Abstract
Purpose
Female patients with pelvic floor diseases may suffer from several sexual disorders and sexual life impairments. The aim of this manuscript was to evaluate sexual dysfunction in female patients presenting with faecal incontinence (FI) and defecation disorder (DD).
Methods
A retrospective review was performed of a prospectively collected database of sexually active women referred to the pelvic floor clinic, who completed the Pelvic Organ Prolapse/Incontinence Sexual Questionnaire-12 (PISQ-12) at first visit. Statistical analysis was performed to evaluate and compare sexual dysfunction between patients with FI and DD and with published data on the general population. Regression analysis was used to identify predictors of sexual dysfunction and surgery.
Results
Three hundred thirteen patients were included, 192 (61%) with FI and 121 (39%) with DD. The patients with DD received more non-gynaecological surgical procedures (p = 0.023). More patients with DD received surgery for their current pelvic floor disease (p < 0.001). Major sexual impairment (PISQ-12 < 30) was found in 100 patients (31.9%). The mean PISQ-12 (33.2 ± 7.2) score was by 5 points lower than those reported in the general population from PISQ-validating studies. Prior anorectal surgery (odds ratio (OR) = 15.4), partner ejaculation problems (PISQ item 11, OR = 2.5), reduced sexual arousal (item 2, OR = 2.1), and orgasm perception (item 13, OR = 2.1) were the strongest predictors of worse sexual function in patients with FI. Patients with DD were almost 15 times more likely to receive subsequent surgery (OR = 14.6, p < 0.001), whereas fear of urine leakage almost doubled the risk.
Conclusions
Sexual dysfunction is prevalent among patients suffering from FI and DD, and questionnaires are useful in recognizing these patients. Subsequent surgery is more common for patients with DD compared to those with FI.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Sexual dysfunction is considered one of the most prevalent psychological disorders in the general population [1]. Female patients with pelvic floor disturbances are reported to suffer from many sexual disorders and sexual life impairments, although they tend not to restrict sexual activities [2, 3]. However, long term, this can lead to abstinence of sexual intercourse with detrimental effect on the relation with partners [3].
The management of these patients is challenging. Treatments must aim to ameliorate their symptoms and to improve their health perception. Improvement of sexual life represents a major endpoint of the management of patients with pelvic floor disturbances, and has resulted in several validated scales and questionnaires aimed at assessing their sexual function [4].
Our aims were to assess the prevalence of sexual dysfunction in patients referred to our pelvic floor clinic with a validated questionnaire, seeking for modifiers of their sexual life and for predictors of subsequent need of surgery for their current problem.
Methods
A retrospective review was performed to identify sexually active female patients presenting at the pelvic floor clinic of the Chelsea and Westminster Hospital, London, between 2008 and 2016, who have filled in the Pelvic Organ Prolapse/Incontinence Sexual Questionnaire-12 (PISQ-12).
Patients were excluded from the study if:
-
1.
They were not sexually active.
-
2.
They refused to fill in the questionnaire.
-
3.
They did not answer more than two of the questions in the questionnaire.
-
4.
They were not able to understand the study and to complete the forms.
This study met the guidelines of Good Clinical Practice.
Definitions and patient groups
Patients were classified as suffering from either faecal incontinence (FI) or structural defecatory disorders (DD) [5].
During history taking, patients were asked regarding previous pregnancies, number and modality of deliveries, and previous surgery. This was classified as abdominal non-gynaecological, gynaecological, anorectal, or pelvic floor surgery. All patients underwent complete clinical examination, and further tests were performed as required. Treatment was planned according to clinical and imaging/manometric findings, and included (1) conservative measures (i.e. observation, medications, biofeedback), (2) semi-invasive measures (i.e. Peristeen, sacral nerve stimulation), and (3) surgery. Follow-up visits were scheduled accordingly.
Data of interest and tools
Patients were asked to fill in the PISQ-12 [4] at their first appointment. The PISQ-12 is the short form of the PISQ-31, a validated tool for assessing sexual function of patients with both urinary and defecatory disorders [4]. It includes 12 questions, and responses are graded on a 5-point Likert scale from 0 (never) to 4 (always). Three subscales can be identified in PISQ-12 based on the assessed dimension, the Behavioural/Emotive, the Physical and the Partner-Related subscale. Scores range between 0 and 48, high scores being associated with better sexual function. The total score was calculated by adding the score from each question and by subtracting the score of questions 1 to 4.
Missing values in ≤2 items were obtained by calculating the number of items by the mean of available items. If more than 2 items were missing, patients were excluded from the analysis [4].
There is no agreement on how to interpret the score of PISQ-12. A study on patients with pelvic floor disturbances defined the minimal important difference in PISQ-31 as the difference in score that is representative of what patients and physicians perceive as beneficial or harmful eventually leading to a proper change in the management, and suggested that this can be estimated as 6 [6]. In terms of PISQ-12, 6 is equal to 2.3 (6 divided by 2.58 [4]). Papers that validated the PISQ-12 with samples from the general population with no diseases reported that the overall scores may range between 34.7 [7] and 42.7 [8]. Patients with an overall score less than 30 were thought to be suffering with severe sexual dysfunction.
Patients were also investigated for associated disturbances of micturition and urine incontinence.
Outcomes
Primary outcomes
-
1.
Sexual function of patients suffering from faecal incontinence and defecation disorders by means of a validated questionnaire
Secondary outcomes
-
1.
To compare sexual function of patients suffering from faecal incontinence with that of patients suffering from defecation disorders
-
2.
To compare sexual function of patients suffering from faecal incontinence and defecation disorders with that of the general population
-
3.
To identify specific questionnaire items that predict sexual impairment in patients suffering from faecal incontinence and defecation disorders
-
4.
To identify other patient- or disease-related factors that predict sexual impairment in patients suffering from faecal incontinence and defecation disorders
-
5.
To identify risk factors that predict subsequent need of surgery of interest
Statistical analysis
Fisher’s exact test was used for comparing dichotomous variables and Mann-Whitney U test for continuous variables. Each item score of the questionnaire was used to compare patients suffering from DD and FI. It is not recommended to evaluate subscales when using PISQ-12.
A control group from the general population was not available, but plenty of studies are available in the literature reporting on PISQ-12 in controls. The differences in the overall scores between the general population and patients suffering from pelvic floor disturbances range between 15 and 3 points. By assuming a lower score by at least 2 points, and assuming a standard deviation of 8 from previous papers, a sample of 314 patients would be able to reject the null hypothesis that the PISQ scores of the groups are equal with power of 88 and 5% type I error probability.
Binomial regression analysis was used to evaluate factors associated with worse sexual function (as defined previously <30) and with the need of subsequent surgery for current pelvic floor disease. p values <0.05 were considered to be statistically significant.
Statistical analysis was performed with the Statistical Package for Social Sciences (SPSS®) 17.0 (SPSS Inc., Chicago, IL).
Results
Patient characteristics
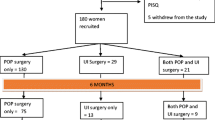
Overall, 401 patients were eligible for inclusion. Sixteen of them were diagnosed with chronic anal pain syndrome and were removed. Seventy-two patients either were discharged without need of further treatments or did not complete at least 10 items of the PISQ-12. Hence, 313 patients (mean age 42.2 ± 10.9 years) were included in the final analyses. Among them, 192 (61%) patients had FI and 121 (39%) had DD. Patient characteristics are depicted in Table 1. Patients with FI were younger than DD patients (p = 0.034). No differences were observed in terms of previous pregnancies or vaginal deliveries. Concerning previous surgery, patients with DD had underwent significantly more abdominal non-gynaecological surgical procedure (7.4 vs 2%, DD vs FI, p = 0.023). Significant urinary disturbances interfering with everyday activities were equally found among groups. Patients suffering from DD were also more likely to undergo subsequent surgery as maximal treatment for their current pelvic floor disorders at our clinic (17.3 vs 2.6%, DD vs FI, p < 0.001).
Sexual dysfunction
Two hundred and twelve patients (67.7%) reported PISQ-12 scores below 38, suggesting presence of sexual function impairment. Major sexual restrictions (as defined by total PISQ-12 <30) were observed in 100 patients (31.9%). No differences were observed between patients suffering from FI and patients suffering from DD (Table 2). The mean overall PISQ-12 score was 33.2 ± 7.2. This was by 5 points lower than the mean of scores reported in the general population from validating PISQ studies [4, 7, 8]. No differences were observed between patients with DD or FI in terms of total PISQ-12 scores and in scores of each variable (Table 2).
Figure 1 depicts the mean PISQ score for each item in FI and DD patients in this study, as well as mean scores of healthy individuals obtained from the literature. Controls had higher mean scores in all of the PISQ-12 items, with the exception of items 10 and 11, related to partner dysfunctions causing sexual restrictions. Subscales mean scores and total PISQ-12 are reported in Fig. 2. Controls had higher total PISQ-12 scores, and higher scores in both behavioural-emotional and structural dimensions, whereas no differences were observed in terms of partner-related variables compared with FI and DD patients.
Independent predictors of worse sexual function and subsequent surgery
Multivariate regression analysis was performed to identify the major factors which may predict severe sexual dysfunction (total PISQ-12 score below 30). The factors included in the multivariate regression analysis were (Tables 3 and 4) age; previous pregnancy/ies (yes vs no); vaginal delivery/ies (yes vs no); urinary disorders (yes vs no); previous abdominal (yes vs no), gynaecological (yes vs no), pelvic floor (yes vs no), or anorectal (yes vs no) surgery; diagnosis (DD vs FI); and all the individual items of PISQ-12.
Previous anorectal surgery (odds ratio (OR) 15.4, p = 0.016) was the strongest predictor of worse sexual function. The individual PISQ-12 items that were the main predictors of severe sexual impairment were partner ejaculation problems (item 11, OR 2.5, p = 0.009), reduced sexual arousal during intercourse (item 2, OR 2.1, p = 0.007), and orgasm perception (item 13, OR 2.1, p = 0.003) (Table 3). The same factors were tested in a multivariate analysis to predict subsequent surgery for current pelvic floor disorders.
Patients suffering from DD were almost 15 times more likely receiving subsequent surgery for current pelvic floor disorders. Restriction of sexual activity due to fear of urine leakage (item 7) almost doubled the risk (Table 4).
Subgroup analyses for predictors of worse sexual function and subsequent surgery
Subgroup regression analyses were performed. In patients with DD, partner erection problems (item 10; OR 6; 95% confidence interval (CI) 1.06, 34.32; p = 0.043), negative emotional feelings (item 10; OR 4.7; 95% CI 1.55, 13.97; p = 0.006), restriction of sexual activity due to fear of urine leakage (item 7; OR 4.6; 95% CI 1.51, 14.05; p = 0.007), and total PISQ-12 score (OR 2.6; 95% CI 1.25, 5.67; p = 0.011) were the strongest predictors for subsequent need of surgery.
Previous anorectal surgery was the strongest predictor of poor sexual function in patients with FI, as well as fear of urine leakage (item 7; OR 2.02; 95% CI 1.08, 3.8; p = 0.028) and partner ejaculation problems (item 11; OR 2.58; 95% CI 1.08, 6.17; p = 0.033).
Discussion
This study investigated the burden of sexual dysfunction in patients referred to a pelvic floor clinic. We demonstrated that many patients have at least a minor impairment of sexual function (67.7%), and almost one out of three suffers from major sexual disturbances, irrespective of baseline condition (FI or DD). The mean total PISQ-12 scores were lower than those reported by healthy controls in available validation papers of the scores from different countries by at least 5 points. Some patient-related and disease-related factors, as well as PISQ-12 items, are independently associated with clinical impairment of sexual function and subsequent need of surgery. These differ among FI and DD.
Female sexual function is recognized as an important aspect of women’s health, and it is being widely investigated over the last years [1, 9]. Studies have estimated that the prevalence of sexual dysfunctions in healthy women is not negligible and can be affected by several factors. The most common disturbances include orgasmic disorder (4–42% [10, 11]), sexual arousal disorder (6–21% [10, 12]), and dyspareunia (3–18% [12, 13]). Women suffering from pelvic floor disorders are at increased risk of sexual problems as compared with the general population, which can negatively affect their relationship with their partners [3, 7, 8]. However, these patients try to conduct a normal sexual life, even if impaired [3]. Hence, the effects of the treatments aimed at resolving FI and DD should take into account sexual function. PISQ-12 is a validated tool to assess sexual function in patients with pelvic floor disease, but there is no agreement on how to interpret the scores, as well as on the clinical relevance of each item. We were able to show a significant prevalence of sexual disorders in patients with pelvic floor disease, irrespective of their baseline condition (FI or DD). Up to 67.7% had at least a minor impairment and 31.9% had relevant restrictions of their sexual habits. Interestingly, the role of partner disturbances can be a less important modifier of sexual restrictions in DD and FI, as compared with the general population (Figs. 1 and 2). However, when other variables are taken into account, partner difficulties with erection and ejaculation were significantly associated with sexual restrictions in both groups (Tables 3 and 4). This may result from either primary male dysfunction or, more likely, the negative effect of female sexual dysfunction on couple life.
Subscales mean scores and total PISQ-12 in FI and DD patients and healthy individuals. As reported, the controls have higher total PISQ-12 scores and higher scores in both behavioural-emotional and structural dimensions, whereas no differences were observed in terms of partner-related variables compared with FI and DD patients. Blue FI, red DD, green controls
No studies have assessed the relevance of each dimension of the score separately and independently either in affecting the total PISQ-12 (and more importantly being clinically relevant for the patients) or in modifying the management of patients. Surgery can ameliorate sexual function in selected patients with pelvic floor disturbances [14–18]. Structural and functional tools are currently used to guide the treatment of patients. However, it has been showed that the restoration of pelvic anatomy does not linearly correlate with function and sexual life improvements [18], hence suggesting that the decision-making process in these patients requires a wider spectrum of influencing variables, among which sexual function may play a relevant role. These factors may differ between FI and DD patients.
Several patient- and disease-related factors have been associated with sexual dysfunction in patients with FI and DD. These include increasing age [19], pregnancy and delivery [20], and urinary disorders [21].
Even if we included age in the regression models to remove confounders, we did not exclude patients from the analysis based on age but rather excluded those who were not sexually active. This was supported by the fact that many women are still sexually active even if older and, more importantly, that pelvic floor diseases rather than ageing negatively affect sexual function in these patients [2]. It can hence be anticipated that age itself does not independently affect sexuality in pelvic floor diseases, which is consistent with our findings (Tables 3 and 4). Moreover, being non-sexually active has been reported as a predictor of failed PISQ-12 completion [22].
Irrespective of number of carriages and modality of delivery, previous pregnancies and delivery are associated with worse sexual function [20]. We were able to show that these factors are not associated with either more relevant impairment of function or subsequent need of surgery. However, they can be important and must be considered, as they can affect sexual life of patients for other than anatomical reasons, rather resulting from long-lasting emotional reactions to pregnancy and delivery [20]. We found that anorectal surgery in these patients may be more clinically relevant in terms of sexual function, due to manipulation of the sphincter complex. This facet should not be overlooked.
Urinary disturbances are often observed in patients with pelvic floor disorders, and are included in two items of the PISQ-12. Nevertheless, PISQ could not be the most appropriate tool to assess concomitant lower urinary tract symptoms [21]. We assessed urinary dysfunctions separately and included them in the analysis. Even if these were not independently associated with worse function or surgery, urinary items of PISQ-12 were. We would hence suggest that PISQ-12 is good enough to assess urinary dysfunctions that are likely to affect sexual function, even if the overall quality of life could be better assessed with other measurements. More importantly, the restrictions in sexual activities due to fear of urine incontinence are associated with higher odds of subsequent surgery (Table 4). This can be due to a more advanced pelvic floor disorder but could also be due to these patients having been keener to undergo surgery. This is suggested by our finding that, even if urine leakage is a major modifier of sexual function in FI, it is strongly associated with subsequent surgery in patients with DD.
The management of patients with pelvic floor disturbances is challenging, and surgery must be advocated selectively. We were able to show that urine dysfunctions can independently justify subsequent surgery in these patients. When assessing patients based on either DD or FI, in the former group, many factors besides urine leakage are useful to guide the surgical management, ranging from partner erection problems (OR 6) to negative emotional feelings (OR 4.7). Notably, the total PISQ-12 score (OR 2.6) can be useful to select DD patients for surgery. This is a new finding, with practical implications. Patients with DD were unsurprisingly more likely to receive surgery, and our findings can have a role in choosing the right treatment. The management of FI is challenging and ultimately needs case-by-case decisions which are poorly eased by standardized observations.
Study limitations
This study has several limitations. We did not establish a control group of patients to match PISQ-12 scores with our patients. Nevertheless, by analysing data from published series, we were able to obtain a more reliable estimation of sexual dysfunction in these patients by including controls from several countries. This is strength, as mean PISQ-12 scores in the general population can widely vary and no validated system is available to quantify the impairments and interpret PISQ scores. Moreover, for this comparison, the clinical importance might be more relevant than the statistical significance. The use of the PISQ-12 questionnaire in patients with FI has been questioned, and other questionnaires have been developed for these patients. Nevertheless, the PISQ-12 is applicable in this study because there is overlap in women with FI and urinary incontinence.
It should be considered that difference in subsequent surgery probably can be justified by the underlying diseases rather than sexual function. DD might be due to prolapse which could be surgically corrected, whereas there are not many successful options for surgically treating FI.
Lastly, our findings might have been influenced by a centre bias, due to specific referral. However, it is likely that they apply to all patients with DD and FI because our data were collected over a period of 8 years.
Strengths of this study include its sample size large enough to justify observation. The study was conducted at a teaching hospital, with several consultant surgeons acquiring data and assessing patients, hence making observations reliable and reproducible. This is the first study to look at predictors of worse sexual function in patients with FI and DD, and factors that may influence the subsequent management of patients.
Combined with a complete clinical assessment and instrumental tests, our findings can be useful in the management of these patients. The burden of sexual dysfunctions in patients with DD and FI was high, suggesting that sexual function should be assessed routinely with objective and validated measurements. It is important to estimate the clinical relevance of sexual problems. This could eventually result in better patient management; disclose unmet needs in these patients, including their relation with their partners; and identify those who may need proactive measures to cope with their dysfunction.
References
Spector IP, Carey MP (1990) Incidence and prevalence of the sexual dysfunctions: a critical review of the empirical literature. Arch Sex Behav 19(4):389–408
Panman CM, Wiegersma M, Talsma MN, Kollen BJ, Berger MY, Lisman-Van Leeuwen Y, Dekker JH (2014) Sexual function in older women with pelvic floor symptoms: a cross-sectional study in general practice. Br J Gen Pract 64(620):e144–e150. doi:10.3399/bjgp14X677518
Cichowski SB, Komesu YM, Dunivan GC, Rogers RG (2013) The association between fecal incontinence and sexual activity and function in women attending a tertiary referral center. Int Urogynecol J 24(9):1489–1494. doi:10.1007/s00192-013-2044-8
Rogers RG, Coates KW, Kammerer-Doak D, Khalsa S, Qualls C (2003) A short form of the Pelvic Organ Prolapse/Urinary Incontinence Sexual Questionnaire (PISQ-12). Int Urogynecol J Pelvic Floor Dysfunct 14(3):164–168 . doi:10.1007/s00192-003-1063-2discussion 168
Haylen BT, Maher CF, Barber MD, Camargo S, Dandolu V, Digesu A, Goldman HB, Huser M, Milani AL, Moran PA, Schaer GN, Withagen MI (2016) An International Urogynecological Association (IUGA) / International Continence Society (ICS) joint report on the terminology for female pelvic organ prolapse (POP). Int Urogynecol J 27(2):165–194. doi:10.1007/s00192-015-2932-1
Mamik MM, Rogers RG, Qualls CR, Morrow JD (2014) The minimum important difference for the Pelvic Organ Prolapse-Urinary Incontinence Sexual Function Questionnaire. Int Urogynecol J 25(10):1321–1326. doi:10.1007/s00192-014-2342-9
Shaaban MM, Abdelwahab HA, Ahmed MR, Shalaby E (2014) Assessment of female sexual function among women with pelvic organ prolapse or urinary incontinence via an Arabic validated short-form sexual questionnaire. Int J Gynaecol Obstet 124(1):24–26. doi:10.1016/j.ijgo.2013.06.031
Santana GW, Aoki T, Auge AP (2012) The Portuguese validation of the short form of the Pelvic Organ Prolapse/Urinary Incontinence Sexual Questionnaire (PISQ-12). Int Urogynecol J 23(1):117–121. doi:10.1007/s00192-011-1505-1
Lowenstein L, Pierce K, Pauls R (2009) Urogynecology and sexual function research. How are we doing? J Sex Med 6(1):199–204. doi:10.1111/j.1743-6109.2008.00968.x
Chandraiah S, Levenson JL, Collins JB (1991) Sexual dysfunction, social maladjustment, and psychiatric disorders in women seeking treatment in a premenstrual syndrome clinic. Int J Psychiatry Med 21(2):189–204
Read S, King M, Watson J (1997) Sexual dysfunction in primary medical care: prevalence, characteristics and detection by the general practitioner. J Public Health Med 19(4):387–391
Líndal E, Stefànsson JG (1993) The lifetime prevalence of psychosexual dysfunction among 55 to 57-year-olds in Iceland. Soc Psychiatry Psychiatr Epidemiol 28(2):91–95
Moody GA, Mayberry JF (1993) Perceived sexual dysfunction amongst patients with inflammatory bowel disease. Digestion 54(4):256–260
Pauls RN, Rogers RG, Parekh M, Pitkin J, Kammerer-Doak D, Sand P (2015) Sexual function in women with anal incontinence using a new instrument: the PISQ-IR. Int Urogynecol J 26(5):657–663. doi:10.1007/s00192-014-2563-y
Shimonov M, Groutz A, Schachter P, Gordon D (2015) Is bariatric surgery the answer to urinary incontinence in obese women? Neurourol Urodyn. doi:10.1002/nau.22909
Ramage L, Georgiou P, Tekkis P, Tan E (2015) Is robotic ventral mesh rectopexy better than laparoscopy in the treatment of rectal prolapse and obstructed defecation? A meta-analysis. Techniques in coloproctology 19(7):381–389. doi:10.1007/s10151-015-1320-7
Meriwether KV, Komesu YM, Craig E, Qualls C, Davis H, Rogers RG (2015) Sexual function and pessary management among women using a pessary for pelvic floor disorders. J Sex Med 12(12):2339–2349. doi:10.1111/jsm.13060
Thibault F, Costa P, Thanigasalam R, Seni G, Brouzyine M, Cayzergues L, De Tayrac R, Droupy S, Wagner L (2013) Impact of laparoscopic sacrocolpopexy on symptoms, health-related quality of life and sexuality: a medium-term analysis. BJU Int 112(8):1143–1149. doi:10.1111/bju.12286
Simons JS, Carey MP (2001) Prevalence of sexual dysfunctions: results from a decade of research. Arch Sex Behav 30(2):177–219
Botros SM, Abramov Y, Miller JJ, Sand PK, Gandhi S, Nickolov A, Goldberg RP (2006) Effect of parity on sexual function: an identical twin study. Obstet Gynecol 107(4):765–770. doi:10.1097/01.AOG.0000207677.03235.76
Cetinkaya SE, Dokmeci F, Dai O (2013) Correlation of pelvic organ prolapse staging with lower urinary tract symptoms, sexual dysfunction, and quality of life. Int Urogynecol J 24(10):1645–1650. doi:10.1007/s00192-013-2072-4
Collins SA, O‘Sullivan DM, LaSala CA (2010) Factors associated with PISQ-12 completion in an academic urogynecology population. Int Urogynecol J 21(5):579–582. doi:10.1007/s00192-009-1079-3
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
For this type of study, formal consent is not required.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
The abstract of this manuscript was accepted as a Lunchtime Poster at the ESCP 11th Scientific & Annual Meeting, Milan, September 28–30, 2016.
Rights and permissions
About this article
Cite this article
Pellino, G., Ramage, L., Simillis, C. et al. Evaluation of sexual dysfunction in female patients presenting with faecal incontinence or defecation disorder. Int J Colorectal Dis 32, 667–674 (2017). https://doi.org/10.1007/s00384-017-2795-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00384-017-2795-7