Abstract
Some species of fish are more tolerant of rotenone, a commonly used non-specific piscicide, than others. This species-specific tolerance to rotenone has been thought to be associated with the uptake and the efficiency at which the chemical is detoxified. However, rotenone stimulates oxidative stress and superoxides, which are also toxic. Understanding the modes in which fish physiologically respond to rotenone is important in developing improved protocols for its application in controlling aquatic nuisance species. Using a molecular approach, we investigated the physiological and molecular mechanisms of rotenone resistance. Species-specific responses were observed when rotenone-sensitive silver, Hypophthalmichthys molitrix, and both rotenone-resistant bighead carp, Hypophthalmichthys nobilis, and bigmouth buffalo, Ictiobus cyprinellus, were exposed to rotenone. Rotenone levels in plasma were highest 90 min after exposure in both silver carp and bigmouth buffalo, but bigmouth buffalo tolerated over twice the burden (ng mL−1 g−1) than silver carp. Expression of genes related with detoxification (cyp1a and gst) increased in silver carp, but either decreased or remained the same in bighead carp. Genes linked with oxidative stress in the cytosol (gpx, cat and sod1) and hsp70 increased only in silver carp after a 6-h exposure. Expression of genes associated with oxidative stress in the mitochondria (sod2 and ucp2) differed between silver carp and bighead carp. Expression of sod2 changed minimally in bighead carp, but expression of ucp2 linearly increased to nearly 85-fold of the level prior to exposure. Expression of sod2 and ucp2 did not change until 6 h in silver carp. Use of sod1 and sod2 to combat oxidative stress results in hydrogen peroxide production, while use of ucp2 produces nitric oxide, a chemical known to inhibit apoptosis. We conclude that the mechanism at which a fish handles oxidative stress plays an important role in the tolerance to rotenone.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Rotenone is commonly used as a piscicide to control undesirable fishes by resource management agencies because of its high toxicity to fishes and low toxicity to mammals (Lennon 1970; Rach and Gingerich 1986). Unfortunately, variability exists in rotenone’s toxicity among species of fishes. According to one manufacturer’s general guide to application (Prentiss, Inc., Floral Park, NY USA; 5% rotenone as active ingredient), twice the concentration of rotenone is required to effectively kill bullheads (Ictalurus spp.) and common carp, Cyprinus carpio, than for other species. Our laboratory has previously shown that under similar concentrations of rotenone, all yellow perch, Perca flavescens, and bluegill, Lepomis macrochirus, died while only half of the channel catfish, Ictalurus punctatus, and bighead carp, Hypophthalmichthys nobilis, died (Rach et al. 2009). Differences in rotenone resistance may be explained by differences in absorption, distribution, metabolism or excretion (Gingerich and Rach 1985) or possibly the ability to combat reactive oxygen species (ROS) caused by exposure to rotenone.
Xenobiotic metabolism is generally considered to include modification and conjugation of the xenobiotic to facilitate elimination. One of the most common modifications is hydroxylation catalyzed by the cytochrome P450 enzymes. Cytochrome P450s, particularly cytochrome P4501A (cyp1a), are commonly used biomarkers of fish exposed to various toxicants (Curtis et al. 1993; Bucheli and Fent 1995; Collier et al. 1995; Eggens et al. 1995; Egaas et al. 1999). Conjugation of the xenobiotics with charged species like glutathione by glutathione S-transferase (gst) provides another commonly used biomarker for xenobiotic exposure in aquatic organisms (Fitzpatrick et al. 1997; Al-Ghais and Ali 1999; Egaas et al. 1999; Sayeed et al. 2003).
Besides metabolizing rotenone, fish must overcome the cellular affect of rotenone. Rotenone inhibits the reoxidation of nicotinamide adenine dinucleotide and, subsequently, the transfer of electrons between Complex 1 and coenzyme Q of the electron transport chain resulting in decreased formation of ATP (Horgan 1968). Studies have demonstrated that the differences in species sensitivity cannot be explained by differential blocking of the nicotinamide adenine dinucleotide (Lindahl 1961; Horgan 1968). The blocking of the electron transport chain and cellular respiration by rotenone causes the cell to become hyperoxic. Since oxygen is present but not being used by the electron transport chain, superoxides (O2 −) are formed resulting in a highly oxidative state and increasing the amount of cellular damage. Resistant species, like common carp, have been found to concentrate rotenone in tissues (e.g., liver) that have a high level of oxidative metabolism (Rach and Gingerich 1986), suggesting a possible link between the ability to metabolize ROS and sensitivity to rotenone.
Cells respond to oxidative stress by converting the ROS to hydrogen peroxide using copper/zinc superoxide dismutase (sod1; cytosol) and iron/manganese superoxide dismutase (sod2; mitochondria). Hydrogen peroxide decomposition is catalyzed by catalase (cat) or glutathione peroxidase (gpx). These four genes are thus commonly used biomarkers to indicate oxidative stress (Regoli et al. 2003; van der Oost et al. 2003; Monteiro et al. 2006). Some organisms can also respond to oxidative stress through an entirely different pathway by limiting ROS production in the mitochondria. This mechanism relies on uncoupling protein 2 (ucp2) to uncouple cellular respiration from phosphorylation. This mechanism is thought to be a powerful method to limit ROS production (Ricquier and Bouillaud 2000). This uncoupling pathway results in the formation of nitric oxide (NO), not H2O2 like that in the dismutase pathway.
Cellular proteins become denatured in the presence of ROS and need to be removed. Heat shock protein 70 (hsp70), another commonly used biomarker for oxidative stress, temporarily binds to the hydrophobic residues of cellular proteins and prevents them from aggregating and allowing them to refold or the denatured proteins are passed to the ubiquitination and proteolysis pathway for recycling (Luders et al. 2000; Clark and Peck 2009; Kayhan and Duman 2010; Roberts et al. 2010). When not participating in stress response, hsp70 inhibits a cascade that would otherwise trigger apoptosis (Mosser et al. 1997; Li et al. 2003).
We hypothesize that resistant fish can withstand higher levels of rotenone because they have lower uptake of rotenone, are more efficient at metabolizing rotenone or respond differently to ROS caused from rotenone exposure. To test this, we performed two separate trials evaluating the effects of rotenone on a susceptible species (silver carp) and a resistant species (bigmouth buffalo and/or bighead carp). The main objective of the first trial was to determine the rate of uptake and the maximum concentration of rotenone in the plasma of rotenone-sensitive silver carp and rotenone-resistant bigmouth buffalo. The objective of the second trial was to determine the transcriptional responses of a few genes responsible for detoxification (cyp1a and gst) and oxidative stress (sod1, sod2, cat, gpx, ucp2 and hsp70) to low-dose rotenone exposure of silver carp and bighead carp. Expression of hypoxia initiation factor 1-α (hif1-α), a commonly used hypoxia biomarker, was included to assess the state of hypoxia within the hepatocytes. Any use of trade, product or company name is for descriptive purposes only and does not imply endorsement by the U.S. Government.
Materials and methods
Plasma rotenone study
To determine the maximum amount of rotenone in plasma and the rate at which it accumulates in the plasma of silver carp and bigmouth buffalo, fish of both species were exposed to a commercial formulation of rotenone and the amount of rotenone determined following a 12-h exposure and compared with pre-exposure levels. Thirty-six mixed-gender bigmouth buffalo (718.9 ± 252.3 g) and thirty-six silver carp (983.9 ± 190.5 g) were stocked into each of two 500-L fiberglass tanks on September 29, 2010 for a 12-h (720-min) rotenone exposure. Tanks were filled with river water from the Illinois River near Havana, IL. River water temperature at the time of the study was 20.4°C; tanks were placed streamside to provide a natural photoperiod. Compressed air was supplied to each tank to maintain constant dissolved oxygen at ≥5 mg L−1. Procedures for handling and euthanasia of test animals were approved by the US Geological Survey Upper Midwest Environmental Sciences Center Animal Care and Use Committee.
Plasma was collected from three fish of each species per tank prior adding rotenone (Prenfish™, 5% rotenone, Prentiss Inc. Alpharetta, GA) at a commonly used exposure concentration, 50 ppb. Three fish of each species were euthanized by cranial concussion at 45, and 90, 180, 360 and 720 min after treatment. Immediately after euthanasia, whole blood was collected by cardiac puncture with a 3-mL syringe with a pre-heparinized 18-gauge needle and then expelled into separate 2-mL heparinized centrifuge tubes. Blood samples were centrifuged at 3000×g for 3 min. Plasma was removed and placed into separate 2-mL cyro-vials and stored at −80°C until plasma rotenone concentrations could be quantified.
A solid phase extraction (SPE) method was developed for the extraction of rotenone from fish plasma. Plasma samples were processed through Phenomenex Strata C18-E (55 μm, 70 Å, 100 mg, 1 mL) SPE columns. Each column was conditioned with acetonitrile (2 mL) followed by 0.5% formic acid (FA, 2 mL). An additional 0.5 mL FA was added to the column bed immediately before sample addition. Plasma sample (0.1–0.5 mL) was loaded on the column and brought to 1 mL total volume with FA. The sample was processed through the column at a rate of 1 mL min−1. The column was washed with 0.5 mL FA water and dried for 1–2 min. The sample was eluted from the column with ethyl acetate (1 mL) and collected in a 2-mL microcentrifuge tube. The sample was evaporated to dryness under nitrogen, reconstituted with 0.25 or 0.5 mL 70:30 acetonitrile/water, vortexed for 1 min, sonicated for 5 min and then filtered with a 0.2-μm PTFE syringe filter into a high-performance liquid chromatography (HPLC) vial.
The sample was then analyzed by HPLC using an Agilent 1200 Series equipped with a Waters Symmetry C18, 4.6 × 150 mm, 3.5 μm column. The HPLC parameters used were as follows: isocratic mobile phase, 70:30 acetonitrile/0.05% FA; flow rate, 1 mL min−1; column temperature, 45°C; injection volume, 20 μL; and detector wavelength, 296 nm.
Gene expression study
How the expression of a few common detoxification and oxidative stress biomarkers changes when silver carp and bighead carp are exposed to rotenone was determined by comparing the level of expression following exposure to that prior to exposure using quantitative PCR. Thirty bighead carp (41.2 ± 9.7 g) and eighteen silver carp (142.3 ± 65.7 g) were exposed to 25-ppb rotenone for 12 h (720 min). This size fish was used in this trial due to limited space and for ease of handling since silver carp jump upon being stressed. Water temperature was maintained at 20°C for a 12-h exposure period. Compressed air was supplied to each tank to maintain constant dissolved oxygen concentrations at ≥5 mg L−1. Procedures for handling and euthanasia of test animals were approved by the US Geological Survey Upper Midwest Environmental Sciences Center Animal Care and Use Committee.
Six bighead carp and 3 silver carp prior (T-0) were euthanized by cranial concussion and livers collected before initiation of exposure to a 12-h static bath of 25-ppb rotenone. Six bighead carp and 4 silver carp were euthanized by cranial concussion and livers taken at 45, 90, 180, 360 and 720 min of being exposed to rotenone. Liver samples were immediately placed in RNALater™ (Applied Biosystems/Ambion, Austin, TX, USA) and stored at −80°C. Before processing, RNALater™ was removed and liver tissues were immediately placed into 1 mL of TRIzol® (Invitrogen, Carlsbad, CA, USA) and then homogenized with a Tissue Tearor™ (Cole-Palmer, Vernon Hills, IL, USA). Total ribonucleic acids (RNA) were extracted according to the manufacturer’s instructions.
Total RNA was quantified spectrophotometrically using a BioPhotometer plus (Eppendorf, Westbury, NY, USA) and then diluted to a concentration of 1 μg total RNA μL−1 in DEPC-treated molecular-grade water. Quality of RNA was determined by gel electrophoresis. Total RNA (5 μg) was treated with DNase I (Fermentas Inc., Glen Burnie, MD, USA) according to the manufacturer’s instructions. Total RNA (2 μg) was then reverse transcribed to cDNA using High-Capacity Reverse Transcription Kit (Applied Biosystems, Foster City, CA, USA) primed with random hexamer primers according to the manufacturer’s instructions. A “no-reverse transcriptase” control was used for testing genomic DNA contamination for each sample.
Eight genes and a single reference gene, elongation factor 1-α (ef1-α), were selected based upon biological function (Table 1). Three commonly used housekeeping genes, 18S ribosomal RNA, β-actin and ef1-α, were evaluated to identify a candidate reference gene for this study. Of the three, ef1-α was the least variable (i.e., lowest CV) and was also found not to differ significantly among exposure times, but did differ between species. Annotated sequences for Hypophthalmichthys were retrieved from the NCBI database (http://www.ncbi.nlm.nih.gov/Genbank). Gene-specific primers sets (Table 1) were designed using Primer 3.0 v.0.4.0 (http://frodo.wi.mit.edu) and specificity for each primer was verified by BLAST analysis. All primers were synthesized by Invitrogen, Inc. (Carlsbad, CA USA).
All quantitative polymerase chain reactions (qPCR) were performed using SYBRgreen® technology. Reactions (total volume—12 μL) were comprised of 6.0 μL of SensiMix SYBR master mix (Bioline USA Inc., Tauton, MA USA), 360 nM of each primer and 16.7 ng of cDNA template and molecular-grade water. For controls, the template was replaced with molecular-grade water or no reverse transcriptase. All assays were carried out in duplicate using a Mastercycler® ep realplex real-time platform (Eppendorf Inc., Westbury, NY USA). The PCR conditions were as follows: 94°C for 15 s and 56°C for 30 s for 40 amplification cycles. A single product was confirmed using melt-curve analysis for every sample.
Data analysis
Morphometric data (i.e., body mass and total length) and plasma rotenone concentrations (Table 2) were compared among sample times using ANOVA and Tukey’s post hoc test. All qPCR results were log transformed and expressed as means ± standard error of the mean (SEM). All gene expression data were normalized to the expression of ef1-α and relative to samples collected at T0 in accordance with the ΔΔ-Ct method (Pfaffl 2001). Relative transcript abundance data were analyzed by 2-way ANOVA and Tukey’s post hoc test with species, sampling time and their interactions as the independent variables. All statistical analyses were performed using SigmaPlot® 11.0 (Systat Software, Inc., San Jose, CA USA) with a significance level of p < 0.05.
Results
Plasma rotenone
Silver carp were much more sensitive to the rotenone treatment than bigmouth buffalo. All silver carp died within the first 360 min of exposure to rotenone compared to no bigmouth buffalo mortality during the same time period. Two bigmouth buffalo were still alive at 720 min. Bigmouth buffalo had higher levels of rotenone in plasma than silver carp (p = 0.02) (Table 2). Rotenone concentrations were greatest in both species within 90 min of being exposed where plasma rotenone concentrations reached 173.3 ng mL−1 in bigmouth buffalo and 127.3 ng mL−1 in silver carp. When normalized to body mass (Table 2), the concentration of rotenone in bigmouth buffalo was near twice that in silver carp (p < 0.01), 0.180 ± 0.017 and 0.098 ± 0.017 ng mL−1 g−1 body mass, respectively.
Gene expression
Expression of genes related with detoxification (i.e., cyp1a and gst) exhibited very different patterns between species. Expression of cyp1a increased following exposure to rotenone in silver carp (p < 0.01) but decreased in bighead carp (p < 0.01) (Fig. 1a). Expression of silver carp cyp1a increased sevenfold (±0.73) in the first 360 min of rotenone exposure (p < 0.01), whereas expression of cyp1a in bighead carp decreased 0.45-fold (±0.20) during the same period. Expression of gst increased 9.85-fold (±1.37) in silver carp and 1.18-fold (±0.35) in bighead carp following 720 min of exposure to rotenone (Fig. 1b).
Relative transcript abundance for genes associated with detoxifying chemicals in livers bighead (n = 6) and silver carp (n = 4) during a 12-h exposure to 25-ppb rotenone. Transcript abundance (Log10 mean ± SEM) of cytochrome P450 1A (a) and glutathione S-transferase (b) are expressed relative to levels prior to rotenone exposure. White bars, light gray bars, medium gray bars, darker gray bars and nearly black bars represent values determined after 45, 90, 180, 360 and 720 min of exposure to rotenone, respectively. Bars with the same letter are not significantly different (p < 0.05)
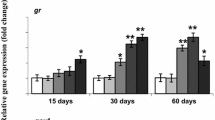
No differences were found in the level of expression of hif1-α in either species except at 360-min exposure (Fig. 2). The expression of hif1-α increased 30.32-fold (±15.99) in silver carp after 360 min of exposure (p < 0.01) but expression of hif1-α did not differ from pre-exposure levels in bighead carp at that same time. Instead in bighead carp, hif1-α steadily increased by 8.15-fold (±1.67) by 720 min (p = 0.02). The expression of hsp70 followed a pattern similar to hif1-α with silver carp expression of hsp70 increasing 31.63-fold (±21.64) after 360 min of rotenone exposure (p = 0.01) compared to the steady increase in bighead carp to 6.60-fold (±1.11) after 720 min of exposure (Fig. 3).
Relative transcript abundance of hypoxia initiation factor 1-α in livers bighead (n = 6) and silver carp (n = 4) during a 12-h exposure to 25-ppb rotenone. Transcript abundance (Log10 mean ± SEM) is expressed relative to levels prior to rotenone exposure. White bars, light gray bars, medium gray bars, darker gray bars and nearly black bars represent values determined after 45, 90, 180, 360 and 720 min of exposure to rotenone, respectively. Bars with the same letter are not significantly different (p < 0.05)
Relative transcript abundance of heat shock protein 70 (hsp70) in livers bighead (n = 6) and silver carp (n = 4) during a 12-h exposure to 25-ppb rotenone. Transcript abundance (Log10 mean ± SEM) is expressed relative to levels prior to rotenone exposure. White bars, light gray bars, medium gray bars, darker gray bars and nearly black bars represent values determined after 45, 90, 180, 360 and 720 min of exposure to rotenone, respectively. Bars with the same letter are not significantly different (p < 0.05)
Not only did hif1-α and hsp70 have high expression levels only at 360-min exposure, but expression of gpx, cat and sod1 was high at that same exposure time (Fig. 4a, b, c). In silver carp, gpx was elevated by 32.38-fold (±21.69), cat by 28.28-fold (±17.36) and sod1 by 26.43-fold (±13.76) 360 min after being exposed to rotenone (p ≤ 0.01) but unlike hif1-α and hsp70, expression of gpx, cat and sod1 did not change in bighead carp throughout the study. Genes linked with oxidative stress in the mitochondria, upc2 and sod2, had a similar expression pattern as gpx, cat and sod1 in silver carp where only the transcript level at 360 min was significantly elevated (Fig. 5a, b). Expression of ucp2 was also highly variable in silver carp (CV = 0.80) compared with bighead carp. However, in bighead carp, expression of ucp2 increased 84.73-fold (±20.01) after 720 min of being exposed (p < 0.01) and exhibited a linear trend (Fig. 5a).
Relative transcript abundance for genes associated with reducing reactive oxygen species in the cytosol in livers bighead (n = 6) and silver (n = 4) carp during a 12-h exposure to 25-ppb rotenone. Transcript abundance of a glutathione peroxidase (gpx), b catalase (cat) and c copper/zinc superoxide dismutase (sod1) relative to transcripts abundance prior to exposure of low levels rotenone. White bars, light gray bars, medium gray bars, darker gray bars and nearly black bars represent values determined after 45, 90, 180, 360 and 720 min of exposure to rotenone, respectively. Bars with the same letter are not significantly different (p < 0.05)
Relative transcript abundance for genes associated with reducing reactive oxygen species in the mitochondria in livers bighead (n = 6) and silver carp (n = 4) during a 12-h exposure to 25-ppb rotenone. Transcript abundance of a uncoupling protein 2 (ucp2) and b iron/manganese superoxide dismutase (sod2) relative to transcripts abundance prior to exposure of low levels rotenone. White bars, light gray bars, medium gray bars, darker gray bars and nearly black bars represent values determined after 45, 90, 180, 360 and 720 min of exposure to rotenone, respectively. Bars with the same letter are not significantly different (p < 0.05)
Discussion
Data from previous studies indicate that rotenone applied according to the manufacturer’s label is toxic to silver and bighead carp (Henderson 1976; Marking and Bills 1976; Chapman et al. 2003; Rach et al. 2009). Our study demonstrated that rotenone, applied according to the manufacturer’s label, is toxic to bigmouth buffalo in a 12-h exposure. We observed that silver carp were more sensitive to rotenone than bigmouth buffalo since all silver carp were dead by 360 min, while none of the bigmouth buffalo were found dead during that same time while exposed to 50-ppb rotenone. Since rotenone blocks cellular respiration, we expected that the molecular response of rotenone-exposed fish would mimic the response to hypoxic conditions. Both silver carp and bigmouth buffalo are known to tolerate low dissolved oxygen levels (Killgore and Hoover 2001). Silver carp in this study were observed gasping at the surface during the exposure. Silver carp have been found to lengthen their lower jaw in response to low dissolved oxygen, which allows them to increase intake of surface water (Adamek and Groch 1993) and potentially occupy what would otherwise be marginal habitat. However, if this morphological change is the only modification enabling silver carp to withstand low oxygen levels, that is, corresponding physiological or metabolic response mechanisms, then there is likely minimal effect given the rapid onset of cellular hypoxic-like conditions after rotenone exposure. Bigmouth buffalo remained below the surface for much of the exposure, suggesting a greater physiological, rather than morphological, adaptation to low oxygen levels. Maximum plasma rotenone concentrations were higher in bigmouth buffalo during the 12-h exposure than the levels found in silver carp, similar to those reported for rotenoid residues in tissues of rotenone-resistant and rotenone-sensitive fishes (Rach and Gingerich 1986). Rotenoid residues in common carp, a rotenone-resistant species, were greater than those of the rotenone-sensitive yellow perch or bluegill (Rach and Gingerich 1986). Rotenoid residues were greatest in the bile and liver (Rach and Gingerich 1986), suggesting that hepatocytes responded to rotenone exposure by modifying the xenobiotic and then excreting it into bile.
Expression of two genes in liver that are associated with physiological response to xenobiotic exposure, cyp1a and gst, differed substantially between silver carp and bighead carp. Expression of cyp1a increased in the liver of silver carp, while decreasing in the liver of bighead carp. These expression patterns suggest both cyp1a and gst may play different roles in silver carp compared with those in bighead carp. Cytochrome P450s are a highly diverse family of enzymes, and different isozymes may be responsible for the detoxification of rotenone in silver and bighead carp. Erickson et al. (1992), for example, suggested that rotenone may be metabolized by more than one cytochrome P450 isozyme in rainbow trout and several cytochrome P450s were found to detoxify cocaine in mullet (Arinc and Bozcaarmutlu 2003). Different isozymes of cytochrome P450 from those used by silver carp may be used by bighead carp to detoxify rotenone. Identification and function of each cytochrome P450 isozyme remain unknown for Hypophthalmichthys and should be studied further.
Like the cytochrome P450s, little is currently known about the different forms of glutathione S-transferases in Hypophthalmichthys. Glutathione S-transferases have been used as a biomarker to assess stress response in common carp, Nile tilapia, Oreochromis niloticus, armored catfish, Pterygoplichthys anisitsi, to a petroleum products and insecticides (Tlili et al. 2010; Nogueira et al. 2011; Puerto et al. 2011; Toni et al. 2011). An injection of microcystin stimulated a transcriptional response in gst in bighead carp (Li et al. 2010). In our study, up-regulation of gst suggests the use of a glutathione detoxification pathway in silver carp. However, no change in gst levels was found in bighead carp, suggesting a different pathway for the conjugation of rotenone in this species.
Not only is the direct detoxification of rotenone important, but since rotenone inhibits cellular respiration by blocking the electron transport chain, it stimulates formation of toxic superoxides. Superoxide dismutases (i.e., sod1 and sod2), cat and gpx are the first line of defense against this type of toxicity (Pandey et al. 2003; Monteiro et al. 2006). Superoxide dismutases (sod) convert superoxides to molecular oxygen or hydrogen peroxide, while catalase (cat) converts hydrogen peroxide to water and oxygen. Thus, the combination of sod and cat have been used as biomarkers to indicate the production of reactive oxygen species (Regoli et al. 2003; van der Oost et al. 2003; Monteiro et al. 2006). Studies have found that rotenone stimulates expression of sod1, cat and gpx (Molina-Jimenez et al. 2005; Shaikh and Nicholson 2009). Shaikh and Nicholson (2009) reported that sod1 increased in human microglia cells exposed to a low dose of rotenone for 4 weeks. In another study, cat and gpx were found to be up-regulated in human neuroblastoma cells after 16 h of incubating in rotenone (Molina-Jimenez et al. 2005). This suggests the length of time an animal/cell is exposed to rotenone is critical to stimulate a transcriptional response. In our study, significant up-regulation of genes linked with ROS in the cytosol in silver carp, specifically sod1, cat and gpx, were only observed following exposure for 360 min. The increased abundance of transcripts at the 360-min time point may indicate that superoxides and lipid peroxides need to reach a critical threshold concentration before inducing a transcriptional response in silver carp. In our experiment, it appears that some critical transcription-induction threshold was met when silver carp were exposed to rotenone between 180 and 360 min. Transcript levels for sod1, cat and gpx in bighead carp, unlike those in silver carp, did not change pre-exposure levels. Superoxides and lipid peroxides may not have accumulated in the cytosol of bighead carp hepatocytes as postulated to have occurred in silver carp, or perhaps another mechanism, possibly at the mitochondrial level, may have limited ROS production in bighead carp.
It has been suggested that uncoupling proteins (e.g., ucp2) could be a powerful system to limit the production of ROS (Ricquier and Bouillaud 2000). Uncoupling protein 2 has been reported to increase NO production (Fong et al. 2011), which plays an important role in redox reactions. Like ucp2, sod2 is also known to reduce ROS associated with the mitochondria through the production of hydrogen peroxide (Sies 1986; Nordberg and Arner 2001). Superoxide dismutase 2 also does not uncouple respiration and phosphorylation. Up-regulation of ucp2 and not sod2 in livers of bighead carp in our study suggests potential uncoupling and the production of NO as a mechanism to combat ROS on mitochondria, whereas in silver carp, sod2 is consistently elevated throughout our study and ucp2 only increases following 6 h of exposure. Not only does NO combat ROS, but it also has been found to inhibit apoptosis in hepatocytes (Kim et al. 1997; Brune 2003) and induces expression of hsp70 to protect from tumor necrosis factor-alpha induced apoptosis (Kim et al. 1997).
Several physiological functions of hsp70 have been identified in fish. Since oxidative stress can increase protein degradation, concomitant increases in hsp70 are expected (Luders et al. 2000; Clark and Peck 2009; Kayhan and Duman 2010; Roberts et al. 2010). Exposure to different xenobiotics (e.g., heavy metals, microcystin) caused increased or variable induction of hsp70 expression in a diverse array of fish (He et al. 2010; Olsvik et al. 2011; Rajeshkumar and Munuswamy 2011). Besides protecting damaged proteins, hsp70 has been also found to bind to apoptosis protease activating factor-1 and block a apoptosis (Mosser et al. 1997; Ravagnan et al. 2001), a pathway that rotenone has been found to initiate (Li et al. 2003). Both functions of hsp70 may exist in bighead and silver carp. In bighead carp, where hsp70 remained at pre-exposure levels, the amount of proteins requiring recycling may have been below the threshold to induce hsp70 expression. Additionally, available hsp70 may have been at levels sufficient to block apoptosis. In silver carp, the up-regulation of hsp70 may have been either to increase protein recycling demand or to block apoptosis or both.
In summary, our studies demonstrate that species absorb rotenone at similar rates, but rotenone-resistant species (e.g., bigmouth buffalo) tolerate higher plasma levels of rotenone than sensitive species like silver carp. Different isoforms of detoxifying enzymes, with potentially varying efficiencies, may be used by these species to detoxify rotenone, as shown by the up-regulation of cyp1a and gst in silver carp but down-regulation of cyp1a and minimal change in gst in bighead carp. Finally, the ability of a species to combat ROS appears to be important to the capacity to tolerate rotenone exposure in that species. Rotenone-resistant bighead carp up-regulate ucp2, in response to rotenone exposure, presumably to combat rotenone-induced ROS in the mitochondria. This may limit the effects of rotenone in the cytosol and thus explain the lack of change in the expression of hsp70. Available hsp70 in bighead carp in combination with the possible increased amounts of NO from uncoupling cellular respiration and phosphorylation could block the apoptotic affects of rotenone. The up-regulation of sod1 and sod2, which produce hydrogen peroxide, is not likely to block apoptosis in silver carp. Up-regulation of hsp70 in silver carp suggests increased cellular protein damage with a potential shift in the use of hsp70 to protein degradation away from inhibition of apoptosis.
References
Adamek Z, Groch L (1993) Morphological adaptations of silver carp (Hypophthalmicthys molitrix) lips as a reaction on hypoxic conditions. Folia Zool 42:179–182
Al-Ghais SM, Ali B (1999) Inhibition of glutathione S-transferase catalyzed xenobiotic detoxication by organotin compounds in tropical marine fish tissues. Bull Environ Contam Toxicol 62:207–213
Arinc E, Bozcaarmutlu A (2003) Catalyzation of cocaine N-demethylation by cytochromes P4502B, P4503A, and P4502D in fish liver. J Biochem Mol Toxicol 17:169–176
Brune B (2003) Nitric oxide: NO apoptosis or turning it ON? Cell Death Differ 10:864–869
Bucheli TD, Fent K (1995) Induction of cytochrome P450 as a biomarker for environmental contamination in aquatic ecosystems. Crit Rev Environ Sci Technol 25:201–268
Chapman D, Fairchild J, Carollo B, Deters J, Feltz J, Witte C (2003) An examination of the sensitivity of bighead carp and silver carp to antimycin-A and rotenone. In: U.S. Geological Survey, Columbia Environmental Research Center, Columbia
Clark MS, Peck LS (2009) Triggers of the HSP70 stress response: environmental responses and laboratory manipulation in an Antarctic marine invertebrate (Nacella concinna). Cell Stress Chaperones 14:649–660
Collier TK, Anulacion BF, Stein JE, Goksoyr A, Varanasi U (1995) A field evaluation of cytochrome P4501A as a biomarker of contaminant exposure in 3 species of flatfish. Environ Toxicol Chem 14:143–152
Curtis LR, Carpenter HM, Donohoe RM, Williams DE, Hedstrom OR, Deinzer ML, Bellstein MA, Foster E, Gates R (1993) Sensitivity of cytochrome P4501–A1 induction in fish as a biomarker for distribution of TCDD and TCDF in the Willamette River, Oregon. Environ Sci Technol 27:2149–2157
Egaas E, Sandvik M, Fjeld E, Kallqvist T, Goksoyr A, Svensen A (1999) Some effects of the fungicide propiconazole on cytochrome P450 and glutathione S-transferase in brown trout (Salmo trutta). Comp Biochem Physiol C-Pharmacol Toxicol Endocrinol 122:337–344
Eggens M, Bergman A, Vethaak D, Vanderweiden M, Celander M, Boon JP (1995) Cytochrome P4501A indexes as biomarkers of contaminant exposure—results of a field study with plaice (Pleuronectes platessa) and flounder (Platichthis flesus) from the southern North Sea. Aquatic Toxicol 32:211–225
Erickson DA, Laib FE, Lech JJ (1992) Biotransformation of rotenone by hepatic microsomes following pretreatment of rainbow trout with inducers of cytochrome P450. Pesticide Biochem Physiol 42:140–150
Fitzpatrick PJ, Ohalloran J, Sheehan D, Walsh AR (1997) Assessment of a glutathione S-transferase and related proteins in the gill and digestive gland of Mytilus edulis (L), as potential organic pollution biomarkers. Biomarkers 2:51–56
Fong RN, Fatehi-Hassanabad Z, Lee SC, Lu H, Wheeler MB, Chan CB (2011) Uncoupling protein-2 increases nitric oxide production and TNFAIP3 pathway activation in pancreatic islets. J Mol Endocrinol 46:193–204
Gingerich WH, Rach JJ (1985) Uptake, biotransformation, and elimination of rotenone by bluegills (Lepomis macrochirus). Aquatic Toxicol 6:179–196
He S, Liang XF, Li RQ, Li GG, Wang L, Shen D (2010) Molecular characterization of heat shock protein 70 genes in the liver of three warm freshwater fishes with differential tolerance to microcystin-LR. J Biochem Mol Toxicol 24:293–302
Henderson S (1976) Tolerance of silver and bighead carp to often used pond treatment chemicals. Completion report. In: Arkansas Game and Fish Commission, Little Rock
Horgan DJ (1968) Studies on the respiratory chain-linked reduced nicotinamide adenine dinucleotide dehydrogenase. XIII. Binding sites of rotenone, piericidin A, and amytal in the respiratory chain. J Biol Chem 243:10
Kayhan FE, Duman BS (2010) Heat shock protein genes in fish. Turkish J Fish Aquatic Sci 10:287–293
Killgore KJ, Hoover JJ (2001) Effects of hypoxia on fish assemblages in a vegetated waterbody. J Aquatic Plant Manage 39:40–44
Kim YM, Talanian RV, Billiar TR (1997) Nitric oxide inhibits apoptosis by preventing increases in caspase-3-like activity via two distinct mechanisms. J Biol Chem 272:31138–31148
Lennon RE (1970) A resume on field applications of antimycin-A to control fish. Investig Fish Control 40:19
Li NY, Ragheb K, Lawler G, Sturgist J, Rajwa B, Melendez JA, Robinson JP (2003) Mitochondrial complex I inhibitor rotenone induces apoptosis through enhancing mitochondrial reactive oxygen species production. J Biol Chem 278:8516–8525
Li L, Xie P, Guo LG (2010) Antioxidant response in liver of the phytoplanktivorous bighead carp (Aristichthys nobilis) intraperitoneally-injected with extracted microcystins. Fish Physiol Biochem 36:165–172
Lindahl PE (1961) The effect of rotenone on the respiration and its point of attack. Exp Cell Res 23:12
Luders J, Demand J, Hohfeld J (2000) The ubiquitin-related BAG-1 provides a link between the molecular chaperones Hsc70/Hsp70 and the proteasome. J Biol Chem 275:4613–4617
Marking LL, Bills TD (1976) Toxicity of rotenone to fish in standardized laboratory tests. Investig Fish Control 72:1–11
Molina-Jimenez MF, Sanchez-Reus MI, Cascales M, Andres D, Benedi J (2005) Effect of fraxetin on antioxidant defense and stress proteins in human neuroblastoma cell model of rotenone neurotoxicity. Comparative study with myricetin and N-acetylcysteine. Toxicol Appl Pharmacol 209:214–225
Monteiro DA, de Almeida JA, Rantin FT, Kalinin AL (2006) Oxidative stress biomarkers in the freshwater characid fish, Brycon cephalus, exposed to organophosphorus insecticide Folisuper 600 (methyl parathion). Comp Biochem Physiol C-Toxicol Pharmacol 143:141–149
Mosser DD, Caron AW, Bourget L, DenisLarose C, Massie B (1997) Role of the human heat shock protein hsp70 in protection against stress-induced apoptosis. Mol Cell Biol 17:5317–5327
Nogueira L, Rodrigues ACF, Tridico CP, Fossa CE, de Almeida EA (2011) Oxidative stress in Nile tilapia (Oreochromis niloticus) and armored catfish (Pterygoplichthys anisitsi) exposed to diesel oil. Environ Monit Assess 180:243–255
Nordberg J, Arner ESJ (2001) Reactive oxygen species, antioxidants, and the mammalian thioredoxin system. Free Radical Biol Med 31:1287–1312
Olsvik PA, Brattas M, Lie KK, Goksoyr A (2011) Transcriptional responses in juvenile Atlantic cod (Gadus morhua) after exposure to mercury-contaminated sediments obtained near the wreck of the German WW2 submarine U-864, and from Bergen Harbor, Western Norway. Chemosphere 83:552–563
Pandey S, Parvez S, Sayeed I, Haque R, Bin-Hafeez B, Raisuddin S (2003) Biomarkers of oxidative stress: a comparative study of river Yamuna fish Wallago attu (Bl. & Schn.). Sci Total Environ 309:11
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:2002–2007
Puerto M, Gutierrez-Praena D, Isabel Prieto A, Pichardo S, Jos A, Luis Miguel-Carrasco J, Vazquez CM, Camean AM (2011) Subchronic effects of cyanobacterial cells on the transcription of antioxidant enzyme genes in tilapia (Oreochromis niloticus). Ecotoxicology 20:479–490
Rach JJ, Gingerich WH (1986) Distribution and accumulation of rotenone in tissue of warm-water fishes. Trans Am Fish Soc 115:214–219
Rach JJ, Boogaard M, Kolar C (2009) Toxicity of rotenone and antimycin to silver carp and bighead carp. N Am J Fish Manage 29:388–395
Rajeshkumar S, Munuswamy N (2011) Impact of metals on histopathology and expression of HSP 70 in different tissues of Milk fish (Chanos chanos) of Kaattuppalli Island, South East Coast, India. Chemosphere 83:415–421
Ravagnan L, Gurbuxani S, Susin SA, Maisse C, Daugas E, Zamzami N, Mak T, Jaattela M, Penninger JM, Garrido C, Kroemer G (2001) Heat-shock protein 70 antagonizes apoptosis-inducing factor. Nat Cell Biol 3:839–843
Regoli F, Winston GW, Gorbi S, Frenzilli G, Nigro M, Corsi I, Focardi S (2003) Integrating enzymatic responses to organic chemical exposure with total oxyradical absorbing capacity and DNA damage in the European eel Anguilla anguilla. Environ Toxicol Chem 22:2120–2129
Ricquier D, Bouillaud F (2000) Mitochondrial uncoupling proteins: from mitochondria to the regulation of energy balance. J Physiol Lond 529:3–10
Roberts RJ, Agius C, Saliba C, Bossier P, Sung YY (2010) Heat shock proteins (chaperones) in fish and shellfish and their potential role in relation to fish health: a review. J Fish Dis 33:789–801
Sayeed I, Parvez S, Pandey S, Bin-Hafeez B, Haque R, Raisuddin S (2003) Oxidative stress biomarkers of exposure to deltamethrin in freshwater fish, Channa punctatus Bloch. Ecotoxicol Environ Saf 56:295–301
Shaikh SB, Nicholson LFB (2009) Effects of chronic low dose rotenone treatment on human microglial cells. Mol Neurodegeneration 4:1–13
Sies H (1986) Biochemistry of oxidative stress. Angew Chem Int Ed Engl 25:1058–1071
Tlili S, Jebali J, Banni M, Haouas Z, Mlayah A, Helal AN, Boussetta H (2010) Multimarker approach analysis in common carp Cyprinus carpio sampled from three freshwater sites. Environ Monitor Assess 168:285–298
Toni C, Ferreira D, Kreutz LC, Loro VL, Gil Barcellos LJ (2011) Assessment of oxidative stress and metabolic changes in common carp (Cyprinus carpio) acutely exposed to different concentrations of the fungicide tebuconazole. Chemosphere 83:579–584
van der Oost R, Beyer J, Vermeulen NPE (2003) Fish bioaccumulation and biomarkers in environmental risk assessment: a review. Environ Toxicol Pharmacol 13:57–149
Acknowledgments
The authors would like to thank Adam Ladwig and Todd Severson for their assistance in dissecting fish and tissue collections. This work was financially supported through funds from the Great Lakes Restoration Initiative.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Amberg, J.J., Schreier, T.M. & Gaikowski, M.P. Molecular responses differ between sensitive silver carp and tolerant bighead carp and bigmouth buffalo exposed to rotenone. Fish Physiol Biochem 38, 1379–1391 (2012). https://doi.org/10.1007/s10695-012-9625-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10695-012-9625-1