Abstract
Context
Habitat fragmentation can exacerbate the negative effects of habitat loss for some species. Mitigating fragmentation is difficult, however, because population responses depend on species-level traits (e.g., dispersal ability, edge sensitivity) and landscape context (e.g., habitat amount). Thus, conservation requires determining not only if, but also where and why, fragmentation matters.
Objectives
We aimed to determine if and where grassland fragmentation affects tallgrass prairie birds, which have declined precipitously due to land-use change. We surveyed four edge-sensitive species at 2250 sites (10,291 total surveys) across eastern Kansas, USA, over two breeding seasons. We assessed how the occurrence of each species varied with different levels of fragmentation in local landscapes comprising different grassland amounts.
Results
Fragmentation clearly mediated positive relationships between occurrence probability and grassland area for all four species. The direct effect of fragmentation was greater than that of grassland area for two species. Moreover, fragmentation reduced the occurrence of each species by at least half in some contexts. Fragmentation effects were most pronounced in landscapes comprising ~ 50–90% grassland, and less pronounced or absent in landscapes comprising < 50% grassland, which were occupied relatively infrequently.
Conclusions
Conservation efforts should minimize ‘perforation’ of large grasslands by woody vegetation and land development, which not only replace grassland, but also often create disproportionately large amounts of grassland edge. Identifying mechanisms responsible for edge effects could further inform species-level conservation. Our results counter assertions that fragmentation does not matter or only matters when habitat is scarce or for species that are dispersal limited.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Humans have “severely altered” more than three-quarters of Earth’s terrestrial environments and now use more than one-third of land surface for crop or livestock production (IPBES 2019). Temperate grasslands and savannas have been particularly hard hit; globally, nearly half (46%) have been replaced by crop agriculture and other land uses (Hoekstra et al. 2005). In North America, more than 97% of tallgrass prairie—an ecosystem that once covered 160 million hectares throughout the Great Plains—has been lost (Samson and Knopf 1994), and most of what remains is either managed intensively for cattle production or unmanaged (With et al. 2008; Rahmig et al. 2009). Shrubs and trees are encroaching on unmanaged grasslands due to fire suppression, climate change, and surrounding land development (Briggs et al. 2005; Kulmatiski and Beard 2013; Ratajczak et al. 2014; Scholtz et al. 2018). Additionally, ever-increasing energy demands mean that oil and gas wells and wind turbines are now common within grasslands (Hovick et al. 2014; Shaffer and Buhl 2016). Only 2% of remaining native grasslands in the USA are publicly owned and managed for conservation (NABCI 2011), reflecting the generally low level of protection that temperate grasslands receive worldwide (Hoekstra et al. 2005).
The widespread loss and fragmentation (e.g., increased edge-to-area ratio; Ewers and Didham 2006; Hargreaves 2019) of grasslands has caused drastic declines of grassland-dependent taxa (Samson and Knopf 1994; Brennan and Kuvlesky 2005; Azpiroz et al. 2012; Grand et al. 2019). Grassland birds have declined more rapidly than any other bird group in North America, and one-third of all grassland bird species are of conservation concern (NABCI 2016). Many grassland bird species have large habitat area requirements and thus do not occur in landscapes without expansive grassland (Ribic et al. 2009; Patten et al. 2011; Winder et al. 2014). Grassland birds also often avoid edges of grassland patches, particularly near woodlands or energy facilities, where they may experience greater risk of adult mortality (Hovick et al. 2014), nest depredation (With 1994; Renfrew and Ribic 2003; Klug et al. 2010), and/or brood parasitism by Brown-headed Cowbirds (Molothrus ater) (Winter et al. 2000). Likewise, empirical studies have found that the occurrence and abundance of some tallgrass prairie birds correlate more strongly with interior or ‘core’ grassland area away from edges than total grassland area per se (e.g., Helzer and Jelinksi 1999; Renfrew and Ribic 2008; Herse et al. 2018). However, few of these studies have assessed the importance of fragmentation while accounting for habitat amount (i.e., area), which could further inform management decisions about when to prioritize increasing total habitat area versus increasing core habitat area (Collinge 2009; Herse et al. 2018).
Empirical studies of population and community responses to habitat amount and fragmentation have mostly focused on determining which of these two landscape factors has a greater overall effect (Fahrig 2003, 2017; Hadley and Betts 2016), and in recent years, divergent views have emerged regarding the importance of fragmentation (Didham et al. 2012; Fahrig 2013; Hanski 2015; Fletcher et al. 2018a; Fahrig et al. 2019). For example, the ‘habitat amount hypothesis’ posits that within local landscapes, relationships between species richness and habitat distribution are determined primarily by habitat amount irrespective of fragmentation (Fahrig 2013). Nevertheless, numerous theoretical and empirical studies indicate that both population and community responses to habitat amount can be mediated by fragmentation, and responses depend on species-level traits (e.g., dispersal ability, sensitivity to habitat edges) and landscape context (e.g., amount of habitat present, edge type) (Ewers and Didham 2006; With 2016; Haddad et al. 2017; Martin 2018; Valente and Betts 2019). For instance, species with small habitat-area requirements and/or low sensitivity to habitat edges may only be affected by fragmentation in landscapes where habitat is scarce (With and King 2001; Villard and Metzger 2014). In contrast, fragmentation may affect species with large habitat area requirements and/or high sensitivity to habitat edges in landscapes where habitat is relatively abundant (With and King 2001; Villard and Metzger 2014). Importantly, it is not always clear whether edge-sensitive species will exhibit landscape-scale responses to fragmentation, because the amount of edge habitat in a landscape is partially a function of the total amount of habitat present, and edge effects could reflect indirect responses to either habitat amount (Ruffell et al. 2016) or fragmentation (With and Pavuk 2012). Thus, understanding if and where populations are affected by fragmentation requires assessing responses to different levels of fragmentation while accounting for habitat amount (Didham et al. 2012; Villard and Metzger 2014).
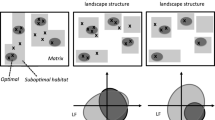
We adopted a landscape-scale approach to test whether variation in the occurrence of four species of declining, edge-sensitive tallgrass prairie birds is driven by grassland area irrespective of fragmentation (consistent with the habitat amount hypothesis; Fahrig 2013) versus grassland area and fragmentation in combination. We further assessed where along a grassland amount gradient (i.e., 20–90% grassland) fragmentation effects occurred by comparing species occurrence probabilities across landscapes containing different levels of grassland fragmentation while accounting for grassland amount. We predicted that grassland area and fragmentation would influence tallgrass prairie birds in one of four possible ways: (A) fragmentation has no effect, such that only grassland amount affects species occurrence (Fig. 1a); (B) fragmentation only matters at low habitat amounts (e.g., < 30%) (Fig. 1c; Andrén 1994; Fahrig 1997); (C) fragmentation only matters at intermediate habitat amounts, where the amount of edge habitat is greatest and varies most (Fig. 1c; Villard and Metzger 2014); or, (D) fragmentation matters across a range of habitat amount, with population responses depending on a species’ edge sensitivity (Fig. 1d; With and King 1999, 2001). We assessed these relationships and tested predictions by modelling the effects of grassland area and fragmentation on species occurrence probabilities across thousands of local landscapes in eastern Kansas, USA.
Plots illustrating predictions about how tallgrass prairie birds would respond to variation in habitat amount (i.e., area) and fragmentation. If total grassland amount irrespective of fragmentation determines species occurrence, we predicted that A responses to a given amount of grassland would not vary between landscapes containing aggregated versus fragmentated grassland configurations. Alternatively, if fragmentation mediates grassland area effects, we predicted that responses to a given amount of grassland would depend on fragmentation B only in landscapes where habitat is scarce, or C only in landscapes containing intermediate amounts of grassland (where fragmentation varies most), or D within a range of habitat amount that depends on a species’ edge sensitivity (grey lines indicate low edge sensitivity whereas black lines indicate high edge sensitivity). We illustrated species-habitat relationships as non-linear functions to make them clearer, but they could also be more linear
Methods
Focal species
We surveyed Grasshopper Sparrows (Ammodramus savannarum), Henslow’s Sparrows (Ammodramus henslowii), Upland Sandpipers (Bartramia longicauda), and Greater Prairie-Chickens (Tympanuchus cupido) because all four species exhibit sensitivity to grassland area and edges (Johnson and Temple 1990; Helzer and Jelinski 1999; Winter et al. 2000; Ribic et al. 2009) and are currently declining across all or most of their geographic ranges (Houston et al. 2011; Rosenberg et al. 2016; Sauer et al. 2017). Both sparrow species are migratory and have male-territorial breeding systems. Grasshopper Sparrows are widely distributed, nesting in both arid and mesic grasslands characterized by moderate vegetative cover (Vickery 1996). Grasshopper Sparrows are recognized as a Common Bird in Steep Decline by Partners in Flight (PIF) because populations have declined by > 60% since 1970 (Rosenberg et al. 2016). Henslow’s Sparrows favor undisturbed mesic grasslands with tall native vegetation and dense litter (Zimmerman 1988). Henslow’s Sparrow population trends are uncertain due to their rarity and elusiveness, but the species is recognized as a Species of Continental Concern (SCC) by PIF because their preferred habitat is extremely scarce (Rosenberg et al. 2016). The Greater Prairie-Chicken is also considered a SCC by PIF and listed as Vulnerable by the International Union for Conservation of Nature because populations have declined by > 50% over the past half-century (BirdLife International 2016; Rosenberg et al. 2016). Prairie-chickens require expansive and heterogeneous grasslands as they use different prairie habitats for lekking (competitive mating displays), nesting, and brood-rearing (Patten et al. 2011). Upland Sandpipers are listed as a species of conservation concern in 22 states and provinces in North America (Houston et al. 2011). A long-distance migrant, the species has a male-territorial breeding system and nests in temperate grasslands in North America characterized by low vegetative cover (Houston et al. 2011).
Study area and bird surveys
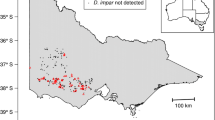
Our study area covered ~ 65,000 km2 of eastern Kansas, USA, including most of the Flint Hills ecoregion and parts of the Central Irregular Plains and Western Corn Belt Plains ecoregions (Omernik 1987) (Fig. 2). Approximately 80% (2 million ha) of North America’s remaining tallgrass prairie is in the Flint Hills, where rocky soils prevent crop agriculture and native perennial grasses are managed for cattle production (With et al. 2008). Areas outside the Flint Hills are dominated by crop agriculture, but also contain hayfields and pastures.
Map of our study area and the 75, 30-point (23.2-km) transects we surveyed for tallgrass prairie birds in eastern Kansas, USA. Blue lines represent portions of transects we surveyed in 2015 and 2016, whereas red lines represent portions we surveyed only in 2016. Thin black and grey lines are boundaries of the Flint Hills ecoregion and state counties, respectively. The rectangular shape in the upper left inset is Kansas, which is in central USA
We conducted point-count bird surveys during the breeding seasons of 2015 and 2016 along North American Breeding Bird Survey (BBS) transects and new transects that we established (Supplementary Material) (Herse et al. 2017, 2018). Both BBS and new transects were primarily along unpaved, infrequently-traveled secondary roads (Supplementary Material), which cause minimal bias in grassland bird surveys (Lituma and Buehler 2016). Eastern Kansas is almost entirely privately owned, and secondary roads provide an efficient means to survey extensive areas while minimizing potential survey bias (Lituma and Buehler 2016). During each 6-min survey, observers recorded presence or absence of each focal species detected within an unlimited radius. In total, we surveyed 1425 points located along 57, 19.2-km transects in 2015. In 2016, we added additional points to all transects, and 18 new 30-point transects, resulting in a total of 2250 survey points located along 75, 23.2-km transects (Fig. 2). Each year we began surveying in early April, which corresponds with sparrow and sandpiper arrival and peak prairie-chicken lekking activity. We finished surveys in late July when most breeding ends. We aimed to survey all transects three times per season during successive ‘rounds’ of surveys. Start and end dates of each survey round overlapped by < 1 week when heavy rains constrained survey schedules. We separated consecutive visits to the same transect by ≥ 2 weeks. ‘Early season’ began 7 April in 2015 and 9 April in 2016, ‘mid season’ began 13 May in 2015 and 20 May in 2016, and ‘late season’ began 15 June in 2015 and 27 June in 2016. Late-season surveys ended by 23 July in 2015 and 29 July in 2016. We describe survey protocols and observer training in more detail in the Supplementary Material.
Landscape factors and spatial scales
Given the study objectives, we considered total grassland area and fragmentation around point-count stations as sources of variation in the occurrence of each species. The broad spatial extent of our study region, coupled with the inherent dynamics of tallgrass prairie ecosystems and private land ownership, precluded mapping finer-scale distributions of each species’ preferred prairie habitat, which varies locally and seasonally with weather and rangeland management (Fuhlendorf et al. 2006; With et al. 2008). Thus, using land-cover data developed by the Kansas Applied Remote Sensing Lab from classified satellite imagery (Peterson et al. 2010), we calculated grassland area as the percent area comprising grassland at different spatial scales (described below; Table S1), combining both warm- and cool-season grasslands because all study species breed in both types (Vickery 1996; Houston et al. 2011; Jaster et al. 2013; Matthews et al. 2013). We used grassland edge density as a measure of grassland fragmentation at each scale (Table S1) because focal species in this study are known to be edge-sensitive (Johnson and Temple 1990; Helzer and Jelinski 1999; Winter et al. 2000; Ribic et al. 2009), and because alternative metrics such as number of grassland patches could obscure variation among landscapes in their proportion of edge habitat. We calculated edge density as the length (m) of grassland edge abutting trees or shrubs, croplands, waterbodies, or developed areas (i.e., commercial, industrial, or residential zones) per ha grassland. The overall accuracy of land-cover classifications was 86.2% (Peterson et al. 2010).
We summarized percent grassland and grassland edge density within spatial scales corresponding with the dispersal distances and space-use areas of the focal species (McGarigal et al. 2016; Fletcher et al. 2018b) using ArcMap 10.3 (Environmental Systems Research Institute, Redlands, CA) and Fragstats 4.2 (McGarigal et al. 2012). We defined the most local scale as the area within 400-m radii (~ 50 ha) of each survey point, which encompassed within-season dispersal distances of > 50% of individuals of each sparrow species (Young 2017; Williams and Boyle 2018), as well as observers’ maximum detection radius (~ 250 m) measured previously (Herse et al. 2017, 2018). Because individuals of both sparrow species sometimes disperse over larger distances within breeding seasons (Young 2017; Williams and Boyle 2018), we doubled the extent while holding the resolution of land-cover data (30 m × 30 m) unchanged, summarizing land cover within 800-m (~ 200 ha) and 1600-m (~ 800 ha) radii. Due to larger detection distances of some prairie-chickens and sandpipers (estimated by observers to sometimes be > 400 m away), we only considered intermediate (800-m radii) and broad (1600-m radii) scales when modelling habitat associations for these species. The intermediate scale corresponds with the minimum patch-area requirements of sandpipers in fragmented landscapes (~ 200 ha; Vickery et al. 1994), whereas the broad scale corresponds with mean breeding-season home range size of sandpipers in the Flint Hills (8.4 km2; Sandercock et al. 2015). Breeding-season home ranges of prairie-chickens are similar or larger in size (> 7–12 km2), with space use and nesting concentrated near leks (Patten et al. 2011; Winder et al. 2014). Thus, because we almost always detected prairie-chickens while lekking, these two scales represent potential core breeding-season home-range areas for this species.
Statistical models
Ecologists often estimate species occurrence probabilities using occupancy models designed to account for imperfect detection (e.g., MacKenzie et al. 2002, 2003). We did not use occupancy models for two main reasons. First, occupancy models estimate site occupancy and detection probabilities by assuming that sites are closed to immigration and emigration between secondary sampling periods within primary sampling periods (e.g., between individual surveys within breeding seasons, such as in this study; Mackenzie et al. 2002, 2003). Thus, if we fit occupancy models to our data, we would have to assume that birds did not move in or out of sites (described below) over entire breeding seasons. However, both focal sparrow species are highly dispersive within breeding seasons (Herse et al. 2017; Williams and Boyle 2018), and sandpipers and prairie-chickens often utilize multiple core areas within large breeding home ranges (Patten et al. 2011; Winder et al. 2014; Hill et al. 2019), which violates the closure assumption of occupancy models and would likely lead to biased estimates (Mackenzie et al. 2002, 2003, 2005; Hayes and Monfils 2015). Second, this study aimed to assess how patterns of species occurrence varied with grassland area and fragmentation, and we did not expect potential imperfect detection to vary systematically with these landscape factors. Thus, we modelled the apparent occurrence of each species using binomial generalized linear mixed models (GLMMs; Bolker et al. 2009), including a random effect of transect in all models to account for spatial non-independence in the survey design. We defined ‘sites’ as areas within which observers could detect each species and classified a site ‘occupied’ by a given species if we detected the species during ≥ 1 survey of either study season or ‘unoccupied’ otherwise. Despite minimizing imperfect detection by hiring seven trained observers (3–4 per season in addition to MRH), rotating observers among transects, surveying each site five times on average (detailed below), and following standardized protocols (see Supplementary Material), we expected that survey effort and timing could have affected detection. Thus, we included fixed effects of survey effort (number of surveys) and the time of morning when we conducted surveys (represented analytically by survey point number because we surveyed points in a consistent order during each visit; Supplementary Material) in all models to account for these potential sources of variation in apparent occurrence. We defined ‘landscapes’ as areas within 400-, 800-, and 1600-m radii of survey points. We only considered landscape factors assayed within 800- and 1600-m radii scales for models of sandpipers and prairie-chickens due to their large detection distances, as described above.
For each species, we first compared alternative models to determine (i) the spatial scale over which percent grassland and grassland edge density affected apparent occurrence most strongly, (ii) whether effects of percent grassland and grassland edge density were interactive versus additive only, and (iii) whether the relationship between apparent occurrence and percent grassland was linear versus non-linear. Constraining individual models to include only landscape variables assayed at a single spatial scale (“pseudo-optimized single scale” approach; McGarigal et al. 2016), we considered alternative models representing all possible combinations of (i) spatial scale, (ii) interactive versus non-interactive effects of landscape variables, and (iii) linear versus quadratic versus logarithmic (i.e., pseudo-threshold) effects of percent grassland. Each model also included fixed effects of survey effort and survey point number, as described above. Correlations among fixed predictor variables (percent grassland, grassland edge density, survey effort, and survey point number) used together were low (r ≤ 0.38; Supplementary Fig. S1). We scaled all predictors to allow direct comparisons of slope estimates (i.e., partial regression coefficients or effect sizes), which are based only on the variation attributed to a given predictor after holding all the others constant (Abdi 2004). Therefore, the slope estimate for a given predictor provides a measure of its relative overall importance for estimating apparent occurrence probability. We fit models using ‘lme4’ package in software R (Bates et al. 2019; R Core Team 2019) and compared competing models using corrected Akaike’s information criterion (∆AICc; Burnham and Anderson 2002). Next, we removed uninformative parameters (i.e., those with slope estimate confidence intervals overlapping zero) from the most parsimonious model if doing so reduced AICc. We then validated the resulting ‘final’ model of each species by verifying that standardized residuals were not spatially autocorrelated (Supplementary Fig. S2) using correlograms (‘ncf’ pacakage; Bjørnstad 2019) and that overdispersion was negligible using χ2 tests (Bolker et al. 2009). As additional measures of fit, we estimated the proportion of variance in apparent occurrence explained by fixed effects only and by each final model including the random effect of transect (Table 1) (‘MuMIn’ package; Barton 2019; Nakagawa et al. 2017).
Assessing where fragmentation affected species occurrence
To determine where fragmentation per se influenced species occurrences most strongly along the grassland-area gradient, we used the final model for each species to assess the predicted relationship between apparent occurrence and percent grassland when grassland configurations were aggregated (i.e., contained small proportions of grassland edge) versus fragmented (i.e., contained large proportions of grassland edge) (Figs. 1, 3). We only predicted relationships across the range of grassland area where fragmentation varied considerably (20–90% grassland; Fig. 3; Supplementary Fig. S1). We used a single value of grassland edge density to represent each configuration type (aggregated versus fragmented) for modelling predictions, basing values on those observed in our study region (Fig. 3). Interdependence between percent grassland and grassland edge density precluded using the overall minimum and maximum observed edge densities to represent aggregated and fragmented configurations, respectively, in all contexts, because grassland edge density varied with grassland amount (Fig. 3; Supplementary Fig. S1). Thus, we chose edge density values that reflected extreme configurations which we observed across the entire gradient of 20–90% grassland. To that end, we identified the minimum and maximum edge densities observed within each 10%-grassland increment from 20–90% grassland at each spatial scale, and then used the highest minimum value of grassland edge density observed across all increments at a given spatial scale to represent ‘aggregated’ configurations, and the lowest maximum edge density to represent ‘fragmented’ configurations (Fig. 3). This approach allowed us to assess the effect of grassland fragmentation on the apparent occurrence of each species in landscapes comprising a given amount of grassland while accounting for the effect of that amount of grassland.
Plot illustrating how we selected edge density values to define aggregated versus fragmented grassland configurations for model predictions. Each circle represents one of 2250 local landscapes (centered on individual survey sites) and the corresponding values of percent grassland and grassland edge density. Black circles represent landscapes comprising 20–90% grassland (the range of percent grassland over which we modelled predictions), whereas light grey circles represent landscapes that fall outside that range. Solid blue and red dots indicate minimum and maximum edge densities, respectively, within each 10%-grassland increment from 20 to 90% grassland (marked by black vertical lines). Arrows and dashed lines indicate the highest minimum and lowest maximum values of grassland edge density observed across all increments, which we used to define ‘aggregated’ and ‘fragmented’ configurations, respectively, in model predictions
Results
We completed 10,291 point-count bird surveys over two breeding seasons (3656 in 2015 and 6635 in 2016) at 2250 sites, visiting most sites 4–5 times in total (mean 4.5, median 5, range 2–6). Henslow’s Sparrows and Greater Prairie-Chickens were each recorded at < 10% of sites and on fewer than half of the transects (Table 1). Upland Sandpipers and Grasshopper Sparrows were each recorded at approximately one-quarter and one-half of sites, respectively, and on most transects. Holding grassland area and fragmentation effects constant, apparent occurrence of all species increased with survey effort. Apparent occurrence of Grasshopper Sparrows and Greater Prairie-Chickens was also higher at sites surveyed earlier in the morning (Table 1). Having accounted for variation in apparent occurrence caused by effects of survey effort and timing, we subsequently regarded residual variation as representing an index of true occurrence.
The occurrence of Grasshopper Sparrows and Henslow’s Sparrows correlated most strongly with landscape factors within 400-m radii, whereas Greater Prairie-Chicken and Upland Sandpiper occurrence correlated with landscape factors most strongly within 1600-m radii (Supplementary Tables S2–S5). Occurrence of all species increased with percent grassland, with both sparrow species responding non-linearly (Table 1; Fig. 4). Grassland edge density was negatively associated with the occurrence of all four focal species (Table 1; Fig. 4). Overall, the direct effect of grassland edge density on species occurrence was greater than that of percent grassland for Grasshopper Sparrows and Upland Sandpipers (Table 1). Moreover, the magnitude of the fragmentation effect on sandpipers depended strongly on percent grassland, as indicated by a significant interaction term (Table 1). Grassland fragmentation reduced the occurrence of sandpipers in landscapes comprising 20–90% grassland, with fragmentation effects being most pronounced in landscapes containing 90% grassland (Fig. 4). Even in the absence of significant interactions, fragmentation reduced the occurrence of the other species by at least half in some contexts (Fig. 4). Fragmentation effects on the occurrence of sparrows were most pronounced in landscapes comprising ~ 50–90% grassland. Confidence limits for the occurrence of prairie-chickens within aggregated versus fragmented grasslands overlapped across the entire grassland-area gradient, but the estimates differed most at 90% grassland (Fig. 4).
Grassland fragmentation mediated relationships between apparent occurrence and percent grassland for four tallgrass prairie bird species. To illustrate where fragmentation effects were most pronounced, we plotted relationships between apparent occurrence and percent grassland in landscapes containing ‘aggregated’ versus ‘fragmented’ grassland configurations. We defined ‘aggregated’ configurations as having low edge density (400-m radii landscapes = 22.2 m/ha; 1600-m radii landscapes = 19.9 m/ha) and ‘fragmented’ configurations as having high edge density (400-m radii landscapes = 98.2 m/ha; 1600-m radii landscapes = 64.2 m/ha). Circles are the raw presence-absence data for each species, offset slightly along the y axis to make them more visible and colored to show variation among landscapes in grassland edge density (ED). Thin lines around estimates indicate 95% confidence limits for fixed effects only. Estimates are based on the most parsimonious model (i.e., lowest AICc) for each species and account for variation in apparent occurrence that is attributable to differences among sites in survey effort and timing
Discussion
Fragmentation clearly mediated the effects of grassland area on the occurrence of four declining tallgrass prairie bird species. Fragmentation effects were most pronounced in landscapes comprising ~ 50–90% grassland (Fig. 4), reflecting the focal species’ strong preferences for large tracts of grassland and high sensitivity to grassland edges (Johnson and Temple 1990; Helzer and Jelinski 1999; Winter et al. 2000; Ribic et al. 2009). Conversely, fragmentation effects were less pronounced or absent in landscapes comprising < 50% grassland as these landscapes were occupied relatively infrequently regardless of the level of fragmentation (Fig. 4). This result suggests that when grassland is scarce, edge habitat is unavoidable for focal species and modifying grassland configuration to reduce habitat edge-to-area ratio may not attract more birds. It could also explain why the focal species are absent from many of North America’s remaining tallgrass prairies, which tend to be confined to relatively small fragments (Herkert 1994; Ribic et al. 2009). Theoretical models have predicted that a species’ response to habitat amount and fragmentation should be influenced more by edge-sensitivity than area-sensitivity (With and King 2001). However, many grassland birds are apparently both edge- and area-sensitive (Ribic et al. 2009; Winter et al. 2000), which has undoubtedly contributed to population collapses following landscape change (Brennan and Kuvlesky 2005; NABCI 2016).
The landscape-scale models developed in this study tended to be better at explaining variation in the occurrence of species that were relatively common (Table 1). The low proportion of variance explained by models of Henslow’s Sparrow occurrence (Table 1) likely reflects both their rarity and stringent habitat preferences that are not simply reflected in total grassland area and grassland edge density. Despite strongly favoring sites embedded in large grasslands with low edge density, Henslow’s Sparrows were absent from hundreds of such sites. Some apparent absences of Henslow’s Sparrows could be due to imperfect detection. However, the remarkable rarity of Henslow’s Sparrows in expansive prairie must be at least partially explained by intensive rangeland management practices in eastern Kansas (Reinking 2005; With et al. 2008), where annual fires, heavy grazing, and frequent haying reduce vegetation height and density in ways that reduce habitat quality for Henslow’s Sparrows (Reinking 2005). Thus, preserving or increasing core grassland area without simultaneously reducing the intensity of management of those grasslands is unlikely to benefit this species (Fuhlendorf et al. 2006; With et al. 2008; Hovick et al. 2015b).
Greater Prairie-Chickens were like Henslow’s Sparrows in that they strongly favored expansive grasslands and were frequently absent from such grasslands, probably due in part to the intensity of rangeland management and resulting lack of vegetative cover. Another aspect of the prairie-chicken results hints that fragmentation effects may depend on edge context; grassland edge negatively affected prairie-chicken occurrence (Table 1), but confidence limits for estimates in aggregated versus fragmented landscapes overlapped over the entire gradient of grassland amount (Fig. 4). This could be caused by prairie-chickens responding differently to grassland-to-cropland versus grassland-to-woodland edges, which collectively comprise most grassland edges in the study region. Although prairie-chickens consistently avoid woody edges (Matthews et al. 2011; McNew et al. 2014), they sometimes select areas near crop fields, which may provide suitable combinations of vegetative cover for nesting and bare ground for feeding (Matthews et al. 2011). Thus, treating cropland and woody edges as equivalent may obscure finer-scale responses to landscape features. We recommend that land managers recognize the potential threats and values associated with different edge types (Winter et al. 2000; Ries et al. 2004; Herse et al. 2018).
Most temperate grasslands worldwide have already been lost to, or fragmented by, agricultural development (Hoekstra et al. 2005). Remaining grasslands are still subject to an array of anthropogenic threats and disturbances. One such threat is the pervasive spread of buildings and energy infrastructures that intersect and dot, or ‘perforate,’ grassland patches (Hovick et al. 2014, 2015a). Because patterns of grassland perforation are spatially irregular, small amounts of grassland loss via perforation can disproportionately increase the edge-to-area ratio of grassland to levels typically associated with greater loss via agricultural development. Thus, for edge-sensitive species with large habitat-area requirements, habitat fragmentation may only matter in landscapes comprising large amounts of habitat (e.g., > 50%) where habitat perforation is the main cause of habitat loss. Conversely, fragmentation may matter less for species such as those we studied where, regardless of spatial configuration, habitat area is insufficient to attract viable numbers of individuals. Thus, future development should avoid expansive tracts of habitat, and when development is unavoidable, minimize the amount of perforation and resulting edge that it creates.
Another major source of habitat perforation in grasslands is woody encroachment. In mesic tallgrass prairies, fire-free periods of > 3 years allow shrubs and trees to establish and grow large enough to survive subsequent fires and ultimately transition to shrublands or woodlands within a few decades (Ratajczak et al. 2014). Furthermore, agricultural development can contribute to woody encroachment by increasing edge area where young shrubs and trees are less susceptible to fires (Scholtz et al. 2018). Thus, woody encroachment can be both a product and driver of grassland loss and fragmentation, and undoubtedly contributed to the fragmentation effects we found. Likewise, preventing woody encroachment, particularly in expansive grasslands such as the Flint Hills of eastern Kansas, is necessary for reversing grassland bird declines in North America (Fuhlendorf et al. 2017; Tack et al. 2017).
Rethinking expectations about where fragmentation matters
Ecologists often expect fragmentation to matter most when habitat is scarce (Fahrig 1998; Hanski and Gaggiotti 2004; Hanski 2015; Supplementary Fig. S3). For example, Hanski (2015) stated, “everybody agrees that in the case of land-coverings such as forests, fragmentation makes little or no difference if the amount of habitat is large, [covering] more than 20–30% of the landscape.” This notion is rooted in island biogeography and metapopulation theories wherein fragmentation negatively affects populations by inhibiting dispersal (MacArthur and Wilson 1967; Levins 1969; Hanski 1998). Indeed, dispersal-limited populations become increasingly vulnerable to isolation as habitat becomes confined to distant patches that are too far away to reach, which often does not occur until more than half of the habitat in a landscape is lost (With and Crist 1995; King and With 2002; Swift and Hannon 2010). However, this is not always the case; theoretical models have shown that species with weak dispersal ability can be influenced by fragmentation where habitat covers as much as 80% of a landscape (King and With 2002). Habitat can also become effectively isolated for vagile species if individuals are unwilling to disperse. For example, some tropical understory birds avoid dispersing across forest clearings despite being able to (Van Houtan et al. 2007). Thus, when fragmentation inhibits dispersal, the range of habitat amount where fragmentation matters depends on a species’ dispersal propensity, which may be influenced by characteristics of the intervening matrix between habitat patches (Ewers and Didham 2006; Boesing et al. 2018).
Importantly, dispersal is not the only mechanism via which fragmentation can negatively affect populations. The focal species of this study are not dispersal-limited within the spatial scales we considered, eliminating dispersal as a mechanism that could explain the negative responses we observed to fragmentation. Instead, fragmentation in local landscapes probably affected grassland birds via negative edge effects (Winter et al. 2000; Herse et al. 2018; Lockhart and Koper 2018). For instance, in expansive North American prairies, energy infrastructure can displace adult Greater Prairie-Chickens and/or increase their mortality via collisions (Hovick et al. 2014). Similarly, the presence of woody vegetation within or beside grasslands can increase nest predation rates by snakes and mid-sized carnivores for Henlsow’s Sparrows, Grasshopper Sparrows, and other grassland-obligate birds such as Dickcissel (Spiza americana) (Winter et al. 2000; Klug et al. 2010). Negative edge effects such as these and others are not unique to tallgrass prairie birds and have been documented in places ranging from North America to Borneo and in taxa ranging from ants to ungulates (Suarez et al. 1998; Ewers and Didham 2006; Brodie et al. 2015; Pérez-Rodríguez et al. 2018). Thus, edge-mediated fragmentation effects can occur in landscapes comprising small, intermediate, and/or large amounts of habitat and depend on edge context and species’ responses to different edge types (Ewers and Didham 2006; Herse et al. 2018).
Conclusions
The results of this study run counter to assertions that fragmentation does not matter (Fahrig 2013) or only matters when habitat is scarce (Fahrig 1998; Hanski and Gaggiotti 2004; Hanski 2015). Fragmentation mediated the effects of habitat area on declining tallgrass prairie birds, likely by increasing proportions of edge habitat, which the focal species often avoid. Fragmentation effects were most pronounced in landscapes comprising large amounts of grassland and less pronounced or absent in landscapes comprising small amounts of grassland as they were inhabited relatively infrequently. In expansive prairie, small amounts of grassland perforation can create disproportionately large amounts of edge habitat that deter birds from settling. Thus, large grasslands are not necessarily suitable if they lack enough core habitat away from edges. Consequently, preventing further land development and woody encroachment in remaining large prairies is crucial to reversing the rapid declines that grassland birds are currently experiencing. Additional research is needed to identify the species-level traits that determine if, where, and why fragmentation matters, to help guide species-level conservation.
Change history
12 January 2021
In the original publication of the article, Table 1 was published incorrectly. The level of significance for the modelled fixed effect of grassland edge density on Grasshopper Sparrow occurrence was omitted from the published table. Grassland edge density had a significant negative effect on Grasshopper Sparrow occurrence (<Emphasis Type="Italic">β</Emphasis> = − 0.71, standard error = 0.09, <Emphasis Type="Italic">p</Emphasis> < 0.001). The corrected version of Table 1 is shown below.
References
Abdi H (2004) Partial regression coefficients. In: Lewis-Beck M, Bryman A, Futing T (eds) Encyclopedia of social sciences research methods. Sage, Thousand Oaks, pp 1–4
Andrén H (1994) Effects of habitat fragmentation on birds and mammals in landscapes with different proportions of suitable habitat: a review. Oikos 71:355–366
Azpiroz AB, Isacch JP, Dias RA, Di Giacomo AS, Fontana CS, Palarea CM (2012) Ecology and conservation of grassland birds in southeastern South America: a review. J Field Ornithol 83:217–246
Barton K (2019) Package “MuMIn” - Multi-model inference version 1.43.6. http://CRAN.R-project.org/package=MuMIn
Bates D, Maechler M, Bolker B, Walker S, Bojesen RH, Singmann H, Dai B, Scheipl F, Grotherndieck G, Green P, Fox J (2019) lme4: Linear mixed-effects models using “Eigen” and S4 version 1.1–21, http://CRAN.R-project.org/package=lme4
BirdLife International (2016) Tympanuchus cupido. The IUCN Red List of Threatened Species 2016: e.T22679514A92817099. https://doi.org/10.2305/IUCN.UK.2016-3.RLTS.T22679514A92817099.en. Accessed 10 Mar 2020
Bjørnstad ON (2019) ncf: Spatial Covariance Functions version 1.2–8. http://CRAN.R-project.org/package=ncf
Boesing AL, Nichols E, Metzger JP (2018) Biodiversity extinction thresholds are modulated by matrix type. Ecography 41:1520–1533
Bolker BM, Brooks ME, Clark CJ, Geange SW, Poulsen JR, Stevens MHH, White JSS (2009) Generalized linear mixed models: a practical guide for ecology and evolution. Trends Ecol Evol 24:127–135
Brennan LA, Kuvlesky WP (2005) North American grassland birds: an unfolding conservation crisis? J Wildl Manag 69:1–13
Briggs JM, Knapp AK, Blair JM, Heisler JL, Hoch GA, Lett MS, McCarron JK (2005) An ecosystem in transition: causes and consequences of the conversion of mesic grassland to shrubland. Bioscience 55:243–254
Brodie JF, Giordano AJ, Ambu L (2015) Differential responses of large mammals to logging and edge effects. Mammal Biol 80:7–13
Burnham K, Anderson D (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edn. Springer, New York
Collinge SK (2009) Ecology of fragmented landscapes. John Hopkins University Press, Baltimore
Didham RK, Kapos V, Ewers RM (2012) Rethinking the conceptual foundations of habitat fragmentation research. Oikos 121:161–170
Ewers RM, Didham RK (2006) Confounding factors in the detection of species responses to habitat fragmentation. Biol Rev 81:117
Fahrig L (1997) Relative effects of habitat loss and fragmentation on population extinction. J Wildl Manag 61:603–610
Fahrig L (1998) When does fragmentation of breeding habitat affect population survival? Ecol Model 105:273–292
Fahrig L (2003) Effects of habitat fragmentation on biodiversity. Annu Rev Ecol Evol Syst 34:487–515
Fahrig L (2013) Rethinking patch size and isolation effects: the habitat amount hypothesis. J Biogeogr 40:1649–1663
Fahrig L (2017) Ecological responses to habitat fragmentation per se. Annu Rev Ecol Evol Syst 48:1–23
Fahrig L, Arroyo-Rodríguez V, Bennett JR, Boucher-Lalonde V, Cazetta E, Currie DJ, Eigenbrod F, Ford AT, Harrison SP, Jaeger JA, Koper N, Martin AE, Martin JL, Metzger JP, Morrison P, Rhodes RJ, Saunders DA, Simberloff D, Smith AC, Tischendorf L, Vellend M, Watling JI (2019) Is habitat fragmentation bad for biodiversity? Biol Conserv 230:179–186. https://doi.org/10.1016/j.biocon.2018.12.026
Fletcher RJ Jr, Reichert BE, Holmes K (2018a) The negative effects of habitat fragmentation operate at the scale of dispersal. Ecology 99:2176–2186
Fletcher RJ Jr, Didham RK, Banks-Leite C, Barlow J, Ewers RM, Rosindell J, Holt RD, Gonzalez A, Pardini R, Damschen EI, Melo FP (2018b) Is habitat fragmentation good for biodiversity? Biol Conserv 226:9–15
Fuhlendorf SD, Harrell WC, Engle DM, Hamilton RG, Davis CA Jr (2006) Should heterogeneity be the basis for conservation? Grassland bird response to fire and grazing. Ecol Appl 16:1706–1716
Fuhlendorf SD, Hovick TJ, Elmore RD, Tanner AM, Engle DM, Davis CA (2017) A hierarchical perspective to woody plant encroachment for conservation of prairie-chickens. Rangel Ecol Manag 70:9–14
Grand J, Wilsey C, Wu JX, Michel NL (2019) The future of North American grassland birds: incorporating persistent and emergent threats into full annual cycle conservation priorities. Conserv Sci Pract 1:e20
Haddad NM, Gonzalez A, Brudvig LA, Burt MA, Levey DJ, Damschen EI (2017) Experimental evidence does not support the habitat amount hypothesis. Ecography 40:48–55
Hadley AS, Betts MG (2016) Refocusing habitat fragmentation research using lessons from the last decade. Curr Landsc Ecol Rep 1:55–66
Hanski I (1998) Metapopulation dynamics. Nature 396:41–49
Hanski I (2015) Habitat fragmentation and species richness. J Biogeogr 42:989–993
Hanski I, Gaggiotti OE (2004) Metapopulation biology: past, present, and future. In: Hanski I, Gaggiotti OE (eds) Ecology, genetics, and evolution of metapopulations. Academic Press, San Diego, pp 3–22
Hargreaves A (2019) Lasting signature of forest fragmentation. Science 366:1196–1197
Hayes DB, Monfils MJ (2015) Occupancy modelling of bird point counts: implications of mobile animals. J Wildl Manag 79:1361–1368
Helzer CJ, Jelinski DE (1999) The relative importance of patch area and perimeter-area ratio to grassland breeding birds. Ecol Appl 9:1448
Herkert JR (1994) The effects of habitat fragmentation on Midwestern grassland bird communities. Ecol Appl 4:461–471
Herse MR, Estey ME, Moore PJ, Sandercock BK, Boyle WA (2017) Landscape context drives breeding habitat selection by an enigmatic grassland songbird. Landsc Ecol 32:2351–2364
Herse MR, With KA, Boyle WA (2018) The importance of core habitat for a threatened species in changing landscapes. J Appl Ecol 55:2241–2252
Hill JM, Sandercock BK, Renfrew RB (2019) Migration patterns of Upland Sandpipers in the Western Hemisphere. Front Ecol Evol 7:426
Hoekstra JM, Boucher TM, Ricketts TH, Roberts C (2005) Confronting a biome crisis: global disparities of habitat loss and protection. Ecol Lett 8:23–29
Houston C, Jackson C, Bowen Jr D (2011) Upland Sandpiper (Bartramia longicauda), version 2.0. In: Poole AF (ed) The Birds of North America Online. Cornell Lab of Ornithology, Ithaca. http://doi.org/10.2173/bna.580. Accessed 10 March 2020
Hovick TJ, Dahlgren DK, Papeş M, Elmore RD, Pitman JC (2015a) Predicting Greater Prairie-Chicken lek site suitability to inform conservation actions. PLoS ONE 10:e0137021
Hovick TJ, Elmore RD, Dahlgren DK, Fuhlendorf SD, Engle DM (2014) Evidence of negative effects of anthropogenic structures on wildlife: a review of grouse survival and behaviour. J Appl Ecol 51:1680–1689
Hovick TJ, Elmore RD, Fuhlendorf SD, Engle DM, Hamilton RG (2015b) Spatial heterogeneity increases diversity and stability in grassland bird communities. Ecol Appl 25:662–672
Intergovernmental Science-Policy Platform on Biodiversity and Ecosystem Services (IPBES) (2019) Global Assessment Summary for Policy Makers. IPBES Secretariat, Bonn
Jaster L, Jensen WE, Forbes AR (2013) Abundance, territory sizes, and pairing success of male Henslow’s Sparrows in restored warm- and cool-season grasslands. J Field Ornithol 84:234–241
Johnson RG, Temple SA (1990) Nest predation and brood parasitism of tallgrass prairie birds. J Wildl Manag 54:106–111
King AW, With KA (2002) Dispersal success on spatially structured landscapes: when do spatial pattern and dispersal behavior really matter? Ecol Model 147:23–39
Klug PE, Jackrel SL, With KA (2010) Linking snake habitat use to nest predation risk in grassland birds: the dangers of shrub cover. Oecologia 162:803–813
Kulmatiski A, Beard KH (2013) Woody plant encroachment facilitated by increased precipitation intensity. Nat Clim Change 3:833–837
Levins R (1969) Some demographic and genetic consequences of environmental heterogeneity for biological control. Bull Entomol Soc Am 15:237–240
Lituma CM, Buehler DA (2016) Minimal bias in surveys of grassland birds from roadsides. Condor 118:715–727
Lockhart J, Koper N (2018) Northern prairie songbirds are more strongly influenced by grassland configuration than grassland amount. Landsc Ecol 33:1543–1558
MacArthur RH, Wilson EO (1967) The theory of island biogeography. Princeton University Press, Princeton
MacKenzie DI, Nichols JD, Hines JE, Knutson MG, Franklin AB (2003) Estimating site occupancy, colonization, and local extinction when a species is detected imperfectly. Ecology 84:2200–2207
MacKenzie DI, Nichols JD, Lachman GB, Droege S, Royle JA, Langtimm CA (2002) Estimating site occupancy rates when detection probabilities are less than one. Ecology 83:2248–2255
MacKenzie DI, Royle A (2005) Designing occupancy studies: general advice and allocating survey effort. J Appl Ecol 42:1105–1114
Martin CA (2018) An early synthesis of the habitat amount hypothesis. Landsc Ecol 33:1831–1835
Matthews TW, Tyre AJ, Taylor JS, Lusk JJ, Powell LA (2011) Habitat selection and brood survival of Greater Prairie-Chickens. In: Sandercock BK, Martin K, Segelbacher G (eds) Ecology, conservation, and management of grouse. Studies in Avian Biology (no. 39), University of California Press, Berkley, pp 179–191
Matthews TW, Tyre AJ, Taylor JS, Lusk JJ, Powell LA (2013) Greater prairie-chicken nest success and habitat selection in southeastern Nebraska. J Wildl Manag 77:1202–1212
McGarigal K, Cushman SA, Ene E (2012) FRAGSTATS v4: Spatial Pattern Analysis Program for Categorical and Continuous Maps. http://www.umass.edu/landeco/research/fragstats/fragstats.html
McGarigal K, Wan HY, Zeller KA, Timm BC, Cushman SA (2016) Multi-scale habitat selection modeling: a review and outlook. Landsc Ecol 31:1161–1175
McNew LB, Hunt LM, Gregory AJ, Wisely SM, Sandercock BK (2014) Effects of wind energy development on nesting ecology of Greater Prairie-Chickens in fragmented grasslands. Conserv Biol 28:1089–1099
Nakagawa S, Johnson PCD, Schielzeth H (2017) The coefficient of determination R2 and intra-class correlation coefficient from generalized linear mixed-effects models revisited and expanded. J R Soc B 14:20170213
North American Bird Conservation Initiative (NABCI) (2016) The State of North America’s Birds 2016. Environment and Climate Change Canada, Ottawa
North American Bird Conservation Initiative (NABCI), U.S. Committee (2011) The State of the Birds 2011 Report on Public Lands and Waters. U.S. Department of Interior, Washington, DC
Omernik JM (1987) Ecoregions of the Conterminous United States. Ann Assoc Am Geogr 77:118–125
Patten MA, Pruett CL, Wolfe DH (2011) Home range size and movements of Greater Prairie-Chickens. In: Sandercock BK, Martin K, Segelbacher G (eds) Ecology, conservation, and management of grouse. Studies in Avian Biology (no. 39), University of California Press, Berkley, pp 51–62
Pérez-Rodríguez A, Khimoun A, Ollivier A, Eraud C, Faivre B, Garnier S (2018) Habitat fragmentation, not habitat loss, drives the prevalence of blood parasites in a Caribbean passerine. Ecography 41:1835–1849
Peterson D, Whistler J, Egbert E, Martinko E (2010) 2005 Kansas land cover patterns: Phase II Final Report. Open-file Report 167. Kansas Biological Survey, Lawrence, KS
R Core Team (2019) R: a language environment for statistical computing. R Foundation for Statistical Computing, Vienna
Rahmig CJ, Jensen WE, With KA (2009) Grassland bird responses to land management in the largest remaining tallgrass prairie. Conserv Biol 23:420–432
Ratajczak Z, Nippert JB, Briggs JM, Blair JM (2014) Fire dynamics distinguish grasslands, shrublands and woodlands as alternative attractors in the Central Great Plains of North America. J Ecol 102:1374–1385
Reinking DL (2005) Fire regimes and avian responses in the central tallgrass prairie. Stud Avian Biol 30:116–126
Renfrew RB, Ribic CA (2003) Grassland passerine nest predators near pasture edges identified on videotape. Auk 120:371–383
Renfrew RB, Ribic CA (2008) Multi-scale models of grassland passerine abundance in a fragmented system in Wisconsin. Landsc Ecol 23:181–193
Ribic CA, Koford RR, Herkert JR, Johnson DH, Niemuth ND, Naugle DE, Bakker KK, Sample DW, Renfrew RB (2009) Area sensitivity in North American grassland birds: patterns and processes. Auk 126:233–244
Ries L, Fletcher RJ Jr, Battin J, Sisk TD (2004) Ecological responses to habitat edges: mechanisms, models, and variability explained. Annu Rev Ecol Evol Syst 35:491–522
Rosenberg KV, Kennedy JA, Dettmers R, Ford RP, Reynolds D, Alexander JD, Beardmore CJ, Blancher PJ, Bogart RE, Butcher GS, Camfield AF (2016) Partners in Flight Landbird Conservation Plan: 2016 Revision for Canada and Continental United States. Partners in Flight Science Committee
Ruffell J, Banks-Leite C, Didham RK (2016) Accounting for the causal basis of collinearity when measuring the effects of habitat loss versus habitat fragmentation. Oikos 125:117–125
Samson F, Knopf F (1994) Prairie conservation in North America. Bioscience 44:418–421
Sandercock BK, Alfaro-Barrios M, Casey AE, Johnson TN, Mong TW, Odom KJ, Strum KM, Winder VL (2015) Effects of grazing and prescribed fire on resource selection and nest survival of upland sandpipers in an experimental landscape. Landsc Ecol 30:325–337
Sauer JR, Niven DK, Hines JE, Ziolkowski Jr DJ, Pardieck KL, Fallon JE, Link WA (2017) The North American Breeding Bird Survey, Results and Analysis 1966–2015. Version 2.07.2017. USGS Patuxtent Wildlife Research Center, Laurel, MA
Scholtz R, Polo JA, Tanner EP, Fuhlendorf SD (2018) Grassland fragmentation and its influence on woody plant cover in the southern Great Plains, USA. Landsc Ecol 33:1785–1797
Shaffer JA, Buhl DA (2016) Effects of wind-energy facilities on breeding grassland bird distributions. Conserv Biol 30:59–71
Suarez AV, Bolger DT, Case TJ (1998) Effects of fragmentation and invasion on native any communities in coastal southern California. Ecology 79:2041–2056
Swift TL, Hannon SJ (2010) Critical thresholds associated with habitat loss: a review of the concepts, evidence, and applications. Biol Rev 85:35–53
Tack JD, Quamen FR, Kelsey K, Naugle DE (2017) Doing more with less: removing trees in a prairie system improves value of grasslands for obligate bird species. J Environ Manag 198:163–169
Tscharntke T, Tylianakis JM, Rand TA, Didham RK, Fahrig L, Batáry P, Bengtsson J, Clough Y, Crist TO, Dormann CF, Ewers RM (2012) Landscape moderation of biodiversity patterns and processes-eight hypotheses. Biol Rev 87:661–685
Valente JJ, Betts MG (2019) Response to fragmentation by avian communities is mediated by species traits. Divers Distrib 25:48–60
Van Houtan KS, Pimm SL, Halley JM, Bierregaard RO, Lovejoy TE (2007) Dispersal of Amazonian birds in continuous and fragmented forest. Ecol Lett 10:219–229
Vickery PD (1996) Grasshopper Sparrow (Ammodramus savannarum), version 2.0. In: Poole AF, Gill FB (eds) The birds of North America online
Vickery PD, Hunter ML, Melvin SM (1994) Effects of habitat area on the distribution of grassland birds in Maine. Conserv Biol 8:1087–1097
Villard M-A, Metzger JP (2014) Beyond the fragmentation debate: a conceptual model to predict when habitat configuration really matters. J Appl Ecol 51:309–318
Williams EJ, Boyle WA (2018) Patterns and correlates of within-season breeding dispersal: a common strategy in a declining grassland songbird. Auk 135:1–14
Winder VL, McNew LB, Gregory AJ, Hunt LM, Wisely SM, Sandercock BK (2014) Space use by female Greater Prairie-Chickens in response to wind energy development. Ecosphere 5:3
Winter M, Johnson DH, Faaborg J (2000) Evidence for edge effects on multiple levels in tallgrass prairie. Condor 102:256–266
With KA (1994) The hazards of nesting near shrubs for a grassland bird, the McCown’s Longspur. Condor 96:1009–1019
With KA (2016) Are landscapes more than the sum of their patches? Landsc Ecol 31:969–980
With KA, Crist TO (1995) Critical thresholds in species’ responses to landscape structure. Ecology 76:2446–2459
With KA, King AW (1999) Extinction thresholds for species in fractal landscapes. Conserv Biol 13:314–326
With KA, King AW (2001) Analysis of landscape sources and sinks: the effect of spatial pattern on avian demography. Biol Conserv 100:75–88
With KA, King AW, Jensen WE (2008) Remaining large grasslands may not be sufficient to prevent grassland bird declines. Biol Conserv 141:3152–3167
With KA, Pavuk DM (2012) Direct versus indirect effects of habitat fragmentation on community patterns in experimental landscapes. Oecologia 170:517–528
Young AC (2017) Seasonal fecundity and post-fledging survival and habitat selection of Henslow’s Sparrow (Ammodramus henslowii). MS Thesis, University of Nebraska
Zimmerman JL (1988) Breeding season habitat selection by the Henslow’s Sparrow (Ammodramus henslowii) in Kansas. Wilson Bull 100:17–24
Acknowledgements
We thank our field crew, K. Courtois, P. Moore, L. Rhine, K. Scott, P. Turner, and E. Wilson, and laboratory assistant S. Replogle-Curnutt. M. Estey provided logistical support, and B. Meiwes and V. Cikanek of Kansas Department of Wildlife, Parks and Tourism provided accommodations at Fall River Wildlife Area. This project was funded by U.S. Fish and Wildlife Service and Eastern Tallgrass Prairie and Big Rivers Landscape Conservation Cooperative.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic Supplementary Material
Below is the link to the electronic Supplementary Material.
Rights and permissions
About this article
Cite this article
Herse, M.R., With, K.A. & Boyle, W.A. Grassland fragmentation affects declining tallgrass prairie birds most where large amounts of grassland remain. Landscape Ecol 35, 2791–2804 (2020). https://doi.org/10.1007/s10980-020-01064-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-020-01064-y