Abstract
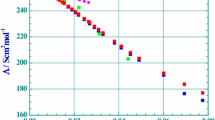
Literature values of the electrical conductivities of dilute aqueous solutions of trivalent rare earth hexacyanoferrates(III) (La, Pr, Nd, Sm, Gd) and of rare earth hexacyanocobaltates(III) (La, Nd, Sm, Y) were reexamined within the framework of the Quint-Viallard conductivity equation in order to obtain a uniform representation of their conductivities. It was observed that the limiting conductances of electrolytes at infinite dilution depend weakly on the applied conductivity equation, whereas the derived ion association equilibrium constants vary considerably and therefore should be treated rather as fitting parameters.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Apelblat, A., Neueder, R., Barthel, J.: Electrolyte Data Collection. Electrolyte Conductivities and Dissociation Constants of Aqueous Solutions of Organic Monobasic Acids CH2O2–C7H14O3, vol. XII, Part 4a. Dechema Chemistry Data Series, Frankfurt (2004)
Apelblat, A., Neueder, R., Barthel, J.: Electrolyte Data Collection. Electrolyte Conductivities and Dissociation Constants of Aqueous Solutions of Organic Monobasic Acids C8H5NO2–C7H14O3, vol. XII, Part 4b. Dechema Chemistry Data Series, Frankfurt (2005)
Apelblat, A., Neueder, R., Barthel, J.: Electrolyte Data Collection. Electrolyte Conductivities and Dissociation Constants of Aqueous Solutions of Organic Dibasic and Tribasic Acids, vol. XII, Part 4c. Dechema Chemistry Data Series, Frankfurt (2006)
Atkinson, G.: Ionic association in polyvalent symmetrical electrolytes. I. Conductances of several rare earth hexacyanocobaltates(III) in dioxane–water mixtures. J. Am. Chem. Soc. 82, 818–820 (1960)
Bhat, D.M., Davies, C.W., Stock, D.I.: Solubility and conductance of lanthanum ferricyanide in water. J. Chem. Soc. (A) 1682–1684 (1969)
Bianchi, H.L., Dujovne, I., Fernández-Prini, R.: Comparison of electrolytic conductivity theories: performance of classical and new theories. J. Solution Chem. 29, 237–253 (2000)
Buchner, R., Barthel, J., Gill, B.: La[Fe(CN)6] ion pairing in aqueous solution. A dielectric relaxation study. Phys. Chem. Chem. Phys. 1, 105–109 (1999)
Conway, B.E.: Ionic interactions and activity behaviour of electrolyte solutions. In: Conway, B.E., Bockris, J. O’M., Yeager, E. (eds.) Comprehensive Treatise of Electrochemistry, vol. 5, pp. 111–222. Plenum Press, New York (1983)
Davies, C.W.: Ion Association. Butterworths, Washington (1962)
Davies, C.W., James, J.C.: The conductivity of aqueous solutions of lanthanum ferricyanide. Proc. R. Soc. Lond. Ser. A, Math. Phys. Sci. 195, 116–123 (1948)
Dunsmore, H.S., James, J.C.: The electrolytic dissociation of magnesium sulphate and lanthanum ferricyanide in mixed solvents. J. Chem. Soc. 2925–2930 (1951)
Dunsmore, H.S., Kelly, T.R., Nancollas, G.H.: Electrochemical studies in the rare earth series. Part 3. Conductances of some 3-3 electrolytes in water and dioxane + water. Trans. Faraday Soc. 59, 2606–2611 (1963)
Fiddler, A.: Conductivity of three trivalent electrolytes. Collect. Czechoslov. Chem. Commun. 37, 758–761 (1972)
Fidler, A.: Conductivity of three trivalent electrolytes. Collect. Czechoslov. Chem. Commun. 37, 458–461 (1972)
Hamann, S.D., Pearce, P.J., Strauss, W.: The effect of pressure on the dissociation of lanthanum ferricyanide ion pairs in water. J. Phys. Chem. 68, 375–380 (1964)
Harned, H.S., Owen, B.B.: The Physical Chemistry of Electrolytic Solutions, 3rd edn. Reinhold, New York (1958)
James, J.C., Monk, C.B.: The conductivities of potassium and lanthanum cobalticyanides and ferricyanides. Trans. Faraday Soc. 46, 1041–1050 (1950)
Kielland, J.: Individual activity coefficients of ions in aqueous solutions. J. Am. Chem. Soc. 59, 1675–1678 (1937)
Lemire, R.J., Lister, M.W.: Conductivity measurements and relative association constants of some 3:1 electrolytes. J. Solution Chem. 5, 171–181 (1976)
Malatesta, F.: Activity coefficients of electrolytes from the emf of liquid membrane cells. IV: Revised activity coefficients of lanthanum hexacyanoferrate(III). J. Solution Chem. 24, 241–252 (1995)
Malatesta, F., Giacomelli, A., Zamboni, R.: Activity coefficients of electrolytes from the emf of liquid membrane cells. III: LaCl3, K3Fe(CN)6, and LaFe(CN)6. J. Solution Chem. 23, 11–36 (1994)
Malatesta, F., Trombella, S., Giacomelli, A., Onor, M.: Activity coefficients of 3:3 electrolytes in aqueous solutions. Polyhedron 19, 2493–2500 (2000)
Monk, C.B.: Conductances of some lanthanide cobalticyanides in dioxane–water. A re-assessment. J. Am. Chem. Soc. 82, 5762–5763 (1960)
Monk, C.B.: Electrolytic Dissociation. Academic Press, London (1961)
Panckhurst, M.H., Woolmington, K.G.: A spectrophotometric study of ionic association in aqueous solutions. Proc. R. Soc. Lond. Ser. A, Math. Phys. Sci. 244, 124–139 (1958)
Quint, J.: Contribution a l’étude de la conductibilité électrique de mélanges d’électrolytes. Ph.D. Thesis, University of Clermont-Ferrand, April (1976)
Quint, J., Viallard, A.: Relaxation field for the general case of electrolyte mixtures. J. Solution Chem. 7, 137–153 (1978)
Quint, J., Viallard, A.: The electrophoretic effect for the case of electrolyte mixtures. J. Solution Chem. 7, 525–531 (1978)
Quint, J., Viallard, A.: Electrical conductance of electrolyte mixtures of any type. J. Solution Chem. 7, 533–548 (1978)
Robinson, R.A., Stokes, R.H.: Electrolyte Solutions, 2nd edn. rev. Butterworths, London (1965)
Spedding, F.H., Dye, J.L.: Conductances, transference numbers and activity coefficients of aqueous solutions of some rare earth chlorides at 25 °C. J. Am. Chem. Soc. 76, 879–881 (1954)
Spedding, F.H., Jaffe, I.S.: Conductances, transference numbers and activity coefficients of aqueous solutions of some rare earth halides at 25 °C. J. Am. Chem. Soc. 74, 4751–4755 (1952)
Spedding, F.H., Jaffe, S.: Conductances, solubilities and ionization constants of some rare earth sulfates in aqueous solutions at 25 °C. J. Am. Chem. Soc. 76, 882–884 (1954)
Spedding, F.H., Jaffe, S.: Conductances, transference numbers and activity coefficients of some rare earth perchlorates and nitrates at 25 °C. J. Am. Chem. Soc. 76, 884–888 (1954)
Tsurko, E.N., Neueder, R., Barthel, J., Apelblat, A.: Conductivity of phosphoric acid, sodium, potassium, and ammonium phosphates in dilute aqueous solutions from 278.15 K to 308.15 K. J. Solution Chem. 28, 973–999 (1999)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Apelblat, A. Representation of Electrical Conductances for Polyvalent Electrolytes by the Quint-Viallard Conductivity Equation. Part 2. Symmetrical 3:3 Type Electrolytes. Dilute Aqueous Solutions of Rare Earth Hexacyanoferrates(III) and Hexacyanocobaltates(III). J Solution Chem 40, 1724–1734 (2011). https://doi.org/10.1007/s10953-011-9748-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-011-9748-5