Abstract
Background
Because of the small numbers of cases in single centers, the indications for and survival benefits of adrenalectomy for adrenal metastasis remain unclear. We evaluated the outcomes of laparoscopic adrenalectomy for patients with adrenal metastasis.
Methods
We retrospectively analyzed the records of 67 patients who underwent laparoscopic adrenalectomy for metastatic disease from 2003 to 2017 at 11 hospitals. Associations of clinical, surgical, and pathologic features with overall survival (OS) and positive surgical margins were evaluated using univariate and multivariate Cox regression analyses and univariate logistic regression analysis.
Results
Lung cancer (30%) and renal cell carcinoma (30%) were the most common primary tumor types. Intraoperative complications were observed in seven patients (10%) and postoperative complications in seven (10%). The surgical margin was positive in 10 patients (15%). The median OS was 3.8 years. Univariate analysis showed that the tumor size, episodes of extra-adrenal metastasis before adrenalectomy, extra-adrenal metastasis at the time of adrenalectomy, and positive surgical margins were significantly associated with shorter OS (p = 0.022, p = 0.005, p < 0.001, and p = 0.022, respectively). Multivariate analysis showed that extra-adrenal metastasis at the time of adrenalectomy and positive surgical margins remained statistically significant (p = 0.022 and p = 0.049, respectively). In the univariate analysis, the tumor size was significantly associated with positive surgical margins (p = 0.039).
Conclusions
Laparoscopic adrenalectomy for adrenal metastasis can be safely performed in selected patients, and patients with isolated adrenal metastasis and negative surgical margins seem to have more favorable outcomes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Because of its rich, sinusoidal blood supply, the adrenal gland is a common site for metastasis of malignancies. In patients with a history of malignancy, metastases to the adrenal gland are reportedly the second most common type of adrenal masses after benign adenomas [1].
The guidelines published by the American Association of Clinical Endocrinologists (AACE) and American Association of Endocrine Surgeons (AAES) indicate that surgical resection may improve disease-free survival in selected patients with good control of their extra-adrenal disease and good performance status [2]. While the effect of adrenalectomy (ADX) on survival differs depending on the cancer site of origin, patients with a solitary adrenal metastasis, smaller tumor size, and longer disease-free interval from the primary tumor diagnosis tend to have more favorable outcomes [1, 3-6]. However, all previous reports of ADX for adrenal metastasis were retrospective series, and only five reports with relatively small numbers of patients performed multivariate analyses to evaluate the associations of clinical features with overall survival (OS) after ADX [6-10]. Therefore, no clinical criteria with which to preoperatively define which patients will benefit from ADX in terms of cure or increased survival have yet been established.
Laparoscopic ADX is a standard procedure for benign adrenal disease. Even for metastatic adrenal disease, several recent studies have demonstrated the safety and similar oncologic outcomes for laparoscopic ADX compared with open ADX in selected patients [3, 6, 7, 9-13]. However, among the studies that involved laparoscopic surgery and more than 50 patients, only one assessed both perioperative complications and surgical margins in patients who underwent laparoscopic ADX for adrenal metastasis [8].
The present study was performed to assess the surgical and oncologic outcomes of laparoscopic ADX for adrenal metastasis and to identify patients who are most likely to have prolonged survival after resection of adrenal metastasis based on multicenter survey in Japan.
Material and methods
This retrospective multicenter study was based on survey data provided by three academic university hospitals and eight teaching hospitals in Japan. After obtaining institutional review board approval from each hospital, a retrospective chart review was conducted to identify patients who underwent laparoscopic ADX for metastasis from 2003 to 2017. Patients who underwent ADX at the time of ipsilateral nephrectomy for surgery for renal cell carcinoma were excluded. The patients’ medical records were reviewed for demographics, type of primary tumor, interval from diagnosis of the primary tumor to ADX, extent of metastatic disease before and at the time of ADX, operative approach [transperitoneal (anterior) or retroperitoneal (posterior) approach], perioperative factors (tumor laterality, tumor size, operative time, blood loss, open conversion, intraoperative and postoperative complications, and surgical margin), and date of last follow-up or death. Based on imaging studies, adrenal only group was defined as the patients with isolated adrenal metastasis, and extra-adrenal group was defined as the patients with other extra-adrenal metastatic site. The data were collected from each hospital using question sheets. The indication of transperitoneal or retroperitoneal laparoscopic approach was determined by each surgeon in each hospital. There was no definitive follow up protocol in terms of further surveillance imaging, and patients were referred back to the team dealing with the primary tumor in many hospitals. Using the Clavien–Dindo classification v2.0 [14], adverse events were recorded when we observed adverse event grade 2 or more. Major complications were defined as adverse events grade 3 or more.
Continuous variables were evaluated using Mann–Whitney U tests, and categorical variables were analyzed using chi-square test to compare clinical and pathological factors between transperitoneal and retroperitoneal laparoscopic approach groups. OS was measured from the date of ADX to the date of death of any cause or last follow-up, calculated using the Kaplan–Meier method, and compared using log rank test. Clinical and pathological factors were evaluated for their potential association with OS using univariate and multivariate Cox regression analyses. Multivariate OS analysis was conducted using stepwise Cox proportional hazards regression modeling and included all factors with a p value of < 0.05 in the univariate analysis. Preoperative factors were also evaluated for their association with positive surgical margins using univariate logistic regression analysis. Log transformation was used for analysis of continuous variables.
All statistical analyses were performed using JMP Pro version 13.0.0 software (SAS Institute Japan Inc., Tokyo, Japan), with p values of < 0.05 considered statistically significant.
Results
Table 1 summarizes patient characteristics according to the surgical approach. A total of 67 patients underwent laparoscopic ADX for adrenal metastasis from 2003 to 2017. Their median age was 68 years [interquartile range (IQR) 64–74], and 46 (69%) were men. The most common primary tumor types were lung cancer (n = 20, 30%) and renal cell carcinoma (n = 20, 30%), followed by liver cancer (n = 10, 15%) and colorectal cancer (n = 6, 9%). The median size of the adrenal metastasis was 2.8 cm (IQR 2.0–4.0). In most patients, adrenal metastasis was diagnosed by computed tomography. Twenty-seven patients (40%) had episodes of extra-adrenal metastasis before ADX. Forty-eight patients (72%) had isolated adrenal metastasis and 19 (28%) had extra-adrenal metastasis at the time of ADX. Of 19 patients who had extra adrenal metastatic disease, the common primary tumor types were lung cancer (n = 5) and renal cell carcinoma (n = 6). Thirteen patients had the plans of additional metastasectomy, radiotherapy or transcatheter arterial embolization for extra-adrenal metastatic disease after ADX. For two patients who had the episode of double cancer, ADX were performed to diagnose the primary site of metastatic disease. For one patient of malignant pheochromocytoma, ADX was performed to control the disease related symptoms. The median time from the primary tumor diagnosis to ADX was 2.4 years (IQR 1.1–4.9).
A retroperitoneal approach was selected in 18 patients (27%) and an intraperitoneal approach in 49 (73%) (Table 1). The median operative time was 175 min (IQR 141–253). Open conversion was performed in five patients (7%). The perioperative outcomes and surgical margin status are listed in Table 2. Intraoperative complications were observed in seven patients (10%). The intraoperative complications were pneumothorax (n = 3), inferior vena cava injury (n = 2), splenic artery injury (n = 1), and intestinal injury (n = 1). Eight postoperative complications were observed in seven patients (10%). The complications observed postoperatively were adrenal insufficiency (n = 2), bacterial peritonitis (n = 1), bacterial pneumonia (n = 1), ileus (n = 1), malignant hypertension (n = 1), rupture of a superior mesenteric artery aneurysm (n = 1), and multiorgan failure (n = 1). Two patients who received ipsilateral ADX during radical nephrectomy for renal cell carcinoma followed by contralateral ADX due to adrenal metastasis had adrenal insufficiency. Perioperative steroid cover was performed at the time of contralateral ADX. Major postoperative complications were observed in two patients (3%), including one who died of multiorgan failure 13 days following ADX for right adrenal metastasis of renal cell carcinoma.
There was no significant difference in clinical and pathological factors between transperitoneal and retroperitoneal laparoscopic approach groups (Tables 1, 2). Retroperitoneal approach was selected in 60% of liver cancer patients. Open conversion and postoperative complications were not observed in retroperitoneal approach group.
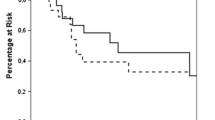
After a median follow-up of 1.5 years (IQR 0.7–2.7), 30 patients (45%) were alive without evidence of disease, 13 (19%) were alive with evidence of disease, and 24 (36%) died of disease or other causes. The median OS was 3.8 years [95% confidence interval (CI) 2.4–11.9]. The 1-, 3-, and 5-year OS rate following ADX were 82%, 58%, and 43%, respectively (Fig. 1).
Univariate analysis (Table 3) showed that the following factors were significantly associated with shorter OS: larger tumor size [hazard ratio (HR) 1.280; 95% CI 1.037–1.564; p = 0.022], episodes of extra-adrenal metastasis before ADX (HR 3.313; 95% CI 1.432–8.056; p = 0.005), extra-adrenal metastasis at the time of ADX (HR 5.777; 95% CI 2.372–14.236; p < 0.001), and positive surgical margins (HR 3.190; 95% CI 1.199–7.753; p = 0.022). In the multivariate analysis, extra-adrenal metastasis at the time of ADX (HR 4.127; 95% CI 1.227–13.783; p = 0.022) and positive surgical margins (HR 2.950; 95% CI 1.003–8.169; p = 0.049) remained statistically significant.
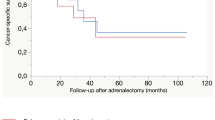
Figure 2 shows the Kaplan–Meier curves of the influence of the extent of disease and surgical margin status on OS after ADX. Patients with isolated adrenal metastasis at the time of ADX had better OS than patients with other extra-adrenal metastasis at the time of ADX (p < 0.0001). Additionally, a negative surgical margin was a statistically significant parameter for favorable OS (p = 0.009).
Preoperative factors associated with positive surgical margins were also evaluated, and the univariate analysis (Table 4) showed that a larger tumor size was significantly associated with positive surgical margins (HR 1.446; 95% CI 1.019–2.086; p = 0.039).
Discussion
Our study suggests that ADX for adrenal metastasis can be safely performed by laparoscopic surgery in carefully selected patients. While the median follow-up was relatively short, our median OS after ADX was 3.8 years and our 3- and 5-year OS estimates were 58% and 43%, respectively. These results are similar to those in other studies involving laparoscopic surgery that reported 5-year survival rates ranging from 31 to 45% after ADX for adrenal metastasis (Table 5) [3, 5, 6, 9-11]. This finding suggests that carefully selected patients may benefit from adrenal metastasectomy by laparoscopy.
Laparoscopic ADX is currently considered the standard procedure for benign adrenal tumors. Laparoscopic ADX for malignant tumors has been adopted more reluctantly because of concern for a high risk of incomplete resection and tumor dissemination. However, several recent reports have demonstrated that laparoscopic ADX for adrenal metastasis is equivalent to open ADX in terms of safety, local recurrence, and OS [3, 6, 7, 9-13]. In the present study, intraoperative complications were observed in seven patients (10%) and postoperative complications were observed in seven patients (10%), including two with severe postoperative complications (3%). These results are similar to those in other studies of laparoscopic ADX for adrenal metastasis, in which the intraoperative and postoperative complication rates ranged from 4 to 20% (Table 5) [3, 8, 9]. Notably, the capsule of adrenal malignant tumors is fragile [15], suggesting that laparoscopic ADX for metastasis should be performed by experienced surgeons.
Ma et al. reported favorable perioperative outcome of retroperitoneal laparoscopic ADX for adrenal metastasis, with surgical margin rates of 7% and complication rates of 8% [8]. Arenas et al. and Glenn et al. reported outcomes of ADX for adrenal metastasis, including open, transperitoneal and retroperitoneal laparoscopic approach [9, 16]. They showed that transperitoneal and retroperitoneal laparoscopic ADX were safe and effective for selected patients; however, the reports which compared transperitoneal and retroperitoneal laparoscopic approach were rare. In the present study, there were no significant difference in clinical and pathological factors between transperitoneal and retroperitoneal laparoscopic approach groups. Retroperitoneal approach was selected in 60% of liver cancer patients, suggesting that retroperitoneal approach might have been preferred in the patients who have undergone open intraperitoneal surgery. Interestingly, open conversion and postoperative complications were not observed in retroperitoneal group. It might be reasonable to select retroperitoneal approach in the patients who have undergone open intraperitoneal surgery and have the risk of severe intraperitoneal adhesions.
Patients with nonsurgically treated adrenal metastasis have a poor prognosis [5]. The current guidelines published by AAES/AACE for the management of adrenal masses suggest that ADX for adrenal metastasis is rarely indicated but may improve disease-free survival in a very select group of patients with good control of their extra-adrenal disease and good performance status [2]. While the effect of ADX on survival differs depending on the cancer site of origin, patients with a solitary adrenal metastasis, smaller tumor size, and longer disease-free interval from the time of the primary tumor diagnosis tend to have a more favorable tumor biology and better outcomes after ADX [1, 3-6]. However, the indications for ADX for adrenal metastasis and the survival benefit have not been clearly defined because all previous reports were retrospective series involving relatively small numbers of patients. Among the reports from non-western countries, this report include the second largest number of patients [8]. Although several retrospective series have identified selected groups of patients whose survival appears to have been prolonged with adrenal metastasectomy, few multivariate regression analyses have been performed to evaluate the associations of clinical and pathological factors with OS after ADX [6-10].
Our univariate analysis showed that larger tumor size, episodes of extra-adrenal metastasis before ADX, extra-adrenal metastasis at the time of ADX, and positive surgical margins were significantly associated with shorter OS after ADX. These clinical and surgical factors are compatible with those in previous reports [5, 6, 8, 17, 18]. Smaller tumor size and non-extra-adrenal metastasis might indicate a more favorable tumor biology. In our multivariate analysis, extra-adrenal metastasis at the time of ADX and positive surgical margins were significantly associated with OS. This is the first report which shows that extra-adrenal metastasis at the time of ADX were significantly associated with shorter OS using multivariate analyses. Because of the small number of the patients, multivariate analyses to evaluate the association of clinical and pathological features with OS after ADX have been performed by only five reports, and four of the reports included the patients with extra-adrenal metastasis at the time of ADX [6-10]. Some of them showed that positive surgical margins and fail of potential cure (positive surgical margins or residual metastasis) by ADX were associated with shorter OS; however, none of them have clearly showed that extra-adrenal metastasis at the time of ADX were associated with shorter OS [6-8].
Although the results may vary depending on the therapies performed before and after ADX, our data suggest that radiological and microscopical tumor free status with the result of ADX might bring favorable outcomes of these cohorts. Moreover, a larger tumor size was significantly associated with positive surgical margins in our study. Our data support the current AAES/AACE guidelines and indicate that patients with a solitary adrenal metastasis that is neither huge nor suspected to involve local invasion into contiguous structures seem to be good candidates for laparoscopic ADX.
The major limitation of our study is its retrospective nature, which may have introduced selection and ascertainment bias. Additionally, the median follow-up was relatively short and the recurrence rate was not assessed in our study, because many patients were referred back to the team dealing with the primary tumor and we couldn’t get the enough data about recurrence after ADX in many cases. Our study was a multicenter, retrospective review of patients who underwent laparoscopic ADX for adrenal metastasis, and all included patients of various primary tumor types were highly selected with no comparison group. Therefore, the effectiveness of open ADX or alternative less invasive treatments (or non-local treatments) is unknown. Favorable results of percutaneous radiofrequency ablation therapy for adrenal metastasis were recently reported [19-21]. To select the appropriate candidates who may gain a survival benefit of ADX for adrenal metastasis, a comprehensive, prospective randomized study comparing ADX with percutaneous ablation and non-local treatment would be ideal. However, such a prospective randomized study would be difficult because of the small numbers of cases in single institutions and the variety of underlying primary malignancies, disease burdens, and treatments provided. Therefore, accumulation of retrospective multicenter studies including patients who have undergone ADX, percutaneous ablation, and non-local treatment is important to perform a future meta-analysis for selection of the most appropriate candidates for ADX.
In conclusion, laparoscopic ADX for adrenal metastasis can be safely performed in selected patients. Patients with isolated adrenal metastasis and negative surgical margins might have more favorable outcomes after ADX.
Abbreviations
- AACE:
-
American Association of Clinical Endocrinologists
- AAES:
-
American Association of Endocrine Surgeons
- ADX:
-
Adrenalectomy
- OS:
-
Overall survival
- IQR:
-
Interquartile range
- CI:
-
Confidence interval
- HR:
-
Hazard ratio
References
Sancho JJ, Triponez F, Montet X et al (2012) Surgical management of adrenal metastases. Langenbeck's Arch Surg 397(2):179–194. https://doi.org/10.1007/s00423-011-0889-1
Zeiger MA, Thompson GB, Duh QY et al (2009) The American Association of Clinical Endocrinologists and American Association of Endocrine Surgeons medical guidelines for the management of adrenal incidentalomas. Endocr Pract 15(Suppl 1):1–20. https://doi.org/10.4158/ep.15.s1.1
Strong VE, D'Angelica M, Tang L et al (2007) Laparoscopic adrenalectomy for isolated adrenal metastasis. Ann Surg Oncol 14(12):3392–3400. https://doi.org/10.1245/s10434-007-9520-7
Gittens PR Jr, Solish AF, Trabulsi EJ (2008) Surgical management of metastatic disease to the adrenal gland. Semin Oncol 35(2):172–176. https://doi.org/10.1053/j.seminoncol.2007.12.006
Vazquez BJ, Richards ML, Lohse CM et al (2012) Adrenalectomy improves outcomes of selected patients with metastatic carcinoma. World J Surg 36(6):1400–1405. https://doi.org/10.1007/s00268-012-1506-3
Moreno P, de la Quintana Basarrate A, Musholt TJ et al (2013) Adrenalectomy for solid tumor metastases: results of a multicenter European study. Surgery 154(6):1215–1222. https://doi.org/10.1016/j.surg.2013.06.021(discussion 1222–1213)
Muth A, Persson F, Jansson S et al (2010) Prognostic factors for survival after surgery for adrenal metastasis. Eur J Surg Oncol 36(7):699–704. https://doi.org/10.1016/j.ejso.2010.04.002
Ma X, Li H, Zhang X et al (2013) Modified anatomical retroperitoneoscopic adrenalectomy for adrenal metastatic tumor: technique and survival analysis. Surg Endosc 27(3):992–999. https://doi.org/10.1007/s00464-012-2553-4
Romero Arenas MA, Sui D, Grubbs EG et al (2014) Adrenal metastectomy is safe in selected patients. World J Surg 38(6):1336–1342. https://doi.org/10.1007/s00268-014-2454-x
Zerrweck C, Caiazzo R, Clerquin B et al (2012) Renal origin and size are independent predictors of survival after surgery for adrenal metastasis. Ann Surg Oncol 19(11):3621–3626. https://doi.org/10.1245/s10434-012-2464-6
Howell GM, Carty SE, Armstrong MJ et al (2013) Outcome and prognostic factors after adrenalectomy for patients with distant adrenal metastasis. Ann Surg Oncol 20(11):3491–3496. https://doi.org/10.1245/s10434-013-3050-2
Sarela AI, Murphy I, Coit DG et al (2003) Metastasis to the adrenal gland: the emerging role of laparoscopic surgery. Ann Surg Oncol 10(10):1191–1196
Adler JT, Mack E, Chen H (2007) Equal oncologic results for laparoscopic and open resection of adrenal metastases. J Surg Res 140(2):159–164. https://doi.org/10.1016/j.jss.2006.08.035
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213. https://doi.org/10.1097/01.sla.0000133083.54934.ae
Zini L, Porpiglia F, Fassnacht M (2011) Contemporary management of adrenocortical carcinoma. Eur Urol 60(5):1055–1065. https://doi.org/10.1016/j.eururo.2011.07.062
Glenn JA, Kiernan CM, Yen TW et al (2016) Management of suspected adrenal metastases at 2 academic medical centers. Am J Surg 211(4):664–670. https://doi.org/10.1016/j.amjsurg.2015.11.019
Bradley CT, Strong VE (2014) Surgical management of adrenal metastases. J Surg Oncol 109(1):31–35. https://doi.org/10.1002/jso.23461
Solaini L, Ministrini S, Tomasoni M et al (2015) Adrenalectomy for metastasis: long-term results and predictors of survival. Endocrine 50(1):187–192. https://doi.org/10.1007/s12020-015-0596-8
Frenk NE, Daye D, Tuncali K et al (2018) Local control and survival after image-guided percutaneous ablation of adrenal metastases. J Vasc Interv Radiol 29(2):276–284. https://doi.org/10.1016/j.jvir.2017.07.026
Hasegawa T, Yamakado K, Nakatsuka A et al (2015) Unresectable adrenal metastases: clinical outcomes of radiofrequency ablation. Radiology 277(2):584–593. https://doi.org/10.1148/radiol.2015142029
Gunjur A, Duong C, Ball D et al (2014) Surgical and ablative therapies for the management of adrenal 'oligometastases'—a systematic review. Cancer Treat Rev 40(7):838–846. https://doi.org/10.1016/j.ctrv.2014.04.001
Acknowledgements
We thank the following members for help in collecting the data: Kodai Hattahara (Otsu City Hospital), Takuro Sunada (Kyoto City Hospital), Ryosuke Suzuki (Kobe City Medical Center General Hospital), Yudai Takahashi (Tenri Hospital), and Yuki Kamiyama (Japanese Red Cross Wakayama Medical Center). We also thank Angela Morben, DVM, ELS, from Edanz Group (https://www.edanzediting.com/ac), for editing a draft of this manuscript.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Informed consent
Informed consent was obtained in the form of opt-out on the web-site. Those who rejected were excluded.
Human rights statement
The study was approved by Kyoto university institutional review board (Number R1218). Data use for the present study was approved by the ethical committees of all participating institutions, and a retrospective chart review was conducted.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Goto, T., Inoue, T., Kobayashi, T. et al. Feasibility of laparoscopic adrenalectomy for metastatic adrenal tumors in selected patients: a retrospective multicenter study of Japanese populations. Int J Clin Oncol 25, 126–134 (2020). https://doi.org/10.1007/s10147-019-01533-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10147-019-01533-8