Abstract
The geochemical distribution and enrichment of ten heavy metals in the surface sediments of Vembanad Lake, southwest coast of India was evaluated. Sediment samples from 47 stations in the Lake were collected during dry and wet seasons in 2008 and examined for heavy metal content (Al, Fe, Mn, Cr, Zn, Ni, Pb, Cu, Co, Cd), organic carbon, and sediment texture. Statistically significant spatial variation was observed among all sediment variables, but negligible significant seasonal variation was observed. Correlation analysis showed that the metal content of sediments was mainly regulated by organic carbon, Fe oxy-hydroxides, and grain size. Principal component analysis was used to reduce the 14 sediment variables into three factors that reveal distinct origins or accumulation mechanisms controlling the chemical composition in the study area. Pollution intensity of the Vembanad Lake was measured using the enrichment factor and the pollution load index. Severe and moderately severe enrichment of Cd and Zn in the north estuary with minor enrichment of Pb and Cr were observed, which reflects the intensity of the anthropogenic inputs related to industrial discharge into this system. The results of pollution load index reveal that the sediment was heavily polluted in northern arm and moderately polluted in the extreme end and port region of the southern arm of the lake. A comparison with sediment quality guideline quotient was also made, indicating that there may be some ecotoxicological risk to benthic organisms in these sediments.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Estuaries are unique systems among the most productive marine ecosystems in the world and receive inputs of pollutants, as they are often situated in the vicinities of highly populated and industrialized areas (Liu et al. 2003). They are extremely important biogeochemical zones, with the capability of altering the flux of materials between the land via rivers to coastal zones and ultimately to the oceans. Heavy metals enter these aquatic environments mainly through natural sources such as weathering of rocks and soil in the catchment and anthropogenic sources like agricultural, municipal, domestic, and industrial wastes (Demirak et al. 2006; Cevik et al. 2009). Heavy metals discharged into these systems, get integrated and amplified by the sediments. Flocculation, adsorption onto inorganic–organic particulates, and subsequent sedimentation are the main processes responsible for the trapping and accumulation of metals in estuarine systems (Liu et al. 2009). Immobilization by sedimentation dominates than diagenitic processes for most of the metal pollutants, thus resulting in elevated level of metals in bottom sediment (Simeonov et al. 2000) and provides a record of catchment inputs into aquatic ecosystems (DelValls et al. 1998; Jonathan et al. 2004). Sediment-associated metals have the potential to be ecotoxic due to their mobility and bioavailability, and this in turn affects both ecosystems and human life through a process of bioaccumulation and biomagnification, respectively (Buccolieri et al. 2006; Ip et al. 2007). Thus, the knowledge on the metal concentrations in sediment is always considered to be a vital face of aquatic environmental assessment studies (Jain et al. 2005; Karbassi et al. 2008), because it provides data for planners and decision makers for its proper management.
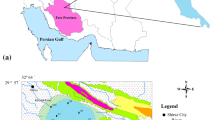
Vembanad Lake (Lat. 9° 30′–10° 10′ N and Long. 76° 10′–76° 25′ E), the largest brackish water system in the southwest coast of India, consist of mangrove patches, water fowl habitats, and supports an unique assemblage of marine, brackish water, and freshwater species (Menon et al. 2000). Due to the international significance of this wetland, it was designated as a Ramsar Site (No. 1214) at the Convention on Wetlands organized by the UNESCO in the Iranian city of Ramsar in 1981. The main socio-economic activities developed in this Lake are fishing, agriculture, aquaculture, coir making, and clam fishing besides the traditional ferry and transport services. This system is particularly vulnerable to input changes due to limited water exchange, and the pollution scenarios are quite confusing (Nair 2002) because of the complicated interactions in this ecosystem. Thus, an advanced method is required to describe the system processes by differentiating natural from anthropogenic impacts. A combination of both geochemical normalization and multivariate statistical methods seem to be a better option (Alkarkhi et al. 2008; Idris 2008) to understand the natural and anthropogenic inputs in the complex estuarine system and the factors responsible for the geochemical distribution. In the present study, the extent of metal contamination was assessed using the pollution load index, enrichment factor, and toxicity by mean sediment quality guideline quotient.
Although many studies have been conducted on heavy metals distribution in the present system (Padmalal and Seralathan 1995; Padmalal et al. 1997; Shajan 2001; Balachandran et al. 2005, 2006; Renjith and Chandramohanakumar 2009; Harikumar et al. 2009; Ratheesh Kumar et al. 2010), most of these studies have focused only on particular zones of the Vembanad Lake. Lack of comprehensive studies in heavy metal behavior of this lake posed restrictions in understanding the overall geochemical characteristics of the lake. This is a comprehensive study covering the entire Vembanad Lake system and its continuum by sampling in 47 stations. The primary objective of this study is to assess the spatial and seasonal distribution pattern of heavy metals in this lake to get a basic idea about the geochemical factors governing the accumulation of heavy metals. It also describes the use of geochemical and multivariate statistical technique for identifying the sources and differentiating the anthropogenic versus natural contribution of metals in sediments. This study also aims to obtain a good evaluation of overall pollution status of sediments in Vemband Lake using Pollution Load index and with Sediment Quality Guidelines for a potential level of biological risk.
Materials and methods
Study area
Vembanad Lake is a complex bar built micro-tidal (<1 m) estuary (Fig. 1), and it occupies an area of 241 km2. It is characterized by an ox-bow shape (Soman 1997) with mud flats, tidal marshes, islands, and a network of canals of width 0.1–6 km and bathymetry of depth varying between 2 and 7 m. The two ship channels at the Cochin harbor region, from bar mouth towards the southern arm, are dredged and maintained at 10–13 m depth (Qasim 2003). This estuary is unique due to its distinct topography and circulation pattern and consists of two arms. Northern arm (30 km) extending from Cochin bar mouth (450 m wide) to Azheekode bar mouth (250 m wide) is generally narrow and shallow. The southern arm (62 km) on the other hand is wider and deeper extending from the Cochin bar mouth to Alappuzha, which is further divided by the construction of the salinity barrier (Thaneermukkom Bund, constructed in 1976) to brackish water downstream and freshwater upstream (Gopalan et al. 1983). The rivers Achankoil, Pampa, Manimala, Meenachil, Muvattupuzha, and Periyar along with large number of canals debouch at different parts of this lake, discharging about 2 × 1010 m3 year−1 of fresh water (Srinivas et al. 2003) and the volume of river inflow to the inner zone of the lake reflects a significant seasonal variation. The hydrological characteristics of the lake is governed by the monsoon regime which results in the variability of estuarine environmental characteristics, including flow, salinity, etc. About 71% of the annual rainfall is contributed by monsoon (Jayaprakash 2002), and the rivers drain major amount of sediment load during the monsoon season (Chandramohan and Balchand 2007). The sediment flux from catchments to this system is about 32 × 106 t y−1 (Thomson 2002). Improper management in the past several decades has resulted in the 40% shrinkage of the Vembanad Lake (Qasim 2003). Rapid industrialization and urbanization has resulted in the discharge of about 1.04 × 105 m3 day−1 of effluents, 260 m3 day−1 of sewage and unspecifiable quantities of wastes from agricultural and aquaculture farms into this system (Balachandran et al. 2005).
Sampling and analytical methods
Seasonal sampling was carried out in 47 locations of the entire Vembanad Lake during April (dry season) and September (wet season) of 2008 (Fig. 1). Among these, 13 samples (stations 1–13) were collected from the freshwater upstream of the southern arm, which is affected by agricultural inputs, tourism activities, domestic sewage, and the four southern rivers. Twenty-one samples collected from the brackish water downstream of the southern arm, representing the influence of domestic settlement, Muvattupuzha River (14–27) and port and shipyard activities near the bar mouth region (28–34). Thirteen samples (stations 35–47) were collected from the northern arm indicating the industrial footprint from the industrial zone, null zone (midway between Cochin and Azheekode bar mouth), Periyar River and sewage discharge. Domestic sewage discharge from Cochin (a city with a population of nearly 1.5 million) into both the northern arm and downstream of the southern arm is also a critical concern.
In situ measurements of pH, salinity, and dissolved oxygen (DO) in the surface water were measured using a multiprobe (TOA–DKK, WQC-24), after calibration with appropriate solutions. Surface sediment samples (0–5 cm) were collected using Van-Veen Grab (0.042 m2), sampled from the middle portion using a teflon-coated spatula and packed in air-tight pre-labeled polyethylene bags and kept frozen until analysis. The sediment samples were oven dried at 50°C and divided into two fractions, one portion for the analysis of sediment texture parameters (sand, silt, clay) and the other portion was powdered using an agate mortar and pestle, sieved through 63 μ sieve and analyzed for organic carbon and heavy metals. All sediment samples were analyzed in duplicate, and all concentrations reported in this work are the average on dry weight basis.
Granulometric analysis was carried out using standard sieve and pipette techniques after inorganic and organic carbon removal (Folk 1974). The inorganic carbon is removed by adding 10% HCl to the sediment and slowly heated until effervescence ceased and then washed with distilled water, and the solution was decanted (Vaasma 2008). Next, 30% H2O2 was added to oxidize and remove the organic carbon. Organic carbon was determined using wet oxidation method (Gaudette et al. 1974), and the accuracy of this titration is ≤5%. For total heavy metal analysis, a known quantity of sediments (0.5 g) was digested in a mixture of HF–HClO4–HNO3 (Loring and Rantala 1992). The complete digestion was confirmed by repeating the acidification until a clear solution was obtained and brought into solution in 0.1 M HCl. After calibration with suitable EMerck elemental standards, samples were analyzed in Flame AAS (Perkin Elmer AA 800) equipped with deuterium background corrector. Graphite furnace method was used for Cd analysis. The accuracy and precision of the analytical methodology was assessed by triplicate analyses of certified reference materials, Sediment T (National Research Council, Canada). Precision was generally good (<4%), and good to excellent recoveries ranging from 87% to 114% were obtained for the analyzed heavy metals (Cd = 114%; Co = 87%; Cu = 93%; Pb = 110%; Ni = 93%; Zn = 98%; Cr = 109%; Mn = 103%; Fe = 98%; Al = 105%) indicating the accuracy of the methodology.
Statistical methods
Statistical analysis was performed using Statistical Program for Social Sciences (SPSS version 13.0). Two-way analysis of variance (ANOVA) without replication was carried out to find out the spatial and seasonal variations of sediment parameters in the study area. Pearson correlation analysis was performed to identify inter-elemental relationship with their sediment properties. Prior to further statistical analysis, the data were Z-score normalized to create uniformity in the units of variables (Shaw 2003). The principal component analytical (PCA) method, which is widely used to detect the hidden structure of sediment sources and to distinguish natural and anthropogenic inputs, was applied here to explore the origin and geochemical factors influencing their distribution (Loska and Wiechula 2003). Analytical results were elaborated by using Geographical Information System (GIS) Arch-GIS 9.3 software to show geochemical indices.
Results and discussion
Basic hydrographic and sediment characteristics
The basic parameters of surface water, texture, organic carbon, and heavy metal concentrations of surface sediment of the Vembanad Lake for both the seasons are presented in Table 1. The spatial variations of salinity during both the dry and wet seasons were quite significant. Bar mouth regions showed higher salinity during dry season (23.6–28.4) when compared to the wet season (5.4–8.3). Salinity gradually decreases towards the southern arm (0.1) during both the seasons. In the northern arm, salinity decreases until the null zone (station 43) and then gradually increases when it approaches near the Azheekode bar mouth. Salinity variation with near fresh water conditions during wet season under the profound influence of monsoon and gradual transformation into brackish condition during dry season was quite apparent. The pH values varied (6.3–8.3), with a high average value observed during dry season. pH (<7) was recorded in the fresh water upstream with a steady transformation to the alkaline (≥8.0) approaching the bar mouth, due to mixing of seawater. DO ranged between (4.3–8.6 mg L−1), with lower values at northern arm stations. Surface water components like salinity, DO, and pH showed significant spatial variation (p < 0.05), whereas salinity and pH showed very significant seasonal variation (p < 0.001) (Table 1). It is established that hydrodynamic conditions in this system mainly depend on the intrusion of seawater associated with tides, influx of fresh water from rivers, and precipitation processes (Joseph 2002).
Grain size generally increased from the inner areas towards the bar mouth and also at the mouths of the rivers Muvattupuzha and Periyar. Spatial distribution of grain size in the study area for both seasons are shown in Fig. 2 and Table 1. The sand content varied widely from 1.7% to 62.5% in the freshwater upstream, 5.3% to 83.2% in the southern arm, and 5.4% to 92.6% in the northern arm, respectively. Silt showed distribution pattern with average values (51.4%, 36.5%, 33.7%) and clay (21.2%, 23.1%, 19.3%) in freshwater upstream, southern arm downstream, and northern arm, respectively. Sediments showed variable textural facies mainly sandy mud, muddy sand, sandy silt, mud, and silt. The sediments were mainly dominated by mud (silt + clay), except at stations near the river mouths and the bar mouth. Very significant spatial variation (ANOVA, p < 0.001) was noted among sand, silt, and clay (Table 1). The uneven admixture of sand, silt, and clay fractions among the stations from the study area reflect variable transportation and accumulation patterns existing across the channels of the lake. Sand was found to be high during monsoon among stations (20–25, 40) due to the high energy condition that exist in these river mouth regions. Among sediment texture, sand and silt shows high values during wet season than dry season. Laluraj et al. (2008) also found the accumulation of alluvium as bed sediment in southern arm during monsoon, and the sediment transport in this system is dominant through suspension. No significant seasonal variation was found among sand and silt, whereas clay shows significant (p < 0.05) seasonal variation (Table 1). Organic carbon (OC) distributions in surface sediments are shown in the (Fig. 2). Higher values of OC (3.0–4.3%) were mainly observed on the sites in southern fresh water zone. The distribution of OC is mainly dependent on the sediment grain size due to the higher surface area of finer fractions (Rodríguez-Barroso et al. 2010). Organic carbon showed significant spatial variation and no significant seasonal variation. Similar distributional characteristics of OC with finer sediments indicate that dominant hydrodynamic processes are the main reason for the concentration of organic matter in the surface sediments (Valdes et al. 2005).
Spatial distribution of grain size, organic carbon, and metal content in the lake sediment for dry and wet seasons. The solid line (metals) represents the shale value (Turekian and Wedepohl 1961) of the metals
Distribution of heavy metals
The pattern of spatial distribution of all heavy metals measured in this study was for both dry and wet seasons as shown in Fig. 2 and Table 1. The distribution pattern of heavy metals in the Vembanad Lake was in the following sequential order: Al>Fe>Mn>Zn>Cr>Ni>Pb>Cu>Co>Cd which is similar to the world average shale (Turekian and Wedepohl 1961) distribution with the exception of Pb being higher than Cu. Lowest concentration of the heavy metals were observed in station 34 (Cochin bar mouth). Al 11.6% and Fe 6.8% showed higher concentration in the southern freshwater upstream than the southern arm downstream (7.7% and 5.0%, respectively) and northern arm (8.9% and 5.0%, respectively). Mn and Co also showed similar pattern like the major elements with average values of 636, 436, 325 μg g−1 for Mn and 27, 17, 15 μg g−1 for Co, in freshwater upstream, southern arm downstream, and northern arm, respectively. Pb concentrations showed no apparent spatial variability; with almost all stations showing levels higher than shale values. Cu and Ni showed similar distribution pattern with not much variation among the three regions of the system. Distribution pattern of Cr shows higher values in the northern arm and near the port region of southern arm compared with other regions. Both Zn (449, 92, 90 μg g−1) and Cd (4.7, 0.7, 0.3 μg g−1) were distributed in the northern arm, southern arm downstream, and freshwater upstream, respectively. Zn and Cd from the northern arm of the Vembanad Lake are enriched than the values recorded in the southern arm, which are mainly from anthropogenic sources. The sediment heavy metal concentrations found in this study were of the same order of magnitude as metal concentrations reported by different authors in sediments of the Cochin estuary (Padmalal and Seralathan 1995; Padmalal et al. 1997; Shajan 2001; Balachandran et al. 2005, 2006; Renjith and Chandramohanakumar 2009; Harikumar et al. 2009). Apart from Cd, Zn, and Pb, all other heavy metals showed highest concentrations in fine-grained sediment regions, and lower concentrations were found in the sandy sediments, suggesting the dependence on texture. Salinity and pH gradient in the downstream of both the arms near the bar mouth also favors the association of metals with fine particles through flocculation and coagulation (Bouezmarni and Wollast 2005). The distribution of all heavy metals in the study area indicate that higher concentration (especially in stations 6–11, 30–42) is due to the anthropogenic activities and recent development of major industries and harbor activities, along with the intense movement of boats in the Vembanad Lake. All heavy metal (Al, Fe, Mn, Cr, Zn, Ni, Pb, Cu, Co, Cd) components showed very significant spatial variation (p < 0.001) (Table 1) and almost similar average concentration during both dry and wet season. There is no significant seasonal variation among most of the heavy metals except in the case of Pb, which shows a significant seasonal variation (p < 0.05) (Table 1). Heavy metals along with sediment texture displayed more significant spatial variations than seasonal variations. It was found that there is not much significant seasonal variation among the sediment geochemical characters, thus further statistical analysis has been done on the combined dataset for both dry and wet seasons.
Correlation analysis
Correlation matrix (Table 2) shows that Cd and Zn are not controlled by any size fraction, whereas all other heavy metals showed negative correlation with sand indicating the adsorption of heavy metals on the finer sediments as observed by Padmalal et al. (1997). Heavy metals like Cu, Ni, Cr, and Fe depicts positive correlation with both silt and clay fraction; whereas, Co, Mn, and Al shows good association with silt. The good correlation of OC with metal reveals the formation of organic complexes with heavy metals as a ligand by flocculation and subsequently influences their distributions, due to its high specific surface area (Marchand et al. 2006; Zourarah et al. 2008). Fe exhibits significant association with heavy metals Cu, Ni, Cr, Co, Mn, and Pb along with OC, which reveals its key control over the linkage of these metals with organic matrix by association as Fe-oxy-hydroxides (Rubio et al. 2000). Balachandran et al. 2005 also reported the coagulation–flocculation of metals as colloids with hydrous iron oxide in Vembanad Lake. The good association of Co with Mn and Fe (0.79, 0.77) suggest the role of both Fe and Mn-oxy-hydroxides as host phase for this metal. Significant correlation exists between Cu, Cr, Ni indicates their similarity, and identical behavior. High-significant correlations of Cd with Zn alone, indicates same point source for these two metals as influenced by the anthropogenic processes. Hence, the sediment geochemistry in this system could be deduced as a strong association of heavy metals (HM) with three matrices (HM–HM, HM–OC, and HM–silt). The relative affinity of the carrier phases with the metals except for anthropogenic influenced Cd and Zn are in the order of Fe>OC>silt>clay, which indicates that OC and Fe hydroxides are the main geochemical carriers of metals in this regions
Principal component analysis
The factor loading matrix accounted by using 14 sediment variables is listed in Table 3. The result of PCA ordination using sediment quality characteristics gives three factors (based on scree plot) explained by 79.7% of the total variance. The strong loading of silt, clay, OC along with Fe, Cu, Cr, Ni, Al in the first component can be interpreted in terms of association of metals as a single source with fine sediments and OC. This factor clearly illustrates the granulometric dependence of these metals with dominant controlling factors by OC and Fe oxy-hydroxides. In the second component high loading of Mn, Co with moderate loading of Fe and Al are associated with geogenic mineral component of the sediment. The good association of Co with Mn and the poor loading with other metals in the second component suggest the role of Mn-oxide as a minor host phase for these heavy metals (Alagarsamy 2006). High loading of Cd and Zn is in the third component, along with moderate loading of Pb points towards purely anthropogenic factors for these metals. The third factor arises mainly from different industrial and domestic effluents, port activities, sewage, and non-associated with the detrital component of sediments. This component includes both point (Cd, Zn) and non-point (Pb) anthropogenic sources. Factor 1 describes that the accumulation pattern of metals is natural source, dependent on OC-granulometry-Fe oxy-hydroxides. Factor 2 is purely natural origin, which includes metals associated with sediment geogenic component, whereas factor 3 comprises metals purely from anthropogenic sources like various industrial and urban activities. Thus, the concentrations and distributions of metals between different sites could be explained using chemical and textural compositions.
Enrichment factor
The large granulometric spectrum and variation in metal deposition with particle size makes it difficult to compare the metal deposits occurring in different regions of the Vembanad Lake. In geochemical studies, normalizing metals relative to Al or Fe is widely used to compensate for both the granulometric and mineralogical variability of metal concentrations in sediment (Chapman and Wang 2001). Statistical methods can also be used to determine the reference element as metal concentration normalizer (Liu et al. 2003). From the Fig. 3, the linear relationship strength of metals with Al and Fe confirms the applicability of Fe as the most appropriate normalizer element in this study area. Several authors have used Fe to normalize heavy metal contaminant in estuarine sediments (Schiff and Weisberg 1999; Neto et al. 2000). EF for each element was calculated using the formula \( {\text{EF}} = \frac{{({M_{\text{s}}}/{\text{F}}{{\text{e}}_{\text{s}}})}}{{({M_{\text{b}}}/{\text{F}}{{\text{e}}_{\text{b}}})}} \) Where M s is the content of the metal in the sample, M b is the world shale average of the metal (Turekian and Wedepohl 1961), Fes is the content of Fe in sample, and Feb is the world shale average of Fe. Shale averages as background level were employed here owing to the difficulty in obtaining a pristine environment value in the study region, due to the complicated sediment texture character and diverse anthropogenic stresses that are observed throughout the Vembanad Lake.
The EF values were plotted using Box whisker plot (Fig. 4) and the EF values ranges for metals were as follows: Cd (0.2–37.1), Zn (0.6–10.0), Pb (0.8–4.6), Cr (0.6–1.8), Co (0.6–1.4), Cu (0.4–1.0), Ni (0.4–0.9), and Mn (0.2–0.8). Following the interpretation of (Birch 2003) Cu, Ni, and Mn shows no enrichment, although very small percentile of Co shows minor enrichment. EFs (<1) for these metals in the sediment indicate its origin predominantly from lithogenous material and suggest the absence of contamination by these metals in the study region. Almost full percentile of Pb and half percentile of Cr in the minor enrichment region indicate the anthropogenic input of these metals. Half percentile of Zn values shows minor enrichment, but in few stations towards the north Zn values, it showed moderate to moderately severe enrichment (EF > 5) which is depicted from the long whisker plot. Cd, which showed minor to moderate enrichment values all through the study area with a long whisker (EF > 25), indicates severe to very severe enrichment in northern stations. The observations suggest that the lake sediment is polluted by Cd, Zn, Pb, and Cr and acts as a sink for heavy metals contributed from a multitude of anthropogenic sources. The distribution maps of the EF values for Zn, Cd, Pb, and Cr reflect the status of the anthropogenic influences is shown in Fig. 5. High EF values of Zn and Cd in the north possibly displays the effluent discharge of nearby chemical industries (fertilizers, heavy metal processing, pesticides, insecticides, petrol refining, chemical and allied industries) and urban activities through Periyar river. In the northern estuary industrial effluents, flow pattern of Periyar river and synchronous tides entering through two barmouths (Cochin and Azheekode) lead to high deposition of Cd and Zn in points (32–42), whereas agricultural and domestic activities influence major portion in the southern part of the estuary makes less pollution compared with the northern arm. The weak flow from the north eastern estuary (Periyar River) towards further north and the dominant effect of tide entering through Azheekode bar mouth leads to less pollution in stations (44–47), and intersystem gradient in northern arm. The higher values of Pb and its similar distribution all through the Vembanad Lake could be from nonpoint sources. Pb content most probably comes from boat exhaust, paints, and indirect sources of atmospheric deposition generated from automobile exhaust emission (Gajghate and Bhanarkar 2005), which is further transferred to the Vembanad Lake by rain, road runoff, and storm water drains. Cr which is mainly concentrated in the northern arm and near port region possibly comes from municipal waste and leather product industrial effluents.
Pollution load index
The assessment of the contamination extent by metals in sediments was calculated using the Pollution Load Index (PLI) (Tomlinson et al. 1980) with the heavy metal data and world shale average values (Turekian and Wedepohl 1961) of the metals (Badr et al. 2009; Ray et al. 2006). It provides a summative indication of the overall level of heavy metal pollution in a particular sample. PLI was calculated for the area under investigation for only eight of the ten studied metals, considering the least toxicity by most abundant metals (Fe, Al).
PLI can be evaluated using the equation \( {\text{PLI}} = \sqrt[n]{{({\text{C}}{{\text{F}}_{{1}}}{ } \times {\text{ C}}{{\text{F}}_{{2}}}{ } \times ........ \times {\text{ C}}{{\text{F}}_n})}} \)
- CF:
-
contamination factor
- n :
-
number of metals
- contamination factor:
-
metal concentration in sediment/shale value of the metal.
The values of PLI recorded for all the sites ranged from 0.4 to 2.2. The lowest PLI values were found in stations ( 17, 18, 20, 21, 22, 34, 46, 47), and the highest values were recorded among stations (32, 35, 36, 37, 40, 41, 42). The spatial distribution of PLI along the Lake is shown in the (Fig. 6). PLI values were <1 for stations 1–22 with few exceptional stations (6, 7, 8, 9, 10) in the freshwater upstream. The high values in the fresh water upstream indicate that the accumulation of metals in these regions were from agricultural and domestic discharges, which build up due to the blockage of tidal flushing by salinity barrier. PLI values from station No. 23 is >1, and it progressively increased to a value >2 for several sites beyond station 32. The PLI reveals the alarming status of the sites on the northern, central part of the lake compared to the less polluted southern region apart from the stations closer to Alappuzha (Fig. 6).
Toxicological significance
The results were compared with universal guidelines on sediment toxicity limits by international environmental authority, considering the dependence of the biotic resource of this system. Sediment Quality Guidelines (SQG) is an informal tool to evaluate and categorize the relative quality of sediments (Long and Macdonald 1998) and also make an initial assessment of sediment toxicity in the absence of direct biological effects data (Birch and Taylor 2002). Mean SQG quotients were determined as the average of the ratios between the chemical concentrations in the samples and the respective effective range median values (ERM), so as to account for the actual concentration of individual metals as well as their combinations (Carr et al. 1996; Long et al. 1998).
Where C i is the sediment concentration of compound i,
- ERMi :
-
is the effect range median for compound i and
- n :
-
is the number of compound i.
The spatial distribution of mean ERM quotient of the sediment samples are shown in Fig. 7. The mean ERM quotient of <0.1 has a 12% probability of being toxic; a mean ERM quotient of 0.11–0.5 has a 30% probability of toxicity; a mean ERM quotient of 0.51–1.5 has a 40% probability of being toxic and a mean ERM quotient of >1.5 has a 74% probability of toxicity (Long et al. 2000; Essien et al. 2009). According to this classification, 85% of the sediment samples studied can be classified as “medium–low priority” sites with 30% probability of toxicity and 15% of the sediment samples sites falling in the medium-high priority sites with 40% probable toxicity. Continual pollution for the past few decades has upset the ecological balance by affecting the species composition and diversity of benthic organisms in this area. Clear evidence of the disappearance of benthic population and the presence of tolerant species from the medium-high priority sites are reported by Martin et al. 2010.
Conclusions
A detailed survey of the sediment quality was evaluated using geochemical and statistical tools to assess the present status of this Ramsar site. High concentrations of heavy metals were observed in the northern part of the Vembanad Lake compared to the less polluted southern part. The geochemical distribution of heavy metals is mainly controlled by the association of Fe, OC, and fine sediments. The extraction of three latent factors using PCA offers a specific interpretation of the major determining source factors in the lake metal geochemistry. Factor analysis denotes that in first two components, OC with sediment texture and geogenic compositions were the different dominant controlling factors of metal concentrations in the surface sediments, and the third component clearly spot the anthropogenic influence. High enrichment factor values in northern arm and spatial distribution of heavy metals in the sediments suggested that the Cd and Zn mainly originated from point sources, while Pb probably came from non-point sources in the Vembanad Lake. On the basis of PLI, northern arm has been identified as critical point of contamination and the lake is receiving pollution stress from all directions. Hence, attention should therefore be drawn to fast developmental activities and habitation in and around the estuarine area. Mean ERM quotient indicates some ecotoxicological risk for benthic organisms and further validation with toxicological work will provide information for the effective management of the lake. It is essential that further studies on the metal fractionation in sediments should be carried out in the near future to identify more vital information regarding bioavailability of metals and their potential toxicity. Proper management is needed to avoid the spreading of contaminants within and outside the Vembanad Lake, by adopting stringent measures to control before any catastrophic event affects this area.
References
Alagarsamy, R. (2006). Distribution and seasonal variation of trace metals in surface sediments of the Mandovi Estuary, West Coast of India. Estuarine, Coastal and Shelf Science, 67(1–2), 333–339.
Alkarkhi, A. F. M., Anees, A., Ismail, N., & Easa, A. M. (2008). Multivariate analysis of heavy metals concentrations in river estuary. Environmental Monitoring and Assessment, 143, 179–186.
Badr, N. B. E., El-Fiky, A. A., Mostafa, A. R., & Al-Mur, B. A. (2009). Metal pollution records in core sediments of some Red Sea coastal areas, Kingdom of Saudi Arabia. Environmental Monitoring and Assessment, 155(1–4), 509–526.
Balachandran, K. K., Laluraj, C. M., Nair, M., Joseph, T., Sheeba, P., & Venugopal, P. (2005). Heavy metal accumulation in a flow restricted, tropical estuary. Estuarine, Coastal and Shelf Science, 65, 361–370.
Balachandran, K. K., Laluraj, C. M., Martin, G. D., Srinivas, K., & Venugopal, P. (2006). Environmental analysis of heavy metal deposition in a flow-restricted tropical estuary and its adjacent shelf. Environmental Forensics, 7, 345–351.
Birch, G. (2003). A scheme for assessing human impacts on coastal aquatic environments using sediments. In C. D. Woodcoffe & R. A. Furness (Eds.), Coastal GIS 2003. Australia: Wollongong University Papers in Center for Maritime Policy.
Birch, G. F., & Taylor, S. E. (2002). Application of sediment quality guidelines in the assessment of contaminated surficial sediments in Port Jackson (Sydney Harbour), Australia. Environmental Management, 29(6), 860–870.
Bouezmarni, R., & Wollast, R. (2005). Geochemical composition of sediments in the Scheldt estuary with emphasis on trace metals. Hydrobiologia, 540, 155–168.
Buccolieri, A., Buccolieri, G., Cardellicchio, N., Dell'Atti, A., Di Leo, A., & Maci, A. (2006). Heavy metals in marine sediments of Taranto Gulf (Ionian Sea, Southern Italy). Marine Chemistry, 99(1–4), 227–235.
Carr, R. S., Long, E. R., Windom, H. L., Chapman, D. C., Thursby, G., Sloane, G. M., et al. (1996). Sediment quality assessment studies of Tampa Bay, Florida. Environmental Toxicology and Chemistry, 15, 1218–1231.
Cevik, F., Goksu, M. Z. L., Derici, O. B., & Findik, O. (2009). An assessment of metal pollution in surface sediments of Seyhan dam by using enrichment factor, geoaccumulation index and statistical analyses. Environmental Monitoring and Assessment, 152, 309–317.
Chandramohan, T., & Balchand, A. N. (2007). Regional sediment yield pattern for the west flowing rivers of Kerala state India. RMZ—Materials and Geoenvironment, 54(4), 501–511.
Chapman, P. M., & Wang, F. (2001). Assessing sediment contamination in estuaries. Environmental Toxicology and Chemistry, 20, 3–22.
DelValls, T. A., Forja, J. M., Gonzalez-Mazo, E., & Gomez-Parra, A. (1998). Determining contamination sources in marine sediments using multivariate analysis. Trends in Analytical Chemistry, 17(4), 181–192.
Demirak, A., Yilmaz, F., Tuna, A. L., & Ozdemir, N. (2006). Heavy metals in water, sediment and tissues of Leuciscus cephalus from a stream in southwestern Turkey. Chemosphere, 63, 1451–1458.
Essien, J. P., Antai, S. P., & Olajire, A. A. (2009). Distribution, seasonal variations and ecotoxicological significance of heavy metals in sediments of Cross river, estuary mangrove swamp. Water, Air, and Soil Pollution, 197, 91–105.
Folk, R. L. (1974). Petrology of sedimentary rocks. TX, Hemphill: Austin. 182.
Gajghate, D. G., & Bhanarkar, A. D. (2005). Characterisation of particulate matter for toxic metals in ambient air of Kochi city, India. Environmental Monitoring and Assessment, 102, 119–129.
Gaudette, H. E., Flight, W. R., Toner, L., & Folger, D. W. (1974). An inexpensive titration method for the determination of organic carbon in recent sediments. Journal of Sedimentary Petrology, 44, 249–253.
Gopalan, U. K., Vengayil, D. T., Udayavarma, P., & Krishnankutty, M. (1983). The shrinking backwaters of Kerala. Journal of Marine Biological Association of India, 25, 131–141.
Harikumar, P. S., Nasir, U. P., & Mujeebu Rahman, M. P. (2009). Distribution of heavy metals in the core sediments of a tropical wetland system. International journal of Environmental Science and Technology, 6(2), 225–232.
Idris, A. M. (2008). Combining multivariate analysis and geochemical approaches for assessing heavy metal level in sediments from Sudanese harbors along the Red Sea coast. Microchemical Journal, 90, 159–163.
Ip, C. C. M., Li, X. D., Zhang, G., Wai, O. W. H., & Li, Y. S. (2007). Trace metal distribution in sediments of the Pearl River Estuary and the surrounding coastal areas, South China. Environmental Pollution, 147, 311–323.
Jain, C. K., Singhal, D. C., & Sharma, U. K. (2005). Metal pollution assessment of sediment and water in the river Hindon, India. Environmental Monitoring and Assessment, 105, 193–207.
Jayaprakash, A. A. (2002). Long term trends in rainfall, sea level and solar periodicity, a case study for forecast of Malabar sole and oil sardine fishery. Journal of Marine Biological Association of India, 44, 163–175.
Jonathan, M. P., Ram-Mohan, V., & Srinivasalu, S. (2004). Geochemical variations of major and trace metals in recent sediments, off the Gulf of Mannar, the southeast coast of India. Environmental Geology, 45, 466–480.
Joseph, P. V. (2002). Dynamics and speciation of heavy metals in the lower reaches of Chitrapuzha—a tropical tidal river. Ph.D. thesis, Cochin University of Science and Technology.
Karbassi, A. R., Monavari, S. M., Nabi Bidhendi, Gh. R., Nouri, J., & Nematpour, V. (2008). Metal pollution assessment of sediment and water in the Shur River. Environmental Monitoring and Assessment, 147, 107–116.
Laluraj, C. M., Gopinath, G., Dinesh Kumar, P. K., & Balachandran, K. K. (2008). Hydrodynamic and geomorphic controls on the morphology of an island ecosystem in the Vembanad Lake, West Coast of India. Journal of Coastal Research, 24(2), 145–150.
Liu, W. X., Li, X. D., Shen, Z. G., Wang, D. C., Wai, O. W. H., & Li, Y. S. (2003). Multivariate statistical study of heavy metal enrichment in sediments of the Pearl River Estuary. Environmental Pollution, 121, 377–388.
Liu, C., Xu, J., Liu, C., Zhang, P., & Dai, M. (2009). Heavy metals in the surface sediments in Lanzhou Reach of Yellow River China. Bulletin Environ Contam Toxicol, 82, 26–30.
Long, E. R., & MacDonald, D. D. (1998). Recommended uses of empirically derived, sediment quality guidelines for marine and estuarine ecosystems. Human and Ecological Risk Assessment, 4(5), 1019–1039.
Long, E. R., Field, L. J., & Macdonald, D. D. (1998). Predicting toxicity in marine sediments with numerical Sediment Quality Guidelines. Environmental Toxicology and Chemistry, 17(4), 714–727.
Long, E. R., MacDonald, D. D., Severn, C. G., & Hong, C. B. (2000). Classifying probabilities of acute toxicity in marine sediments with empirically derived sediment quality guidelines. Environmental Toxicology and Chemistry, 19, 2598–2601.
Loring, D. H., & Rantala, R. T. T. (1992). Manual for the geochemical analyses of marine sediments and suspended particulate matter. Earth-Science Reviews, 32, 235–283.
Loska, K., & Wiechula, D. (2003). Application of principal component analysis for the estimation of source of heavy metal contamination in surface sediments from the Rybnik reservoir. Chemosphere, 51(8), 723–733.
Marchand, C., Lallier-Vergés, E., Baltzer, F., Albéric, P., Cossa, D., & Baillif, P. (2006). Heavy metals distribution in mangrove sediments along the mobile coastline of French Guiana. Marine Chemistry, 98(1–2), 1–17.
Martin, G. D., Nisha, P. A., Balachandran, K. K., Madhu, N. V., Nair, M., Shaiju, P., et al. (2010). Eutrophication induced changes in benthic community structure of a flow-restricted tropical estuary (Cochin backwaters), India. Environmental Monitoring and Assessment. doi:10.1007/s10661-010-1594-1.
Menon, N. N., Balchand, A. N., & Menon, N. R. (2000). Hydrobiology of the Cochin backwater system—a review. Hydrobiologia, 430, 149–183.
Nair, K.K.C. (2002). Breathing Cochin Backwaters. Need for conservation. http://drs.nio.org/drs/bitstream/2264/204/1/gbwmp_1st_rd_35.pdf. Accessed 10 November 2010.
Neto, B. J. A., Smith, B. J., & McAllister, J. J. (2000). Heavy metal concentrations in surface sediments in a nearshore environment Jurujuba Sound Southeast Brazil. Environmental Pollution, 109(1), 1–9.
Padmalal, D., & Seralathan, P. (1995). Geochemistry of Fe and Mn in surficial sediments of a tropical river and estuary, India—a granulometric approach. Environmental Geology, 25, 270–276.
Padmalal, D., Maya, K., & Seralathan, P. (1997). Geochemistry of Cu, Co, Ni, Zn, Cd and Cr in the surficial sediments of a tropical river and estuary, southwest coast of India—a granulometric approach. Environmental Geology, 31, 85–93.
Qasim, S. Z. (2003). Indian estuaries. Mumbai: Allied Publication Pvt. Ltd.
Ratheesh Kumar, C. S., Joseph, M. M., Gireesh Kumar, T. R., Renjith, K. R., Manju, M. N., & Chandramohanakumar, N. (2010). Spatial variability and contamination of heavy metals in the inter-tidal systems of a tropical environment. International Journal of Environmental Research, 4(4), 691–700.
Ray, A. K., Tripathi, S. C., Patra, S., & Sarma, V. V. (2006). Assessment of Godavary estuarine mangrove ecosystem through trace metal studies. Environment International, 32, 219–223.
Renjith, K. R., & Chandramohanakumar, N. (2009). Distribution of heavy metals in the surficial sediments of a complex micro-tidal estuarine system in Southwest India. Research Journal of Chemistry and Environment, 13(2), 34–44.
Rodríguez-Barroso, M. R., García-Morales, J. L., Coello Oviedo, M. D., & Quiroga Alonso, J. M. (2010). An assessment of heavy metal contamination in surface sediment using statistical analysis. Environmental Monitoring and Assessment, 163(1–4), 489–501.
Rubio, B., Nombela, M. A., & Vilas, F. (2000). Geochemistry of major and trace metals in sediments of the Ria de Vigo (NW Spain), an assessment of metal pollution. Marine Pollution Bulletin, 40, 968–980.
Schiff, K. C., & Weisberg, S. B. (1999). Iron as a reference element for determining trace metal enrichment in Southern California coast shelf sediments. Marine Environmental Research, 48(2), 161–176.
Shajan, K. P. (2001). Geochemistry of bottom sediments from a river-estuary- shelf mixing zone on the tropical southwest coast of India. Bulletin of the Geological Survey of Japan, 52, 371–382.
Shaw, P. J. A. (2003). Multivariate statistics for the environmental sciences. New York: Arnold Publishers Oxford University Press Inc.
Simeonov, V., Massart, D. L., Andreev, G., & Tsakovski, S. (2000). Assessment of metal pollution based on multivariate statistical modeling of ‘hot spot’ sediments from the Black Sea. Chemosphere, 41, 1411–1417.
Soman, K. (1997). Geology of Kerala. Bangalore: Geological Society of India Publishers.
Srinivas, K., Revichandran, C., Maheswaran, P. A., Md Ashraf, T. T., & Nuncio, M. (2003). Propagation of tides in the Cochin estuarine system, southwest coast of India. Indian Journal of Marine Science, 32, 14–24.
Thomson, K. T. (2002). Economic and social issues of biodiversity loss in Cochin backwaters. Technical report: Cochin University of Science and Technology, Cochin, India.
Tomlinson, D. L., Wilson, J. G., Harris, C. R., & Jeffrey, D. W. (1980). Problems in the assessment of heavy-metal levels in estuaries and the formation of a pollution index. Helgolander Meeresuntersuchungen Helgolander Meeresunters, 33, 566–575.
Turekian, K. K., & Wedepohl, K. H. (1961). Distribution of the elements in some major units of the earth’s crust. Bulletin of Geological Society of America, 72, 175–191.
Vaasma, T. (2008). Grain-size analysis of lacustrine sediments: a comparison of pre-treatment methods. Estonian Journal of Ecology, 57(4), 231–243.
Valdes, J., Vargas, G., Sifeddine, A., Ortlieb, L., & Guiñez, M. (2005). Distribution and enrichment evaluation of heavy metals in Mejillones Bay (23°S), Northern Chile, geochemical and statistical approach. Marine Pollution Bulletin, 50(12), 1558–1568.
Zourarah, B., Maanan, M., Robin, M., & Carruesco, C. (2008). Sedimentary records of anthropogenic contribution to heavy metal content in Oum Er Bia estuary (Morocco). Environmental Chemistry Letters, 7(1), 67–78.
Acknowledgements
One of the authors gratefully acknowledges Dr. K.R. Renjith, Cochin University of Science and Technology and Dr. S. Srinivasalu and Dr. B. Senthilkumar, Anna University for their helpful comments at various stages while writing manuscript. In addition, we wish to thank Mr. Rajkumar, Mr. Sathishkumar, and Ms. Thulasibai from our institute, for their help in preparing geochemical maps. We are sincerely grateful for the positive journal corrections made by the anonymous reviewer that have improved the clarity of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Selvam, A.P., Priya, S.L., Banerjee, K. et al. Heavy metal assessment using geochemical and statistical tools in the surface sediments of Vembanad Lake, Southwest Coast of India. Environ Monit Assess 184, 5899–5915 (2012). https://doi.org/10.1007/s10661-011-2389-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10661-011-2389-8