Abstract
The contamination levels and ecological risks of heavy metals in the sediments of the Nansi Lake were investigated. The contents of Cd, Cr, Cu, Pb, Zn, Ni, and Co in the surface sediments collected at 20 sites ranged from 0.08 to 1.12, 58.92 to 135.62, 38.09 to 78.65, 24.51 to 53.95, 110.51 to 235.36, 11.30 to 65.40, and 4.12 to 20.14 mg/kg, respectively. The results of partitioning analysis revealed that the proportions of soluble and exchangeable fraction were less than 1 %, the proportions of carbonate, amorphous oxides, organic matter, and crystalline oxides fraction were less than 10 %, and 10.52 % of Cd was associated with carbonate. The average proportions in the residual fraction ranged from 48.62 % for Cu to 73.76 % for Ni, indicating low mobility and bioavailability. The geoaccumulation index (I geo), relative enrichment factor (REF), sediment pollution index (SPI), and potential effect concentration quotient (PECQ) values of the heavy metals in the sediments were not in agreement with each another. The average REF values of Cd and Zn were higher than those of other metals. However, the average PECQ values were higher for Cr and Ni than those of other metals, indicating that these two metals would cause higher adverse biological effects. Therefore, it is suggested that future management and pollution control might focus on Cd, Zn, Cr, and Ni in the sediments of the Nansi Lake.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Heavy metals contamination in sediments has become a critical area of growing concern worldwide in recent years, because it is one of the greatest threats to water quality, the aquatic ecosystem, and human health. Trace element pollution is regarded as a serious threat to the environment due to pollutants’ toxicity, environmental persistence, and the ability to be incorporated into food chains (Long et al. 2006; Sadiq et al. 2007; Sakan et al. 2009). Sediment has been widely recognized as the main sink for pollutants including trace metallic and metalloid elements (Förstner and Wittmann 1981; Jain et al. 2005). Heavy metals accumulate in sediments via several pathways: disposal of liquid effluents, terrestrial runoff, and leachate carrying chemicals originating from numerous urban, industrial, and agricultural activities, as well as atmospheric deposition (Rivail Da Silva et al. 1996; Maxwell and Kastenberg 1999; Karageorgis et al. 2002; Mucha et al. 2003). Metal-contaminated sediments may release metals into the overlying water column and thus pose a risk to aquatic life and ecosystems (Calmano et al. 1990). Sediment can reflect the quality of water system, and its capacity to accumulate contaminants is an important factor to assess environmental impact on aquatic ecosystems (Silva and Rezende 2002; Jain et al. 2008). Therefore, sediments have been widely used as environmental indicators, and their monitoring can present important information on the degree of pollution in a given region (Singh et al. 2002).
Heavy metals discharged into a river system either by natural or anthropogenic sources (Ouyang et al. 2002; Choi et al. 2007; Yao et al. 2007). Anthropogenic sources include industrial and energy production, vehicle exhaust, waste disposal, fossil fuel combustion, and fertilizer and pesticide use. A large proportion of heavy metals deposited in aquatic sediments are adsorbed to and co-precipitated with sediments (Gaur et al. 2005; Varol 2011). Numerous studies have been conducted to assess and establish the extent of heavy metals in sediments worldwide (Samecka-Cymerman and Kempers 2001; Mays and Edwards 2001; Bragato et al. 2006), including lakes (Ozmen et al. 2004; Kamala-Kannan et al. 2008; Zhang et al. 2009; Selvam et al. 2012; Suresh et al. 2012; Kankilic et al. 2013; Iqbal et al. 2013; Li et al. 2014; Kukrer et al. 2014; Swarnalatha et al. 2014), rivers (Fan et al. 2002; Audry et al. 2004; Singh et al. 2003), and marine areas (Yuan et al. 2012; Dou et al. 2013; Liang et al. 2004).
Additionally, the overall behaviors of heavy metals in an aquatic environment are strongly influenced by the association of metals with various geochemical phases in sediments, which determine their mobilization capacity, and bioavailability (Tessier et al. 1979). Aquatic sediments consist of several geochemical phases, mainly carbonates, sulfides, organic matter, iron, and manganese oxides, and clays, which act as scavengers of trace metals in the environment (Tessier and Campbell 1987). Therefore, sequential selective extraction (SSE) techniques have been developed to separate the total metal content into these forms (Tessier et al. 1979; Ure et al. 1993; Han and Banin 1999). The most widely used methods are based on sequential extraction procedures whereby several reagents are used consecutively to extract operationally defined phases from the sediment in a sequence.
The present study investigated the total contents and the chemical partitioning of heavy metals in the sediments of the Nansi Lake. The objectives of this study were to assess the enrichment, contamination, and potential ecological risk of heavy metals in the sediments of the Nansi Lake. These results from this study provide an initial judgment on the cleanup and dredging operation and management of the sediments in this region.
Materials and methods
Study area
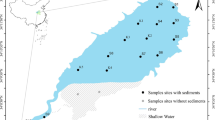
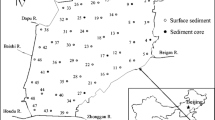
The Nansi Lake (34°27′–35°20′N, 116°34′–117°21′E) is a typical shallow lake with an area of 1,266 km2 and average depth of 1.46 m, including four lakes: the Nanyang Lake, the Dushan Lake, the Zhaoyang Lake, and the Weishan Lake (Fig. 1). It is the largest freshwater lake in the southwest of Shandong province and become one of the largest buffering reservoirs in the East Route of China’s South-North Water Transfer Project. After the building of a dam in 1960s, Nansi Lake was divided into two parts: the upper lake and lower lake. The upper lake including the Nanyang Lake, the Dushan Lake, and the Zhaoyang Lake drains an area of more than 1,000 km2. The lower lake referring to the Weishan Lake drains an area of more than 200 km2.
The urban streams are often contaminated by effluents and other wastes from industrial and domestic sources. There has been a rapid economic development in the region around the Nansi Lake since 1970s and this rapid industrialization and urbanization might lead to an excessive release of pollutants into the Nansi Lake. Generally, the untreated effluents from industrial and municipal activities, the runoff from mining sites and agricultural land, and the deposition of air pollutants all contribute to the increase of heavy metal levels in the sediments. According to the “Water pollution Prevention Planning of the South-to-North Water Diversion Project (east route) of Shandong Section”, water quality of the lake should be better than the Grade III of the “China surface water quality standard”. However, previous researches indicated that the sediments of the Nansi Lake and its main inflow rivers were polluted by heavy metals (Liu et al. 2007).
Sediment sampling
In this study, surface sediment samples of the Nansi Lake were collected at 20 sites in May 2013 (Fig. 1). At each site, the surface sediment samples (from 0 to 15 cm depth) from five locations were collected using cable operated sediment samplers (Van Veen grabs, Eijkelkamp), mixed together to make composite samples to improve site representation, and placed in dark-colored polyethylene bags. Then they were taken to the laboratory immediately after collection. All sediment samples were freeze-dried (FD-1A, China), slightly crushed, passed through a two millimeter sieve, and stored at 4 °C in glass bottles in the dark before analysis.
Analytical methods
The pH value of the sediment samples was measured in a 1:10 solid/liquid ratio suspension using a combination pH electrode (Orion, USA). The amount of sediment organic matter (SOM) was determined by the potassium dichromate dilution heat colorimetric method (Bao 1999). Carbonate content was measured by the manometric method following the addition of dilute HCl to dissolve carbonates (Loeppert and Suarez 1996). The grain size distribution of the sediment samples was analyzed using a LS 230 laser diffraction particle analyzer (Beckman Coulter) and the percentages of clay (<2 μm), silt (2–20 μm), and sand fractions (20–2,000 μm) were calculated. Sediment samples were digested using the method of HClO4–HNO3–HF (GB/T 17138, 17141-1997). The contents of Cr, Mn, Ti, Al, Fe, Ca, Mg, Na, and K in the digestion solution were measured by inductively coupled plasma atomic emission spectrometry (ICP-AES) (IRIS Intrepid II, Thermo Electron Co., USA). While Cd, Co, Cu, Ni, Pb, and Zn were measured with an inductively coupled plasma mass spectrometer (ICP-MS) (X Series II, Thermo Electron).
The accuracy of the method was checked by replicate analyses of the elements in the certified reference materials GSS1 and GSS3 from the Institute of Geophysical and Geochemical Exploration, Chinese Academy of Geological Science. The results demonstrated that the analytical error (accuracy) for all elements ranged from −8.5 to 11.4 %. In details, the analytical error was −8.5 to 8.3 % for Cd, −4.1 to 11.4 % for Co, −6.4 to 7.9 % for Cr, −4.2 to 3.2 % for Cu, −5.0 to 1.9 % for Mn, −6.6 to 7.2 % for Ni, −0.4 to 8.8 % for Pb, −3.6 to 2.5 % for Ti, −0.7 to 2.5 % for Zn, −5.3 to 4.4 % for Al, −3.9 to 5.8 % for Fe, −2.8 to −1.9 % for Mg, −1.7 to 5.8 % for Ca, −3.5 to −0.9 % for Na, and −1.2 to −0.4 % for K.
Taking into account the behavior of heavy metals in the sediment samples, the six-step sequential extraction procedure from Han and Banin (1995) was performed using 1 g of dry sediment. The extraction procedures were designed to separate specific environmental compartments of heavy metals and other trace metals in the sediment samples (Table 1). The chemical forms were operationally fractioned into six phases: (1) the soluble and exchangeable fraction (EXC); (2) bound to carbonate minerals (CARB); (3) bound to easily reducible oxides (ERO); (4) bound to organic matter (OM); (5) bound to reducible oxides (RO); and (6) the residual fraction (RES). In this study, only the first five forms displayed in Table 1 were extracted and measured, and the contents of meals in the residual fraction were calculated by subtracting the sum of the first five forms from the total content.
Evaluation method of ecological toxicity of heavy metals in sediments of the Nansi Lake
Geoaccumulation index (I geo)
In this study, the index of geoaccumulation proposed by Müller (1969) and described by Förstner et al. (1990) was applied as a quantitative measure of the degree of metal pollution in aquatic sediment [Eq. (1)].
where C n is the measured content in the sediment, B n is the background content in the shale sediment (Table 2), and 1.5 is a correction factor due to lithogenic effluents.
The following classification is given for the geoaccumulation index by Förstner et al. (1990): <0 = practically unpolluted, 0–1 = unpolluted to moderately polluted, 1–2 = moderately polluted, 2–3 = moderately to strongly polluted, 3–4 = strongly polluted, 4–5 = strongly to very strongly polluted, and >5 = very strongly polluted.
Relative enrichment factor (REF)
For a better estimation of anthropogenic input, a REF was calculated for each trace element by dividing its ratio to a normalizing element by the same ratio found in the chosen baseline. The upper continental crust (UCC) has often been used as the baseline and Al has often been used as the normalizing element to calculate the enrichment factors of trace metals (TM) (Mil-Homens et al. 2007; Pasternack and Brown 2006). In this study, the REF for each trace element was calculated from the formulae:
where the concentrations of TM and Al in the UCC were taken from Wedepohl (1995) (Tables 2, and 3); REF values of <2, 2–5, 5–20, 20–40, >40 suggest no or minimal, moderate, significant, strong, and extreme enrichment or pollution, respectively (Sutherland 2000).
Sediment pollution index (SPI)
Singh et al. (2002) introduced the SPI concept. The SPI is a multi-metal approach for an assessment of sediment quality with respect to trace metal concentrations and toxicity. The SPI in this study can be expressed as:
where EF is the ratio between total sediment content in a given sample and the average shale concentration of a metal m (Table 2) and W m is the toxicity weight for a metal m. A toxicity weight 1 was assigned to Cr and Zn, a weight of 2 was assigned to Cu and Ni, a weight of 5 was assigned to Pb, and a weight of 300 was assigned to Cd (Singh et al. 2002). The following classification is given for the SPI: 0–2 = natural sediment, 2–5 = low polluted sediment, 5–10 = moderately polluted sediment, 10–20 = highly polluted sediment, and >20 = dangerously polluted sediment.
Probable effect concentration quotient (PECQ)
The probable effect concentrations (PECs) were intended to identify contaminant concentrations above which harmful effects on sediment-dwelling organisms were expected to frequently occur. The PECs, developed by Ingersoll et al. (2001), have been adopted as an informal tool to evaluate sediment chemical data in relation to possible adverse effects on aquatic biota. A PEC quotient (PECQ) was calculated for each chemical in each sample by dividing the concentration of a chemical by the PEC for that chemical (Ingersoll et al. 2001). The mean PECQs (mPECQ) were then calculated for each sample by summing the individual quotient for each chemical and dividing this sum by the number of reliable PECs evaluated in Table 2 (Ingersoll et al. 2001).
where TMsample is the content of a give trace metal in a sample, TMPEC is the PEC vale for a give trace metal (Table 3), PECQ i is the PECQ value for ith trace metal, n is the total number of trace metal.
The mPECQ values of ≤0.1, >0.1–1.0, and >1.0 indicate relatively uncontaminated sediments, moderately contaminated sediments, and highly contaminated sediments, respectively (Long et al. 2006).
Results and discussion
General properties of the sediments in the Nansi Lake
A selection of general physical and chemical characteristics of the sediments is shown in Table 2. The amount of sediment organic matter (SOM) ranged from 4.32 to 14.32 %. Higher OM content was observed at some sites located in the Nanyang Lake and Dushan Lake, especially in the Dushan Lake. A long-term monitor indicated that annually about 2,900 million tons of untreated domestic sewage and industrial wastewater from the Jining, Zaozhuang, and Heze cities discharged into the Nansi Lake through the thirteen main inflow rivers flowing into the Nanyang Lake and Dushan Lake, and the contents of chemical oxygen demand (COD) and biological oxygen demand (BOD) in these rivers were higher than the Grade V of the “China surface water quality standard” (Zhang et al. 1999). In addition, inorganic and organic materials are carried and deposited by water from the upper lake to the lower lake under natural conditions. Therefore, the content of SOM was higher in the upper lake sediments than that in the lower lake sediments. Faster development of aquaculture as well as richness in aquatic plants in the Dushan Lake than other lakes might also contribute to high SOM contents in the Dushan Lake sediments (Qu et al. 2001). The pH values of the sediments in the Nansi Lake were in the range of 7.01–8.21, showing an alkaline characteristic. The carbonate content ranged from 0.01 to 1.17 %. The amounts of Al and Fe in the sediments of the Nansi Lake ranged from 4.10 to 7.46 and 2.74 to 4.13 %, respectively (Table 2). For all the sediment samples of the Nansi Lake, the Al and Fe contents were in the range of 4.46–7.49 and 2.82–4.3 %, respectively, had no big change. The content of Ca in the sediments of the Nansi Lake was in the range of 2.00–15.43 %. It was observed that the Ca content of the sediments at sites 4–11 was higher than that of other sites. The average content of Ca of the sediments was the highest in the Dushan Lake. The limestone is widespread in the eastern mountain of the Dushan Lake, which might contribute to the high content of Ca in the sediments nearby the mountain (Lang 1983). Silt particles were found to be dominant in all of the samples (40.84–55.48 %), followed by sand (22.68–36.52 %) and clay (12.24–25.36 %). The average contents of clay and silt in the sediments were 19.82 and 52.09 %, respectively, had no big differences for all the sediment samples of the Nansi Lake, though it fluctuated at some sampling sites. In summary, the sediments of the Nansi Lake were generally alkaline and silt, with low level of carbonate and moderate levels of Fe and Al and high levels of SOM.
Contents of heavy metals in the sediments of the Nansi Lake
The contents of Cd, Cr, Cu, Pb, Zn, Ni, and Co in the surface sediments of the Nansi Lake were showed in Table 3. The average levels of Cd, Cr, Cu, Pb, Zn, Ni, and Co in the sediments of the upper lake including Nanyang Lake, Dushan Lake, and Zhaoyang Lake were 0.38, 94.73, 60.18, 46.17, 166.66, 26.89, and 11.07 mg/kg, respectively. Whereas the contents of these trace elements in the Weishan Lake were 0.15, 67, 38.99, 28.86, 119.58, 16.8, and 9.77 mg/kg, respectively. Therefore, the concentrations of heavy metals in the sediment samples of the upper lakes were generally higher than that of the Weishan Lake. In addition, from the results of this study, the average and median contents of heavy metals except Ni and Co were higher than those generally found in sediments globally; however, the minimum concentrations of Cu, Pb, and Zn in the sediments of the Nansi Lake were higher than the global average concentration (Table 3). This comparison may indicate that most of the sediment samples from the Nansi Lake are polluted heavily by these metals.
Industries such as manufacture of pharmacy, fertilizer, textile, mining and quarrying, power, smelting and pressing of ferrous metal, raw chemical material and chemical products, and printing developed extremely quickly in the Jining, Zaozhuang, and Heze cities, which might result in the emission of heavy metal pollutants. It was reported that 9.34 × 105, 7.41 × 105, and 1.98 × 105 t of untreated industrial wastewater were discharged into the Nansi Lake from Jining, Zaozhuang, and Heze cities annually, respectively (Zhang et al. 1999). A long-term monitor indicated that thirteen inflow rivers were all polluted by heavy metals due to various industries (Liu et al. 2007). Thus, industrial effluents were one important reason that resulted in the relatively high content of heavy metals in the sediments of the Nansi Lake, especially in the upper lake. In addition, the rapid development of fisheries and aquaculture also favored the accumulation of heavy metals such as Cr, Cu, and Pb in the sediments of the Nansi Lake. The elements Pb and Zn were generally regarded as the identified elements of traffic pollution (Paterson et al. 1999; Dai 2001). Diesel boats are among the main vehicles in the Nansi Lake, vehicle exhaust and leached diesel to water body contain some heavy metals. Therefore, traffic transportation might be a potential source of heavy metals in the study area. In addition, Pb, Zn, and their compounds might originate from domestic and industrial sewage from cities around the Nansi Lake and the heavy application of agricultural chemicals in the adjacent arable fields, as they are contained in different manufactured goods (e.g., paints, cosmetics, automobile tires, and batteries) and in agricultural fertilizers (Mico et al. 2006). The coal resources are very rich in the region around the Nansi Lake. There are some big coal-fired power plants such as Jining, Zoucheng, Jiaxiang, and Liyan power plants nearby the Nansi Lake, and their total installed capacity is more than 10 million kilowatts. The coal mining waste might be an important pollution source of heavy metals for the sediment of the Nansi Lake. In addition, water flows from the upper lake to the lower lake in the Nansi Lake, so the sediments of the upper lake are affected firstly by the effluents. After aggradation and decontamination in the upper lake, sediments in the lower lake are at a corresponding low-grade contamination level. Therefore, the heavy metal contents of the sediments in the upper lake are higher than that of the lower lake.
Partitioning of heavy metals in the sediments of Nansi Lake
The behaviors including chemical interaction, mobility, biological availability, and potential toxicity of heavy metals in sediments are strongly influenced by their chemical forms. It is important to identify and quantify their association with various geochemical phases in sediments to evaluate processes of downstream transport, deposition, and release under changing environmental conditions and gain a more precise understanding of the potential and actual impact of elevated levels of heavy metals in sediment (Singh et al. 2005). The fractional distribution of heavy metals in samples from the 20 sites was shown in Fig. 2.
Soluble and exchangeable fraction (EXC): The form of heavy metals had high immediate mobility and bioavailability. The proportion of heavy metals in these forms was generally less than 1 %, indicating the low immediate metal reservoir for aquatic organisms.
Bound to carbonate minerals (CARB): This proportion of heavy metals in this fraction, on average, displayed the following order: Cd (10.52 %) > Zn (9.42 %) > Pb (7.78 %) > Cu (6.51 %) > Co (5.32 %) > Ni (4.2 %). As the size of Cd ion is similar to that of the Ca ion, Cd is preferentially associated with calcite minerals (Zachara et al. 1991). The partitioning pattern found for Cd was not unusual; high percentages of total Cd have been found to be associated with carbonate phase by many studies (Han and Banin 1995; Singh et al. 2005; Nasrabadi et al. 2010; Wang et al. 2011).
Bound to easily reducible oxides (ERO): The average proportion of heavy metals in the ERO fraction (the amorphous Fe/Mn oxide phase) displayed the following order: Co (10.21 %) > Cu (9.25 %) > Zn (8.56 %) > Cr (7.23 %) > Pb (6.52 %) > Cd (5.84 %) > Ni (2.64 %).
Bound to organic matter (OM): The average proportion of heavy metals in the OM phase displayed the following order: Cu (18.03 %) > Zn (12.75 %) > Co (11.84 %) > Cd (10.02 %) > Ni (9.85 %) > Cr (8.25 %) > Pb (5.11 %).
Bound to reducible oxides (RO): The average proportion of heavy metals in the RO fraction (crystalline Fe/Mn oxides) displayed the following order: Cu (18.03 %) > Zn (12.11 %) > Pb (13.35 %) > Cr (10.35 %) > Ni (9.13 %) > Co (6.21 %) > Cd (3.72 %).
Residual fraction (RES): Metals found in the RES fraction are expected to be chemically stable and biologically inactive. Most of the metals, including Cr (68.77 %), Ni (73.36 %), Pb (67.0 %), Zn (56.32 %), Co (65.8 %), Cu (48.62 %), and Cd (69.05 %), were found in the residual fraction.
In summary, these results suggest that the average proportions of heavy metals in residual fraction ranged from 48.62 % for Cu to 77.76 % for Ni; these proportions are generally not susceptible to mobility and bioavailability even if fluvial conditions change. However, approximately 10.52 % of the Cd in the Nansi Lake sediments was bound to carbonate minerals; thus, the mobility and bioavailability of Cd in the sediments were higher in comparison to the other heavy metals.
Potential ecological risk assessment of heavy metals in the sediments of the Nansi Lake
The overall mean I geo for Cr, Ni, and Co was negative, showing that the mean levels of these elements in the Nansi Lake sediments are lower than the average shale concentrations (Table 4). For other elements including Cd, Cu, Pb, and Zn, the values of I geo were positive in some sampling sites, revealing that the mean concentrations of these heavy metals in the Nansi Lake sediments are higher than the average shale concentrations (Table 4). The maximal value of I geo was 1.76 for Cd at site 1, indicating that the sediment at the site 1 was moderately polluted by Cd.
To quantify potential anthropogenic impacts, REF values were calculated for each metal and site (Table 4). The average REF values of Cd, Cr, Cu, Pb, Zn, Ni, and Co were 4.44, 3.32, 2.16, 3.29, 3.95, 1.79, and 1.24, respectively. These values suggest significant enrichment of the heavy metals in the lake sediments.
The results of SPI and mPECQ are shown in Fig. 3. The highest SPI value was 4.99 at site 1, 2.04 at site 2, 2.93 at site 3, 2.44 at site 5, 2.10 at site 10, 3.06 at site 12. These values indicate that the sediments at these sites had low pollution levels. The mPECQ value ranged from 0.23 to 0.55 for all the sites. Therefore, heavy metals in the Nansi Lake sediments may have a moderate potential for adverse biological effects.
From the results mentioned above, the I geo, REF, SPI, and PECQ values were not in agreement with one another. The average I geo values were much higher for Pb and Cu, while average REF values were much higher for Cd and Zn (Table 4). The average I geo and REF values were not correlated. Because the REF takes sediment heterogeneity into account, it is expected to be more accurate than I geo for the contamination assessments of sediments at the watershed scale. In contrast, the average PECQ value was much higher for Cr and Ni than for other metals, suggesting that these two metals would cause higher adverse biological effects than other metals. The SPI values was significantly correlated to and mainly attributed to the Cd content in the sediments, because its toxicity weight of 300 was too substantially higher than that of the other metals. Therefore, whereas I geo and SPI showed that sediments at most sites were not contaminated by heavy metals. REF and mPECQ suggested that toxic trace elements were enriched in the Nansi Lake sediments and might have a moderated potential for adverse biological effects. In addition, it was found that the sediments of the Nansi Lake were formed by bedload from flooding of the Yellow River (Lang 1983). Considering the parameters in the evaluation methods in this study, the REF and PECQ are more suitable to show the contamination and biological effects of heavy metals in sediments at a watershed scale, respectively.
Conclusions
The average levels of Cd, Cr, Cu, Pb, Zn, Ni, and Co in the sediments of the Nansi Lake were 0.35, 53.33, 27.01, 25.52, 63.17, 25.38, and 10.88 mg/kg, respectively. The levels of heavy metals in the sediments of the upper lake were generally higher than that in the Weishan Lake. The average and median concentrations of Cd, Cr, Cu, Pb, and Zn in the sediments of the Nansi Lake were higher than that in sediments globally, while Ni and Co were lower. Industry, metallurgy, coal burning, and using of agricultural fertilizer and pesticide with heavy metals might favor the accumulation of heavy metals in the sediments in this region. In addition, the heavy metals were mostly bound in the residual fraction, ranging from 48.62 % for Cu to 73.76 % for Ni, indicating low mobility and bioavailability. Among these heavy metals, Cd was the most mobile metal because it presented the highest percentages bound to carbonate and the lowest percentage in the residual fraction. From the results of ecologic risk assessment, these heavy metals were minimal or moderate enrichment in the Nansi Lake sediments and might have a moderate potential for adverse biological effects. Average REF values of Cd and Zn and average PECQ values of Cr and Ni were higher among these heavy metals. Therefore, it is necessary to focus more attention on Cd, Cr, Zn, and Ni in the sediments in the future management and pollution control of the Nansi Lake.
References
Audry, S., Schafer, J., Blanc, G., & Jouanneau, J. M. (2004). Fifty-year sedimentary record of heavy metal pollution (Cd, Zn, Cu, Pb) in the Lot River reservoirs (France). Environmental Pollution, 132, 413–426.
Bao, S. D. (1999). In S. D. Bao, R. F. Jiang, & C. G. Yang (Eds.), Analysis of agri-chemistry in soil (3rd ed., pp. 34–35). Beijing: Chinese Agricultural Science and Technology Press. In Chinese.
Bowen, H. J. M. (1979). Environmental chemistry of the elements. London: Academic.
Bragato, C., Brix, H., & Malagoli, M. (2006). Accumulation of nutrients and heavy metals in Phragmites australis (Cav.) Trin. ex Steudel and Bolboschoenus maritimus (L.) Palla in a constructed wetland of the Venice lagoon watershed. Environmental Pollution, 144, 967–975.
Calmano, W., Wolfgang, A., & Förstner, U. (1990). Exchange of heavy metals between sediment components and water. In: Broekaert, J.A.C., Gucer, S., Adams, F. (Eds.) Metal speciation in the environment. NATO ASI Ser, Ser G 23, Berlin, pp 503–522.
Choi, M. S., Yi, H. I., Yang, S. Y., Lee, C. B., & Cha, H. J. (2007). Identification of Pb sources in Yellow Sea sediments using stable Pb isotope ratios. Marine Chemistry, 107, 255–274.
Dai, S. (2001). Environmental chemistry. Beijing: Beijing, Higher Education Press (In Chinese).
Dou, Y., Li, J., Zhao, J., Hu, B., & Yang, S. (2013). Distribution, enrichment and source of heavy metals in surface sediments of the eastern Beibu Bay, South China Sea. Marine Pollution Bulletin, 67, 137–145.
Fan, C., Zhu, Y., Ji, Z., Zhang, L., & Yang, L. (2002). Characteristics of the pollution of heavy metals in the sediments of Yilihe River, Taihu Basin. Journal of Lake Science, 14, 235–241 (In Chinese with English Abstract).
Förstner, U., & Wittmann, G. T. W. (1981). Metal pollution in the aquatic environment. Berlin: Springer. 486.
Förstner, U., Ahlf, W., Calmano, W., & Kersten, M. (1990). Sediment criteria development—contributions from environmental geochemistry to water quality management. In D. Heling, P. Rothe, U. Förstner, & P. Stoffers (Eds.), Sediments and environmental geochemistry: selected aspects and case studies (pp. 311–338). Berlin: Springer.
Gaur, V. K., Gupta, S. K., Pandey, S., Gopal, K., & Misra, V. (2005). Distribution of heavy metals in sediment and water of river Gomti. Environmental Monitoring and Assessment, 102, 419–433.
Han, F. X., & Banin, A. (1995). Selective sequential dissolution techniques for trace metals in arid-zone soils: the carbonate dissolution step. Communications in Soil Science and Plant Analysis, 26, 553–576.
Han, F. X., & Banin, A. (1999). Long-term transformations and redistribution of potentially toxic heavy metals in arid-zone soils: II, Under field capacity regime. Water, Air, and Soil Pollution, 114, 221–250.
Ingersoll, C. G., MacDonald, D. D., Wang, N., Crane, J. L., Field, L. J., Haverland, P. S., Kemble, N. E., Lindskoog, R. A., Severn, C., & Smorong, D. E. (2001). Predictions of sediment toxicity using consensus-based freshwater sediment quality guidelines. Archives of Environmental Contamination and Toxicology, 41, 8–21.
Iqbal, J., Tirmizi, S. A., & Shah, M. H. (2013). Statistical apportionment and risk assessment of selected metals in sediments from Rawal Lake (Pakistan). Environmental Monitoring and Assessment, 185, 729–743.
Jain, C. K., Singhal, D. C., & Sharma, M. K. (2005). Metal pollution assessment of sediment and water in the River Hindon, India. Environmental Monitoring and Assessment, 105, 193–207.
Jain, C. K., Gupta, H., & Chakrapani, G. J. (2008). Enrichment and fractionation of heavy metals in bed sediments of River Narmada, India. Environmental Monitoring and Assessment, 141, 35–47.
Kamala-Kannan, S., Prabhu Dass Batvari, B., Lee, K. J., Kannan, N., Krishnamoorthy, R., Shanthi, K., & Jayaprakash, M. (2008). Assessment of heavy metals (Cd, Cr and Pb) in water, sediment and seaweed (Ulva lactuca) in the Pulicat Lake, Southeast India. Chemosphere, 7, 1233–1240.
Kankilic, G. B., Tuzun, I., & Kadioglu, Y. K. (2013). Assessment of heavy metal levels in sediment samples of Kapulukaya Dam Lake (Kirikkale) and lower catchment area. Environmental Monitoring and Assessment, 185, 6739–6750.
Karageorgis, A. P., Sioulas, A. I., & Anagnostou, C. L. (2002). Use of surface sediments in Pagassitikos Gulf, Greece, to detect anthropogenic influence. Geo-Marine Letters, 21, 200–211.
Kukrer, S., Seker, S., Abaci, Z. T., & Kutlu, B. (2014). Ecological risk assessment of heavy metals in surface sediments of northern littoral zone of Lake Cildir, Ardahan, Turkey. Environmental Monitoring and Assessment, 186, 3847–3857.
Lang, L. R. (1983). Study on formation of Nansi Lake. Transactions of Oceanology and Limnology, 4, 31–38 (In Chinese with English Abstract).
Li, F., Huang, J. H., Zeng, G. M., Huang, X. L., Li, X. D., Liang, J., Wu, H. P., Wang, X. Y., & Bai, B. (2014). Integrated source apportionment, screening risk assessment, and risk mapping of heavy metals in surface sediments: a case study of the Dongting Lake, Middle China. Human and Ecological Risk Assessment, 20, 1213–1230.
Liang, L. N., He, B., Jiang, G. B., Chen, D. Y., & Yao, Z. W. (2004). Evaluation of mollusks as biomonitors to investigate heavy metal contaminations along the Chinese Bohai Sea. Science of the Total Environment, 324, 105–113.
Liu, E. F., Shen, J., Yang, L. Y., Zhu, Y. X., Sun, Q. Y., & Wang, J. J. (2007). Chemical fractionation and pollution characteristics of heavy metals in the sediment of Nansi Lake and its main inflow rivers, China. Environmental Science, 28, 1377–1383 (In Chinese with English Abstract).
Loeppert, R. H., & Suarez, D. L. (1996). Carbonate and gypsum. In D. L. Sparks et al. (Eds.), Methods of soil analysis, part 3. Chemical methods (pp. 437–474). Madison: SSSA and ASA.
Long, E. R., Inqersoll, C. G., & MacDonald, D. D. (2006). Calculation and uses of mean sediment quality guideline quotients: a critical review. Environmental Science & Technology, 40, 1726–1736.
Maxwell, R. M., & Kastenberg, W. E. (1999). A model for assessing and managing the risks of environmental lead emissions. Stochastic Environmental Research and Risk Assessment, 13, 232–250.
Mays, P., & Edwards, G. (2001). Comparison of heavy metal accumulation in a natural wetland and constructed wetlands receiving acid mine drainage. Ecological Engineering, 16, 487–500.
Mico, C., Recatala, L., Peris, M., & Sanchez, J. (2006). Assessing heavy metal sources in agricultural soils of an European Mediterranean area by multivariate analysis. Chemosphere, 65, 863–872.
Mil-Homens, M., Stevens, R. L., Cato, I., & Abrantes, F. (2007). Regional geochemical baselines for Portuguese shelf sediments. Environmental Pollution, 148, 418–427.
Mucha, A. P., Vasconcelos, M. T. S. D., & Bordalo, A. A. (2003). Macrobenthic community in the Douro Estuary: relations with trace metals and natural sediment characteristics. Environmental Pollution, 121, 169–180.
Müller, G. (1969). Index of geoaccumulation in sediments of the Rhine River. Geosciences Journal, 2, 108–118.
Nasrabadi, T., Bidhendi, G. N., Karbassi, A., & Mehrdadi, N. (2010). Partitioning of metals in sediments of the Haraz River (Southern Caspian Sea basin). Environmental Earth Sciences, 59, 1111–1117.
Ouyang, Y., Higman, J., Thompson, J., O’Toole, T., & Campbell, D. (2002). Characterization and spatial distribution of heavy metals in sediment from Cedar and Ortega rivers subbasin. Journal of Contaminant Hydrology, 54, 19–35.
Ozmen, H., Kulahci, F., Cukurovali, A., & Dogru, M. (2004). Concentrations of heavy metal and radioactivity in surface water and sediment of Hazar Lake (Elazig, Turkey). Chemosphere, 55, 401–408.
Pasternack, G. B., & Brown, K. J. (2006). Natural and anthropogenic geochemical signatures of floodplain and deltaic sedimentary strata, SacramentoeSan Joaquin Delta, California, USA. Environmental Pollution, 141, 295–309.
Paterson, E., Sanka, M., & Clark, L. (1999). Urban soil as pollutant sinks—a case study from Aberdeen, Scotland. Applied Geochemistry, 11, 129–131.
Qu, W. C., Dickman, M., & Wang, S. M. (2001). Mutivariate analysis of heavy metal and nutrient concentrations in sediments of Taihu Lake, China. Hydrobiological, 450, 83–89.
Rivail Da Silva, M., Lamotte, M., Donard, O. F. X., Soriano-Sierra, E. J., & Robert, M. (1996). Metal contamination in surface sediments of mangroves, lagoons and Southern Bay in Florianopolis Island. Environmental Technology, 17, 1035–1046.
Sadiq, R., Rodriguez, M. J., Imran, S. A., & Najjaran, H. (2007). Communicating human health risks associated with disinfection by-products in drinking water supplies: a fuzzy-based approach. Stochastic Environmental Research and Risk Assessment, 21, 341–353.
Sakan, S. M., Ðorđević, D. S., Manojlović, D. D., & Predrag, P. S. (2009). Assessment of heavy metal pollutants accumulation in the Tisza river sediments. Journal of Environmental Management, 90, 3382–3390.
Samecka-Cymerman, A., & Kempers, A. (2001). Concentrations of heavy metals and plant nutrients in water, sediments and aquatic macrophytes of anthropogenic lakes (former open cut brown coal mines) differing in stage of acidification. Science of the Total Environment, 281, 87–98.
Selvam, A. P., Priya, S. L., Banerjee, K., Hariharan, G., Purvaja, R., & Ramesh, R. (2012). Heavy metal assessment using geochemical and statistical tools in the surface sediments of Vembanad Lake, Southwest Coast of India. Environmental Monitoring and Assessment, 184, 5899–5915.
Silva, M. A. L., & Rezende, C. E. (2002). Behavior of selected micro and trace elements and organic matter in sediments of freshwater system in south-east Brazil. Science of the Total Environment, 292, 121–128.
Singh, M., Müller, G., & Singh, I. B. (2002). Heavy metals in freshly deposited stream sediments of rivers associated with urbanization of the Ganga Plain, India. Water, Air, and Soil Pollution, 141, 35–54.
Singh, M., Müller, G., & Singh, I. B. (2003). Geogenic distribution and baseline concentration of heavy metals in sediments of the Ganges River, India. Journal of Geochemical Exploration, 80, 1–17.
Singh, K. P., Mohan, D., Singh, A. K., & Malik, A. (2005). Studies on distribution and fractionation of heavy metals in Gomti river sediments—a tributary of the Ganges, India. Journal of Hydrology, 312, 14–27.
Suresh, G., Sutharsan, P., Ramasamy, V., & Venkatachalapathy, R. (2012). Assessment of spatial distribution and potential ecological risk of the heavy metals in relation to granulometric contents of Veeranam lake sediments, India. Ecotoxicology and Environmental Safety, 84, 117–124.
Sutherland, R. A. (2000). Bed sediment-associated trace metals in an urban stream, Oahu, Hawaii. Environmental Geology, 39, 611–627.
Swarnalatha, K., Letha, J., & Ayoob, S. (2014). Effect of seasonal variations on the surface sediment heavy metal enrichment of a lake in South India. Environmental Monitoring and Assessment, 186, 4153–4168.
Tessier, A., & Campbell, P. G. C. (1987). Partitioning of trace metals in sediments: Relationship with bioavailability. Hydrobiologia, 149, 43–52.
Tessier, A., Campbell, P. G. C., & Bisson, M. (1979). Sequential extraction procedure for the speciation of particulate trace metals. Analytical Chemistry, 51, 844–851.
Turekian, K. K., & Wedepohl, K. H. (1961). Distribution of the elements in some major units of the earth’s crust. Geological Society of America Bulletin, 72, 175–192.
Ure, A., Quevaullier, P., Muntau, H., & Griepink, B. (1993). Speciation of heavy metals in soils and sediments: an account of the improvement and harmonization of extraction techniques undertaken under the auspices of the BCR of the Commission of the European Communities. International Journal of Environmental Analytical Chemistry, 51, 135–151.
Varol, M. (2011). Assessment of heavy metal contamination in sediments of the Tigris River (Turkey) using pollution indices and multivariate statistical techniques. Journal of Hazardous Materials, 195, 355–364.
Wang, S. L., Lin, C., & Cao, X. Z. (2011). Heavy metals content and distribution in the surface sediments of the Guangzhou section of the Pearl River, Southern China. Environmental Earth Sciences, 64, 1593–1605.
Wedepohl, K. H. (1995). The composition of the continental crust. Geochimica et Cosmochima Acta, 59, 1217–1239.
Yao, Q. Z., Zhang, J., Wu, Y., & Xiong, H. (2007). Hydrochemical processes controlling arsenic and selenium in the Changjiang River (Yangtze River) system. Science of the Total Environment, 377, 93–104.
Yuan, H., Song, J., Li, X., Li, N., & Duan, L. (2012). Distribution and contamination of heavy metals in surface sediments of the South Yellow Sea. Marine Pollution Bulletin, 64, 2151–2159.
Zachara, J. M., Cowan, C. E., & Resch, C. T. (1991). Sorption of divalent metals on calcite. Geochimica et Cosmochimica Acta, 55, 1549–1562.
Zhang, Z. L., Sun, Q. Y., Peng, L. M., Niu, Z. G., & Wu, A. M. (1999). Water environment problems in the Nansi Lake area. Journal of Lake Science, 11, 86–90 (In Chinese with English Abstract).
Zhang, M., Cui, L., Sheng, L., & Wang, Y. (2009). Distribution and enrichment of heavy metals among sediments, water body and plants in Hengshuihu Wetland of Northern China. Ecological Engineering, 35, 563–569.
Acknowledgments
This study is supported by the Encouraging Foundation for Outstanding Youth Scientists of Shandong Province (no. BS2012HZ006), the National Nature Science Foundation of China (41301532), the First Class General Financial Grant from the China Postdoctoral Science Foundation (2013M540103), the Financial Grant from the State Environmental Protection Key Laboratory of Microorganism Application and Risk Control of China (MARC2012D005), and the National Key Research Project on Water Environmental Pollution Control in China (2012ZX07301-001).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Xuezhi Cao and Yan Shao contributed equally to this work.
Rights and permissions
About this article
Cite this article
Cao, X., Shao, Y., Deng, W. et al. Spatial distribution and potential ecologic risk assessment of heavy metals in the sediments of the Nansi Lake in China. Environ Monit Assess 186, 8845–8856 (2014). https://doi.org/10.1007/s10661-014-4048-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10661-014-4048-3