Abstract
Purpose
To investigate structure–function associations between retinal thickness, visual acuity (VA), and contrast sensitivity (CS), using the quantitative contrast sensitivity function (qCSF) method in patients with idiopathic epiretinal membrane (ERM).
Methods
Retrospective, cross-sectional observational study. Patients with a diagnosis of idiopathic ERM were included. Patients underwent complete ophthalmic examination, spectral-domain optical coherence tomography imaging (SD-OCT) (SPECTRALIS® Heidelberg), and CS testing using the qCSF method. Outcomes included area under the log CSF (AULCSF), contrast acuity (CA), and CS thresholds at 1, 1.5, 3, 6, 12, and 18 cycles per degree (cpd).
Results
A total of 102 eyes of 79 patients were included. Comparing standardized regression coefficients, retinal thickness in most ETDRS sectors was associated with larger reductions in AULCSF, CA, and CS thresholds at 3 and 6 cpd than those in logMAR VA. These differences in effect on VA and CS metrics were more pronounced in the central subfield and inner ETDRS sectors. Among the retinal layers, increased INL thickness had the most detrimental effect on visual function, being significantly associated with reductions in logMAR VA, AULCSF, CA, and CS thresholds at 3 and 6 cpd (all p < .01), as well as at 1.5 and 12 cpd (p < .05).
Conclusion
Retinal thickness seems to be associated with larger reductions in contrast sensitivity than VA in patients with ERM. Measured with the qCSF method, contrast sensitivity may serve as a valuable adjunct visual function metric for patients with ERM.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.

Introduction
Epiretinal membrane (ERM) formation is a common retinal condition caused by fibrocellular proliferation on the surface of the inner retina [1]. As the condition progresses in its severity, the membrane contracts, causing increasing distortion of retinal layers [1, 2]. Despite nonsignificant change in visual acuity (VA), which measures the ability to distinguish targets in high contrast, patients with ERM still experience a decrease in contrast sensitivity (CS), which measures the ability to detect differences of light and dark between the target and its background [3]. Moreover, patients who undergo vitrectomy for ERM may experience a limited improvement in VA but experience a significant improvement in CS [4]. However, clinical implementation of traditional CS tests has been limited by their time-consuming nature and poor test–retest reliability [5, 6]. As a promising alternative, the quantitative CS function (qCSF) method employs an intelligent, novel active learning algorithm to measure CS across multiple spatial frequencies in a time-efficient manner and with high sensitivity and test–retest reliability [7]. The qCSF has been employed in multiple retinal conditions including macular degeneration [8, 9], retinal detachment [10], retinal vein occlusion [11, 12], central serous chorioretinopathy [13], and diabetic retinopathy [14]. Structure–function associations of qCSF-measured contrast sensitivity and imaging biomarkers are currently being investigated.
Spectral-domain optical coherence tomography (SD-OCT) is the standard of care imaging modality used in the routine clinical practice to obtain a detailed view of the retinal layers in eyes with ERM. Associating structural changes on the objective clinical assessment with functional outcomes will help clinicians to better understand the degree of subjective visual impairment experienced by the patients. To date, structure–function studies on ERMs have mainly focused on associations between OCT metrics and VA, reporting that ellipsoid zone disruption [15,16,17,18], central foveal thickness (CFT) [19, 20], and retinal cysts [17] are associated with decreased VA. Meanwhile, only few studies have studied the relationship of SD-OCT structural biomarkers and contrast sensitivity, employing the Optec 6500 vision testing system, CSV-1000E chart, and CGT-2000 [4, 21, 22] sine wave grating tests that have been shown to have poor test–retest reliability, especially at lower spatial frequencies [5, 6, 8].
Our group has previously used the qCSF method to show that in maculopathy patients with VA as good as 20/25 or even VA of 20/20–1 (logMAR 0.020), significant reductions in contrast sensitivity may still be observed [23]. Herein, we present a retrospective study using the qCSF method and SD-OCT to investigate structure–function associations between retinal thickness, VA, and contrast sensitivity in patients with ERM.
Methods
Study design
This was a retrospective, cross-sectional observational study performed at Massachusetts Eye and Ear (MEE). It adhered to the tenets of the Declaration of Helsinki. The institutional review board (IRB) of MEE and partners approved the study protocol.
Study subjects
Patients with a diagnosis of idiopathic unilateral or bilateral ERM were recruited during their scheduled clinical appointments from November 2017 to December 2021. Exclusion criteria included VA less than 20/200, presence of any other co-existing retinal disease, diagnosis of glaucoma, history of vitreoretinal or other intraocular surgery besides cataract, and improvement of VA more than 1 line with pinhole.
qCSF testing methodology and study outcomes
Prior to pupil dilation, study subjects underwent contrast sensitivity testing using the qCSF method on the Manifold Contrast Vision Meter (Adaptive Sensory Technology, San Diego, CA, USA) following a protocol previously described by our group [8]. In brief, the device uses an active learning algorithm to select and display personalized optotypes of various contrasts and spatial frequencies based on each individual subject’s prior responses, hence maximizing information gain [7]. This active learning system allows for CS testing over a wide range of contrast levels (128 possible contrasts, 0.0002% to 100%) and spatial frequencies (19 optotypes sizes, approximately 1–27 cycles per degree [cpd]) in a relatively quick testing time (2–5 min per eye), while operating with a great test–retest reliability [24].
The main outcomes of the qCSF method include area under the logarithm contrast sensitivity curve (AULCSF), contrast acuity (CA), and CS thresholds at six spatial frequencies (1 cpd, 1.5 cpd, 3 cpd, 6 cpd, 12 cpd, and 18 cpd). AULCSF represents a global measure of CSF, and CA measures the smallest optotype at the highest level of contrast (i.e., the spatial frequency where CS threshold is 100%, illustrated by the intersection of the CSF curve with the x-axis) [7]. Each CS threshold represents the lowest amount of contrast that can be seen at each spatial frequency [7, 8].
Spectral-domain OCT imaging and image analysis
SD-OCT images were obtained after pupil dilation, using the Heidelberg Spectralis (Spectralis HRA + OCT, Heidelberg Engineering, Inc., Heidelberg, Germany) on a 30° × 30° scanning protocol, during the same clinical visit that the qCSF was employed. Complete ophthalmological examination was performed, including Snellen VA measurement and application tonometry, and the lens status was graded as either normal, pseudophakic, or by stage of nuclear sclerosis (NS). Snellen VA without correction was used in the analysis given that qCSF testing also does not involve pinhole correction.
OCT scans were either automatically or manually segmented using the Heidelberg Eye Explorer review software according to the International Nomenclature for Optical Coherence Tomography Panel [25]. All images were manually evaluated to ensure sufficient quality and confirm proper placement of segmentation lines and Early Treatment of Diabetic Retinopathy Study (ETDRS) grid (comprised of inner and outer rings; diameters 1 mm, 3 mm, 6 mm). One image was excluded due to poor quality. Global and sectoral thicknesses of each retinal layer, including the nerve fiber layer (NFL), ganglion cell layer (GCL), inner plexiform layer (IPL), inner nuclear layer (INL), outer plexiform layer (OPL), and outer nuclear layer (ONL), were exported from the machine using a custom software developed at MEE.
Statistical analysis
Analysis was carried out using R V.1.3.959. Normally distributed data were described as mean with SD, while data which were not normally distributed were reported as median with IQR. Snellen VAs converted into LogMAR for analysis purposes. Mixed-effects multivariate linear regression models fit by restricted maximum likelihood were performed to account for the correlation of both eyes of the same patients. These mixed-effects multivariate models were used to assess the following qCSF outcomes as dependent variables: AULCSF, CA, CS thresholds at 1–18 cpd. A separate model was run for each ETDRS subfield and each retinal layer as an independent variable. The statistical significance level was set as p ≤ 0.250 on the initial univariate analyses for assessing all the potential confounders (including age, lens status, gender, race, and eye separately). A backward stepwise elimination procedure, based on the Akaike Information Criteria and statistical significance (p ≤ 0.05), was then used to achieve the final multivariate mixed-effects models presented. Unstandardized regression coefficients were converted to standardized regression coefficients to allow for comparisons between LogMAR VA and contrast sensitivity metrics. Lastly, a subgroup analysis using the same procedures was performed on patients with VA of 20/20 to examine changes in contrast sensitivity metrics.
Results
Demographic and clinical characteristics
A total of 102 eyes of 79 patients were enrolled in the study. Baseline demographic and clinical information are summarized in Table 1. The mean age of the study cohort was 68.2 ± 8.5 years. The median logMAR VA was 0.22 (0.07, 0.37), which translates to a 20/33 Snellen equivalent.
Associations between VA, contrast sensitivity, and retinal thickness per ETDRS subfield
When controlling for age and race, standardized regression coefficients (b) for associations of total retinal thickness with visual acuity and contrast sensitivity metrics per ETDRS subfield are shown in Fig. 1A. An increase in retinal thickness was associated with LogMAR VA at the outer temporal region (unstandardized regression coefficient, B = 1.38; p = 0.015). Contrast metrics which showed the greatest number of associations with regional retinal thicknesses were AULCSF, CA, and CS thresholds at 3 cpd and 6 cpd. Retinal thickness significantly associated with AULCSF in the outer temporal (B = − 1.81, p = 0.010), inner temporal (B = 1.01, p = 0.039), outer superior (B = − 1.42, p = 0.042), inner superior (B = − 1, p = 0.043), and outer nasal region (B = − 1.58, p. 0.036). Retinal thickness was associated with CA at the outer inferior (B = − 1.28, p = 0.023), outer temporal (B = − 1.11, p = 0.021), and outer nasal region (B = − 1.07, p = 0.036). Further, retinal thickness was associated with CS threshold at 3 cpd at the outer temporal (B = − 1.63, p = 0.016), inner temporal (B = − 1, p = 0.024), and inner superior (B = − 0.66, p = 0.021). Retinal thickness was associated with CS threshold at 6 cpd at the outer inferior (B = − 0.46, p = 0.014), outer temporal (B = − 0.47, p = 0.014), outer superior (B = − 0.42, p = 0.022), and outer inferior region (B = − 0.43, p = 0.037).
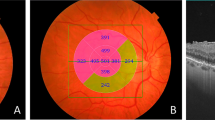
A Standardized regression coefficients (b) for associations of total retinal thickness with visual acuity and contrast sensitivity metrics (AULCSF curve, CA, and contrast sensitivity CS thresholds) per ETDRS subfield in the total cohort of ERM patients and B in a subgroup analysis of patients with BCVA 20/30–3.. The left side of each grid represents the temporal retina, and the right side represents the nasal retina. * signifies statistically significant association at the level of p < .05
Associations between VA, contrast sensitivity, and individual retinal layer thicknesses
Standardized regression coefficients (b) for the association of logMAR VA or contrast sensitivity metrics are outlined in Table 2. When controlling for race (none of the other variables were found to be significant enough to be included in our final model), mixed-effects multiple linear regression analysis showed that increased INL thickness was significantly associated with worse logMAR VA (B = 0.011, p = 0.001), AULCSF (B = − 0.016, p = 0.003), CA (B = − 0.01, p = 0.006), and decreased CS thresholds at 1.5 cpd (B = − 0.008, p = 0.036), 3 cpd (B = − 0.014, p = 0.005), 6 cpd (B = − 0.022, p = 0.004), and 12 cpd (B = − 0.01, p = 0.041). Additionally, increased OPL thickness was found to be significantly associated with worse LogMAR VA (B = 0.014, p = 0.010) and decreased CS threshold at 3 cpd (B = − 0.017, p = 0.025). Increased ONL thickness was associated with decreased CA (B = − 0.004, p = 0.039). The results are summarized in Fig. 2.
When investigating the effect of individual retinal layer thicknesses in specific ETDRS subfields on contrast sensitivity and VA, INL showed significant associations with visual function metrics in the greatest number of ETDRS sectors compared to any other retinal layer (Supplemental Table 1). The INL showed significant regional associations with logMAR VA, AULCSF, and CS thresholds at 1 cpd, 3 cpd, and 6 cpd and 12 cpd (Supplemental Table 1).
Subgroup analysis
A subgroup analysis was performed in patients with BCVA 20/20–3 or better. The subset included 23 eyes of 21 patients. Baseline demographics for this group are listed in Table 1. When controlling for eye (no other variable was significant enough to be included in the final multivariate model), associations of total retinal thickness with contrast sensitivity metrics per ETDRS subfield are shown in Fig. 1B. Additionally, when controlling for eye, increased INL thickness was significantly associated with worse CS thresholds at 3 cpd and 6 cpd (Table 3, Fig. 2).
Discussion
In this retrospective cross-sectional observational study, we present a cohort of 102 ERM eyes investigating structure–function associations between retinal thickness (total, regional and individual layer thickness), visual acuity, and contrast sensitivity. This is the first ERM study where contrast sensitivity is measured by employing the active-learning qCSF platform, which is reliable, sensitive, and feasible for clinical practice. This study offers valuable insight into the contribution of both regional and individual retinal thicknesses on contrast sensitivity and provides a deeper understanding of the implications of structural changes on visual function in patients with ERM.
Overall, our results show that in patients with ERM, increased total retinal thickness seems to be associated with larger reductions in contrast sensitivity than in VA, suggesting that contrast sensitivity may be a valuable adjunct metric of visual function for routine clinical practice. This derives from the fact that the absolute values of the standardized regression coefficients for the central ETDRS subfield are more than double for the associations with AULCSF and CS thresholds at 1.5, 3, 6, and 12 cpd compared to the respective regression coefficient for VA (Fig. 1A). Moreover, the absolute value of the standardized regression coefficients for the four inner ETDRS subfields are consistently larger for the associations with AULCSF, CA, and CS thresholds at 3 and 6 cpd compared to that for VA (Fig. 1A).
Increased total retinal thickness in the outer ETDRS subfields was found to be significantly associated with reductions in contrast sensitivity (more pronounced in AULCSF and CS thresholds at 6 cpd), while significant association with decreased VA was present only in the outer temporal subfield. Further, increased total retinal thickness in the inner temporal ETDRS subfield was significantly associated with reduced AULCSF and CS threshold at 3 cpd but not with significantly reduced VA (Fig. 1A). Lastly, even in patients with BCVA ≥ 20/20–3, retinal thickness is still significantly associated with decreased AULCSF and CS thresholds at 3 and 6 cpd (Fig. 1B).
This is one of the first studies, that we know of, to investigate regional retinal thickness in relation to contrast sensitivity in patients with ERM. Previous literature on the effect of CFT on visual acuity rendered mixed results [16, 17, 19, 20, 26]. Only one study showed a reduction in contrast sensitivity in patients with non-foveal ERM; however, this study focused on a population only with multifocal intraocular lenses [22], which are known to be associated with reduced contrast sensitivity [27]. Moreover, this study used the CGT-2000 instrument (Takagi, Seiko, Japan), which tests only CS thresholds at ranging from 0.64 to 6.3 cpd [6] and has been shown to have poor repeatability [6]. Using the qCSF method, our results suggest that CFT may not be the most appropriate indicator of visual dysfunction in all patients with ERM, which is clinically intuitive as the area of traction caused by the ERM is not always limited to the fovea.
When investigating the effect of individual retinal layer thickness on functional outcomes, our results suggest that of all the retinal layers affected by ERMs, increased INL thickness is the most detrimental to visual function. In specific, increased INL thickness is significantly associated with worse LogMAR VA, as well as decreased AULCSF, contrast acuity, and decreased CS thresholds at 1.5, 3, 6, and 12 cpd. Further, increased OPL thickness is significantly associated with decreased VA and decreased CS threshold at 3 cpd. Comparing standardized regression coefficients derived from our mixed-effects regression models for the above associations, it seems that increased INL thickness in ERMs is associated with changes in contrast sensitivity outcomes (AULCSF and CS thresholds at 3 and 6 cpd) nearly equal to changes in VA (Table 2). Additionally, even in a subgroup analysis of patients with BCVA ≥ 20/20–3, increased INL thickness is still significantly associated with decreased CS thresholds at 3 and 6 cpd.
To the best of our knowledge, literature on individual retinal layer thickness and contrast sensitivity has been scarce. So far, VA has been mainly associated with thickening of the INL and OPL [28,29,30]. Other studies have correlated metamorphopsia with INL thickness [30,31,32]. Our study offers unique insight into the effect of these individual retinal layers on contrast sensitivity.
Among the limitations of this analysis is that it did not account for ERM stage, or other morphological changes that may occur in the presence of ERM, such as presence of retinal cysts, lamellar holes, cystoid macular edema, or ellipsoid zone disruption. These characteristics could be the focus of future studies. Some characteristics present in the eyes of this study (i.e., ectopic foveal inner layers [2], cotton ball sign [33], and microcystic edema could have had an effect on the retinal layer segmentation for our study. However, these scans were manually checked to ensure accurate delineation of retinal layers. Second, as this was a cross-sectional study, no information on the effect of vitrectomy/ERM peeling on contrast sensitivity could be drawn; future work on that would be valuable. Furthermore, given the existing qCSF protocol in place at our institution, we compared VA and CS data without pinhole correction, but it would be interesting to see how the relative correlation between retinal thickness and these visual outcomes differ when correction is applied.
In conclusion, total retinal thickness seems to be associated with larger reductions in contrast sensitivity than in VA, especially in the central subfield and four inner ETDRS subfields. When investigating the effect of individual retinal layer thicknesses on visual function, increased INL thickness seems to be most detrimental to contrast sensitivity and VA. qCSF-measured contrast sensitivity seems to be a valuable adjunct metric of visual function in patients with ERM.
Data availability
Data are available upon reasonable request. Not applicable.
References
Hiscott P, Hagan S, Heathcote L et al (2002) 2002) Pathobiology of epiretinal and subretinal membranes: possible roles for the matricellular proteins thrombospondin 1 and osteonectin (SPARC. Eye 164(16):393–403. https://doi.org/10.1038/sj.eye.6700196
Govetto A, Lalane RA, Sarraf D et al (2017) Insights into epiretinal membranes: presence of ectopic inner foveal layers and a new optical coherence tomography staging scheme. Am J Ophthalmol 175:99–113. https://doi.org/10.1016/J.AJO.2016.12.006
Nishi Y, Shinoda H, Uchida A et al (2013) Detection of early visual impairment in patients with epiretinal membrane. Acta Ophthalmol 91:e353–e357. https://doi.org/10.1111/AOS.12060
Sugiura Y, Okamoto F, Okamoto Y et al (2014) Contrast sensitivity and foveal microstructure following vitrectomy for epiretinal membrane. Invest Ophthalmol Vis Sci 55:7594–7600. https://doi.org/10.1167/IOVS.14-14035
Richman J, Spaeth GL, Wirostko B (2013) Contrast sensitivity basics and a critique of currently available tests. J Cataract Refract Surg 39:1100–1106. https://doi.org/10.1016/J.JCRS.2013.05.001
Pesudovs K (2007) Takagi Glare Tester CGT-1000 for contrast sensitivity and glare testing in normal individuals and cataract patients. J Refract Surg 23:492–498. https://doi.org/10.3928/1081-597X-20070501-13
Lesmes LA, Lu ZL, Baek J, Albright TD (2010) Bayesian adaptive estimation of the contrast sensitivity function: the quick CSF method. J Vis 10:17–17. https://doi.org/10.1167/10.3.17
Vingopoulos F, Wai KM, Katz R et al (2021) Measuring the contrast sensitivity function in non-neovascular and neovascular age-related macular degeneration: the quantitative contrast sensitivity function test. J Clin Med 10:10. https://doi.org/10.3390/JCM10132768
Ou WC, Lesmes LA, Christie AH et al (2021) Normal- and low-luminance automated quantitative contrast sensitivity assessment in eyes with age-related macular degeneration. Am J Ophthalmol 226:148–155. https://doi.org/10.1016/J.AJO.2021.01.017
Thomas M, Silverman RF, Vingopoulos F et al (2020) Active learning of contrast sensitivity to assess visual function in macula-off retinal detachment: J Vitreoretin Dis 5:313–320. https://doi.org/10.1177/2474126420961957
Silverman RF, Kasetty M, Vingopoulos F et al (2020) Measuring contrast sensitivity function with active learning in retinal vein occlusion: a new endpoint of visual function. Ophthalmic Surg Lasers Imaging Retina 51:392–400. https://doi.org/10.3928/23258160-20200702-04
Wang J, Cui Y, Vingopoulos F et al (2022) Disorganisation of retinal inner layers is associated with reduced contrast sensitivity in retinal vein occlusion. Br J Ophthalmol 106:241–245. https://doi.org/10.1136/BJOPHTHALMOL-2020-317615
Vingopoulos F, Garg I, Kim EL et al (2022) Quantitative contrast sensitivity test to assess visual function in central serous chorioretinopathy. Br J Ophthalmol. https://doi.org/10.1136/BJOPHTHALMOL-2021-320415
Joltikov KA, de Castro VM, Davila JR, et al (2017) Multidimensional functional and structural evaluation reveals neuroretinal impairment in early diabetic retinopathy. Invest Ophthalmol Vis Sci 58:BIO277–BIO290. https://doi.org/10.1167/IOVS.17-21863
Inoue M, Morita S, Watanabe Y et al (2011) Preoperative inner segment/outer segment junction in spectral-domain optical coherence tomography as a prognostic factor in epiretinal membrane surgery. Retina 31:1366–1372. https://doi.org/10.1097/IAE.0B013E318203C156
Watanabe K, Tsunoda K, Mizuno Y et al (2013) Outer retinal morphology and visual function in patients with idiopathic epiretinal membrane. JAMA Ophthalmol 131:172–177. https://doi.org/10.1001/JAMAOPHTHALMOL.2013.686
Michalewski J, Michalewska Z, Cisiecki S, Nawrocki J (2007) Morphologically functional correlations of macular pathology connected with epiretinal membrane formation in spectral optical coherence tomography (SOCT). Graefes Arch Clin Exp Ophthalmol 245:1623–1631. https://doi.org/10.1007/S00417-007-0579-4
Koo HC, Rhim W, Lee EK (2012) Morphologic and functional association of retinal layers beneath the epiretinal membrane with spectral-domain optical coherence tomography in eyes without photoreceptor abnormality. Graefes Arch Clin Exp Ophthalmol 250:491–498. https://doi.org/10.1007/S00417-011-1848-9
Lo D, Heussen F, Ho HK et al (2012) Structural and functional implications of severe foveal dystopia in epiretinal membranes. Retina 32:340–348. https://doi.org/10.1097/IAE.0B013E31821DBB35
Romano MR, Cennamo G, Amoroso F et al (2017) Intraretinal changes in the presence of epiretinal traction. Graefes Arch Clin Exp Ophthalmol 255:31–38. https://doi.org/10.1007/s00417-016-3413-z
Liu L, Wang Y, Liu J, Liu W (2018) Retinal-image quality and contrast sensitivity function in eyes with epiretinal membrane: a cross-sectional observational clinical study. BMC Ophthalmol 18. https://doi.org/10.1186/S12886-018-0957-1
Kim B, Kwon S, Choi A, Jeon S (2021) Influence of mild non-foveal involving epiretinal membrane on visual quality in eyes with multifocal intraocular lens implantation. Graefes Arch Clin Exp Ophthalmol 259:2723–2730. https://doi.org/10.1007/S00417-021-05225-W
Wai KM, Vingopoulos F, Garg I, et al (2021) Clinical science contrast sensitivity function in patients with macular disease and good visual acuity. Br J Ophthalmol 0:1–6. https://doi.org/10.1136/bjophthalmol-2020-318494
F H, LA L, W K, et al (2016) Evaluating the performance of the quick CSF method in detecting contrast sensitivity function changes. J Vis 16. https://doi.org/10.1167/16.6.18
Staurenghi G, Sadda S, Chakravarthy U, Spaide RF (2014) Proposed lexicon for anatomic landmarks in normal posterior segment spectral-domain optical coherence tomography: the IN•OCT consensus. Ophthalmology 121:1572–1578. https://doi.org/10.1016/J.OPHTHA.2014.02.023
Kinoshita T, Imaizumi H, Okushiba U et al (2012) Time course of changes in metamorphopsia, visual acuity, and OCT parameters after successful epiretinal membrane surgery. Invest Ophthalmol Vis Sci 53:3592–3597. https://doi.org/10.1167/IOVS.12-9493
de Silva SR, Evans JR, Kirthi V, et al (2016) Multifocal versus monofocal intraocular lenses after cataract extraction. Cochrane database Syst Rev 12. https://doi.org/10.1002/14651858.CD003169.PUB4
Cacciamani A, Cosimi P, Ripandelli G et al (2021) Epiretinal membrane surgery: structural retinal changes correlate with the improvement of visual function. J Clin Med 10:1–10. https://doi.org/10.3390/JCM10010090
Cacciamani A, Cosimi P, Di Nicola M et al (2019) Correlation between outer retinal thickening and retinal function impairment in patients with idiopathic epiretinal membranes. Retina 39:331–338. https://doi.org/10.1097/IAE.0000000000001971
Kim JH, Kang SW, Kong MG, Ha HS (2013) Assessment of retinal layers and visual rehabilitation after epiretinal membrane removal. Graefes Arch Clin Exp Ophthalmol 251:1055–1064. https://doi.org/10.1007/S00417-012-2120-7
Ichikawa Y, Imamura Y, Ishida M (2018) Inner nuclear layer thickness, a biomarker of metamorphopsia in epiretinal membrane, correlates with tangential retinal displacement. Am J Ophthalmol 193:20–27. https://doi.org/10.1016/J.AJO.2018.06.001
Okamoto F, Sugiura Y, Okamoto Y et al (2015) Inner nuclear layer thickness as a prognostic factor for metamorphopsia after epiretinal membrane surgery. Retina 35:2107–2114. https://doi.org/10.1097/IAE.0000000000000602
Tsunoda K, Watanabe K, Akiyama K et al (2012) Highly reflective foveal region in optical coherence tomography in eyes with vitreomacular traction or epiretinal membrane. Ophthalmology 119:581–587. https://doi.org/10.1016/J.OPHTHA.2011.08.026
Funding
NIH P30 EY003790 (TE and MW), NIH R00 EY028631 (MW), Alcon Young Investigator Grant (MW).
Author information
Authors and Affiliations
Contributions
Concept and design: RZ, FV, LAK, DGV, DH, JBM. Data collection: RZ, FV, AB, HEW, GB, RK, TK. Data analysis: RZ, MW, TE. Original manuscript draft: RZ, FV. Critical revision of manuscript: RZ, FV, MW, AB, HEW, GB, RK, TK, TE, LAK, DGV, DH, JBM. Guarantors: RZ, FV, JBM.
Corresponding author
Ethics declarations
Ethics approval
This study involves human participants and was approved by the institutional review board at Massachusetts Eye and Ear.
Consent for publication
Consent was waived due to the retrospective nature of the study.
Competing interests
LAK has received research support from the National Eye Institute (R01EY027739) and CureVac AG and has a financial arrangement with Pykus Therapeutics. DH is a consultant for Allergan, Genentech, Omeicos Therapeutics and has received financial support from the National Eye Institute, Lions VisionGift, Commonwealth Grant, Lions International, Syneos LLC, and the Macular Society. DGV is a consultant for Valitor and OliX Pharmaceuticals and has received financial support from the National Eye Institute and by grants from the National Institute of Health (R01EY025362 and R21EY0203079), Research to Prevent Blindness, Loeffler’s Family Foundation, Yeatts Family Foundation, and Alcon Research Institute. JBM is a consultant for Alcon, Allergan, Carl Zeiss, Sunovion, and Genentech.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zeng, R., Vingopoulos, F., Wang, M. et al. Structure–function association between contrast sensitivity and retinal thickness (total, regional, and individual retinal layer) in patients with idiopathic epiretinal membrane. Graefes Arch Clin Exp Ophthalmol 261, 631–639 (2023). https://doi.org/10.1007/s00417-022-05819-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-022-05819-y