Abstract
To determine the mycorrhizal status of pteridophyte gametophytes in diverse taxa, the mycorrhizal colonization of wild gametophytes was investigated in terrestrial cordate gametophytes of pre-polypod leptosporangiate ferns, i.e., one species of Osmundaceae (Osmunda banksiifolia), two species of Gleicheniaceae (Diplopterygium glaucum, Dicranopteris linearis), and four species of Cyatheales including tree ferns (Plagiogyriaceae: Plagiogyria japonica, Plagiogyria euphlebia; Cyatheaceae: Cyathea podophylla, Cyathea lepifera). Microscopic observations revealed that 58 to 97 % of gametophytes in all species were colonized with arbuscular mycorrhizal (AM) fungi. Fungal colonization was limited to the multilayered midrib (cushion) tissue in all gametophytes examined. Molecular identification using fungal SSU rDNA sequences indicated that the AM fungi in gametophytes primarily belonged to the Glomeraceae, but also included the Claroideoglomeraceae, Gigasporaceae, Acaulosporaceae, and Archaeosporales. This study provides the first evidence for AM fungal colonization of wild gametophytes in the Plagiogyriaceae and Cyatheaceae. Taxonomically divergent photosynthetic gametophytes are similarly colonized by AM fungi, suggesting that mycorrhizal associations with AM fungi could widely occur in terrestrial pteridophyte gametophytes.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Although recent studies have shown that most land plant groups, from bryophytes to angiosperms, are mycorrhizal (Wang and Qiu 2006), the status of mycorrhizal symbioses remains unknown in many plant species, especially in “lower” land plants (Read et al. 2000). For example, the mycorrhizal status of the gametophytes of pteridophytes (ferns and lycophytes) is largely unknown, as the simple structure of the tiny gametophytes makes it difficult to identify plant species using morphological features. In contrast, the mycorrhizal associations of pteridophyte sporophytes have been surveyed in many lineages worldwide (Cooper 1976; Gemma et al. 1992; Zhi-wei 2000; Lehnert et al. 2009; Kessler et al. 2010; Muthukumar and Prabha 2013; Muthuraja et al. 2014; Lara-Pérez et al. 2015). Since mycorrhizal associations influence plant nutrient uptake (Marschner and Dell 1994), plant responses to stress (Finlay et al. 2008), and the structure and composition of plant communities (van der Heijden et al. 2008), characterizing the mycorrhizal associations of pteridophytes is essential for a more thorough understanding of their ecology and evolution.
To date, the mycorrhizal fungi of pteridophyte gametophytes have been identified as being primarily arbuscular mycorrhizal (AM) fungi (Read et al. 2000), with a few exceptions, such as Diphasiastrum alpinum (Lycopodiaceae) associated with basidiomycetes (Horn et al. 2013). Gametophytes grow in divergent habitats ranging from underground, terrestrial on the ground, epilithic on rocks, and epiphytic on tree trunks (Farrar et al. 2008). The underground gametophytes of the Lycopodiaceae, Psilotaceae, and Ophioglossaceae are achlorophyllous and depend on mycorrhizal fungi for their carbon supply (Bierhorst 1971). The associations of such mycoheterotrophic gametophytes with AM fungi have been surveyed since the early twentieth century (Campbell 1908a; Boullard 1979; Schmid and Oberwinkler 1993; Winther and Friedman 2007, 2008, 2009). In contrast, the terrestrial and chlorophyllous gametophytes of ferns, which are generally cordate, have been considered devoid of mycorrhiza, with a few exceptions in the Marattiales, Osmundales, Gleicheniales (Campbell 1908a; Boullard 1979; Schmid and Oberwinkler 1995), and Polypodiales (Turnau et al. 2005, 2013). In a recent study, we found that more than 90 % of field-collected gametophytes were colonized by AM fungi in Angiopteris lygodiifolia (Marattiaceae) and Osmunda japonica (Osmundaceae) (Ogura-Tsujita et al. 2013). However, mycorrhizal colonization of gametophytes has only been documented in a few families, and the status of mycorrhizal associations remains largely unknown in pteridophyte gametophytes, especially in aboveground photosynthetic gametophytes.
The main objective of this study was to survey the mycorrhizal status of field-collected gametophytes of entire groups of leptosporangiate ferns. Phylogenetically, the leptosporangiate ferns comprise seven clades: osmundaceous, filmy, gleichenioid, schizaeoid, heterosporous, and tree- and polypod-ferns (sensu Pryer et al. 2004). The mycorrhizal status of the gametophytes of polypod ferns, the most derived and largest group of leptosporangiate ferns (Pryer et al. 2004), will be reported elsewhere since preliminary studies suggested that they exhibit a wide variation in fungal colonization levels. Among the “pre-polypod leptosporangiate ferns” (i.e. the leptostorangiate ferns excluding the polypod), filmy ferns and heterosporous ferns were not examined in this study because their gametophytes are non-cordate. Among the schizaeoid ferns, the Lygodiaceae have typical cordate gametophytes, but natural samples of lygodiaceous gametophytes unfortunately were not obtained at this time. For osmundaceous and gleichenioid ferns, the mycorrhizal status of O. japonica has been addressed elsewhere (Ogura-Tsujita et al. 2013) but other osmundaceous and gleichenioid ferns have not been analyzed using molecular identification techniques, although anatomical observations have been conducted in a few species (Campbell 1908a; Boullard 1979; Schmid and Oberwinkler 1995). Thus, in the present study, the mycorrhizal colonization status of the wild gametophytes of one species of osmundaceous fern (Osmunda banksiifolia (C. Presl) Kuhn), two species of gleichenioid ferns (Diplopterygium glaucum (Houtt.) Nakai and Dicranopteris linearis (Burm. f.) Underw.), and four species of the tree-fern clade (Plagiogyria japonica Nakai and Plagiogyria euphlebia (Kunze) Mett. of the Plagiogyriaceae and Cyathea podophylla (Hook.) Copel. and Cyathea lepifera (J. Sm. ex Hook.) Copel. of the Cyatheaceae) were assessed using anatomical observations, and fungal lineages were identified by molecular methods.
Cordate gametophytes are composed of a multilayered midrib (cushion) and one-cell-thick wing portions (Bower 1923; Raghavan 1989). In a previous study, we documented that mycorrhizal colonization was limited to the cushion in both A. lygodiifolia and O. japonica gametophytes (Ogura-Tsujita et al. 2013), suggesting that the gametophyte cushion structure is key to associations with AM fungi. Therefore, anatomical observations to examine the site(s) of fungal colonization and cushion thickness in the gametophytes were also conducted.
Materials and methods
Sample collection
A recent survey of the natural distribution of fern gametophytes and sporophytes at a field site in Japan indicated that cordate gametophytes were without exception accompanied by their conspecific sporophytes at nearby sites, whereas the sporophytic counterparts of non-cordate (strap- or ribbon-like) gametophytes were often absent or rare near the sites (Ebihara et al. 2013). Therefore, field expeditions were conducted at locations where a large number of sporophytes of the seven studied fern species were growing. Wild gametophytes were collected from sites around which conspecific sporophytes were growing between May 2008 and November 2012 (Table 1). These study sites were located in subtropical evergreen forests (for Osmundaceae, Gleicheniaceae, and Cyatheaceae) or temperate deciduous forests (for Plagiogyriaceae). Most gametophytes were growing on the soil of slopes along trails.
All collected gametophytes were washed in water using a soft fine brush under a stereomicroscope, and images were taken using a digital camera (DP70, Olympus) attached to a light microscope (Olympus BX51). Immature gametophytes lacking archegonia were not used for examinations. Mature gametophytes were cut sagittally into two (right and left) pieces in the middle of the cushion using a razor blade. One piece was used for microscopic observations to confirm fungal colonization, and the other piece was used for DNA analysis to identify the species of both plant and AM fungi (Fig. S1). In addition, one gametophyte each of C. podophylla, P. euphlebia, and Dip. glaucum and two gametophytes of O. banksiifolia were cut transversally into two pieces: the upper two thirds and basal one third for use in microscopic observations and DNA analysis, respectively.
Microscopic observations
Gametophyte pieces were fixed in FAA (50 % ethanol/formaldehyde/acetic acid, 90:5:5) for more than 24 h. Pieces were either stained using trypan blue or resin-sectioned (Table 2). For trypan blue staining, fixed pieces were cleared using 10 % KOH for 5 min and stained with acidic glycerol/trypan blue (0.05 %) (Koske and Gemma 1989). Materials stained using trypan blue were dehydrated in a graded ethanol series and mounted with Permount (Fischer Scientific, Morris Plains, NJ, USA). For resin-sectioning, the fixed materials were dehydrated through a graded ethanol series, embedded in Technovit 7100 (Heraeus, Kulzer, Germany), cut longitudinally or rarely transversely into 3-μm-thick sections, and stained using modified Sharman’s solution (Jernstedt et al. 1992). For the longitudinal and transverse sections, the thickest portion of the cushion was measured as the cushion thickness of individuals. Prepared slides of trypan blue-stained and resin-sectioned samples were observed under a light microscope (Olympus BX51). Colonization levels were calculated as the total number of gametophytes infected by AM fungi divided by the total number of gametophytes investigated. All prepared slides were deposited at the herbarium of the National Museum of Nature and Science as voucher specimens.
Fungal and plant DNA analysis
DNA was extracted from fresh gametophyte tissues using a DNeasy Plant Mini Kit (Qiagen, Valencia, CA, USA). Gametophytes for which fungal colonization was confirmed via microscopic observation were used for fungal DNA analysis. The fungal small subunit (SSU) rDNA region was amplified and sequenced using a universal eukaryotic primer NS31 (Simon et al. 1992) and an AM fungal specific primer AML2 (Lee et al. 2008). PCR was performed using PrimeStarGXL DNA Polymerase (Takara-Bio, Shiga, Japan). PCR conditions and sequencing were conducted as described by Ogura-Tsujita et al. (2013). Most PCR products were cloned using the pGEM-T Vector System I (Promega, Madison, WI, USA), while a portion of samples was directly sequenced without cloning (see Supplementary information Table S1). Ten to 20 colonies were chosen from each PCR product for sequencing. Fungal sequences were assessed for similarities against the MaarjAM database (Öpik et al. 2010) and then assigned to either a virtual taxon (VT) based on sequence similarity of ≥97 % or an operational taxonomic unit (OTUs) for similarity <97 %. To avoid PCR-generated chimera, sequences from multiple individuals or the above three clones were assigned to OTUs. The DNA Data Bank of Japan (http://www.ddbj.nig.ac.jp) accession numbers of the fungal sequences determined in this study are given in Fig. 2.
The rbcL-a region or upper 700 bp of the chloroplast rbcL gene was amplified from all collected gametophytes to confirm the identity of the plant species, as described by Ebihara et al. (2008), using the primer combination rbcL1-1 (Hasebe et al. 1995) and rbcLHIR1 (Ebihara et al. 2002). Sequences were then compared to the DNA barcoding data set for Japanese pteridophytes (Ebihara et al. 2010).
Phylogenetic analysis
One representative sequence was selected for each VT or OTU for use in the phylogenetic analysis. Fungal SSU sequences from gametophytes were aligned manually against the data matrix in Schwarzott et al. (2001) using MacClade v. 4.06 (Maddison and Maddison 2003). Classification of Glomeromycota followed Schüßler and Walker (2010) and Krüger et al. (2012).
Bayesian analysis was performed using MrBayes 3.1.2 (Ronquist and Huelsenbeck 2003) employing the GTR + I + G model of DNA substitution. Four Markov chain Monte Carlo chains were run for ten million generations with sampling every 1000 generations, and the first 2500 samples were discarded as burn-in. The bootstrap method (Felsenstein 1985) was used with 1000 replicates to assess the relative robustness of the branches. The SSU sequences of Mortierella polycephala (X89436) and Endogone pisiformis (X58724) were used as outgroup taxa.
Results
Gametophyte morphologies and fungal colonization
The wild gametophytes of the seven study species all exhibited the typical cordate shape with a deeply seated notch between nearly equally developed right and left wings (Fig. 1a, d, g, j, m, p; image of C. lepifera not shown), although the ratio between the width and length of the wings varied somewhat with age and individual, even within the same species. The cushion ran from the notch to the base. Archegonia occurred along both lateral sides of the cushion in O. banksiifolia (data not shown), whereas they arose from the entire surface of the cushion in the other species examined (e.g., small dots on the upper portion of the cushion of Dic. linearis, Fig. 1g). Antheridia were found on both wings in all species examined (data not shown).
Fern gametophytes. a–c Osmunda banksiifolia, d–f Diplopterygium glaucum, g–i Dicranopteris linearis, j–l Plagiogyria japonica, m–o Plagiogyria euphlebia, p–r Cyathea podophylla. a, d, g, j, m, p Microscope images of entire gametophytes, bars = 1 mm; b, e, h, k, n, q resin sections in sagittal plane of the cushion, bars = 100 μm; c, f, i, l, o, r enlarged figures of cells colonized by AM fungi, indicated by arrows in b, e, h, k, n, and q, bars = 10 μm
Serial longitudinal sections of gametophytes in the sagittal plane revealed that fungal hyphae, if present, occupied the posterior (basal) portion of the cushion. The area of fungal colonization covered up to half, and sometimes two thirds, of the area from the base end of the gametophytes (e.g., Fig. 1e, h, n). Hyphae were absent from the anterior portion just below the notch. Cells with fungal colonization were not always continuous but were sometimes found in separate island areas (Fig. 1b). In the typical case, fungi were not found in the dorsal surface cell layer but were primarily located in the inner tissue and ventral surface cells of the cushion (Fig. 1e, n). In some cases, however, the dorsal surface cells were also colonized by fungi (Fig. 1k). The fungal hyphae were sometimes found in rhizoids, which arose from the ventral surface cells. Serial transverse sections of gametophytes clearly indicated the absence of fungal colonization in the one-cell-thick wing portions (data not shown).
Fungal hyphae in the cushion tissue cells were aseptate and sometimes showed finely branched arbuscules (Fig. 1c, f, i, l, o, r), which are typical of AM fungi. Cells colonized by AM fungi contained very few plastids, whereas non-mycorrhizal cells had relatively large numbers of plastids (data not shown).
Fungal colonization levels and cushion thickness of gametophytes
Fungal colonization levels, which were calculated using the total number of trypan blue-stained and resin-sectioned preparations for each species, are summarized in Table 2. For all seven species examined, 58 to 97 % of gametophytes exhibited AM fungal colonization. High colonization (≥90 %) was observed in O. banksiifolia and P. japonica, whereas colonization was relatively low in Dip. glaucum (58 %) and C. lepifera (63 %).
Cushion thickness and the number of cushion cell layers were measured in samples that did not experience shrinkage in fixative solution. Cushion thickness ranged from 208 to 306 μm, and the number of cell layers ranged from 5.6 to 8.4 (Table 2). Cushion structure (either thickness or the number of cushion cell layers) did not appear to be correlated with the percentage of AM fungal colonization. Notably, resin-sectioned samples always contained fungi (i.e., 100 % colonization) for all species examined, whereas samples stained with trypan blue sometimes lacked fungi. In particular, when fungi were limited to the basal area with dense rhizoidal hairs, they were sometimes overlooked in trypan blue-stained samples but not in resin-sectioned samples.
DNA analysis
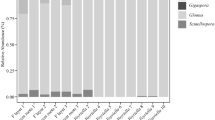
Fungal sequences obtained from the gametophytes were grouped into 40 VTs and two OTUs (Table 3). Individual gametophytes harbored 1–6 VTs or OTUs. Most VT sequences from the gametophytes belonged to the Glomeraceae (former Glomus group A), although Claroideoglomeraceae, Acaulosporaceae, Archaeosporaceae, and Gigasporaceae sequences were also identified. The most frequently observed VTs (≥20 instances) were VT 80, 166, 191, and 219; VT 80 and 219 were found in all of the fern taxa examined.
The phylogenetic analysis revealed that the Glomeromycota sequences isolated from gametophytes were widely divergent (Fig. 2). The OTU sequences, which shared less similarity (≤96 %) with VT sequences in the MaarjAM BLAST analysis, were included in the phylogenetic analysis, resulting in ambiguous phylogenetic positions for OTU1 and OTU2.
Discussion
Using microscopic and molecular techniques, the present study has demonstrated that field-collected gametophytes of the Osmundaceae, Gleicheniaceae, Plagiogyriaceae, and Cyatheaceae are colonized by AM fungi. To our knowledge, this study provides the first evidence of AM fungal colonization in wild gametophytes of the tree fern clade (sensu Pryer et al. 2004), i.e., the Plagiogyriaceae and Cyatheaceae where fungal colonization levels were high (63–97 %). Cooper (1976) sowed spores of several fern species, including Cyathea, into field soils and found that fungal colonization only sporadically occurred in gametophytes. Although Boullard (1979) reported that the gametophytes of the Cyatheaceae and Loxomataceae were non-mycorrhizal, the present study indicates that the Cyatheales include AM-associated gametophytes. Concerning the Osmundaceae and Gleicheniaceae, the gametophytes of O. banksiifolia and the two species Dip. glaucum and Dic. linearis were colonized by AM fungi at intermediate to high levels (58–91 %) suggesting that diverse taxa of the Osmundaceae and Gleicheniaceae harbor AM fungi in gametophytes. Mycorrhizal gametophytes have already been documented in Osmunda (Campbell 1908a; Boullard 1957) and Todea (Cooper 1976), and we have reported AM fungal colonization in 95 % of wild O. japonica gametophytes (Ogura-Tsujita et al. 2013). Endophytic fungal colonization of aboveground Gleicheniaceae gametophytes has been previously reported (Campbell 1908b; Boullard 1957), and Schmid and Oberwinkler (1995) observed intracellular hyphal coils with arbuscules and vesicles in field-collected gametophytes of the Gleicheniaceae using light and electron microscopy.
The molecular identification of AM fungi in the present study suggests that photosynthetic gametophytes of pteridophytes are associated with a broad range of fungi in the Glomeromycota, particularly the Glomeraceae. Pteridophyte sporophytes are well known to associate with a wide range of AM fungi (Zhang et al. 2004; West et al. 2009), whereas mycoheterotrophic gametophytes, such as Botrychium (Winther and Friedman 2007) and Psilotum (Winther and Friedman 2009), exhibit very specific associations with particular members of the Glomeraceae. In the present study, the most frequently observed (≥20 total instances) VTs were VT 80, 166, 191, and 219 of the Glomeraceae. VT 166 has been recorded on six continents (Africa, North America, South America, Oceania, Europe, Asia), while VT 191 and 219 have been recorded on five continents (Öpik et al. 2010). These observations demonstrate that these VTs are globally distributed and extremely common taxa, accounting for their frequent occurrence among the gametophyte samples examined in the present study.
Colonization by AM fungi was limited to parts of the cushion in all observed gametophytes, similar to previous findings (Ogura-Tsujita et al. 2013). These results suggest that fungal colonization is limited to the cushion tissue in aboveground cordate gametophytes, as has been shown for liverwort thalli where associate AM fungi primarily colonize the inner parenchyma (Ligrone et al. 2007).
Among the gametophytes of the six species examined, AM fungal colonization levels did not appear to be correlated with cushion thickness or the number of cushion cell layers. The gametophytes of P. japonica exhibited highest colonization (97 %) but had the fourth thickest cushion (252 μm thick and 6.0 cell layers). Conversely, Dic. linearis (Gleicheniaceae) had the thickest cushion (306 μm thick and 8.4 cell layers) but exhibited only the third highest colonization level (80 %). However, Angiopteris and Osmunda gametophytes exhibited colonization levels of over 95 % and much thicker cushions compared to the gametophytes of Plagiogyriaceae, Gleicheniaceae, and Cyatheaceae: 807 μm thick and 14 cell layers for A. lygodiifolia and 470 μm thick and 10 cell layers for O. japonica (Ogura-Tsujita et al. 2013). In addition, preliminary examinations indicated that the terrestrial cordate gametophytes of some species of polypods, whose cushions are relatively thin, have low levels of AM fungal colonization. For example, Polystichum tripteron (Dryopteridaceae), with a thin cushion of 4.3 cell layers, exhibited only 16 % colonization (4/25 individuals) (Ayako Suzuki et al. unpublished data). Together, these findings suggest that the level of AM fungal colonization may indeed be related to cushion thickness. However, the data available for gametophytes of leptosporangiate ferns are quite limited. More extensive investigations across a wide spectrum of polypod groups (sensu Pryer et al. 2004) are needed to clarify the interrelationship between AM fungal colonization and cushion thickness,.
To our knowledge, the present study is the first report of the morphology of wild gametophytes in the Plagiogyriaceae and Cyatheaceae. Studies of the comparative morphology of fern gametophytes have typically used gametophytes cultured from spores, rather than wild gametophytes, because field-collected gametophytes are difficult to identify to the species, genus, or even family level due to insufficient morphological characters. However, recently established DNA bar-coding techniques have facilitated the identification of field-collected gametophytes (Ebihara et al. 2013). The present examination of wild gametophytes showed that their morphology is nearly identical to cultured gametophytes (Stokey and Atkinson 1956a for O. banksiifolia; Stokey 1950 for Gleicheniaceae; Stokey and Atkinson 1956b for Plagiogyria; Khare and Chandra 1995 for Cyathea). Cultured and wild gametophytes did not differ in the locations of reproductive organs, archegonia, or antheridia. Cushion thickness tended to be thinner in wild compared to cultured gametophytes: 7 vs. 18 cell layers for O. banksiifolia; 8 vs. 10–12 or 7–9 cell layers, respectively, for Gleicheniaceae; 6 vs. 4–7 or 6–7 cell layers for Plagiogyria; and 6 vs. 7–8 cell layers for Cyathea (Stokey and Atkinson 1956a for O. banksiifolia; Stokey 1950 for Gleicheniaceae; Stokey and Atkinson 1956b for Plagiogyria; Khare and Chandra 1995 for Cyathea).
In conclusion, previous mycorrhizal research on pteridophyte gametophytes has primarily focused on underground mycoheterotrophic gametophytes, such as the Lycopodiaceae and Ophioglossaceae (Duckett and Ligrone 1992; Schmid and Oberwinkler 1993; Winther and Friedman 2007), whereas the mycorrhizal associations of aboveground photosynthetic gametophytes remain largely unknown. The present study of five genera from four families of the Osmundales and Gleicheniales, and the Cyatheales, showed that taxonomically divergent photosynthetic gametophytes, which were mainly terrestrial and ground-dwelling, are colonized by AM fungi. These results suggest that mycorrhizal associations with AM fungi could widely occur among pteridophyte gametophytes, although more taxonomically and ecologically divergent species including epiphytic and aquatic gametophytes need to be examined. Because the tiny gametophytes exhibit simple rhizoid structures instead of root systems, AM fungal associations may play an important role in water and nutrient uptake in pteridophyte gametophytes.
References
Bierhorst DW (1971) Morhology of vascular plants. Macmillan, New York
Boullard B (1957) La mycotrophie chez les pteridophytes. Sa fréquence, ses caractères, sa signification. Botaniste 41:5–185
Boullard B (1979) Considération sur la symbiose fongique chez les pteridophytes. Syllogeus 19:1–59
Bower FO (1923) The ferns (Filicales). Vol. 1. Analytical examination of the criteria of comparison. Cambridge Univ Press, Cambridge
Campbell DH (1908a) Symbiosis in fern prothallia. Amer Nat 42:154–165
Campbell DH (1908b) The prothallium of Kaulfussia and Gleichenia. Ann Jard Bot Buitenzorg 8:69–102
Cooper KM (1976) A field survey of mycorrhizas in New Zealand ferns. New Zealand J Bot 14:169–181
Duckett JG, Ligrone R (1992) A light and electron microscope study of the fungal endophytes in the sporophyte and gametophyte of Lycopodium cernuum with observations on the gametophyte–sporophyte junction. Can J Bot 70:58–72
Ebihara A, Iwatsuki K, Kurita S, Ito M (2002) Systematic position of Hymenophyllum rolandi-principis Rosenst. or a monotypic genus Rosenstockia Copel. (Hymenophyllaceae) endemic to New Caledonia. Acta Phytotax Geobot 53:35–49
Ebihara A, Farrar DR, Ito M (2008) The sporophyte-less filmy fern of eastern North America Trichomanes intricatum (Hymenophyllaceae) has the chloroplast genome of an Asian species. Am J Bot 95:1645–1651
Ebihara A, Nitta JH, Ito M (2010) Molecular species identification with rich floristic sampling: DNA barcoding the pteridophyte flora of Japan. PLoS ONE 5:e15136
Ebihara A, Yamaoka A, Mizukami N, Sakoda A, Nitta JH, Imaichi R (2013) A survey of the fern gametophyte flora of Japan: frequent independent occurrences of noncordiform gametophytes. Am J Bot 100:735–743
Farrar DR, Dassler C, Watkins JE Jr, Skelton C (2008) Gametophyte ecology. In: Ranker TA, Haufler CH (eds) Biology and evolution of ferns and lycophytes. Cambridge Univ Press, Cambridge, pp 222–256
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 39:783–791
Finlay RD, Lindahl BD, Taylor AFS (2008) Responses of mycorrhizal fungi to stress. In: Avery S, Stratford M, van West P (eds) Stress in yeasts and filamentous fungi. Elsevier, Amsterdam, pp 201–220
Gemma JN, Koske RE, Flynn T (1992) Mycorrhizae in Hawaiian pteridophytes: occurrence and evolutionary significance. Am J Bot 79:843–852
Hasebe M, Wolf PG, Pryer KM, Ueda K, Ito M, Sano R, Gastony GJ, Yokoyama J, Manhart JR, Murakami N, Crane EH, Haufler CH, Hauk WD (1995) Fern phylogeny based on rbcL nucleotide sequences. Am Fern J 85:134–181
Horn K, Franke T, Unterseher M, Schnittler M, Beenken L (2013) Morphological and molecular analyses of fungal endophytes of achlorophyllous gametophytes of Diphasiastrum alpinum (Lycopodiaceae). Am J Bot 100:2158–2174
Jernstedt JA, Cutter EG, Gifford EM, Lu P (1992) Angle meristem origin and development in Selaginella martensii. Ann Bot 69:351–363
Kessler M, Jonas R, Strasberg D, Lehnert M (2010) Mycorrhizal colonization of ferns and lycophytes on the island of La Reunion in relation to nutrient availability. Basic Appl Ecol 11:329–336
Khare PB, Chandra S (1995) Gametophyte morphology of an endangered species of Cyathea SM. Indian Bot Soc 174:103–106
Koske RE, Gemma JN (1989) A modified procedure for staining roots to detect V-A mycorrhizas. Mycol Res 92:486–488
Krüger M, Krüger C, Walker C, Stockinger H, Schüßler A (2012) Phylogenetic reference data for systematics and phylotaxonomy of arbuscular mycorrhizal fungi from phylum to species level. New Phytol 193:970–984
Lara-Pérez LA, Valdés-Baizabal MD, Noa-Carrazana JC, Zulueta-Rodríguez R, Lara-Capistrán L, Andrade-Torres A (2015) Mycorrhizal associations of ferns and lycopods of central Veracruz, Mexico. Symbiosis 65:85–92
Lee J, Lee S, Young JPW (2008) Improved PCR primers for the detection and identification of arbuscular mycorrhizal fungi. FEMS Microbiol Ecol 65:339–349
Lehnert M, Kottke I, Setaro S, Pazmiño LF, Suárez JP, Kessler M (2009) Mycorrhizal associations in ferns from southern Ecuador. Am Fern J 99:292–306
Ligrone R, Carafa A, Lumini E, Bianciotto V, Bonfante P, Duckett JG (2007) Glomeromycotean associations in liverworts: a molecular, cellular and taxonomic analysis. Am J Bot 94:1756–1777
Maddison DR, Maddison WP (2003) MacClade: analysis of phylogeny and character evolution, Version 4.06. Sinauer Associates, Sunderland
Marschner H, Dell B (1994) Nutrient uptake in mycorrhizal symbiosis. Plant Soil 159:89–102
Muthukumar T, Prabha K (2013) Arbuscular mycorrhizal and septate endophyte fungal associations in lycophytes and ferns of south India. Symbiosis 59:15–33
Muthuraja R, Muthukumar T, Sathiyadash K, Uma E, Priyadharsini P (2014) Arbuscular mycorrhizal (AM) and dark septate endophyte (DSE) fungal association in lycophytes and ferns of the Kolli Hills, Eastern Ghats, Southern India. Am Fern J 104:67–102
Ogura-Tsujita Y, Sakoda A, Ebihara A, Yukawa T, Imaichi R (2013) Arbuscular mycorrhiza formation in cordate gametophytes of two ferns, Angiopteris lygodiifolia and Osmunda japonica. J Plant Res 126:41–50
Öpik M, Vanatoa A, Vanatoa E, Moora M, Davison J, Kalwij JM, Reier Ü, Zobel M (2010) The online database MaarjAM reveals global and ecosystemic distribution patterns in arbuscular mycorrhizal fungi (Glomeromycota). New Phytol 188:223–241
Pryer KM, Schuettpelz E, Wolf PG, Schneider H, Smith AR, Cranfill R (2004) Phylogeny and evolution of ferns (Monilophytes) with a focus on the early leptosporangiate divergence. Am J Bot 91:1582–1598
Raghavan V (1989) Developmental biology of fern gametophytes. Cambridge Univ Press, Cambridge
Read DJ, Duckett JG, Francis R, Ligrone R, Russell A (2000) Symbiotic fungal associations in ‘lower’ land plants. Phil Trans R Soc Lond B 355:815–831
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574
Schmid E, Oberwinkler F (1993) Mycorrhiza-like interaction between the achlorophyllous gametophyte of Lycopodium clavatum L. and its fungal endophyte studied by light and electron microscopy. New Phytol 124:69–81
Schmid E, Oberwinkler F (1995) A light- and electron-microscopic study on a vesicular–arbuscular host–fungus interaction in gametophytes and young sporophytes of the Gleicheniaceae (Filicales). New Phytol 129:317–324
Schüßler A, Walker C (2010) The Glomeromycota; a species list with new families and new genera. http://www.amf-phylogeny.com
Schwarzott D, Walker C, Schüßler A (2001) Glomus, the largest genus of the arbuscular mycorrhizal fungi (Glomales), is nonmonophyletic. Mol Phylogenet Evol 21:190–197
Simon L, Lalonde M, Bruns TD (1992) Specific amplification of 18S fungal ribosomal genes from vesicular–arbuscular endomycorrhizal fungi colonizing roots. Appl Environ Microbiol 58:291–295
Stokey AG (1950) The gametophyte of the Gleicheniaceae. Bull Torrey Club 34:243–263
Stokey AG, Atkinson LR (1956a) The gametophyte of the Osmundaceae. Phytomorphology 6:19–40
Stokey AG, Atkinson LR (1956b) The gametophytes of Plagiogyria glauca (BL.) Mett. and P. semicordata (PR.) Christ. Phytomorphology 6:239–249
Turnau K, Anielska T, Jurkiewicz A (2005) Mycothallic/mycorrhizal symbiosis of chlorophyllous gametophytes and sporophytes of a fern, Pellaea viridis (Forsk.) Prantl (Pellaeaceae, Pteridales). Mycorrhiza 15:121–128
Turnau K, Przybyłowicz WJ, Ryszka P, Orłowska E, Anielska T, Mesjasz-Przybyłowicz J (2013) Mycorrhizal fungi modify element distribution in gametophytes and sporophytes of a fern Pellaea viridis from metaliferous soils. Chemosphere 92:1267–1273
van der Heijden MGA, Bardgett RD, van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310
Wang B, Qiu YL (2006) Phylogenetic distribution and evolution of mycorrhizas in land plants. Mycorrhiza 16:299–363
West B, Brandt J, Holstien K, Hill A, Hill M (2009) Fern-associated arbuscular mycorrhizal fungi are represented by multiple Glomus spp.: do environmental factors influence partner identity? Mycorrhiza 19:295–304
Winther JL, Friedman WE (2007) Arbuscular mycorrhizal symbionts in Botrychium (Ophioglossaceae). Am J Bot 94:1248–1255
Winther JL, Friedman WE (2008) Arbuscular mycorrhizal associations in Lycopodiaceae. New Phytol 177:790–801
Winther JL, Friedman WE (2009) Phylogenetic affinity of arbuscular mycorrhizal symbionts in Psilotum nudum. J Plant Res 122:485–496
Zhang Y, Guo LD, Liu RJ (2004) Arbuscular mycorrhizal fungi associated with common pteridophytes in Dujiangyan, southwest China. Mycorrhiza 14:25–30
Zhi-wei ZW (2000) The arbuscular mycorrhizas of pteridophytes in Yunnan, southwest China: evolutionary interpretations. Mycorrhiza 10:145–149
Acknowledgments
The authors thank Kurama-dera Temple, Kyoto, for the permission to collect Plagiogyria gametophytes from Mt. Kurama, Prof. Masayuki Takamiya of Kumamoto University and Mr. Tadakatsu Maruno for helping a field expedition in Kagoshima, and Assistant Prof. Masahide Yamato of Chiba University for the helpful advice on AM fungal phylogeny. This study was funded by JSPS KAKENHI Grant Number 24570116 (to Ryoko Imaichi) and 2440185 (to Yuki Ogura-Tsujita).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ogura-Tsujita, Y., Hirayama, Y., Sakoda, A. et al. Arbuscular mycorrhizal colonization in field-collected terrestrial cordate gametophytes of pre-polypod leptosporangiate ferns (Osmundaceae, Gleicheniaceae, Plagiogyriaceae, Cyatheaceae). Mycorrhiza 26, 87–97 (2016). https://doi.org/10.1007/s00572-015-0648-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-015-0648-1