Abstract
Liver cancer is the sixth most common cancer and the fourth leading cause of cancer deaths in the world. The most common type of liver cancers is hepatocellular carcinoma (HCC). Autophagy is the cellular digestion of harmful components by sequestering the waste products into autophagosomes followed by lysosomal degradation for the maintenance of cellular homeostasis. The impairment of autophagy is highly associated with the development and progression of HCC although autophagy may be involved in tumour-suppressing cellular events. In regards to its protecting role, autophagy also shelters the cells from anoikis- a programmed cell death in anchorage-dependent cells detached from the surrounding extracellular matrix which facilitates metastasis in HCC. Liver cancer stem cells (LCSCs) have the ability for self-renewal and differentiation and are associated with the development and progression of HCC by regulating stemness, resistance and angiogenesis. Interestingly, autophagy is also known to regulate normal stem cells by promoting cellular survival and differentiation and maintaining cellular homeostasis. In this review, we discuss the basal autophagic mechanisms and double-faceted roles of autophagy as both tumour suppressor and tumour promoter in HCC, as well as its association with and contribution to self-renewal and differentiation of LCSCs.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Autophagy is an intracellular self-digestive mechanism that degrades damaged cellular materials and wastes through lysosomal degradation [1]. It is essential for cell survival and is highly conserved across various species [2]. In Saccharomyces cerevisiae, the autophagic machinery was first identified and was then described in other mammalian models [3, 4] This process involves a variety of autophagy-related (ATG) proteins and require diverse signalling pathways as it starts off with the initiation phase, elongation of the phagophore and maturation of the vesicle. The matured autophagosome then fuses with the lysosome and subsequent degradation occurs [5]. Over the years, autophagy has been linked to the pathophysiology of various diseases including myopathies, ageing, cancer and metabolic diseases such as diabetes, liver and heart diseases [6]. In fact, the dysregulation of autophagy had been observed in various cancers including breast, ovarian, and liver cancers [7].
Liver cancer is the sixth most common cancer and the fourth leading cause of cancer-related deaths in the world. The incidence and mortality rates are approximately 2 to 3 times higher in males than in females [8]. Hepatocellular carcinoma (HCC) is the most common form of liver malignancy which accounts for 85–90% of the total cases [9]. The aetiology of HCC is linked to dietary, lifestyle, environmental factors and genetic predispositions [10]. Chronic hepatitis B virus (HBV) and hepatitis C virus (HCV) infection, alcohol consumption, and non-alcoholic fatty liver remain the major risk factors for HCC. Approximately 75% of HCC cases are related to chronic HBV and HCV infections [11]. HBV can induce activation of oncogenes and genetic instability [12] while HCV damages liver cells by causing hepatic necrosis, accumulation of oxidative stress and steatosis prior to the development of HCC [13]. Other risk factors of HCC include the exposure of aflatoxin B1, primary biliary cirrhosis, genetically associated predispositions including hereditary hemochromatosis, alpha1-antitrypsin deficiency, glycogen storage disease type-1, autoimmune hepatitis and type-2 diabetes [14].
HCC commonly emerges from chronic liver cirrhosis which is a progressive, fibrosing and nodular condition that disrupts the structure and functions of the liver [15]. Typically, liver cirrhosis develops after persistent chronic inflammation and could contribute to carcinogenesis by inducing chromosomal instability and molecular alterations including disruption of cell cycle checkpoints and activation of oncogenic pathways [14]. HCC is usually detected either by active periodical screening, or due to clinical presentations such as abdominal pain, and/or liver decompensation [16]. This disease is often diagnosed at late stages which significantly affects the treatment efficacies [17]. Treatment options include surgical resection, tumour ablation, liver transplantation, percutaneous treatments and chemoembolization [18, 19]. For HCC detected in early stages, 5-year survival of patients after resection and transplantation can achieve 60–70%. Patients with intermediate-stage HCC have a 3-year survival rate of 50%, while advanced stage have a 3-year survival rate of 8%. Patients who are diagnosed in the late stages have a very poor prognosis and can have less than 6 months life expectancy [17, 18].
HCC tumour suppression and promotion are modulated by several regulatory components and pathways, of which some of them confers both fates depending on the tumour niche and cancer progression. The double-sided role of autophagy as a tumour suppressor or promoter in HCC has been previously reported and is known to play a part in cancer progression [20]. Under normal circumstances, autophagy is responsible for maintaining homeostasis by degrading abnormal proteins and damaged organelles, thus preventing abnormal cell growth which eventually leads to tumour development [21]. However, this autophagic mechanism seems to be “hijacked” by cancer cells to recycle and reuse the nutrients produced by the waste metabolic products to support cancer growth [22]. The energy generated from this process is then used to strive and counteract against environmental stress such as hypoxia, oxidative stress and energy deprivation which consequently contributes to the survival and proliferation of liver cancer cells. In addition, autophagy also contributes to the promotion of drug resistance [23]. Signalling pathways involving proto-oncogenes such as PI3K, AKT and mTOR, and tumour suppressors such as BECN1 and TP53, as well as non-coding RNAs are involved in autophagy regulation. These oncogenes can either promote or suppress autophagy, which eventually contributes to HCC or otherwise [24].
In recent years, a comprehensive understanding of cancer as a disease and the underlying mechanisms driving cancer progression has been developed. Intriguingly, cancer stem cells (CSCs) are found to be one of the key drivers of tumour progression and aggressiveness which leads to failure of treatments [25]. These are small subpopulations of cells found within the tumours in various cancers such as leukaemia, breast cancer, glioblastoma, prostate, lung, gastric and ovarian cancer [26]. CSCs possess unique characteristics including self-renewal, differentiation and chemoresistance [27]. They are enriched with unique markers such as CD133, CD90, CD13, CD44, and EpCAM [27, 28]. The number of CSCs present in solid tumours varies largely and its role in cancer regulation may vary depending on the cancer type and disease staging [29]. CSCs are also readily found in HCC tumours and have shown to contribute to the metastasis, tumour recurrence and drug-resistant nature of HCC [27, 30].
In liver cancer stem cells (LCSCs), autophagy was found to be involved in the stem cell regulation for cell maintenance, self-renewal, and stem cell differentiation [31]. The interplay of autophagy and the different signalling pathways involved in stem cell regulation provides a clearer picture of how it can influence HCC development and progression. Thus, understanding the role of autophagy in LCSCs in HCC progression and developing treatment targeting CSCs have emerged as an important effort. In this review, we aim to discuss the role of autophagy and CSCs in HCC progression and aggressiveness. We also attempt to provide insights on the interplay between autophagy and LCSCs in promoting HCC.
Role of autophagy in hepatocellular carcinoma
Basic mechanism and functions of autophagy
Autophagy was first described and coined in 1963 by biochemist, Christian de Duve following the observation of the degradation of the mitochondria and other intracellular structures in rat liver shortly after the injection of glucagon [32]. The mechanisms of protein degradation in the cells were soon delineated after the discovery of lysosomes from the cell fractionation of rat liver homogenate which contained acid phosphatase and other hydrolytic enzymes [33]. The autophagosome, a double membrane structure that contains organelles and cytoplasmic components, was formed in the early stages of autophagy. Ensuing this process, the structure was later observed and termed as autophagolysosome which embodies various proteins and organelles for degradation by hydrolytic enzymes [34].
Autophagy possesses sequential pathways which ultimately leads to the degradation of damaged proteins and cytoplasmic constituents to maintain cellular homeostasis and survival [35]. It is also activated in response to physiological stress in cells such as oxidative stress and nutrient starvation [36]. In mammals, autophagy induction is primarily regulated by class III phosphatidylinositol 3-kinase (PI3K) complexes such as vacuolar protein sorting 34 (VPS34) and ATG6/Beclin-1 complex [37], and mammalian target of rapamycin complex (mTORC1) (Fig. 1). In the event of a growth-favourable environment, mTORC1 phosphorylates and represses unc-51 like kinase 1 (ULK1), mammalian autophagy-related protein 13 (ATG13), and focal adhesion kinase family-interacting protein of 200 kDa (FIP200) which form a kinase complex that is required to activate the autophagy pathway [38]. When the cells undergo nutrient deprivation, ULK1 alternatively dissociates with 5` adenosine monophosphate-activated protein kinase (AMPK) to induce autophagy [39]. ATG13 binds to ULK1/2 and FIP200 and subsequently, ULK1/2 phosphorylates FIP200 along with the ULK1-ATG13-FIP200 complex. This event activates a cascade, whereby the PI3K complexes consisting of Beclin-1, ATG14L, AMBRA1, VPS34 and p150 are activated [40]. Following that, VPS34 facilitates the formation of phosphatidylinositol triphosphate (PI3P) to recruit other complexes for subsequent phagophore elongation and the formation of the autophagosome [41].
Autophagic processes in a cell. Stress signals generated from nutrient or growth factor deprivation and mTORC1 inhibition lead to the activation of (1) ULK complexes including ULK1/2, ATG13 and FIP200. ULK phosphorylates Beclin-1, activating the PI3K complexes consisting of Beclin-1, ATG14L, VPS34, AMBRA and P150 followed by phagophore formation. (2) The source of membrane used to form the phagophore assembled from the mitochondria, Golgi complex, recycling endosomes and plasma membrane. (3) Two ubiquitin-like conjugation systems: ATG12-5-16L1 complex and the LC3 conjugation system are involved in the elongation of the phagophore. The catalysation of LC3 to LC3-II and its conjugation to phosphatidylethanolamine (PE) forms the LC3 conjugation system. (4) Both are then incorporated into the phagophore for elongation. (5) LC3-II exists as a site for binding of adaptor proteins such as SQSTM1/p62 and NIX with respective cargos allowing cargo sequestration. (6) The sealing of the phagophore marks the end of autophagosome formation and it eventually fuses with the lysosome which allows the release of acid proteases. (7) The lysosome is completely fused with the autophagosome to form the autolysosome. (8) Proteins or other cellular components are degraded, and the nutrients released from the hydrolysed waste products are recycled to generate additional energy for the maintenance of cell viability and proliferation including in cancer cells
Autophagy is initiated from a phagophore formation that originates and assembles from the plasma membrane, Golgi complex, endosomes and endoplasmic reticulum (ER) [42] (Fig. 1). As the phagophore elongates, there are two types of ubiquitylation-like conjugation systems that are involved. These systems are the ATG12-ATG5-ATG16L1 and ATG8 system. ATG12 is first conjugated to ATG5 by E1-like activating enzyme ATG7 and E2-like conjugating enzyme ATG10 followed by the conjugation of ATG16L1. The conjugated ATG12-ATG5-ATG16L1 acts as an E3-like enzyme which plays a role in the second conjugation system by promoting lipidation of ATG8 for cargo recognition and autophagosome formation [43]. ATG12-ATG5-ATG16L1 is also involved in membrane tethering and the aggregation of vesicles for phagophore elongation [44]. Microtubule-associated protein 1A/1B-light chain 3 (LC3) are distributed throughout the mammalian cells and they are the ortholog of ATG8 in yeast [45]. In the second conjugation system, the LC3 protein is cleaved by cysteine protease ATG4 to form the cytosolic LC3-I protein. LC3-I is then conjugated to phosphatidylethanolamine (PE) by E1-like enzyme ATG7 and E2-like enzyme ATG3 to form lipidated LC3-II [45]. LC3-II then binds to the phagophore membrane to form an autophagosome. ATG18/WIPI proteins are also involved in the elongation of the phagophore by recognizing and binding to PI3P and assembling the two conjugation systems [46]. Transmembrane protein ATG9, which is accumulated at the trans-Golgi and late endosomes, also facilitates membrane transfer and phagophore elongation [47].
Cargo recognition is enabled by autophagy adaptor proteins and cargo receptors such as sequestosome-1/ubiquitin-binding protein p62 (SQSTM1/p62), the receptor for mitochondrial protein (NIX), optineurin (OPTN), NDP52/CALCOCO2 and NBR1. These cargo receptors allow binding to specific target cargos and ubiquitination. The cargo receptors then interact with receptor protein LC3-II on the autophagosome membrane to direct the cargo delivery to the autophagosome [48, 49]. After the cargoes are delivered, the autophagosome is elongated until it is sealed. The lysosome then fuses with the autophagosome to mature into an autolysosome. In this process, the lysosome-associated membrane protein 2 (LAMP2) and LC3-II facilitate the fusion by interacting with Golgi reassembly-stacking protein 55 (GRASP55) [50]. The bidirectional movement of lysosomes and autophagosomes are mediated by motor protein dynein and kinesin [51]. The degraded metabolites, including monosaccharides and amino acids, are then released out through the lysosomal efflux transporters on the lysosomal membrane [52]. The completion of autophagy triggers lysosomal reformation. This marks the end of the autophagic process as vesicles are extruded and proto-lysosomal tubules extend from the autolysosome to mature into fully functional lysosomes [53] (Fig. 1).
As previously mentioned, the autophagosome biogenesis and membrane nucleation is initiated by a plethora of signalling pathways which ultimately involves the modulation of AMPK and mTORC1 activity in initiating the autophagic process as a whole [38,39,40,41,42]. The autolysosome however, is formed from either the direct fusion of the autophagosome with a lysosome or the fusion of the intermediate amphisome (formed when an endosome fuses with the autophagosome) with a lysosome [54]. The result of this process would be the degradation of cargos within the autolysosome [55].
Autophagy in the liver
The basal autophagy machinery plays an important role in maintaining normal liver homeostasis. The liver is responsible for several functions from the metabolism of carbohydrate, protein and fat to the secretion of bile [56]. In order to compensate for the high energy demand, hepatocytes rely on autophagy for energy production. A disruption in normal function can alter the amino acid levels in the blood, resulting in metabolic disorders such as fatty liver, impaired liver function and the development of diseases such as fibrosis, cirrhosis and HCC [57]. Autophagy plays important roles in the physiological processes in the normal liver [58]. Autophagy in bulk provides a supply of amino acids for the sustenance of protein synthesis [59]. Further starvation or shortage of nutrients would lead to the breakdown of lipids and glycogen. Lipid droplet turnover occurs through autophagy is termed lipophagy. It is responsible in supplying free fatty acids for energy and ketone production [60]. In glycophagy, autophagosomes envelop glycogen granules and is subsequently catabolized into glucose by α-glycosidase present in lysosomes [61]. In addition, autophagy is associated with the catabolism of inclusion bodies including Mallory-Denk bodies (MDBs) which are found in the cytoplasm of hepatocytes [62, 63]. MDBs are hyaline inclusions that are found in various liver diseases such as fatty liver and HCC as the result of a malfunction in protein quality control. Protein misfolding, p62 expression and autophagy are found to contribute to MDBs formation [63].
Cumulative studies suggest that autophagy has double-faceted roles in HCC [64], these will be discussed in detail in the following sections.
Autophagy in HCC suppression and prevention
Autophagy exerts tumour-suppressing activity by removing damaged mitochondria, abnormal proteins, protein aggregates and oncogenic proteins [21, 65] (Fig. 2). A study has shown that autophagy plays an important role in regulating lipid content, especially in the liver where the storage of triglycerides takes place. During nutrient starvation, the autophagy mechanism of lipid, known as lipophagy, is required to break down the fat storage to supply fatty acids as an alternate energy source [60]. Autophagy impairment can lead to the shortage of fatty acids source as well as slowing down the amino acid recovery which is essential for the synthesis of nucleic acid and protein as well as ATP generation to support the cellular metabolism, thus causing tissue damage and death in normal liver cells [21]. Chronic inflammation of the liver, which leads to oxidative stress, nitrosative stress and aldehydes through the generation of reactive oxygen species (ROS) and reactive nitrogen species (RNS), is associated with more than 90% of HCC cases. These generated aldehydes could induce mutations in the genomic DNA [66]. Oxidative stress is the key driver for both hepatocarcinogenesis and the progression of HCC [66, 67]. In HCC with persistent inflammation, several studies suggested that inhibiting the autophagy in these cells could worsen the inflammation in the liver and further promote HCC, which highlights the role of autophagy in tumour suppression (Fig. 2). One of the mechanisms in which autophagy functions as a tumour suppressor in response to oxidative stress is through mitophagy- a selective degradation of mitochondria through autophagy [67]. Parkin is a cytosolic E3 ubiquitin ligase that is associated with the maintenance of mitochondria. Parkin regulates mitochondrial maintenance by translocating to the location of the dysfunctional mitochondria and facilitating ubiquitylation [68]. NIX proteins are then recruited to the mitochondria to initiate mitophagy [69]. A study has demonstrated that PARK2 (encodes for Parkin) deletion in mice resulted in a high proliferation rate of hepatocytes and the development of hepatic tumours. The deficiency of parkin also caused the inhibition of caspase activity which prevented apoptosis in hepatocytes [70]. This study highlights the importance of Parkin-induced mitophagy in the suppression of HCC.
Role of autophagy in hepatocellular carcinoma (HCC). Autophagy acts as a tumour suppressor in the early stages of cancer by removing damaged mitochondria and abnormal proteins, regulating lipid metabolism in liver cells, and repressing inflammation (represented by the red line). However, once cancer has established, autophagy turns into the tumour promoter by regulating the metabolism and maintaining oxygen homeostasis to support the survival of cancer cells. Autophagy also induces anoikis resistance and epithelial-mesenchymal transition (EMT) in promoting the disease progression
Several other studies have also shown that the knockout or deletion of various autophagic genes could induce HCC, highlighting the role of autophagy in preventing cancer. ATG5 and ATG7 are important proteins that function in the ubiquitin conjugation systems in the process of autophagy [43]. A report showed that mice with ATG5 and ATG7 deletion developed multiple liver adenomas as the result of impaired autophagy with the accumulation of high-level oxidative stress and DNA damages [71]. In another study, allelic deletion of BECN1 and ATG5 also led to deficient autophagy in the cells, resulting in the accumulation of p62 which drove DNA damage, genomic instability and tumour development [72]. The accumulation of p62 and ubiquitinated proteins are characteristics of impaired autophagy. Autophagy deficiency resulted in the inhibition of nuclear factor erythroid 2-related factor 2-kelch-like ECH-associated protein 1 (Nrf2-Keap1) system due to the p62 accumulation as reported by Inami and colleagues [73]. Nrf2 is a transcription factor that binds to Keap1 which is a ubiquitin ligase adaptor [74]. Under normal circumstances, Nrf2 is degraded through protein catabolism in the ubiquitin–proteasome pathway. In the event of autophagy impairment, the accumulation of p62 resulted in the inhibition of the Nrf2 and Keap1 interaction, thus stabilizing and activating Nrf2 which eventually led to the development of hepatoma [73].
Autophagy in HCC progression
On the other hand, autophagy may also promote tumour growth by allowing the cancer cells to strive and adapt the stressful conditions such as hypoxia, nutrient starvation and metabolic burden (Fig. 2). Tumours in HCC patients can undergo hypoxia and oxidative stress due to the lack of oxygen and the increase of ROS, which subsequently induced autophagy in hepatocytes [75]. The hypoxia-inducible factors (HIFs) are involved in promoting glycolysis for adaptation of HCC under hypoxic stress through the regulation of various glycolytic genes [76]. HIF1α belongs to the family of HIFs and it is vital for oxygen homeostasis. HIFα is also known to initiate hypoxia-induced autophagy. A study has shown that HIF1α upregulated pro-apoptotic Bcl-2/adenovirus E1B 19-kDa interacting protein 3 (BNIP3) and Bcl-2/adenovirus E1B 19-kDa interacting protein 3-like (BNIP3L/NIX) proteins which activated autophagy for cell survival [77, 78]. In normal circumstances, Bcl-2, a cell death regulating protein, inhibited the progression of autophagy by binding to Beclin-1 via the BH3 domain to form the Bcl-2-Beclin-1 complex [79]. However, the upregulation of BNIP3 and BNIP3L by HIF1α-induced autophagy could dissociate the Bcl-2-Beclin-1 complex, and hence, supporting autophagy processes by facilitating the assembly of autophagosome structures [77]. On top of that, both BNIP3 and HIF1α have also been found to play an essential role in promoting tumorigenicity and invasion in HCC. The silencing of BNIP3 could decrease the level of LC3-II which is the autophagy protein in HCC [80].
As a result of the adaptation of HCC to hypoxia, the aberrant expression of either HIF isoforms, HIF1 or HIF2 are frequently found in tumour tissues [81]. HIF2α, an isoform of HIF1α, was also found to be involved in HCC cell survival. A study by Mendrad et al. demonstrated that the downregulation of HIF2α in HCC spheroids resulted in an upregulation of HIF1α, anti-apoptotic Bcl-XL (Bcl-2 protein), and BNIP3 which upregulates autophagy and prevents apoptotic events [82]. In this instance, the mechanistic action of BNIP3 in autophagy induction is similar to the dissociation of Bcl-2-Beclin-1 complex in HIF1α-induced autophagy as previously mentioned [77, 82]. BNIP3 acts by releasing Beclin-1 through the binding to Beclin-1-Bcl-XL, localized in the ER, which induces the formation of the autophagosome [83]. In contrast, the localization of Bcl-XL in the mitochondria leads to apoptotic inhibitory effects [84]. Besides that, it was reported that the knockdown of HIF2α resulted in an upregulation of HIF1α and vice versa, which suggests that both HIF1α and HIF2α are important in HCC progression [82]. A knockdown of HIF2α diminished cell invasion, metastasis and angiogenesis in HCC cells. In addition, the cyclooxygenase-2/prostaglandin E(2) (COX-2/PGE2) axis is shown to be responsible for the nuclear translocation of HIF2α through MAPK signalling, activates TGFα/EFGR and regulates HIF2α, thereby promoting HCC progression [85].
High expression of autophagic proteins is consistently observed in HCC tissues. From a cohort study of 156 HCC patients, high LC3-II expression was observed in the tissues and was associated with vascular invasion, lymph node metastasis and tumour, node, metastasis (TNM) staging [86]. Increased autophagy events were also observed in advanced stage HCC and were associated with poor prognosis [86, 87]. The role of autophagy in both tumour suppression and promotion is summarised in Fig. 2.
Autophagy in HCC metastasis
Metastasis is the development of secondary malignancy away from the primary cancer site. From the primary tumour, the cancer cells invade into the lymphatic or circulatory system as circulating tumour cells (CTCs), infiltrate and proliferate in the distal organs [88, 89]. Anoikis are a form of apoptotic cell death that occurs when anchorage-dependent cells detach from the extracellular matrix [90]. Both epithelial-mesenchymal transition (EMT) and anchorage-independent growth abilities contribute to anoikis resistance which mediates metastasis [91]. Anoikis resistance is regulated by a plethora of complex signalling pathways including apoptotic-related pathways and pathways involving oncogenic Ras, PI3K/Akt or integrin-linked kinase [92]. Cancer cells that acquire resistance would enhance invasiveness and metastasis.
Owing to the tumour-promoting characteristic of autophagy, several studies have suggested that autophagy can contribute to the survival of tumour cells and support tumour invasiveness [93]. In a study conducted by Peng et al., autophagy was inhibited through the silencing of BECN1 and ATG5 genes in HCC cells using a lentivirus system. The autophagy inhibition in HCC cell lines led to enhanced anoikis in vitro. In the histopathological examination, the inhibition of autophagy significantly reduced pulmonary metastases incidence, anoikis resistance and colonization of HCC in vivo [94]. Furthermore, the upregulation of BNIP3 in detached HCC cells via the ERK/HIF1α pathway also contributed to anoikis resistance and metastasis through autophagy induction [80].
Transforming growth factor beta (TGF-β) is a growth factor that is involved in generating multiple cellular responses affecting apoptosis, cell proliferation and differentiation [95]. TGF-β is widely known to be pro-apoptotic in many different cell types, and apoptosis is induced through the upregulation of the cyclin-dependent kinase inhibitors and the suppression of c-Myc, a transcriptional activator of cell proliferation [96]. In contrast to the pro-apoptotic effects of TGF-β, studies have shown that TGF-β can also promote cell growth and metastasis in late stages of cancer [97]. TGF-β is now known as an important component in EMT with the interplay of autophagy [98, 99]. EMT is a cellular phenomenon whereby epithelial cells change their characteristics to acquire mesenchymal-like characteristics involving cytoskeleton reorganization, loss of cell–cell junctions and the expression of mesenchymal markers [100]. EMT enhances tumour cell metastatic capacity of epithelial cells and is involved in cancer cell invasion, metastasis and HCC progression, and this can be affected by autophagy (Fig. 2) [98, 101]. Increasing studies have suggested that autophagy can induce EMT during starvation by promoting the cell invasion through TGF-β/Smad3 signalling [98, 100]. In a study conducted by Li and colleagues [99], autophagy downregulated the expression of epithelial markers E-cadherin and CK18 and upregulated the mesenchymal markers fibronectin and MMP-9, suggesting the induction of EMT by autophagy. The siRNA targeting ATG3 and ATG7 genes inhibited the autophagy in HepG2 and BEL7402 cells and successfully reversed invasion in HCC [99]. The TGF-β1 expression and Smad3 phosphorylation were also found to be suppressed following the autophagy gene-silencing which suggests the involvement of the TGF-β/Smad3 pathway in autophagy-mediated EMT [99]. It is well established that autophagy works as an EMT promoter, however, the repression of autophagy can also promote cell migration and invasion through EMT in cancer cells [102]. DNA-damage regulated autophagy modulator 1 (DRAM1) is a transmembrane protein and a target of p53-mediated autophagy. DRAM1 was found to mediate the autophagy-EMT pathway. When DRAM1 was knocked down, an upregulation of EMT protein E-cadherin and a downregulation of Vimentin, a filament protein expressed in mesenchymal cells, were observed [103]. Interestingly, the downregulation of DRAM1 also significantly inhibited cell migration, invasion and metastasis through the autophagy-EMT pathway [103]. In another HCC model, the induction of autophagy promoted EMT as a result of an upregulation of TGF-β through the increased activation of cAMP response element-binding protein (CREB) and cAMP/PKA/CREB signalling [104]. On the other hand, the inhibition of autophagy could also promote EMT in cancer cells. As demonstrated by Wang and colleagues, impeded autophagy in RAS-mutated cancer cells promoted EMT with the upregulation of transcription factors involved in EMT partially through the activation of the NFKB/NF-κB pathway. Furthermore, the knockdown of autophagy synergized with RAS activation to induce EMT [105]. Another example would be in the ovarian cancer cells in which EMT is promoted through the ROS/heme oxygenase-1 (HO-1) pathway due to autophagic inhibition and subsequently enhanced invasion and migration [106].
Autophagy in HCC treatment resistance
Cancer cells thrive and survive anticancer therapy by developing drug resistance through different mechanisms, which includes autophagy [24]. The administration of therapeutic drugs has been known to induce and upregulate autophagy in HCC. Cisplatin is a platinum-based anti-cancer drug that is used as a chemotherapeutic drug. The cytotoxicity of the drug owes mainly to its ability to react with the DNA to form DNA adducts at purine bases and resulting DNA–protein or DNA-DNA crosslinks [107, 108]. The resulting ER stress from cisplatin treatment triggers an unfolded protein response (UPR) in the attempts of overcoming ER stress and promoting cell viability. In HCC cells, UPR was found to cytoprotective properties against apoptosis induced by cisplatin. In the same study, results have shown that UPR with heat shock protein 27 (HSP27) promoted cell survival through autophagic regulation and activation [109]. Oxaliplatin is another platinum-based drug with a similar chemotherapeutic mechanism of action to cisplatin [108]. For example, oxaliplatin-treated HepG2 cells exhibited upregulated LC3 lipidation and puncta formation which are key processes in autophagy [110]. This response confers resistance and promotes cell survival [110]. Similarly, DNA topoisomerase II inhibitor [111] doxorubicin treatment was also able to induce autophagy in HCC [112]. 5-Fluorouracil (5-FU) is an inhibitor of thymidylate synthase which is essential for DNA biosynthesis and is used in HCC treatment [113]. p53 is shown to modulate autophagy induction and promoting cell survival with chemoresistance in nutrient-deprived cells. This study suggests that p53 plays a part in the mechanism conferring drug resistance [114] (Table 1).
Autophagy can be targeted to restore the chemosensitivity to various chemotherapeutic drugs in HCC. For example, siRNA-silenced ATG7 and chloroquine treatment increased the susceptibility of HCC to oxaliplatin [110]. In another study, miR-101 that inhibited autophagy by targeting RAB5A, STMN1 and ATG4D which are important genes in regulating autophagosome formation, increased the sensitivity of HepG2 to cisplatin [122]. Similarly, miR-233 that inhibited autophagy by targeting FOXO3a, which is a transcription factor associated with autophagy regulation, also increased the susceptibility of HCC cells to doxorubicin [112]. This study suggests that autophagy induced by doxorubicin treatment plays a part in conferring drug resistance [112].
Transarterial embolization (TAE) and transarterial chemoembolization (TACE) remain a standard therapeutic option for unresectable HCC [123]. TAE involves the embolization of the hepatic artery with the absence of chemotherapy drug usage, while TACE involves the injection of chemotherapeutic drug preceding TAE [124]. Both of the vascular embolization techniques aim to deprive cancer cells of nutrients and oxygen, resulting in ischemic necrosis and halting cell growth [125]. HCC cells subjected to severe ischemia reduced sensitivity to the cell cycle chemotherapy drug doxorubicin and mitomycin C, which is correlated to induced quiescence in surviving cells [126]. In the same study, cell survival under severe ischemia was mediated by the activation of autophagy as a significant upregulation of mRNA expression of autophagic-related genes were observed [126].
Early growth response 1 (Egr-1) protein is a zinc finger transcription factor that is induced when the cells undergo environmental stress such as oxidative stress [121]. Egr-1 is associated with the promotion of drug resistance in various cancers [121,127]. In HCC, Egr-1 is highly expressed and is known to regulate hypoxia-induced autophagy by upregulating LC3 conversion and Beclin-1. The regulation of autophagy by Egr-1 is conferred through its binding with the promoter region of LC3 [121]. Egr-1 expression induced autophagy and subsequently resulted in chemoresistance of cisplatin and epirubicin in HCC [121]. Interestingly, its expression is also involved in conferring radio-resistance in HCC through the regulation of autophagy [128]. Upon ionizing radiation (IR) on HCC cells, increased expression of Egr-1 was observed, and autophagy was induced by transcriptional activation of ATG4B [128]. ATG4B is a cysteine protease that is involved in LC3 lipidation which is an indispensable element in autophagy [129]. The inhibition of Egr-1 was shown to decrease the IR-induced autophagy and increase the radiosensitivity in HCC [128].
Sorafenib is one of the commonly-used drug and a first-line systemic therapy used for treating HCC [130]. It is a multikinase inhibitor that functions by inhibiting vascular endothelial growth factor receptors (VEGFR), platelet-derived growth factor receptor β (PDGFR-β) and serine-threonine kinase (Raf kinases) [115]. However, sorafenib resistance tends to develop within 6 months of administration in HCC [131]. HCC develops resistance through a variety of mechanism. Phase II metabolic enzyme glutathione transferase Mu 1 (GSTM1) is thought to be involved in the drug resistance mechanism in HCC. GSTM1 acts by hydrolysing the chemo-drug and may be involved in mediating drug resistance to both sorafenib and oxaliplatin through autophagic activation [116]. The activation of Akt is thought to mediate sorafenib resistance as its inhibition reversed the resistance as a result of shifting from cytoprotective autophagy to autophagic cell death [116]. Sorafenib administration causes an upregulation of HIF2α which contributes to resistance through the activation of the TGFα/EFGR pathway [118]. Intriguingly, another study revealed that the induced hypoxia from sorafenib treatment confers resistance to the drug via HIF1α and NF-κB activation [119]. Increased level of LC3-II and ATG5 which facilitate autophagosomal formation during autophagy has been observed in sorafenib treatment in HCC. Sorafenib induced ER stress which then activated the autophagic process [132]. Targeting autophagy by autophagy inhibitor, chloroquine in combination with sorafenib has successfully reduced the tumour volume and inhibited the tumour growth in HCC using mouse models [132].
Role of cancer stem cells in hepatocellular carcinoma
Liver cancer stem cell surface markers
Cancer stem cells (CSCs) are a subpopulation of cells in the tumour that have the ability to self-renew and differentiate into heterogeneous lineages. They reside in a microenvironment that comprises a variety of cells that support their survival [133]. In HCC, liver CSCs (LCSCs) have been identified as precursor cells for cancer progression [134] however, the origins of these LCSCs remains unclear. Yamashita and Wang suggested that LCSCs may originate from non-CSCs such as hepatocyte and cholangiocyte through the process of “de-differentiation” [135]. The expression of distinguished markers on the cell surface can define the heterogeneity of LCSCs [28]. Cell surface markers epithelial cell adhesion molecules (EpCAM), CD133, CD44, CD90, CD13, OV-6 and K19 are among those that are commonly expressed on CSCs [27, 28]. Surface markers with their phenotype and their clinical characteristics are summarised in Table 2.
EpCAM is a transmembrane glycoprotein on the cell surface that has been extensively studied as a marker for LCSCs. Yamashita et al. demonstrated that EpCAM + alpha-fetoprotein (AFP) + hepatocellular carcinoma subtype showed stemness properties, activation of stem cell-associated genes, high metastasis occurrence and associated with poor prognosis [136]. The expression of EpCAM in LCSCs also correlated with the tumour growth and cell invasiveness in HCC [137]. CD133 has been reported as a CSC marker for the brain, prostate, colon and liver. Ma et al. demonstrated that Huh7 cell lines that are CD133 + showed a higher proliferative rate and tumorigenicity ability in comparison to CD133- cells. The tumours arisen from CD133 + cells were larger in size and greater in colony number [138]. Another LCSC marker, CD44 is a glycoprotein that is involved in cell–cell adhesion, migration and invasion of liver cancer cells [139]. CD44 + cells co-expressing CD133 and CD90 were more aggressive than cells presenting either CD133 or CD90 alone [139, 140]. CD90, a small 25–37 kDa glycosylphosphatidylinositol (GPI)-anchored conserved cell surface protein, has also been identified in CSCs. Expression of CD90 positively correlated with tumorigenicity capabilities and metastatic potentials of several HCC cell lines including MHCC97L and MHCC97H [140]. CD13 or aminopeptidase N is a membranous glycoprotein that is associated with cancer progression and drug resistance [141]. CD13 are expressed in slow-growing or semi-quiescent LCSCs which contributes to tumour formation in HCC. From the limiting dilution analysis of Huh7, CD133 + CD13 + cells were able to form tumours from 100 cells while CD133 + CD13- cells were able to form tumours from 1000 cells. Interestingly, no tumour was formed from CD133- CD13- cells even up to 5,000 cells which highlighted the importance of CD13 and CD133 expressing LCSCs in HCC [141]. In another study, OV6 + cell could be isolated from HCC and the OV6 + cells possess greater chemotherapeutic resistance and tumorigenic ability compared to OV6- Cells [142]. Keratin (K)19, another LCSCs marker was detected in HCC with poor prognosis [143]. The K19 expression exhibits a strong correlation with increased EMT-related proteins, tumorigenicity, metastasis and invasiveness in HCC [143].
To date, there is a great variation of LCSCs surface marker expression across different cell types and diseases. According to Zheng et al. cells pre-dominantly expressing single markers could develop into a population with mixed CSCs markers due to the phenotypic heterogeneity. This forms a huge challenge in the identification and classification of CSCs [144]. Zhu and colleagues revealed that only a minority of CD133 + isolate from HCC cell lines are stem cells, although CD133 + cancer cells possessed some stem cell-like properties, including higher proliferative potential, greater colony‐forming efficiency, self‐renewal and differentiating capacity when compared to CD133 − counterparts [139]. Interestingly, it was found that CD44 was consistently and preferentially expressed in CD133 + cells. From the same experiment, Zhu identified that CD133 + CD44 + are more tumorigenic and more resistant to chemotherapy agents compared to CD133 + CD44- counterparts [139]. Hence, a double positive subpopulation is more likely to represent the actual LCSCs.
Liver cancer stem cells in HCC progression
Cumulative evidence showed that the initiation and development of HCC can be accelerated by the presence of LCSCs [145]. LCSCs might also contribute to the disease metastasis and recurrence after the tumour resection [27]. The underlying pathways in regulating the proliferation of CSCs include Wnt/β-catenin, Notch, fibroblast growth factors (FGF) and TGF-β signalling [146,147,148,149]. Similar to cancer cells, genetic aberrations in stem cells can cause them to divide uncontrollably and lead to tumorigenesis [150]. CSCs regulate their stemness properties and are actively involved in angiogenesis to support tumour growth and metastasis [25]. CSCs also recruit, transform and modify the functions of neighbouring cells and surrounding extracellular matrix (ECM) to promote tumour growth [151]. A study by Gao et al. revealed that HCC cells co-expressing CSC markers- CD44 and EMT markers: N-cadherin and Vimentin possessed accelerated cell migration and metastatic rate [152]. Although the clear relationship between EMT and cancer stemness is not well understood, the speculation is the stem cell traits were acquired during EMT and aggravated the intrahepatic metastasis [153]. Furthermore, patients with LCSCs have a relatively poor prognosis by having more frequent recurrence after the curative therapies [140, 154]. Specifically, a study demonstrated that patients with CD133 + HCC subpopulation cells had a poorer prognosis and more frequently presented with multiple tumour lesions [155]. In another study, a population of CD13high subpopulation cells resulted in a larger size of tumour and earlier cancer recurrence as compared to CD13low cells [156]. Taken together, it is highly evident that CSCs play a key role in driving cancer progression and relapse.
Liver cancer stem cells in drug resistance
One of the primary concerns of CSCs in the ineffectiveness of cancer therapy is its role in driving drug resistance. This can occur through several cellular events such as hypoxia, decreased ROS production, upregulation of drug-efflux pumps and increased expression of aldehyde dehydrogenase (ALDH) enzyme [157]. CSCs are mainly found close to the hypoxic zone of the tumour core, hence only a small proportion of CSCs is exposed to the anticancer agent since hypoxic tumour zones are distant from the blood supply [158]. The activation of HIF1α can promote drug resistance by decreasing ROS production which enhanced cancer cell survival [159]. For instance, Baumann and colleagues demonstrated that a higher radiation dose was required to kill the tumour cells with low levels of ROS [159]. On the other hand, hypoxia drives CSCs to enter the quiescence state to evade cancer therapies that target actively dividing cells [160]. LCSCs are also known to upregulate ATP-binding cassette (ABC) transporters in the efflux of anticancer drugs which then leads to multidrug resistance [161]. For example, an elevated number of CD90 + CD133 + LCSCs and upregulation of ABCG2 were found in the HCC tissues and were responsible for drug resistance [162, 163]. ALDH plays a role in suppressing apoptosis in cancer cells that are induced by DNA damage [155]. ALDH takes part in the metabolism of 4-hydroxy-2-nonenal (4-HNE), converting it to 4-hydroxynon-2-enoic acid. This aids in detoxifying 4-HNE and act as a defence against oxidative stress [164], as 4-HNE is a product of lipid peroxidation which is triggered by oxidative stress. It can cause various protein disruption, DNA damage and alterations in mitochondrial metabolism [165, 166]. Since chemotherapy leads to elevated oxidative stress, the function of ALDH as a defence confers chemoresistance [165]. CSCs can repair DNA damage by promoting the expression of nucleotide excision repair protein (ERCC1) [167]. Besides promoting drug resistance through DNA repair systems, CSCs can escape the apoptotic pathway through mutations or inactivation of the apoptotic gene, p53 and its isoform p63 and p73 [168].
Role of SIRT1 in stemness maintenance and drug resistance in LCSCs
Mammalian sirtuins (encoded by SIRT1-7 genes) are nicotinamide adenosine dinucleotide (NAD)-dependent deacetylases that are involved in many biological processes such as cell survival, senescence, proliferation, apoptosis, DNA repair, cell metabolism and caloric restriction [169]. The sirtuin protein is characterized by a conserved 275 amino acid catalytic core domain with additional N-terminal and/or C-terminal sequences of variable length [170]. SIRT1 was the first SIRT family member that was discovered in the nucleus [170]. Later, it was found there are 7 members in which there have specific and overlapping functions mainly regulating metabolism, cell fates via epigenetic and gene expression modulation depending on their localisation, either cytoplasm, mitochondria or nucleus [171]. SIRT1 has been reported in regulating the functions of stem cells that are crucial for normal embryonic development and adult tissue homeostasis through self-renewal and differentiation [172]. SIRT1 is overexpressed in various cancers including human myeloid leukaemia, colon cancer, prostate cancer and liver cancer [173]. Conversely, SIRT1 expression is reduced in ovarian cancers and glioblastoma [174]. Intriguingly, SIRT1 is found to act as a tumour promoter and suppressor via the Sirt1-p53 axis, thought to be localisation dependent [171]. However, the mechanism in which SIRT1 regulate tumorigenesis warrants an investigation [169].
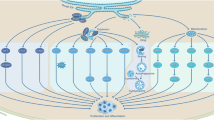
In HCC cells and LCSCs, the SIRT1 gene was observed to be overly expressed [173]. This is attributed due to the regulatory role of SIRT1 in self-renewal, stemness maintenance, cell survival and proliferation in HCC. The underlying mechanisms that may be involved are Wnt/β-catenin, Notch, mitogen-activated protein kinase (MEK) and mitochondrial ribosomal protein S5 (MRPS5) deacetylation (Fig. 3) [175]. Wnt/β-catenin is activated through the binding of Wnt to the frizzled receptors leading to the inhibition of the degradation complex, thus stabilizing β-catenin. SIRT1 promotes Wnt/β-catenin signalling through inhibition of the pathway antagonist and stabilizing β-catenin [172]. Chen et al. demonstrated that SIRT1 deacetylates β-catenin to promote its nuclear accumulation, which then activates the Wnt/β-catenin signalling pathway that is involved in the maintenance of stemness in the tissue and progression of various cancers [176].
Role of SIRT 1 and the related signalling pathways in stemness maintenance and drug resistance. (1) In the Wnt/β-catenin signalling pathway, SIRT1 deacetylates and stabilizes β-catenin leading to its translocation into the nucleus. β-catenin then activates the expression of downstream proteins such as c-Myc, EpCAM and ABCB 1 that improve stemness and induce the drug resistance of LCSCs. (2) The notch signalling pathway involves the destabilization of NICD by SIRT1, hence inhibiting the translocation of NICD into the nucleus. In this event, the expression of downstream proteins hes1 and hey1 is halted. This inhibits cell differentiation in LCSCs. (3) Notch signalling also activates SIRT1 leading to its translocation into the nucleus. SIRT1 deacetylates and activates LSD 1, leading to the transcription of the SOX2 gene, hence increasing the stemness of cells. (4) Lastly, SIRT1 deacetylates MRPS5 which leads to its translocation into mitochondria. SIRT1 also reduces the ROS through the activation of mitochondria unfolded protein response (UPRmt) activity. AC Acetyl, TCF/LEF T-cell factor/lymphoid enhancer factor, CSL CBF-1, Suppressor hairless Lag-1, MAML mastermind like protein, NICD notch intracellular domain, NECD notch extracellular domain, LSD 1 lysine demethylase 1, UPRmt mitochondria unfolded protein response, ROS: reactive oxygen species
Notch signalling is known to modulate cell proliferation, cell differentiation and cell survival [177], but its role in HCC is highly debatable. As reported by Qi et al. the overexpression of Notch1 was able to inhibit the growth of HCC cells in vitro and in vivo [178]. O’Callaghan et al. demonstrated that SIRT1 was highly expressed in LCSCs and downregulated the Notch signalling pathway through the inhibition of nuclear translocation of notch intracellular domain (NCID). This then led to the inhibition of downstream effector such as Hes1 and Hey1 which are involved in cell differentiation [172].
As opposed to Qi et al. [178] and O’Callaghan et al. [172], Ning and colleagues indicated that Notch expression was highly activated in HCC in comparison to the adjacent normal tissue and its upregulation promoted the tumorigenicity of HCC [179]. As demonstrated by Farcas et al. Notch signalling induced the SIRT1 expression and activated the lysine demethylase 1 (LSD1), thus promoting LCSCs self-renewal and improved stemness through transcription of the SOX2 gene [175]. SOX2 is an embryonic transcription factor that is associated with the self-renewal ability of CSCs [175]. Likewise, Liu et al. demonstrated that SIRT1 is recruited to the promoter of SOX2 gene and regulates transcription of SOX2 gene by chromatin-based epigenetic changes [180]. SOX2 is a well-known factor to promote cell proliferation, survival, drug resistance and cancer stemness [181]. These evidence illustrated that SIRT1 promotes tumorigenesis of LCSCs by upregulating the SOX-2 expression through the activation notch signalling pathway.
Mitochondrial ribosomal protein S5 (MRPS5) supports the mitochondria function of LCSCs to promote the progression of cancer [182]. The overexpressed SIRT1 in LCSCs deacetylates MRPS5 to promote its translocation into mitochondria, thus inducing oxidative phosphorylation and production of ROS. SIRT1 also promotes mitochondria unfolded protein response (UPRmt) activity that reduces the ROS levels, consequently improving mitochondrial function that helps in maintaining the stemness of LCSC [175]. Furthermore, there is a positive interaction between mitogen-activated protein kinase 1 (MEK1) signalling activation and SIRT1 expression with the protein stability [183]. MAPK1/MEK1 are enzymes that phosphorylate threonine and tyrosine residues within the activation loop of their MAP kinase substrates [184]. Based on the study by Cheng et al., reduced MEK1 expression led to degradation of SIRT1 and resulted in the decrease in the stem cell markers expression in LCSC such as SOX2, Oct4 and NANOG that significantly contributes to self-renewal [183].
Interplay between autophagy and liver cancer stem cells in HCC progression
Autophagy and hepatic/liver progenitor cells in HCC
Autophagy is known to regulate the multiple processes in stem cells as it plays a role in the removal of damaged organelles and proteins during stem cell renewal, differentiation and growth [185]. Under normal conditions, autophagy inhibition can disrupt the growth and proliferation of hepatic progenitor cells (HPCs) or liver progenitor cells (LPCs). HPCs/LPCs are the progeny of stem cells that reside in the liver which differentiate into hepatocytes and cholangiocytes, which are the two most abundant epithelial cell types in the liver [186]. A study demonstrated that the lentiviral-mediated knockdown of the autophagic gene, ATG5 and BECN1 resulted in the reduced cell proliferation and differentiation of LPCs. ATG5 or BECN1 knockdown impaired stemness, suggesting that autophagy is involved in conferring LPC function [187]. The autophagy deficiency in HPCs also reduced the spheroid or colony formations and delayed liver regeneration in rats which suggest the importance of autophagy in preventing dysfunction of self-renewal and stemness [188]. The same study also demonstrated that autophagy inhibition resulted in the accumulation of damaged mitochondria and mitochondrial ROS while decreasing the mitochondrial membrane potential [188].
Autophagy and liver cancer stem cells in HCC
The exact origin of CSCs remains unclear to our knowledge, however since CSCs have similar characteristics/markers as normal stem cells, it is presumed that CSCs could be derived from normal stem cells [189]. A study has shown that progenitor cells from the mouse liver were able to develop into cancer, like that of human HCC following genetic manipulation and transplantation into recipient mice [190]. Similarly, with the inactivation of TGFβ signalling and interleukin-6 associated transformation, progenitor cells from the mouse liver led to carcinogenesis [191].
As the autophagic process has been proven to be pivotal in HPCs, it is also known to be important in LCSC survival through mitophagy [192]. In order for cancer stem cells to survive in the harsh tumour microenvironment of HCC, the breakdown of damaged mitochondria is important in adaptation to environmental stresses such as hypoxia. Results show that mitophagy is involved in the suppression of p53 which leads to positive LCSC regulation and hepatocarcinogenesis [178, 192]. The inhibition of mitophagy led to p53 phosphorylation by PINK1 (PTEN-induced putative kinase) and nuclear translocation and subsequent suppression of transcription factor NANOG which is responsible for stemness and self-renewal in CSCs [192]. Axin2, a negative regulator of Wnt/β-catenin, is also involved in stem cell regulation. Dysregulation of autophagy in cirrhosis demonstrated increased expression of hepatocyte growth factor (HGF) activating the Met/JNK and Met/STAT3 pathway in Axin2/EGFP + hepatic cells and resulted in the following transition to Axin2/EGFP + /CD90 + cells having acquired stem cells characteristics. The activation of Met/JNK and Met/STAT3 pathway via HGF expression is concluded to be an important process in carcinogenesis in the liver [193].
In HCC, autophagy is also closely linked to the stem cell characteristic of self-renewal and the maintenance of stemness, which is evident by observing sphere formation [194]. Results revealed that CD133 + was shown to have higher autophagic levels with increased GFP-LC3 puncta. Autophagy inhibition by chloroquine enhanced apoptosis and diminished proliferation ability with reduced sphere-forming ability of liver cancer cell line, Huh7. Inversely, the same study demonstrates an increase in the sphere-forming capacity of CD133 + cells following the induction of autophagy [194].
As previously mentioned, the interplay between autophagy and CSC survival and self-renewal had been established. Yet, little is known about the interrelation between autophagy and LCSC differentiation. CCND1, which encodes for cyclin D1, is responsible for the phosphorylation and inactivation of the retinoblastoma protein [31]. Upregulated expression of CCND1 is often seen in HCC and is associated with poor prognosis and cancer recurrence [195]. It was shown that the overexpression of cyclin D1 enhanced the expression of the autophagic LC3-II and Beclin-1. On the contrary, both the expression of LC3-II and Beclin-1 was reduced when CCND1 was silenced, suggesting that autophagy plays a role in LCSC differentiation, thereby contributing to the progression of HCC. Overall, CCND1 silencing inhibited LCSC differentiation through the suppression of autophagy [195].
Crosstalks of autophagy and LCSC signalling pathways
There are a variety of different CSC-signalling pathways that are also involved in the regulation of autophagy including FGF signalling, TGF-β signalling, Notch signalling pathway and Wnt/β-catenin signalling (Fig. 4).
Role of different CSC-related signalling pathways in autophagy. (1) FGF signalling pathway. FGF signalling suppresses the initiation of autophagy by activating mTOR which subsequently inhibits the recruitment of the ULK complex. (2) TGF-β signalling pathway. TGF-β facilitates phagophore elongation. (3) Both TGF-β and Cyclin D1 is involved in the upregulation of LC3-II and mediates the autophagosome formation. (4) Notch signalling pathway. Autophagy is involved in inhibiting the Notch signalling pathway through the degradation of Notch via uptake of ATG16L1-positive autophagosomes (5) Wnt/β-catenin signalling pathway. β-catenin is found to inhibit autophagosome formation and repressing p62 expression
FGFs and their receptors fibroblast growth factor receptors (FGFRs) play an important role in HCC progression [196]. There are 22 known human FGFs and four FGFR types identified which act as transmembrane tyrosine kinase [197]. In the majority of HCC cases reported by Gauglhofer et al., 82% presented upregulation of FGFs and/or FGFRs. Further study revealed that FGF17 and FGF18 promoted HCC-derived myofibroblasts proliferation, while FGF8, as well as FGF17 and FGF18, promoted the growth of hepatic endothelial cells. This indicates that FGF8, FGF17 and FGF18 are involved in HCC malignancy and angiogenesis [198]. Besides, the upregulation of FGF19 led to elevated cyclin D1 levels through the activation of β-catenin. FGF19 and CCND1 play an important role in tumorigenesis in HCC [199]. Furthermore, it is certain that the FGF signalling is involved in many cellular pathways and there is a growing interest as a therapeutic target for HCC. FGFR4 inhibitor, BLU9931, was shown to have a binding affinity to FGFR4, and as a result, inhibited HCC proliferation in vitro. Likewise, BLU9931 also exhibited antitumor activity in HCC xenograft in mice [200]. Anti-FGFR4 neutralizing antibody LD1 was generated by French and colleagues. LD1 demonstrated colony inhibition of HCC cell lines, and tumour growth in vivo via modulation of FGFR4 downstream genes. This establishes the role of FGFR4 in HCC progression [201]. In CSCs, FGFR modulated proliferation of hepatoblasts and tumour initiating stem cells/CSCs via AKT-β-catenin-CBP pathway [202]. Additionally, FGF signalling is indeed found to be related to autophagy. The inhibition of FGF signalling promoted LC3 conversion to LC3-II. FGF signalling negatively regulates autophagy through activation of mTOR [203]. FGF/FGFR signalling was identified as a crucial element in autophagy regulation in bone growth [204]. Similarly, the inhibition of FGFR1 promoted autophagy, while the activation of FGFR1 resulted in the autophagy suppression via Beclin-1 silencing which led to increased apoptosis after FGFR inhibitor, AZ4547 treatment in lung cancer [205]. Although FGF signalling is related to autophagy in other cell types, its role in relation to autophagy in LCSCs and HCC is not well established.
TGF-β signalling is also very much involved in autophagic regulation and cellular processes including survival, growth and differentiation [206]. TGF-β was reported to induce autophagosomal formation and potentiate the conversion of LC3-I to LC3-II in HCC [206]. As a result, autophagic flux was increased. The study also revealed the involvement of Smad signalling which comprises of Smad downstream effectors of Type I and Type II receptors that typically binds to the signalling molecule TGF-β [206]. Thus, the facilitation of the autophagic pathway in the TGF-β-related growth inhibition in HCC cells was recognised [206]. This phenomenon correlates to the tumour-suppressing role of autophagy in HCC. On the contrary, TGF-β1, a subgroup/isoform of TGF-β, was found to induce autophagy and increase the capability for cellular invasion of HCC cells. With TGF-β1 treatment, elevated LC3 and Beclin-1 levels were observed. Besides, combination treatment with autophagy inhibitor 3-methyladenine significantly inhibited cell invasion [207].
The Notch signalling pathway is involved in the differential process of HPCs into biliary cells and in turn, contribute to the bile duct and liver development [208]. Autophagy was found to be involved in regulating biliary differentiation of HPCs through Notch1 signalling. Biliary differentiation was induced in rat HPC cell line and a low autophagy level was observed in both early and later stage of differentiation. Autophagy was found to suppress biliary differentiation of HPCs through the inhibition of the Notch1 signalling pathway [209]. Notch is typically degraded/inhibited by autophagy through the uptake of ATG16L1-positive autophagosomes [210]. Sustained Notch signalling is dependent on mTOR activation and subsequent autophagy inhibition [211]. Besides, Notch1 is also involved in HCC development as the expression of Notch1 and its ligand Jagged1 were reported to be higher in normal liver tissue than in HCC, which implies that Notch1 can act as a tumour suppressor in HCC [212]. Similarly, the knockout of Notch1 led to uncontrollable growth of liver cells which suggests that defective Notch1 may contribute to hepatocarcinogenesis [213].
The activation of Wnt/β-catenin signalling is often seen in HCC and is highly associated with poor prognosis [214]. Its relationship with autophagy and HCC as well as LCSCs has been discovered. Wnt/β-catenin pathway inhibitor, 2,5-dichloro-N-(2-methyl-4-nitrophenyl) benzenesulfonamide (FH535), targets β-catenin and were used to treat HCC cell lines. This led to an upregulation of LC3-II and p62 which corresponds to the build-up of autophagosomes in the cells as a result of impaired lysosomal degradation [215]. The synergistic treatment of FH535 and sorafenib greatly disrupted the autophagic flux and increases apoptosis, which suggests that Wnt/β-catenin signalling is vital for the survival of HCC cells [215]. In contrast, Wnt/β-catenin signalling was found to negatively regulate autophagy by inhibiting autophagosome formation and p62 expression [216]. Reciprocally, autophagy activates Wnt/β-catenin as increased expression of β-catenin upon autophagic induction was observed in HCC [217]. HPC/stem cell differentiation is facilitated by autophagy through the Wnt/β-catenin signalling pathway [218]. Autophagy was observed to be highly upregulated during the cell differentiation of HPC [218]. The deactivation of autophagy through downregulation of the ATG5 gene was shown to disrupt HPC differentiation by inhibiting the Wnt/β-catenin signalling pathway which is important in cell proliferation and renewal of CSCs [218]. The hampering of Wnt/β-catenin led to a decrease in cell viability and self-renewal of LCSCs as well [219]. Hence, autophagy and Wnt/β-catenin remains an attractive target for HCC treatment. Although the relationship between autophagy and LCSCs with the Wnt/β-catenin pathway is known, the mechanistic involvement of autophagy with Wnt/β-catenin signalling is still poorly understood to our knowledge.
The four signalling pathways mentioned in this section are also involved in the maintenance and survival of CSCs. Since autophagy is involved in the regulation of these signalling pathways, we can speculate that autophagy also plays a role in CSCs via these signalling pathways.
Above we described the role of autophagy in cancer, including tumour suppression, promotion and metastasis. Some of the results from model studies may contribute to the linkage between autophagy and tumours growth, but the contribution of these autophagy-related pathway activities toward human cancer need extensive research. Numerous studies have found an association of autophagy with cell survival, stemness as well as the differentiation of LCSCs, however, the specific and detailed mechanisms as to how autophagy is involved in different metabolic pathways or molecular processes remains unknown to our knowledge. To date, drug resistance still represents the most challenging issue in anti-cancer treatment. Autophagy and CSC remain as an attractive target for treatment regimen, but doubts are not resolved. Drugs such as chloroquine or hydroxychloroquine that function as autophagy modulators bring about some undesired side effects including retinopathy, inflammation, and impact on white blood cells [220]. Moreover, none of the clinical trials using autophagy modulators assures the hypothesis that inhibition of autophagy produces a great therapeutic effect in cancer patients [221]. Yet, autophagy is important for survival, proliferation and in some cases, inhibition of tumour progression. Long-period inhibition of autophagy (both local and systemic) could probably lead to unwanted outcomes in cancer patients since it is not clear whether the drug is sufficiently specific. Also, the understanding of CSC and its applicability in anti-cancer is not so thorough. Although CSC has similar characteristics with normal stem cells, it is not well explained with the origin of CSC, as not all CSC originate from stem cells that turned malignant. Instead, tumours enriched with CSC may give rise to non-CSC populations, while non-CSC tumours also produce CSC populations [221]. The diversity of CSC, and the possible evolution of their protective measure after treatment will be one of the challenges in developing treatment strategies. We learned that many studies are conducted to clarify LCSC stemness features, but the biological difference between normal and cancer stem cells is not well-established. In order to produce a method that eradicates only CSCs without affecting normal stem cells, the exploration of uniqueness in CSCs must be performed in-depth.
Conclusions
In conclusion, autophagy is a conserved pathway which involves the intracellular self-digestion mechanism that degrades damaged cellular materials and wastes through lysosomal degradation. The role of autophagy has been implicated in various cellular processes which are important for cell survival and tumour suppression in liver cells. Aberrant autophagic regulation is linked to hepatocarcinogenesis, HCC progression and malignancy. Although we now know that autophagy is associated with the various cellular pathways that are related to HPC and CSC regulation, the exact mechanism in which autophagy is involved is not well established. In normal stem cells, strict regulation of protein turnover mechanisms is vital and required for cellular differentiation, renewal and regeneration, without which malignant transformation would ensue. With the advancement of research, the relationship of LCSCs with autophagy in HCC will be progressively established. A deeper understanding of the role of autophagy in the survival and the biology of LCSCs in HCC may advance and improve diagnostics and contribute to the development of new treatment methods through the identification of novel biomarkers and drug targets. By identifying and determining crucial elements that contribute to carcinogenesis and HCC progression, we will be equipped to tackle the issue of recurring disease and improve prognosis.
Abbreviations
- HCC:
-
Hepatocellular carcinoma
- HBV:
-
Hepatitis B virus
- HCV:
-
Hepatitis C virus
- CSC:
-
Cancer stem cell
- PI3K:
-
Phosphatidylinositol 3-kinase
- VPS34:
-
Vacuolar protein sorting 34
- mTORC1:
-
Mammalian target of rapamycin complex
- ULK1:
-
Unc-51 like kinase 1
- ATG:
-
Autophagy related protein
- FIP200:
-
Focal adhesion kinase family-interacting protein 200 kDa
- AMPK:
-
5` Adenosine monophosphate-activated protein kinase
- PI3P:
-
Phosphatidylinositol triphosphate
- LC3:
-
Microtubule-associated protein 1A/1B-light chain 3
- PE:
-
Phosphatidylethanolamine
- SQSTM1/p62:
-
Sequestosome-1/ubiquitin-binding protein p62
- NIX:
-
BNIP3L/Receptor for mitochondrial protein
- OPTN:
-
Optineurin
- LAMP2:
-
Lysosome-associated membrane protein 2
- GRASP55:
-
Golgi reassembly-stacking protein 55
- EMS:
-
Eukaryotic endomembrane system
- ILVs:
-
Intraluminal vesicles
- MVBs:
-
Multivesicular bodies
- MDB:
-
Mallory-Denk bodies
- ATP:
-
Adenosine triphosphate
- ROS:
-
Reactive oxygen species
- RNS:
-
Reactive nitrogen species
- Nrf2-Keap1:
-
Nuclear factor erythroid 2-related factor 2-kelch-like ECH-associated protein 1
- HIF:
-
Hypoxia inducible factor
- BNIP3:
-
BCL2 and adenovirus E1B 19-kDa-interacting protein 3
- BNIP3L:
-
BCL2: adenovirus E1B 19-kDa-interacting protein 3-like
- COX2:
-
Cyclooxygenase-2
- PGE2:
-
Prostaglandin E(2)
- TNM:
-
Tumour, node, metastasis
- CTC:
-
Circulatory tumour cells
- EMT:
-
Epithelial-mesenchymal transition
- TGF-β:
-
Transforming growth factor β
- TRAF6:
-
Tumour necrosis factor receptor-associated factor-6
- TAK1:
-
TGF-β activated kinase 1
- DRAM1:
-
DNA damage modulator 1
- CREB:
-
CAMP response element binding
- HO-1:
-
ROS/heme oxygenase 1
- UPR:
-
Unfolded protein response
- HSP:
-
Heat shock protein
- 5-FU:
-
5-Fluorouracil
- TAE:
-
Transarterial embolization
- TACE:
-
Transarterial chemoembolization
- Egr-1:
-
Early growth response-1
- IR:
-
Ionizing radiation
- VEGFR:
-
Vascular endothelial growth factor receptor
- PDGFR-β:
-
Platelet-derived growth factor receptor β
- GSTM1:
-
Glutathione transferase Mu 1
- LCSC:
-
Liver cancer stem cells
- AFP:
-
Alpha fetoprotein
- FGFs:
-
Fibroblast growth factors
- ECM:
-
Extracellular matrix
- SIRT1 :
-
Gene encoding Sirtuin1 protein
- ALDH:
-
Aldehyde dehydrogenase
- ABC:
-
ATP-binding cassette
- 4-HNE:
-
4-Hydroxy-2-nonenal
- NAD:
-
Nicotinamide adenosine dinucleotide
- MEK:
-
Mitogen-activated protein kinase
- MRPS5:
-
Mitochondrial ribosomal protein S5
- AC:
-
Acetyl
- TCF/LEF:
-
T-cell factor/lymphoid enhancer factor
- CSL, CBF-1:
-
Suppressor hairless lag-1
- MAML:
-
Mastermind-like protein
- NICD:
-
Notch intracellular domain
- NECD:
-
Notch extracellular domain
- LSD1:
-
Lysine demethylase 1
- UPRmt:
-
Mitochondrial unfolded protein response
- HPC/LPC:
-
Hepatic/liver progenitor cells
- PINK1:
-
PTEN-induced putative kinase
- HGF:
-
Hepatocyte growth factor
- FGFR:
-
Fibroblast growth factor receptor
References
Mathew R, Karantza-Wadsworth V, White E (2007) Role of autophagy in cancer. Nat Rev Cancer 7:961–967. https://doi.org/10.1038/nrc2254
King JS (2012) Autophagy across the eukaryotes: is S. cerevisiae the odd one out? Autophagy 8:1159–1162. https://doi.org/10.4161/auto.20527
Tsukada M, Ohsumi Y (1993) Isolation and characterization of autophagy-defective mutants of Saccharomyces cerevisiae. FEBS Lett 333:169–174. https://doi.org/10.1016/0014-5793(93)80398-e
Abounit K, Scarabelli TM, McCauley RB (2012) Autophagy in mammalian cells. World J Biol Chem 3:1–6. https://doi.org/10.4331/wjbc.v3.i1.1
Weidberg H, Shvets E, Elazar Z (2011) Biogenesis and cargo selectivity of autophagosomes. Annu Rev Biochem 80:125–156. https://doi.org/10.1146/annurev-biochem-052709-094552
Lee MS (2014) Role of islet β cell autophagy in the pathogenesis of diabetes. Trends Endocrinol Metab 25:620–627. https://doi.org/10.1016/j.tem.2014.08.005
Yun CW, Lee SH (2018) The roles of autophagy in cancer. Int J Mol Sci 19:3466. https://doi.org/10.3390/ijms19113466
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Raihan R, Azzeri A, Shabaruddin FH, Mohamed R (2018) Hepatocellular carcinoma in Malaysia and its changing trend. Euroasian J Hepatogastroenterol 8:54–56. https://doi.org/10.5005/jp-journals-10018-1259
Montella M, Crispo A, Giudice A (2011) HCC, diet, and metabolic factors. Hepat Mon 11:159–162
Bartosch B (2010) Hepatitis B and C viruses and hepatocellular carcinoma. Viruses 2:1504–1509. https://doi.org/10.3390/v2081504
Chisari FV, Isogawa M, Wieland SF (2010) Pathogenesis of hepatitis B virus infection. Parodontol 58:258–266. https://doi.org/10.1016/j.patbio.2009.11.001
Irshad M, Mankotia DS, Irshad K (2013) An insight into the diagnosis and pathogenesis of hepatitis C virus infection. World J Gastroenterol 19:7896–7909. https://doi.org/10.3748/wjg.v19.i44.7896
El-Serag HB, Rudolph KL (2007) Hepatocellular carcinoma: Epidemiology and molecular carcinogenesis. Gastroenterology 132:2557–2576. https://doi.org/10.1053/j.gastro.2007.04.061
Heidelbaugh JJ, Bruderly M (2006) Cirrhosis and chronic liver failure: Part I. Diagnosis and evaluation. Am Fam Physician 74:756–762
Sengupta S, Parikh ND (2017) Biomarker development for hepatocellular carcinoma early detection: current and future perspectives. Hepatic Oncol 4:111–122. https://doi.org/10.2217/hep-2017-0019
Llovet JM, Zucman-Rossi J, Pikarsky E, Sangro B, Schwartz M, Sherman M, Gores G (2016) Hepatocellular carcinoma. Nat Rev Dis Prim 2:16018. https://doi.org/10.1038/nrdp.2016.18
Llovet JM, Fuster BJ (2004) The Barcelona approach: diagnosis, staging, and treatment of hepatocellular carcinoma. Liver Transplant 10:S115-120. https://doi.org/10.1002/lt.20034
Santopaolo F, Lenci I, Milana M, Manzia TM, Baiocchi L (2019) Liver transplantation for hepatocellular carcinoma: Where do we stand? World J Gastroenterol 25:2591–2602. https://doi.org/10.3748/wjg.v25.i21.2591
Di Fazio P, Matrood S (2018) Targeting autophagy in liver cancer. Transl Gastroenterol Hepatol 3:39. https://doi.org/https://doi.org/10.21037/tgh.2018.06.09
White E, Karp C, Strohecker AM, Guo Y, Mathew R (2010) Role of autophagy in suppression of inflammation and cancer. Curr Opin Cell Biol 22:212–217. https://doi.org/10.1016/j.ceb.2009.12.008
Akkoç Y, Gözüaçık D (2018) Autophagy and liver cancer. Turkish J Gastroenterol 29:270–282. https://doi.org/10.5152/tjg.2018.150318
Sheng J, Qin H, Zhang K, Li B, Zhang X (2018) Targeting autophagy in chemotherapy-resistant of hepatocellular carcinoma. Am J Cancer Res 8:354–365
Huang F, Wang BR, Wang YG (2018) Role of autophagy in tumorigenesis, metastasis, targeted therapy and drug resistance of hepatocellular carcinoma. World J Gastroenterol 24:4643–4651. https://doi.org/10.3748/wjg.v24.i41.4643
Ayob AZ, Ramasamy TS (2018) Cancer stem cells as key drivers of tumour progression. J Biomed Sci 25:20. https://doi.org/10.1186/s12929-018-0426-4
Desai A, Yan Y, Gerson SL (2019) Concise reviews: cancer stem cell targeted therapies: toward clinical success. Stem Cells Transl Med 8:75–81. https://doi.org/10.1002/sctm.18-0123
Ji J, Wang XW (2012) Clinical implications of cancer stem cell biology in hepatocellular carcinoma. Semin Oncol 39:461–472. https://doi.org/10.1053/j.seminoncol.2012.05.011
Wang N, Wang S, Li MY, Hu BG, Liu LP, Yang SL, Yang S, Gong Z, Lai PBS, Chen GG (2018) Cancer stem cells in hepatocellular carcinoma: An overview and promising therapeutic strategies. Ther Adv Med Oncol 10:1758835918816287. https://doi.org/10.1177/1758835918816287
Visvader JE, Lindeman GJ (2012) Cancer stem cells: current status and evolving complexities. Cell Stem Cell 10:717–728. https://doi.org/10.1016/j.stem.2012.05.007
Wang K, Sun D (2018) Cancer stem cells of hepatocellular carcinoma. Prim Liver Cancer Challenges Perspect 9:23306–23314
Zhang H (2020) CCND1 silencing suppresses liver cancer stem cell differentiation through inhibiting autophagy. Hum Cell 33:140–147. https://doi.org/10.1007/s13577-019-00295-9
Deter RL, De Duve C (1967) Influence of glucagon, an inducer of cellular autophagy, on some physical properties of rat liver lysosomes. J Cell Biol 33:437–449. https://doi.org/10.1083/jcb.33.2.437
De Duve C, Wattiaux R (1966) FUNCTIONS OF LYSOSOMES. Annu Rev Physiol 28:435–492. https://doi.org/10.1146/annurev.ph.28.030166.002251
Ohsumi Y (2014) Historical landmarks of autophagy research. Cell Res 24:9–23. https://doi.org/10.1038/cr.2013.169
Das G, Shravage BV, Baehrecke EH (2012) Regulation and function of autophagy during cell survival and cell death. Cold Spring Harb Perspect Biol 4:1–14. https://doi.org/10.1101/cshperspect.a008813
Filomeni G, De Zio D, Cecconi F (2015) Oxidative stress and autophagy: the clash between damage and metabolic needs. Cell Death Differ 22:377–388. https://doi.org/10.1038/cdd.2014.150
Backer JM (2008) The regulation and function of Class III PI3Ks: novel roles for Vps34. Biochem J 410:1–17. https://doi.org/10.1042/BJ20071427
Ganley IG, Lam DH, Wang J, Ding X, Chen S, Jiang X (2009) ULK1·ATG13·FIP200 complex mediates mTOR signaling and is essential for autophagy. J Biol Chem 284:12297–12305. https://doi.org/10.1074/jbc.M900573200
Shang L, Chen S, Du F, Li S, Zhao L, Wang X (2011) Nutrient starvation elicits an acute autophagic response mediated by Ulk1 dephosphorylation and its subsequent dissociation from AMPK. Proc Natl Acad Sci U S A 108:4788–4793. https://doi.org/10.1073/pnas.1100844108
Kroemer G, Mariño G, Levine B (2010) Autophagy and the integrated stress response. Mol Cell 40:280–293. https://doi.org/10.1016/j.molcel.2010.09.023
Yang Z, Klionsky DJ (2010) Mammalian autophagy: Core molecular machinery and signaling regulation. Curr Opin Cell Biol 22:124–131. https://doi.org/10.1016/j.ceb.2009.11.014
Axe EL, Walker SA, Manifava M, Chandra P, Roderick HL, Habermann A, Griffiths G, Ktistakis NT (2008) Autophagosome formation from membrane compartments enriched in phosphatidylinositol 3-phosphate and dynamically connected to the endoplasmic reticulum. J Cell Biol 182:685–701. https://doi.org/10.1083/jcb.200803137
Noda NN, Fujioka Y, Hanada T, Ohsumi Y, Inagaki F (2013) Structure of the Atg12-Atg5 conjugate reveals a platform for stimulating Atg8-PE conjugation. EMBO Rep 14:206–211. https://doi.org/10.1038/embor.2012.208
Walczak M, Martens S (2013) Dissecting the role of the Atg12-Atg5-Atg16 complex during autophagosome formation. Autophagy 9:424–425. https://doi.org/10.4161/auto.22931
Tanida I, Ueno T, Kominami E (2004) LC3 conjugation system in mammalian autophagy. Int J Biochem Cell Biol 36:2503–2518. https://doi.org/10.1016/j.biocel.2004.05.009
Müller AJ, Proikas-Cezanne T (2015) Function of human WIPI proteins in autophagosomal rejuvenation of endomembranes? FEBS Lett 589:1546–1551. https://doi.org/10.1016/j.febslet.2015.05.008
Feng Y, Klionsky DJ (2017) Autophagic membrane delivery through ATG9. Cell Res 27:161–162. https://doi.org/10.1038/cr.2017.4
Ying H, Yue BYJT (2016) Optineurin: the autophagy connection. Exp Eye Res 144:73–80. https://doi.org/10.1016/j.exer.2015.06.029
Viret C, Rozières A, Faure M (2018) Novel insights into NDP52 autophagy receptor functioning. Trends Cell Biol 28:255–257. https://doi.org/10.1016/j.tcb.2018.01.003
Zhang X, Wang Y (2018) GRASP55 facilitates autophagosome maturation under glucose deprivation. Mol Cell Oncol 5:e1494948. https://doi.org/10.1080/23723556.2018.1494948
Zhao YG, Zhang H (2019) Autophagosome maturation: an epic journey from the ER to lysosomes. J Cell Biol 218:757–770. https://doi.org/10.1083/jcb.201810099
Tong J, Yan X, Yu L (2010) The late stage of autophagy: cellular events and molecular regulation. Protein Cell 1:907–915. https://doi.org/10.1007/s13238-010-0121-z
Yu L, McPhee CK, Zheng L, Mardones GA, Rong Y, Peng J, Mi N, Zhao Y, Liu Z, Wan F, Hailey DW, Oorschot V, Klumperman J, Baehrecke EH, Lenardo MJ (2010) Autophagy termination and lysosome reformation regulated by mTOR. Nature 465:942–946. https://doi.org/10.1038/nature09076
Berg TO, Fengsrud M, Strømhaug PE, Berg T, Seglen PO (1998) Isolation and characterization of rat liver amphisomes: evidence for fusion of autophagosomes with both early and late endosomes. J Biol Chem 273:21883–21892. https://doi.org/10.1074/jbc.273.34.21883
Nakamura S, Yoshimori T (2017) New insights into autophagosome-lysosome fusion. J Cell Sci 130:1209–1216. https://doi.org/10.1242/jcs.196352
Mitra V, Metcalf J (2012) Metabolic functions of the liver. Anaesth Intensive Care Med 13:54–55. https://doi.org/10.1016/j.mpaic.2011.11.006
Lavallard VJ, Gual P (2014) Autophagy and non-alcoholic fatty liver disease. Biomed Res Int 2014:120179. https://doi.org/10.1155/2014/120179
Madrigal-Matute J, Cuervo AM (2016) Regulation of liver metabolism by autophagy. Gastroenterology 150:328–339. https://doi.org/10.1053/j.gastro.2015.09.042
Ezaki J, Matsumoto N, Takeda-Ezaki M, Komatsu M, Takahashi K, Hiraoka Y, Taka H, Fujimura T, Takehana K, Yoshida M, Iwata J, Tanida I, Furuya N, Zheng DM, Tada N, Tanaka K, Kominami E, Ueno T (2011) Liver autophagy contributes to the maintenance of blood glucose and amino acid levels. Autophagy 7:727–736. https://doi.org/10.4161/auto.7.7.15371
Singh R, Kaushik S, Wang Y, Xiang Y, Novak I, Komatsu M, Tanaka K, Cuervo AM, Czaja MJ (2009) Autophagy regulates lipid metabolism. Nature 458:1131–1135. https://doi.org/10.1038/nature07976
Akman HO, Raghavan A, Craigen WJ (2011) Animal models of glycogen storage disorders. Prog Mol Biol Transl Sci 100:369–388. https://doi.org/10.1016/B978-0-12-384878-9.00009-1
Ke PY (2019) Diverse functions of autophagy in liver physiology and liver diseases. Int J Mol Sci 20:300. https://doi.org/10.3390/ijms20020300
Afifiyan N, Tillman B, French BA, Sweeny O, Masouminia M, Samadzadeh S, French SW (2017) The role of Tec kinase signaling pathways in the development of Mallory Denk bodies in balloon cells in alcoholic hepatitis. Exp Mol Pathol 103:191–199. https://doi.org/10.1016/j.yexmp.2017.09.001
Ni HM, Woolbright BL, Williams J, Copple B, Cui W, Luyendyk JP, Jaeschke H, Ding WX (2014) Nrf2 promotes the development of fibrosis and tumorigenesis in mice with defective hepatic autophagy. J Hepatol 61:617–625. https://doi.org/10.1016/j.jhep.2014.04.043
Galluzzi L, Pietrocola F, Bravo‐San Pedro JM, Amaravadi RK, Baehrecke EH, Cecconi F, Codogno P, Debnath J, Gewirtz DA, Karantza V, Kimmelman A, Kumar S, Levine B, Maiuri MC, Martin SJ, Penninger J, Piacentini M, Rubinsztein DC, Simon HU, Simonsen A, Thorburn AM, Velasco G, Ryan KM, Kroemer G (2015) Autophagy in malignant transformation and cancer progression. EMBO J 34: 856-880. https://doi.org/https://doi.org/10.15252/embj.201490784
Fu Y, Chung FL (2018) Oxidative stress and hepatocarcinogenesis. Hepatoma Res 4:39. https://doi.org/https://doi.org/10.20517/2394-5079.2018.29
Ciccarone F, Castelli S, Ciriolo MR (2019) Oxidative stress-driven autophagy across onset and therapeutic outcome in hepatocellular carcinoma. Oxid Med Cell Longev 2019:6050123. https://doi.org/10.1155/2019/6050123
Jin SM, Youle RJ (2012) PINK1-and Parkin-mediated mitophagy at a glance. J Cell Sci 125:795–799. https://doi.org/10.1242/jcs.093849
Zhang T, Xue L, Li L, Tang C, Wan Z, Wang R, Tan J, Tan Y, Han H, Tian R, Billiar TR, Tao WA, Zhang Z (2016) BNIP3 protein suppresses PINK1 kinase proteolytic cleavage to promote mitophagy. J Biol Chem 291:21616–21629. https://doi.org/10.1074/jbc.M116.733410
Fujiwara M, Marusawa H, Wang HQ, Iwai A, Ikeuchi K, Imai Y, Kataoka A, Nukina N, Takahashi R, Chiba T (2008) Parkin as a tumor suppressor gene for hepatocellular carcinoma. Oncogene 27:6002–6011. https://doi.org/10.1038/onc.2008.199
Takamura A, Komatsu M, Hara T, Sakamoto A, Kishi C, Waguri S, Eishi Y, Hino O, Tanaka K, Mizushima N (2011) Autophagy-deficient mice develop multiple liver tumors. Genes Dev 25:795–800. https://doi.org/10.1101/gad.2016211
Mathew R, Karp CM, Beaudoin B, Vuong N, Chen G, Chen HY, Bray K, Reddy A, Bhanot G, Gelinas C, DiPaola RS, Karantza-Wadsworth V, White E (2009) Autophagy suppresses tumorigenesis through elimination of p62. Cell 137:1062–1075. https://doi.org/10.1016/j.cell.2009.03.048
Inami Y, Waguri S, Sakamoto A, Kouno T, Nakada K, Hino O, Watanabe S, Ando J, Iwadate M, Yamamoto M, Lee MS, Tanaka K, Komatsu M (2011) Persistent activation of Nrf2 through p62 in hepatocellular carcinoma cells. J Cell Biol 193:275–284. https://doi.org/10.1083/jcb.201102031
Jaramillo MC, Zhang DD (2013) The emerging role of the Nrf2-Keap1 signaling pathway in cancer. Genes Dev 27:2179–2191. https://doi.org/10.1101/gad.225680.113
Yazdani H, Huang H, Tsung A (2019) Autophagy: Dual response in the development of hepatocellular carcinoma. Cells 8:91. https://doi.org/10.3390/cells8020091
Chen C, Lou T (2017) Hypoxia inducible factors in hepatocellular carcinoma. Oncotarget 8:46691–46703. https://doi.org/https://doi.org/10.18632/oncotarget.17358
Bellot G, Garcia-Medina R, Gounon P, Chiche J, Roux D, Pouysségur J, Mazure NM (2009) Hypoxia-induced autophagy is mediated through hypoxia-inducible factor induction of BNIP3 and BNIP3L via their BH3 domains. Mol Cell Biol 29:2570–2581. https://doi.org/10.1128/MCB.00166-09
Zhang J, Ney PA (2009) Role of BNIP3 and NIX in cell death, autophagy, and mitophagy. Cell Death Differ 16:939–946. https://doi.org/10.1038/cdd.2009.16
Decuypere JP, Parys JB, Bultynck G (2012) Regulation of the autophagic Bcl-2/Beclin 1 interaction. Cells 1:284–312. https://doi.org/10.3390/cells1030284
Sun L, Li T, Wei Q, Zhang Y, Jia X, Wan Z, Han L (2014) Upregulation of BNIP3 mediated by ERK/HIF-1α pathway induces autophagy and contributes to anoikis resistance of hepatocellular carcinoma cells. Future Oncol 10:1387–1398. https://doi.org/10.2217/fon.14.70
Keith B, Johnson RS, Simon MC (2011) HIF1α and HIF2α: Sibling rivalry in hypoxic tumor growth and progression. Nat Rev Cancer 12:9–22. https://doi.org/10.1038/nrc3183
Menrad H, Werno C, Schmid T, Copanaki E, Deller T, Dehne N, Brüne B (2010) Roles of hypoxia-inducible factor-1α (HIF-1α) versus HIF-2α in the survival of hepatocellular tumor spheroids. Hepatology 51:2183–2192. https://doi.org/10.1002/hep.23597
Mazure NM, Pouysségur J (2009) Atypical BH3-domains of BNIP3 and BNIP3L lead to autophagy in hypoxia. Autophagy 5:868–869. https://doi.org/10.4161/auto.9042
Leber B, Lin J, Andrews DW (2007) Embedded together: The life and death consequences of interaction of the Bcl-2 Family with membranes. Apoptosis 12:897–911. https://doi.org/10.1007/s10495-007-0746-4
Dong XF, Liu TQ, Zhi XT, Zou J, Zhong JT, Li T, Mo XL, Zhou W, Guo WW, Liu X, Chen YY, Li MY, Zhong XG, Han YM, Wang ZH, Dong ZR (2018) COX-2/PGE2 axis regulates HIF2a activity to promote hepatocellular carcinoma hypoxic response and reduce the sensitivity of sorafenib treatment. Clin Cancer Res 24:3204–3216. https://doi.org/10.1158/1078-0432.CCR-17-2725
Wu DH, Jia CC, Chen J, Lin ZX, Ruan DY, Li X, Lin Q, Min-Dong MXK, Wan XB, Cheng N, Chen ZH, Xing YF, Wu XY, Wen JY (2014) Autophagic LC3B overexpression correlates with malignant progression and predicts a poor prognosis in hepatocellular carcinoma. Tumour Biol 35:12225–12233. https://doi.org/10.1007/s13277-014-2531-7
Lazova R, Camp RL, Klump V, Siddiqui SF, Amaravadi RK, Pawelek JM (2012) Punctate LC3B expression is a common feature of solid tumors and associated with proliferation, metastasis and poor outcome. Clin Cancer Res 18:370–379. https://doi.org/10.1158/1078-0432.CCR-11-1282
Langley RR, Fidler IJ (2011) The seed and soil hypothesis revisited - the role of tumor-stroma interactions in metastasis to different organs. Int J Cancer 128:2527–2535. https://doi.org/10.1002/ijc.26031
Seyfried TN, Huysentruyt LC (2013) On the origin of cancer metastasis. Crit Rev Oncog 18:43–73. https://doi.org/10.1615/critrevoncog.v18.i1-2.40
Kim YN, Koo KH, Sung JY, Yun UJ, Kim H (2012) Anoikis resistance: an essential prerequisite for tumor metastasis. Int J Cell Biol 2012:306879. https://doi.org/10.1155/2012/306879
Paoli P, Giannoni E, Chiarugi P (2013) Anoikis molecular pathways and its role in cancer progression. Biochim Biophys Acta 1833:3481–3498. https://doi.org/10.1016/j.bbamcr.2013.06.026
Yang J, Zheng Z, Yan X, Li X, Liu Z, Ma Z (2013) Integration of autophagy and anoikis resistance in solid tumors. Anat Rec 296:1501–1508. https://doi.org/10.1002/ar.22769
Avivar-Valderas A, Salas E, Bobrovnikova-Marjon E, Diehl JA, Nagi C, Debnath J, Aguirre-Ghiso JA (2011) PERK integrates autophagy and oxidative stress responses to promote survival during extracellular matrix detachment. Mol Cell Biol 31:3616–3629. https://doi.org/10.1128/MCB.05164-11
Peng YF, Shi YH, Ding ZB, Ke AW, Gu CY, Hui B, Zhou J, Qiu SJ, Dai Z, Fan J (2013) Autophagy inhibition suppresses pulmonary metastasis of HCC in mice via impairing anoikis resistance and colonization of HCC cells. Autophagy 9:2056–2068. https://doi.org/10.4161/auto.26398
Kubiczkova L, Sedlarikova L, Hajek R, Sevcikova S (2012) TGF-β - an excellent servant but a bad master. J Transl Med 10:183. https://doi.org/10.1186/1479-5876-10-183
Scandura JM, Boccuni P, Massagué J, Nimer SD (2004) Transforming growth factor β-induced cell cycle arrest of human hematopoietic cells requires p57KIP2 up-regulation. Proc Natl Acad Sci U S A 101:15231–15236. https://doi.org/10.1073/pnas.0406771101
Inman GJ (2011) Switching TGFβ from a tumor suppressor to a tumor promoter. Curr Opin Genet Dev 21:93–99. https://doi.org/10.1016/j.gde.2010.12.004
Tong H, Yin H, Hossain MA, Wang Y, Wu F, Dong X, Gao S, Zhan K, He W (2019) Starvation-induced autophagy promotes the invasion and migration of human bladder cancer cells via TGF-β1/Smad3-mediated epithelial-mesenchymal transition activation. J Cell Biochem 120:5118–5127. https://doi.org/10.1002/jcb.27788
Li J, Yang B, Zhou Q, Wu Y, Shang D, Guo Y, Song Z, Zheng Q, Xiong J (2013) Autophagy promotes hepatocellular carcinoma cell invasion through activation of epithelial-mesenchymal transition. Carcinogenesis 34:1343–1351. https://doi.org/10.1093/carcin/bgt063
Roche J (2018) The epithelial-to-mesenchymal transition in cancer. Cancers (Basel) 10:52. https://doi.org/10.3390/cancers10020052
Wang J, Chen L, Li Y, Guan XY (2011) Overexpression of cathepsin Z contributes to tumor metastasis by inducing epithelial-mesenchymal transition in hepatocellular carcinoma. PLoS ONE 6:e24967. https://doi.org/10.1371/journal.pone.0024967
Chen HT, Liu H, Mao MJ, Tan Y, Mo XQ, Meng XJ, Cao MT, Zhong CY, Liu Y, Shan H, Jiang GM (2019) Crosstalk between autophagy and epithelial-mesenchymal transition and its application in cancer therapy. Mol Cancer 18:101. https://doi.org/10.1186/s12943-019-1030-2
Chen C, Liang QY, Chen HK, Wu PF, Feng ZY, Ma XM, Wu HR, Zhou GQ (2018) DRAM1 regulates the migration and invasion of hepatoblastoma cells via autophagy-EMT pathway. Oncol Lett 16:2427–2433. https://doi.org/10.3892/ol.2018.8937
Hu S, Wang L, Zhang X, Wu Y, Yang J, Li J (2018) Autophagy induces transforming growth factor-β-dependent epithelial-mesenchymal transition in hepatocarcinoma cells through cAMP response element binding signalling. J Cell Mol Med 22:5518–5532. https://doi.org/10.1111/jcmm.13825
Wang Y, Xiong H, Liu D, Hill C, Ertay A, Li J, Zou Y, Miller P, White E, Downward J, Goldin RD, Yuan X, Lu X (2019) Autophagy inhibition specifically promotes epithelial-mesenchymal transition and invasion in RAS-mutated cancer cells. Autophagy 15:886–899. https://doi.org/10.1080/15548627.2019.1569912
Zhao Z, Zhao J, Xue J, Zhao X, Liu P (2016) Autophagy inhibition promotes epithelial-mesenchymal transition through ROS/HO-1 pathway in ovarian cancer cells. Am J Cancer Res 6:2162–2177
Shaaban S, Negm A, Ibrahim EE, Elrazak AA (2014) Chemotherapeutic agents for the treatment of hepatocellular carcinoma: efficacy and mode of action. Oncol Rev 8:246. https://doi.org/10.4081/oncol.2014.246
Riddell IA, Lippard SJ (2018) Cisplatin and oxaliplatin: Our current understanding of their actions. In: Sigel A, Sigel H, Freisinger E, Sigel RKO (ed) Metallo-drugs: Development and action of anticancer agents, De Gruyter, Berlin Munich Boston, pp 1–42. https://doi.org/https://doi.org/10.1515/9783110470734-001
Chen R, Dai RY, Duan CY, Liu YP, Chen SK, Yan DM, Chen CN, Wei M, Li H (2011) Unfolded protein response suppresses cisplatin-induced apoptosis via autophagy regulation in human hepatocellular carcinoma cells. Folia Biol (Praha) 57:87–95
Du H, Yang W, Chen L, Shi M, Seewoo V, Wang J, Lin A, Liu Z, Qiu W (2012) Role of autophagy in resistance to oxaliplatin in hepatocellular carcinoma cells. Oncol Rep 27:143–150. https://doi.org/10.3892/or.2011.1464
Nitiss JL (2009) Targeting DNA topoisomerase II in cancer chemotherapy. Nat Rev Cancer 9:338–350. https://doi.org/10.1038/nrc2607
Zhou Y, Chen E, Tang Y, Mao J, Shen J, Zheng X, Xie S, Zhang S, Wu Y, Liu H, Zhi X, Ma T, Ni H, Chen J, Chai K, Chen W (2019) miR-223 overexpression inhibits doxorubicin-induced autophagy by targeting FOXO3a and reverses chemoresistance in hepatocellular carcinoma cells. Cell Death Dis 10:843. https://doi.org/10.1038/s41419-019-2053-8
Jin J, Huang M, Wei HL, Liu GT (2002) Mechanism of 5-fluorouracil required resistance in human hepatocellular carcinoma cell line Bel7402. World J Gastroenterol 8:1029–1034. https://doi.org/10.3748/wjg.v8.i6.1029
Guo XL, Hu F, Zhang SS, Zhao QD, Zong C, Ye F, Guo SW, Zhang JW, Li R, Wu MC, Wei LX (2014) Inhibition of p53 increases chemosensitivity to 5-FU in nutrient-deprived hepatocarcinoma cells by suppressing autophagy. Cancer Lett 346:278–284. https://doi.org/10.1016/j.canlet.2014.01.011
Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, Schwartz M, Porta C, Zeuzem S, Bolondi L, Greten TF, Galle PR, Seitz JF, Borbath I, Häussinger D, Giannaris T, Shan M, Moscovici M, Voliotis D, Bruix J, SHARP Investigators Study Group (2008) Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 359:378–390. https://doi.org/10.1056/NEJMoa0708857
Fu XT, Song K, Zhou J, Shi YH, Liu WR, Tian MX, Jin L, Shi GM, Gao Q, Ding ZB, Fan J (2018) Autophagy activation contributes to glutathione transferase Mu 1-mediated chemoresistance in hepatocellular carcinoma. Oncol Lett 16:346–352. https://doi.org/10.3892/ol.2018.8667
Zhai B, Hu F, Jiang X, Xu J, Zhao D, Liu B, Pan S, Dong X, Tan G, Wei Z, Qiao H, Jiang H, Sun X (2014) Inhibition of Akt reverses the acquired resistance to sorafenib by switching protective autophagy to autophagic cell death in hepatocellular carcinoma. Mol Cancer Ther 13:1589–1598. https://doi.org/10.1158/1535-7163.MCT-13-1043
Zhao D, Zhai B, He C, Tan G, Jiang X, Pan S, Dong X, Wei Z, Ma L, Qiao H, Jiang H, Sun X (2014) Upregulation of HIF-2α induced by sorafenib contributes to the resistance by activating the TGF-α/EGFR pathway in hepatocellular carcinoma cells. Cell Signal 26:1030–1039. https://doi.org/10.1016/j.cellsig.2014.01.026
Liang Y, Zheng T, Song R, Wang J, Yin D, Wang L, Liu H, Tian L, Fang X, Meng X, Jiang H, Liu J, Liu L (2013) Hypoxia-mediated sorafenib resistance can be overcome by EF24 through Von Hippel-Lindau tumor suppressor-dependent HIF-1α inhibition in hepatocellular carcinoma. Hepatology 57:1847–1857. https://doi.org/10.1002/hep.26224
Tang W, Chen Z, Zhang W, Cheng Y, Zhang B, Wu F, Wang Q, Wang S, Rong D, Reiter FP, De Toni EN, Wang X (2020) The mechanisms of sorafenib resistance in hepatocellular carcinoma: theoretical basis and therapeutic aspects. Signal Transduct Target Ther 5:87. https://doi.org/10.1038/s41392-020-0187-x
Peng WX, Xiong EM, Ge L, Wan YY, Zhang CL, Du FY, Xu M, Bhat RA, Jin J, Gong AH (2016) Egr-1 promotes hypoxia-induced autophagy to enhance chemo-resistance of hepatocellular carcinoma cells. Exp Cell Res 340:62–70. https://doi.org/10.1016/j.yexcr.2015.12.006
Xu Y, An Y, Wang Y, Zhang C, Zhang H, Huang C, Jiang H, Wang X, Li X (2013) miR-101 inhibits autophagy and enhances cisplatin-induced apoptosis in hepatocellular carcinoma cells. Oncol Rep 29:2019–2024. https://doi.org/10.3892/or.2013.2338
Lanza E, Donadon M, Poretti D, Pedicini V, Tramarin M, Roncalli M, Rhee H, Park YN, Torzilli G (2017) Transarterial therapies for hepatocellular carcinoma. Liver Cancer 6:27–33. https://doi.org/10.1159/000449347
Pleguezuelo M, Marelli L, Misseri M, Germani G, Calvaruso V, Xiruochakis E, Manousou P, Burroughs AK (2008) TACE versus TAE as therapy for hepatocellular carcinoma. Expert Rev Anticancer Ther 8:1623–1641. https://doi.org/10.1586/14737140.8.10.1623
Rammohan A, Sathyanesan J, Ramaswami S, Lakshmanan A, Senthil-Kumar P, Srinivasan UP, Ramasamy R, Ravichandran P (2012) Embolization of liver tumors: past, present and future. World J Radiol 4:405–412. https://doi.org/10.4329/wjr.v4.i9.405
Gade TPF, Tucker E, Nakazawa MS, Hunt SJ, Wong W, Krock B, Weber CN, Nadolski GJ, Clark TWI, Soulen MC, Furth EE, Winkler JD, Amaravadi RK, Simon MC (2017) Ischemia induces quiescence and autophagy dependence in hepatocellular carcinoma. Radiology 283:702–710. https://doi.org/10.1148/radiol.2017160728
Tao W, Shi JF, Zhang Q, Xue B, Sun YJ, Li CJ (2013) Egr-1 enhances drug resistance of breast cancer by modulating MDR1 expression in a GGPPS-independent manner. Biomed Pharmacother 67:197–202. https://doi.org/10.1016/j.biopha.2013.01.001
Peng WX, Wan YY, Gong AH, Ge L, Jin J, Xu M, Wu CY (2017) Egr-1 regulates irradiation-induced autophagy through Atg4B to promote radioresistance in hepatocellular carcinoma cells. Oncogenesis 6:e292. https://doi.org/10.1038/oncsis.2016.91
Yang Z, Wilkie-Grantham RP, Yanagi T, Shu CW, Matsuzawa SI, Reed JC (2015) ATG4B (Autophagin-1) phosphorylation modulates autophagy. J Biol Chem 290:26549–26561. https://doi.org/10.1074/jbc.M115.658088
Wang Z, Han W, Sui X, Fang Y, Pan H (2014) Autophagy: a novel therapeutic target for hepatocarcinoma (Review). Oncol Lett 7:1345–1351. https://doi.org/10.3892/ol.2014.1916
Sun T, Liu H, Ming L (2017) Multiple roles of autophagy in the sorafenib resistance of hepatocellular carcinoma. Cell Physiol Biochem 44:716–727. https://doi.org/10.1159/000485285
Shi YH, Ding ZB, Zhou J, Hui B, Shi GM, Ke AW, Wang XY, Dai Z, Peng YF, Gu CY, Qiu SJ, Fan J (2011) Targeting autophagy enhances sorafenib lethality for hepatocellular carcinoma via ER stress-related apoptosis. Autophagy 7:1159–1172. https://doi.org/10.4161/auto.7.10.16818
Prieto-Vila M, Takahashi RU, Usuba W, Kohama I, Ochiya T (2017) Drug resistance driven by cancer stem cells and their niche. Int J Mol Sci 18:2574. https://doi.org/10.3390/ijms18122574
Nio K, Yamashita T, Kaneko S (2017) The evolving concept of liver cancer stem cells. Mol Cancer 16:4. https://doi.org/10.1186/s12943-016-0572-9
Yamashita T, Wang XW (2013) Cancer stem cells in the development of liver cancer. J Clin Invest 123:1911–1918. https://doi.org/10.1172/JCI66024
Yamashita T, Ji J, Budhu A, Forgues M, Yang W, Wang HY, Jia H, Ye Q, Qin LX, Wauthier E, Reid LM, Minato H, Honda M, Kaneko S, Tang ZY, Wang XW (2009) EpCAM-positive hepatocellular carcinoma cells are tumor initiating cells with stem/progenitor cell features. Gastroenterology 136:1012–1024. https://doi.org/10.1053/j.gastro.2008.12.004
Terris B, Cavard C, Perret C (2010) EpCAM, a new marker for cancer stem cells in hepatocellular carcinoma. J Hepatol 52:280–281. https://doi.org/10.1016/j.jhep.2009.10.026
Ma S, Chan KW, Hu L, Lee TKW, Wo JYH, Ng IOL, Zheng BJ, Guan XY (2007) Identification and characterization of tumorigenic liver cancer stem/progenitor cells. Gastroenterology 132:2542–2556. https://doi.org/10.1053/j.gastro.2007.04.025
Zhu Z, Hao X, Yan M, Yao M, Ge C, Gu J, Li J (2010) Cancer stem/progenitor cells are highly enriched in CD133 +CD44+ population in hepatocellular carcinoma. Int J Cancer 126:2067–2078. https://doi.org/10.1002/ijc.24868
Yang FZ, Ngai P, Ho DW, Yu WC, Ng MNP, Lau CK, Li MLY, Tam KH, Lam CT, Poon RTP, Fan ST (2008) Identification of local and circulating cancer stem cells in human liver cancer. Hepatology 47:919–928. https://doi.org/10.1002/hep.22082
Haraguchi N, Ishii H, Mimori K, Tanaka F, Ohkuma M, Kim HM, Akita H, Takiuchi D, Hatano H, Nagano H, Barnard GF, Doki Y, Mori M (2010) CD13 is a therapeutic target in human liver cancer stem cells. J Clin Invest 120:3326–3339. https://doi.org/10.1172/JCI42550
Qiu L, Li H, Fu S, Chen X, Lu L (2018) Surface markers of liver cancer stem cells and innovative targeted-therapy strategies for HCC. Oncol lett 15:2039–2048. https://doi.org/10.3892/ol.2017.7568
Govaere O, Komuta M, Berkers J, Spee B, Janssen C, de Luca F, Katoonizadeh A, Wouters J, van Kempen LC, Durnez A, Verslype C, De Kock J, Rogiers V, van Grunsven LA, Topal B, Pirenne J, Vankelecom H, Nevens F, van den Oord J, Pinzani M, Roskams T (2014) Keratin 19: a key role player in the invasion of human hepatocellular carcinomas. Gut 63:674–685. https://doi.org/10.1136/gutjnl-2012-304351
Zheng H, Pomyen Y, Hernandez MO, Li C, Livak F, Tang W, Dang H, Greten TF, Davis JL, Zhao Y, Mehta M, Levin Y, Shetty J, Tran B, Budhu A, Wang XW (2018) Single cell analysis reveals cancer stem cell heterogeneity in hepatocellular carcinoma. Hepatalogy 68:127–140. https://doi.org/10.1002/hep.29778
Magee JA, Piskounova E, Morrison SJ (2012) Cancer stem cells: Impact, heterogeneity, and uncertainty. Cancer Cell 21:283–296. https://doi.org/10.1016/j.ccr.2012.03.003
Reya T, Clevers H (2005) Wnt signalling in stem cells and cancer. Nature 434:843–850. https://doi.org/10.1038/nature03319
Weinmaster G, Kopan R (2016) A garden of Notch-ly delights. Development 133:3277–3282. https://doi.org/10.1242/dev.02515
Gotoh N (2009) Control of stemness by fibroblast growth factor signaling in stem cells and cancer stem cells. Curr Stem Cell Res Ther 4:9–15. https://doi.org/10.2174/157488809787169048
Derynck R, Zhang YE (2003) Smad-dependent and Smad-independent pathways in TGF-β family signalling. Nature 425:577–584. https://doi.org/10.1038/nature02006
Sell S (2010) On the stem cell origin of cancer. Am J Pathol 176:2584–2594. https://doi.org/10.2353/ajpath.2010.091064
Lau EYT, Ho NPY, Lee TKW (2017) Cancer stem cells and their microenvironment: biology and therapeutic implications. Stem Cells Int 2017:3714190. https://doi.org/10.1155/2017/3714190
Gao Y, Ruan B, Liu W, Wang J, Yang X, Zhang Z, Li X, Duan J, Zhang F, Ding R, Tao K, Dou K (2015) Knockdown of CD44 inhibits the invasion and metastasis of hepatocellular carcinoma both in vitro and in vivo by reversing epithelial-mesenchymal transition. Oncotarget 6:7828–7837. https://doi.org/10.18632/oncotarget.3488
van Zijl F, Zulehner G, Petz M, Schneller D, Kornauth C, Hau M, Machat G, Grubinger M, Huber H, Mikulits W (2009) Epithelial-mesenchymal transition in hepatocellular carcinoma. Futur Oncol 5:1169–1179. https://doi.org/10.2217/fon.09.91
Cazet AS, Hui MN, Elsworth BL, Wu SZ, Roden D, Chan CL, Skhinas JN, Collot R, Yang J, Harvey K, Johan MZ, Cooper C, Nair R, Herrmann D, McFarland A, Deng N, Ruiz-Borrego M, Rojo F, Trigo JM, Bezares S, Caballero R, Lim E, Timpson P, O’Toole S, Watkins DN, Cox TR, Samuel MS, Martín M, Swarbrick A (2018) Targeting stromal remodeling and cancer stem cell plasticity overcomes chemoresistance in triple negative breast cancer. Nat Commun 9:2897. https://doi.org/10.1038/s41467-018-05220-6
Dai XM, Yang SL, Zheng XM, Chen GG, Chen J, Zhang T (2018) CD133 expression and α-fetoprotein levels define novel prognostic subtypes of HBV-associated hepatocellular carcinoma: A long-term follow-up analysis. Oncol Lett 15:2985–2991. https://doi.org/10.3892/ol.2017.7704
Yamanaka C, Wada H, Eguchi H, Hatano H, Gotoh K, Noda T, Yamada D, Asaoka T, Kawamoto K, Nagano H, Doki Y, Mori M (2018) Clinical significance of CD13 and epithelial mesenchymal transition (EMT) markers in hepatocellular carcinoma. Jpn J Clin Oncol 48:52–60. https://doi.org/10.1093/jjco/hyx157
Batlle E, Clevers H (2017) Cancer stem cells revisited. Nat Med 23:1124–1134. https://doi.org/10.1038/nm.4409
Trédan O, Galmarini CM, Patel K, Tannock IF (2007) Drug resistance and the solid tumor microenvironment. J Natl Cancer Inst 99:1441–1454. https://doi.org/10.1093/jnci/djm135
Baumann M, Krause M, Hill R (2008) Exploring the role of cancer stem cells in radioresistance. Nat Rev Cancer 8:545–554. https://doi.org/10.1038/nrc2419
Bai X, Ni J, Beretov J, Graham P, Li Y (2018) Cancer stem cell in breast cancer therapeutic resistance. Cancer Treat Rev 69:152–163. https://doi.org/10.1016/j.ctrv.2018.07.004
Sun YL, Patel A, Kumar P, Chen ZS (2012) Role of ABC transporters in cancer chemotherapy. Chin J Cancer 31:51–57. https://doi.org/10.5732/cjc.011.10466
Zhang G, Wang Z, Luo W, Jiao H, Wu J, Jiang C (2013) Expression of potential cancer stem cell marker ABCG2 is associated with malignant behaviors of hepatocellular carcinoma. Gastroenterol Res Pract 2013:782581. https://doi.org/10.1155/2013/782581
Jia Q, Zhang X, Deng T, Gao J (2013) Positive correlation of Oct4 and ABCG2 to chemotherapeutic resistance in CD90+CD133+ liver cancer stem cells. Cell Reprogram 15:143–150. https://doi.org/10.1089/cell.2012.0048
Ranji P, T. Salmani Kesejini T, Saeedikhoo S, Alizadeh AM, (2016) Targeting cancer stem cell-specific markers and/or associated signaling pathways for overcoming cancer drug resistance. Tumor Biol 37:13059–13075. https://doi.org/10.1007/s13277-016-5294-5
Vassalli G (2019) Aldehyde dehydrogenases: Not just markers, but functional regulators of stem cells. Stem Cells Int 2019:3904645. https://doi.org/10.1155/2019/3904645
Carnero A, Garcia-Mayea Y, Mir C, Lorente J, RubioLLeonart ITME (2016) The cancer stem-cell signaling network and resistance to therapy. Cancer Treat Rev 49:25–36. https://doi.org/10.1016/j.ctrv.2016.07.001
Tsai LL, Yu CC, Lo JF, Sung WW, Lee H, Chen SL, Chou MY (2012) Enhanced cisplatin resistance in oral-cancer stem-like cells is correlated with upregulation of excision-repair cross-complementation group 1. J Dent Sci 7:111–117. https://doi.org/10.1016/j.jds.2012.03.006
Kim NH, Kim HS, Li XY, Lee I, Choi HS, Kang SE, Cha SY, Ryu JK, Yoon D, Fearon ER, Rowe RG, Lee S, Maher CA, Weiss SJ, Yook JI (2011) A p53/miRNA-34 axis regulates Snail1-dependent cancer cell epithelial-mesenchymal transition. J Cell Biol 195:417–433. https://doi.org/10.1083/jcb.201103097
Carafa V, Rotili D, Forgione M, Cuomo F, Serretiello E, Hailu GS, Jarho E, Lahtela-Kakkonen M, Mai A, Altucci L (2016) Sirtuin functions and modulation: from chemistry to the clinic. Clin Epigenetics 8:61. https://doi.org/10.1186/s13148-016-0224-3
Michan S, Sinclair D (2007) Sirtuins in mammals: Insights into their biological function. Biochem J 404:1–13. https://doi.org/10.1042/BJ20070140
Ong ALC, Ramasamy TS (2018) Role of Sirtuin1-p53 regulatory axis in aging, cancer and cellular reprogramming. Ageing Res Rev 43:64–80. https://doi.org/10.1016/j.arr.2018.02.004
O’Callaghan C, Vassilopoulos A (2017) Sirtuins at the crossroads of stemness, aging, and cancer. Aging Cell 16:1208–1218. https://doi.org/10.1111/acel.12685
Liu T, Liu PY, Marshall GM (2009) The critical role of the class III histone deacetylase SIRT1 in cancer. Cancer Res 69:1702–1705. https://doi.org/10.1158/0008-5472.CAN-08-3365
Wang RH, Sengupta K, Li C, Kim HS, Cao L, Xiao C, Kim S, Xu X, Zheng Y, Chilton B, Jia R, Zheng ZM, Appella E, Wang XW, Reid T, Deng CX (2008) Impaired DNA damage response, genome instability, and tumorigenesis in SIRT1 mutant mice. Cancer Cell 14:312–323. https://doi.org/10.1016/j.ccr.2008.09.001
Farcas M, Gavrea AA, Gulei D, Ionescu C, Irimie A, Catana CS, Berindan-Neagoe I (2019) SIRT1 in the development and treatment of hepatocellular carcinoma. Front Nutr 6:148. https://doi.org/10.3389/fnut.2019.00148
Chen X, Huan H, Liu C, Luo Y, Shen J, Zhuo Y, Zhang Z, Qian C (2019) Deacetylation of β-catenin by SIRT1 regulates self-renewal and oncogenesis of liver cancer stem cells. Cancer Lett 463:1–10. https://doi.org/10.1016/j.canlet.2019.07.021
Kopan R (2012) Notch signaling. Cold Spring Harb Perspect Biol 4:a011213. https://doi.org/10.1101/cshperspect.a011213
Qi R, An H, Yu Y, Zhang M, Liu S, Xu H, Guo Z, Cheng T, Cao X (2003) Notch1 signaling inhibits growth of human hepatocellular carcinoma through induction of cell cycle arrest and apoptosis. Cancer Res 63:8323–8329
Ning L, Wentworth L, Chen H, Weber SM (2009) Down-regulation of Notch1 signaling inhibits tumor growth in human hepatocellular carcinoma. Am J Transl Res 1:358–366
Liu L, Liu C, Zhang Q, Shen J, Zhang H, Shan J, Duan G, Guo D, Chen X, Cheng J, Xu Y, Yang Z, Yao C, Lai M, Qian C (2016) SIRT1-mediated transcriptional regulation of SOX2 is important for self-renewal of liver cancer stem cells. Hepatology 64:814–827. https://doi.org/10.1002/hep.28690
Zhang S, Xiong X, Sun Y (2020) Functional characterization of SOX2 as an anticancer target. Sig Transduct Target Ther 5:135. https://doi.org/10.1038/s41392-020-00242-3
Kim HJ, Maiti P, Barrientos A (2017) Mitochondrial ribosomes in cancer. Semin Cancer Biol 47:67–81. https://doi.org/10.1016/j.semcancer.2017.04.004
Cheng J, Liu C, Liu L, Chen X, Shan J, Shen J, Zhu W, Qian C (2016) MEK1 signaling promotes self-renewal and tumorigenicity of liver cancer stem cells via maintaining SIRT1 protein stabilization. Oncotarget 7:20597–20611. https://doi.org/10.18632/oncotarget.7972
Pearson G, Robinson F, Gibson TB, Xu BE, Karandikar M, Berman K, Cobb MH (2001) Mitogen-activated protein (MAP) kinase pathways: Regulation and physiological functions. Endocr Rev 22:153–183. https://doi.org/10.1210/edrv.22.2.0428
Mortensen M, Simon AK (2010) Nonredundant role of Atg7 in mitochondrial clearance during erythroid development. Autophagy 6:423–425. https://doi.org/10.4161/auto.6.3.11528
Miyajima A, Tanaka M, Itoh T (2014) Stem/progenitor cells in liver development, homeostasis, regeneration, and reprogramming. Cell Stem Cell 14:561–574. https://doi.org/10.1016/j.stem.2014.04.010
Cheng Y, Wang B, Zhou H, Dang S, Jin M, Shi Y, Hao L, Yang Z, Zhang Y (2015) Autophagy is required for the maintenance of liver progenitor cell functionality. Cell Physiol Biochem 36:1163–1174. https://doi.org/10.1159/000430287
Xue F, Hu L, Ge R, Yang L, Liu K, Li Y, Sun Y, Wang K (2016) Autophagy-deficiency in hepatic progenitor cells leads to the defects of stemness and enhances susceptibility to neoplastic transformation. Cancer Lett 371:38–47. https://doi.org/10.1016/j.canlet.2015.11.022
Bu Y, Cao D (2012) The origin of cancer stem cells. Front Biosci (Schol Ed) 4:819–830. https://doi.org/10.2741/s302
Zender L, Spector MS, Xue W, Flemming P, Cordon-Cardo C, Silke J, Fan ST, Luk JM, Wigler M, Hannon GJ, Mu D, Lucito R, Powers S, Lowe SW (2006) Identification and validation of oncogenes in liver cancer using an integrative oncogenomic approach. Cell 125:1253–1267. https://doi.org/10.1016/j.cell.2006.05.030
Tang Y, Kitisin K, Jogunoori W, Li C, Deng CX, Mueller SC, Ressom HW, Rashid A, He AR, Mendelson JS, Jessup JM, Shetty K, Zasloff M, Mishra B, Reddy EP, Johnson L, Mishra L (2008) Progenitor/stem cells give rise to liver cancer due to aberrant TGF-beta and IL-6 signaling. Proc Natl Acad Sci U S A 105:2445–2450. https://doi.org/10.1073/pnas.0705395105
Liu K, Lee J, Kim JY, Wang L, Tian Y, Chan ST, Cho C, Machida K, Chen D, Ou JHJ (2017) Mitophagy controls the activities of tumor suppressor p53 to regulate hepatic cancer stem cells. Mol Cell 68:281-292.e5. https://doi.org/10.1016/j.molcel.2017.09.022
Li J, Hu SB, Wang LY, Zhang X, Zhou X, Yang B, Li JH, Xiong J, Liu N, Li Y, Wu YZ, Zheng QC (2017) Autophagy-dependent generation of Axin2+ cancer stem-like cells promotes hepatocarcinogenesis in liver cirrhosis. Oncogene 36:6725–6737. https://doi.org/10.1038/onc.2017.272
Song YJ, Zhang SS, Guo XL, Sun K, Han ZP, Li R, Zhao QD, Deng WJ, Xie XQ, Zhang JW, Wu MC, Wei LX (2013) Autophagy contributes to the survival of CD133+ liver cancer stem cells in the hypoxic and nutrient-deprived tumor microenvironment. Cancer Lett 339:70–81. https://doi.org/10.1016/j.canlet.2013.07.021
Li Z, Jiao X, Di Sante G, Ertel A, Casimiro MC, Wang M, Katiyar S, Ju X, Klopfenstein DV, Tozeren A, Dampier W, Chepelev I, Jeltsch A, Pestell RG (2019) Cyclin D1 integrates G9a-mediated histone methylation. Oncogene 38:4232–4249. https://doi.org/10.1038/s41388-019-0723-8
Zheng N, Wei W, Wang Z (2016) Emerging roles of FGF signaling in hepatocellular carcinoma. Transl Cancer Res 5:1–6
Ocker M (2020) Fibroblast growth factor signaling in non-alcoholic fatty liver disease and non-alcoholic steatohepatitis: paving the way to hepatocellular carcinoma. World J Gastroenterol 26:279–290. https://doi.org/10.3748/wjg.v26.i3.279
Gauglhofer C, Sagmeister S, Schrottmaier W, Fischer C, Rodgarkia-Dara C, Mohr T, Stättner S, Bichler C, Kandioler D, Wrba F, Schulte-Hermann R, Holzmann K, Grusch M, Marian B, Berger W, Grasl-Kraupp B (2011) Up-regulation of the fibroblast growth factor 8 subfamily in human hepatocellular carcinoma for cell survival and neoangiogenesis. Hepatology 53:854–864. https://doi.org/10.1002/hep.24099
Sawey ET, Chanrion M, Cai C, Wu G, Zhang J, Zender L, Zhao A, Busuttil RW, Yee H, Stein L, French DM, Finn RS, Lowe SW, Powers S (2011) Identification of a therapeutic strategy targeting amplified FGF19 in liver cancer by oncogenomic screening. Cancer Cell 19:347–358. https://doi.org/10.1016/j.ccr.2011.01.040
Hagel M, Miduturu C, Sheets M, Rubin N, Weng W, Stransky N, Bifulco N, Kim JL, Hodous B, Brooijmans N, Shutes A, Winter C, Lengauer C, Kohl NE, Guzi T (2015) First selective small molecule inhibitor of FGFR4 for the treatment of hepatocellular carcinomas with an activated FGFR4 signaling pathway. Cancer Discov 5:424–437. https://doi.org/10.1158/2159-8290.CD-14-1029
French DM, Lin BC, Wang M, Adams C, Shek T, Hötzel K, Bolon B, Ferrando R, Blackmore C, Schroeder K, Rodriguez LA, Hristopoulos M, Venook R, Ashkenazi A, Desnoyers LR (2012) Targeting FGFR4 inhibits hepatocellular carcinoma in preclinical mouse models. PLoS ONE 7:e36713. https://doi.org/10.1371/journal.pone.0036713
Mavila N, James D, Utley S, Cu N, Coblens O, Mak K, Rountree CB, Kahn M, Wang KS (2012) Fibroblast growth factor receptor-mediated activation of AKT-β-catenin-CBP pathway regulates survival and proliferation of murine hepatoblasts and hepatic tumor initiating stem cells. PLoS ONE 7:e50401. https://doi.org/10.1371/journal.pone.0050401
Zhang J, Liu J, Liu L, McKeehan WL, Wang F (2012) The fibroblast growth factor signaling axis controls cardiac stem cell differentiation through regulating autophagy. Autophagy 8:690–691. https://doi.org/10.4161/auto.19290
Cinque L, Forrester A, Bartolomeo R, Svelto M, Venditti R, Montefusco S, Polishchuk E, Nusco E, Rossi A, Medina DL, Polishchuk R, De Matteis MA, Settembre C (2015) FGF signalling regulates bone growth through autophagy. Nature 528:272–275. https://doi.org/10.1038/nature16063
Yuan H, Li ZM, Shao J, Ji WX, Xia W, Lu S (2017) FGF2/FGFR1 regulates autophagy in FGFR1-amplified non-small cell lung cancer cells. J Exp Clin Cancer Res 36:72. https://doi.org/10.1186/s13046-017-0534-0
Kiyono K, Suzuki HI, Matsuyama H, Morishita Y, Komuro A, Kano MR, Sugimoto K, Miyazono K (2009) Autophagy is activated by TGF-β and potentiates TGF-β-mediated growth inhibition in human hepatocellular carcinoma cells. Cancer Res 69:8844–8852. https://doi.org/10.1158/0008-5472.CAN-08-4401
Ma CL, Qiao S, Li YC, Wang XF, Sun RJ, Zhang X, Qian RK, Song SD (2017) TGF-β1 promotes human hepatic carcinoma HepG2 cells invasion by upregulating autophagy. Eur Rev Med Pharmacol Sci 21:2604–2610
Zong Y, Panikkar A, Xu J, Antoniou A, Raynaud P, Lemaigre F, Stanger BZ (2009) Notch signaling controls liver development by regulating biliary differentiation. Development 136:1727–1739. https://doi.org/10.1242/dev.029140
Zeng J, Jing Y, Shi R, Pan X, Lai F, Liu W, Li R, Gao L, Hou X, Wu M, Wei L (2016) Autophagy regulates biliary differentiation of hepatic progenitor cells through Notch1 signaling pathway. Cell Cycle 15:1602–1610. https://doi.org/10.1080/15384101.2016.1181234
Wu X, Fleming A, Ricketts T, Pavel M, Virgin H, Menzies FM, Rubinsztein DC (2016) Autophagy regulates Notch degradation and modulates stem cell development and neurogenesis. Nat Commun 7:10533. https://doi.org/10.1038/ncomms10533
Vujovic F, Hunter N, Farahani RM (2019) Notch pathway: a bistable inducer of biological noise? Cell Commun Signal 17:133. https://doi.org/10.1186/s12964-019-0453-0
Wang M, Xue L, Cao Q, Lin Y, Ding Y, Yang P, Che L (2009) Expression of Notch1, Jagged1 and beta-catenin and their clinicopathological significance in hepatocellular carcinoma. Neoplasma 56:533–541. https://doi.org/10.4149/neo_2009_06_533
Croquelois A, Blindenbacher A, Terracciano L, Wang X, Langer I, Radtke F, Heim MH (2005) Inducible inactivation of Notch1 causes nodular regenerative hyperplasia in mice. Hepatology 41:487–496. https://doi.org/10.1002/hep.20571
Khalaf AM, Fuentes D, Morshid AI, Burke MR, Kaseb AO, Hassan M, Hazle JD, Elsayes KM (2018) Role of Wnt/β-catenin signaling in hepatocellular carcinoma, pathogenesis, and clinical significance. J Hepatocell Carcinoma 5:61–73. https://doi.org/10.2147/JHC.S156701
Turcios L, Chacon E, Garcia C, Eman P, Cornea V, Jiang J, Spear B, Liu C, Watt DS, Marti F, Gedaly R (2019) Autophagic flux modulation by Wnt/β-catenin pathway inhibition in hepatocellular carcinoma. PLoS One 14:e0212538. https://doi.org/10.1371/journal.pone.0212538
Petherick KJ, Williams AC, Lane JD, Ordóñez-Morán P, Huelsken J, Collard TJ, Smartt HJM, Batson J, Malik K, Paraskeva C, Greenhough A (2013) Autolysosomal β-catenin degradation regulates Wnt-autophagy-p62 crosstalk. EMBO J 32:1903–1916. https://doi.org/10.1038/emboj.2013.123
Fan Q, Yang L, Zhang X, Ma Y, Li Y, Dong L, Zong Z, Hua X, Su D, Li H, Liu J (2018) Autophagy promotes metastasis and glycolysis by upregulating MCT1 expression and Wnt/β-catenin signaling pathway activation in hepatocellular carcinoma cells. J Exp Clin Cancer Res 37:9. https://doi.org/10.1186/s13046-018-0673-y
Ma Z, Li F, Chen L, Gu T, Zhang Q, Qu Y, Xu M, Cai X, Lu L (2019) Autophagy promotes hepatic differentiation of hepatic progenitor cells by regulating the Wnt/β-catenin signaling pathway. J Mol Histol 50:75–90. https://doi.org/10.1007/s10735-018-9808-x
Kim JY, Lee HY, Park KK, Choi YK, Nam JS, Hong IS (2016) CWP232228 targets liver cancer stem cells through Wnt/β-catenin signaling: A novel therapeutic approach for liver cancer treatment. Oncotarget 7:20395–20409. https://doi.org/10.18632/oncotarget.7954
Bhat P, Kriel J, Shubha Priya B, Basappa SNS, Loos B (2018) Modulating autophagy in cancer therapy: advancements and challenges for cancer cell death sensitization. Biochem Pharmacol 147:170–182. https://doi.org/10.1016/j.bcp.2017.11.021
Galluzzi L, Bravo-San Pedro JM, Levine B, Green DR, Kroemer G (2017) Pharmacological modulation of autophagy: Therapeutic potential and persisting obstacles. Nat Rev Drug Discov 16:487–511. https://doi.org/10.1038/nrd.2017.22
Acknowledgements
We would like to thank Sunway University Postgraduate Degree by Research Scholarship for supporting M. M-.T.W. and H-.Y.C. This work was partly funded by Ministry of Higher Education Malaysia (FRGS/1/2019/SKK08/SYUC/02/1); and Sunway Medical Centre Research Funds (SRC/002/2017/FR and SRC/003/ 2017/FR).
Author information
Authors and Affiliations
Contributions
SYT, TSR and SCP conceived and designed the analysis, MMTW and HYC wrote the paper, JJB, NAA, ESC and SA reviewed and edited the paper. All authors have read and agreed to the manuscript submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wong, M.MT., Chan, HY., Aziz, N.A. et al. Interplay of autophagy and cancer stem cells in hepatocellular carcinoma. Mol Biol Rep 48, 3695–3717 (2021). https://doi.org/10.1007/s11033-021-06334-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06334-9