Abstract
Aims
LncRNA SNHG16 and Toll-like receptor-4 (TLR4) participate in diabetes nephropathy. This study investigated whether SNHG16 regulates diabetic renal injury (DRI) via TLR4 and its related mechanism.
Methods
Diabetic mice and high glucose (HG)-induced HRMCs were used to examine the expressions of SNHG16 and TLR4. The SNHG16 expression, cytokines, reactive oxygen species, MDA, SOD, GSH, and fibrosis-related proteins were evaluated in HG-induced HRMCs transfected with sh-NC or sh-SHNG16. RNA immunoprecipitation and RNA pull-down determined the interaction between SNHG16 and EIF4A3 or TLR4 and EIF4A3. We used HG-treated HRMCs or diabetic mice to investigate the roles of TLR4 or SNHG16 in renal injuries.
Results
Both SNHG16 and TLR4 were upregulated in diabetic conditions. HG increased serum Scr and BUN, led to significant fibrosis, increased inflammation- and renal fibrosis-related proteins in mice, and increased ROS, MDA, and decreased SOD and GSH in HRMCs. SNHG16 silencing diminished HG-upregulated SNHG16, decreased HG-increased cytokines secretion, ROS, MDA, and fibrosis but increased SOD and GSH. RIP and RNA pull-down confirmed that SNHG16 recruits EIF4A3 to stabilize TLR4 mRNA. TLR4 knockdown alleviated HG-induced renal injuries by suppressing RAS and NF-κB-mediated activation of NLRP3 inflammasomes. SNHG16 knockdown alleviated HG-induced renal injuries in HG-induced HRMCs or diabetic mice. Interestingly, TLR4 overexpression reversed the effects of SNHG16 knockdown. Mechanistically, SNHG16 knockdown alleviated HG-induced renal injuries by suppressing TLR4.
Conclusion
SNHG16 accelerated HG-induced renal injuries via recruiting EIF4A3 to enhance the stabilization of TLR4 mRNA. The SNGHG16/ELF4A3/TLR4 axis might be a novel target for treating DRI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetic nephropathy (DN) is a severe complication of diabetes which can lead to end-stage renal deficits [22]. Due to the complex diabetic conditions, the rising frequency of DN is one of the most reasons for diabetes-related kidney failures [23]. Although the exact reason for DN remains unknown, diabetes-related high blood glucose and pressure are among the factors contributing to DN, characterized by hyperfiltration, increased albuminuria, and subsequent cell apoptosis and functional defects in podocytes [26]. Recent studies also showed that high glucose could induce high levels of cytokines, such as IL-8, in patients with T2D, which might induce podocyte damage by activating IL-8/CXCR1/2 [17]. Although many effective interventions are applied in controlling DN progressions, such as blockers for the renin–angiotensin–aldosterone system (RAAS), blood pressure and glucose management, and avoiding alcohol drinking and smoking [26]. Developing a new therapeutic method for DN remains challenging to reduce the high frequency of DN.
Several long non-coding RNA (lncRNA) is involved in the pathophysiological progression of DN. For instance, lncRNA MALAT1 induced podocyte apoptosis [35]. LncRNA lnc-ISG20 enhanced renal fibrosis via miR-486-5p/NFAT5 during the development of diabetic nephropathy [4]. LncRNAs such as PVT1 [30] and NEAT1 ([29] regulate oxidative stress, inflammation, and accumulation of extracellular matrix in DN. Similarly, the previous studies showed that LncRNA SNHG16 contributed to HG-induced podocyte injuries and fibrogenesis [10, 12]. A recent experiment showed that overexpression of SNHG16 in retinal endothelial cells facilitated the endothelial cell proliferation, migration, and angiogenesis via the activation of NF-κB and PI3K/AKT pathways [2]. Additional studies pointed out that SNHG16 aggravates DN through damaging cellular proliferation [10] and enhanced fibrogenesis [12]. However, there are fewer reports related to inflammation. Therefore, more investigations are required to uncover the functions of SNHG16 in HG-induced inflammatory response in DN.
Toll-like receptors (TLRs) are widely expressed in human tissues, including the kidney. A previous study found that TLR1/2/3/4 and TLR6 are mainly expressed in human kidneys’ tubular epithelial cells and mesangial cells [1]. During renal injuries and overexpression of TLR4, a significant member of the TLR family was found in the population of penetrating inflammatory cells [1]. In the renal parenchymal cells, the activated TLR4 could activate the p38 MAPK pathway to synthesize inflammatory cytokines, including TNF-α and interleukin (IL)-1β and -6 [33]. The activation of TLR4/NF-κB signaling plays a critical role in LPS-induced inflammatory response in cardiomyocytes via increasing the NLRP3 inflammasome [25]. Besides inflammatory response, NLRP3 also regulates Th cell fate. Its dysregulation is closely related with the poor outcomes in cardiac-transplanted patients [3]. Therefore, the investigation of NRLP3 signaling is a novel target to delineate how inflammation influences the progression of diabetic kidney diseases. Although the molecular mechanisms of TLR4 in DN remain elusive, the previous studies showed that TLR4 activation significantly contributed to the progression of diabetic inflammation and renal fibrosis in DN [7, 15]. The renin-angiotensin system (RAS) controls blood pressure and body fluid homeostasis [9]. A recent study indicated that RAS blocking improved the outcome of DN-related renal injuries [16]. Increasing evidence showed that RAS upregulated TLR4 to activate the p38 MAPK signaling pathway to produce proinflammatory cytokines, chemokines, and profibrotic elements in DN. As feedback, the TLR4/MAPK pathway also induces the expression of RAS molecules, including angiotensin-converting enzyme (ACE) and angiotensin (AT). However, the definite mechanism related to the interaction of RAS and TLR4 is still unknown.
EIF4A3 is an RNA helicase of the DEAD-box family, which can control mRNA splicing, supervise mRNA quality and regulate RNA metabolism [32]. Evidence showed that EIF4A3 contributed to the pathological progression of diseases like HPV-related cancer [19] and circRNA circPPMIF-modulated M1 macrophage activation in type 1 diabetes mellitus [34]. The online bioinformatics analysis found that TLR4 interacted with eukaryotic initiation factor 4A-III (EIF4A3), which lncRNAs can recruit to stabilize proteins and participate in RNA splicing and influencing the downstream cascades [32]. Therefore, we hypothesized that the interaction between TLR4 and EIF4A3 possibly participated in the pathological progression of DN.
This study aimed to disclose whether SNHG16 contributed to HG-induced renal injuries by EIF4A3-mediated TLR4 expression and the involved potential mechanism.
Materials and methods
Animals
We purchased C57BL6 background db/db male mice and their littermates (db/m) from the Animal Center at the Dongguan Tung wah Hospital, Guangdong, China. The db/db mice comprised five groups: db/db group (n = 8), db/db + sh-NC(n = 8, db/db mice receiving sh-NC lentivirus), db/db + sh-SNHG16 (n = 8, db/db mice receiving sh-SNHG16 lentivirus), db/db + sh-SNHG16 + oe-NC (n = 8, db/db mice receiving sh-SNHG16 lentivirus and pcDNA3.1 empty vector), and db/db + sh-SNHG16 + oe-TLR4 (n = 8, db/db mice receiving sh-SNHG16 lentivirus and pcDNA3.1-TLR4 vector), The control group db/m mice received the injection of sterile 1 × phosphate-buffered saline (PBS). All mice were housed in environment-controlled conditions and had free access to water and food. All experiments were supervised by the Animal Welfare and Ethics Committee of Dongguan Tung wah Hospital. (1) Mice were sacrificed at 12-week to evaluate the expressions of SNHG16 and TLR4 under diabetic conditions. (2) The mice subjected to vehicle control, sh-NC, sh-SNHG16, sh-SNHG16 + pcDNA3.1 vector, or sh-SNHG16 + pcDNA3.1-TLR4 at the age of 12 weeks were killed at the end of 20 weeks. After sacrifice, the kidneys and blood were sampled for further analysis.
Measurement of blood glucose
Blood was sampled from the caudal vein puncture of mice on days 6, 10, 14, 18, and 22. The blood glucose was monitored using veterinary blood glucose meters (DSI, MN, USA).
Biochemical analysis
To determine the characteristics of the diabetic animals, we used an automatic IDEXX VetTest biochemical analyzer (IDEXX Laboratories, Maine, USA) to measure urine proteins and blood serine creatinine (Scr) and urea nitrogen (BUN).
Hematoxylin–eosin (H&E) and periodic acid-Schiff (PAS) staining
After harvesting from each mouse, the kidneys were fixed in 4% paraformaldehyde (PFA). After embedding in paraffin, the kidneys were sectioned into 5 µm thickness. H and E were performed using a commercial kit (H-3502, Vector Laboratories, Inc, CA, USA). Periodic acid-Schiff staining was conducted according to the detailed protocol of the PAS stain kit (C0142M, Beyotime Biotechnology, Shanghai, China). After counterstaining, acidic ethanol differentiation, permeabilization, and the slides were imaged using an OMAX Binocular Laboratory Compound Microscope (M82EZ-C50S, AmScope, CA, USA).
Human renal mesangial cells
Human renal mesangial cells (HRMCs) were purchased from creative bioarray (CSC-7719W, NY, USA). HRMCs were maintained in RMPI-1640 medium plus 10% fetal bovine serum (FBS-02-0050, Rockland Immunochemicals, Inc, NJ, USA) and 1 × penicillin-streptomycin solution (15140122, Thermo Fisher Scientific, CA, USA) at 37 °C 5% CO2 incubator. Cells were used between the 3rd and 7th passages in low serum (2% FBS) after serum starvation for 24 h.
Cell transfection
We purchased commercial constructs including pLKO.1 empty vector (sh-NC), pLKO.1-SNHG16 (sh-SNHG16 (#1, #2, and #3), pcDNA3.1 empty vector, pcDNA3.1-SNHG16, pcDNA3.1-TLR4, pSuper.puro empty vector (si-NC), pSuper.puro-NC vector (si-NC), pSuper.puro-EIF4A3 vector (si-EIF4A3) from Sangon Biotech Co., Ltd. (Shanghai, China). For lentivirus production, LV-MAX™ Lentiviral Production System (A35684) was purchased from Thermo Fisher Scientific, CA, USA. pcDNA3.1 vector was transfected into HRMCs using Lipofectamine 3000 reagent.
qRT-PCR
Total RNAs were isolated from mice kidney tissues or HRMCs using TRIzol reagent (15596026, Thermo Fisher Scientific, CA, USA). To study the effect of SNHG16 on TLR4 mRNA stability, we treated RNA extraction with actinomycin D (10 µg/mL), then collected samples at 0, 3, 6, 9, 12 h. After RNA extraction, we detected the levels of TLR4 mRNA. 500 ng of total RNAs were reversely transcripted into cDNA. The used primers listed in Table 1 were synthesized by Generay Biotech Co Ltd (Guangzhou, China). β-actin and U6 snRNA were the internal controls of the TLR4 gene and the SNHG16, respectively. The levels of genes were quantitatively analyzed by the 2−ΔΔCT method.
Measurement of ROS, MDA, SOD, and GSH
We purchased OxiSelect Intracellular ROS Assay Kit from Amyjet Scientific Inc (Green Fluorescence, KA4074, Wuhan, Hubei, China), and Lipid Peroxidation (MDA) Assay Kit (S0131S), Total Superoxide Dismutase Assay Kit (S0088), Total Glutathione Assay Kit (S0052) from Beyotime Biotechnology (Shanghai, China). To detect ROS, we incubated cells with 2′, 7′-Dichlorodihydrofluorescin diacetate (DCFH-DA) at a final concentration of 100 µM DCFH-DA for 60 min at 37 °C. After removing the solution, cells were washed with 1 × DPBS 3 times. Subsequently, cells were imaged by an inverted OM900-T microscope (AmScope, CA, USA). For the measurement of MDA, SOD, and GSH, the serums from the whole blood of mice were separated, or the cells were harvested with cell lysate. Then, the concentrations of MDA, SOD, and GSH were detected according to the detailed protocols from the commercial kits.
ELISA assay
We detect tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), interleukin-1β (IL-1β), and interleukin-8 (IL-8) in the serum from mice or cell culture media to using the commercial enzyme-linked immunosorbent assay (ELISA) kit purchased from Beyotime Biotechnology (Guangzhou, Guangdong, China).
RNA immunoprecipitation (RIP) assays
To determine whether lncRNA SNHG16 or TLR4 could interact with EIF4A3, we performed the RNA immunoprecipitation using an EZ-Magna RNA-binding protein immunoprecipitation kit (17–700, Sigma-Aldrich, MO, USA) according to the manufacture’s instruction. The antibodies for RIP assays were rabbit anti-EIF4A3 antibody (ab32485) and rabbit anti-human IgG (ab6715) from Abcam (MA, USA).
RNA pull-down
We performed the RNA pull-down assay using a magnetic RNA-protein pull-down kit (20164, Thermo Scientific™, CA, USA). Briefly, HRMCs were transfected with biotinylated-SNHG16, -TLR4, or -control. After 48 h, the extracted total RNA was incubated with 100 nmol streptavidin magnetic beads. The bound RNAs were extracted using TRIzol reagent. The levels of EIF4A3 were detected by qRT-PCR.
Western blot
Total proteins were extracted after the different groups of treatments by RIPA buffer (Beyotime, Nantong, China) supplemented with 100 µM phenylmethylsulfonyl fluoride (PMSF) (P0013C, Beyotime Biotechnology, Nantong, China). The protein concentrations were measured using a bicinchoninic acid (BCA) assay kit (Beyotime Biotechnology, Nantong, China). Twenty micrograms of total proteins (20 µg) were dissolved on 12% SDS-PAGE gel and transferred to nitrocellulose membrane (FFN03, Beyotime, Nantong, China). After blocking, the individual primary antibody was applied. The used primary antibodies were mouse anti-TLR4 (ab22048, CA, USA), rabbit anti-collagen I (ab260043, CA, USA), rabbit anti- CTGF (ab6992, CA, USA), rabbit anti-fibronectin (ab2413, CA, USA), rabbit anti-TGF beta 1 (ab215715, CA, USA), rabbit anti-ACE2 (ab108252, CA, USA), rabbit anti-angiotensin II (Ang II, ab239995, CA, USA), rabbit anti-angiotensin II Type 1 (ab239995, CA, USA),rabbit anti-NF-kB p65 (phosphor-S536) (ab76302, CA, USA), rabbit anti- NF-kB p65 [E379] (ab32536, CA, USA), rabbit anti- NLRP3 [EPR23094-1] (ab263899, CA, USA), rabbit anti-cleaved-caspase1 (89332, Cell Signaling Technology, Inc, MA, USA), and rabbit anti-GAPDH [EPR16891] (ab181602, CA, USA). After three washes by 1 × TBST, the membranes were incubated with goat anti-rabbit IgG H&L (HRP) (ab7090, Abcam, MA, USA) and goat anti-mouse IgG H and L (DyLight® 488) (ab96879, Abcam, MA, USA). After three washes by 1 × TBST, the membranes were visualized using an ECL Substrate Kit (ab133409, Abcam, MA, USA). The protein bands were analyzed with ImageJ 1.58 (NIH, USA).
Statistical analysis
All data were expressed as means ± standard deviation (Mean ± SD). The unpaired student t test was used to analyze the difference between the two groups using SPSS 20.0 (SPSS, Chicago, IL, USA). One-way ANOVA analyzed the differences among multiple groups. P < 0.05 was considered statistically significant.
Results
SNHG16 and TLR4 expressions are upregulated in the diabetic kidney
To test the roles of SNHG16 and TLR4 in the development of diabetic kidneys, we developed the db/m and db/db mice. The db/db mice exhibited significantly higher blood glucose (Fig. 1A), urinary protein, (Fig. 1B), and blood Scr and BUN (Fig. 1C) compared with those of db/m mice. H and E and PAS staining showed db/db mice exhibited increased tubular and interstitial fibrosis, hyperplasia of the mesangial matrix, and thickness of basement membrane relative to those of db/m mice (Fig. 1D). Meanwhile, db/db mice exhibited enhanced inflammatory cytokines (Fig. 1E), upregulated levels of SNHG16 and TLR4 mRNA (Fig. 1F), as well as protein levels of TLR4, collagen 1, CTGF, FN, and TGF-β1 (Fig. 1G). All these data suggested that the expression of SNHG16 and TLR4 might participate in the progression of diabetic nephropathy.
SNHG16 and TLR4 expressions are upregulated in the diabetic kidney. Blood samples were collected on day 6, 10, 14, 18, and 22 from the caudal vein puncture of db/m or db/db mice. Then, an automatic biochemical analyzer measured blood glucose levels (A), protein content in mice urine (B), serine creatinine, and BUN (C). Renal tissues were examined by HE and PAS staining (D). The levels of cytokines in sera were detected by ELISA©. The gene expressions of SNHG16 and TLR4 in the rental tissues were measured by qRT-PCR (F). The protein levels of TLR4, collagen 1, CTGF, FN, and TGF-β1 in renal tissues were detected by western blot (G). Data are representative of three independent experiments. *P < 0.05, **P < 0.01, ***P < 0.001 versus the corresponding control
High glucose upregulates the expressions of SNHG16 and TLR4 in human renal mesangial cells (HRMCs)
Subsequently, we developed the in vitro human diabetic nephropathy models using HRMCs after exposure to HG. Our results revealed that HRMCs after exposure to HG exhibited the increased secretions of cytokines (Fig. 2A), remarkably increased production of reactive oxygen species (ROS) (Fig. 2B), and lipid peroxidation MDA level (Fig. 2C), decreased levels of SOD and GSH (Fig. 2C). Additionally, we found that HG exposure induced higher protein levels of collagen 1, CTGF, FN, TGF-β1 (Fig. 2D), and TLR4 (Fig. 2F), as well as the mRNA levels of SNHG16 and TLR4 (Fig. 2E) relative to control. HG exposure led to the increased expressions of SNHG16 and TLR4.
High glucose upregulates the expressions of SNHG16 and TLR4 in human renal mesangial cells (HRMCs). Human Renal Mesangial Cells (HRMCs) were treated with normal or high glucose for 24 h. The levels of cytokines were measured by ELISA assay (A). ROS production was detected in the normal or high glucose-treated HRMCs (B). The synthesis of malondialdehyde, superoxide dismutase, and glutathione were detected using commercial kits (C). The protein levels of collagen 1, connective tissue growth factor (CTGF), fibronectin (FN), and transforming growth factor-beta 1 (TGF-β1) were detected by western blot (D). SNHG16 and TLR4 mRNA levels were detected by qRT-PCR (E). TLR4 protein level was detected by western blot (F). *P < 0.05, **P < 0.01, ***P < 0.001 versus the corresponding control. Data are representative of three independent experiments
Knockdown of SNHG16 alleviates HG-induced injuries in HRMCs
Subsequently, we transfected an SNHG16 knockdown plasmid (pLKO.1-SNHG16) or its parallel pLKO.1 empty vector into HRMCs with or without HG-treatment and investigated the effect of SNHG16 on the pathological conditions of HRMCs. Our data showed that SNHG16 was significantly decreased after transfection of pLKO.1-SNHG16 compared with that of the empty vector group (Fig. 3A). Further experiments showed that the expression of SNHG16 was significantly upregulated after HG exposure, while knockdown of SNHG16 remarkably downregulated SNHG16 in HG-treated HRMCs (Fig. 3B). Subsequent assays indicated that HG exposure induced high levels of inflammatory cytokines (Fig. 3C), leading to the increased accumulation of reactive oxygen species (ROS) (Fig. 3D) and MDA (Fig. 3E) and decreased expressions of SOD and GSH (Fig. 3E). In contrast, knockdown of SNHG16 reversed the effects of HG in HRMCs (Fig. 3C–E). Further western blot assay revealed that HG caused a high level of renal fibrosis-related proteins (collagen 1, CTGF, FN, and TGF-β1), SNHG16 knockdown reversed the effects of HG (Fig. 3F). All those results suggested that knockdown of SNHG16 alleviated HG-induced injuries in HRMCs.
Knockdown of SNHG16 alleviates high glucose-induced injuries in HRMCs. HRMCs were transfected with shNC or shSNHG16 #1/#2/#3 for 48 h. Then, cells were treated with normal or high glucose for 24 h. The transfection efficiency was measured by qRT-PCR (A). SNHG16 mRNA levels were measured in each group by qRT-PCR (B). The cytokines were measured by ELISA (C). ROS levels were evaluated by DCFDA staining using a ROS detection assay kit (D). The levels of malondialdehyde (MDA), superoxide dismutase (SOD), and glutathione (GSH) were measured using the commercial kits (E). The protein levels of collagen 1, CTGF, FN, and TGF-β1 were detected by western blot (F). *P < 0.05, **P < 0.01, ***P < 0.001 versus the corresponding control. Data are representative of three independent experiments
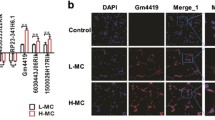
SNHG16 stabilizes TLR4 mRNA by recruiting EIF4A3
We performed the RIP and RNA pull-down assays to find out the potential downstream molecules of SNGH16. The RIP assay showed that both SNHG16 and TLR4 bound to EIF4A3 proteins (Fig. 4A). Meanwhile, the RNA pull-down assay indicated that the ELF4A3 remarkably was enriched by both bio-SNHG16 and bio-TLR4 relative to the bio-control group (Fig. 4B), meaning that both SNHG16 and TLR4 interacted with EIF4A3. After overexpression of SNHG16, we found that the enrichment of TLR4 mRNAs by EIF4A3 was markedly increased, while the enrichment of TLR4 mRNA by EIF4A3 was significantly decreased when SNHG16 was knockout, indicating that SNHG16 increased the TLR4 levels by EIF4A3 protein (Fig. 4C). The additional experiment showed that overexpression of SNHG16 promoted TLR4 expression, which was significantly attenuated by EIF4A3 knockdown (Fig. 4D–E). The mRNA stability assay using transcription inhibitor actinomycin D showed that overexpression of SNHG16 stabilized the TLR4 mRNA, while knockdown of EIF4A3 promoted the degradation of TLR4 mRNA (Fig. 4F), indicating that SNHG16 stabilized TLR4 mRNA by recruiting EIF4A3 protein.
SNHG16 stabilizes TLR4 mRNA by recruiting EIF4A3. HRMCs were transfected with pcDNA3.0, pcDNA3.0-SNHG16, pcDNA3.0-TLR4, shNC or shSNHG16, siNC or siEIF4A3 for 48 h. The interactions between SNHG16 and EIF4A3 or between TLR4 and EIF4A3 were determined by RNA immunoprecipitation (A). The bindings of SNHG16 or TLR4 to EIF4A3 were determined by RNA pull-down assay (B). TLR4 mRNA levels were evaluated by RIP assay using anti-IgG and anti-EIF4A3 antibodies (C). TLR4 mRNA or protein levels were detected by qRT-PCR or western blot (D, E). The stabilities of TLR4 mRNA were detected in actinomycin-treated HRMCs after transfection of pcDNA3.1, pcDNA3.1-SNHG16 single or together with si-EIF4A3 (F). *P < 0.05, **P < 0.01, ***P < 0.001 versus the corresponding control. Data are representative of three independent experiments
Knockdown of TLR4 alleviates the HG-induced injuries in HRMCs by suppressing RAS and NF-κB to attenuate the activation of the NLRP3 inflammasome
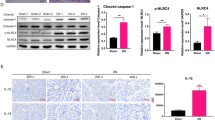
Then, we further characterized the role of TLR4 in HG-induced renal injuries. qRT-PCR confirmed that the transfection of TLR4 siRNA significantly decreased the TLR4 expression (Fig. 5A). HG significantly upregulated TLR4 expression at both mRNA and protein levels, but TLR4 silencing antagonized the upregulation of TLR4 at both mRNA and protein levels (Fig. 5B, C). The ELISA assays showed that HG led to significant secretion of cytokines, but knockdown of TLR4 diminished the levels of inflammatory cytokines (Fig. 5D). Further experiments exhibited that HG led to significantly increased productions of ROS and MDA but decreased expressions of SOD and GSH in HRMCs (Fig. 5E, F). However, when TLR4 was knockdown, the effects of HG on ROS, MDA, SOD, and GSH were reversed (Fig. 5E, F). Similarly, HG-induced high levels of renal fibrosis-related proteins (collagen 1, CTGF, FN, and TGF-β1), but TLR4 knockdown diminished HG-induced renal fibrosis (Fig. 5G). To further discover the underlying mechanism related to TLR4 in HG-induced renal injuries, we detected HG-induced pathological changes in the renin-angiotensin system (RAS) and the inflammasome. Our western blot assays exhibited that HG caused high activations of ACE, Ang II, AT1R, p-p65, and NLRP3, as well as cleaved-caspase1. TLR4 knockdown reversed the trend (Fig. 5H, I), indicating that knockdown of TLR4 protected HG-induced injuries by disrupting the blood system and increasing inflammation.
Knockdown of TLR4 alleviates the high glucose-induced injuries in HRMCs by suppressing RAS and NF-κB to attenuate the activation of the NLRP3 inflammasome. HRMCs were transfected with siNC or siTLR4 for 48 h, then administrated by normal or high glucose for a further 24 h. The TLR4 mRNA levels were measured (A). TLR4 mRNA or protein levels were measured by qRT-PCR or western blot (B, C). The protein levels of cytokines were analyzed by ELISA (D). DCFDA staining evaluated the ROS levels (E). MDA, SOD, and GSH levels were assessed using commercial kits (F). Western blot detected the protein levels of collagen 1, CTGF, FN and TGF-β1 (G), ACE, Ang II and AT1R (H), p-p65, p65, NLRP3, cleaved-caspase1, and total-caspase 1 (I). *P < 0.05, **P < 0.01, ***P < 0.001 versus the corresponding control. Data are representative of three independent experiments
SNHG16 promotes high glucose-induced cell injuries in HRMCs by regulating TLR4-mediated RAS and NF-κB to activate the NLRP3 inflammasome
To disclose the mechanism of SHNG16 in HG-induced renal injuries, we performed siRNA-based SNHG16 knockdown in HG-induced HRMCs and found that SNHG16 knockdown largely diminished HG-increased SNHG16 (Fig. 6A). Meanwhile, overexpression of TLR4 could not rescue SNHG16 silencing-induced low levels of SNHG16 (Fig. 6A). Interestingly, SNHG16 knockdown significantly decreased HG-upregulated TLR4, which was complemented by TLR4 overexpression (Fig. 6B, C). Additional experiments showed that knockdown of SNHG16 markedly reduced inflammatory cytokines, ROS productions, and MDA and increased the synthesis of SOD and GSH proteins (Fig. 6D–F). However, overexpression of TLR4 attenuated the protective roles of SNHG16 in HG-treated HRMCs by the increased ROS, cytokines, and MDA and the decreased SOD and GSH (Fig. 6D–F). Furthermore, we found that SNHG16 knockdown distinctly diminished HG-induced renal fibrosis and RAS-related proteins, but those protective roles were reversed when TLR4 was overexpressed (Fig. 6G, H). Importantly, we identified that knockdown of SNHG16 abolished HG-induced the increased p-p65, NLRP3, and cleaved caspase1. However, overexpression of TLR4 reversed the inhibitory effects of SNHG16 knockdown (Fig. 6I). Therefore, those results suggested that SNHG16 promotes renal injuries in HRMCs under HG conditions, possibly through directly regulating TLR4-mediated RAS and NF-κB- drove NLRP3-inflammasome.
SNHG16 promotes high glucose-induced cell injuries in HRMCs by regulating TLR4-mediated RAS and NF-κB to activate the NLRP3 inflammasome. HRMCs were transfected with shNC, shSNHG16, shSNHG16 + ov-NC or shSNHG16 + ov-TLR4 for 48 h, then were treated with normal or high glucose for a further 24 h. The SNHG16 mRNA levels were detected by qRT-PCR (A). TLR4 mRNA and protein levels were detected by qRT-PCR or western blot (B, C). The protein levels of cytokines TNF-α, IL-6, IL-1β, and IL-8 were detected by ELISA assay (D). ROS, MDA, SOD, and GSH were analyzed using a corresponding commercial kit (E, F). Western blot detected the protein levels of collagen 1, CTGF, FN, and TGF-β1 (G), ACE, Ang II, and AT1R (H), p-p65, p65, NLRP3, cleaved- and total-caspase 1 (I). *P < 0.05, **P < 0.01, ***P < 0.001 versus the corresponding control. Data are representative of three independent experiments
Knockdown of SNHG16 alleviates kidney injuries in db/db mice by suppressing TLR4
Subsequently, we generated the diabetic mice models and investigated the effects of SNHG16 in vivo. Our data showed that SNHG16 knockdown decreased the blood glucose levels in mice, but TLR4 overexpression compensated for SNHG16 knockdown-induced the decreased glucose level (Fig. 7A). The biochemical assays found that diabetic mice exhibited higher levels of urinary protein, Scr and BUN relative to those of control (Fig. 7B, C). Knockdown of SNHG16 decreased the levels of urinary protein, Scr and BUN in diabetic mice, but overexpression of TLR4 observably increased the urinary protein, Scr and BUN when SNHG16 was silenced (Fig. 7B, C). H and E and PAS staining showed that HG led to noticeable tubular and interstitial fibrosis, hyperplasia of the mesangial matrix, and increased thickness of basal membrane of mice renal tissue. Meanwhile, the knockdown of SNHG16 alleviated HG-induced damages. However, TLR4 overexpression reversed the protective role of SNHG16 knockdown (Fig. 7D). Our ELISA assay further found that knockdown of SNHG16 distinctly attenuated the inflammatory cytokine levels in diabetic mice compared with the control group. Relative to the control group, overexpression of TLR4 markedly enhanced the cytokine secretion in diabetic mice subjected to SNHG16 knockdown (Fig. 7E). We also measured the expressions of SNHG16 and TLR4. Our qRT-PCR results validated that SNHG16 and TLR4 were significantly increased in diabetic conditions, but knockdown of SNHG16 decreased both SNHG16 and TLR4. However, overexpression of TLR4 could not upregulate SNHG16 but significantly upregulated TLR4 (Fig. 7F). Our western blot further revealed that diabetes mice exhibited higher levels of TLR4, collagen 1, CTGF, FN, TGF-β1, RAS proteins ACE, Ang II, AT1R, p-p65, NLRP3, and cleaved caspase-1 than those of normal mice (Fig. 7G–I). SNHG16 silencing dramatically attenuated HG-induced effects. Significantly, TLR4 overexpression observably diminished the protective roles of SNHG16 knockdown (Fig. 7G–I). All those results suggested that knockdown of SNHG16 alleviated renal injuries in diabetic mice probably via suppressing TLR4.
Knockdown of lncRNA SNHG16 alleviates kidney injuries in db/db mice by suppressing TLR4. The db/db mice were injected with sh-NC, sh-SNHG16, sh-SNHG16 + VE, or sh-SNHG16 + TLR4 lentivirus. A glucometer detected the blood glucose levels (A). An automatic biochemical analyzer analyzed the urine protein contents of mice (B). An automatic biochemical analyzer analyzed serum creatinine (SCR) or blood urea nitrogen (BUN) (C). Hematoxylin and eosin staining (HE) and periodic acid-Schiff (PAS) assay analyzed the changes in histological structures and glycogen accumulation (D). The cytokines in the sera of mice were determined by ELISA assay (E). The SNHG16 and TLR4 mRNA levels were detected by qRT-PCR (F). Western blot detected the protein levels of TLR4, collagen 1, CTGF, FN, and TGF-β1 (G), ACE, Ang II, and AT1R (H), p-p65, p65, NLRP3, cleaved-caspase1, and total-caspase1 (I).*P < 0.05, **P < 0.01, ***P < 0.001 versus the corresponding control. Data are representative of three independent experiments
Discussions
DN is one of the chronic inflammation and microvascular deficits. The previous studies evidenced that inflammation is a significant cause of DN development [27]. Our data revealed that lncRNA SNHG16 contributed to renal injury via regulating TLR4, thus activating the RAS system and NF-κβ-mediated NLRP3 inflammasome. Our findings further identified that SNHG16 upregulated TLR4 expression by recruiting EIF4A3 to stabilize TLR4 mRNA and decreased the degradation of TLR4 mRNA. Knockdown of SNHG16 or TLR4 alleviated HG-induced renal injuries, providing a new therapeutic target for ND treatment.
LncRNAs are non-coding RNAs that act as primary regulators to regulate gene expressions at transcriptional, post-transcriptional, translational, or post-translational levels [36]. Recently, accumulating evidence found that lncRNAs participate in many pathological progressions of DN, such as podocyte apoptosis [13], renal fibrosis [8], oxidative stress [21], and inflammation [30]. In the current study, our experiments revealed that SNHG16 was dramatically increased in both diabetic renal tissues and HG-induced HRMCs. Further investigations found that knockdown of SNHG16 decreased the release of cytokines, ROS, MDA, and fibrosis-related proteins and increase SOD and GSH, indicating that SNHG16 might be a potential target for diabetic renal injuries.
LncRNAs exhibit functional roles, possibly through direct binding its downstream mRNA sequences to silence gene expression or directly interacting with proteins to regulate the downstream genes (X. [36]. Recently, it was found that lncRNA NEAT1 promoted the TLR4 expression by directly binding and inhibiting miR-302b-3p [6]. This study found that both SNHG16 and TLR4 were upregulated in diabetic conditions. Hence, we wondered whether SNHG16 regulated TLR4. Renin-angiotensin system (RAS) supervises blood pressure and fluid homeostasis. During diabetic conditions, HG can significantly upregulate RAS-related angiotensin II, contributing to the progression of diabetic angiopathy [24]. A previous study verified that HG-induced high levels of angiotensin II increased the expression of TLR4 [20]. Later experiments also showed that high glucose upregulated TLR4 expression, subsequently regulating the Ang II expression [5]. Increasing evidence identified that inflammatory response might be the critical regulator linking the mutual regulation between TLR4 and Ang II activations [5]. Our study found that HG upregulated both TLR4 and RAS-related enzymes ACE, Ang II, and AT1R, and knockdown of TLR4 decreased the ACE, Ang II, and AT1R. All those results indicated that HG-induced activation of RAS and TLR4 exhibited mutual regulation and promotion. However, their regulatory relationship still needs to be further explored.
Several studies confirmed that inflammation-related signaling pathways such as IL-8-CXCR1/2 axis [17] and TLR4 signaling were potential therapeutic target for diabetic kidney disease. A recent study showed that some drugs like panobinostat might attenuate inflammation-related glomerulonephritis through regulating the differentiation of human renal progenitor cells into podocytes to restore the glomerular filtration barrier [18]. Accumulating studies indicate that the NLRP3 inflammasome is a promising therapeutic target for treating ND [31]. Suppression of NLRP3 inflammasome alleviated podocyte damage [28] and autophagy [11]. A previous study had validated that high glucose mediates the activation of NLRP3 inflammasome via the TLR4/NF-κB signaling pathway [14]. Our experiments showed that HG-induced NLRP3 inflammasome correlated with the level of TLR4. After TLR4 was silenced in HG-induced HRMCs, we found HG-induced NLRP3 was remarkably downregulated. In contrast, when TLR4 was overexpressed, NLRP3 expression was reversed in HG-induced HRMCs and diabetic renal tissues. All those results revealed that the activation of TLR4 contributed to the development of NLRP3 inflammasome under diabetic conditions.
In conclusion, our study validated that lncRNA SNHG16 aggravated DN via the activations of RAS and TLR4, which subsequently promote NF-κB-dependent activation of the NLRP3 inflammasome. Mechanistically, SNHG16 upregulated TLR4 mRNA by recruiting EIF4A3 and increasing the stabilization of TLR4 mRNA. Our findings suggest that SNHG16/TLR4/NRLP3 axis might be a novel therapeutic target for treating DN.
Data availability
All data collected and analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- LncRNA:
-
Long non-coding RNAs
- SNHG16:
-
Small nucleolar RNA host gene 16
- RAS:
-
Renin-angiotensin system
- HG:
-
High glucose
- TLR4:
-
Toll-like receptor 4
- NRLP3:
-
NLR family pyrin domain containing 3
- EIF4A3:
-
Eukaryotic translation initiation factor 4A3
- DRI:
-
Diabetic renal injury
- DN:
-
Diabetes nephropathy
- RIP:
-
RNA immunoprecipitation
- HRMCs:
-
Human renal mesangial cells
- ROS:
-
Reactive oxygen species
- MDA:
-
Malondialdehyde
- SOD:
-
Superoxide dismutase
- GSH:
-
Glutathione
- MAPK:
-
Mitogen-activated protein kinase
- ACE:
-
Angiotensin-converting enzyme
- AT:
-
Angiotensin
- DCFH-DA:
-
Dichlorodihydrofluorescin diacetate
- FN:
-
Fibronectin
References
Batsford S, Duermueller U, Seemayer C, Mueller C, Hopfer H, Mihatsch M (2011) Protein level expression of Toll-like receptors 2, 4 and 9 in renal disease. Nephrol Dial Transplant 26(4):1413–1416. https://doi.org/10.1093/ndt/gfq752
Cai F, Jiang H, Li Y, Li Q, Yang C (2021) Upregulation of long non-coding RNA SNHG16 promotes diabetes-related RMEC dysfunction via activating NF-κB and PI3K/AKT pathways. Mol Ther Nucleic Acids 24:512–527. https://doi.org/10.1016/j.omtn.2021.01.035
D’Addio F, Vergani A, Potena L et al (2018) P2X7R mutation disrupts the NLRP3-mediated Th program and predicts poor cardiac allograft outcomes. J Clin Invest 128(8):3490–3503. https://doi.org/10.1172/jci94524
Duan YR, Chen BP, Chen F et al (2021) LncRNA lnc-ISG20 promotes renal fibrosis in diabetic nephropathy by inducing AKT phosphorylation through miR-486-5p/NFAT5. J Cell Mol Med 25(11):4922–4937. https://doi.org/10.1111/jcmm.16280
Feng Q, Liu D, Lu Y, Liu Z (2020) The interplay of renin-angiotensin system and Toll-like receptor 4 in the inflammation of diabetic nephropathy. J Immunol Res 2020:6193407. https://doi.org/10.1155/2020/6193407
Fu D, Ju Y, Zhu C, Pan Y, Zhang S (2022) LncRNA NEAT1 promotes TLR4 expression to regulate lipopolysaccharide-induced trophoblastic cell pyroptosis as a molecular sponge of miR-302b-3p. Mol Biotechnol. https://doi.org/10.1007/s12033-021-00436-2
Garibotto G, Carta A, Picciotto D, Viazzi F, Verzola D (2017) Toll-like receptor-4 signaling mediates inflammation and tissue injury in diabetic nephropathy. J Nephrol 30(6):719–727. https://doi.org/10.1007/s40620-017-0432-8
Ge Y, Wang J, Wu D et al (2019) lncRNA NR_038323 suppresses renal fibrosis in diabetic nephropathy by targeting the miR-324-3p/DUSP1 axis. Mol Ther Nucleic Acids 17:741–753. https://doi.org/10.1016/j.omtn.2019.07.007
Gurley SB, Coffman TM (2007) The renin-angiotensin system and diabetic nephropathy. Semin Nephrol 27(2):144–152. https://doi.org/10.1016/j.semnephrol.2007.01.009
He X, Zeng X (2020) LncRNA SNHG16 aggravates high glucose-induced podocytes injury in diabetic nephropathy through targeting miR-106a and thereby up-regulating KLF9. Diabetes Metab Syndr Obes 13:3551–3560. https://doi.org/10.2147/dmso.S271290
Hou Y, Lin S, Qiu J et al (2020) NLRP3 inflammasome negatively regulates podocyte autophagy in diabetic nephropathy. Biochem Biophys Res Commun 521(3):791–798. https://doi.org/10.1016/j.bbrc.2019.10.194
Jiang X, Ru Q, Li P et al (2020) LncRNA SNHG16 induces proliferation and fibrogenesis via modulating miR-141-3p and CCND1 in diabetic nephropathy. Gene Ther 27(12):557–566. https://doi.org/10.1038/s41434-020-0160-x
Lei X, Zhang L, Li Z, Ren J (2018) Astragaloside IV/lncRNA-TUG1/TRAF5 signaling pathway participates in podocyte apoptosis of diabetic nephropathy rats. Drug Des Dev Ther 12:2785–2793. https://doi.org/10.2147/dddt.S166525
Leng B, Zhang Y, Liu X et al (2019) Astragaloside IV suppresses high glucose-induced NLRP3 inflammasome activation by inhibiting TLR4/NF-κB and CaSR. Mediat Inflamm 2019:1082497. https://doi.org/10.1155/2019/1082497
Lin M, Yiu WH, Wu HJ et al (2012) Toll-like receptor 4 promotes tubular inflammation in diabetic nephropathy. J Am Soc Nephrol 23(1):86–102. https://doi.org/10.1681/asn.2010111210
Liu X, Ma L, Li Z (2020) Effects of renin-angiotensin system blockers on renal and cardiovascular outcomes in patients with diabetic nephropathy: a meta-analysis of randomized controlled trials. J Endocrinol Invest 43(7):959–972. https://doi.org/10.1007/s40618-020-01179-8
Loretelli C, Rocchio F, D’Addio F et al (2021) The IL-8-CXCR1/2 axis contributes to diabetic kidney disease. Metabolism 121:154804. https://doi.org/10.1016/j.metabol.2021.154804
Melica ME, Antonelli G, Semeraro R et al (2022) Differentiation of crescent-forming kidney progenitor cells into podocytes attenuates severe glomerulonephritis in mice. Sci Transl Med 14(657):eabg3277. https://doi.org/10.1126/scitranslmed.abg3277
Meznad K, Paget-Bailly P, Jacquin E et al (2021) The exon junction complex core factor eIF4A3 is a key regulator of HPV16 gene expression. Biosci Rep. https://doi.org/10.1042/bsr20203488
Nair AR, Ebenezer PJ, Saini Y, Francis J (2015) Angiotensin II-induced hypertensive renal inflammation is mediated through HMGB1-TLR4 signaling in rat tubulo-epithelial cells. Exp Cell Res 335(2):238–247. https://doi.org/10.1016/j.yexcr.2015.05.011
Qin B, Cao X (2021) LncRNA PVT1 regulates high glucose-induced viability, oxidative stress, fibrosis, and inflammation in diabetic nephropathy via miR-325-3p/Snail1 axis. Diabetes Metab Syndr Obes 14:1741–1750. https://doi.org/10.2147/dmso.S303151
Samsu N (2021) Diabetic nephropathy: challenges in pathogenesis, diagnosis, and treatment. Biomed Res Int 2021:1497449. https://doi.org/10.1155/2021/1497449
Satirapoj B, Adler SG (2015) Prevalence and management of diabetic nephropathy in western countries. Kidney Dis (Basel) 1(1):61–70. https://doi.org/10.1159/000382028
Shaw S, Wang X, Redd H, Alexander GD, Isales CM, Marrero MB (2003) High glucose augments the angiotensin II-induced activation of JAK2 in vascular smooth muscle cells via the polyol pathway. J Biol Chem 278(33):30634–30641. https://doi.org/10.1074/jbc.M305008200
Su Q, Li L, Sun Y, Yang H, Ye Z, Zhao J (2018) Effects of the TLR4/Myd88/NF-κB signaling pathway on NLRP3 inflammasome in coronary microembolization-induced myocardial injury. Cell Physiol Biochem 47(4):1497–1508. https://doi.org/10.1159/000490866
Sugahara M, Pak WLW, Tanaka T, Tang SCW, Nangaku M (2021) Update on diagnosis, pathophysiology, and management of diabetic kidney disease. Nephrology (Carlton) 26(6):491–500. https://doi.org/10.1111/nep.13860
Wada J, Makino H (2013) Inflammation and the pathogenesis of diabetic nephropathy. Clin Sci (Lond) 124(3):139–152. https://doi.org/10.1042/cs20120198
Wu M, Yang Z, Zhang C et al (2021) Inhibition of NLRP3 inflammasome ameliorates podocyte damage by suppressing lipid accumulation in diabetic nephropathy. Metabolism 118:154748. https://doi.org/10.1016/j.metabol.2021.154748
Wu X, Fan D, Chen B (2022) LncRNA NEAT1 accelerates the proliferation, oxidative stress, inflammation, and fibrosis and suppresses the apoptosis through the miR-423-5p/GLIPR2 axis in diabetic nephropathy. J Cardiovasc Pharmacol 79(3):342–354. https://doi.org/10.1097/fjc.0000000000001177
Xu Y, Zhan X (2021) lncRNA KCNQ1OT1 regulated high glucose-induced proliferation, oxidative stress, extracellular matrix accumulation, and inflammation by miR-147a/SOX6 in diabetic nephropathy (DN). Endocr J. https://doi.org/10.1507/endocrj.EJ21-0514
Yang M, Wang X, Han Y et al (2021) Targeting the NLRP3 inflammasome in diabetic nephropathy. Curr Med Chem 28(42):8810–8824. https://doi.org/10.2174/0929867328666210705153109
Ye J, She X, Liu Z et al (2021) Eukaryotic initiation factor 4A-3: a review of its physiological role and involvement in oncogenesis. Front Oncol. https://doi.org/10.3389/fonc.2021.712045
Zhang B, Ramesh G, Uematsu S, Akira S, Reeves WB (2008) TLR4 signaling mediates inflammation and tissue injury in nephrotoxicity. J Am Soc Nephrol 19(5):923–932. https://doi.org/10.1681/ASN.2007090982
Zhang C, Han X, Yang L et al (2020) Circular RNA circPPM1F modulates M1 macrophage activation and pancreatic islet inflammation in type 1 diabetes mellitus. Theranostics 10(24):10908–10924. https://doi.org/10.7150/thno.48264
Zhang H, Yan Y, Hu Q, Zhang X (2021) LncRNA MALAT1/microRNA let-7f/KLF5 axis regulates podocyte injury in diabetic nephropathy. Life Sci 266:118794. https://doi.org/10.1016/j.lfs.2020.118794
Zhang X, Wang W, Zhu W et al (2019) Mechanisms and functions of long non-coding RNAs at multiple regulatory levels. Int J Mol Sci 20(22):5573. https://doi.org/10.3390/ijms20225573
Funding
This work was supported by Guangdong Provincial Basic and Applied Basic Research Fund Regional Joint Fund Project (Youth Fund Project) (Project Number: 2020A1515110130).
Author information
Authors and Affiliations
Contributions
YL contributed to conceptualization; methodology; supervision; MZ contributed to validation; formal analysis; HZ contributed to investigation; resources; NX contributed to data curation; YW contributed to writing—original draft; SD contributed to visualization; XS contributed to writing—review and editing; project administration; funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
All experiments were supervised by the Animal Welfare and Ethics Committee of Dongguan Tung wah Hospital.
Consent for publication
All the authors approved the publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the topical collection Diabetic Nephropathy, managed by Giuseppe Pugliese.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Zhang, M., Zhong, H. et al. LncRNA SNHG16 regulates RAS and NF-κB pathway-mediated NLRP3 inflammasome activation to aggravate diabetes nephropathy through stabilizing TLR4. Acta Diabetol 60, 563–577 (2023). https://doi.org/10.1007/s00592-022-02021-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-022-02021-8