Abstract
We aimed to investigate the preventive and therapeutic effect of apocynin (APO) on bleomycin (BLC)-induced lung injury in rats. Rats were assigned into groups as follows: control group; APO group, 20 mg/kg APO was given intraperitoneal for 29 days; BLC-1 and BLC-2 groups, a single intratracheal injection of BLC (2.5 mg/kg); APO+BLC-preventive group, 20 mg/kg APO was administered 12 h before the intratracheal BLC injection and continued for 14 days; BLC+APO-treatment group, 20 mg/kg APO was given on the 14th day after the intratracheal BLC injection and continued to sacrifice. The BLC-1 group was sacrificed on the 14th day of BLC administration to validate BLC-induced lung inflammation and fibrosis on the 14th of study initiation. All other groups were sacrificed on the 29th day after BLC administration. The semiquantitative histopathological assessment, tissue levels of malondialdehyde (MDA), superoxide dismutase, catalase (CAT), glutathione peroxidase (GPx), reduced glutathione (GSH), total antioxidant capacity, total oxidant status (TOS), and oxidative stress index (OSI) were measured. An addition to the serum myeloperoxidase (MPO), the cell count and cytokines (IL-1β, IL-6, and IL-8) of bronchoalveolar lavage (BAL) fluid were assayed. BLC-provoked histological changes were significantly detected compared to the control group. APO restored these histological damages in different quantity in the treatment and prevention groups. BLC caused a significant decrease in GSH, CAT, and GPX, which were accompanied with significantly the increased MDA, TOS levels, and OSI in the lung tissue concomitant with increased levels of the cellular account and proinflammatory cytokines in the BAL fluid. Otherwise, APO administration, both before and after BLC, reversed all biochemical markers and cytokine as well as histopathological changes induced by BLC. Interestingly, APO treatment reversed MPO activity in serum increased by BLC. In this study, both protective and therapeutic effects of APO against BLC-induced lung fibrosis were demonstrated for the first time.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Pulmonary fibrosis is a chronic and progressive lung disease. The disease can be idiopathic or developed as a complication of many pulmonary and systemic diseases. Up to one half of the patients with pulmonary fibrosis are idiopathic pulmonary fibrosis (IPF). It is characterized by fibroblast proliferation, excessive collagen accumulation and other deposition of extracellular matrix proteins within the pulmonary interstitial resulting in a loss of pulmonary function, and eventually, respiratory failure [1, 2]. Although the etiology of IPF has not been clearly explained yet, inflammation, oxidative stress, and damages from cytokines are involved in the pathogenesis machinery [2, 3]. It is well established that the first response following injury is inflammation. Activated alveolar macrophages and neutrophils accumulate in the lower respiratory tract, releasing harmful amount of reactive oxygen species (ROS) and diversity of harmful cytokines. Activated neutrophils can also release myeloperoxidase (MPO), an enzyme that interacts with hydrogen peroxide (H2O2), to form the highly toxic hydroxyl radicals [3]. In this respect, target inhibition of inflammation, oxidative stress, and cytokine release represent possible strategic points for therapeutic intervention.
Up to now, even though investigation, there has been no efficient treatment for IPF and other pulmonary fibrosis. In the absence of the lung transplantation, the mortality rates of the 3-and 5-year are 50 and 80 % (respectively) [1, 4]. Thence, treating patients with agents that have anti-inflammatory, antioxidants, and anti-fibrotic properties as an adjuvant or neoadjuvant therapy may have useful effects in prevention or treatment of IPF.
Bleomycin (BLC)-induced lung fibrosis is commonly used as an animal model of human IPF, as intratracheal BLC administration causes an inflammatory response alveolar cell damage, fibroblast proliferation, and collagen content deposition in rat [5–7]. Indeed, the role of ROS in the formation of lung injury in this model is known [3, 6]. There are also a lot of studies which tested diverse antioxidant agents such as N-acetylcysteine [5], erdosteine [6], aminoguanidine [7], molsidomine [8], melatonin [9], ginkgo biloba [10], resveratrol [11], dexpanthenol [12], and crysin [13] in BLC-induced lung fibrosis in rats as a specially prophylactic setting, and found these substances usually reduced or prevented lung fibrosis according to the semiquantitative histopathological assessment and lung hydroxyproline content.
However, in the clinical practice, the majority of patients with IPF are recognized during late stage of the disease. Therefore, it is clear that new studies are needed to investigate the effect of the drugs in BLC-induced lung fibrosis model simulating human IPF at the both early and late stages as a therapeutic model.
NADPH-oxidase (NOX) is a major enzyme that uses NADPH to generate superoxide, initial ROS molecule. Apocynin (APO) (4-hydroxy-3-methoxy-acetophenone), a natural occurring methoxy-substituted catechol, inhibits NOX in activated leukocytes, preventing production of ROS [14]. It was extracted from the roots of Apocynum cannabinum (Canadian hemp) and Picrorhiza kurroa (Scrophulariaceae) [15]. Several in vivo researches have demonstrated that APO can diminish neutrophil oxidative burst and neutrophil chemotaxis, and thus reduce neutrophil-mediated cell injury [15]. It has also been revealed to decline monocyte–endothelium interaction in vitro via stimulation with cytokines [16]. In addition, APO has been used to treat dropsy and heart troubles in India and currently seems to be an encouraging therapy for hypertension in the light of many experimental studies [17, 18].
Since BLC, one of the clinically significant causative agents in the pulmonary fibrosis is widely used in experimental models of human disease resembling pulmonary fibrosis [10]. The aim of this study was to investigate the protective and therapeutic effects of APO on lung fibrosis induced by the BLC administration by using histopathological semiquantitative evaluation, oxidative stress markers, and some cytokine in damaged lung tissue in rats.
MATERIAL AND METHODS
Study Design
This animal experimental study designed to ARRIVE guidelines [19]. This study was approved by Animal Ethics Committee (reference number 2012/A-60) and was conducted in accordance with the “Animal Welfare Act and the Guide for the Care and Use of Laboratory animals (NIH publication no. 5377–3, 1996), Animal Ethics Committee.”
Animals and Groups
For this study, a total of 48 female Wistar Albino rats of 10–12 weeks of age and weighing 250–300 g were obtained from Inonu University Laboratory Animals Research Center and placed in a temperature- (21 ± 2 °C) and humidity- (60 ± 5 %) controlled room in which a 12:12-h light/dark cycle was maintained. The animals were divided into six groups, each with eight rats, according to their experimental treatment as follows: (1) control group; (2) APO group, 20 mg/kg APO in 0.25 ml phosphate-buffered saline (Apocynin, Sigma-Aldrich) was given intraperitoneal (i.p.) for 29 days; (3) BLC-1 group, a single intratracheal injection of BLC (2.5 mg/kg body weight in 0.25 ml phosphate-buffered saline). This group was sacrificed on the 14th day of BLC administration to validate BLC-induced lung inflammation and fibrosis on the 14th of study initiation. (4) BLC-2 group, a single intratracheal injection of BLC (2.5 mg/kg body weight in 0.25 ml phosphate-buffered saline); (5) APO+BLC-preventive group, 20 mg/kg APO was administered 12 h before the intratracheal BLC injection and continued for 14 days i.p. (6) BLC+APO-treatment group, 20 mg/kg APO was administered on the 14th day after the intratracheal BLC injection and continued to sacrifice.
BLC-Induced Lung Fibrosis
The rats were weighted and then anesthetized with ketamine (75 mg/kg) and xylazine (5 mg/kg) i.p. followed by a single intratracheal injection of BLC hydrochloride (2.5 mg/kg body weight in 0.25 ml phosphate-buffered saline (PBS), Nippon Kayaku, Japan). The control group received the same amount of intratracheal saline by the same route. All rats (not BLC-1 group) were sacrificed after 29 days of the BLC injection. The BLC-1 group was sacrificed on the 14th of BLC administration to validate BLC-induced lung fibrosis on the 14th of the experiment. After scarification by an overdose of the anesthesia, the lung tissue specimens were quickly and meticulously harvested for biochemical and histopathological analysis. Right section of the lung was placed in liquid nitrogen and stored at −70 °C until assayed for thiobarbituric acid-reactive substances (TBARS), a lipid peroxidation product, superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), reduced glutathione (GSH), total antioxidant capacity (TAC), total oxidant status (TOS) contents, and oxidative stress index (OSI), and the left part of the lung was placed in formaldehyde solution for routine histopathological examination by light microscopy. Blood was drawn from the abdominal aorta for serum MPO. The dosage of BLC and APO was chosen depending on the previous dose–response studies that have been reported to cause lung fibrosis and marked antioxidative and anti-inflammatory effects in rats, respectively [8, 12, 17, 20].
Bronchoalveolar Lavage Fluid
After the anesthesia, the lungs were prepared for lavage by calculating the trachea with a blunt needle attached by a syringe. The lung lavage was obtained by washing the lung four times with 5 ml aliquots of saline through a tracheal cannula. Cell suspensions were concentrated by low speed centrifugation and the cell pellet resuspended. Also, alveolar macrophages were counted in stained preparations with May–Grünwald–Giemsa (MGG) and neutrophils were counted in stained preparations with Papanicolaou stain (PAP) from bronchoalveolar lavage (BAL) fluid and in the 10 microscopic under ×40 objective magnification using Leica Q Win Image Analysis System (Leica Micros Imaging Solution Ltd. Cambridge, UK).
Interleukin (IL)-1β, IL-6, and IL-8 were quantified using enzyme-linked immunosorbent assay (ELISA) kits specifically for the previously mentioned rat cytokines according to the manufacturer’s instruction and guideline (Biosource International, Nivelles, Belgium) in the BAL fluid. The particular assay kits were selected because of their high degree of sensitivity, specificity, inter- and intra-assay precision, and small amount of plasma sample required to conduct the assay.
Biochemical Analyses
Two hundred milligrams of frozen lung tissue specimens cut into pieces on dry ice, were homogenized in 1.15 % KCl buffer (1:9, w/v) using a manual glass homogenizer for approximately 5 min, and flushed with centrifugation for approximately 10 s to remove large debris. The supernatant was used for analysis.
Determination of Serum TBARS Representing MDA Content
The TBARS contents of the homogenates were determined by TBARS [21]. Three milliliters of 1 % phosphoric acid and 1 ml 0.6 % thiobarbituric acid solution were added to 0.5 ml of plasma pipetted into a tube. The mixture was heated in boiling water for 45 min. After the mixture had cooled, the color was extracted into 4 ml of n-butanol. The absorbance was measured by a spectrophotometer (UV-1601; Shimadzu, Kyoto, Japan) at 532 nm. The amount of lipid peroxides was calculated as TBARS of lipid peroxidation. The results were expressed in nanomoles per gram (nmol/g tissue) according to a standard graph, which was prepared from the measurements of standard solutions (1,1,3,3-tetramethoxypropane).
Determination of SOD
Total (Cu-Zn and Mn) SOD (EC 1.15.1.1) activity was determined based on the method of Sun et al. [22]. The principle of the method is the inhibition of nitroblue tetrazolium (NBT) reduction of the xanthine–xanthine oxidase system as a superoxide (O2·–) generator. One unit of SOD was defined as the enzyme amount causing 50 % inhibition in the NBT reduction rate. SOD activity was expressed as units per milligram protein (U/g protein).
Determination of CAT
CAT (EC 1.11.1.6) activity was determined with respect to Aebi’s method [23]. The principle of the assay is based on the determination of the rate constant (k, s − 1) or the H2O2 decomposition rate at 240 nm. Results were expressed as k per gram protein (K/g protein).
Determination of GPx
Determination of GPx activity (EC 1.6.4.2) was measured by the method of Paglia and Valentine [24]. An enzymatic reaction in a tube containing NADPH, GSH, sodium azide, and glutathione reductase was initiated with the addition of H2O2, and the change in absorbance at 340 nm was monitored by a spectrophotometer. Activity was given in units per gram protein (U/mg protein).
Determination of GSH Content
The GSH content in the lung tissue as non-protein sulfhydryls was analyzed following a previously described method [25]. Aliquots of tissue homogenate were mixed with distilled water and 50 % trichloroacetic acid in glass tubes and centrifuged at 3,000 rpm for 15 min. The supernatants were mixed with Tris buffer (0.4 M, pH 8.9) and 5,5′-dithiobis(2-nitrobenzoic acid) (DTNB, 0.01 M) was added. After shaking the reaction mixture, its absorbance was measured at 412 nm within 5 min by the addition of DTNB against blank with no homogenate. The absorbance values were extrapolated from a glutathione standard curve and expressed as GSH (μmol/g tissue).
Determination of MPO Activity
All experiments were performed within 1 week of a collected serum. MPO activity, the index of polymorphonuclear infiltration, was measured according to the method described by Bai et al. [26]. Absorbance was read at 460 nm using a microplate reader. MPO was expressed in milliunits per gram protein of serum, where one unit corresponds to the activity required to degrade 1 mmol of hydrogen peroxide in 1 min at 24 °C.
Measurement of TAC
TAC levels were determined using a novel automated colorimetric measurement method developed by Erel [27]. In this method, the hydroxyl radical, the most potent biological radical, is produced by the Fenton reaction and reacts with the colourless substrate O-dianisidine to produce the dianisyl radical, which is bright yellowish-brown in color. Upon the addition of sample, the oxidative reactions initiated by the hydroxyl radicals present in the reaction mix are suppressed by the antioxidant components of the sample, preventing the color change and thereby providing an effective measure of the total antioxidant capacity of the sample. The assay has excellent precision values, which are lower than 3 %. The results were expressed as millimole Trolox equivalent/liter.
Measurement of TOS
TOS was determined using a novel automated measurement method, developed by Erel [28]. Oxidants present in the sample oxidize the ferrous ion-O-dianisidine complex to ferric ion. The oxidation reaction is enhanced by glycerol molecules, which are abundantly present in the reaction medium. The ferric iron makes a colored complex with xylenol orange in an acidic medium. The color intensity, which can be measured spectrophotometrically, is related to the total amount of oxidant molecules present in the sample. The assay was calibrated with hydrogen peroxide and the results were expressed in terms of micromole H2O2 equivalent/liter.
Measurement of OSI
The percentage ratio of the TOS to TAC yields the OSI, an indicator of the degree of oxidative stress [28]. OSI (arbitrary unit) = TOS/TAC. The OSI value of the lung samples was also calculated as an OSI (arbitrary unit).
Histological Evaluations
The lung tissues were fixed in 10 % formalin and were embedded in paraffin. Tissue sections were cut at 5 μm, mounted on slides, and stained with hematoxylin-eosin (H-E) for general lung structure, periodic acid Schiff (PAS) to identify alveolar macrophage, and Masson’trichrome for connective tissue. Lung injury was scored for each of the following tissue injury criteria: lymphocytes, eosinophils, and macrophages accumulation; alveolar hemorrhage; increase in connective tissue in the parenchyma were scored on a scale of 0–3: 0 for normal tissue, 1 for, 25 % injury involvement, 2 for 26–75 % injury involvement, and 3 for 75 % injury involvement. The total histology score is the sum score of all parameters. Also, alveolar macrophages were counted in stained preparations with MGG and neutrophils were counted in stained preparations with PAP from BAL fluid and in the 10 microscopic under ×40 objective magnification using Leica Q Win Image Analysis System (Leica Micros Imaging Solution Ltd. Cambridge, UK).
Statistics
For detecting even minor effects, the required sample sizes used in this experiment were identified using statistical power analysis. The sample sizes necessary for a power of 0.80 were estimated using NCSS software. Data were analyzed using the IBM SPSS Statistics software program for Windows, version 22.0 (SPSS Inc., Chicago, IL). The normality of the distribution was confirmed using the Kolmogorov–Smirnov test. According to the results obtained from the normality test, one-way analysis of variance (ANOVA) and Kruskal–Wallis H test were used for the statistical analysis, as appropriate. Multiple comparisons were carried out by Tukey’s test after the ANOVA test and Mann–Whitney U test with Bonferroni correction after the Kruskal–Wallis H test. The results are expressed as mean ± standard deviation (SD) and median with min and max values where appropriate. P values less than 0.05 were regarded as statistically significant.
RESULTS
Histological Findings
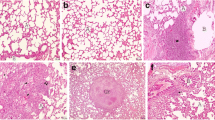
The lungs of rats in the control group showed normal lung structure and there were no lesions (Fig. 1a). APO alone-treated group was similar to that of the control group (Fig. 1b). No sign of connective tissue deposition was observed with Masson’s trichrome staining methods (Fig. 1c, d) and the appearance of macrophages was normal with PAS-stained methods in the lung of control and APO groups (Fig. 1e, f). However, there were considerable histological changes in the lung tissues in the BLC groups. Alveolar hemorrhage was seen in these groups (Fig. 2a, b). Moreover, lymphocytes (Fig. 2c, d), eosinophils (Fig. 2e, f), and macrophages accumulations (Fig. 2g, h) were observed in BLC groups. The other most remarkable finding was an increase in the connective tissue in the BLC groups compared to control and APO groups. There were statistically significant changes in the connective tissue deposition in the BLC-2 group when compared to the BLC-1 group (P = 0.01) (Fig. 3a, b). On the other hand, growth of connective tissue was found to be significantly decreased in the treatment groups when compared to the BLC-2 group (P < 0.001) (Fig. 3c, d). While alveolar hemorrhage in APO+BLC group was significantly lower than BLC groups, there was no statistically significant change in the BLC+APO group when compared to the BLC groups (P > 0.05). Although lung damage was alleviated in the BLC+APO and APO+BLC groups, inflammatory alterations such as moderate accumulation of lymphocytes (Fig. 4a, b), eosinophils (Fig. 4c, d), and macrophages were still present (Fig. 4e, f). However, there was no statistically significant changes between APO+BLC group and BLC+APO group in terms of the histological damage score (P > 0.05). The results of semiquantitative histological scores in all groups were shown in the Table 1.
BLC-1 (a) and BLC-2 (b) groups: congestion of the parenchyma (star). BLC-1 (c) and BLC-2 (d): visible lymphocytes accumulation (arrows); H-E, ×20. BLC-1 (e) and BLC-2 (f) groups notice an eosinophils accumulation (arrows); H-E, ×40. BLC-1 (g) and BLC-2 (h) groups: the appearance of alveolar macrophage accumulation (arrows); PAS, ×40.
Inflammatory Cell Count and Cytokines (IL-1β, IL-6, and IL-8) in BAL Fluid
The cells in BAL fluid were analyzed to evaluate the effect of APO on the accumulation of inflammatory cells to the lung induced by BLC. BLC treatment caused a significant increase in the number of cells in the BAL fluid as compared to control rats and this enhancement was significantly reduced by the treatment of APO (P < 0.05). The differential cell count showed that neutrophils (Fig. 5a–d) and macrophage (Fig. 6a–d) were markedly augmented in the lungs of rats which were exposed to BLC and this augmentation was significantly reduced by the treatment of APO (P < 0.05). The effect of APO on BAL differential cell count was shown in Table 2.
In order to assess the pulmonary inflammation, proinflammatory cytokines such as IL-1β, IL-6, and IL-8 levels were detected in the BAL fluid. Compared to the control and APO groups, levels of IL-6 and IL-8 were significantly increased in the BLC groups (P < 0.05). As shown in Table 3, increased in these two cytokines were significantly reduced by administration of APO both before and after BLC treatment. However, although the level of IL-1β in the BLC groups did not reach statistical significance when compared to the control group, it was found to have a tendency to increase (Table 3). The APO combined with BLC treatment improved the level of IL-1β; however, this improvement was not statistically significant (Table 3).
Changes in Oxidative Stress Parameters
The levels of SOD, GPx, CAT, GSH, malondialdehyde (MDA), TAC, TOS, OSI, and MPO are given in Table 4. There was no statistically differentiation between BLC-1 and BLC-2 groups in terms of oxidative stress in all parameters. Likewise, APO alone treatment did not cause a change in these parameters when compared to the control group (P > 0.05).
The BLC treatment caused a significant increase in MDA and TOS levels, and a significant decrease in GPx and CAT activities in the rat lung tissues compared to control and APO groups. Although the level of SOD and TAC in the BLC groups did not reach statistical significance when compared to the control group, it was found to have a tendency to decrease (Table 4). When compared to the control group, there was a greater decrease in the GSH levels of the BLC-2 group (P = 0.016).
Additionally, it was determined that APO combined (both protection and therapeutic purpose) with BLC generally lead to a significant decrease in lipid peroxidations, caused by BLC, via decline of MDA and TOS levels and a significant increase of antioxidant status via improved in SOD, CAT, and GPx levels. Although the APO combined with BLC improved the level of TAC and GSH; however, this improvement was not statistically significant (Table 4).
In addition, OSI was increased by BLC instillation compared to the control groups (P < 0.0001). Increased OSI were significantly reduced in the APO+BLC and BLC+APO groups compared to the BLC administration groups (Table 4).
As shown in Table 4, MPO, a marker of neutrophil accumulation, content was significantly increased in rats administrated with BLC alone as compared to the controls. The rise in the serum MPO activity was significantly prevented by the APO administration of both before and after BLC treatment.
DISCUSSION
In the current study, we evaluated the effect of intraperitoneal administration of APO, an inhibitor of NOX inhibitory, in the protection and treatment of BLC-induced lung fibrosis in rats. Lung injury was quantitatively assessed biochemically (oxidant and anti-oxidant of lung tissue), histopathologically (semi-quantitative assessment of lung inflammation and fibrosis), different cell account and some cytokine in BAL fluid. In addition to administration of APO prevented BLC-induced early lung injury and fibrosis as assessed through histopathological findings, it successfully treated BLC-induced settled fibrosis. Furthermore, we found that administration of APO after and before BLC had reversed the BLC-induced increases in the some cytokines (IL-1β, IL-6, and IL-8) and account of different inflammatory cells (neutrophils and macrophage) in the BAL fluid, MDA levels, and the BLC-induced reduction in the antioxidant enzyme activities in the damaged lung tissue of the rats. Interestingly, APO prevented the increased serum MPO levels in the BLC+APO and APO+BLC groups (Table 4).
Currently, pulmonary fibrosis is considered to result from the recurrent epithelial damage and apoptosis followed by insufficient re-epithelialization and aberrant wound healing [29, 30]. Essential to the abnormal fibrotic reaction are transforming growth factor (TGF)-β, a major profibrotic mediator, and myofibroblasts, the key cell type of overdone extracellular matrix deposition and distorted lung architecture, which constitute the typical features of IPF [29]. There are certain difficulties in disease modeling of IPF, including the difficulty to reproduce the chronic nature of the disease in animal models and the lack of triggering fibrosis to cause appropriate lung injury as the underlying cause of IPF remains unknown. However, BLC-induced lung fibrosis is widely used as an experimental model for researches of new treatments and pathogenesis of IPF. In this model, it is used as a single intratracheal dose of the drug during the first 7 days to induce an inflammatory response [31] and increase the epithelial apoptosis [32], closely mimicking acute lung injury (ALI). The intratracheal BLC application immediately promotes the release of proinflammatory cytokines such as tumor necrosis factor-α (TNF-α), IL-1β, IL-6 (also, a profibrotic), and IL-8. Subsequently, the inflammation resolves and fibrosis is detected during the following transitional period of 3 days. In the previous studies, it was shown that fibrotic changes occur in approximately 9 days after application of BLC [31, 32]. The late (fibrotic) stage persists for 3–4 weeks following BLC administration and is characterized by intense deposition of extracellular matrix, resulting in the fibrotic tissue [33]. Therefore, studies related to the BLC-induced pulmonary fibrosis are classified as preventive when the tested agent was given before BLC and as therapeutic when they are given 7 days after BLC [31].
The development of lung fibrosis is related to and likely preceded by an increase accumulation of inflammatory cells within the lung parenchyma. Carre et al. [34] shown that the neutrophil recruitment and the content of cytokines, such as IL and interferons, were augmented in the lung tissue. Similarly, in one study which evaluated the role of cytokines in BLC-induced lung fibrosis, it was suggested that BLC caused the expression of IL-6, and these cytokines could have aggravated BLC injury [35]. Recently, studies have shown that expression of TNF-α, IL-1β, IL-6, and IL-8 was increased by BLC administration while antioxidants such as N-acetylcysteine and resveratrol attenuate the inflammatory reactions, therefore preventing of cellular injury and the subsequent development of BLC-induced lung fibrosis in animal models [36, 37]. In accordance with these findings, in the current study, it was demonstrated that the proinflammatory cytokines, IL-1β, IL-6, and IL-8, in the BAL fluid were increased in the BLC groups. The increases of IL-6 and IL-8 levels were statistically significant. Additionally, these results were verified using both biochemical markers and semiquantitative histological assessment. Furthermore, application of the APO for both preventative and therapeutic effects reversed all these injury parameters and the levels of inflammatory markers. The inhibitory effect of APO on the production of proinflammatory cytokines was previously demonstrated in the ventilator-induced lung injury models, where the treatment with APO repaired the structural lung injury [38]. Similarly, in an another experimental study, APO treatment reversed the increased levels of IL-8, a potent neutrophil attractant and activator, due to toxic smoke inhalation injury where a semiquantitative histological score of lung inflammation was alleviated [39].
Apocynin is a strong inhibitor of NOX activity and concomitant ROS production in activated neutrophils [14, 40]. Interestingly, it does not appear to interfere with the neutrophils’ other defense mechanism, as it does not influence phagocytosis or intracellular killing [14]. The superoxide anion (O2 −) is an essential and a preliminary ROS molecule and transform to more reactive and toxic-free radicals, such as peroxynitrite, H2O2, or hydroxyl radical, under situation of oxidative stress [14]. Although the mechanism that APO inhibits the NOX activity is not clearly understood, it is suggested to act by preventing the translocation of its cytosolic components, p47phox, to gp91phox [15, 41, 42]. In addition to NOX inhibition, in the previous studies, it has been demonstrated that APO may inhibit thromboxane synthase, cytochrome P450, and COX-2 expression [43, 44]. APO that has low toxicity and specificity may be capable as potential therapy for asthma, arthritis, neurological, and cardiovascular diseases via anti-inflammatory and antioxidant effects [17, 45–47]. In addition, it has been used in many experimental studies related to ALI. In these studies, APO was found to be effective in protection of the tissue against the lipopolysaccharide- or ventilator-induced lung injury [15, 38, 48]. In another study, Altintas et al. [40] evaluated APO on ischemia–reperfusion-induced renal injury in an experimental model and reported helpful effects related to the reduction in oxidative stress.
It has been well known that ROS can injure lipids, protein, and DNA and therefore may cause the decreased of enzyme activity or structural damage in the BLC-induced lung fibrosis. Lipid peroxidation induced by oxidant causes a loss of membrane constancy and integrity, leading to augmented transepithelial permeability [33, 37]. We have observed that elevated inflammatory cell count in the BAL fluid support this hypothesis based on significant increase in the MDA levels in the lung tissue. Many studies have also demonstrated the increased of lipid peroxidation products, such as MDA, in the lung tissue of BLC-administered rats [8, 9, 12, 37]. Additionally, in the current study, BLC administration caused the prominent TOS and OSI, which are novel very important indexes of oxidative stress, in the lung tissues. Likewise, MPO activity, indicating the increased neutrophils activity, in the serum was significantly increased by BLC treatment. These findings seem to be related to the induction of lung injury, consistent with other studies, which used to BLC-induced lung fibrosis models [7, 29, 30, 33]. The BLC-induced lung damage comprises, as a primary event, the generation of oxidant species of iron-dependent mechanism [33]. Further damage is probably elicited by augmented amounts of ROS and reactive nitrogen space produced by activating inflammatory cells recruited into the damaged lung induced by BLC administration [7]. On the other hand, in the current study, APO treatment reversed the increased MDA, TOS, OSI, and MPO by inducing BLC. Likewise, implicating membrane protective effect of this antioxidant agent causes dramatic decreases in various inflammatory cell numbers in the BAL fluid.
A balance between intracellular and extracellular oxidants and antioxidants is a precondition for normal pulmonary homeostasis. The defense system of enzymatic antioxidant is the native protection against lipid peroxidation that contains SOD, CAT, and GPx. Some native or synthetic compound with antioxidant features may reduce lung injury partially or totally. Thus, there is a significant protective effect of agents which scavenging O2 − and OH· against a variety of diseases [5, 7, 13]. In the current study, a significant decrease activity of CAT and GPx was observed in the lung tissue of BLC-treated rats when compared with the control group. However, the decreased level of SOD in the BLC groups did not reach a statistically significant when compared to the control group. In addition, APO supplementation for the BLC-treated groups significantly increased the levels of all antioxidant enzymes reduced by BLC administration. In many previous studies, it was reported that APO has protective effects in different organ or tissue via increasing the activity of those antioxidant enzymes, which decreased due to various toxic agents [40, 48]. Our results are in accordance with those observations.
However, limited studies assessed the role of testing agents in the late phase of lung fibrosis in rats that are established as an animal model of human IPF. In one of these studies [46], the effect of follistatin was examined which is an inhibitor of TGF-β, given during early and late phases on histological changes, and hydroxyproline content of the tissue to estimate collagen deposit in the lung. This study demonstrated that follistatin significantly decreased the hydroxyproline content and there is no significant difference in the reduction of hydroxyproline content between the early-phase and late-phase treatment groups. In addition, BLC-induced lung fibrosis was noticeably recovered in follistatin-treated groups. In another experimental study, Kakugawa et al. [47] showed a considerable improvement in lung fibrosis score and hydroxyproline substance induced by intratracheal BLC instillation into the lung of mice by the treatment pirfenidone, an antifibrotic drug, taken 14 days after BLC instillation. Also, more recently, our study group found that molsidomine treatment markedly improved the histopathological changes of early and late phase of BLC-induced lung fibrosis [8].
Although the anti-inflammatory and antioxidant properties of APO are well recognized, the ability of anti-fibrosis is not clear. To the best of our knowledge, only in one study that it was shown that APO inhibited the reproduction of human fibroblast cells in vitro conditions [49]. In the present study, for the first time, in addition to its protective effect against the lung fibrosis, it was shown that APO downgraded lung fibrosis that has already established in the experimental model. Thus, for the first time, the antifibrotic property of APO was demonstrated through this study’s in vivo condition. We think that APO could treat BLC-induced lung fibrosis via inhibition of TGF-β and IL-6. In the present study, APO could decrease the increase of IL-6, a profibrotic cytokine, induced by BLC. On the other hand, it could not be analyzed because of difficulties in the supply of ELISA kit for TGF-β.
Although our research is well designed, there are some limitations. The BLC-induced lung fibrosis model fails to summarize several important characteristics of usual interstitial pneumonia, namely the lack of fibroblastic foci and hyperplastic epithelium. In addition, an inflammatory, neutrophil-rich process is observed following the injury caused by a single dose of BLC, and this result is more indicative of an ALI model than a fibrosis model. Another limitation is that TGF-β and TNF-α, which are an important profibrotic and proinflammatory cytokines, in BAL fluid could have not been studied because of difficulties in the supply of ELISA kit for these cytokines.
In conclusion, the findings of the current study showed for the first time that APO, with its potent free radical scavenging, antioxidant, and anti-inflammatory properties, seems to be a highly promising agent not only in preventing but also in treatment BLC-induced lung inflammation and fibrosis. Furthermore, for the first time, anti-fibrotic property of APO in in vivo condition was demonstrated. However, further studies are required to evaluate the role of different doses of APO in the treatment of lung fibrosis and the effect of systemic administration. If our data is supported by further studies, APO may offer a new and promising therapy for IPF and other fibrotic disease, which has no curative treatment, in the future.
References
Raghu, G., H.R. Collard, J.J. Egan, et al. 2011. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. American Journal of Respiratory and Critical Care Medicine 183: 788–824.
Gross, T.J., and G.W. Hunninghake. 2001. Idiopathic pulmonary fibrosis. The New England Journal of Medicine 345: 517–525.
Dedon, P.C., and I.H. Goldberg. 1992. Free-radical mechanisms involved in the formation of sequence-dependent bistranded DNA lesions by the antitumor antibiotics bleomycin, neocarzinostatin, and calicheamicin. Chemical Research in Toxicology 5: 311–332.
Cottin, V. 2013. The role of pirfenidone in the treatment of idiopathic pulmonary fibrosis. Respiratory Research 14: 5–9. doi:10.1186/1465-9921-14- S1-S5.
Mata, M., A. Ruíz, M. Cerdá, M. Martinez-Losa, et al. 2003. Oral N-acetylcysteine reduces bleomycin-induced lung damage and mucin Muc5ac expression in rats. The European Respiratory Journal 22: 900–905.
Sogut, S., H. Ozyurt, F. Armutcu, et al. 2004. Erdosteine prevents bleomycin-induced pulmonary fibrosis in rats. European Journal of Pharmacology 494: 213–220.
Yildirim, Z., Y. Turkoz, M. Kotuk, et al. 2004. Effects of aminoguanidine and antioxidant erdosteine on bleomycin-induced lung fibrosis in rats. Nitric Oxide 11: 156–165.
Kilic, T., H. Parlakpinar, A. Polat, et al. 2014. Protective and therapeutic effect of molsidomine on bleomycin-induced lung fibrosis in rats. Inflammation 37: 1167–1178. doi:10.1007/s10753-014-9841-1.
Yildirim, Z., M. Kotuk, H. Erdogan, et al. 2006. Preventive effect of melatonin on bleomycin-induced lung fibrosis in rats. Journal of Pineal Research 40: 27–33.
Iraz, M., H. Erdogan, M. Kotuk, et al. 2006. Ginkgo biloba inhibits bleomycin-induced lung fibrosis in rats. Pharmacological Research 53: 310–316.
Akgedik, R., S. Akgedik, H. Karamanli, et al. 2012. Effect of resveratrol on treatment of bleomycin-induced pulmonary fibrosis in rats. Inflammation 35: 1732–1741.
Ermis, H., H. Parlakpinar, G. Gulbas, et al. 2013. Protective effect of dexpanthenol on bleomycin-induced pulmonary fibrosis in rats. Naunyn-Schmiedeberg's Archives of Pharmacology 386: 1103–1110. doi:10.1007/s 00210-013-0908.
Kilic, T., O. Osman Ciftc, A. Cetin, et al. 2014. Protective effect of chrysin on bleomycin-induced lung fibrosis in rats. Inflammation. doi:10.1007/s10753-014-9946-6.
Stefanska, J, and R. Pawliczak. 2008. Apocynin: molecular aptitudes. Mediators of Inflammation 106507
Impellizzeri, D., E. Esposito, E. Mazzon, et al. 2011. Effect of apocynin, a NADPH oxidase inhibitor, on acute lung inflammation. Biochemical Pharmacology 81: 636–648.
De Caterina, R., and M. Massaro. 2005. Omega-3 fatty acids and the regulation of expression of endothelial proatherogenic and pro-inflammatory genes. The Journal of Membrane Biology 206: 103–116.
Taylor, N.E., P. Glocka, M. Liang, et al. 2006. NADPH oxidase in the renal medulla causes oxidative stress and contributes to salt-sensitive hypertension in Dahl S rats. Hypertension 47: 692–698.
Jin, L., R.A. Beswick, T. Yamamoto, et al. 2006. Increased reactive oxygen species contributes to kidney injury in mineralocorticoid hypertensive rats. Journal of Physiology and Pharmacology 57: 343–357.
Colak, C., and H. Parlakpınar. 2012. Hayvan Deneyleri: in vivo Denemelerin Bildirimi: ARRIVE Kılavuzu-Derleme. Journal Turgut Ozal Medical Center 19(2): 128–131.
Connell, B.J., M.C. Saleh, B.V. Khan, et al. 2011. Apocynin may limit total cell death following cerebral ischemia and reperfusion by enhancing apoptosis. Food and Chemical Toxicology 49: 3063–3069.
Uchiyama, M., and M. Mihara. 1978. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Analytical Biochemistry 34: 271–278.
Sun, Y., L. Oberley, and Y. Li. 1988. A simple method for clinical assay of superoxide dismutase. Clinical Chemistry 34: 497–500.
Aebi, H. 1974. Catalase. In Methods of enzymatic analysis, ed. H.U. Bergmeyer, 673–677. New York: Academic.
Paglia, D.E., and W.N. Valentine. 1967. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. Journal of Laboratory and Clinical Medicine 70: 158–170.
Ellman, G.L. 1959. Tissue sulfhydryl groups. Archives of Biochemistry and Biophysics 82: 70–77.
Bai, A., P. Hu, J. Chen, et al. 2007. Blockade of STAT3 by antisense oligonucleotide in TNBS-induced murine colitis. International Journal of Colorectal Disease 22: 625–635.
Erel, O. 2004. A novel automated direct measurement method for total antioxidant capacity using a new generation, more stable ABTS radical cation. Clinical Biochemistry 37: 277–285.
Erel, O. 2005. A new automated colorimetric method for measuring total oxidant status. Clinical Biochemistry 38: 1103–1111.
Coward, W.R., G. Saini, and G. Jenkins. 2010. The pathogenesis of idiopathic pulmonary fibrosis. Therapeutic Advances in Respiratory Disease 4: 367–388.
Harari, S., and A. Caminati. 2010. IPF: new insight on pathogenesis and treatment. Allergy 65: 537–553.
Chaudhary, N.I., A. Schnapp, and J.E. Park. 2006. Pharmacologic differentiation of inflammation and fibrosis in the rat bleomycin model. American Journal of Respiratory and Critical Care Medicine 173: 769–776.
Mungunsukh, O., A.J. Griffin, Y.H. Lee, et al. 2010. Bleomycin induces the extrinsic apoptotic pathway in pulmonary endothelial cells. American Journal of Physiology - Lung Cellular and Molecular Physiology 298: 696–703.
Mouratis, M.A., and V. Aidinis. 2011. Modeling pulmonary fibrosis with bleomycin. Current Opinion in Pulmonary Medicine 17: 355–361.
Carre, P., and P. Leophonte. 1993. Cytokines and pulmonary fibroses. Revue des Maladies Respiratoires 10: 193–207.
Underwood, D.C., R.R. Osborn, S. Bochnowicz, et al. 2000. SB 239063, a p38 MAPK inhibitor, reduces neutrophilia, inflammatory cytokines, MMP-9, and fibrosis in lung. American Journal of Physiology - Lung Cellular and Molecular Physiology 279: L895–L902.
Gon, Y., S. Hashimoto, T. Nakayama, et al. 2000. N-Acetyl-l-cysteine inhibits bleomycin-induced interleukin-8 secretion by bronchial epithelial cells. Respirology 5: 309–313.
Sener, G., N. Topaloglu, A.O. Sehirli, et al. 2007. Resveratrol alleviates bleomycin-induced lung injury in rats. Pulmonary Pharmacology & Therapeutics 20: 642–649.
Chiang, C.H., C.H. Chuang, S.L. Liu, et al. 2011. Apocynin attenuates ventilator-induced lung injury in an isolated and perfused rat lung model. Intensive Care Medicine 37: 1360–1367.
Perg, D.W., T.M. Chang, J.Y. Wang, et al. 2013. Inflammatory role of AMP-activated protein kinase signaling in an experimental model of toxic smoke inhalation injury. Critical Care Medicine 41: 120–132.
Altintas, R., A. Polat, N. Nigar Vardi, et al. 2013. The protective effects of apocynin on kidney damage caused by renal ischemia/reperfusion. Journal of Endourology 27: 617–624.
Peters, E.A., J.T.N. Hiltermann, and J. Stolk. 2001. Effect of apocynin on ozone-induced airway hyperresponsiveness to methacholine in asthmatics. Free Radical Biology and Medicine 31: 1442–1447.
Ximenes, V.F., M.P. Kanegae, S.R. Rissato, et al. 2007. The oxidation of apocynin catalyzed by myeloperoxidase: proposal for NADPH oxidase inhibition. Archives of Biochemistry and Biophysics 457: 134–141.
Smit, H.F., B.H. Kroes, A.J.J. van den Berg, et al. 2000. Immunomodulatory and anti-inflammatory activity of Picrorhiza scrophulariiflora. Journal of Ethnopharmacology 73: 101–109.
Barbieri, S.S., V. Cavalca, S. Eligini, et al. 2004. Apocynin prevents cyclooxygenase 2 expression in human monocytes through NADPH oxidase and glutathione redox dependent mechanisms. Free Radical Biology and Medicine 37: 156–165.
Stefanska, J., A. Sarniak, A. Wlodarczyk, et al. 2012. Apocynin reduces reactive oxygen species concentrations in exhaled breath condensate in asthmatics. Experimental Lung Research 38: 90–99.
Hougee, S., A. Hartog, A. Sanders, et al. 2006. Oral administration of the NADPH-oxidase inhibitor apocynin partially restores diminished cartilage proteoglycan synthesis and reduces inflammation in mice. European Journal of Pharmacology 531: 264–269.
Tang, L.L., K. Ye, X.F. Yang, et al. 2007. Apocynin attenuates cerebral infarction after transient focal ischaemia in rats. Journal of International Medical Research 35: 517–522.
Xu, L., Y. Li, S. Wan, Y. Wang, and P. Yu. 2014. Protective effects of apocynin nitrone on acute lung injury induced by lipopolysaccharide in rats. International Immunopharmacology 20: 377–382.
Antao-Menes, A., E.A. Turpin, P.C. Post, et al. 2008. STAT-1 signaling in human lung fibroblast in induced vanadium pentoxide through an IFN-beta autocrine loop. Journal of Immunology 180: 4200–4207.
Conflict of Interest
The authors have no conflict of interest to declare. This work has been supported by the scientific research unit of Inonu University (Project number:2013/112 ).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kilic, T., Parlakpinar, H., Taslidere, E. et al. Protective and Therapeutic Effect of Apocynin on Bleomycin-Induced Lung Fibrosis in Rats. Inflammation 38, 1166–1180 (2015). https://doi.org/10.1007/s10753-014-0081-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-014-0081-1