Abstract
Previous studies of the effects of body fat on bone mineral density (BMD) have shown conflicting results depending on the age, sex, and menopausal status of the subjects. The purpose of the present study was to investigate the effects of body fat on BMD using data from the Fifth Korea National Health and Nutrition Examination Survey, which was conducted in 2010. Our data were drawn from a relevant population within the survey’s range of participants, yielding a total of 4,138 subjects aged 20–93 years (1,133 men <50 years, 946 men ≥50 years, 1,332 premenopausal women, and 727 postmenopausal women). Percent body fat, fat mass (FM), lean mass (LM), and BMD of the total hip, lumbar spine, and arm were measured in the original survey using dual-energy X-ray absorptiometry. Relationships between FM and BMD were evaluated using multiple linear regression analyses. LM was the strongest determinant of BMD. Although FM also showed a significant positive correlation, even after adjusting for LM, with total hip and lumbar spine BMD in men ≥50 years and pre- and postmenopausal women, this positive correlation was not observed in the arm. In an analysis of obese and non-obese subgroups, the positive correlation between FM and BMD disappeared in obese subjects. Furthermore, FM and BMD exhibited a negative correlation in premenopausal obese women. These results suggest that FM contributes to BMD primarily through mechanical loading. FM, then, is not beneficial and may be detrimental to bone health in obese people.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Osteoporosis is a serious problem in the elderly because it can lead to osteoporotic fractures, which are associated with increased morbidity and mortality [1]. Previous studies have demonstrated that bone mineral density (BMD) is a better long-term predictor of mortality than either blood pressure or serum cholesterol levels [2, 3]. Many genetic and environmental factors influence BMD and the development of osteoporosis, and body weight is one of these [4]. Because of the adaptive response of bone to mechanical loading, a high body weight is widely considered to be a protective factor in bone health [4].
Many researchers have focused on the effects of body composition, rather than simply body weight, on BMD. Measuring body composition involves breaking down body weight into its two main components, lean mass (LM) and fat mass (FM). In most studies, LM has shown a positive correlation with BMD of the spine, hip, and whole body [5–7]. This positive correlation has been observed regardless of the age, sex, or menopausal status of the subjects. In early studies, FM was also considered to have a beneficial effect on BMD [8]. However, subsequent studies of the effects of FM on BMD have shown conflicting results [7–10]. Some studies have reported that FM is an important determinant of BMD [8]. However, other studies have reported a negative association [7, 9, 10] or no association [11, 12] between FM and BMD.
These discrepancies in the reported effects of FM on BMD can be explained by differences in the study design. In these studies, the populations studied differed in age, sex, and menopausal status [5, 13–17], and most data were drawn from local or regional areas [5, 10, 13] or medical records from healthcare centers [7, 16]. The definition of body fat (percentage or absolute weight) also differed among the studies, and most studies used only a single parameter to measure it [e.g., percent body fat (%BF) or FM] [5, 11, 13, 16]. Moreover, these studies used different adjusted covariables and analytical methods. To resolve the ambiguities created by these variations, our investigation used measures of both %BF and FM at various body sites drawn from data collected in a nationwide survey.
Materials and methods
Data source and study population
The data used in this study were drawn from the Fifth Korea National Health and Nutrition Examination Survey (KNHANES V-1), conducted in 2010 by the Korea Centers for Disease Control and Prevention. KNHANES V-1 was a nationwide, representative survey of 8,958 people in 3,840 households. From this pool of subjects, those aged 20 years or older who had completed surveys on height, body weight, body composition, BMD, serum 25-hydroxyvitamin D levels, menopausal status, smoking, alcohol consumption, and physical activity were selected. Subjects who had a history of stroke, myocardial infarction, angina, rheumatoid arthritis, thyroid disease, renal failure, liver cirrhosis, chronic obstructive pulmonary disease, or bilateral oophorectomy, and subjects who had undergone estrogen treatment for longer than a month, had lost more than 10 kg weight in the preceding year, or had been bedridden for more than 20 days in the preceding month were excluded. We also excluded subjects who had tuberculosis, cancer, or osteoporosis treatment at the time of the survey. Ultimately, data from 4,138 subjects, all of whom provided informed consent, were used. This study was approved by the institutional review board of Seoul National University Hospital (IRB No. E-1207-001-414).
Measurements
In the original survey, height, body weight, and waist circumference were measured using standard protocols. Body mass index (BMI) was calculated as weight in kilograms divided by height in meters squared (kg/m2). Obesity was defined as a BMI greater than 25 kg/m2.
Body composition and BMD were measured using dual-energy X-ray absorptiometry (DXA; DISCOVERY-W fan-beam densitometer; Hologic, Bedford, MA, USA) and standard procedures. FM was defined as whole-body FM. LM was defined as body mass minus FM. Soft lean mass (SLM) was defined as LM minus bone mineral content; in the extremities, SLM indicated muscle mass. Arm BMD was measured in the course of a whole-body scan for body composition analysis. The left arm was selected for analysis. The lumbar spine (L1–L4) and total hip BMD were measured in individual sessions conducted for detecting osteoporosis. Total hip BMD was measured in the left hip except in subjects who had received a left hip implant. Serum 25-hydroxyvitamin D concentration was measured with a radioimmunoassay kit (DiaSorin, Stillwater, MN, USA).
Smoking status, alcohol consumption, and physical activity were assessed with a self-administered questionnaire. Smoking status was divided into two categories, current smoker and non-smoker. High-risk alcohol consumption was defined as consuming seven or more drinks for men or five or more drinks for women on a single occasion at least once a week. Physical activity was evaluated using the Korean version of the International Physical Activity Questionnaire–Short Form (IPAQ-SF), which contains three domains: vigorous activity, moderate-intensity activity, and walking. Vigorous activity is defined as at least 20 min per day of vigorous activity on 3 or more days per week. Moderate-intensity activity is defined as at least 30 min of moderate-intensity activity on 5 or more days per week. Walking is defined as at least 30 min per day of walking on 5 or more days per week. These categorizations were based on the classification scheme in KNHANES V-1.
Statistical analysis
All subjects were categorized into the following groups: men <50 years, men ≥50 years, premenopausal women, and postmenopausal women. The data are presented as mean ± standard deviation for continuous variables and as a percentage for categorical variables. Differences between different age groups of the same sex were compared using Student’s t test for continuous variables. Pearson’s χ2 test was used to compare differences in distribution between categorical variables. Pearson’s correlation analysis was performed between body size or body composition variables and BMD.
Multiple linear regression models were used to evaluate the effects of body fat on BMD. Model 1 included body weight and %BF and model 2 included FM and LM as independent variables for evaluating the effects of pure body fat on BMD. To assess the effects of FM in the presence and absence of obesity, subgroup analyses of obese and non-obese subjects were performed. To evaluate the effects of abdominal obesity on BMD, waist circumference was added to model 2. The effects of the relative distribution of fat and muscle mass on regional BMD were also evaluated. In the arm BMD analysis, arm FM to total FM (AFM/TFM) ratio and arm SLM to total SLM (ASLM/TSLM) ratio were included as independent variables in a multiple linear regression model adjusted by FM and LM. Similarly, leg FM to total FM (LFM/TFM) ratio and leg SLM to total SLM (LSLM/TSLM) ratio were included as independent variables in the total hip BMD analysis. To adjust for other factors affecting BMD, all the aforementioned regression models accounted for age, height, serum 25-hydroxyvitamin D level, smoking status, alcohol consumption status, and physical activity. All the regression analyses used the enter method to compare the effects of body composition on BMD among the various groups and body sites. PASW Statistics version 18 (SPSS, Chicago, IL, USA) was used to conduct all analyses. P < 0.05 was considered statistically significant.
Results
General characteristics
A total of 4,138 subjects, including 1,133 men <50 years, 946 men ≥50 years, 1,332 premenopausal women, and 727 postmenopausal women, were examined in this study. The subjects’ general characteristics are shown in Table 1. Men <50 years exhibited a higher BMI and a greater prevalence of obesity than men ≥50 years, but waist circumference and %BF did not differ between younger and older men. Postmenopausal women showed a higher BMI, prevalence of obesity, waist circumference, and %BF than premenopausal women.
Effects of body composition on BMD
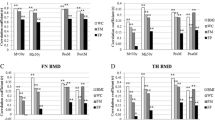
Body weight, %BF, FM, and LM showed significant positive correlations with all areas of BMD except %BF in men <50 years with all areas of BMD and %BF in men ≥50 years with arm BMD (Table 2). Waist circumference also showed significant positive correlations with all areas of BMD except the lumbar spine BMD in men <50 years (Table 2). To evaluate the effects of pure body fat on BMD, %BF was adjusted by body weight (model 1), and FM was adjusted by LM (model 2). In model 1, body weight was positively correlated with BMD in all three sites, and %BF was negatively correlated with BMD in all three sites except the lumbar spine of postmenopausal women (Table 3). In model 2, which included FM and LM instead of body weight and %BF, FM exhibited a positive correlation with total hip and lumbar spine BMD in men ≥50 years and pre- and postmenopausal women (Table 3). When controlled for LM, FM showed the strongest correlation with lumbar spine BMD in premenopausal women (β = 0.147, P < 0.001). LM, on the other hand, exhibited a positive correlation with BMD in all three sites, and the β-values for LM were stronger than for FM. Additional data about the correlation of adjusted covariables such as age and height with BMD are given in Supplementary Table 1 (Online Resource 1).
Different effects of FM on BMD depending on the presence of obesity
Subgroup analyses evaluated whether the effects of FM are different in obese and non-obese subjects (Table 4). In non-obese subjects, FM was positively correlated with total hip and lumbar spine BMD in men ≥50 years and both pre- and postmenopausal women. However, no significant correlation between FM and total hip or lumbar spine BMD was observed in obese subjects of any age or either sex. For arm BMD, there was no significant correlation between FM and BMD, except for the negative correlation in premenopausal obese women (β = −0.186, P < 0.01).
Effects of abdominal obesity on BMD
Waist circumference, a surrogate marker of abdominal obesity, was positively correlated with BMD (Table 2). However, when controlled for FM and LM, the correlation between waist circumference and BMD was not observed, except for a positive correlation with arm BMD in premenopausal women (β = 0.163, P < 0.01) (Table 5). In a subgroup analysis according to the presence of obesity, this positive correlation was maintained only in the non-obese premenopausal women (β = 0.133, P < 0.01); no significant correlation between waist circumference and BMD was observed in obese premenopausal women (data not shown in table).
Regional effects of fat on BMD
The correlations between regional FM and muscle mass and regional BMD are shown in Table 6. The LSLM/TSLM ratio exhibited a positive correlation with total hip BMD in all men and postmenopausal women. The ASLM/TSLM ratio displayed a positive correlation with arm BMD in all men and premenopausal women. However, neither the LFM/TFM nor the AFM/TFM ratio showed an association with any area of BMD except arm BMD in premenopausal women, which showed a negative correlation with the AFM/TFM ratio (β = −0.064, P < 0.01).
Discussion
The effects of body composition on BMD were investigated using data from a well-organized, large-scale nationwide survey that included representative groups of different ages and sexes. In this study, LM was the strongest determinant of BMD. Although FM also showed a significant positive association with BMD, even after adjusting for LM, this relationship was not observed in the arm, a non-weight-bearing site, and it was attenuated in obese subjects. The local effects of FM were also not observed when evaluated with the LSFM/TSFM or ASFM/TSFM ratio.
Conflicting results have been reported in the literature on the effects of body fat on BMD [7–10]. These conflicts may have been caused by the different definitions of body fat in the studies. In previous studies, %BF showed a negative correlation with BMD [18, 19], whereas FM showed inconsistent correlations with BMD [5, 7, 9, 10]. We evaluated both of two different variables, %BF and FM, in the same subjects. In our two different multiple linear regression models, these variables displayed opposite patterns of correlation; %BF was correlated negatively and FM positively with BMD. This discrepancy may be caused by the stronger relationship of BMD with LM than with FM [20]. For a given body weight, an increase in %BF indicates not only an increase of FM but also a decrease of LM, which can lead to a decrease in BMD. Therefore, a negative association between %BF and BMD does not necessarily mean that body fat exerts a negative effect on BMD. In addition, it suggests that FM, the absolute weight of body fat, may be a more appropriate parameter for evaluating the independent effects of body fat on BMD.
In this study, FM was positively correlated with BMD, and this correlation was maintained in men ≥50 years and pre- and postmenopausal women after adjusting for LM. FM is believed to contribute differently to BMD according to age and sex [5, 13–17]. It appears that FM is a more important determinant of BMD in women than in men [13–15] and in older people than in younger people [5, 16, 17]. These results are consistent with our data. Several hypotheses have been suggested to account for the beneficial effects of FM on BMD. First, FM can contribute to BMD by increasing mechanical loading on the bone. In this study, although FM showed a significant association with BMD in the lumbar spine and total hip, it showed no such association with BMD in the arm. Because the arm is a minimally weight-bearing site, these results support that mechanical loading is a major contributing factor to the effects of FM on BMD. Second, FM can have a role as an important source of estrogen in postmenopausal women. Adipose tissues can synthesize estrogens from androgen precursors. Increased estrogen production by adipose tissues is believed to have a protective effect on BMD, especially in postmenopausal women [14, 21]. It is also reported that in postmenopausal women, FM was positively correlated with BMD, but hormone replacement therapy attenuated the effect of FM on BMD [22]. Last, insulin and insulin-like growth factor 1 have been proposed as mediators of the effects of FM on bone [23, 24].
In this study, the positive associations between FM and BMD disappeared in obese subjects whereas LM consistently showed a positive association with BMD (Table 3). In contrast, in obese premenopausal women, FM was negatively correlated with arm BMD (Table 4). These results are consistent with recent studies reporting a higher risk of humerus fracture in obese postmenopausal women [25, 26]. This observation suggests that obesity might not be protective against osteoporosis and fracture. One possible explanation for the harmful effects of body fat is that adipose tissues secrete adipokines such as adiponectin and leptin [27]. Negative correlations between serum adiponectin or leptin levels and BMD have been reported [28–30], and it may be speculated that adipokines counterbalance the mechanically beneficial effects of FM on BMD in obese people.
The effects of FM on BMD can differ depending on the type and location of the FM. Although most previous studies have reported a negative association between visceral fat and BMD [31, 32], some studies have reported a positive association [32, 33]. In this study, abdominal obesity was evaluated using waist circumference. Waist circumference cannot distinguish visceral fat from subcutaneous fat. Therefore, when controlled for total FM, waist circumference was not correlated with BMD except arm BMD in premenopausal women. In premenopausal women, waist circumference was positively correlated with arm BMD. Because visceral fat is relatively low in premenopausal women [34, 35], waist circumference cannot accurately represent it in this group, in contrast to men and postmenopausal women [36].
LM and FM may have systemic or local effects on BMD. To ascertain the local effects of muscle and fat on BMD, a task that few studies have attempted, regional LM and FM ratios were calculated. LSLM/TSLM and ASLM/TSLM ratios were positively correlated with total hip and arm BMD, respectively, but the LFM/TFM and AFM/TFM ratios did not show any significant correlations except in premenopausal women. This result means that muscle may have a local beneficial effect but fat does not. Muscle contraction may provide an explanation for the local effects of muscle on bone. Because larger muscles exert higher tensile forces on the bones to which they are attached, regional muscle can increase regional BMD [37]. This explanation is supported by the positive correlation between muscle strength and BMD in both upper and lower limbs [38–40]. This regional effect of LM explains the stronger correlation of LM and BMD compared to FM and BMD.
There are a few limitations to the present study. Because it is cross-sectional, it cannot establish a causal relationship between FM and BMD. Other factors such as nutrition and occupation, not included in this study, also influence BMD. In addition, waist circumference is a relatively imprecise indicator of visceral fat compared to measurements made by computed tomography (CT) or magnetic resonance imaging (MRI) scans.
In conclusion, this study demonstrated a positive correlation between FM and total hip and lumbar spine BMD in men ≥50 years and pre- and postmenopausal women. However, a positive relationship between FM and BMD was not observed in obese subjects. FM was not associated with arm BMD in men or premenopausal women. Furthermore, FM showed a negative association with arm BMD in obese premenopausal women. These results suggest that FM contributes to BMD mainly by means of mechanical loading, and that FM is not beneficial and might be harmful to bone health in obese individuals.
References
Sambrook P, Cooper C (2006) Osteoporosis. Lancet 367:2010–2018
Johansson C, Black D, Johnell O, Oden A, Mellstrom D (1998) Bone mineral density is a predictor of survival. Calcif Tissue Int 63:190–196
von der Recke P, Hansen MA, Hassager C (1999) The association between low bone mass at the menopause and cardiovascular mortality. Am J Med 106:273–278
Felson DT, Zhang Y, Hannan MT, Anderson JJ (1993) Effects of weight and body mass index on bone mineral density in men and women: the Framingham study. J Bone Miner Res 8:567–573
Cui LH, Shin MH, Kweon SS, Park KS, Lee YH, Chung EK, Nam HS, Choi JS (2007) Relative contribution of body composition to bone mineral density at different sites in men and women of South Korea. J Bone Miner Metab 25:165–171
Ho-Pham LT, Nguyen ND, Lai TQ, Nguyen TV (2010) Contributions of lean mass and fat mass to bone mineral density: a study in postmenopausal women. BMC Musculoskelet Disord 11:59
Kim CJ, Oh KW, Rhee EJ, Kim KH, Jo SK, Jung CH, Won JC, Park CY, Lee WY, Park SW, Kim SW (2009) Relationship between body composition and bone mineral density (BMD) in perimenopausal Korean women. Clin Endocrinol (Oxf) 71:18–26
Reid IR, Ames R, Evans MC, Sharpe S, Gamble G, France JT, Lim TM, Cundy TF (1992) Determinants of total body and regional bone mineral density in normal postmenopausal women—a key role for fat mass. J Clin Endocrinol Metab 75:45–51
Zhao LJ, Liu YJ, Liu PY, Hamilton J, Recker RR, Deng HW (2007) Relationship of obesity with osteoporosis. J Clin Endocrinol Metab 92:1640–1646
Kim JH, Choi HJ, Kim MJ, Shin CS, Cho NH (2011) Fat mass is negatively associated with bone mineral content in Koreans. Osteoporos Int 23:2009–2016
Liu JM, Zhao HY, Ning G, Zhao YJ, Zhang LZ, Sun LH, Xu MY, Chen JL (2004) Relationship between body composition and bone mineral density in healthy young and premenopausal Chinese women. Osteoporos Int 15:238–242
Leslie WD, Weiler HA, Lix LM, Nyomba BL (2008) Body composition and bone density in Canadian White and Aboriginal women: the First Nations Bone Health Study. Bone (NY) 42:990–995
Lim S, Joung H, Shin CS, Lee HK, Kim KS, Shin EK, Kim HY, Lim MK, Cho SI (2004) Body composition changes with age have gender-specific impacts on bone mineral density. Bone (NY) 35:792–798
Douchi T, Oki T, Nakamura S, Ijuin H, Yamamoto S, Nagata Y (1997) The effect of body composition on bone density in pre- and postmenopausal women. Maturitas 27:55–60
Reid IR, Plank LD, Evans MC (1992) Fat mass is an important determinant of whole body bone density in premenopausal women but not in men. J Clin Endocrinol Metab 75:779–782
Ijuin M, Douchi T, Matsuo T, Yamamoto S, Uto H, Nagata Y (2002) Difference in the effects of body composition on bone mineral density between pre- and postmenopausal women. Maturitas 43:239–244
Cheng Q, Zhu YX, Zhang MX, Li LH, Du PY, Zhu MH (2012) Age and sex effects on the association between body composition and bone mineral density in healthy Chinese men and women. Menopause 19:448–455
Yoo HJ, Park MS, Yang SJ, Kim TN, Lim KI, Kang HJ, Song W, Baik SH, Choi DS, Choi KM (2012) The differential relationship between fat mass and bone mineral density by gender and menopausal status. J Bone Miner Metab 30:47–53
Lu H, Fu X, Ma X, Wu Z, He W, Wang Z, Allison DB, Heymsfield SB, Zhu S (2011) Relationships of percent body fat and percent trunk fat with bone mineral density among Chinese, black, and white subjects. Osteoporos Int 22:3029–3035
Gjesdal CG, Halse JI, Eide GE, Brun JG, Tell GS (2008) Impact of lean mass and fat mass on bone mineral density: the Hordaland Health Study. Maturitas 59:191–200
Reid IR (2002) Relationships among body mass, its components, and bone. Bone (NY) 31:547–555
Jensen LB, Vestergaard P, Hermann AP, Gram J, Eiken P, Abrahamsen B, Brot C, Kolthoff N, Sorensen OH, Beck-Nielsen H, Nielsen SP, Charles P, Mosekilde L (2003) Hormone replacement therapy dissociates fat mass and bone mass, and tends to reduce weight gain in early postmenopausal women: a randomized controlled 5-year clinical trial of the Danish Osteoporosis Prevention Study. J Bone Miner Res 18:333–342
Reid IR, Evans MC, Cooper GJ, Ames RW, Stapleton J (1993) Circulating insulin levels are related to bone density in normal postmenopausal women. Am J Physiol 265:E655–E659
Bredella MA, Torriani M, Ghomi RH, Thomas BJ, Brick DJ, Gerweck AV, Harrington LM, Breggia A, Rosen CJ, Miller KK (2011) Determinants of bone mineral density in obese premenopausal women. Bone (NY) 48:748–754
Prieto-Alhambra D, Premaor MO, Fina Aviles F, Hermosilla E, Martinez-Laguna D, Carbonell-Abella C, Nogues X, Compston JE, Diez-Perez A (2012) The association between fracture and obesity is site-dependent: a population-based study in postmenopausal women. J Bone Miner Res 27:294–300
Gnudi S, Sitta E, Lisi L (2009) Relationship of body mass index with main limb fragility fractures in postmenopausal women. J Bone Miner Metab 27:479–484
Biver E, Salliot C, Combescure C, Gossec L, Hardouin P, Legroux-Gerot I, Cortet B (2011) Influence of adipokines and ghrelin on bone mineral density and fracture risk: a systematic review and meta-analysis. J Clin Endocrinol Metab 96:2703–2713
Lenchik L, Register TC, Hsu FC, Lohman K, Nicklas BJ, Freedman BI, Langefeld CD, Carr JJ, Bowden DW (2003) Adiponectin as a novel determinant of bone mineral density and visceral fat. Bone (NY) 33:646–651
Blum M, Harris SS, Must A, Naumova EN, Phillips SM, Rand WM, Dawson-Hughes B (2003) Leptin, body composition and bone mineral density in premenopausal women. Calcif Tissue Int 73:27–32
Wu N, Wang QP, Li H, Wu XP, Sun ZQ, Luo XH (2010) Relationships between serum adiponectin, leptin concentrations and bone mineral density, and bone biochemical markers in Chinese women. Clin Chim Acta 411:771–775
Gilsanz V, Chalfant J, Mo AO, Lee DC, Dorey FJ, Mittelman SD (2009) Reciprocal relations of subcutaneous and visceral fat to bone structure and strength. J Clin Endocrinol Metab 94:3387–3393
Sheu Y, Cauley JA (2011) The role of bone marrow and visceral fat on bone metabolism. Curr Osteoporos Rep 9:67–75
Warming L, Ravn P, Christiansen C (2003) Visceral fat is more important than peripheral fat for endometrial thickness and bone mass in healthy postmenopausal women. Am J Obstet Gynecol 188:349–353
Abdulnour J, Doucet E, Brochu M, Lavoie JM, Strychar I, Rabasa-Lhoret R, Prud’homme D (2012) The effect of the menopausal transition on body composition and cardiometabolic risk factors: a Montreal-Ottawa New Emerging Team group study. Menopause 19:760–767
Stevens J, Katz EG, Huxley RR (2010) Associations between gender, age and waist circumference. Eur J Clin Nutr 64:6–15
Kim HI, Kim JT, Yu SH, Kwak SH, Jang HC, Park KS, Kim SY, Lee HK, Cho YM (2011) Gender differences in diagnostic values of visceral fat area and waist circumference for predicting metabolic syndrome in Koreans. J Korean Med Sci 26:906–913
Schoenau E (2005) From mechanostat theory to development of the “Functional Muscle-Bone-Unit”. J Musculoskelet Neuronal Interact 5:232–238
Blain H, Vuillemin A, Teissier A, Hanesse B, Guillemin F, Jeandel C (2001) Influence of muscle strength and body weight and composition on regional bone mineral density in healthy women aged 60 years and over. Gerontology 47:207–212
Di Monaco M, Di Monaco R, Manca M, Cavanna A (2000) Handgrip strength is an independent predictor of distal radius bone mineral density in postmenopausal women. Clin Rheumatol 19:473–476
Frank AW, Lorbergs AL, Chilibeck PD, Farthing JP, Kontulainen SA (2010) Muscle cross sectional area and grip torque contraction types are similarly related to pQCT derived bone strength indices in the radii of older healthy adults. J Musculoskelet Neuronal Interact 10:136–141
Acknowledgments
This research was supported by the Bio & Medical Technology Development Program of the National Research Foundation (NRF) funded by the Korean government (MEST) (No. 2011-0030135).
Conflict of interest
All authors have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Kim, W., Chung, S.G., Kim, K. et al. The relationship between body fat and bone mineral density in Korean men and women. J Bone Miner Metab 32, 709–717 (2014). https://doi.org/10.1007/s00774-013-0545-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-013-0545-7