Abstract
Objective
To determine the effects of artificial disulfide bridges on the thermostability and catalytic efficiency of chitosanase EAG1.
Results
Five artificial disulfide bridges were designed based on the structural information derived from the three-dimensional (3-D) model of chitosanase EAG1. Two beneficial mutants (G113C/D116C, A207C-L286C) were located in the flexible surface loop region, whereas the similar substitutions introduced in α-helices regions had a negligible effect. Mut5, the most active mutant, had a longer half-life at 50 °C (from 10.5 to 69.3 min) and a 200 % higher catalytic efficiency (K cat/K m) than that of the original EAG1.
Conclusions
The contribution of disulfide bridges to enzyme thermostability is mainly dependent on its location within the polypeptide chain. Strategical placement of a disulfide bridge in flexible regions provides a rigid support and creation of a protected microenvironment, which is effective in improving enzyme’s thermostability and catalytic efficiency.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Chitosan is a cationic amino polysaccharide with a linear structure of β-1,4-linked D-glucosamine (GlcN) residues (Kim and Rajapakse 2005). Chitosan can be degraded into chitosanoligosaccharides (COSs) by chemical or enzymatic methods. The functional properties of COSs depend on their molecular weight. COSs from 0.5 to 5.5 kDa exhibit promising biological activities including antitumor, antifungal and antibacterial effects (Jeon et al. 2001; Shen et al. 2009; Vo et al. 2011). In the preparation of COSs, chemical hydrolysis of chitosan has some drawbacks to be commercialized due to the production of a large amount of monosaccharides and the higher risk associated with the environmental pollution. Enzymatic-based approaches are preferable because they are more controllable and the amount of acidic waste is limited (Cheng et al. 2006). In industrial enzyme applications, thermostability is a key factor. In general, high thermostability is often accompanied by a longer half-life (Ferdjani et al. 2011). A 2–15 °C increase of thermostability often corresponds to a tenfold longer life time (Korkegian et al. 2005). A number of protein engineering techniques, such as random mutagenesis, DNA shuffling, truncation and circularization, have been developed for improving the thermostability of proteins (Eijsink et al. 2004; Tokuriki and Tawfik 2009; Zhao et al. 2010). However, it is difficult to acquire beneficial mutants because the screening of positive clones is often laborious and usually requires the creation of large enzyme mutant libraries. This triggers a quest for more focused methods to reduce the screening effort required to find a desired enzyme.
EAG1 is a classical glycoside hydrolase from Bacillus ehimensis. Not only can it hydrolyze the β-1,4-glycosidic bonds of chitosan to COS in different sizes but also keeps a high catalytic activity in organic solvents and with metal ions. However, EAG1 is relatively unstable and its activity decreases abruptly above 50 °C (Kouichi et al. 1999). Several factors, including hydrogen bonds, packing density and the number of ion pairs, contribute to the protein thermostability. Strong covalent disulfide bonds are one of the important factors, which stabilize the proteins mostly through an entropic effect, by decreasing the conformational chain entropy of the denatured protein (Clarke et al. 2000; Matsumura et al. 1989). Many mutant proteins have distinct thermostability because of the introduction of artificial disulfide bridges (Joo et al. 2011; Siadat et al. 2006). In this paper, we constructed the three-dimensional (3-D) structure model of EAG1 using a homology model and designed different disulfide bridges through the strategy of computational design to investigate the effects of disulfide bridges on thermostability and catalytic efficiency.
Materials and methods
Strains and media
The EAG1 encoding region gene (GenBank accession number AB008788) from B. ehimensis (IF015659) was synthesized by Sangon Inc (Shanghai, China). The plasmid, pPIC9K, and Pichia pastoris GS115 were from Invitrogen (Shanghai, China). T4 DNA ligase, along with other restriction endonucleases, was from Takara Bio. Co. (Dalian, China). P. pastoris, harboring the mutated EAG1 gene, were cultured in BMGY medium and transferred to BMMY medium for protein expression. BMGY/BMMY medium: 10 g yeast extract l−1, 20 g peptone l−1, 13.4 g yeast nitrogen base l−1, 4 × 10−4 g biotin l−1, 100 mM potassium phosphate pH 6, and 10 ml glycerol l−1 or 5 ml methanol l−1.
Homology modeling
Homology modeling techniques and molecular dynamics (MD) simulations were utilized to construct and refine the 3-D structure of EAG1. A sequence similarity search for EAG1 against amino acid sequences from the Protein Data Bank (PDB) was performed on the BLAST online server (http://blast.ncbi.nlm.nih.gov). After excluding the redundant results, the structure of EAG1 was modelled based on the structure of chitosanase MH-K1 from Bacillus circulans (1.6 Å resolution). MD simulation was carried out at normal pressure and temperature to examine the quality of model structures.
Design of disulfide bridges and plasmid construction
Sites for insertion of disulfide bridges were selected using the program Disulfide by Design (Dombkowski, Wayne State University, USA), which recognizes cysteine pairs that are in the proper orientation to form a disulfide bridge. For EAG1, all residue pairs are rapidly assessed for proximity and geometry consistent with disulfide bridge formation, assuming the residues were mutated to cysteines. Mutated EAG1 gene with no signal peptide coding region (1–43 amino acids) was obtained through substituting target residue to cysteine using overlap extension PCR. To facilitate the purification of the recombinant enzyme, hexahistidine (6×His)-tagged was designed at the C-terminal of EAG1. Mutated PCR product was digested with the EcoRI and AvrII and inserted into the multicloning site of the expression vector pPIC9k.
Enzyme preparation and assay
The recombinant plasmids were linearized with BglII and transformed into P. pastoris for protein expression. Positive transformants were initially grown in BMGY medium (25 ml) in 100 ml flasks at 28 °C with shaking (200 rpm) until the OD600 reached 4.5. The culture was centrifuged at 3000×g for 10 min, the resuspended pellets were added to 1 l BMMY medium. To maintain expression, methanol was added every 24 h to give 0.5 % (v/v). After 120 h induction, about 1 l supernatant was concentrated to 80 ml by ultrafiltration (10 kDa cut-off). One ml concentrated crude enzyme was diluted in 5 ml of buffer A (20 mM sodium phosphate containing 500 mM NaCl and 20 mM imidazole, pH 8) and applied to Ni2+-charged 1 ml HisTrap FF crude column (GE Healthcare) equilibrated with buffer A. The recombinant EAG1 were eluted with buffer B (buffer A plus 400 mM imidazole). Chitosanase activity was determined at 40 °C by estimating the amount of the reducing ends of sugars using a modified dinitrosalicyclic acid (DNS) method with glucosamine·HCl as the calibration standard. One unit of chitosanase was defined as the amount of enzyme required to liberate 1 μmol reducing sugar per min under the conditions described above. Three replicates were performed per analysis.
Thermal inactivation
The deactivation constants (k d) was calculated from the semi-logarithmic plot of residual activity versus time (Eq. 1), where Et is the residual enzyme activity after heat treatment for time, E0 is the initial enzyme activity before heat treatment. The half-lives of the wild-type and mutant EAG1 were calculated using Eq. 2.
Michaelis–Menten kinetics
The kinetic parameters were determined with chitosan from 5 to 20 mg ml−1 at 40 °C for 10 min. The K m and the turnover number (K cat) were calculated using the nonlinear least-square fitting procedure for the Michaelis–Menten standard protocol in Prism software (version 5.0 for Windows, San Diego, CA, USA).
Results
Homology modeling
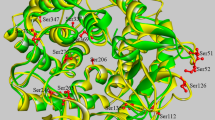
Taking the amino acid sequence of EAG1 as a standard, high homology templates were scouted in the PDB database. B. circulans MH-K1 chitosanase (PDB: 1QG1) shared 91 % sequence identity with EAG1, while others shared no less than 50 % sequence identity. Thus, MH-K1 was selected as a template for the homology modeling. After the MD energy minimization, the check process was carried out. The Ramachandran plot showed that 92.4 % of residues were in the most favored allowed regions and 4 % were in the additional allowed region (Supplementary Fig. 1). Verify-3D graphs showed that 86.3 % of the residues had average 3D-1D scores of no less than 0.2. These results showed that side chain environments in the selected model were acceptable. Meanwhile, the overall quality factor of our model was 75.657. Thus, the selected model was acceptable for further analysis. The overall molecular structure of EAG1 is shown in Fig. 1. EAG1 is composed of two globular domains including an upper domain and a lower domain. As a backbone, α6 links to the two independent domains. EAG1 has 13 α-helices and 3 β-strands. Long loops with short β-strands protrude from the end of the outer helices and fold towards the inner helices, forming the substrate-binding pockets. Six acidic residues, including Glu80, Glu94, Asp98, Asp120, Gly121 and Thr186, participate in substrate recognition. Two cysteine pairs 93/167 form a disulfide bridge to stabilize the molecule. Glu80 and Asp98, assigned as a proton donor and an acceptor respectively, are the conserved catalytic residues.
Models of the simulated structure of EAG1. EAG1 is labeled with red at the beginning of the N-terminus, and then goes through the rainbow to become purple at the C-terminus. The six substrate recognition residues are shown as stick models and colored by element. Cys93 and Cys167 that form the disulfide bridge are shown as ball-and-stick representations with amino acid coloring
Design of disulfide bridges
In the design of novel disulfide bridges, all the residue pairs of EAG1 were assessed by proximity and geometry. 39 residue pairs including one original disulfide bridges (residues 93/167) satisfied the geometric constraints for disulfide bridges. To select the most promising candidates, the mutant sites were further selected in accordance with the principle that they had relatively low bond energy with Chi3 (×3 torsion angle) value between ±90 and ±110. Fifteen residue pairs for possible disulfide bridges satisfied the energy constraints. In order to prevent the loss of catalytic activity and get more reliable residue pairs for the formation of beneficial mutations, no disulfide bridge was designed close to the catalytic residues and substrate binding sites. Finally, five disulfide bridges were designed in silico (Table 1).
Thermostability and thermal inactivation analysis of the EAG1 mutants
The effects of disulfide bridges on thermostability were investigated in all the expressed and purified mutants. Among the five mutants, two had higher thermostability than that of the wild-type EAG1. As shown in Fig. 2, the wild-type EAG1 had a significant enzyme activity loss at 50 °C and less than 37 % of the initial activity after 30 min. While the Mut1 (G113C/D116C) and Mut5 (A207C-L286C) had markedly improved enzyme stability and remained 62.0 and 81.0 % of residual enzyme activity respectively. Structural analysis showed that Mut1 and Mut5 were located in surface-exposed loop regions (Fig. 3).
Thermostability of wild type and the mutants. Thermostability of the wild-type and mutants was tested by preincubating the enzyme (0.1 mg protein ml−1) at 40–60 °C for 30 min, the residual activities ofter heat treatment were measured immediately. The activity of the pre-incubated enzyme at 4 °C was taken as 100 %
Why the mutations strongly enhance protein thermostability, and similar substitutions introduced elsewhere (such as α-helices region) have a negligible effect? From the structural view, we speculate that the strongly stable mutations are usually located at the spot where the protein starts to unfold. The surface-exposed loop regions are highly flexible and more likely to unfold at early stage, cross-linking of these regions via a disulfide bridge has a higher chance to stabilize the folded state. The enzyme activity is affected when mutations appear in α-helices regions. Excessive rigidity caused by steric contacts or loss of favorable interactions with substitution of a cysteine residue decreased the ability of conformational changes at high temperature and influenced the enzyme activity.
Mut5, one of the most active mutants, was selected for thermal inactivation test. The half-life of Mut5 was monitored at a constant temperature. As shown in Fig. 4 (a), the wild-type EAG1 lost almost 50 % activity at 50 °C over 10 min. Over 50 min, residual enzyme activity dropped to about 5 % of the original. Mut5 had substantially improved enzyme stability due to the formation of the new disulfide bridge, the residual chitosanase activity still kept 67 % of the original at 50 °C over 50 min. The deactivation constants (k d) for the first-order thermal deactivation were calculated using Eq. (1). As shown in Fig. 4 (b), the k d of the Mut5 at 50 °C was 10−2 min−1 in comparison with 6.6 × 10−2 min−1 of wild-type EAG1. The half-lives of Mut5 and EAG1 were determined according to Eq. (2). The Mut5 had a half-life of 69.3 min, which was higher than that of the wild-type EAG1 (10.5 min).
Thermal deactivation curves (a) and thermal deactivation constants (b) of EAG1 and Mut5. The thermal inactivation at 50 °C of enzyme was determined up to 0–80 min. the residual activities ofter heat treatment were measured immediately. The activity of the pre-incubated enzyme at 4 °C was taken as 100 %
Michaelis–Menten kinetics
To investigate the importance of disulfide bridge in the catalytic activity, the kinetic parameters of EAG1 and Mut5 (1.5 mg protein ml−1) toward chitosan were determined (Table 2). Compared with the EAG1, Mut5 had a 141 % increase in V max. A previous report has suggested that only approximately 7 % of disulfide bridges available in the PDB database are involved in allosteric regulation of enzymes and substrate binding during catalysis (Pace and Weerapana 2013). Our results showed that no significant change occurred in the K m value between the EAG1 and the Mut5. This result may be explained by the position of Cys residues. The disulfide bridge between residues C207 and C286 forms at the middle of the loop regions, and it is far away from the substrate-binding pockets, which only increases the rigidity of the two flexible regions and has no significant influence on substrate binding. The K cat values of the EAG1 and the Mut5 were 2.5 and 4.9, respectively. The catalytic efficiency (K cat/K m) was 0.52 for EAG1 and 1.03 for Mut5, respectively. The disulfide bridge did not affect the K m, but increased K cat and the overall catalytic efficiency of Mut5.
Discussion
How to improve enzyme thermostability to resist extremely harsh conditions is a meaningful target in protein engineering research. In this study, the effects of strategical placement of a disulfide bridge on thermostability and catalytic efficiency of chitosanase EAG1 were investigated. Five artificial disulfide bridges were introduced into the EAG1 according to the geometry and energy comparative analysis. It is more likely that beneficial mutants can be obtained when amino acids residues located in flexible surface loop region are mutated to cysteines. This means that the contribution of different disulfide bridges to enzyme thermostability mainly depends on its location within the polypeptide chain. Compared with the high rigidity region, the loops are likely to accommodate a variety of stabilizing mutations without disturbing the 3-D structure of proteins.
On the relationship between enzymatic rigidity and activity, some researchers believe that high flexibility of a protein is associated with a high affinity (Joseph et al. 2008) and, introducing a disulfide bridge could lead to the decrease of the V max and the affinity (Yanget al. 2007). Another view is that improving the rigidity of the protein can increase the affinity and catalytic activity (Chen et al. 2013). Chen et al. (2013) introduced a disulfide bridge into xylanase resulting in the affinity and catalytic efficiency of mutant, AoXyn11AC5–C32, improving 263 and 168 %, respectively, compared to the wild-type AoXyn11A. We believe that the stability of the protein is sometimes not at the cost of enzyme affinity and activity. In some extent, localized rigidity may be compatible with the overall flexibility. Increasing rigidity, enhancing affinity and improving catalytic efficiency are not contradicted.
Even though engineering a disulfide bridge is an effective method to improve protein stability, there are still some factors limiting its prevalence. Firstly, the disulfide bridge introduction also brings to detrimental effects, or even destabilization. Molecular flexibility is essential both for substrates binding and catalysis, while stability is needed to ensure the appropriate geometry to avoid denaturation. How to keep the balance between molecular stability and structural flexibility is still a challenge. Secondly, acquiring the crystals and understanding the 3-D structure information of proteins are necessary to use the appropriate computational strategy to design disulfide bridge in a sensible fashion. For some low similarity enzymes, little structure information affected the applicatioon of this technique.
References
Chen Z, Zhang H, Wang J, Tang C, Wu J, Wu M (2013) Engineering the thermostability of a xylanase from Aspergillus oryzae by an enhancement of the interactions between the N-terminus extensionand the β-sheet A2 of the enzyme. Biotechnol Lett 35:2073–2079
Cheng CY, Chang CH, Wu YJ, Li YK (2006) Exploration of glycosyl hydrolase family 75, a chitosanase from Aspergillus fumigates. J Biol Chem 281:3137–3144
Clarke J, Hounslow AM, Bond CJ, Fersht AR, Daggett V (2000) The effects of disulfide bonds on the denatured state of barnase. Protein Sci 9:2394–2404
Eijsink VGH, Bjørk A, Ga SS, Sireva GR, Synstad B, Van DB, Vriend G (2004) Rational engineering of enzyme stability. J Biotechnol 113:105–120
Ferdjani S, Ionita M, Roy B, Dion M, Djeghaba Z, Rabiller C, Tellier C (2011) Correlation between thermostability and stability of glycosidases in ionic liquid. Biotechnol Lett 33:1215–1219
Jeon YJ, Park PJ, Kim SK (2001) Antimicrobial effect of chitooligosacchardies produced by bioreactor. Carbohydr Polym 44:71–76
Joo JC, Pack SP, Kim YH, Yoo YJ (2011) Thermostabilization of Bacillus circulans xylanase: computational optimization of unstable residues based on thermal fluctuation analysis. J Biotechnol 151:56–65
Joseph B, Ramteke PW, Thomas G (2008) Cold active microbial lipases: some hot issues and recent developments. Biotechnol Adv 26:457–470
Kim SK, Rajapakse N (2005) Enzymatic production and biological activities of chitosan oligosacch- arides (COS): a review. Carbohydr Polym 62:357–368
Korkegian A, Black ME, Baker D, Stoddard BL (2005) Computational thermostabilization of an enzyme. Science 308:857–860
Kouichi A, Fujita T, Kuroshima KI, Sakane T, Yakota A, Takata R (1999) Purification and gene cloning of a chitosanase from Bacillus ehimensis EAG1. J Biosci Bioeng 87:383–385
Matsumura M, Signor G, Matthews BW (1989) Substantial increase of protein stability by multiple disulphide bonds. Nature 342:291–293
Pace NJ, Weerapana E (2013) Diverse functional roles of reactive cysteines. ACS Chem Biol 8:283–296
Shen KT, Chen MH, Chan HY, Jeng JH, Wang YJ (2009) Inhibitory effects of chitooligosaccharides on tumor growth and metastasis. Food Chem Toxicol 47:1864–1871
Siadat OR, Lougarre A, Lamouroux L, Ladurantie C, Fournier D (2006) The effect of engineered disulfide bonds on the stability of Drosophila melanogaster acetylcholinesterase. BMC Biochem 7:12
Tokurik N, Tawfik DS (2009) Stability effects of mutations and protein evolvability. Curr Opin Struct Biol 19:596–604
Vo TS, Kong CS, Kim SK (2011) Inhibitory effects of chitooligosaccharides on degranulation and cytokine generation in rat basophilic leukemia RBL-2H3 cells. Carbohydr Polym 84:649–655
Yang HM, Yao B, Meng K, Wang YR, Bai YG, Wu NF (2007) Introduction of a disulfide bridge enhances the thermostability of a Streptomyces olivaceoviridis xylanase mutant. J Ind Microbiol Biotechnol 34:213–218
Zhao Z, Ma X, Li L, Zhang W, Ping S, Xu MQ, Lin M (2010) Protein cyclization enhanced thermostability and exopeptidase resistance of green fluorescent protein. J Microbiol Biotechnol 20:460–466
Acknowledgments
This study was supported by the grants from basic research funds of Central Government and Public Research Institutes (No. 2014C04XK01 and 20603022013016).
Supporting information
Supplementary Figure 1—Ramachandran plot of EAG1.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sheng, J., Ji, X., Zheng, Y. et al. Improvement in the thermostability of chitosanase from Bacillus ehimensis by introducing artificial disulfide bonds. Biotechnol Lett 38, 1809–1815 (2016). https://doi.org/10.1007/s10529-016-2168-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10529-016-2168-2