Abstract
Context
Interactions between landscape-scale processes and fine-grained habitat heterogeneity are usually invoked to explain species occupancy in fragmented landscapes. In variegated landscapes, however, organisms face continuous variation in micro-habitat features, which makes necessary to consider ecologically meaningful estimates of habitat quality at different spatial scales.
Objectives
We evaluated the spatial scales at which forest cover and tree quality make the greatest contribution to the occupancy of the long-horned beetle Microplophorus magellanicus (Coleoptera: Cerambycidae) in a variegated forest landscape.
Methods
We used averaged data of tree quality (as derived from remote sensing estimates of the decay stage of single trees) and spatially independent pheromone-baited traps to model the occurrence probability as a function of multiple cross-scale combinations between forest cover and tree quality (with scales ranging between 50 and 400 m).
Results
Model support and performance increased monotonically with the increasing scale at which tree quality was measured. Forest cover was not significant, and did not exhibit scale-specific effects on the occurrence probability of M. magellanicus. The interactive effect between tree quality and forest cover was stronger than the independent (additive) effects of tree quality and particularly forest cover. Significant interactions included tree quality measured at spatial scales ≥200 m, but cross-scale interactions occurred only in four of the seven best-supported models.
Conclusions
M. magellanicus respond to the high-quality trees available in the landscape rather than to the amount of forest per se. Conservation of viable metapopulations of M. magellanicus should consider the quality of trees at spatial scales >200 m.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Habitat quality is one of the main factors influencing space use, habitat selection, population/metapopulation dynamics, and distribution of wild animals (e.g., Thomas 2000; Morrison et al. 2006; Mortelliti et al. 2010). Evolutionary and ecological studies predict that animals should be more likely to occupy habitat patches as the biotic and abiotic conditions at these patches become more suitable for reproductive success and survival (Fretwell and Lucas 1970; Cody 1985; Hall et al. 1997; Johnson 2007; Mortelliti et al. 2010). Consequently, conservation efforts and ecological research have been oriented toward understanding how species respond to natural and human-induced changes in habitat quality (Sala et al. 2000; Griffen and Drake 2008).
Although habitat degradation (sensu Lindenmayer and Fischer 2007) is well known to affect invertebrates (Wettstein and Schmid 1999; Heisswolf et al. 2009), vertebrates (Verboom et al. 1991; Mortelliti et al. 2012) and plants (Adriaens et al. 2009), assessing how changes in habitat quality alter occupancy patterns requires addressing some critical conceptual and methodological issues. First, we need to know at which spatial scales organisms perceive and respond to habitat heterogeneity (Johnson 1980; Hutto 1985; Wiens 1989; O’Neill et al. 1989; Kristan 2006). Distinguishing the spatially hierarchical levels of habitat structure (i.e., micro-habitats, habitat patches and landscape) can be appropriate for species responding hierarchically to habitat conditions (Boyce 2006; Davis et al. 2007; Doligez et al. 2008; Mayor et al. 2009). However, such a spatial design may not be suited for landscapes with some level of habitat variegation (sensu McIntyre and Barrett 1992), where habitat resources are not distributed patchily, but rather vary gradually across the landscape (Fischer and Lindenmayer 2007; Lindenmayer and Fischer 2007; Drielsma and Ferrier 2009). In this case, occupancy patterns of wild animals should respond to continuous variations in habitat quality resulting from multi-scale processes, such as broad-scale environmental gradients or fine-grained disturbances (Lindenmayer and Fischer 2007). Second, landscape-scale studies require reliable and robust estimates of habitat quality. Although field-based estimates are generally more realistic than using gross proxies (e.g., broad vegetation or land-use classes), estimates of habitat quality are sensitive to the trade-off between accuracy and sample size (Mortelliti et al. 2010). Recent developments in high-resolution remote sensing systems have improved identification of fine grained habitat patches that vary in the amount of resources they provide to wildlife, such as individual trees used by tree specialist species (Mason et al. 2003; Waser et al. 2014; Jacobsen et al. 2015; Soto et al. 2016). Third, processes occurring at broad landscape or regional scales can produce nonlinear (threshold) effects on fine-grained processes (Peters et al. 2004; Schooley and Branch 2007). Top-down constraints can involve habitat preferences becoming weaker as the amount of habitat and connectivity decrease in the landscape (Schooley and Branch 2007; Vergara and Armesto 2009; Mortelliti 2013; Vergara et al. 2015). Cross-scale linkages between habitat quality and quantity, however, depend on how organisms perceive, or respond to, multi-scale heterogeneity in habitat conditions (Schooley and Branch 2007). Thus, species occupancy patterns may be misunderstood if ecologically meaningful measures of habitat quality are not available for large spatial scales.
In this study we evaluated at which spatial scales forest cover and tree quality make the largest contribution to the occupancy pattern of the long-horned beetle Microplophorus magellanicus (Coleoptera: Cerambycidae). The woodboring larvae of M. magellanicus are specialized on wood with advanced decay, which is typically present in weakened, dying, or dead trees of southern beech species (genus Nothofagus) that dominate Magellanic subpolar forests (Cerda 1986; Barriga et al. 1993; Artigas 1994; Cerda and Angulo 2002; Zuñiga-Reinoso 2013). Thus, tree quality for M. magellanicus may be considered as being proportional to the availability of decaying wood microhabitat (e.g., see Grove 2002; Siitonen 2012). Although these beetles have not been categorized with a conservation status, they perform an important trophic role as the main prey of the charismatic and keystone species, the Magellanic woodpecker (Campephilus magellanicus), also endemic to the southern beech forests (Vergara and Schlatter 2004). Furthermore, M. magellanicus contributes significantly to the decomposition of dead wood in these forests (Giganti and Dapoto 1990; Zuñiga-Reinoso 2013). Therefore, understanding how this long-horned beetle species responds to habitat degradation at different spatial scales may contribute to the improvement of the conservation strategies for subpolar forest ecosystems.
Numerous studies in the northern hemisphere indicate that long-horned beetle species are sensitive to deforestation at different spatial scales (e.g., Økland et al. 1996; Holland et al. 2004, 2005; Saint-Germain et al. 2006; Sverdrup-Thygeson et al. 2014). These species respond negatively to declining availability of decayed trees because these trees supply long-horned beetles with decaying wood microhabitat required for larval development and pupation (Grove 2002; Jonsson et al. 2006; Buse et al. 2007; Russo et al. 2011; Siitonen 2012; Thorn et al. 2014). Long-horned beetles are usually affected by disturbances occurring at nested spatial levels, including individual trees, forest patches and landscapes (Gibb et al. 2006; Boulanger et al. 2010; Russo et al. 2011; Saint-Germain and Drapeau 2011; Sverdrup-Thygeson et al. 2014). For instance, adult beetles prefer trees that promote the emergence of their progeny (e.g., old decayed trees), but these tree preferences can respond to top-down constraints, with tree choices being influenced by the characteristics of forest patches they had previously selected (e.g., Saint-German et al. 2006; Boulanger et al. 2010). In variegated landscapes, however, long-horned beetles may respond to continuous variation in micro-habitat features and its interaction with the amount of trees available in the landscape. Accordingly, we assessed the occupancy pattern of M. magellanicus as a function of forest cover and remote-sensing-derived tree quality estimates (e.g., Vergara et al. 2016), with both being measured at spatial scales. Our approach involved sampling the occurrence of adult individuals of M. magellanicus using pheromone-baited traps (for increasing detectability) with a sampling design appropriate for multiscale assessment. We used high-resolution remote sensing data to classify individual trees according to their decay stage, thus providing accurate estimates of tree quality at different spatial scales. Lastly, to identify the critical spatial scales that best predict the occupancy probability of M. magellanicus we tested several alternative models that contained the variables forest cover and tree quality measured at different spatial scales.
Methods
Study landscape and species background
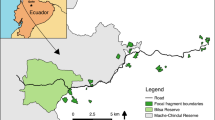
This study was conducted on the northern part of the Navarino Island in the southernmost part of Chile (55°05′S, 67°40′W; Fig. 1). The climate is oceanic, with a mean annual temperature of 6 °C and nearly 500 mm of precipitation. The study area (ca. 480 km2) comprises a variegated forest landscape, with a relatively continuous canopy cover (ca. 68 % covered with native forest) across the landscape exhibiting low fragmentation levels (Fig. 1). Forest vegetation includes stands of deciduous Nothofagus pumilio, mixed stands of N. pumilio-Nothofagus betuloides, and small stands of Nothofagus antarctica around water courses (Pisano 1977). Non-forest areas include peat fields, ponds created by the introduced North American beaver (Castor canadensis), and human-deforested areas covered by open scrub of Berberis microphylla and Junellia tridens.
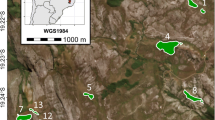
The studied variegated landscape in the northern part of the Navarino Island, in the extreme south of Chile. Bottom Plant Senescence Reflectance Index (PSRI) used as a proxy of tree quality for M. magellanicus (see main text). The trees were identified from a segmentation process carried out using a high-resolution WorldView-2 satellite images. Center Sampling design used to collect adult individuals of M. magellanicus, where intercept traps were arranged in the center of square grid cells located more than 800 m apart. Top Details of two 400 m × 400 m grid cells (with a small and large amount of forest cover) and a cross-vane panel intercept trap hanging from an austral beech (Nothofagus) tree. A male M. magellanicus is shown (photo Mauricio Cid)
Although more than 180 species of long-horned beetles belonging to the Cerambycidae family have been described for Chile (Elgueta 2000), only two long-horned beetle species have been observed in the Navarino Island: M. magellanicus and Callisphyris leptopus. Of these two species, M. magellanicus would be the most common, with their larvae being reported in decaying wood of live or dying trees of N. pumilio, N. betuloides and N. antarctica (Cerda 1986; Barriga et al. 1993; Zúñiga-Reinoso Zuñiga-Reinoso 2013). The studied landscape comprises mature stands with decayed trees covering nearly one-third of the total forest wood (Lombardi et al. 2011).
Field surveys
We collected long-horned beetles using traps in a sampling design based on a 400 m by 400 m square grid (Fig. 1). The sampling procedure consisted in sequential random selection of 50 grid cells, excluding all of its eight neighboring cells in the next cell choice. Each trap (n = 50; see below) was set in the middle of the selected cells. Thus, sampling locations were more than 800 m apart in order to ensure the spatial independence of data while providing enough distance for creating non-overlapping buffers around the traps (see tree quality section below). This sampling design was consistent with the space use pattern of the Magellanic woodpecker, which has a specialized diet based on larvae and adults of M. magellanicus. Specifically, Magellanic woodpeckers have stable home ranges in the Navarino Island, ranging in size from 90 to 250 ha (Soto et al. 2012, 2015; see also Ojeda and Chazarreta 2014).
Adult long-horned beetles were captured using cross-vane panel flight intercept traps, which have proven to be an effective trapping technique for cerambycid beetles (McIntosh et al. 2001; Graham et al. 2010). Since the phenology of M. magellanicus in Navarino Island has not been documented yet, we covered the entire period in which adult long-horned beetles have been reported in Nothofagus forests (see details in Cerda 1986; Zuñiga-Reinoso 2013). Thus, captures were carried out during two seasons: austral spring (Oct–Dec) 2014 and austral summer (Jan–March) 2015. Intercept traps consisted of two transparent plastic panels (35.5 cm wide; 122.0 cm high), crossed perpendicularly and coated with a slippery film of polysiloxane liquid to improve capture efficiency (Fig. 1). Traps were hung from a branch of a N. pumilio or N. betuloides trees ca. 1 m above the ground (Fig. 1). The traps included a plastic funnel that guided beetles into a plastic container which was filled with 300 mL of diluted antifreeze (ethylene glycol) solution, in order to break the surface tension of the water while preserving the collected specimens. Traps were baited with a blend of synthetic racemic pheromone 3R-hydroxyhexan-2-one, designed to attract a broad range of long-horned beetle species (see details in Millar et al. 2009; Graham et al. 2010; Handley et al. 2015). Pheromone lures consisted of polyethylene press-seal sachets (Chem Tica Internacional, S.A.) suspended in the trap centers and replaced at the end of spring 2014. All collected beetles were preserved in 70 % ethyl alcohol for transport to the laboratory for identification at the species level.
Tree quality
Tree quality was quantified through a high-resolution (0.50 and 2.00 m pixel sizes for panchromatic and 8-band multispectral imagery, respectively) WorldView-2 satellite image taken during the austral spring, when deciduous austral beech trees exhibit leaves. The image was firstly preprocessed for radiance and reflectance conversion (Updike and Comp 2010). Then, we improved the contrast and color resolution of the image to identify vegetation cover classes (see above) developed by using a digital supervised classification scheme in ENVI 5.0 (as described in Soto et al. 2012, 2015, 2016 and Vergara et al. 2016).
To identify the crowns of the individual trees, we applied object-based image analysis on the 8-band pan-sharpened images ERDAS Imagine 9.0 (Erdas Inc., Norcross, GA, USA). Following Waser et al. (2014) and using eCognition Developer 8.7 OBIA software (Trimble Germany GmbH, Munich, Germany), we applied a multi-resolution segmentation algorithm based on scale and homogeneity criteria. Image segmentation involved grouping neighboring pixels into regions based on similarity criteria (Waser et al. 2014). The tree crowns derived from the segmentation process were successfully identified when compared with the photo-interpreted and digitized crowns of individual trees. We used a GIS database of the Plant Senescence Reflectance Index (PSRI) to assess the decay stage of individual trees identified from the segmentation process (Soto et al. 2016). PRSI makes the link between the multispectral properties of trees and biochemical-biophysical senescence resulting from degradation of chlorophyll and retention of carotenoids (Merzlyak et al. 1999). Larger values of PSRI indicate a more advanced stage of wood decay (Merzlyak et al. 1999; Yang et al. 2014). PRSI values were averaged on all pixels contained in the crowns of individual trees, with these values being interpreted as a proxy for the quality of each tree for long-horned beetles (Soto et al. 2015, 2016). Based on a parallel study (Soto et al. 2016), tree-level PRSI values were statistically validated by comparing them with the fraction of dead wood observed in the field, as estimated using the method proposed by Vergara and Schlatter (2004) for Nothofagus subpolar forests (Fig. S1, supplementary material).
Habitat characterization
Explanatory habitat variables were estimated at the trap (local) level and the landscape level. First, using 10-m radius circular plots centered on each trap (see above), we quantified attributes of all the nearest trees surrounding the trap (including the tree where it was hung). Tree attributes included: (1) the diameter at breast height (DBH, in cm); (2) tree height (m); (3) tree species composition (% N. pumilio trees); (4) field estimation of dead wood (see above); and (5) pecking intensity, i.e., the number of pecking holes made by Magellanic woodpeckers for removing larvae from the wood (Vergara and Schlatter 2004; Table 1). At the landscape level, forest cover and tree quality were measured at different spatial scales by creating circular buffers around each intercept trap. We considered five different spatial scales: 50, 100, 200, 300 and 400 m, with these scales ensuring data independence among traps (i.e., traps were ≥800 m apart; see above). As indicated above, such a range of spatial scales (50–400 m) is consistent with the space use of the Magellanic woodpecker. Using the resulting vegetation classification (as explained above), we calculated the amount of forest cover as the fraction of Nothofagus forest in the buffer area (Table 1). We estimated the weighted average of the tree-level PRSI values for each spatial scale and used these PRSI estimates as a measured of tree quality in the landscape. Weights were assigned to each individual tree as the relative area covered by its crown into the buffer (Table 1).
Data analysis
We used a Bayesian hierarchical single-species model to assess the occurrence probability (\(\varPsi_{i}\)) of M. magellanicus while accounting for imperfect detection through repeated trapping surveys (Kéry and Schaub 2011). The model assumes that the presence or absence of M. magellanicus at the trap i = 1, 2, N (with N = 50) is a Bernoulli distributed latent variable, \(z_{i} \sim Ber(\varPsi_{i} )\), with z i = 1 if M. magellanicus is present and z i = 0 if it is absent, while \(\varPsi_{i}\) is the probability of M. magellanicus to occur at trap i. The observed presence of M. magellanicus in a trap (\(y_{i}\)) is modelled by assuming a Bernoulli distributed variable, \(y_{ij} \sim Ber(z_{i} \cdot \theta_{i,j} )\), where θ i,j is the detection probability of M. magellanicus during the season j (j = 1 for spring and j = 2 for summer). We assumed that the detection probability of M. magellanicus varies seasonally depending on the phenology of these beetles. Thus, the detection function was specified by a categorical factor representing each surveyed season.
We developed a set of candidate occupancy models assessing the independent (additive) and interactive (multiplicative) effects of forest cover and tree quality. Models involved all possible cross-combinations of forest cover and tree quality at different spatial scales (i.e., 52 models; see model selection procedure below). In all candidate models we retained the effect of local-scale habitat variables (Table 1), but those variables were not used to analyse cross-scale interactions because our model selection procedure required including the same variable over different spatial scales. We used principal component analysis (PCA) to reduce the number of local-scale variables (Table 1) and avoid potential multicollinearity problems. Only PCA factors with eigenvalues (standard deviations) greater than one were used in posterior analysis (Table S1, supplementary material). The resulting PCA scores showed that only the first factor can be considered to be meaningful (as indicated by its eigenvalue >1; Table S1). Such a first PCA factor, which had a moderate loading (>0.5) on tree height, tree DBH, and dead wood (Table S1), was included as a model covariate representing the local habitat conditions. Tree quality values were transformed to vary between 0 and 1 in order to make easy comparisons among the effects of forest cover, tree quality and their interaction (see the distribution of these covariates in Fig. S2, supplementary material). Thus, the occurrence probability of M. magellanicus in trap i (\(\varPsi_{i}\)) was modelled by using the following logit function:
where \(\beta_{1}\) (l = 0, 1,.., 4) are the fixed-effect model coefficients (including an intercept) and \(\in_{i}\) is a spatially structured random intercept for each trap. Covariates in Eq.(1) are as follows: (1) \(TQ(k)_{i}\) is the tree quality at spatial scale k; (2) \(FC(h)_{i}\) is the forest cover at spatial scale h; (3)\(TQ(k)_{i} \times FC(h)_{i}\) is the interaction between tree quality and forest cover measured on scales k and h, respectively; and (4) \(LH_{i}\) represents local habitat conditions around trap i. The \(TQ(k) \times FC(h)\) terms included cross-scale interactions (for h ≠ k) and the product between forest cover and tree quality measured at the same spatial scale (h = k). Forest cover and tree quality estimates (at whatever spatial scale) were not correlated (r < 0.32; Table S2), thus satisfying the orthogonality condition required to evaluate the interaction between independent covariates. Since \(TQ(k)\) represents the expected (mean) quality of all trees at scale k, its interaction (product) with \(FC(h)\), \(TQ(k) \times FC(h)\), was interpreted as proportional to the number of high-quality trees available at these spatial scales.
We used a model-selection procedure intended to compare the goodness of fit of models representing the additive and interactive effects of forest cover and tree quality at different spatial scales. The support of the 25 candidate models (i.e., including all 5 × 5 cross-scale combinations) was assessed by using the Deviance Information Criteria (DIC). For each candidate model, its difference in DIC (ΔDIC) with the best-supported model (i.e., the model with the least DIC value) was used to interpret its strength of evidence, with models with ΔDIC < 2 being considered to be equally supported by the data. In order to determine if the candidate models supported by the data achieved satisfying predictions, we use the Area Under the estimated ROC Curve (AUC), which represents the ability of the model to correctly classify, or discriminate between, the traps with and without detections of M. magellanicus. In a subsequent analysis, we assessed how ΔDIC and AUC values of the candidate models changed with spatial scale by using penalized regression splines (mgcv-package of R, R Core Team 2015). This smoothing method uses cross-validation to determine the effective degrees of freedom (df) of the models, with df near 0 indicating a linear relationship (Wood 2006).
The importance of fixed effect coefficients of the best supported models was evaluated for each model separately by examining its Bayesian Credible Intervals (BIC) and its significance level (i.e., p value), both estimated from the posterior distribution of parameters sampled from MCMC (Markov Chain Monte Carlo) chains. In particular, p-values were estimated as the proportion of the sampled coefficients that were >0 (if the mean coefficient was positive) or <0 (otherwise). We assumed a normal distribution for the logit function in Eq.(1). We used vague non-informative prior distributions for all model parameters. Parameter distributions were based on three MCMC chains, each with 20,000 iterations, discarding the first 10,000 iterations and thinning by 3. We used the Potential Scale Reduction factor and visual observation to examine MCMC Convergence. Models were run using OpenBUGS via the R2OpenBUGS package of R.
Results
Data from two of the 50 traps were discarded from the analysis because these traps were damaged during the spring. A total of 14 individuals of M. magellanicus were recorded in 12 of the 48 (25 %) intercept traps, while the overall occurrence probability (ψ) was 0.46 (Table 2). No other species of long-horned beetle were recorded in the traps. During spring M. magellanicus individuals were detected in only three (6.3 %) traps, while in the austral summer they were captured in 11 (23 %) traps, with detection probability increasing 3.2 times from spring to summer (Table 2).
We found similar support (ΔDIC ≤ 2) for seven models explaining the \(\varPsi\) of M. magellanicus. The best-supported candidate models included the tree quality at scales ≥200 m only. Conversely, forest cover did not exhibit scale-specific effects on \(\varPsi\), with the best-supported models including forest cover measured at all spatial scales studied (50, 100, 200, 300 and 400 m; Fig. 2; Table S3, supplementary material). Four of the seven best-supported models included some cross-scale combination of tree quality (TC) and forest cover (FC), i.e., TQ(300 m)–FC(50 m), TQ(200 m)–FC(400 m), TQ(300 m)–FC(100 m), TQ(200 m)–FC(300 m) (Table S3; Fig. 2). The remaining three best-supported models included tree quality and forest cover measured at the same scale: TQ(200 m)–FC(200 m), TQ(300 m)–FC(300 m) and TQ(400 m)–FC(400 m) (Table S3; Fig. 2). The Area under the ROC curve (AUC) was consistent, and positively associated, with ΔDIC values. Indeed, the best-supported models had AUC values ≥0.94, which on the average were larger than the AUC values of the unsupported (ΔDIC > 2) models, whose AUC values averaged 0.92 and ranged between 0.89 and 0.94 (Table S3; Fig. 2). Fitted smooth curves and the effective degree of freedom of penalized splines indicated that model support and performance, as estimated through ΔDIC and AUC values, respectively, increased monotonically with the increasing scale at which tree quality was measured (Fig. 2). Conversely, we did not find a significant association between the scale at which forest cover was quantified with the ΔDIC and AUC values (Fig. 2).
Differences in deviance information criteria (ΔDIC) (on the left) and the area under the Roc curve (AUC) (on the left) for candidate Bayesian models explaining the occurrence probability of M. magellanicus as a function of tree quality and forest cover measured at five different spatial scales (50, 100, 200, 300 and 400 m). Top A two dimensional diagram representing ΔDIC and AUC values of the 25 candidate models. Center scatter plots of the association of forest cover on both ΔDIC and AUC values, including a loess (local regression) model fitted to the data. A degree of freedom (df) near zero indicates a linear relationship with the p value representing the significance of the effect. Below scatter plots for the association of tree quality with ΔDIC and AUC values, such as explained for the forest cover plots
Coefficients from the best-supported models indicated that the effect of the TQ × FC interaction was stronger than the additive effect of tree quality, and particularly than the additive effect of forest cover, as shown by their p-values and 95 % Bayesian Credible Intervals (BCI in Table 3; Fig. 3). The TQ × FC effect was significant, or marginally significant (i.e., with p < 0.062), only in the models that included tree quality at spatial scales ≥200 m (Table 3; Fig. 3). However, the p-value of the TQ × FC effect decreased more markedly (from p = 0.024 to p = 0.037) with increasing forest cover than with increasing the tree quality, as shown by the smoothing regression fitted to the coefficient’s p values of all candidate models (Fig. 4). The additive effect of tree quality was not significant, except for tree quality averaged at a scale of 300 m, which had a marginally significant positive effect on Ψ (Table 3; Fig. 3). The additive forest cover effect was not significant for any of the five spatial scales studied, while we did not find a significant local habitat effect (Table 3; Fig. 3). The difference between the interactive and additive effects of forest cover and tree quality can be observed in the contour plots of Fig. 5, where the predicted ψ values are plotted for the first and second best-supported models (i.e., ΔDIC ≤ 1; Table S3). In the model including a marginally significant effect of tree quality (Fig. 5a), Ψ increases markedly (from 0.1 to 1.0) when tree quality exceeds the average quality in the landscape, but it remains relatively invariant with increasing the forest cover. In the model including a significant TQ × FC effect (Fig. 5b), contour isolines of Ψ show that a reduction in forest cover is compensated by high tree quality values, and vice versa. However, the convex shape of the isolines in Fig. 5b also indicates that increased Ψ values should occur with a simultaneous and balanced increase in both forest cover and tree quality.
Model coefficients from the seven best supported (ΔDIC < 2) Bayesian models explaining the occurrence probability of M. magellanicus as a function of tree quality and forest cover measured at five different spatial scales (50, 100, 200, 300 and 400 m). The coefficients are those associated with the additive effect of tree quality (TQ), forest cover (FC), and the cross-scale interactions between these two covariates (TQ × FC)
A three-dimensional smoothing scatter-plot regression for the significance (p-value) of cross-scale (TQ × FC) interactions as a function of tree quality (TQ) and forest cover (FC) at different spatial scales (from 0 to 400 m). We used p-values from Bayesian models explaining the occurrence probability of M. magellanicus (Table 2)
Contour plots showing model-predicted occurrence probabilities (ψ) of M. magellanicus as a function of tree quality, forest cover, and the cross-scale interaction between these two covariates (see Eq. 1). Contour isolines indicate combinations of the two covariates predicting a particular ψ level, while vertical and horizontal dashed lines indicate the mean value of tree quality, forest cover, as measured for the corresponding spatial scale
Discussion
The results of this study suggest that the occupancy pattern of M. magellanicus in a variegated forest landscape is consistent with the quantity of high-quality trees available in the landscape, as interpreted from the positive and significant effect of the interaction between tree quality and forest cover (TQ × FC). Our results also indicate that the TQ × FC interaction has a stronger effect than tree quality and forest cover, but also that the TQ × FC effect varies with the spatial scale at which tree quality and forest cover are measured. Ecological processes emerging from these findings can be understood as follow:
First, although forest cover contributed to explain the occurrence probability ψ of M. magellanicus through its interaction with tree quality, occupancy models showed that the additive effect of forest cover is negligible. The lack of such a forest cover effect contrasts with the plethora of studies supporting the strong association of saproxylic long-horned beetles with forest amount (e.g., see Økland et al. 1996; Holland et al. 2004, 2005; Saint-Germain et al. 2006; Sverdrup-Thygeson et al. 2014). These studies, however, have usually assessed forest cover at spatial scales larger than those used in this study (e.g., Holland et al. 2005 and herein). Some methodological techniques have been developed to assess the effect of forest cover at large spatial scales without compromising the independence of the data (Holland et al. 2004). Unfortunately, to date no approach has been developed to model the complex patterns of occurrence and detection probabilities. Although we do not know the effect of forest cover at spatial scales greater than 400 m, we did not see an increase in the support (ΔDIC) and performance (AUC) of the models when the spatial scale for forest cover was increased (see Fig. 2). Since the results indicate that M. magellanicus individuals are not sensitive to the amount of forest per se, we suggest that their sensitivity to forest loss should be assessed in fragmented landscapes, with forest cover reaching critical values for M. magellanicus.
Second, the positive response of M. magellanicus to the significant TQ × FC interactions suggests that its abundance and population size is directly associated with the quantity of old, decayed and dying Nothofagus trees available at scales ≥200 m (see Table S3). These results are consistent with saproxylic long-horned beetles of the northern hemisphere, which are sensitive to the quality and quantity of decayed trees (e.g., Similä et al. 2002; Jacobs et al. 2007; Cobb et al. 2011). Since those decayed trees act as an obligate substrate for the larvae and pupae of M. magellanicus (Cerda and Angulo 2002; Zuñiga-Reinoso 2013), the positive TQ × FC effect should result from different ecological processes occurring at the individual and population levels, including habitat selection, dispersal, recruitment and survival. Adult long-horned beetles may select forest stands containing high-quality trees after dispersing on long distances (Russo et al. 2011). Similarly, the scales of the significant TQ × FC interactions suggest that adult M. magellanicus could move over distances >200 m before selecting an individual tree, so they would make tree selection decisions based on the number of high-quality trees available at these spatial scales. Indeed, the variegated landscape studied here, which exhibits a continuous rather than a patchy structure, can promote the functional connectivity for adult M. magellanicus (e.g., see Schiegg 2000). Indeed, an increased number of high quality trees in the landscape not only may lead to the massive emergence of adult beetles, but also it could have consequences on metapopulation dynamics when combined with a high functional connectivity. Such a high dispersal rate through a connected landscape should contribute to the demographic rescue of local populations occupying mature stands with decayed trees (e.g., Hanski 2009).
Studies addressing the habitat degradation effects on saproxylic long-horned beetles rely on accurate estimations of the decay stage of the trees. These studies usually include variables measured either directly on small field plots (e.g., Russo et al. 2011) or extracted from GIS land-cover data (e.g., Laaksonen et al. 2008). The former studies do not provide habitat quality estimates at the landscape scale, while the latter can lead to spurious relationships. Recent development of remote sensing systems, however, has provided landscape-scale metrics based on fine-grained estimates of habitat quality for species with specialized habitat requirements (e.g., Santos et al. 2010; Jacobsen et al. 2015). Our findings supported the use of PSRI as a proxy of the habitat quality for long-horned beetles, at least at the landscape level. The advantages of habitat quality estimation derived from remote sensing systems, as used in this study, are its applications in landscape management for organisms sensitive to forest degradation. We suggest that management strategies intended to conserve M. magellanicus in variegated forest landscapes should be based on the quality of trees at spatial scales >200 m. Firewood extraction and the presence of the introduced beavears (Castor canadensis) represent the main causes of forest disturbances in the study landscape. Thus, the conservation of viable metapopulations of M. magellanicus involves maintaining old-growth forest conditions through reducing firewood extraction while controlling the beaver-caused forest loss. Furthermore, such a landscape planning could also contribute to preserve the population of other species that interact with M. magellanicus, such as the Magellanic woodpeckers.
References
Adriaens D, Jacquemyn H, Honnay O, Hermy M (2009) Conservation of remnant populations of Colchicum autumnale: the relative importance of local habitat quality and habitat fragmentation. Acta Oecol 35:69–82
Artigas J (1994) Entomología Económica: Insectos de interés agrícola, forestal, médico y veterinario (nativos, introducidos y susceptibles de ser introducidos). Ediciones Universidad de Concepción. 2 volúmenes
Barriga J, Curkovic T, Fichet T, Henríquez J, Macaya J (1993) Nuevos antecedentes de coleópteros xilófagos y plantas hospederas en Chile, con una ecopilación de citas previas. Rev Chile Entomol 20:65–91
Boulanger Y, Sirois L, Hebert C (2010) Distribution of saproxylic beetles in a recently burned landscape of the northern boreal forest of Quebec. For Ecol Manage 260:1114–1123
Boyce MS (2006) Scale for resource selection functions. Divers Distrib 12:269–276
Buse J, Schröder B, Assmann T (2007) Modelling habitat and spatial distribution of an endangered longhorn beetle—a case study for saproxylic insect conservation. Biol Conserv 137:372–381
Cerda M (1986) Lista sistemática de los cerambícidos chilenos (Coleoptera: Cerambycidae). Rev Chile Entomol 14:29–39
Cerda L, Angulo A (2002) Insectos asociados a bosques del centro sur de Chile. En: Baldini A. y Pancel, L. 2002. Agentes de Daño en el Bosque nativo. Editorial Universitaria, pp 201–268
Cobb TP, Morissette JL, Jacobs JM, Koivula MJ, Spence JR, Langor DW (2011) Effects of post fire salvage logging on deadwood-associated beetles. Conserv Biol 25:94–104
Cody ML (1985) Habitat selection in birds. Academic Press, Orlando
Davis J, Debinski D, Danielson B (2007) Local and landscape effects on the butterfly community in fragmented Midwest USA prairie habitats. Landscape Ecol 22:1341–1354
Doligez B, Berthouly A, Doligez D, Tanner M, Saladin V, Bonfils D, Richner H (2008) Spatial scale of local breeding habitat quality and adjustment of breeding decisions. Ecology 89:1436–1444
Drielsma M, Ferrier S (2009) Rapid evaluation of metapopulation persistence in highly variegated landscapes. Biol Conserv 142:529–540
Elgueta M (2000) Coleoptera de Chile. In: Martín-Piera F, Morrone JJ, y Melic A (eds) Hacia un Proyecto CYTED para el Inventario y Estimación de la Diversidad Entomológica en Iberoamérica: Monografías Tercer Milenio vol. 1. SEA, Zaragoza, pp 145–154
Fischer J, Lindenmayer DB (2007) Landscape modification and habitat fragmentation: a synthesis. Glob Ecol Biogeogr 16:265–280
Fretwell SD, Lucas HL Jr (1970) On territorial behavior and other factors influencing habitat distribution in birds. I. Theoretical development. Acta Biotheor 19:16–36
Gibb H, Hjalten J, Ball JP, Atlegrim O, Pettersson RB, Hilszczanski J, Johansson T, Danell K (2006) Effects of landscape composition and substrate availability on saproxylic beetles in boreal forests: a study using experimental logs for monitoring assemblages. Ecography 29:191–204
Giganti H, Dapoto G (1990) Coleoptera of the native forests in the Department of Aluminé (Neuquén-Argentina). Bosque 11:37–44
Graham EE, Mitchell RF, Reagel PF (2010) Treating panel traps with a fluoropolymer enhances their efficiency in capturing cerambycid beetles. J Econ Entomol 103:641–647
Griffen BD, Drake JM (2008) Effects of habitat quality and size on extinction in experimental populations. Proc R Soc B 275:2251–2256
Grove SJ (2002) Saproxylic insect ecology and the sustainable management of forests. Annu Rev Ecol Syst 33:1–23
Hall LS, Krausman PR, Morrison ML (1997) The habitat concept and a plea for standard terminology. Wildl Soc Bull 25:171–182
Handley K, Hough-Goldstein J, Hanks LM, Millar JG, D’amico V (2015) Species richness and phenology of cerambycid beetles in urban forest fragments of northern Delaware. Ann Entomol Soc Am 108(3):251–262
Hanski I (2009) Incorporating the spatial configuration of the habitat into ecology and evolutionary biology. In: Ecology Spatial (ed) Cantrell S, Cosner C, Shigui Ruan S. Chapman and Hall/CRC New York, NewYork, pp 167–188
Heisswolf A, Reichmann S, Poethke HJ, Schrader B, Obermaier E (2009) Habitat quality matters for the distribution of an endangered leaf beetle and its egg parasitoid in a fragmented landscape. J Insect Conserv 13:165–175
Holland JD, Bert DG, Fahrig L (2004) Determining the spatial scale of species’ response to habitat. Bioscience 54:227–233
Holland JD, Fahrig L, Cappuccino N (2005) Body size affects the spatial scale of habitat-beetle interactions. Oikos 110:101–108
Hutto RL (1985) Habitat selection by nonbreeding, migratory land birds. In: Cody ML (ed) Habitat selection in birds. Academic Press, Orlando, pp 455–476
Jacobs J, Spence JR, Langor DW (2007) Influence of forest succession and dead wood qualities on boreal saproxylic beetles. Agric For Entomol 9:3–16
Jacobsen RM, Sverdrup-Thygeson A, Birkemoe T (2015) Scale-specific responses of saproxylic beetles: combining dead wood surveys with data from satellite imagery. J Insect Conserv 19:1053–1062
Johnson DH (1980) The comparison of usage and availability measurements for evaluating resource preference. Ecology 61:65–71
Jonsson M, Ranius T, Ekvall H, Bostedt G, Dahlberg A, Ehnström B, Nordén B, Stokland JN (2006) Cost-effectiveness of silvicultural measures to increase substrate availability for redlisted wood-living organisms in Norway spruce forests. Biol Conserv 127:443–462
Johnson MD (2007) Measuring habitat quality: a review. Condor 109:489–504
Kéry M, Schaub M (2011) Bayesian population analysis using winbugs—a hierarchical perspective. Academic Press, Waltham
Kristan WB III (2006) Sources and expectations for hierarchical structure in bird-habitat associations. Condor 108:5–12
Laaksonen M, Peuhu E, Varkonyi G, Siitonen J (2008) Effects of habitat quality and landscape structure on saproxylic species dwelling in boreal spruce-swamp forests. Oikos 117:1098–1110
Lindenmayer DB, Fischer J (2007) Tackling the habitat fragmentation panchreston. Trends Ecol Evol 22:127–132
Lombardi F, Cocozza C, Lasserre B, Tognetti R, Marchetti M (2011) Dendrochronological assessment of the time since death of dead wood in an old growth Magellan’s beech forest, Navarino Island (Chile). Austral Ecol 36:329–340
Mason DC, Anderson GQA, Bradbury RB, Cobby DM, Davenport IJ, Vandepoll M, Wilson JD (2003) Measurement of habitat predictor variables for organism-habitat models using remote sensing and image segmentation. Int J Remote Sens 24:2515–2532
Mayor SJ, Schneider DC, Schaefer JA, Mahoney SP (2009) Habitat selection at multiple scales. Ecoscience 16:238–247
McIntosh RL, Katinic PJ, Allison JD (2001) Comparative efficacy of five types of trap for woodborers in the Cerambycidae, Buprestidae and Siricidae. Agric For Entomol 3:113–120
McIntyre S, Barrett GW (1992) Habitat variegation, an alternative to fragmentation. Conserv Biol 6(1):146–147
Merzlyak MN, Gitelson AA, Chivkunova OB, Rakitin VYU (1999) Non-destructive optical detection of pigment changes during leaf senescence and fruit ripening. Physiol Plant 106:135–141
Millar JG, Hanks LM, Moreira JA, Barbour JD, Lacey ES (2009) Pheromone chemistry of cerambycid beetles. In: Nakamuta K, Millar JG (eds) Chemical ecology of wood-boring insects. Forestry and Forest Products Research Institute, Ibaraki, pp 52–79
Morrison ML, Marcot BG, Mannan RW (2006) Wildlife-habitat relationships: concepts and applications, 3rd edn. Island Press, Washington, DC
Mortelliti A (2013) Targeting habitat management in fragmented landscapes: a case study with forest vertebrates. Biodivers Conserv 22:187–207
Mortelliti A, Amori G, Boitani L (2010) The role of habitat quality in fragmented landscapes: a conceptual overview and prospectus for future research. Oecologia 163:535–547
Mortelliti a, Sozio G, Boccacci F, Ranchelli E, Cecere JG, Battisti C, Boitani L (2012) Effect of habitat amount, configuration and quality in fragmented landscapes. Acta Oecol 45:1–7
O’Neill RV, Johnson AR, King AW (1989) A hierarchical framework for the analysis of scale. Landscape Ecol 3:193–205
Ojeda V, Chazarreta L (2014) Home range and habitat use by Magellanic woodpeckers in an old-growth forest of Patagonia. Can J For Res 44:1265–1273
Økland B, Bakke A, Hagvar S (1996) What factors influence the diversity of saproxylic beetles? A multiscaled study from a spruce forest in southern Norway. Biodiver Conserv 5:75–100
Peters DPC, Peilke RA Sr, Bestelmeyer BT, Allen CD, Munson-McGee S, Havstad KM (2004) Cross scale interactions, nonlinearities, and forecasting catastrophic events. Proc Natl Acad Sci USA 101:15130–15135
Pisano E (1977) Fitogeografía de Fuego-Patagonia chilena I.- Comunidades vegetales entre las latitudes 52° y 56°S. 8:121–250. Anales del Instituto de la Patagonia (Chile) 8:121–250
R Core Team (2015) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria
Russo D, Cistrone L, Garonna AP (2011) Habitat selection by the highly endangered long-horned beetle Rosalia alpina in southern Europe: a multiple spatial scale assessment. J Insect Conserv 15:685–693
Saint-Germain M, Buddle CM, Drapeau P (2006) Sampling saproxylic Coleoptera: scale issues and the importance of behavior. Environ Entomol 35:478–487
Saint-Germain M, Drapeau P (2011) Response of saprophagous wood-boring beetles (Coleoptera: Cerambycidae) to severe habitat loss due to logging in an aspen-dominated boreal landscape. Landscape Ecol 26:573–586
Sala OE, Chapin Iii FS, Armesto JJ, Berlow E, Bloomfield J, Dirzo R, Huber-Sanwald E, Huenneke LF, Jackson RB, Kinzig A, Leemans R, Lodge DM, Mooney HA, Oesterheld M, Poff NL, Sykes MT, Walker BH, Walker M, Wall DH (2000) Global biodiversity scenarios for the year 2100. Science 287(5459):1770–1774
Santos MJ, Greenberg JA, Ustin SL (2010) Using hyperspectral remote sensing to detect and quantify southeastern pine senescence effects in Red-cockaded woodpecker (Picoides borealis) habitat. Remote Sens Environ 114:1242–1250
Schiegg K (2000) Effects of dead wood volume and connectivity on saproxylic insect species diversity. Écoscience 7:290–298
Schooley RL, Branch LC (2007) Spatial heterogeneity in habitat quality and cross-scale interactions in metapopulations. Ecosystems 10:846–853
Siitonen J (2012) Microhabitats. In: Stokland J, Siitonen J, Jonsson BG (eds) Biodiversity in dead wood. Cambridge University Press, United Kingdom, pp 151–182
Similä M, Kouki J, Martikainen P, Uotila A (2002) Conservation of beetles in boreal pine forests: effects of forest age and naturalness on species assemblages. Biol Conserv 106:19–27
Soto GE, Perez-Hernández CG, Hahn IJ, Rodewald AD, Vergara PM (2016) Tree senescence as a direct measure of habitat quality: linking red-edge vegetation indices to space use by magellanic woodpeckers. Remote Sens Environ (in press)
Soto GE, Vergara PM, Lizama ME, Celis C, Rozzi R, Duron Q, Hahn IJ, Jiménez JE (2012) Do Beavers improve the habitat quality for Magellanic woodpeckers? Bosque 33:271–274
Soto GE, Vergara PM, Perez-Hernández CG (2015) Home-range scale’s attributes limiting the habitat selection pattern on magellanic woodpecker. In: 100th ESA Annual Meeting, Baltimore, USA, 9–14 Aug 2015
Sverdrup-Thygeson A, Gustafsson L, Kouki J (2014) Spatial and temporal scales relevant for conservation of dead-wood associated species: current status and perspectives. Biodivers Conserv 23:513–535
Thomas CD (2000) Dispersal and extinction in fragmented landscapes. Proc R Soc Lond B 267:139–145
Thorn S, Bässler C, Gottschalk T, Hothorn T, Bussler H, Raffa K, Müller J (2014) New insights into the consequences of post-windthrow salvage logging revealed by functional structure of saproxylic beetles assemblages. PLoS One 9(7):e101757. http://doi.org/10.1371/journal.pone.0101757
Updike T, Comp C (2010) Radiometric use of Worldview-2 imagery, DigitalGlobe. Technical Note. DigitalGlobe®, Colorado
Verboom J, Schotman A, Opdam P, Metz JAJ (1991) European Nuthatch metapopulations in a fragmented agricultural landscape. Oikos 61:149–156
Vergara PM, Armesto JJ (2009) Responses of Chilean forest birds to anthropogenic habitat fragmentation across spatial scales. Landscape Ecol 24:25–38
Vergara PM, Saura S, Pérez-Hernández C, Soto GE (2015) Hierarchical spatial decisions in fragmented landscapes: modeling the foraging movements of woodpeckers. Ecol Model 300:114–122
Vergara PM, Schlatter RP (2004) Magellanic woodpecker (Campephilus magellanicus) abundance and foraging in Tierra del Fuego, Chile. J Ornithol 145:343–351
Vergara PM, Soto GE, Moreira-Arce D, Rodewald AD, Meneses LO, Perez-Hernandez CG (2016) Foraging behaviour in magellanic woodpeckers is consistent with a multi-scale assessment of tree quality. PLoS One 11(7):e0159096
Waser LT, Küchler M, Jütte K, Stampfer T (2014) Evaluating the potential of WorldView-2 data to classify tree species and different levels of ash mortality. Remote Sens 6:4515–4545
Wettstein W, Schmid B (1999) Conservation of arthropod diversity in montane wetlands effect of altitude, habitat quality and habitat fragmentation on butterflies and grasshoppers. J Appl Ecol 36:363–373
Wiens JA (1989) Spatial scaling in ecology. Funct Ecol 3:385–397
Wood SN (2006) Generalized additive models: an introduction with R. CRC Press, Boca Raton
Yang X, Tang J, Mustard J (2014) Beyond leaf color: comparing camera-based phenological metrics with leaf biochemical, biophysical, and spectral properties throughout the growing season of a temperate deciduous forest. J Geophys Res 119:181–191
Zuñiga-Reinoso A (2013) Review of the Longhorns (Coleoptera: cerambycidae) from Magellanes region: illustrated checklist. Anales Instituto Patagonia (Chile) 41:53–59
Acknowledgments
This study was supported by FONDECYT Grant 1131133. GES acknowledges Wesley Hochachka from the Cornell Lab of Ornithology and Mario Nazar for their technical support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Vergara, P.M., Meneses, L.O., Grez, A.A. et al. Occupancy pattern of a long-horned beetle in a variegated forest landscape: linkages between tree quality and forest cover across spatial scales. Landscape Ecol 32, 279–293 (2017). https://doi.org/10.1007/s10980-016-0443-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10980-016-0443-1