:233–245
Brandt LA, King JY, Hobbie SE, Milchunas DG, Sinsabaugh RL (2010) The role of photodegradation in surface litter decomposition across a grassland ecosystem precipitation gradient. Ecosystems 13:1–17
Carreiro MM, Sinsabaugh RL, Repert DA, Parkhurst DF (2000) Microbial enzyme shifts explain litter decay responses to simulated nitrogen deposition. Ecology 81:2359–2365
Cui J, Zhou J, Peng Y, He Y, Yang H, Mao J, Zhang M, Wang Y, Wang S (2014) Atmospheric wet deposition of nitrogen and sulfur in the agroecosystem in developing and developed areas of southeastern China. Atmos Environ 89:102–108
Fang H, Mo J, Peng S, Li Z, Wang H (2007) Cumulative effects of nitrogen additions on litter decomposition in three tropical forests in southern China. Plant Soil 297:233–242
Fog K (1988) The effect of added nitrogen on the rate of decomposition of organic matter. Biol Rev 63:433–462
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai Z, Freney JR, Martinelli L, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889–892
Gosz JR, Likens GE, Bormann FH (1973) Nutrient release from decomposing leaf and branch litter in the Hubbard brook Forest, New Hampshire. Ecol Monogr 43:173–191
Hobbie SE (2008) Nitrogen effects on decomposition: a five-year experiment in eight temperate sites. Ecology 89:2633–2644
Hobbie SE, Eddy WC, Buyarski CR, Adair EC, Ogdahl ML, Weisenhorn P (2012) Response of decomposing litter and its microbial community to multiple forms of nitrogen enrichment. Ecol Monogr 82:389–405
Jackson RB, Mooney HA, Schulze ED (1997) A global budget for fine root biomass, surface area, and nutrient contents. Proc Natl AcadSci USA 94:7362–7366
Jia Y, Yu G, He N, Zhan X, Fang H, Sheng W, Zuo Y, Zhang D, Wang Q (2014) Spatial and decadal variations in inorganic nitrogen wet deposition in China induced by human activity. Sci Rep 4:3763
Jiang P, Wang H, Wu J, Xu Q, Zhou G (2009) Winter mulch increases soil CO2 efflux under Phyllostachys Praecox stands. J Soils Sediments 9:511–514
Keeler BL, Hobbie SE, Kellogg L (2009) Effects of long-term nitrogen additions on soil and litter microbial enzyme activity in eight forested and grassland sites–implications for litter and SOM decomposition. Ecosystems 12:1–15
Knorr M, Frey SD, Curtio PS (2005) Nitrogen additions and litter decomposition: a meta-analysis. Ecology 86:3252–3257
Li H, Lei Y (2010) Estimation and evaluation of forest biomass carbon storage in China. Chinese Forestry Press, Beijing
Li R, Werger MJA, de Kroon H, During HJ, Zhong Z (2000) Interactions between shoot age structure, nutrient availability and physiological integration in the giant bamboo Phyllostachys pubescens. Plant boil 2:437–446
Li Q, Song X, Gu H, Gao F (2016) Nitrogen deposition and management practices increase soil microbial biomass carbon but decrease diversity in Moso bamboo plantations. Sci Rep 6:28235
Lin C, Yang Y, Guo J, Chen G, Xie J (2011) Fine root decomposition of evergreen broadleaved and coniferous tree species in mid-subtropical China: dynamics of dry mass, nutrient and organic fractions. Plant Soil 338:311–327
Liu X, Zhang Y, Han W, Tang A, Shen J, Cui Z, Vitousek P, Erisman JW, Goulding K, Christie P, Fangmeier A, Zhang F (2013) Enhanced nitrogen deposition over China. Nature 494:459–462
Liu L, Zhang X, Wang S, Lu X, Ouyang X (2016) A review of spatial variation of inorganic nitrogen (N) wet deposition in China. PLoSONE11:e0146051
Manning P, Saunders M, Bardgett RD, Bonkowski M, Bradford MA, Ellis RJ, Kandeler E, Marhan S, Tscherko D (2008) Direct and indirect effects of nitrogen deposition on litter decomposition. Soil Biol Biochem 40:688–698
Manzoni S, Jackson RB, Trofymow JA, Porporato A (2008) The global stoichiometry of litter nitrogen mineralization. Science 321:684–686
Manzoni S, Trofymow JA, Jackson RB, Porporato A (2010) Stoichiometric controls on carbon, nitrogen, and phosphorus dynamics in decomposing litter. Ecol Monogr 80:89–106
Mary B, Recous S, Darwis D, Robin D (1996) Interactions between decomposition of plant residues and nitrogen cycling in soil. Plant Soil 181:71–82
Matson PA, McDowell WH, Townsend AR, Vitousek PM (1999) Theglobalization of N deposition: ecosystem consequences in tropical environments. Biogeochemistry 46:67–83
Mo J, Brown S, Xue J, Fang Y, Li Z, Li D, Dong S (2007) Response of nutrient dynamics of decomposing pine (Pinusmassoniana) needles to simulated N deposition in a disturbed and a rehabilitated forest in tropical China. Ecol Res 22:649–658
Nadelhoffer KJ, Raich JW (1992) Fine root production estimates and belowground carbon allocation in forest ecosystems. Ecology 73:1139–1147
Olson JS (1963) Energy storage and the balance of producers and decomposition in ecological system. Ecology 44:322–331
Ostertag R, Hobbie SE (1999) Early stages of root and leaf decomposition in Hawaiian forest: effect of nutrient availability. Oecologia 121:564–573
Parton W, Silver WL, Burke IC, Grassens L, Harmon ME, Currie WS, King JY, Adair EC, Brand LA, Hart SC (2007) Global-scale similarities in nitrogen release patterns during long-term decomposition. Science 315:361–364
Reay DS, Dentener F, Smith P, Grace J, Feely RA (2008) Global nitrogen deposition and carbon sinks. Nat Geosci 1:430–437
Silver WL, Miya RK (2001) Global patterns in root decomposition: comparisons of climate and litter quality effects. Oecologia 129:407–419
Sinsabaugh RL (2010) Phenol oxidase, peroxidase and organic matter dynamics of soil. Soil Biol Biochem 42:391–404
Song X, Zhou G, Jiang H, Yu S, Fu J, Li W, Wang W, Ma Z, Peng C (2011) Carbon sequestration by Chinese bamboo forests, and their ecological benefits: assessment of potential, problems, and future challenges. Environ Rev 19:418–428
Song X, Jiang H, Zhang Z, Zhou G, Zhang S, Peng C (2014) Interactive effects of elevated UV-B radiation and N deposition on Moso bamboo litter decomposition. Soil Biol Biochem 69:11–16
Song X, Zhou G, Gu H, Qi L (2015) Management practices amplify the effects of N deposition on leaf litter decomposition of the Moso bamboo forest. Plant Soil 395:391–400
Song X, Gu H, Wang M, Zhou G, Li Q (2016) Management practices regulate the response of Moso bamboo foliar stoichiometry to nitrogen deposition. Sci Rep 6:24107
Sun T, Mao Z, Han Y (2013) Slow decomposition of very fine roots and some factors controlling the process: a 4-year experiment in four temperate tree species. Plant Soil 372:445–458
Thirukkumaran CM, Parkinson D (2000) Microbial respiration, biomass, metabolic quotient and litter decomposition in a lodgepole pine forest floor amended with nitrogen and phosphorous fertilizers. Soil Biol Biochem 32:59–66
Tian H, Chen G, Lu C, Xu X, Ren W, Zhang B, Banger K, Tao B, Pan S, Liu M, Zhang C, Bruhwiler L, Wofsy S (2015) Global methane and nitrous oxide emissions from terrestrial ecosystems due to multiple environmental changes. Ecosyst Health Sustain 1:4
Tu L, Peng Y, Chen G, Hu H, Xiao Y, Hu T (2015) Direct and indirect effects of nitrogen additions on fine root decomposition in a subtropical bamboo forest. Plant Soil 389:273–288
Van Groenigen KJ, Six J, Hungate BA, de Graaff MA, van Breemen N, van Kessel C (2006) Element interactions limit soil carbon storage. Proc Natl AcadSci USA 103:6571–6574
Xie Y, Zhang S, Zhao X, Xiong Z, Xing G (2008) Seasonal variation patterns of NH4 +-N/NO3 −-N ratio and δ15 NH4 + value in rainwater in Yangtze river delta. Chinese. J Appl Ecol 19:2035–2041
Xu R (2013) Amelioration principles and technologies for acidified red soils. Science Press, Beijing
Xu X, Hirata E (2005) Decomposition patterns of leaf litter of seven common canopy species in a subtropical forest: N and P dynamics. Plant Soil 273:279–289
Xu Q, Liu L, Hong Y (1998) Analysis on enzyme activities of high-yield and low-yield Phyllostachys pubescens stands. J Bamboo Res 17:37–40
Xu Q, Jiang P, Xu Z (2008) Soil microbial functional diversity under intensively managed bamboo plantations in southern China. J Soils Sediments 8:177–183
Yang Y, Chen G, Guo J, Lin P (2004) Decomposition dynamic of fine roots in a mixed forest of Cunninghamialanceolataand Tsoongiodendronodorumin midsubtropics. Ann ForSci61:65–72
Zhang D, Hui D, Luo Y, Zhou G (2008) Rates of litter decomposition in terrestrial ecosystems: global patterns and controlling factors. J Plant Ecol 1:85–93
Zhou B, Fu M (2004) Review on bamboo’s underground rhizome-root system research. For Res 17:533–540
Zhou B, Fu M (2008) Fine root production and turnover of Phyllostachys pubescens stands in Miaoshanwu nature reserve. Acta Agri UnivJiangxiensis 2:239–245
Zhou G, Jiang P (2004) Density, storage and spatial distribution of carbon in Phyllostachy pubescens forest. SciSilvaeSinicae 40:20–24
Zhou G, Xu J, Jiang P (2006) Effect of management practices on seasonal dynamics of organic carbon in soils under bamboo plantations. Pedosphere 16:525–531
Zhou G, Jiang P, Xu Q (2010) Carbon fixing and transition in the ecosystem of bamboo stands. Science Press, Beijing
Acknowledgments
This study was funded by the “948” Project of the State Forestry Bureau of China (Grant No. 2013-4-55), the National Natural Science Foundation of China (Grant No. 31270517, 31470529), and the Pandeng Project for Young & Middle-aged Discipline Leaders of Zhejiang Province (Grant No. pd2013234).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no competing financial interests. All of the authors have contributed substantially to the manuscript and approved the final submission. All previous work cited in the manuscript has been fully acknowledged.
This study did not involve human participants, specimens or tissue samples, or vertebrate animals, embryos or tissues.
This manuscript has not been submitted elsewhere in whole or in part and is not currently under consideration for publication in any other journal before a decision has been made by Plant and Soil.
Additional information
Responsible Editor: Alfonso Escudero.
Rights and permissions
About this article
Cite this article
Song, X., Li, Q. & Gu, H. Effect of nitrogen deposition and management practices on fine root decomposition in Moso bamboo plantations. Plant Soil 410, 207–215 (2017). https://doi.org/10.1007/s11104-016-2997-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-016-2997-8