Abstract
Radiotherapy (RT) is considered as a standard in the treatment of most solid cancers, including glioblastoma, lung, breast, rectal, prostate, colorectal, cervical, esophageal, and head and neck cancers. The main challenge in RT is tumor cell radioresistance associated with a high risk of locoregional relapse and distant metastasis. Despite significant progress in understanding mechanisms of radioresistance, its prediction and overcoming remain unresolved. This review presents the state-of-the-art for the potential universal biomarkers correlated to the radioresistance and poor outcome in different cancers. We describe radioresistance biomarkers functionally attributed to DNA repair, signal transduction, hypoxia, and angiogenesis. We also focus on high throughput genetic and proteomic studies, which revealed a set of molecular biomarkers related to radioresistance. In conclusion, we discuss biomarkers which are overlapped in most several cancers.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Radiation therapy (RT) is the most effective method of cytotoxic treatment based on ionizing radiation (Baskar and Itahana 2017). RT plays a key role in the treatment of many cancers, and approximately 50% of cancer patients are estimated to receive RT (Harrington et al. 2011). Indications for the appointment of RT include (a) radical treatment, (b) adjuvant therapy after surgery to eliminate residual disease, and (c) palliative care (Harrington et al. 2011). As an independent treatment strategy, RT can be used at earlier stages of the disease if surgical intervention is impossible (Das et al. 2010; Swanton et al. 2021). The primary purpose of radical RT is to achieve complete eradication of tumor cells by delivering sufficient doses of radiation. The levels of radical doses depend on the histological type of tumor, mitotic activity, and the degree of cell differentiation.
Tumor-specific radioresistance is a critical factor for RT failure and the development of locoregional relapse and distant metastases (Huang and Zhou 2020). The frequency of relapses after RT or chemoradiation therapy (CRT) varies between different cancers. It is 5.2% in head and neck cancer (Lindegaard et al. 2020), 16–20% in prostate cancer (Grün et al. 2020), 55% in cervical cancer (Ning et al. 2018), 8.63% and 4.31% (local recurrence and regional recurrence after RT) in breast cancer, and 6.5% in rectal cancer (Yu et al. 2008; Couch and Hemingway 2016; Huang et al. 2017).
The search for effective clinical, morphological, and molecular criteria for predicting the success of the treatment at the initial and follow-up steps is one of the key tasks in oncology. Despite the main and common molecular mechanisms of radioresistance being clear, the establishment of effective biomarkers is still a significant challenge. The antigen of squamous cell carcinoma (SCC-Ag) was recently established for determining a residual disease after treatment and the effectiveness of prescribed therapy, but it is not related to radioresistance (Yagi et al. 1987; Petrelli et al. 1988; Yoshimura et al. 1990). The development of novel radioresistance biomarkers universal for all cancers seems relevant and demanded.
Radiation induces different alterations related to DNA damage and repair, cell cycle regulation, reactive oxygen species (ROS), hypoxia, tumor microenvironment (TME), angiogenesis, and epigenetic regulation (Willers et al. 2013; Tang et al. 2018). Plenty of existing studies investigates the radiation effects in vitro and in vivo. An increasing number of studies have also been trying to establish prognostic and predictive biomarkers of radioresistance in cancer patients.
In the present review, we analyze the available data on the potential biomarkers of radioresistance in different cancers defined as an unfavorable outcome in most cases. In particular, we focus on clinical studies demonstrating molecular factors associated with radioresistance. We also review genomic, transcriptomic, and proteomic studies that revealed the sets of molecular factors prognostic for cancer after RT. In conclusion, we discuss the complications in the search for a single biomarker and explain which attempts should be made in the future to overcome cancer radioresistance.
DNA damage and repair
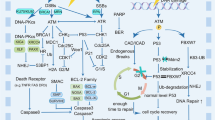
In general, the success or failure of RT is determined by five R radiobiology: repair of DNA damage, cell redistribution in the cell cycle, repopulation, reoxygenation of hypoxic tumor areas, and radiosensitivity (Pajonk et al. 2010; Goedegebuure et al. 2019). A common feature of ionizing radiation is the induction of DNA damage directly leading to cancer cell death. Critical mechanisms of DNA damage include cleavage at the sugar-phosphate linkage region of a DNA polynucleotide chain (single-strand break, SSB), breaks on both DNA chains in adjacent or nearly adjacent sugar-phosphate-binding sites (double-strand break, DSB), intramolecular and intermolecular linkage between DNA or DNA-protein, degradation of organic bases, loss of a purine or pyrimidine base, and breaking of hydrogen bonds resulting in permanent deformation of DNA structure (Liu et al. 2020b). Among them, DSBs are the most lethal lesions which trigger a series of cellular DNA damage responses (DDRs), including the activation of DNA damage sensing and early transduction pathways, cell cycle arrest, and DNA repair (Huang and Zhou 2020). DSBs can be repaired either by homologous recombination (HR) or via nonhomologous end joining (NHEJ). HR is a crucial pathway for the accurate repair of DSBs and maintaining genomic stability (Shrivastav et al. 2008).
Several factors responsible for DNA repair are associated with radioresistance in several clinical studies (Table 1). Pretreatment protein expression of XRCC2, involved in NHEJ-based repair of DSBs, negatively correlates with 3-year overall survival (OS) after RT in patients with locally advanced rectal cancer (Qin et al. 2015). Among patients who achieved a pathological response, 66.7% of cases were negative for XRCC2 expression, and 33.3% were positive for XRCC2 expression (Qin et al. 2015). In NSCLC patients, XRCC2 SNPs were associated with RT response and OS (Yin et al. 2011; Yang and Liu 2020). The protein expression of BRCC3 and YB-1, which are involved in DNA repair, was upregulated in pretreatment biopsies of patients with nasopharyngeal carcinoma (NPC) who had worse OS and a higher risk of recurrence (Tay et al. 2009; Tu et al. 2015). Increased protein expression of Ku80 (XRCC5), a key mediator of DSB repair, correlates with locoregional recurrence in post-RT specimens of patients with HPV-negative head and neck squamous cell carcinoma (HNSCC) (Moeller et al. 2011).
In NPC patients treated with conventional RT, the radioresistance-associated biomarkers belonging to the heterotrimeric replication protein A (RPA) complex were demonstrated. The expression of DNA repair marker RPA3 in post-treatment specimens was higher in radioresistant NPC patients who experienced local recurrence. Analysis of TCGA data indicated that high pretreatment RPA3 expression also correlates with poor OS and a high recurrence rate in patients with HNSCC after RT (Qu et al. 2017).
The impact of copy number alterations in DDR genes on clinical outcome based on biochemical recurrence (defined as a rise in PSA level) was examined in patients with prostate cancer (PC) receiving radical RT (Berlin et al. 2014). Evaluation of pre-RT biopsies by a comparative genomic hybridization revealed CNAs predominantly in DDR-sensing genes: NBN (15.1%), ATR (8.6%), PRKDC (7.9%), and ATM (5.8%). In addition, NBN gain was an independent prognostic factor for 5-year biochemical recurrence (Berlin et al. 2014).
The molecular mechanisms of DDR are well known, and many in vitro studies have established them as first-line processes to protect tumors from irradiation inducing radioresistance. Above mentioned evidence indicates that increased pre-and post-treatment expression of DDR-associated factors correlates to poor prognosis; however, this correlation is not specific to cancer type. Only XRCC2 expression was predictive for radioresistance in several cancers (Fig. 1, Table 1). As far as DNA repair is complex, clinical investigation of other DDR-associated factors can help search for sensitive and effective biomarkers or signatures for tumor response to RT.
Overlapping radioresistance biomarkers between different human cancers. Nine common biomarkers are shown in the seven most representative cancer types treated with RT. BC breast cancer, CC cervical cancer, ESCC esophageal squamous cell carcinoma, HNSCC head and neck squamous cell carcinoma, NSCLC non-small cell lung carcinoma, PC prostate cancer, RC rectal cancer
Tyrosine kinases and cell cycle control
Tyrosine kinases (TKs) are enzymes that regulate cell survival and cell proliferation in response to stress (Bhattacharya et al. 2018). TKs are primarily classified into receptor tyrosine kinases (RTKs) (e.g., EGFR, PDGFR, HER-2, FGFR, and IGF-1R) and non-receptor tyrosine kinases (NRTK): SRC, ABL, FAK, AKT, and Janus kinase (Paul and Mukhopadhyay 2004). They play a crucial role in radiation-activated DNA repair and cell survival (Bhattacharya et al. 2018) (Table 1).
HER-2 overexpression is related to distant metastasis of rectal cancer after neoadjuvant RT, especially in patients with poor response to treatment (Yao et al. 2014). Rectal, breast, and prostate cancer patients with low pretreatment protein and negative mRNA expression of IGF-1R display improved sensitivity to RT and decreased post-RT recurrence rate (Turner et al. 1997; Wu et al. 2014; Aleksic et al. 2017). High protein expression of Pim-1 and gene expression of FAK/PTK2 in post-treatment samples is associated with poor prognosis and worse DFS in patients with HNSCC treated with RT (Peltola et al. 2009; Skinner et al. 2016). A high level of phosphorylated AKT (pAKT), regulating DDR, in samples obtained after RT correlates with poor OS/PFS in patients with squamous cell cervical carcinoma, glioblastoma, and advanced HNSCC (Kim et al. 2006; Suzuki et al. 2007; Freudlsperger et al. 2015). Oppositely, a high pretreatment level of pAKT is associated with a lower incidence of recurrence and better RT response in breast and rectal cancers (Davies et al. 2011; Sjöström et al. 2020).
The outcome of irradiation depends on the regulation of the cell cycle (Otani et al. 2016). Cells in the G2/M phase are more vulnerable to irradiation than cells in G1 or S phases, where rapid DNA repair can successfully result in radioresistance (Pawlik and Keyomarsi 2004). Tumors with high expression of checkpoint serine/threonine kinase 1 (Chk1), a key cell cycle mediator involved in DNA repair, display radioresistant phenotype. DNA damage induces the activation of Chk1, facilitating the DDR and initiation of the cell cycle checkpoints (Patil et al. 2013). High expression of Chk1 in pretreatment samples is significantly associated with shorter progression-free survival (PFS) and shorter time to local recurrence in patients with gastric cancer and breast cancer treated with CRT and RT, respectively (Alsubhi et al. 2016; Bargiela-Iparraguirre et al. 2016). High protein expression of P16INK4A, a cyclin-dependent kinase (CDK) inhibitor belonging to tumor suppressors, in pre-RT samples is prognostic for the improved 5-year OS and DFS rates in patients with cervical cancer receiving adjuvant RT or concurrent CRT (Fu et al. 2018). CDKs are essential for cell cycle progression through the G1-S phases and initiation of DNA repair. CDK inhibition combined with RT is proposed to diminish the radioresistance development (Johnson and Shapiro 2010). In cervical cancer, high expression of KLF4 (cell cycle regulator in G1–S phases) after treatment is found in radiation-resistant patients and associated with increased rates of local recurrence and distant metastases (Liu et al. 2017a; Hou et al. 2017; Yang et al. 2020; Köster et al. 2020).
Thus, IGF-1R, belonging to RTKs, and pAKT, a non-receptor tyrosine kinase, possess predictive RT value in several cancers (Fig. 1, Table 1). However, it is questionable whether TKs can be effective biomarkers as they regulate multiple signaling pathways. Nevertheless, due to the involvement of RTKs and cell cycle control proteins in DNA repair, combining RT with TK inhibitors can be a promising approach to increase RT efficacy. Recent studies showed that RTKs and CDK inhibitors display remarkable anti-tumor efficacy in lung, colorectal, hepatocellular, renal, and breast cancers, as well as leukemia and melanoma (Pottier et al. 2020; Zhang et al. 2021).
Hypoxia and metabolism
Hypoxia is a common feature of solid tumors resulting from the imbalance between oxygen availability and consumption (Wang et al. 2019b). Hypoxia generates an intratumoral oxygen gradient that contributes to tumor plasticity and heterogeneity and activates the DDR pathways (Jing et al. 2019). Thus, hypoxic cancer cells acquire a more aggressive and metastatic phenotype and become resistant to any cytotoxic treatment, including RT and CRT (Zhu et al. 2016). On the contrary, molecular oxygen may react with radiation-induced DNA radicals to generate DNA damage. Therefore, well-oxygenated cancer cells are more sensitive to irradiation than hypoxic ones (Willers et al. 2013).
Hypoxia-inducible factor-1α (HIF-1α) is an important transcription factor, which is increased in hypoxic conditions. RT promotes HIF-1 activation by vascular damage or reactive oxygen species (ROS) (Huang and Zhou 2020). In cervical cancer, HIF-1α expression after CRT is absent in cases with complete response (CR) and is found in 63% of patients with partial response (Zhu et al. 2016). High HIF-1α expression before CRT is considered a predictive biomarker for poor response to preoperative CRT in squamous cervical, oropharyngeal, esophageal, and laryngeal cancers, but for better prognosis in oral squamous carcinoma (Sun et al. 2013; Zhu et al. 2016; Swartz et al. 2021). Increased level of HIF-1α in pre-CRT/RT samples is a poor prognostic biomarker for OS and metastasis-free survival in patients with cervical cancer and NPC (Hui et al. 2002; Kim et al. 2013; Chen et al. 2014).
ROS, such as superoxide anion (O2−), hydroxyl radicals (OH−), and hydrogen peroxide (H2O2), are generated by water radiolysis in extracellular environments and are toxic to cancer cells and adjacent normal tissues (Zou et al. 2017). ROS can induce genetic instability (Perillo et al. 2020) (Figure 2). The high expression of ROS modulator 1 (ROMO1) after RT is associated with worse PFS and OS and shorter locoregional recurrence in NSCLC patients treated with definitive RT (Kong et al. 2019). Expression of coenzyme A synthase (COASY) measured after RT is associated with radioresistance in rectal cancer patients. Patients with no response to CRT have significantly higher COASY expression than other patients (Ferrandon et al. 2020). Low expression of pH2AX, a damage-associated protein, and MAP17, a ROS-related protein, is associated with better OS (Rivero et al. 2018). In pretreatment biopsy samples, protein expression of oxidative stress-associated factor RKIP is significantly downregulated, while the level of NRF2 and NQO1 is upregulated in radioresistant NPC. A low level of RKIP and a high level of NRF2 and NQO1 correlate to reduced OS (Huang et al. 2020). In patients with locally advanced esophageal squamous-cell carcinoma (ESCC) and rectal cancer, high NRF2 expression indicates a poor response to RT/CRT and unfavorable survival (Wang et al. 2020; O’Cathail et al. 2021).
Overlapping radioresistance biomarkers among the key processes related to radioresistance. Irradiation results in ROS generation and DNA damage. In response to irradiation, DNA repair is activated, accompanied by increased tyrosine kinase activity via multiple signal transduction pathways. The effects of ionizing radiation depend on the oxygenation/hypoxia metabolic balance in the tumor. Oxygenation induces ROS generation, which triggers DNA damage, but can activate the expression of pro-angiogenic genes. In contrast, hypoxia interferes with the effect of RT and can also stimulate the upregulation of angiogenic factors and immunosuppressive immune responses. The suppression of anti-tumor immune response is followed by the RT-induced metabolic and transcriptional changes in the pro-tumor phenotypes of tumor-associated macrophages (TAMs), cancer-associated fibroblasts (CAFs), and Tregs. TAMs and Tregs additionally promote angiogenesis. Boxes in blue reflect intracellular processes, boxes in grey demonstrate extracellular components, which trigger radioresistance in tumor cells. Common radioresistance biomarkers are given in the corresponding boxes and highlighted in red
Analysis of TCGA data revealed that HNSCC patients with high ACLY (ATP citrate lyase) expression experience poor OS (Göttgens et al. 2019). Expression of IGF-1 and GLUT1 in post-treatment samples correlates with poor OS in patients with cervical cancer who underwent RT/CRT (Moreno-Acosta et al. 2017). In breast cancer, expression of p-S6K1, a critical downstream effector of the mTOR pathway, before treatment correlates with decreased locoregional recurrence-free survival (Choi et al. 2020b).
Expression of COX-2, regulating oxidative phosphorylation, is a poor prognostic factor for patients with cervical cancer. Five‐year OS and DFS are decreased in COX-2-positive patients (Kim et al. 2002; Chen et al. 2005). CC patients with the double expression of iNOS- and COX-2 after RT have the poorest survival rates (Chen et al. 2005). COX-2 expression negatively correlates with complete response to RT and local recurrence in rectal cancer and ESCC (Zhang et al. 2017b; Berbecka et al. 2021).
Thus, tumor metabolic status determines radiotherapeutic sensitivity through tumor oxygenation and ROS production. Since ROS is non-specifically induced by radiation, it is challenging to use ROS level as a predictive biomarker. Oppositely, hypoxia-associated factors, such as HIF-1α and COX-2, and NRF2, a transcriptional activator of antioxidant genes, could be used to predict failed RT response in several human cancers (Figure 1, Table 1).
Tumor microenvironment
Radiation affects not only cancer cells but also the tumor microenvironment (TME) (Barker et al. 2015) (Fig. 2). The TME comprises diverse cell types, including cancer-associated fibroblasts (CAFs), endothelial cells, tumor-associated macrophages (TAMs), and other immune cells (Balkwill et al. 2012; Fridman et al. 2017). Complex crosstalk between cancer cells and components of TME facilitates tumor growth, angiogenesis, invasion, and metastasis (Stakheyeva et al. 2017; Larionova et al. 2019).
CAFs, a significant component of tumor stroma, promote cancer cell recovery and tumor relapse after radiotherapy (Wang et al. 2017). CAFs are responsible for the synthesis of extracellular matrix (ECM) proteins, including matrix metallopeptidases (MMPs), and secretion of cytokines and growth factors that regulate tumor proliferation, invasion, and metastasis (Wang et al. 2019b; Ansems and Span 2020). CAFs can recruit macrophages and potentiate their biological functions by modifying the ECM. The interaction of CAFs and TAMs establishes immunosuppressive conditions in TME (Ansems and Span 2020). Radiation-treated fibroblasts display increased expression of factors involved in cell cycle arrest, DNA repair, ROS scavenging, ECM remodeling, and Wnt and IGF signaling pathways (Rødningen et al. 2005; Wang et al. 2019b). Different factors expressed by CAFs after RT mediate subsequent fibrosis, EMT/invasion, and treatment resistance. Fibrosis, in turn, supplies proliferation and invasion signals to cancer cells, leading to radioresistance and tumor progression (Ansems and Span 2020).
CAF-secreted factors are associated with the prognosis of different human cancers (Ham et al. 2021). High gene expression of stromal CXCL12 and FAP, essential for the fibrotic process, correlates with poor OS in rectal cancer with preoperative CRT (Saigusa et al. 2010, 2011). High protein expression of TGF-β in CAFs is associated with poor OS in ESCC patients treated with CRT (Zhang et al. 2017a). High post-treatment expression of PLOD3 (procollagen-lysine, 2-oxoglutarate 5-dioxygenase 3), involved in fibrotic processes and tissue remodeling, is associated with poor 5-year survival in lung cancer patients treated with RT (Baek et al. 2019).
TAMs are a crucial component of innate immunity in TME, promoting tumor growth, angiogenesis, metastasis, and tumor regrowth after chemo- and radiotherapy (Larionova et al. 2019, 2020). Numerous tumor models demonstrate that irradiation induces monocyte/macrophage infiltration via CXCL12, CCL2, and CSF1-dependent mechanisms, and accumulation of M2-like TAMs promotes tumor vasculogenesis and immunosuppression that limits RT efficacy (Xu et al. 2013; Genard et al. 2017; Wu et al. 2017). Increased CD68+ macrophage infiltration is associated with poor OS and DFS in OSCC patients who received radiation (Ai et al. 2021). High expression of pro-tumor M2 macrophage markers indicates poor survival and metastasis (Larionova et al. 2020). Cervical cancer and HNSCC patients with increased M2 marker CD163 show unfavorable disease outcomes after RT/CRT (Balermpas et al. 2014; Lippens et al. 2020). High expression of HIF-2α in TAMs correlates to low DFS rates and increased risk of local recurrence (Kawanaka et al. 2008).
CD8+ cytotoxic T lymphocytes (CTLs) are the essential immune cells for killing cancer cells presenting MHC I molecules (Farhood et al. 2019). In NSCLC, increased CD8+ T-cell counts, CD4/Treg ratio, and higher CD103+ cell infiltration after RT/CRT are observed in responsive patients (complete and partial response vs. stable disease) and associated with better OS and DFS (Komdeur et al. 2017; Liu et al. 2019; Boulle et al. 2020). The cervical cancer‐specific survival is significantly higher in patients with increased CD8 post-treatment scores, high CD8/CD4 and CD163/CD68 ratios, or PD-L1 expression in more than 5% of immune cells (Lippens et al. 2020). Contradictory results are obtained for the PD-L1 expression in NSCLC. Programmed cell death-1 (PD-1) is a co-stimulatory receptor of the CD28 family that plays a crucial role in tumor cell tolerance (Keir et al. 2006). PD-1/PD-L1 interaction inhibits T-lymphocyte proliferation, survival, and effector functions, induces apoptosis of antigen-presenting T cells, promotes differentiation of CD4+ T cells into FoxP3+ regulatory cells, and provides resistance of tumor cells to a cytotoxic response (Iwai et al. 2002; Wang et al. 2008). In patients with NSCLC who received RT/CRT, lower baseline soluble PD-L1 level correlates to the most prolonged OS and objective response to treatment (Zhao et al. 2017; Sui et al. 2021). Oppositely, high post-treatment PD-L1 expression in lung cancer tissue predicts a greater radiosensitivity and better outcome (Fiedler et al. 2018).
Other immune molecules belonging to different function classes were found to be associated with prognosis and to predict the response to RT. NF-κB is a transcriptional factor that regulates multiple aspects of innate and adaptive immune functions and serves as a pivotal mediator of the inflammatory response (Liu et al. 2017b). The expression of NF-κB positively correlates with increased rates of distant metastases, locoregional failure, and overall (local and distant) relapse in cervical cancer patients (Garg et al. 2010). Increased pretreatment expression of CCR6, an inflammatory CC chemokine receptor, is observed in the non-pCR patients with rectal cancer who received CRT (Chang et al. 2018). Negative expression of ICAM-3, an adhesion molecule for leukocytes, was found in normal tissue compared to tumor tissue, and in 65% of radiosensitive cervical cancer cases. In comparison, ICAM-3 was detected in 83% of radioresistant tumors and associated with poor 5-year PFS (Chung et al. 2005). High pretreatment protein expression of CD59, a membrane-bound complement regulatory protein, indicates poor OS and DFS in ESCC patients who received RT (Zhou et al. 2018). The possible mechanism of CD59-mediated radioresistance can be related to induced DDR in cancer cells (Zhou et al. 2018). The level of secreted protein CD166, belonging to the immunoglobulin superfamily, was detected in the serum of NPC patients before treatment. The concentration of CD166 is higher in the radioresistant cases possessing local recurrent disease after RT (Lin et al. 2017). The expression of annexin A1 (ANXA1), a glucocorticoid-induced anti-inflammatory protein, after treatment, oppositely, is significantly decreased in radioresistant NPC compared to radiosensitive tumors (Liao et al. 2018).
RT induces tissue damage and triggers activation of stromal components that can synergistically function along with therapy to kill cancer cells or support tumors. The balance shift to the tumor-supportive activity can be crucial for radioresistance development. However, there are still not enough data to address whether diverse factors produced by key players of TME—CAFs, TAMs, and T-lymphocytes—modulate RT response (Fig. 2). Further studies should focus on the detailed investigation of changes in the expression of growth factors, cytokines, soluble mediators, and surface receptors during RT and their role in radioresistance.
Pro-angiogenic factors
Effects of ionizing radiation strongly depend on the oxygenation of the tumor. As mentioned above, the well-vascularized and perfused tumors are more sensitive to RT due to the enhanced generation of ROS. Vice versa, hypoxic tumors with a lack of blood vessels prevent treatment efficiency (Overgaard 2007; Goedegebuure et al. 2019). Irradiation with different doses leads to varying effects on the vascular system. High dose irradiation (above 10 Gy) induces acute vascular damage caused by endothelial cell apoptosis (Park et al. 2012). It can lead to hypoxia and in the switch to the pro-angiogenic pathways. Fractionated low dose radiotherapy (per 2 Gy daily) positively affects the tumor vasculature and tissue perfusion due to decreased oxygen consumption (Park et al. 2012; Goedegebuure et al. 2019).
The critical factor regulating tumor angiogenesis is VEGF (Larionova et al. 2021) (Fig. 2). Angiogenesis is also associated with metalloproteinases (MMP-2, MMP-9, and MMP-14), tissue inhibitors of metalloproteinases (TIMP-1 and TIMP-2), thymidine phosphorylase (TP), urokinase plasminogen activator (uPA), osteopontin (OPN), and other molecules (Shih et al. 2002; DuBois and Demetri 2007; Larionova et al. 2021). Most of them are also crucial regulators of cancer invasion and metastasis.
Here, we collected the studies demonstrating that pro-angiogenic factors may serve as markers for radioresistance. Pretreatment VEGF serum level and tumor expression negatively correlate with the RT efficacy and recurrence-free, metastasis-free, and overall survival in NSCLC, cervical, and breast cancer patients (Manders et al. 2003; Fu et al. 2014). The contradictory results were shown in ESCC, where high pretreatment VEGF expression significantly correlates with a complete response to CRT (Yoon et al. 2011).
High plasma levels of hypoxic and angiogenic biomarkers OPN, VEGF, CEA, IL-6, CYFRA 21-1, and CA IX before CRT/RT are independent predictors for poor OS in patients with NSCLC (Dehing-Oberije et al. 2011; Ostheimer et al. 2014, 2018; Fu et al. 2014). Elevated pre-RT serum IL-6 level is associated with worse 2-year survival in NPC patients (Chow et al. 2003). In ESCC, IL-6 tumor overexpression correlates to poor CRT response, locoregional failure, and distant metastasis (Chen et al. 2013). In patients with inoperable or metastasized NSCLC who received RT/CRT, a low level of urokinase plasminogen activator PAI-1 before treatment is associated with significantly reduced OS and PFS. Combined low level of PAI-1 and high level of OPN demonstrate an additive prognostic impact on unfavorable NSCLC prognosis with increased risk of death (Ostheimer et al. 2018). In prostate cancer patients, increased pretreatment protein expression of HIF-1α, VEGF, and OPN is significantly associated with a higher risk of biochemical failure after RT (Vergis et al. 2008). Elevated pretreatment plasma and tumor OPN levels indicate poor RT response and higher locoregional and disease-specific mortality in HNSCC, NPC, ESCC, and cervical cancer (Overgaard et al. 2005; Hui et al. 2008; Snitcovsky et al. 2009; Etiz et al. 2013; Chiu et al. 2018).
In cervical cancer patients treated with CRT, high pretreatment protein expression of MMP-2 and TIMP-2 in tumor stroma and MMP-9 in tumor nest and stroma are significantly associated with poorer 12-year survival (Azevedo Martins et al. 2020). Increased pretreatment expression of pro-angiogenic chitinase-like protein YKL-40 is significantly associated with resistance to RT in glioblastoma patients (Pelloski et al. 2005).
Anti-angiogenic drugs (e.g., anti-VEGF agent bevacizumab) are widely used in first- and second-line therapy to improve cancer treatment efficacy (Larionova et al. 2021). Anti-angiogenic treatment is also proposed to increase RT efficiency (Goedegebuure et al. 2019). The accumulating data demonstrate that at least three pro-angiogenic factors, VEGF, IL-6, and OPN, are strongly related to RT failure in numerous solid tumors, making them promising universal biomarkers (Figs. 1 and 2, Table 1).
Cancer stem cells
Cancer stem cell (CSC) repopulation is considered an adaptive response to the cytotoxic effects of radiation (Willers et al. 2013). CSCs exhibit self-renewal, differentiation, and proliferation capacities similar to normal hematopoietic stem cells (Aponte and Caicedo 2017; Peitzsch et al. 2017). Self-renewal and metastatic potential of CSCs, which are crucial mechanisms of tumorigenesis, are regulated by multiple cytokines and growth factors produced by TME. Hypoxia protects CSCs from radiation, preventing the detrimental effect of ROS by upregulation of ROS scavengers (Peitzsch et al. 2014; Najafi et al. 2019). Key activities of CSCs are regulated by Wnt/β-catenin, Notch, Sonic Hedgehog, PTEN, TGF-ß, and other signaling pathways (Olivares-Urbano et al. 2020). Activation of Notch signaling by irradiation results in entering breast CSCs into the cell cycle and acquiring EMT and self-renewal properties (Venkatesh et al. 2018). In cervical cancer, posttreatment high expression of delta-like ligand 4 (DLL4), a transmembrane Notch ligand, is found in radiation-resistant patients (Liu et al. 2017a; Venkatesh et al. 2018; Wang et al. 2019c).
The findings of the CSC role in radioresistance are scarce, and further studies are warranted. Moreover, CSCs are highly heterogeneous and represented by different cell populations with specific phenotypical and functional features. Nevertheless, the question remains open whether the radioresistance degree varies between CSC subsets.
Epigenetic mechanisms
DNA methylation
Epigenetic mechanisms control diverse processes in tumors and have a crucial role in developing radiation resistance (Fardi et al. 2018). Radiotherapy changes the epigenetic landscape of cancer cells towards radioresistance and disease progression. DNA methylation, histone post-translational modifications, and chromatin remodeling are implicated in the control of gene expression related to DNA repair, cell survival and proliferation, evasion of apoptosis, and EMT regulation, thus protecting cancer cells from the cytotoxic effect of radiation (Cabrera-Licona et al. 2021).
DNA methyltransferases (DNMTs) regulate the chromosome stability and genome integrity, while overexpression of DNMTs in various tumors results in hypermethylation and oncogenic activation (Zhang and Xu 2017). High gene expression of DNMT3B predicts shorter OS in patients with NPC and HNSCC (Wu et al. 2020). In pretreatment samples, the nuclear protein expression of RUNX3, a downstream target of the TGF-β signaling pathway, is associated with complete or partial response to RT in ESCC patients. The hypermethylation and transcriptional repression of RUNX3 are found in 96.7% of irradiated ESCCs patients and are associated with low survival (Sakakura et al. 2007). Promoter methylation of tumor suppressor genes RASSF1/RASSF2A and the consequent activation of Ras/PI3K/Akt signaling are significantly related to poorer DFS in OSCC patients who underwent RT after surgery (Huang et al. 2009). The panel of eight hypermethylated genes, controlling cell proliferation and adhesion and associated with metastasis development, was found for determining complete response after RT in oropharyngeal cancer patients (Kurokawa et al. 2020). Pretreatment methylation of ROBO1, ULK4P3, MYOD1, LBX1, CACNA1A, IRX4, DPYSL3, and ELAVL2 genes distinguishes patients with a long time of DFS and OS and patients with tumor recurrence or progression after RT (Kurokawa et al. 2020).
Histone modification
Histones, central chromatin components, acquire diverse post-translational modifications, including acetylation/deacetylation, methylation, and phosphorylation, to form the epigenetic patterns underlying transcriptional processes (Cabrera-Licona et al. 2021).
The expression of histone deacetylases HDAC4 and HDAC6 negatively correlates with OS in patients with glioblastoma treated with RT (Marampon et al. 2017). H3K4me3 demethylase KDM5B, playing an essential role in repairing DSB, correlates with poor response to radiation in patients with lung cancer (Bayo et al. 2018). Hypoxia-induced KDM3A overexpression results in radioresistance in vitro and can indicate RT failure in ESCC patients (Macedo-Silva et al. 2020). Impaired gene expression of NuRD complex (the nucleosome remodeling and deacetylase) subunits (CHD4, CHD3, HDAC1, HDAC2, MTA2, MBD3, RBBP4, and RBBP7), involved in chromatin remodeling and histone deacetylase activity, is identified in patients with rectal cancer treated with CRT (Wang et al. 2019a). Post-treatment expression of CHD3 and CHD4 is higher in non-responding rectal cancer patients than in responders. CHD4 overexpression is also an independent prognostic factor for metastasis-free survival (Wang et al. 2019a).
A recent study indicated that HDAC activity significantly varies between different cancers (Sharda et al. 2020). Therefore, identifying histone code in each cancer type is essential to predict response to RT. Moreover, discovering epigenetic targets is needed to counteract RT in refractory cancers.
MicroRNAs and lncRNAs
Non-coding RNAs, including long non-coding RNAs (lncRNAs) and microRNAs (miRNAs), have been identified as key molecules in radiotherapy failure in many cancer types (Podralska et al. 2020). Recent studies emphasized extracellular miRNAs as potential liquid biomarkers of radioresistance (Nowicka et al. 2019).
The current data indicates the involvement of both serum and intratumor miRNAs in RT response and tumor progression in patients with lung, rectal, cervical, head and neck, and esophageal cancers. Post-treatment expression levels of miR-96, miR-130a, miR-25, miR-196a, and miR-191* are increased in the serum of patients with RT-treated NSCLC compared to healthy controls and associated with recurrence and low survival rate (Suh et al. 2015; Lv et al. 2020; Zheng et al. 2021). MiR-622 is upregulated in rectal cancer patients without tumor regression before CRT (Ma et al. 2015). In cervical cancer, the expression of oncogenic miR-21 after CRT is higher in resistant patients (Liu et al. 2020c). ESCC patients with high pretreatment miR-205 expression have a poorer OS (Pan et al. 2017). The serum level of miR-504 is upregulated during RT and higher in radioresistant NPC patients (Zhao et al. 2015).
More evidence shows the involvement of miRNA overexpression in high RT efficacy in lung cancer. The post-treatment plasma level of miR‐18a‐5p, hsa-miR-98-5p, hsa-miR-302e, hsa-miR-495-3p, and hsa-miR-613 is significantly higher in patients with unresectable stage III and IV NSCLC demonstrating complete or partial response than in cases with refractory tumors (Chen et al. 2016, 2018). The low miR-29c level correlates with shorter RFS of NSCLC patients treated with RT (Arechaga-Ocampo et al. 2017). The high expression of miR-148b before RT and baseline serum level of let-7 significantly correlate with better survival in patients with lung cancer (Wang et al. 2016; Xie et al. 2016). At the same time, the post-treatment level of let-7 is significantly lower in serum of RT-sensitive lung cancer patients with brain metastasis (Liu et al. 2018).
The same findings were found in other cancers. The low miR-203 expression in pretreatment biopsies correlates with local larynx cancer recurrence after RT (de Jong et al. 2015). The low miR-339-5p level is significantly associated with poor OS and DFS in ESCC patients treated with RT (Luo et al. 2019). The positive response to RT is associated with high expression of hsa-miR-1281 and hsa-miR-6732-3p in NPC, miR-339-5p in ESCC, and miR-125a in cervical carcinoma (Pedroza-Torres et al. 2018; Luo et al. 2019; Li et al. 2020).
Thus, no data demonstrate common epigenetic radioresistance biomarkers in different cancers. It can be explained by high epigenetic instability and variability of cancer cells, and insufficient findings. Further studies should indicate whether radioresistance of various cancers can be related to universal molecular mechanisms.
Gene and protein signatures
As multiple processes are associated with RT response, the panels of biomarkers with diverse functional activity can predict radioresistance and determine the cancer prognosis. Recent studies reporting the prognostic value of gene signatures for different cancers have attracted widespread interest (Cantini et al. 2017). Gene signatures are developed to predict therapy response and disease outcome based on the expression of a relatively small number of genes. The sensitivity and specificity of multiple gene models are significantly higher than single biomarkers. Additionally, gene signatures can be used for identifying molecular targets (Gönen 2009).
Several prognostic gene signatures were established in cervical cancer. The eight-gene signature (CCDC136, ABCG2, CYP26A1, TNNI3, CXCL5, SYT13 FOXC2, ITGB3, and TMEM233) predicts OS in cervical cancer patients following RT (Xie et al. 2019). Improved OS is associated with hypermethylation of the CCDC136, ABCG2, CYP26A1, and TNNI3 genes, while poor OS correlates with hypomethylation of the SYT13, FOXC2, CXCL5, and TMEM233 genes (Xie et al. 2019). The seven gene signature (UBE2C, MMP3, DCUN1D5, SDCCAG8, IGF2BP2, CCL18, and FST) predicts the risk of poor DFS in cervical cancer patients following RT with 64% sensitivity and 100% specificity (Rajkumar et al. 2009). A proteomic panel of ERCC1, CD133, HER2, BCL2, and CAIX predicts DFS and OS in cervical cancer patients treated with CRT (Choi et al. 2020a).
Numerous predictive models have been revealed for head and neck cancers. The gene panel of VEGF, BCL-2, CLAUDIN-4, YAP-1, and c-MET was developed to predict no response/partial response to RT in HNSCC. High YAP-1 and BCL-2 and low CLAUDIN-4 expression before RT significantly predict poor recurrence-free survival. YAP-1, BCL-2, and VEGF overexpression correlates to poor cause-specific survival (Akervall et al. 2014). Another predictive panel includes nine genes: CHAC2, CLEC9A, GNG10, JCHAIN, KLRB1, NOG, OLR1, PRELID2, SYT1, VWCE, ZNF443. The high expression of CHAC2, GNG10, JCHAIN, OLR1, KLRB1, PRELID2, SYT1, and ZNF443 in peripheral blood mononuclear cells is related to poor survival, while the upregulation of CLEC9A, NOG, and VWCE—with improved survival in HNSCC patients (Liu et al. 2020a). Combined expression of IGF1R, LAMC2, ITGB1, and IL-6 genes predicts worse radiotherapeutic outcome in HNSCC (You et al. 2019). The analysis of the CHIT1, PDGFB, PNKD, RP2, SERPINC1, SLC4A, STIM1, and THPO proteins expression together with the VEGFA gene variant rs69947 in post-treatment samples predict HNSCC radiosensitivity (Drobin et al. 2020). Post-treatment serum levels of SPARC, SERPIND1, C4B, PPBP, PODXL, SRGN, PPIB, S100A4, and CTSF are significantly higher in the radioresistant patients with NPC. The ERAP1, GC, ITIH1, NRP1, MINPP1, F13A1, C1QB, ITIH2, IGFBP6, and FAM173A proteins are significantly downregulated in the radioresistant cases. The panel based on the SPARC, SERPIND1, C4B, PPIB, and FAM173A proteins predicts the RT response in NPC patients with the sensitivity of 94% and the specificity of 92.6% (Zhang et al. 2019).
Thus, different gene and protein panels have been developed to predict RT response and prognosis in cervical and HNSCC patients. Further studies are needed to determine whether these panels are valid in other cancers or propose some new ones that are universal for different malignancies. Nevertheless, gene and protein panels are generally expensive and time-consuming and thus are challenging to translate to clinical practice.
Conclusion
Based on the above findings, we summarize that no universal radioresistance biomarkers exist. Some biomarkers, mainly VEGF, OPN, and pAKT, are described as associated with RT efficacy simultaneously in breast, rectal, prostate, head and neck, lung, cervical, and esophageal cancers. The involvement of these proteins in radioresistance is confirmed by the studies showing that blood vessel normalization and TK inhibition enhance the clinical benefit of radiotherapy (Goedegebuure et al. 2019; Pottier et al. 2020; Zhang et al. 2021). Further research should be directed toward elucidating the involvement of these biomarkers in radioresistance in other cancers.
References
Ai D, Dou Y, Nan Z et al (2021) CD68(+) macrophage infiltration associates with poor outcome of hpv negative oral squamous carcinoma patients receiving radiation: poly(I:C) enhances radiosensitivity of CAL-27 cells but promotes macrophage recruitment through HMGB1. Front Oncol 11:740622. https://doi.org/10.3389/fonc.2021.740622
Akervall J, Nandalur S, Zhang J et al (2014) A novel panel of biomarkers predicts radioresistance in patients with squamous cell carcinoma of the head and neck. Eur J Cancer 50:570–581. https://doi.org/10.1016/j.ejca.2013.11.007
Aleksic T, Verrill C, Bryant RJ et al (2017) IGF-1R associates with adverse outcomes after radical radiotherapy for prostate cancer. Br J Cancer 117:1600–1606. https://doi.org/10.1038/bjc.2017.337
Alsubhi N, Middleton F, Abdel-Fatah TMA et al (2016) Chk1 phosphorylated at serine345 is a predictor of early local recurrence and radio-resistance in breast cancer. Mol Oncol 10:213–223. https://doi.org/10.1016/j.molonc.2015.09.009
Ansems M, Span PN (2020) The tumor microenvironment and radiotherapy response; a central role for cancer-associated fibroblasts. Clin Transl Radiat Oncol 22:90–97. https://doi.org/10.1016/j.ctro.2020.04.001
Aponte PM, Caicedo A (2017) Stemness in cancer: stem cells, cancer stem cells, and their microenvironment. Stem Cells Int. https://doi.org/10.1155/2017/5619472
Arechaga-Ocampo E, Lopez-Camarillo C, Villegas-Sepulveda N et al (2017) Tumor suppressor miR-29c regulates radioresistance in lung cancer cells. Tumour Biol J Int Soc Oncodev Biol Med 39:1010428317695010. https://doi.org/10.1177/1010428317695010
Azevedo Martins JM, Rabelo-Santos SH, do Amaral Westin MC, Zeferino LC, (2020) Tumoral and stromal expression of MMP-2, MMP-9, MMP-14, TIMP-1, TIMP-2, and VEGF-A in cervical cancer patient survival: a competing risk analysis. BMC Cancer 20:660. https://doi.org/10.1186/s12885-020-07150-3
Baek J-H, Yun HS, Kwon GT et al (2019) PLOD3 suppression exerts an anti-tumor effect on human lung cancer cells by modulating the PKC-delta signaling pathway. Cell Death Dis 10:156. https://doi.org/10.1038/s41419-019-1405-8
Balermpas P, Rödel F, Liberz R et al (2014) Head and neck cancer relapse after chemoradiotherapy correlates with CD163+ macrophages in primary tumour and CD11b+ myeloid cells in recurrences. Br J Cancer 111:1509–1518. https://doi.org/10.1038/bjc.2014.446
Balkwill F, Capasso M, Hagemann T (2012) The tumor microenvironment at a glance. J Cell Sci 125:5591–5596. https://doi.org/10.1242/JCS.116392
Bargiela-Iparraguirre J, Prado-Marchal L, Fernandez-Fuente M et al (2016) CHK1 expression in Gastric Cancer is modulated by p53 and RB1/E2F1: implications in chemo/radiotherapy response. Sci Rep 6:21519. https://doi.org/10.1038/srep21519
Barker HE, Paget JTE, Khan AA, Harrington KJ (2015) The tumour microenvironment after radiotherapy: mechanisms of resistance and recurrence. Nat Rev Cancer 15:409–425. https://doi.org/10.1038/nrc3958
Baskar R, Itahana K (2017) Radiation therapy and cancer control in developing countries: can we save more lives? Int J Med Sci 14:13–17. https://doi.org/10.7150/ijms.17288
Bayo J, Tran TA, Wang L et al (2018) Jumonji inhibitors overcome radioresistance in cancer through changes in H3K4 methylation at double-strand breaks. Cell Rep 25:1040-1050.e5. https://doi.org/10.1016/j.celrep.2018.09.081
Berbecka M, Forma A, Baj J et al (2021) A systematic review of the cyclooxygenase-2 (COX-2) expression in rectal cancer patients treated with preoperative radiotherapy or radiochemotherapy. J Clin Med. https://doi.org/10.3390/jcm10194443
Berlin A, Lalonde E, Sykes J et al (2014) NBN gain is predictive for adverse outcome following image-guided radiotherapy for localized prostate cancer. Oncotarget 5:11081–11090. https://doi.org/10.18632/oncotarget.2404
Bhattacharya P, Shetake NG, Pandey BN, Kumar A (2018) Receptor tyrosine kinase signaling in cancer radiotherapy and its targeting for tumor radiosensitization. Int J Radiat Biol 94:628–644. https://doi.org/10.1080/09553002.2018.1478160
Boulle G, Velut Y, Mansuet-Lupo A et al (2020) Chemoradiotherapy efficacy is predicted by intra-tumour CD8+/FoxP3+ double positive T cell density in locally advanced N2 non-small-cell lung carcinoma. Eur J Cancer 135:221–229. https://doi.org/10.1016/j.ejca.2020.04.040
Cabrera-Licona A, Pérez-Añorve IX, Flores-Fortis M et al (2021) Deciphering the epigenetic network in cancer radioresistance. Radiother Oncol J Eur Soc Ther Radiol Oncol 159:48–59. https://doi.org/10.1016/j.radonc.2021.03.012
Cantini L, Calzone L, Martignetti L et al (2017) Classification of gene signatures for their information value and functional redundancy. Npj Syst Biol Appl 4:2. https://doi.org/10.1038/s41540-017-0038-8
Chang H, Wei J-W, Tao Y-L et al (2018) CCR6 is a predicting biomarker of radiosensitivity and potential target of radiosensitization in rectal cancer. Cancer Res Treat 50:1203–1213. https://doi.org/10.4143/crt.2017.538
Chen HHW, Su W-C, Chou C-Y et al (2005) Increased expression of nitric oxide synthase and cyclooxygenase-2 is associated with poor survival in cervical cancer treated with radiotherapy. Int J Radiat Oncol Biol Phys 63:1093–1100. https://doi.org/10.1016/j.ijrobp.2005.03.062
Chen M-F, Chen P-T, Lu MS et al (2013) IL-6 expression predicts treatment response and outcome in squamous cell carcinoma of the esophagus. Mol Cancer 12:26. https://doi.org/10.1186/1476-4598-12-26
Chen Y, Li X, Wu S et al (2014) Expression of HIF-1α and CAIX in nasopharyngeal carcinoma and their correlation with patients’ prognosis. Med Oncol 31:304. https://doi.org/10.1007/s12032-014-0304-1
Chen X, Xu Y, Liao X et al (2016) Plasma miRNAs in predicting radiosensitivity in non-small cell lung cancer. Tumour Biol 37:11927–11936. https://doi.org/10.1007/s13277-016-5052-8
Chen X, Wu L, Li D et al (2018) Radiosensitizing effects of miR-18a-5p on lung cancer stem-like cells via downregulating both ATM and HIF-1α. Cancer Med 7:3834–3847. https://doi.org/10.1002/cam4.1527
Chiu T-J, Lu H-I, Chen C-H et al (2018) Osteopontin expression is associated with the poor prognosis in patients with locally advanced esophageal squamous cell carcinoma receiving preoperative chemoradiotherapy. Biomed Res Int 2018:9098215. https://doi.org/10.1155/2018/9098215
Choi CH, Chung J-Y, Kang JH et al (2020a) Chemoradiotherapy response prediction model by proteomic expressional profiling in patients with locally advanced cervical cancer. Gynecol Oncol 157:437–443. https://doi.org/10.1016/j.ygyno.2020.02.017
Choi J, Yoon YN, Kim N et al (2020b) Predicting radiation resistance in breast cancer with expression status of phosphorylated S6K1. Sci Rep 10:641. https://doi.org/10.1038/s41598-020-57496-8
Chow K-C, Chiou S-H, Ho S-P et al (2003) The elevated serum interleukin-6 correlates with the increased serum butyrate level in patients with nasopharyngeal carcinoma. Oncol Rep 10:813–819
Chung YM, Kim B-G, Park C-S et al (2005) Increased expression of ICAM-3 is associated with radiation resistance in cervical cancer. Int J Cancer 117:194–201. https://doi.org/10.1002/ijc.21180
Couch DG, Hemingway DM (2016) Complete radiotherapy response in rectal cancer: a review of the evidence. World J Gastroenterol 22:467–470. https://doi.org/10.3748/wjg.v22.i2.467
Das M, Abdelmaksoud MHK, Loo BWJ, Kothary N (2010) Alternatives to surgery for early stage non-small cell lung cancer-ready for prime time? Curr Treat Options Oncol 11:24–35. https://doi.org/10.1007/s11864-010-0119-z
Davies JM, Trembath D, Deal AM et al (2011) Phospho-ERK and AKT status, but not KRAS mutation status, are associated with outcomes in rectal cancer treated with chemoradiotherapy. Radiat Oncol 6:114. https://doi.org/10.1186/1748-717X-6-114
de Jong MC, Ten Hoeve JJ, Grénman R et al (2015) Pretreatment microRNA expression impacting on epithelial-to-mesenchymal transition predicts intrinsic radiosensitivity in head and neck cancer cell lines and patients. Clin Cancer Res 21:5630–5638. https://doi.org/10.1158/1078-0432.CCR-15-0454
Dehing-Oberije C, Aerts H, Yu S et al (2011) Development and validation of a prognostic model using blood biomarker information for prediction of survival of non-small-cell lung cancer patients treated with combined chemotherapy and radiation or radiotherapy alone (NCT00181519, NCT00573040, and NCT. Int J Radiat Oncol Biol Phys 81:360–368. https://doi.org/10.1016/j.ijrobp.2010.06.011
Drobin K, Marczyk M, Halle M et al (2020) Molecular profiling for predictors of radiosensitivity in patients with breast or head-and-neck cancer. Cancers (basel). https://doi.org/10.3390/cancers12030753
DuBois S, Demetri G (2007) Markers of angiogenesis and clinical features in patients with sarcoma. Cancer 109:813–819. https://doi.org/10.1002/cncr.22455
Etiz D, Ataizi FC, Bayman E et al (2013) Prognostic value of osteopontin in patients treated with primary radiotherapy for head and neck cancer. Asian Pac J Cancer Prev 14:5175–5178. https://doi.org/10.7314/apjcp.2013.14.9.5175
Fardi M, Solali S, Farshdousti Hagh M (2018) Epigenetic mechanisms as a new approach in cancer treatment: an updated review. Genes Dis 5:304–311. https://doi.org/10.1016/j.gendis.2018.06.003
Farhood B, Najafi M, Mortezaee K (2019) CD8+ cytotoxic T lymphocytes in cancer immunotherapy: a review. J Cell Physiol. 234:8509–8521. https://doi.org/10.1002/jcp.27782
Ferrandon S, DeVecchio J, Duraes L et al (2020) CoA synthase (COASY) mediates radiation resistance via pi3k signaling in rectal cancer. Cancer Res 80:334–346. https://doi.org/10.1158/0008-5472.CAN-19-1161
Fiedler M, Weber F, Hautmann MG et al (2018) Biological predictors of radiosensitivity in head and neck squamous cell carcinoma. Clin Oral Investig 22:189–200. https://doi.org/10.1007/s00784-017-2099-x
Freudlsperger C, Horn D, Weißfuß S et al (2015) Phosphorylation of AKT(Ser473) serves as an independent prognostic marker for radiosensitivity in advanced head and neck squamous cell carcinoma. Int J Cancer 136:2775–2785. https://doi.org/10.1002/ijc.29328
Fridman WH, Zitvogel L, Sautès-Fridman C, Kroemer G (2017) The immune contexture in cancer prognosis and treatment. Nat Rev Clin Oncol 14:717–734. https://doi.org/10.1038/nrclinonc.2017.101
Fu Z-Z, Sun X-D, Li P et al (2014) Relationship between serum VEGF level and radiosensitivity of patients with nonsmall cell lung cancer among asians: a meta-analysis. DNA Cell Biol 33:426–437. https://doi.org/10.1089/dna.2013.2249
Fu H-C, Chuang I-C, Yang Y-C et al (2018) Low P16(INK4A) expression associated with high expression of cancer stem cell markers predicts poor prognosis in cervical cancer after radiotherapy. Int J Mol Sci. https://doi.org/10.3390/ijms19092541
Garg AK, Jhingran A, Klopp AH et al (2010) Expression of nuclear transcription factor kappa B in locally advanced human cervical cancer treated with definitive chemoradiation. Int J Radiat Oncol Biol Phys 78:1331–1336. https://doi.org/10.1016/j.ijrobp.2009.09.044
Genard G, Lucas S, Michiels C (2017) Reprogramming of tumor-associated macrophages with anticancer therapies: radiotherapy versus chemo- and immunotherapies. Front Immunol 8:828. https://doi.org/10.3389/fimmu.2017.00828
Goedegebuure RSA, de Klerk LK, Bass AJ et al (2019) Combining radiotherapy with anti-angiogenic therapy and immunotherapy; a therapeutic triad for cancer? Front Immunol 9:3107. https://doi.org/10.3389/fimmu.2018.03107
Gönen M (2009) Statistical aspects of gene signatures and molecular targets. Gastrointest Cancer Res 3:S19–S21
Göttgens E-L, van den Heuvel CN, de Jong MC et al (2019) ACLY (ATP Citrate Lyase) mediates radioresistance in head and neck squamous cell carcinomas and is a novel predictive radiotherapy biomarker. Cancers (basel). https://doi.org/10.3390/cancers11121971
Grün A, Kuhnt T, Schlomm T et al (2020) Repeat radiation for local recurrence of head and neck tumors and in prostate cancer. Dtsch Arztebl Int 117:167–174. https://doi.org/10.3238/arztebl.2020.0167
Ham I-H, Lee D, Hur H (2021) Cancer-associated fibroblast-induced resistance to chemotherapy and radiotherapy in gastrointestinal cancers. Cancers (basel) 13:1172. https://doi.org/10.3390/cancers13051172
Harrington KJ, Billingham LJ, Brunner TB et al (2011) Guidelines for preclinical and early phase clinical assessment of novel radiosensitisers. Br J Cancer 105:628–639. https://doi.org/10.1038/bjc.2011.240
Hou H, Er P, Cheng J et al (2017) High expression of FUNDC1 predicts poor prognostic outcomes and is a promising target to improve chemoradiotherapy effects in patients with cervical cancer. Cancer Med 6:1871–1881. https://doi.org/10.1002/cam4.1112
Huang R-X, Zhou P-K (2020) DNA damage response signaling pathways and targets for radiotherapy sensitization in cancer. Signal Transduct Target Ther 5:60. https://doi.org/10.1038/s41392-020-0150-x
Huang K-H, Huang S-F, Chen I-H et al (2009) Methylation of RASSF1A, RASSF2A, and HIN-1 is associated with poor outcome after radiotherapy, but not surgery, in oral squamous cell carcinoma. Clin Cancer Res 15:4174–4180. https://doi.org/10.1158/1078-0432.CCR-08-2929
Huang X, Qian Y, Wu H et al (2015) Aberrant expression of osteopontin and E-cadherin indicates radiation resistance and poor prognosis for patients with cervical carcinoma. J Histochem Cytochem 63:88–98. https://doi.org/10.1369/0022155414561329
Huang L, Chen S, Yang WT, Shao Z (2017) Risk factors of locoregional relapse in locally advanced breast cancer treated with neoadjuvant chemotherapy following mastectomy and radiotherapy. Oncotarget 8:39703–39710. https://doi.org/10.18632/oncotarget.14407
Huang W, Shi G, Yong Z et al (2020) Downregulation of RKIP promotes radioresistance of nasopharyngeal carcinoma by activating NRF2/NQO1 axis via downregulating miR-450b-5p. Cell Death Dis 11:504. https://doi.org/10.1038/s41419-020-2695-6
Hui EP, Chan ATC, Pezzella F et al (2002) Coexpression of hypoxia-inducible factors 1alpha and 2alpha, carbonic anhydrase IX, and vascular endothelial growth factor in nasopharyngeal carcinoma and relationship to survival. Clin Cancer Res 8:2595–2604
Hui EP, Sung FL, Yu BKH et al (2008) Plasma osteopontin, hypoxia, and response to radiotherapy in nasopharyngeal cancer. Clin Cancer Res 14:7080–7087. https://doi.org/10.1158/1078-0432.CCR-08-0364
Iwai Y, Ishida M, Tanaka Y et al (2002) Involvement of PD-L1 on tumor cells in the escape from host immune system and tumor immunotherapy by PD-L1 blockade. Proc Natl Acad Sci USA 99:12293–12297. https://doi.org/10.1073/pnas.192461099
Jing X, Yang F, Shao C et al (2019) Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol Cancer 18:157. https://doi.org/10.1186/s12943-019-1089-9
Johnson N, Shapiro GI (2010) Cyclin-dependent kinases (cdks) and the DNA damage response: rationale for cdk inhibitor–chemotherapy combinations as an anticancer strategy for solid tumors. Expert Opin Ther Targets 14:1199. https://doi.org/10.1517/14728222.2010.525221
Kawanaka T, Kubo A, Ikushima H et al (2008) Prognostic significance of HIF-2alpha expression on tumor infiltrating macrophages in patients with uterine cervical cancer undergoing radiotherapy. J Med Invest 55:78–86. https://doi.org/10.2152/jmi.55.78
Keir ME, Liang SC, Guleria I et al (2006) Tissue expression of PD-L1 mediates peripheral T cell tolerance. J Exp Med 203:883–895. https://doi.org/10.1084/jem.20051776
Kim YB, Kim GE, Cho NH et al (2002) Overexpression of cyclooxygenase-2 is associated with a poor prognosis in patients with squamous cell carcinoma of the uterine cervix treated with radiation and concurrent chemotherapy. Cancer 95:531–539. https://doi.org/10.1002/cncr.10684
Kim T-J, Lee J-W, Song SY et al (2006) Increased expression of pAKT is associated with radiation resistance in cervical cancer. Br J Cancer 94:1678–1682. https://doi.org/10.1038/sj.bjc.6603180
Kim BW, Cho H, Chung J-Y et al (2013) Prognostic assessment of hypoxia and metabolic markers in cervical cancer using automated digital image analysis of immunohistochemistry. J Transl Med 11:185. https://doi.org/10.1186/1479-5876-11-185
Komdeur FL, Prins TM, van de Wall S et al (2017) CD103+ tumor-infiltrating lymphocytes are tumor-reactive intraepithelial CD8+ T cells associated with prognostic benefit and therapy response in cervical cancer. Oncoimmunology 6:e1338230. https://doi.org/10.1080/2162402X.2017.1338230
Kong M, Sung J-Y, Lee SH (2019) Reactive oxygen species modulator 1 as an adverse prognostic marker in stage III non-small cell lung cancer treated with radiotherapy: a retrospective pilot study. Onco Targets Ther 12:8263–8273. https://doi.org/10.2147/OTT.S217514
Köster F, Sauer L, Hoellen F et al (2020) PSMD9 expression correlates with recurrence after radiotherapy in patients with cervical cancer. Oncol Lett 20:581–588. https://doi.org/10.3892/ol.2020.11622
Koyama FC, Lopes Ramos CM, Ledesma F et al (2018) Effect of Akt activation and experimental pharmacological inhibition on responses to neoadjuvant chemoradiotherapy in rectal cancer. Br J Surg 105:e192–e203. https://doi.org/10.1002/bjs.10695
Kurokawa T, Nakagawa T, Matsusaka K et al (2020) Establishment of epigenetic markers to predict irradiation efficacy against oropharyngeal cancer. Cancer Sci 111:1407–1416. https://doi.org/10.1111/cas.14338
Larionova I, Cherdyntseva N, Liu T et al (2019) Interaction of tumor-associated macrophages and cancer chemotherapy. Oncoimmunology 8:1596004. https://doi.org/10.1080/2162402X.2019.1596004
Larionova I, Tuguzbaeva G, Ponomaryova A et al (2020) Tumor-associated macrophages in human breast, colorectal, lung Ovarian and Prostate Cancers. Front Oncol 10:566511. https://doi.org/10.3389/fonc.2020.56651
Larionova I, Kazakova E, Gerashchenko T, Kzhyshkowska J (2021) New angiogenic regulators produced by TAMs: perspective for targeting tumor angiogenesis. Cancers (basel). https://doi.org/10.3390/cancers13133253
Li K, Zhu X, Li L et al (2020) Identification of non-invasive biomarkers for predicting the radiosensitivity of nasopharyngeal carcinoma from serum microRNAs. Sci Rep 10:5161. https://doi.org/10.1038/s41598-020-61958-4
Liao L, Yan W-J, Tian C-M et al (2018) Knockdown of annexin A1 enhances radioresistance and inhibits apoptosis in nasopharyngeal carcinoma. Technol Cancer Res Treat 17:1533034617750309. https://doi.org/10.1177/1533034617750309
Lin H, Chen Z-T, Zhu X-D et al (2017) Serum CD166: A novel biomarker for predicting nasopharyngeal carcinoma response to radiotherapy. Oncotarget 8:62858–62867. https://doi.org/10.18632/oncotarget.16399
Lindegaard AM, von Buchwald C, Rasmussen JH et al (2020) Outcome in patients with isolated regional recurrence after primary radiotherapy for head and neck cancer. Head Neck 42:3161–3170. https://doi.org/10.1002/hed.26361
Lippens L, Van Bockstal M, De Jaeghere EA et al (2020) Immunologic impact of chemoradiation in cervical cancer and how immune cell infiltration could lead toward personalized treatment. Int J Cancer 147:554–564. https://doi.org/10.1002/ijc.32893
Liu H-X, Li N, Wei L et al (2017a) High expression of Kruppel-like factor 4 as a predictor of poor prognosis for cervical cancer patient response to radiotherapy. Tumour Biol J Int Soc Oncodevelopmental Biol Med 39:1010428317710225. https://doi.org/10.1177/1010428317710225
Liu T, Zhang L, Joo D, Sun S-C (2017b) NF-κB signaling in inflammation. Signal Transduct Target Ther 2:17023. https://doi.org/10.1038/sigtrans.2017.23
Liu J-K, Liu H-F, Ding Y, Gao G-D (2018) Predictive value of microRNA let-7a expression for efficacy and prognosis of radiotherapy in patients with lung cancer brain metastasis: A case-control study. Medicine (baltimore) 97:e12847. https://doi.org/10.1097/MD.0000000000012847
Liu C, Hu Q, Hu K et al (2019) Increased CD8+CD28+ T cells independently predict better early response to stereotactic ablative radiotherapy in patients with lung metastases from non-small cell lung cancer. J Transl Med 17:120. https://doi.org/10.1186/s12967-019-1872-9
Liu G, Zeng X, Wu B et al (2020a) RNA-Seq analysis of peripheral blood mononuclear cells reveals unique transcriptional signatures associated with radiotherapy response of nasopharyngeal carcinoma and prognosis of head and neck cancer. Cancer Biol Ther 21:139–146. https://doi.org/10.1080/15384047.2019.1670521
Liu J, Bi K, Yang R et al (2020b) Role of DNA damage and repair in radiation cancer therapy: a current update and a look to the future. Int J Radiat Biol 96:1329–1338. https://doi.org/10.1080/09553002.2020.1807641
Liu Q, Liu S, Wang D (2020c) Overexpression of microRNA-21 decreased the sensitivity of advanced cervical cancer to chemoradiotherapy through SMAD7. Anticancer Drugs 31:272–281. https://doi.org/10.1097/CAD.0000000000000871
Loncaster JA, Cooper RA, Logue JP et al (2000) (2000) Vascular endothelial growth factor (VEGF) expression is a prognostic factor for radiotherapy outcome in advanced carcinoma of the cervix. Br J Cancer 835(83):620–625. https://doi.org/10.1054/bjoc.2000.1319
Luo A, Zhou X, Shi X et al (2019) Exosome-derived miR-339-5p mediates radiosensitivity by targeting Cdc25A in locally advanced esophageal squamous cell carcinoma. Oncogene 38:4990–5006. https://doi.org/10.1038/s41388-019-0771-0
Lv J, An J, Zhang Y-D et al (2020) A three serum miRNA panel as diagnostic biomarkers of radiotherapy-related metastasis in non-small cell lung cancer. Oncol Lett 20:236. https://doi.org/10.3892/ol.2020.12099
Ma W, Yu J, Qin X, et al (2015) Radiation-induced microrna-622 causes radioresistance in colorectal cancer cells by down-regulating Rb. Oncotarget. 6:15984–15994. https://doi.org/10.18632/oncotarget.3762
Macedo-Silva C, Miranda-Gonçalves V, Lameirinhas A et al (2020) JmjC-KDMs KDM3A and KDM6B modulate radioresistance under hypoxic conditions in esophageal squamous cell carcinoma. Cell Death Dis 11:1068. https://doi.org/10.1038/s41419-020-03279-y
Manders P, Sweep FCGJ, Tjan-Heijnen VCG et al (2003) Vascular endothelial growth factor independently predicts the efficacy of postoperative radiotherapy in node-negative breast cancer patients. Clin Cancer Res 9:6363–6370
Marampon F, Megiorni F, Camero S et al (2017) HDAC4 and HDAC6 sustain DNA double strand break repair and stem-like phenotype by promoting radioresistance in glioblastoma cells. Cancer Lett 397:1–11. https://doi.org/10.1016/j.canlet.2017.03.028
Moeller BJ, Yordy JS, Williams MD et al (2011) DNA repair biomarker profiling of head and neck cancer: Ku80 expression predicts locoregional failure and death following radiotherapy. Clin Cancer Res 17:2035–2043. https://doi.org/10.1158/1078-0432.CCR-10-2641
Moreno-Acosta P, Vallard A, Carrillo S et al (2017) Biomarkers of resistance to radiation therapy: a prospective study in cervical carcinoma. Radiat Oncol 12:120. https://doi.org/10.1186/s13014-017-0856-2
Najafi M, Mortezaee K, Majidpoor J (2019) Cancer stem cell (CSC) resistance drivers. Life Sci 234:116781. https://doi.org/10.1016/j.lfs.2019.116781
Ning MS, Ahobila V, Jhingran A et al (2018) Outcomes and patterns of relapse after definitive radiation therapy for oligometastatic cervical cancer. Gynecol Oncol 148:132–138. https://doi.org/10.1016/j.ygyno.2017.10.017
Nowicka Z, Stawiski K, Tomasik B, Fendler W (2019) Extracellular miRNAs as biomarkers of head and neck cancer progression and metastasis. Int J Mol Sci 20:4799. https://doi.org/10.3390/ijms20194799
O’Cathail SM, Wu C-H, Thomas R et al (2021) NRF2 mediates therapeutic resistance to chemoradiation in colorectal cancer through a metabolic switch. Antioxidants 10:1380
Olivares-Urbano MA, Griñán-Lisón C, Marchal JA, Núñez MI (2020) CSC radioresistance: a therapeutic challenge to improve radiotherapy effectiveness in cancer. Cells 9:1651. https://doi.org/10.3390/cells9071651
Ostheimer C, Bache M, Güttler A et al (2014) A pilot study on potential plasma hypoxia markers in the radiotherapy of non-small cell lung cancer. Osteopontin, carbonic anhydrase IX and vascular endothelial growth factor. Strahlenther Onkol 190:276–282. https://doi.org/10.1007/s00066-013-0484-1
Ostheimer C, Evers C, Bache M et al (2018) Prognostic implications of the co-detection of the urokinase plasminogen activator system and osteopontin in patients with non-small-cell lung cancer undergoing radiotherapy and correlation with gross tumor volume. Strahlenther Onkol 194:539–551. https://doi.org/10.1007/s00066-017-1255-1
Otani K, Naito Y, Sakaguchi Y et al (2016) Cell-cycle-controlled radiation therapy was effective for treating a murine malignant melanoma cell line in vitro and in vivo. Sci Rep 6:30689. https://doi.org/10.1038/srep30689
Overgaard J (2007) Hypoxic radiosensitization: adored and ignored. J Clin Oncol 25:4066–4074. https://doi.org/10.1200/JCO.2007.12.7878
Overgaard J, Eriksen JG, Nordsmark M et al (2005) Plasma osteopontin, hypoxia, and response to the hypoxia sensitiser nimorazole in radiotherapy of head and neck cancer: results from the DAHANCA 5 randomised double-blind placebo-controlled trial. Lancet Oncol 6:757–764. https://doi.org/10.1016/S1470-2045(05)70292-8
Pajonk F, Vlashi E, McBride WH (2010) Radiation resistance of cancer stem cells: the 4 R’s of radiobiology revisited. Stem Cells 28:639–648. https://doi.org/10.1002/stem.318
Pan F, Mao H, Bu F et al (2017) Sp1-mediated transcriptional activation of miR-205 promotes radioresistance in esophageal squamous cell carcinoma. Oncotarget 8:5735–5752. https://doi.org/10.18632/oncotarget.13902
Park HJ, Griffin RJ, Hui S et al (2012) Radiation-induced vascular damage in tumors: implications of vascular damage in ablative hypofractionated radiotherapy (SBRT and SRS). Radiat Res 177:311–327. https://doi.org/10.1667/rr2773.1
Patil M, Pabla N, Dong Z (2013) Checkpoint kinase 1 in DNA damage response and cell cycle regulation. Cell Mol Life Sci 70:4009–4021. https://doi.org/10.1007/s00018-013-1307-3
Paul MK, Mukhopadhyay AK (2004) Tyrosine kinase—role and significance in cancer. Int J Med Sci 1:101–115. https://doi.org/10.7150/ijms.1.101
Pawlik TM, Keyomarsi K (2004) Role of cell cycle in mediating sensitivity to radiotherapy. Int J Radiat Oncol 59:928–942. https://doi.org/10.1016/j.ijrobp.2004.03.005
Pedroza-Torres A, Campos-Parra AD, Millan-Catalan O et al (2018) MicroRNA-125 modulates radioresistance through targeting p21 in cervical cancer. Oncol Rep 39:1532–1540. https://doi.org/10.3892/or.2018.6219
Peitzsch C, Perrin R, Hill RP et al (2014) Hypoxia as a biomarker for radioresistant cancer stem cells. Int J Radiat Biol 90:636–652. https://doi.org/10.3109/09553002.2014.916841
Peitzsch C, Tyutyunnykova A, Pantel K, Dubrovska A (2017) Cancer stem cells: the root of tumor recurrence and metastases. Semin Cancer Biol 44:10–24. https://doi.org/10.1016/j.semcancer.2017.02.011
Pelloski CE, Mahajan A, Maor M et al (2005) YKL-40 expression is associated with poorer response to radiation and shorter overall survival in glioblastoma. Clin Cancer Res 11:3326–3334. https://doi.org/10.1158/1078-0432.CCR-04-1765
Peltola K, Hollmen M, Maula S-M et al (2009) Pim-1 kinase expression predicts radiation response in squamocellular carcinoma of head and neck and is under the control of epidermal growth factor receptor. Neoplasia 11:629–636. https://doi.org/10.1593/neo.81038
Perillo B, Di Donato M, Pezone A et al (2020) ROS in cancer therapy: the bright side of the moon. Exp Mol Med 52:192–203. https://doi.org/10.1038/s12276-020-0384-2
Petrelli NJ, Shaw N, Bhargava A et al (1988) Squamous cell carcinoma antigen as a marker for squamous cell carcinoma of the anal canal. J Clin Oncol 6:782–785. https://doi.org/10.1200/JCO.1988.6.5.782
Podralska M, Ciesielska S, Kluiver J et al (2020) Non-coding RNAs in cancer radiosensitivity: micrornas and lncRNAs as regulators of radiation-induced signaling pathways. Cancers (basel). https://doi.org/10.3390/cancers12061662
Pottier C, Fresnais M, Gilon M et al (2020) Tyrosine kinase inhibitors in cancer: breakthrough and challenges of targeted therapy. Cancers (basel) 12:731. https://doi.org/10.3390/cancers12030731
Qin C-J, Song X-M, Chen Z-H et al (2015) XRCC2 as a predictive biomarker for radioresistance in locally advanced rectal cancer patients undergoing preoperative radiotherapy. Oncotarget 6:32193–32204. https://doi.org/10.18632/oncotarget.4975
Qu C, Zhao Y, Feng G et al (2017) RPA3 is a potential marker of prognosis and radioresistance for nasopharyngeal carcinoma. J Cell Mol Med 21:2872–2883. https://doi.org/10.1111/jcmm.13200
Rajkumar T, Vijayalakshmi N, Sabitha K et al (2009) A 7 gene expression score predicts for radiation response in cancer cervix. BMC Cancer 9:365. https://doi.org/10.1186/1471-2407-9-365
Rivero M, Peinado-Serrano J, Muñoz-Galvan S et al (2018) MAP17 (PDZK1IP1) and pH2AX are potential predictive biomarkers for rectal cancer treatment efficacy. Oncotarget 9:32958–32971. https://doi.org/10.18632/oncotarget.26010
Rødningen OK, Overgaard J, Alsner J et al (2005) Microarray analysis of the transcriptional response to single or multiple doses of ionizing radiation in human subcutaneous fibroblasts. Radiother Oncol J Eur Soc Ther Radiol Oncol 77:231–240. https://doi.org/10.1016/j.radonc.2005.09.020
Saigusa S, Toiyama Y, Tanaka K et al (2010) Stromal CXCR4 and CXCL12 expression is associated with distant recurrence and poor prognosis in rectal cancer after chemoradiotherapy. Ann Surg Oncol 17:2051–2058. https://doi.org/10.1245/s10434-010-0970-y
Saigusa S, Toiyama Y, Tanaka K et al (2011) Cancer-associated fibroblasts correlate with poor prognosis in rectal cancer after chemoradiotherapy. Int J Oncol 38:655–663. https://doi.org/10.3892/ijo.2011.906
Sakakura C, Miyagawa K, Fukuda K-I et al (2007) Frequent silencing of RUNX3 in esophageal squamous cell carcinomas is associated with radioresistance and poor prognosis. Oncogene 26:5927–5938. https://doi.org/10.1038/sj.onc.1210403
Sharda A, Rashid M, Shah SG et al (2020) Elevated HDAC activity and altered histone phospho-acetylation confer acquired radio-resistant phenotype to breast cancer cells. Clin Epigenetics 12:4. https://doi.org/10.1186/s13148-019-0800-4
Shih S-C, Robinson GS, Perruzzi CA et al (2002) Molecular profiling of angiogenesis markers. Am J Pathol 161:35–41. https://doi.org/10.1016/S0002-9440(10)64154-5
Shrivastav M, De Haro LP, Nickoloff JA (2008) Regulation of DNA double-strand break repair pathway choice. Cell Res 18:134–147. https://doi.org/10.1038/cr.2007.111
Sjöström M, Veenstra C, Holmberg E et al (2020) Expression of HGF, pMet, and pAkt is related to benefit of radiotherapy after breast-conserving surgery: a long-term follow-up of the SweBCG91-RT randomised trial. Mol Oncol 14:2713–2726. https://doi.org/10.1002/1878-0261.12803
Skinner HD, Giri U, Yang L et al (2016) Proteomic profiling identifies PTK2/FAK as a driver of radioresistance in HPV-negative head and neck cancer. Clin Cancer Res 22:4643–4650. https://doi.org/10.1158/1078-0432.CCR-15-2785
Snitcovsky I, Leitão GM, Pasini FS et al (2009) Plasma osteopontin levels in patients with head and neck cancer undergoing chemoradiotherapy. Arch Otolaryngol Head Neck Surg 135:807–811. https://doi.org/10.1001/archoto.2009.103
Stakheyeva M, Riabov V, Mitrofanova I et al (2017) Role of the immune component of tumor microenvironment in the efficiency of cancer treatment: perspectives for the personalized therapy. Curr Pharm Des 23:4807–4826. https://doi.org/10.2174/1381612823666170714161703
Suh Y-E, Raulf N, Gäken J et al (2015) MicroRNA-196a promotes an oncogenic effect in head and neck cancer cells by suppressing annexin A1 and enhancing radioresistance. Int J Cancer 137:1021–1034. https://doi.org/10.1002/ijc.29397
Sui X, Jiang L, Teng H et al (2021) Prediction of clinical outcome in locally advanced non-small cell lung cancer patients treated with chemoradiotherapy by plasma markers. Front Oncol 10:625911. https://doi.org/10.3389/fonc.2020.625911
Sun G, Hu W, Lu Y, Wang Y (2013) A meta-analysis of HIF-1α and esophageal squamous cell carcinoma (ESCC) risk. Pathol Oncol Res 19:685–693. https://doi.org/10.1007/s12253-013-9631-3
Suzuki Y, Shirai K, Oka K et al (2007) Prognostic impact of pAkt in glioblastoma treated with radiation therapy. Int J Radiat Oncol Biol Phys 69:S239–S240. https://doi.org/10.1016/j.ijrobp.2007.07.1235
Swanton C, Marcus S, Jayamohan J et al (2021) Can adjuvant pelvic radiation therapy after local excision or polypectomy for T1 and T2 rectal cancer offer an alternative option to radical surgery? Clin Transl Radiat Oncol 31:97–101. https://doi.org/10.1016/j.ctro.2021.10.002
Swartz JE, Wegner I, Noorlag R et al (2021) HIF-1a expression and differential effects on survival in patients with oral cavity, larynx, and oropharynx squamous cell carcinomas. Head Neck 43:745–756. https://doi.org/10.1002/hed.26530
Tang L, Wei F, Wu Y et al (2018) Role of metabolism in cancer cell radioresistance and radiosensitization methods. J Exp Clin Cancer Res 37:87. https://doi.org/10.1186/s13046-018-0758-7
Tay W-L, Yip GW-C, Tan P-H et al (2009) Y-Box-binding protein-1 is a promising predictive marker of radioresistance and chemoradioresistance in nasopharyngeal cancer. Mod Pathol 22:282–290. https://doi.org/10.1038/modpathol.2008.181
Tu Z, Xu B, Qu C et al (2015) BRCC3 acts as a prognostic marker in nasopharyngeal carcinoma patients treated with radiotherapy and mediates radiation resistance in vitro. Radiat Oncol 10:123. https://doi.org/10.1186/s13014-015-0427-3
Turner BC, Haffty BG, Narayanan L et al (1997) Insulin-like growth factor-I receptor overexpression mediates cellular radioresistance and local breast cancer recurrence after lumpectomy and radiation. Cancer Res 57:3079–3083
Venkatesh V, Nataraj R, Thangaraj GS et al (2018) Targeting notch signalling pathway of cancer stem cells. Stem Cell Investig 5:5. https://doi.org/10.21037/sci.2018.02.02
Vergis R, Corbishley CM, Norman AR et al (2008) Intrinsic markers of tumour hypoxia and angiogenesis in localised prostate cancer and outcome of radical treatment: a retrospective analysis of two randomised radiotherapy trials and one surgical cohort study. Lancet Oncol 9:342–351. https://doi.org/10.1016/S1470-2045(08)70076-7
Wang L, Pino-Lagos K, de Vries VC et al (2008) Programmed death 1 ligand signaling regulates the generation of adaptive Foxp3+CD4+ regulatory T cells. Proc Natl Acad Sci U S A 105:9331–9336. https://doi.org/10.1073/pnas.0710441105
Wang R, Ye F, Zhen Q et al (2016) MicroRNA-148b is a potential prognostic biomarker and predictor of response to radiotherapy in non-small-cell lung cancer. J Physiol Biochem 72:337–343. https://doi.org/10.1007/s13105-016-0485-5
Wang Y, Gan G, Wang B et al (2017) Cancer-associated fibroblasts promote irradiated cancer cell recovery through autophagy. EBioMedicine 17:45–56. https://doi.org/10.1016/j.ebiom.2017.02.019
Wang H-C, Chou C-L, Yang C-C et al (2019a) Over-expression of CHD4 Is an independent biomarker of poor prognosis in patients with rectal cancers receiving concurrent chemoradiotherapy. Int J Mol Sci. https://doi.org/10.3390/ijms20174087
Wang H, Jiang H, Van De Gucht M, De Ridder M (2019b) Hypoxic radioresistance: can ROS be the key to overcome It? Cancers (basel) 11:112. https://doi.org/10.3390/cancers11010112
Wang Y, Kim TH, Fouladdel S et al (2019c) PD-L1 expression in circulating tumor cells increases during radio(chemo)therapy and indicates poor prognosis in non-small cell lung cancer. Sci Rep 9:566. https://doi.org/10.1038/s41598-018-36096-7
Wang Z, Zhang J, Li M et al (2020) The expression of p-p62 and nuclear Nrf2 in esophageal squamous cell carcinoma and association with radioresistance. Thorac Cancer 11:130–139. https://doi.org/10.1111/1759-7714.13252
Willers H, Azzoli CG, Santivasi WL, Xia F (2013) Basic mechanisms of therapeutic resistance to radiation and chemotherapy in lung cancer. Cancer J 19:200–207. https://doi.org/10.1097/PPO.0b013e318292e4e3
Wu X-Y, Wu Z-F, Cao Q-H et al (2014) Insulin-like growth factor receptor-1 overexpression is associated with poor response of rectal cancers to radiotherapy. World J Gastroenterol 20:16268–16274. https://doi.org/10.3748/wjg.v20.i43.16268
Wu Q, Allouch A, Martins I et al (2017) Macrophage biology plays a central role during ionizing radiation-elicited tumor response. Biomed J 40:200–211. https://doi.org/10.1016/J.BJ.2017.06.003
Wu C, Guo E, Ming J et al (2020) Radiation-induced DNMT3B promotes radioresistance in nasopharyngeal carcinoma through methylation of p53 and p21. Mol Ther Oncolytics 17:306–319. https://doi.org/10.1016/j.omto.2020.04.007
Xie P, Li X, Tan X et al (2016) Sequential serum Let-7 Is a novel biomarker to predict accelerated reproliferation during fractional radiotherapy in lung cancer. Clin Lung Cancer 17:e95–e101. https://doi.org/10.1016/j.cllc.2016.03.010
Xie F, Dong D, Du N et al (2019) An 8-gene signature predicts the prognosis of cervical cancer following radiotherapy. Mol Med Rep 20:2990–3002. https://doi.org/10.3892/mmr.2019.10535
Xu J, Escamilla J, Mok S et al (2013) CSF1R signaling blockade stanches tumor-infiltrating myeloid cells and improves the efficacy of radiotherapy in prostate cancer. Cancer Res 73:2782–2794. https://doi.org/10.1158/0008-5472.CAN-12-3981
Yagi H, Danno K, Maruguchi Y et al (1987) Significance of squamous cell carcinoma (SCC)-related antigens in cutaneous SCC. A Preliminary Report. Arch Dermatol 123:902–906
Yang Z, Liu Z (2020) Potential functional variants in DNA repair genes are associated with efficacy and toxicity of radiotherapy in patients with non-small-cell lung cancer. J Oncol 2020:3132786. https://doi.org/10.1155/2020/3132786
Yang S-S, Yu D-Y, Du Y-T et al (2020) Inhibition of Delta-like Ligand 4 enhances the radiosensitivity and inhibits migration in cervical cancer via the reversion of epithelial-mesenchymal transition. Cancer Cell Int 20:344. https://doi.org/10.1186/s12935-020-01434-1
Yao Y-F, Du C-Z, Chen N et al (2014) Expression of HER-2 in rectal cancers treated with preoperative radiotherapy: a potential biomarker predictive of metastasis. Dis Colon Rectum 57:602–607. https://doi.org/10.1097/DCR.0000000000000107
Yin M, Liao Z, Huang Y-J et al (2011) Polymorphisms of homologous recombination genes and clinical outcomes of non-small cell lung cancer patients treated with definitive radiotherapy. PLoS ONE 6:e20055–e20055. https://doi.org/10.1371/journal.pone.0020055
Yoon MS, Nam T-K, Lee J-S et al (2011) VEGF as a predictor for response to definitive chemoradiotherapy and COX-2 as a prognosticator for survival in esophageal squamous cell carcinoma. JKMS 26:513–520. https://doi.org/10.3346/jkms.2011.26.4.513
Yoshida K, Suzuki S, Sakata J et al (2018) The upregulated expression of vascular endothelial growth factor in surgically treated patients with recurrent/radioresistant cervical cancer of the uterus. Oncol Lett 16:515–521. https://doi.org/10.3892/ol.2018.8610
Yoshimura Y, Oka M, Harada T (1990) Squamous cell carcinoma-antigen for detection of squamous cell and mucoepidermoid carcinoma after primary treatment: a preliminary report. J Oral Maxillofac Surg 48:1283–1288. https://doi.org/10.1016/0278-2391(90)90484-j
You G-R, Cheng A-J, Lee L-Y et al (2019) Prognostic signature associated with radioresistance in head and neck cancer via transcriptomic and bioinformatic analyses. BMC Cancer 19:64. https://doi.org/10.1186/s12885-018-5243-3
Yu T-K, Bhosale PR, Crane CH et al (2008) Patterns of locoregional recurrence after surgery and radiotherapy or chemoradiation for rectal cancer. Int J Radiat Oncol Biol Phys 71:1175–1180. https://doi.org/10.1016/j.ijrobp.2007.11.018
Zhang W, Xu J (2017) DNA methyltransferases and their roles in tumorigenesis. Biomark Res 5:1. https://doi.org/10.1186/s40364-017-0081-z
Zhang H, Xie C, Yue J et al (2017a) Cancer-associated fibroblasts mediated chemoresistance by a FOXO1/TGFβ1 signaling loop in esophageal squamous cell carcinoma. Mol Carcinog 56:1150–1163. https://doi.org/10.1002/mc.22581
Zhang Y, Dong S, Xu R et al (2017b) Prognostic and predictive role of COX-2, XRCC1 and RASSF1 expression in patients with esophageal squamous cell carcinoma receiving radiotherapy. Oncol Lett 13:2549–2556. https://doi.org/10.3892/ol.2017.5780
Zhang G, Zhang K, Li C et al (2019) Serum proteomics identify potential biomarkers for nasopharyngeal carcinoma sensitivity to radiotherapy. Biosci Rep. https://doi.org/10.1042/BSR20190027
Zhang M, Zhang L, Hei R et al (2021) CDK inhibitors in cancer therapy, an overview of recent development. Am J Cancer Res 11:1913–1935
Zhao L, Tang M, Hu Z et al (2015) miR-504 mediated down-regulation of nuclear respiratory factor 1 leads to radio-resistance in nasopharyngeal carcinoma. Oncotarget 6:15995–16018. https://doi.org/10.18632/oncotarget.4138
Zhao J, Zhang P, Wang J et al (2017) Plasma levels of soluble programmed death ligand-1 may be associated with overall survival in nonsmall cell lung cancer patients receiving thoracic radiotherapy. Medicine (baltimore) 96:e6102–e6102. https://doi.org/10.1097/MD.0000000000006102
Zheng Q, Ding H, Wang L et al (2021) Circulating exosomal miR-96 as a novel biomarker for radioresistant non-small-cell lung cancer. J Oncol. https://doi.org/10.1155/2021/5893981
Zhou Y, Chu L, Wang Q et al (2018) CD59 is a potential biomarker of esophageal squamous cell carcinoma radioresistance by affecting DNA repair. Cell Death Dis 9:887. https://doi.org/10.1038/s41419-018-0895-0
Zhu P, Ou Y, Dong Y et al (2016) Expression of VEGF and HIF-1α in locally advanced cervical cancer: potential biomarkers for predicting preoperative radiochemotherapy sensitivity and prognosis. Onco Targets Ther 9:3031–3037. https://doi.org/10.2147/OTT.S104142
Zlobec I, Vuong T, Compton CC et al (2008) Combined analysis of VEGF and EGFR predicts complete tumour response in rectal cancer treated with preoperative radiotherapy. Br J Cancer 98:450–456. https://doi.org/10.1038/sj.bjc.6604172
Zou Z, Chang H, Li H, Wang S (2017) Induction of reactive oxygen species: an emerging approach for cancer therapy. Apoptosis 22:1321–1335. https://doi.org/10.1007/s10495-017-1424-9
Acknowledgements
No funds, grants, or other support were received.
Funding
No funding was secured for this manuscript.
Author information
Authors and Affiliations
Contributions
IL and ED had the idea for the article, IL, MR, EI and YT performed the literature search and data analysis, IL, AC and ED drafted and critically revised the work.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Larionova, I., Rakina, M., Ivanyuk, E. et al. Radiotherapy resistance: identifying universal biomarkers for various human cancers. J Cancer Res Clin Oncol 148, 1015–1031 (2022). https://doi.org/10.1007/s00432-022-03923-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-03923-4