Abstract
Human-modified landscapes play an important role in supporting the survival of primate populations, but they may also facilitate human–primate interactions, possibly leading to negative outcomes. We conducted a scalar investigation of the role of landscape structure in shaping the intensity of crop feeding by a generalist primate species, the rhesus macaque (Macaca mulatta) in northern India. At the species level, we used behavioral sampling techniques to assess habitat selection by two groups of rhesus macaques. At the landscape level, we used GIS-based analyses to quantify landscape structure in terms of class-level and patch-level metrics and correlated it with macaque crop feeding intensities. We found that, on average, both study groups spent only 15% of their total feeding time on cultivars. However, they spent a large proportion of their feeding (BH: 75% and CH: 36%) and ranging (BH: 72% and CH: 39%) time in agroecosystem habitats. Landscape level analysis showed that crop feeding intensity was not related to total area under cultivation. Instead, macaque crop feeding intensity was positively correlated to the arrangement of deciduous forest patches beside cultivated area patches. Our findings call for careful appraisal of landscape management practices as a potential mitigation strategy for primate crop-depredation in such human-modified landscapes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The use of human-modified areas by different nonhuman primate species (hereafter primates) is well documented. Primate species often use anthropogenically modified areas for feeding, temporary refuge, or movement between patches (Anderson et al. 2007; Hockings and McLennan 2012; LaFleur and Gould 2009; Onderdonk and Chapman 2000; Pozo-Montuy et al. 2013). This increases the potential for interactions between humans and primates (Hockings and McLennan 2012; Onderdonk and Chapman 2000; Priston and McLennan 2013). The form and nature of human–primate interactions are shaped by many social and ecological factors, including the material landscape (Li and Essen 2020), and although some interactions may result in neutral or beneficial outcomes for both groups, others often have negative consequences, leading to a state of conflict between the two groups (Radhakrishna et al. 2013). Certain types of anthropogenic land use such as shaded coffee, cocoa, rubber, and timber plantations may support primate species without extensive damage to human-cultivated resources (Estrada and Coates-Estrada 1996; Williams-Guillén et al. 2006). However, other types of land use contain resources (such as crops and plantations) that are susceptible to damage by primate species (Goldman et al. 2008; Hockings and McLennan 2012; Priston and McLennan 2013; Ruppert et al. 2018). Primates feeding on human-cultivated resources is a major cause of human–primate conflict and this poses a serious threat to the survival and coexistence of primate species (Boulton et al. 1996; Hill 2005; Meijaard et al. 2011; Nekaris et al. 2013).
Studies of the ecological drivers of primate crop feeding observe that intensities of crop feeding vary in space (Linkie et al. 2007; Warren 2008) and that the intensities decrease with increasing distance of farms from forest edge (Lemessa et al. 2013; Linkie et al. 2007; Saj et al. 2001). Research on habitat selection by primates indicates that the extent and intensity of crop feeding is also driven by landscape composition and configuration (Mochizuki and Murakami 2011a,b, 2013; Yamada and Muroyama 2010). Landscape composition refers to the different types and extent of land covers present in the landscape, while landscape configuration refers to their relative arrangements in the landscape (Turner and Gardner 2015). Long-term changes in landscape configuration leading to spatial redistribution of food resources have led to increased crop feeding by Japanese macaques (Agetsuma 2007; Mochizuki and Murakami 2011a,b, 2013). This suggests that negative human–primate interactions arising from primate crop feeding activities may be directly linked to specific landscape compositions and configuration.
The enormous expansion of agricultural activities has led to the emergence of agroecosystems as one of the most common types of human-modified environments. Agroecosystems refer to ecosystems where natural flora and/or fauna have been replaced (partially or completely) by human crop plants and livestock animals (Altieri and Koohafkan n.d.). Depending on existing management regimes, agroecosystems may vary in form and characteristics (Estrada et al. 2012; Weibull et al. 2003). Land use practices such as mono-cropping and clear-cutting produce simple agroecosystems like pasturelands that are largely devoid of natural vegetation. In contrast, a land use practice such as agroforestry produces a complex agroecosystem that retains native vegetation in and around farm complexes (Altieri and Koohafkan n.d.; Vandermeer 2003). Complex agroecosystems are diverse in structure and heterogeneous in habitat composition and support the survival of a number of primate species (Anderson et al. 2007; Estrada 2006; Estrada et al. 2012; LaFleur and Gould 2009; Raboy et al. 2004). Understanding how landscape composition and configuration influence habitat selection and use by primate species in complex agroecosystems would lend greater insights into the management of primate crop feeding in such landscapes (Mekonnen et al. 2020).
The rhesus macaque (Macaca mulatta) is a generalist species and is known to exploit a wide variety of habitats (Gupta 2001; Jaman and Huffman 2013; Riley and Wade 2016). Consumption of cultivars by rhesus macaques leads to negative interactions between humans and rhesus macaques in many locations across the geographical range of the species (Anand et al. 2018; Huang et al. 2018; Regmi et al. 2013). Studies of human–rhesus macaque interactions have addressed varied aspects such as quantification of crop damage (Engeman et al. 2010; Sahoo and Mohnot 2004; Singh and Thakur 2012), attitudes and perceptions of affected communities (Ahsan and Uddin 2014; Anand et al. 2018; Chauhan and Pirta 2010; Saraswat et al. 2015), impacts of human provisioning on ecological functions of rhesus macaques (Sengupta et al. 2015) and mitigation techniques to address negative interactions between rhesus macaques and humans (Dhiman and Mohan 2014; Imam et al. 2002; Sambyal et al. 2009; Pebsworth and Radhakrishna 2020). Less is understood about how the topography of the landscape that is cohabited by rhesus macaques and humans influences crop feeding levels by rhesus macaques. Therefore, the aim of the current study was to understand how landscape composition and configuration influence the intensity of crop feeding by rhesus macaques (Macaca mulatta), in an agroecosystem-dominated landscape in northern India. Previous studies of the influence of landscape topography on macaque crop feeding underscore the importance of considering multiple spatial scales for a holistic understanding of this issue (Mochizuki and Murakami 2011a,b, 2013). Hence, we conducted our investigation at two spatial scales: species’ habitat selection at the home range/local scale and effect of landscape heterogeneity at the regional/state-level scale. The objectives of our study were to 1) assess the degree of use of different habitat types by rhesus macaques and evaluate its impacts on the intensity of crop feeding and 2) investigate the relationship between landscape structure and intensity of crop feeding by rhesus macaques.
Methods
Study Area
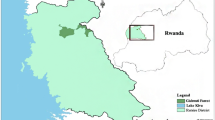
We conducted our study in Solan district (30.70–31.25 N and 76.70–77.33 E, elevation: 300–3000 m) in the state of Himachal Pradesh, northern India (Fig. 1). Solan district is in the southwestern part of the state that falls within the Shiwalik region. Solan experiences significant crop damage due to rhesus macaques and has been classified as high conflict zone in the state (Singh and Thakur 2012).
Habitat Types in the Study Area
The land cover in the study site was a matrix of forest patches, cultivated land, and open land. We used the 2014–15 land use/land cover (LULC) map prepared by the Indian Space Research Organization (ISRO) to define the habitat types in the study area. The 2014–15 ISRO-LULC map identified the following land use/land cover classes in the study area: evergreen, plantation (fruit species such as plum, pears, pomegranate, and/or timber species such as Acacia catechu, Bauhinia variegate, Celtis australis, Cedrella toona, built up, deciduous, degraded/scrub, wasteland, grassland, zaid (summer) crop (cucumber, brinjal , tomato, bitter gourd, bottle gourd), double/triple crop, rabi (winter) crop (wheat, barley, gram, mustard, potato, onion), kharif (monsoon) crop (maize, tomato, beans, capsicum), current fallow. As one of the goals of our study was to evaluate how rhesus macaques used different habitat types, we categorized the various land use classes into three main habitat types: evergreen or pine forest, agroecosystem, and scrubland/grassland. Pine forest refers to the homogeneous area consisting predominantly of evergreen pine trees (Pinus roxburghii). Agroecosystem included different types of cultivated land (zaid crop, double/triple crop, rabi crop, kharif crop), current fallow, deciduous vegetation patches, and plantation areas. Scrubland/grassland comprised degraded/scrubland areas, wasteland, grassland, and built-up areas.
Study Group
We selected two groups of rhesus macaques (BH and CH) that regularly engaged in feeding on cultivars for our behavioral observations. Group size was 46 for BH group (adult male: 8; adult female: 10, adult female with infant: 6; subadult male: 8; subadult female: 2; juvenile: 12) and 40 for CH group (adult male: 4; adult female: 6, adult female with infant: 6; subadult male: 5; subadult female: 5; subadult with unidentified sex: 2, juvenile: 12). Before beginning behavioral sampling, we spent about 2 weeks locating and habituating the study groups to observer presence. Both study groups ranged in human-dominated environment and appeared accustomed to human presence; possibly because of this, they did not avoid observer presence.
Data Collection Methods
From March 2016 to November 2016 and January 2017 to June 2017, we followed the macaque groups (BH and CH) and collected data on their activity and ranging pattern. We typically conducted follows from 08:00 h to 18:00 h every day for about 5 days every week. The mean hours of observation per day for both groups was 3.60 hours (BH: range = 0.25–7.67 h, CH: range = 0.25–8.16 h). The mean hours of observation per month for BH group was 46.45 h (range: 14.65–116.8 h) and for CH group it was 9.85 h (range: 2.00–20.42 h). We used instantaneous scan sampling with 15 min intervals to record the following activities of visible individuals: moving, resting, social interactions, feeding, and other (Altmann 1974). (Electronic Supplementary Material Table SI provides a description of activities.) When feeding activity occurred, we recorded the name of the plant species and its category, i.e., cultivated or wild food. We included only adult and subadult individuals in the scans. To determine the ranging pattern of study individuals, we used a hand-held GPS Garmin eTrex 30X to record the location of the approximate geographic center of the group at 15-min intervals (Fashing 2001). If the group stayed at a location for >15 min, we recorded the next location point after the group moved. We also recorded the habitat types in which rhesus macaque individuals were present.
For the landscape level analysis, we used published records of rhesus macaque-caused crop damage (Singh and Thakur 2012) in the different districts of Himachal Pradesh and GIS-based analysis of land use/land cover in the state. The 2014–15 LULC map used for this was prepared by ISRO at the scale 1:50,000. The overall accuracy of land use/land cover classes varies from 79% to 97% (https://bhuvan-app1.nrsc.gov.in/thematic/thematic/index.php).We used FRAGSTATS software to calculate the following three indices to quantify landscape structure: Class Area, Number of Patches, and Euclidean Nearest Neighbor. Class Area is the area occupied by different land use classes in the landscape, while Number of Patches refers to the total number of patches belonging to a specific land use class. Euclidean Nearest Neighbor distance is the “shortest straight-line distance between the focal patch and its nearest neighbour of the same class” (McGarigal 2015, p. 133). We used Class Area and Number of Patches to quantify the presence of different types of land uses (i.e., evergreen pine forest, deciduous vegetation, plantations, cultivated area, scrubland/grassland) and Euclidean Nearest Neighbor metric to quantify the arrangement of cultivated areas in the different districts of the state.
Data Analysis
We calculated the mean daily proportions of activity categories of study macaques to estimate the overall activity budget. We used one sample proportion test to check for the differences in the feeding time on wild food resources and cultivars. Following Mekonnen et al. (2017), we calculated the home ranges of study groups using kernel density estimation (KDE) and minimum convex polygon (MCP) methods. To estimate home ranges, we used ad hoc fixed KDE with 95% isopleths (contour lines) and core area with 50% isopleths. For home range analysis using MCP, we used 95% and 100% MCPs. To analyze habitat selection by study groups, we calculated the habitat selection ratio within the 95% KDE home range by dividing the frequency of observed use by the frequency of expected use (Manly et al. 2002). We defined frequency of observed use as percentage of observed scans in each habitat type (pine forest, agroecosystem, and scrubland/grassland). We defined frequency of expected use as number of scans expected to fall in each habitat type according to its area proportion. We considered a habitat selection ratio value close to 1 as indicative of No Selection, more than 1 to indicate Selection, and less than 1 to indicate Avoidance (Mekonnen et al. 2017). We calculated the area under each habitat type using the 2014–15 LULC map prepared by ISRO.
For landscape-level analyses, we considered district as the unit of analysis, in accordance with the published records of rhesus macaque crop damage (Singh and Thakur 2012) in the state. We considered the proportion of villages affected by rhesus macaque crop damage in a district (from Singh and Thakur 2012) as a measure of macaque crop feeding intensity for that district. To assess the relationship between Class Area and Number of Patches of different land use types and macaque crop feeding intensity, we first summed all the Class Areas and Number of Patches of each land use class in a district and then did a pairwise correlation between these values for all districts and crop feeding intensity values for the districts using Spearman’s correlation test. To assess the relationship between the arrangement of cultivated area patches and crop feeding intensities, we first estimated the Euclidean nearest neighbor distances for cultivated areas for all districts. We then classified districts into three categories of macaque crop feeding intensity: low (proportion of affected villages <50%), medium (proportion of affected villages 50%–75%), and high (proportion of affected villages >75%) and used ANOVA followed by Tukey post hoc tests to check for statistically significant differences between Euclidean nearest neighbor distances values for different categories of districts.
Ethical Note
The study adhered to the International Primatological Society (IPS) Code of Best Practices for Field Primatology. The research protocol used to collect data for this study was approved by institutional ethics committee of National Institute of Advanced Studies. We also obtained required study permits from Himachal Pradesh Forest Department for carrying out the field study (No. WL/Research-Study/228).
Data Availability
The datasets of the current study are available from the corresponding author on reasonable request.
Results
Habitat Selection
We collected 1947 scans (28951 individual records) for BH group and 1088 scans (14939 individual records) for CH group, amounting to a total of 325 h of observation for BH group and 181 h of observation for CH group. Both groups spent similar amounts of time in different activities; the largest proportion of time was taken up by feeding and foraging (BH: 40%, CH: 39%), followed by resting (BH: 23%, CH: 21%), locomotion (BH: 16%, CH: 17%), social activities (BH: 14%, CH: 16%), and other activities (BH: 7%, CH: 8%). Both groups spent a significantly higher proportion of their feeding time (BH: 85%, 5868/6903 feeding records; CH: 85%, 2576/3031 feeding records) on wild-food resources as compared to the feeding time on cultivars (15%, BH: 1035/6903 feeding records, CH: 455/3031) (one sample proportion test, z = 71.379, P < 0.001). The groups differed in the amount of feeding time spent in different parts of the habitat. While BH spent most (75%, 5196/6903 feeding records) of their feeding time in agroecosystems, CH spent most of their feeding time in scrubland and grassland habitats (56%, 1701/3031 feeding records). CH spent 36% (1080/3031 feeding records) of their feeding time in agroecosystems and BH spent 21% (4161/6903 feeding records) of their time in scrubland and grassland habitats. Both groups spent little time in pine forests (BH: 4%; 247/6903 feeding records; CH: 8%; 251/3031 feeding records).
Overall, rhesus individuals from both groups fed on 42 different plant species (34 wild species and 8 cultivars) belonging to 27 plant families and a variety of grasses (Table I). The groups differed in the number of plant species that they fed on: BH group fed on all 42 plant species and a variety of grasses, whereas CH group fed on a total of 35 plant species (30 wild species and 5 cultivars) and a variety of grasses. We did not distinguish grasses into different species, so we present them as a single category. Although study individuals fed on a variety of plant species, they spent much of their feeding time on a few species: three and four species (including grasses) accounted for more than 50% of the total feeding time of BH and CH group respectively (Table I). Grasses accounted for ca. 25% of total feeding time for both groups (BH: 25.83%, CH: 24.25%). More than three-fourths (76.74%) of the plant species (N = 33 (deciduous species: 24, cultivars: 8, grasses: 1) fed on by rhesus macaques were present in agroecosystem habitats, while the remaining were present in scrubland/grassland area (deciduous species: nine) and forest area (coniferous species: 1).
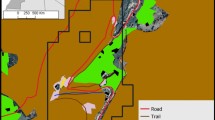
The 95% KDE home range for BH and CH groups was 7.03 km2 and 3.66 km2, respectively (Fig. 2). The proportion of different habitat types differed across the groups’ home ranges. Approximately two-thirds (65.57%) of BH’s home range was composed of agroecosystem (4.61 km2), followed by scrubland/grassland (26.5%, 1.86 km2) and pine forest (8%, 0.56 km2). For CH group, scrubland/grassland was the prominent habitat type occupying 50% of their home range (1.84 km2). Agroecosystem accounted for 38% of the home range (1.40 km2), followed by pine forest (11%, 0.41 km2) (Table II). The time spent by both groups in different habitat types differed. BH group spent 72% of their ranging time in agroecosystem, while for CH group it was 39%. Scrubland amounted for 24% of ranging time for BH group and 50% of ranging time for CH. Both groups spent the least amount of time in pine forest (BH: 4%, CH: 11%). The habitat selection ratio (Table III) indicated that BH group selected agroecosystem and avoided pine forest. No selection was observed in the case of scrubland/grassland habitat type. We did not observe selection for any habitat type in the case of CH group (Table III).
Landscape Structure and Intensity of Crop Feeding
There was no relationship between crop feeding intensity and total area under cultivation (Pearson’s correlation test, r = 0.50, P = 0.13, N = 10). The intensity of crop feeding increased when the number of cultivated patches increased with respect to the number deciduous vegetation patches (Pearson’s correlation test, r = 0.632, P = 0.05, N=10) and crop feeding intensity was negatively correlated with mean patch area of degraded/scrubland (Pearson’s correlation test, r = −0.77, P = 0.009, N = 10). The distribution of cultivated area patches, measured in the terms of Euclidean nearest neighbor distance, differed significantly across the three categories of districts (i.e., crop feeding intensity categories) (one way ANOVA, df (2), F = 69.75, P < 0.001). The mean Euclidean nearest neighbor for low-intensity districts was significantly higher (mean: 175.0 ± SD 234.77, range: 103.03–8781.90, N = 9097) than the medium-intensity (mean: 163.31 ± SD 165.51, range: 103.03–6788.89, N = 27316) and high-intensity districts (mean: 151.66 ± SD 128.22, range: 103.08–5478.77, N = 22,023) (Tukey post hoc test, all P < 0.001).
Discussion
Investigations into the landscape drivers of spatial patterns of primate crop feeding typically highlight the role of factors such as distance of farms from forest edge and crop type (Lemessa et al. 2013; Linkie et al. 2007). Very few studies have paid attention to the relationship between relative landscape composition and crop feeding intensities (Mochizuki and Murakami 2011a,b, 2013; Yamada and Muroyama 2010). Findings from the current study support the view that landscape composition and configuration critically impact the intensity of crop feeding by primates.
The results of the state-level geospatial analyses provide interesting insights into macaque crop feeding intensities as a function of landscape structure. First, we did not find a significant relationship between macaque crop feeding intensity and total area under cultivation. This result implies that although increase in agricultural land is strongly linked with primate crop feeding (Sillero-Zubiri and Switzer 2001), factors such as the location of the agricultural patch and its association with other landscape variables may be more important drivers for primate crop feeding than crop type and quantity.
Our second finding regarding a negative correlation between mean patch size of degraded/scrub vegetation and crop feeding intensity indicates that landscapes that are largely composed of sparse vegetation without tall trees experience little crop damage, possibly because they do not support higher numbers of rhesus macaques. This may be because such areas do not provide suitable refuge sites to primates. This hypothesis is supported by our study group observations, that CH group showed no active selection for scrubland habitat, although it was the predominant habitat type in the group’s home range.
Third, the Euclidean nearest neighbor distance analysis of cultivated area patches showed that landscapes with a greater proportion of area experiencing macaque crop feeding exhibited less segregation of cultivated area patches. Low segregation of cultivated patches means high contiguity between cultivated patches, leaving almost no area available for other land uses. Our field observations confirm that in such scenarios, the intervening spaces are typically occupied by thin strips of deciduous vegetation, as farming practices in the study region involve the maintenance of thin strips of deciduous vegetation between farms to provide fodder/fuelwood resources. Rhesus macaques depend on these deciduous vegetation patches for their feeding requirements (as supported by our study group observations); hence, such landscapes act as a major attractant for the species and therefore these habitats are characterized by high rates of negative interactions between farmers and macaques. By acting as a convenient refuge site for macaques engaged in crop feeding, the deciduous patches also decrease the effectiveness of traditional crop guarding methods such as chasing. In comparison, districts with lower proportion of area experiencing macaque crop feeding exhibit higher segregation of cultivated area patches. Relatively larger areas between cultivated area patches tend to be occupied by coniferous forest patches or scrubland/grassland. As both these habitats have low resource abundance, they do not support a higher number of macaques and hence such landscapes display comparatively lower degree of crop damage by rhesus macaques.
Although the landscape-level analysis provides a general understanding of how landscape structure and composition drive macaque crop feeding intensity patterns across the region, the study site observations are crucial to understand the variations that exist within. Our site observations showed that, depending on the proportion of agroecosystem habitats within their home ranges, study macaque groups differentially used agroecosystems for feeding and ranging. However, both groups spent the same, comparatively small amount of time feeding on cultivars. These findings suggest that rhesus macaque-caused crop damage is not primarily driven by macaques feeding on crops. Instead, access to agroecosystem habitats (through their proximity to forest patches), and the relative ease in navigating through them, due to the presence of the deciduous vegetation patches, may drive macaque crop damage intensities.
Persistent visibility of rhesus macaques during their use of agroecosystem resources increases farmers’ perceptions of rhesus macaque crop feeding. Studies of human–wildlife conflict show that people usually blame big and highly conspicuous species for all the crop depredations they experience, although the actual role of the species may be limited or even misunderstood (Hill 1997; Hockings 2016; Holzner et al. 2019; Knight 2000; Linkie et al. 2007). In the case of crop damage by rhesus macaques too, the conspicuous presence of rhesus macaques in and around farms results in farmers believing that macaques cause heavy crop depredations which, in turn, may lead to heightened perceptions of human–rhesus conflict. Studies have shown that while documentation of farmers’ perceptions regarding primate caused crop damage and observations of primate crop feeding can provide insights into the sources of negative interactions between human and primates (Kansky and Knight 2014; Riley 2007; Treves et al. 2006), the data obtained through such methods do not accurately represent actual crop damage (Mishra 1997; Naughton-Treves and Treves 2005). For a more comprehensive understanding of actual crop damage, independent assessments of crop loss or participatory risk mapping techniques combined with studies of farmers’ perceptions of crop damage can help identify misconceptions about the impact of primates in agroecosystems (Holzner et al. 2019; Riley 2007; Ruppert et al. 2018).
Feeding data from the study groups clearly indicate that the presence of deciduous vegetation plays a major role in the active selection of agroecosystems by macaques. Other studies have underscored the importance of broad-leaved, deciduous vegetation in providing required resources for macaques (Mochizuki and Murakami 2013; Yamada and Muroyama 2010). The study macaques’ dependency on these deciduous vegetation patches for their feeding requirements may be driven by the gymnosperm-dominated forest vegetation in the landscape, which is generally considered low-quality habitat in terms of feeding resources for primates (Imaki et al. 1994). The greater proportion of their ranging time in agroecosystems may also be driven by the safe refuge sites that these deciduous vegetation patches offer (Imaki et al. 1994). Our field observations showed that unlike the tall trees in agroecosystem habitats, deciduous vegetation patches in scrubland/grassland habitats were typically shrubs or herbs and did not offer any refuge to rhesus individuals. This may explain why the study macaques spent less time in scrubland/grassland habitat as compared to agroecosystem habitat, although they contain some deciduous vegetation. The presence of safe refuge sites is particularly important in locations such as the current study site, as rhesus macaques are habitually chased by farmers and dogs as a part of farm guarding measures.
Overall, the results of our study highlight the role of particular configurations of landscape components in driving primate crop feeding levels in a mosaic landscape. These configurations may be the product of region-specific land management practices and therefore it is necessary to investigate landscape structure at multiple spatial scales for a comprehensive understanding of how landscape topography can influence primate crop feeding. Findings from our study thus have important implications for the management of human–rhesus macaque conflict in Himachal Pradesh and in other landscapes of similar characteristics and for human conflict with other taxa. While removal of these deciduous patches may reduce the intensity of crop feeding (Hocking et al. 2015), farming communities actively maintain such vegetative patches, as they provide several direct (fodder, fuelwood, and timber) and indirect benefits (prevention of soil erosion and retention of soil moisture and organic matters) (Anderson et al. 2007). An alternate strategy would be to focus on the feeding preferences of rhesus macaques (Sengupta and Radhakrishna 2016). Our results show that a few wild plant species accounted for most of the feeding time of study rhesus individuals. Replacing some of these tree species in the vegetation patches with plant species that are not significantly consumed or preferred by rhesus macaques would reduce incentives for rhesus macaques to visit the crop–field complexes. It would also enable farmers to continue obtaining resources from these patches. Additionally, plant species used by macaques for feeding can be planted near forest edges and between forest–scrubland transition zones, to increase natural resource availability for macaques (Hill 2017; Hocking and Humle 2009; Mikich and Liebsch 2014).We also recommend a cautious appraisal of the relative costs and benefits of existing farming methods and agroecosystem management practices before any drastic measures (such as macaque culling, sanctioned by the government “vermin” order—Ministry of Environment, Forest and Climate Change 2017) are taken to mitigate conflict intensities. Carefully evaluated habitat modification measures may be the most reliable long-term mitigation strategy for rhesus crop feeding in such human-modified landscapes.
References
Agetsuma, N. (2007). Ecological function losses caused by monotonous land use induce crop raiding by wildlife on the island of Yakushima, southern Japan. Ecological Research, 22, 390–402. https://doi.org/10.1007/978-4-431-73238-9_4.
Ahsan, M. F., & Uddin, M. M. (2014). Human-rhesus monkey conflict at Rampur Village under Monohardi Upazila in Narsingdi District of Bangladesh. Journal of Threatened Taxa, 6(6), 5905–5908.
Altieri, M. A., & Koohafkan, P. (n.d.). Globally Important Ingenious Agricultural Heritage Systems (GIAHS): Extent, significance, and implications for development. http://www.fao.org/docrep/015/ap021e/ap021e.pdf
Altmann, J. (1974). Observational study of behavior: Sampling methods. Behaviour, 49(3/4), 227–267.
Anand, S., Binoy, V. V., & Radhakrishna, S. (2018). The monkey is not always a god: Attitudinal differences toward crop-raiding macaques and why it matters for conflict mitigation. Ambio, 47(6), 711–720. https://doi.org/10.1007/s13280-017-1008-5.
Anderson, J., Rowcliffe, J. M., & Cowlishaw, G. (2007). Does the matrix matter? A forest primate in a complex agricultural landscape. Biological Conservation, 135(2), 212–222. https://doi.org/10.1016/j.biocon.2006.10.022.
Boulton, A. M., Horrocks, J. A., & Baulu, J. (1996). The Barbados vervet monkey (Cercopithecus aethiops sabaeus): Changes in population size and crop damage, 1980–1994. International Journal of Primatology, 17(5), 831–844. https://doi.org/10.1007/BF02735267.
Chauhan, A., & Pirta, R. S. (2010). Agonistic interactions between humans and two Species of Monkeys (rhesus monkey Macaca mulatta and Hanuman langur Semnopithecus entellus) in Shimla, Himachal Pradesh. Journal of Psychology, 1(1), 9–14.
Dhiman, S. P., & Mohan, L. (2014). Prospects of managing human-rhesus monkey conflict in Himachal Pradesh, India. In Human-wildlife conflict in the mountains of SAARC region: Compilation of successful management strategies and practices (pp. 48–60). SAARC Forestry Centre.
Engeman, R. M., Laborde, J. E., Constantin, B. U., Shwiff, S. A., Hall, P., Duffiney, A., & Luciano, F. (2010). The economic impacts to commercial farms from invasive monkeys in Puerto Rico. Crop Protection, 29(4), 401–405. https://doi.org/10.1016/j.cropro.2009.10.021.
Estrada, A. (2006). Human and non-human primate co-existence in the Neotropics: A preliminary view of some agricultural practices as a complement for primate conservation. Ecological and Environmental Anthropology, 2(2), 17–29 http://digitalcommons.unl.edu/icwdmeea/3/.
Estrada, A., & Coates-Estrada, R. (1996). Tropical rain forest fragmentation and wild populations of primates at Los Tuxtlas, Mexico. International Journal of Primatology, 17(5), 759–783. https://doi.org/10.1007/BF02735263.
Estrada, A., Raboy, B. E., & Oliveira, L. C. (2012). Agroecosystems and primate conservation in the tropics: A review. American Journal of Primatology, 74(8), 696–711. https://doi.org/10.1002/ajp.22033.
Fashing, P. J. (2001). Activity and ranging patterns of guerezas in the Kakamega Forest: Intergroup variation and implications for intragroup feeding competition. International Journal of Primatology, 22(4), 549–577. https://doi.org/10.1023/A:1010785517852.
Goldman, A., Hartter, J., Southworth, J., & Binford, M. (2008). The human landscape around the Island Park: Impacts and responses to Kibale National Park. In R. Wrangham & E. Ross (Eds.), Science and conservation in African forests: The benefits of long-term research (pp. 129–144). Cambridge University Press. https://doi.org/10.1017/CBO9780511754920.014
Gupta, A. K. (2001). Status of the primates in Tripura. In ENVIS bulletin: Wildlife and protected areas, non-human primates of India (pp. 127–135). Wildlife Institute of India.
Hill, C. M. (1997). Crop-raiding by wild vertebrates: The farmer’s perspective in an agricultural community in western Uganda. International Journal of Pest Management, 43(1), 77–84. https://doi.org/10.1080/096708797229022.
Hill, C. M. (2005). People, crops and primates: A conflict of interests. In J. Paterson & J. Wallis (Eds.), Commensalism and conflict: The human-primate interface (pp. 40–59). American Society of Primatologists.
Hill, C. M. (2017). Crop raiding. In The International Encyclopedia of Primatology, 1–5. https://doi.org/10.1002/9781119179313.wbprim0109
Hockings, K. J. (2016). Mitigating human-nonhuman primate conflict. In The International Encyclopaedia of Primatology, 1–8. https://doi.org/10.1002/9781119179313.wbprim0053
Hockings, K. J., Anderson, J. R., & Matsuzawa, T. (2009). Use of wild and cultivated foods by chimpanzees at Bossou, Republic of Guinea: Feeding dynamics in a human-influenced environment. American Journal of Primatology, 71(8), 636–646. https://doi.org/10.1002/ajp.20698.
Hockings, K. J., & McLennan, M. R. (2012). From forest to farm: Systematic review of cultivar feeding by chimpanzees—management implications for wildlife in anthropogenic landscapes. PLoS ONE, 7(4). https://doi.org/10.1371/journal.pone.0033391.
Hockings, K. J., McLennan, M. R., Carvalho, S., Ancrenaz, M., Bobe, R., Byrne, R. W., Dunbar, R. I. M., Matsuzawa, T., McGrew, W. C., Williamson, E. A., Wilson, M. L., Wood, B., Wrangham, R. W., & Hill, C. M. (2015). Apes in the Anthropocene: Flexibility and survival. Trends in Ecology and Evolution. https://doi.org/10.1016/j.tree.2015.02.002
Holzner, A., Ruppert, N., Swat, F., Schmidt, M., Weiß, B.M., Villa, G., Mansor, A., Sah, S.A.M., Engelhardt, A., Kühl, H. and Widdig, A. (2019). Macaques can contribute to greener practices in oil palm plantations when used as biological pest control. Current Biology, 29(5), R1066-R1067. https://doi.org/10.1016/j.cub.2019.09.011.
Huang, C., Li, X. Y., Shi, L. J., & Jiang, X. L. (2018). Patterns of human-wildlife conflict and compensation practices around Daxueshan Nature Reserve, China. Zoological Research, 39(6), 406–412. https://doi.org/10.24272/j.issn.2095-8137.2018.056.
Imaki, H., Koganezawa, M., & Maruyama, N. (1994). Habitat selection and forest edge use by Japanese monkeys in the Nikko and Imaichi Area, Central Honshu, Japan. Biosphere Conservation, 7(2), 87–96.
Imam, E., Yahya, H. S. A., & Malik, I. (2002). A successful mass translocation of commensal rhesus monkeys Macaca mulatta in Vrindaban, India. Oryx, 36(1), 87–39. https://doi.org/10.1017/S0030605301000011.
Jaman, M. F., & Huffman, M. A. (2013). The effect of urban and rural habitats and resource type on activity budgets of commensal rhesus macaques (Macaca mulatta) in Bangladesh. Primates, 54(1), 49–59. https://doi.org/10.1007/s10329-012-0330-6.
Kansky, R., & Knight, A. T. (2014). Key factors driving attitudes towards large mammals in conflict with humans. Biological Conservation, 179, 93–105.
Knight, J. (2000). Natural enemies: People-wildlife conflicts in anthropological perspective. Routledge.
Lafleur, M., & Gould, L. (2009). Feeding outside the forest: The importance of crop raiding and an invasive weed in the diet of gallery forest ring-tailed lemurs (Lemur catta) following a cyclone at the Beza Mahafaly Special Reserve, Madagascar. Folia Primatologica, 80(3), 233–246. https://doi.org/10.1159/000240968.
Lemessa, D., Hylander, K., & Hambäck, P. (2013). Composition of crops and land-use types in relation to crop raiding pattern at different distances from forests. Agriculture, Ecosystems and Environment, 167, 71–78. https://doi.org/10.1016/j.agee.2012.12.014.
Li, W., & Essen, E. V. (2020). Guarding crops from monkey troops: farmer-monkey interaction near a nature reserve in Guangxi, China. Environmental Sociology, 7(1), 12–24. https://doi.org/10.1080/23251042.2020.1811004.
Linkie, M., Dinata, Y., Nofrianto, A., & Leader-Williams, N. (2007). Patterns and perceptions of wildlife crop raiding in and around Kerinci Seblat National Park, Sumatra. Animal Conservation, 10(1), 127–135. https://doi.org/10.1111/j.1469-1795.2006.00083.x.
Manly, B.F.J., McDonald, L.L., Thomas, D.L., McDonald, T.L., Erickson, W.P., (2002). Resource Selection by Animals: Statistical Design and Analysis for Field Studies (2nd ed). Kluwer Academic Publishers.
McGarigal, K. (2015). FRAGSTATS help. http://www.umass.edu/landeco/research/fragstats/documents/fragstats.help.4.2.pdf
Meijaard, E., Buchori, D., Hadiprakarsa, Y., Utami-Atmoko, S. S., Nurcahyo, A., Tjiu, A., Prasetyo, D., Nardiyono, Christie, L., Ancrenaz, M., Abadi, F., Antoni, I. N. G., Armayadi, D., Dinato, A., Ella, Gumelar, P., Indrawan, T. P., Kussaritano, Munajat, C., Priyono, C. W. P., et al. (2011). Quantifying killing of orangutans and human-orangutan conflict in Kalimantan, Indonesia. PLoS ONE, 6(11), 1–10. https://doi.org/10.1371/journal.pone.0027491
Mekonnen, A., Fashing, P. J., Bekele, A., Hernandez-Aguilar, R. A., Rueness, E. K., Nguyen, N., & Stenseth, N. C. (2017). Impacts of habitat loss and fragmentation on the activity budget, ranging ecology and habitat use of Bale monkeys (Chlorocebus djamdjamensis) in the southern Ethiopian Highlands. American Journal of Primatology, 79(7), 1–13. https://doi.org/10.1002/ajp.22644.
Mekonnen, A., Fashing, P. J., Bekele, A., & Stenseth, N. C. (2020). Use of cultivated foods and matrix habitat by Bale monkeys in forest fragments: Assessing local human attitudes and perceptions. American Journal of Primatology, 82(4), 1–12. https://doi.org/10.1002/ajp.23074.
Mikich, S. B., & Liebsch, D. (2014). Assessment of food supplementation and surveillance as techniques to reduce damage caused by black capuchin monkeys Sapajus nigritus to forest plantations. Current Zoology, 60(5), 581–590. https://doi.org/10.1093/czoolo/60.5.581.
Ministry of Environment Forest and Climate Change. (2017). Notification for declaration of rhesus macaque as a vermin species. http://hpforest.nic.in/upload/contents/File-148.pdf
Mishra, C. (1997). Livestock depredation by large carnivores in the Indian trans-Himalaya: conflict perceptions and conservation prospects. Environmental Conservation, 24, 338–343. https://doi.org/10.1017/S0376892997000441.
Mochizuki, S., & Murakami, T. (2011a). Change in habitat selection by Japanese macaques (Macaca fuscata) and habitat fragmentation analysis using temporal remotely sensed data in Niigata Prefecture, Japan. International Journal of Applied Earth Observation and Geoinformation, 13(4), 562–571. https://doi.org/10.1016/j.jag.2011.02.006.
Mochizuki, S., & Murakami, T. (2011b). The influence of forest boundary shape and environmental variables on farmland invasion by wild Japanese macaques. Journal of Forest Planning, 16, 223–231.
Mochizuki, S., & Murakami, T. (2013). Scale dependent effects in resource selection by crop-raiding Japanese macaques in Niigata Prefecture, Japan. Applied Geography, 42, 13–22. https://doi.org/10.1016/j.apgeog.2013.04.016.
Naughton-Treves, L., & Treves, A. (2005) Socio-ecological factors shaping local support for wildlife: Crop-raiding by elephants and other wildlife in Africa. In R. Woodroffe, S. Thirgood, & A. Rabinowitz (Eds.), People and wildlife: Conflict or coexistence? (pp. 253–277). Cambridge University Press.
Nekaris, K. A. I., Boulton, A., & Nijman, V. (2013). An ethnoprimatological approach to assessing levels of tolerance between human and commensal non-human primates in Sri Lanka. Journal of Anthropological Sciences, 91, 219–231. https://doi.org/10.4436/JASS.91008.
Onderdonk, D. A., & Chapman, C. A. (2000). Coping with forest fragmentation: The primates of Kibale National Park, Uganda. International Journal of Primatology, 21(4), 587–611. https://doi.org/10.1023/A:1005509119693.
Pebsworth, P., & Radhakrishna, S. (2020). Using conditioned taste aversion to reduce human-nonhuman primate conflict: A comparison of four potentially illness-inducing drugs. Applied Animal Behaviour Science, 225(May 2019), 104948. https://doi.org/10.1016/j.applanim.2020.104948.
Pozo-Montuy, G., Serio-Silva, J. C., Chapman, C. A., & Bonilla-Sánchez, Y. M. (2013). Resource use in a landscape matrix by an arboreal primate: Evidence of supplementation in black howlers (Alouatta pigra). International Journal of Primatology, 34(4), 714–731. https://doi.org/10.1007/s10764-013-9691-y.
Priston, N. E. C., & McLennan, M. R. (2013). Managing humans, managing macaques: Human–macaque conflict in Asia and Africa. In S. Radhakrishna, M. A. Huffman, & A. Sinha (Eds.), The macaque connection: Cooperation and conflict between humans and macaques (pp. 225–250). Developments in Primatology: Progress and Prospects. Springer Science+Business Media.
Raboy, B. E., Christman, M. C., & Dietz, J. M. (2004). The use of degraded and shade cocoa forests by Endangered golden-headed lion tamarins Leontopithecuschrysomelas. Oryx, 38(1), 75–83. https://doi.org/10.1017/S0030605304000122.
Radhakrishna, S., Huffman, M. A., & Sinha, A., Eds. (2013). The macaque connection: Cooperation and conflict between humans and macaques. Developments in Primatology: Progress and Prospects. Springer Science+Business Media.
Regmi, G. R., Nekaris, K. A. I., Kandel, K., & Nijman, V. (2013). Crop-raiding macaques: Predictions, patterns and perceptions from Langtang National Park, Nepal. Endangered Species Research, 20(3), 217–226. https://doi.org/10.3354/esr00502.
Riley, E. P. (2007). The human–macaque interface: Conservation implications of current and future overlap and conflict in Lore Lindu National Park, Sulawesi, Indonesia. American Anthropologist, 109(3), 473–484. https://doi.org/10.1525/AA.2007.109.3.473.474.
Riley, E. P., & Wade, T. W. (2016). Adapting to Florida’s riverine woodlands: The population status and feeding ecology of the silver river rhesus macaques and their interface with humans. Primates, 57(2), 195–210. https://doi.org/10.1007/s10329-016-0517-3.
Ruppert, N., Holzner, A., See, K. W., Gisbrecht, A., & Beck, A. (2018). Activity budgets and habitat use of wild southern pig-tailed macaques (Macaca nemestrina) in oil palm plantation and forest. International Journal of Primatology, 39, 237–251. https://doi.org/10.1007/s10764-018-0032-z.
Sahoo, S. K., & Mohnot, S. M. (2004). A survey of crop damage by rhesus monkeys and Hanuman langur in Himachal Pradesh, India. Tiger Paper, 31, 1–5. https://doi.org/10.1017/CBO9781107415324.004.
Saj, T. L., Sicotte, P., & Paterson, J. D. (2001). The conflict between vervet monkeys and farmers at the forest edge in Entebbe, Uganda. African Journal of Ecology, 39(2), 195–199. https://doi.org/10.1046/j.0141-6707.2000.00299.x.
Sambyal, P., Kumar, S., & Sahi, D. N. (2009). Translocation of urban rhesus monkeys (Macaca mulatta) of Mubarak Mandi Jammu to forest areas of Nagrot. Tiger Paper, 36(2), 11–13.
Saraswat, R., Sinha, A., & Radhakrishna, S. (2015). A god becomes a pest? Human-rhesus macaque interactions in Himachal Pradesh, northern India. European Journal of Wildlife Research, 61, 435–443. https://doi.org/10.1007/s10344-015-0913-9.
Sengupta, A., McConkey, K. R., & Radhakrishna, S. (2015). Primates, provisioning and plants: Impacts of human cultural behaviours on primate ecological functions. PLoS ONE, 10(11). https://doi.org/10.1371/journal.pone.0140961.
Sengupta, A., & Radhakrishna, S. (2016). Influence of fruit availability on fruit consumption in a generalist primate, the rhesus macaque Macaca mulatta. International Journal of Primatology, 37(6), 703–717. https://doi.org/10.1007/s10764-016-9933-x.
Sillero-Zubiri, C., & Switzer, D. (2001). Crop raiding primates: searching for alternative, humane ways to resolve conflict with farmers in Africa. People and Wildlife Initiative. Wildlife Conservation Research Unit, Oxford University.www.peopleandwildlife.org.uk/crmanuals/CropRaidingPrimatesP&WManual
Singh, V., & Thakur, M. (2012). Rhesus macaque and associated problems in Himachal Pradesh-India. TAPROBANICA, 4(2), 112–116 http://www.sljol.info/index.php/TAPRO/article/viewFile/5066/4045.
Treves, A., Wallace, R. B., Naughton-Treves, L., & Morales, A. (2006). Co-managing human-wildlife conflicts: A review. Human Dimensions of Wildlife, 11, 383–396.
Turner, M.G. & Gardner, R. H. (2015). Landscape Ecology in Theory and Practice. Springer-Verlag.
Vandermeer, J. H., Ed. (2003). Tropical agroecosystems. CRC Press.
Warren, Y. (2008). Crop-raiding baboons (Papio anubis) and defensive farmers: A West African perspective. West African Journal of Applied Ecology, 14, 1–11. https://doi.org/10.4314/wajae.v14i1.44705.
Weibull, A. C., Östman, Ö., & Granqvist, Å. (2003). Species richness in agroecosystems: The effect of landscape, habitat and farm management. Biodiversity and Conservation, 12(7), 1335–1355. https://doi.org/10.1023/A:1023617117780.
Williams-Guillén, K., McCann, C., Martínez Sánchez, J. C., & Koontz, F. (2006). Resource availability and habitat use by mantled howling monkeys in a Nicaraguan coffee plantation: Can agroforests serve as core habitat for a forest mammal? Animal Conservation, 9(3), 331–338. https://doi.org/10.1111/j.1469-1795.2006.00042.x.
Yamada, A., & Muroyama, Y. (2010). Effects of vegetation type on habitat use by crop-raiding Japanese macaques during a food-scarce season. Primates, 51(2), 159–166. https://doi.org/10.1007/s10329-009-0183-9.
Acknowledgments
We would like to express our sincere thanks to Vijay Raput and Jagdish Chand for their assistance in fieldwork. We are also extremely thankful for the support provided by all the villagers in our study area during the period of field data collection. SA was supported by an institutional fellowship from National Institute of Advanced Studies during the period of this study.
Author information
Authors and Affiliations
Contributions
SA and SR conceived the study and developed methodology. SA conducted field work and performed statistical analyses. SV collaborated in data analysis. SA and SR wrote the manuscript.
Corresponding author
Additional information
Handling Editor: Joanna M. Setchell.
Supplementary Information
ESM 1
(DOCX 16 kb)
Rights and permissions
About this article
Cite this article
Anand, S., Vaidyanathan, S. & Radhakrishna, S. The Role of Landscape Structure in Primate Crop Feeding: Insights from Rhesus Macaques (Macaca mulatta) in Northern India. Int J Primatol 42, 764–780 (2021). https://doi.org/10.1007/s10764-021-00238-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10764-021-00238-y