Abstract
To investigate the effects of stocking rates on nitrification activity and active nitrifying communities in a typical steppe grazing system, we conducted a laboratory incubation study using soil from a 10-year-old grassland gradient grazing experiment with sheep. A combination of molecular methods, such as DNA-based stable-isotope probing (DNA-SIP), real-time quantitative PCR, and high-throughput sequencing, was used to identify changes of nitrification activity and active nitrifying communities under different stocking rates (0 (SR0), 3 (SR3), 6 (SR6), and 9 (SR9) sheep per ha). The nitrification activity of soils was significantly increased by light grazing (SR3), while it was significantly decreased by heavy grazing (SR9). Nitrososphaera viennensis lineage of ammonia-oxidizing archaea (AOA) functionally predominated over ammonia-oxidizing bacteria (AOB) in nitrification in the SR3 soil, while the Nitrosospira cluster 3 of AOB was the major player in the SR9 soil. Therefore, stocking rates altered the distribution of active nitrifying communities by affecting soil chemical and physical conditions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Nitrification, the microbial oxidation of ammonia (NH4 +-N) to nitrite (NO2 −-N) and then to nitrate (NO3 −-N), is a critical component in the global nitrogen (N) cycling and occurs in a wide range of environments (Galloway et al. 2008). The NO3 −-N produced by nitrification is an essential N source for many plants in agricultural soils, while excessive NO3 −-N derived from synthetic nitrogen fertilizer causes water pollution through runoff from soil (Morimoto et al. 2011). This leads to a serious waste of resources. Nitrous oxide (N2O), which is both a potent greenhouse gas and an ozone-depletion gas, is produced as a by-product of nitrification (Conrad 1996; Di et al. 2014).
Ammonia-oxidizing bacteria (AOB) and ammonia-oxidizing archaea (AOA) are generally understood to be the two groups of microbes carrying out the first and rate-limiting step of nitrification (Könneke et al. 2005). The higher number of AOA amoA copy numbers than those of AOB seems to be a common feature in a wide variety of environments (Szukics et al. 2012; Sterngren et al. 2015; Zhong et al. 2016). A growing body of evidence shows an important role of AOA in nutrient-depleted soils, but a more dominant role by AOB in N-rich soils (Di et al. 2009, 2014; Sterngren et al. 2015; Wang et al. 2016; Zhong et al. 2016). The second step of the nitrification process, nitrite oxidation, has received much less attention than ammonia oxidation, as the physiological diversity of nitrite-oxidizing bacteria (NOB) remains poorly understood (Sorokin et al. 2012). NOB are composed of six genera: Nitrospira, Nitrobacter, Nitrococcus, Nitrotoga, Nitrolancetus, and Nitrospina (Alawi et al. 2007; Attard et al. 2010; Daims et al. 2011; Sorokin et al. 2012). The first two species are believed to be the two major players for nitrite producers in terrestrial ecosystems (Bartosch et al. 2002; Cebron and Garnier 2005; Noll et al. 2005). Nitrobacter-like NOB were hypothesized as r-strategists favored under high N availability, while Nitrospira-like NOB as K-strategists with high nitrite affinity (Attard et al. 2010). It has been proposed that nitrite oxidation was mainly performed by Nitrospira instead of Nitrobacter in paddy soils (Wang et al. 2015).
Grassland is one of the largest terrestrial ecosystems in the world, accounting for up to 40% of the total land area in China (Nan 2005). In order to meet the rising demand for food as a result of human population growth, stocking rates in grazed grassland are increasing. This land use intensification also has environmental consequences. Specifically, heavy grazing decreases plant species diversity (Reeder and Schuman 2002), significantly deteriorates the physical and chemical parameters of steppe topsoil and accelerates soil erosion (Steffens et al. 2008), exerts significant impact on plant N uptake (Jiang et al. 2016), adversely impacts the global C and N balance, and further contributes to triggering climate change in the future (Lal 2004; He et al. 2011; Mcsherry and Ritchie 2013; Zhou et al. 2017). For example, urine-N from grazing livestock is not only a major source of N for plant uptake; it can also lead to increased NO3 −-N leaching and N2O emissions (Ryden et al. 1984; Di and Cameron 2002, 2016).
In grassland soils, mounting studies have focused on the effects of grazing on nitrification-related functional microbes. Community compositions of AOA and AOB were distinctly influenced by grazing (Patra et al. 2006; Le Roux et al. 2008). Grazing also increased the abundances of AOA and AOB in grazed temperate grassland soils (Le Roux et al. 2008; Xie et al. 2014). Nitrosospira cluster 3a of AOB and group І.1b of AOA were previously identified as of key importance in grassland soils (Le Roux et al. 2008; Pan et al. 2016). Nonetheless, it remains elusive whether ammonia oxidation is exclusively or predominantly linked to archaea as archaeal amoA genes are greatly outnumbered by their bacterial counterparts in grassland (Zhong et al. 2014; Xie et al. 2014; Pan et al. 2016). It is an important step forward for sustainable development of grassland soil to understand the nitrification activity and active nitrifying community under different stocking rates (Vitousek and Howarth 1991). Besides, our understanding is very limited on how stocking rates may affect the nitrification activity and distribution of active nitrifiers as well as their relative roles in N cycling in grazed grassland soil.
We therefore conducted an incubation experiment to study nitrification activity and the community abundance and composition of active AOA, AOB, and NOB communities in an Inner Mongolia grassland soil under different stocking rates. DNA-stable isotope probe (SIP) and high-throughput fingerprinting of 16S ribosomal RNA (rRNA) techniques were used to identify active nitrifiers in these soils. Both labeled CO2 and urea were used to avoid the dilution of labeled CO2 by CO2 produced during decomposition of urea, although the amount of CO2 produced from urea was small compared with the added labeled CO2. The objectives were (1) to investigate the effects of long-term stocking rates on nitrification activity and active nitrifying communities; (2) to identify the active nitrifiers and real players in nitrification under long-term stocking rates; and (3) to assess the main abiotic factors to explain observed changes in active nitrifying communities. Findings of this work will be helpful for us to understand and better manage the N cycle in grazed grassland ecosystems.
Materials and methods
Site description and soil sampling
The experimental area was established in 2005 at the Inner Mongolia Grassland Ecosystem Research Station (IMGERS, 43° 37′ N, 116° 43′ E) on the Xilingol steppe of the Xilin River basin, bounded by the west side of the Daxing-An Mountain. The region is at an altitude of 1220–1280 m above sea level and has a temperate semiarid climate, with very cold winters and cool summers. The mean annual temperature is 0.3 °C, with the minimum and maximum monthly mean temperatures ranging from − 21.6 °C in January to 19 °C in July. The average annual precipitation is 337 mm, distributed unevenly across seasons, falling mainly during May–August which coincides with the plant growing season. The annual potential evapotranspiration ranges from 1600 to 1800 mm. The dominant plant species in the study area is Leymus chinensis with some Stipa grandis and Cleistogenes squarrosa. The soil type is dark chestnut (calcic Chernozem according to the ISSS Working Group RB, 1998) (Bai et al. 2010).
Four different stocking rates were established in 2005, of zero, three, six, and nine sheep per hectare. The four stocking rates were classified as no grazing (SR0), light grazing (SR3), moderate grazing (SR6), and heavy grazing (SR9). Simultaneously, the four stocking rates were set to simulate the development of livestock intensity in this region from 1950 to recent years (Tong et al. 2003; Ren et al. 2015). These treatments were arranged in the field in a randomized block design with three replicate plots. We separated each plot into three parts, and collected from the upper 10-cm layer from three random locations within each part using a 5-cm-diameter soil auger in August 2015. So nine samples were collected from each plot, and the nine samples from each plot were bulked into a single composite sample, packed with ice packs, and transported to the laboratory. Soil samples were then passed through a 2-mm sieve and immediately used for incubation experiment. Subsamples were air-dried for physiochemical analysis.
Soil pH was measured with a soil to water ratio of 1:2.5 using a pH meter (Mettler Toledo, Switzerland); gravimetric soil moisture content was analyzed by oven-drying at 105 ± 2 °C for 24 h. Soil bulk density was measured by the volumetric ring method according to Lampurlanés and Cantero-Martinez (2003). Soil organic matter (SOM) was determined by dichromate digestion (Kalembasa and Jenkinson 1973). Total C (TC) and N (TN) were determined by dry combustion in a Vario Max CNS analyzer (Elementar Instruments, Mt. Laurel, NJ). Olsen P was extracted by 0.5 M NaHCO3 (pH = 8.5) and determined with the molybdenum blue method (Olsen 1954). Available potassium (AK) was extracted by 1 M ammonium acetate and determined by flame emission spectrophotometry. NO3 − and exchangeable NH4 + were extracted with 1 M KCl and determined by a flow injection analyzer (SAN++, Skalar, Holland).
Construction and sampling of soil microcosms
Soil microcosms were constructed as described by Wang et al. (2015). Briefly, 10 g of fresh soil was pre-incubated in a 120-mL serum bottle for 20 days at 25 °C in darkness at 40% maximum water-holding capacity before the incubation experiment. The purpose of the pre-incubation was to stabilize the background respiration of soil, so that dilution of 13CO2 by soil-respired 12CO2 would be reduced and maintained at a constant level over the entire incubation period as reported previously (Jia and Conrad 2009; Wang et al. 2015). Assuming an effective soil depth of 20 cm, and that the grazing event lasts from mid-June to mid-September for 3 months (He et al. 2011), the amount of N amended to soils was nearly equivalent to the amount reported by Whitehead (1986), who estimated that as much as 300 kg N ha−1 year−1 returned to grassland soil in the form of livestock urine. Therefore, after pre-incubation, approximately 100 μg urea-N g−1 dry weight soil (d.w.s) was added once a week for 8 weeks through dropwise addition of freshly made urea solution to achieve 60% maximum water-holding capacity to establish a substrate-rich environment for nitrifying communities (Zhao et al. 2015). Microcosms were then flushed with synthetic air (20% O2, 80% N2) for 1 min to maintain oxic conditions. The bottles were then sealed with rubber stoppers and aluminum caps. 12CO2/13CO2/13CO2 + C2H2 (5% (vol/vol) CO2, 100 Pa C2H2) were injected into each microcosm through the rubber septum; 100 Pa C2H2 was used to inhibit ammonia oxidation. The 13CO2 and 13CO2 + C2H2 microcosms were amended with 100 mg 13C-urea-N kg−1 soil, respectively, whereas the 12CO2 treatments received 100 mg 12C-urea-N kg−1 soil. Application of urea and 5% of CO2 and/or 100 Pa C2H2 was renewed weekly. The 12C-urea, 13C-urea, 12CO2, and 13CO2 (99 atom%13C) were purchased from the Shanghai Engineering Research Center of Stable Isotopes (Shanghai, China). Soil samples at day 0 were collected immediately after the 20-day pre-incubation and urea addition for subsequent analysis. Destructive sampling was carried out at days 7, 14, 28, and 56. About 2 g of soil sample from each triplicate microcosm was transferred immediately to a − 80 °C freezer for molecular analysis. NO3 − and NH4 + were extracted with 1 M KCl and determined by a flow injection analyzer (SAN++, Skalar, Holland).
Nucleic acid extraction and SIP fraction
DNA was extracted from 0.5 g (fresh weight) of soil with a FastDNA SPIN Kit for Soil (MP Biomedicals, LLC, Solon, OH, USA), in accordance with the manufacturer’s protocol. The DNA size and integrity were checked by electrophoresis on a 0.7% agarose gel, and the quantity and purity were estimated using a NanoDrop® ND-2000 UV-Vis Spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA). The extracted DNA was stored at − 20 °C until analysis.
SIP fractionation was carried out as described previously (Wang et al. 2015). Extracted DNA from 12C-, 13C-CO2, and 13C-CO2 + C2H2 incubations was subjected to isopycnic density gradient centrifugation to separate the 13C-labeled DNA from the 12C-labeled DNA in triplicate SIP microcosms. For each sample, ~ 3.0 μg of the DNA extract was mixed well with CsCl stock solution to achieve an initial CsCl buoyant density of 1.725 g mL−1, by adding small amounts of gradient buffer (GB) or CsCl solution. The isopycnic density gradient centrifugation was performed using a 5.1-mL Quick-Seal polyallomer ultracentrifugation tube in a Vti65.2 vertical rotor (Beckman Coulter Inc., Palo Alto, CA, USA) at 177,000 g for 44 h at 20 °C. CsCl density gradients displaced with sterile water from the top of the ultracentrifuge tube were fractionated into equal volumes using an NE-1000 single syringe pump (New Era Pump Systems Inc., Farmingdale, NY, USA) with a precisely controlled flow rate of 0.38 mL min−1. A total of 14 DNA gradient fractions were obtained with about 380 μL in each fraction, and 65 μL of each fraction was used for the refractive index measurement using an AR200 digital hand-held refractometer (Reichert Inc., Buffalo, NY, USA). The fractionated DNA was purified and dissolved in 30 μL of sterile water as described previously (Zhang et al. 2010; Wang et al. 2015).
Quantitative PCR (qPCR) of the amoA genes
qPCR assays targeting bacterial and archaeal amoA genes of soil DNA extracts and DNA gradient fractions were carried out in triplicate by qPCR with LightCycler 480 (Roche Applied Science). Primers used for all qPCR assays as well as details of cycling conditions used can be found in Table S1.
Each 20-μL reaction mixture contained 10 μL SYBR Premix Ex Taq (TaKaRa, Dalian, China), 1 μL of each primer and 1 μL of DNA template (1–10 ng), and 7.9 μL Milli-Q water. Melting curve analysis was performed at the end of each real-time PCR run to confirm PCR product specificity, by measuring fluorescence continuously with the temperature increasing from 50 to 99 °C. High efficiencies of 80.7–84.5% were obtained for the functional gene amplification, with R values ranging between 0.994 and 0.998.
Cloning, MiSeq, and phylogenetic analysis
The heavy fractions (fractions 3–7), obtained from isopycnic centrifugation, were amplified for cloning and sequencing. The cloning procedure was developed by Di et al. (2009). Briefly, each gene fragment was PCR-amplified. The details of primers and PCR conditions are shown in Table S1. Triplicate PCR amplicons were subsequently pooled and purified with a PCR cleanup kit (TransGen Biotech, Beijing, China) before being cloned into the pGEM-T Easy Vector (TransGen Biotech, Beijing, China). The resulting ligation mix was transformed in Escherichia coli JM109 competent cells (TransGen Biotech, Beijing, China) according to the manufacturer’s instructions. The correct inserts were checked by re-amplification of each target gene and positive ones (each containing 30 clones) were sequenced with an ABI PRISM® 3730 Genetic Analyzer (Applied Biosystems, Foster City, CA, USA), after cycle sequencing reactions using a BigDye® Terminator v3.1 Cycle Sequencing Kit (Applied Biosystems).
High-throughput sequencing of 16S rRNA genes encompassing the V4 region was used to assess the community shift of nitrifying phylotypes as usual (Lu et al. 2012; Wang et al. 2015). The MiSeq was carried out using the total DNA extracted from soil microcosms in the presence of 13CO2 at day 0 and day 56. In addition, MiSeq was conducted using the amplicons synthesized with a universal 515F-907R primer of the heavy fractions (fractions 3–7) of the labeled (day 56-13CO2) and control (day 56-12CO2 and day 56-13CO2 + C2H2) microcosms.
The sequences were clustered into operational taxonomic units (OTUs) at 97% cutoff using the Mothur software (Schloss et al. 2009), and the OTUs containing less than 10 reads were removed (Xia et al. 2011). A representative sequence was then used from each OTU of the 16S rRNA or amoA gene for phylogenetic analysis. Phylogenetic analysis of archaeal and bacterial amoA genes and 16S rRNA genes was then conducted by Molecular Evolutionary Genetic Analysis software (MEGA6.06) with 1000-fold bootstrap support. The DNA sequences used for phylogenetic tree construction were deposited in the DNA Data Bank of Japan (DDBJ) under the accession number LC179807–LC179818 (AOA) and LC179819–179831 (AOB) for the 13C-archaeal and bacterial amoA genes, respectively. The entire dataset of 16S rRNA gene reads was deposited in NCBI’s Sequence Read Archive (SRA) under accession number SRP089745.
Measurement of 13C
The 13C incorporation into soils were analyzed at day 0 in 12CO2 microcosms and day 56 in 13CO2 microcosms to investigate whether ammonia oxidizers assimilated 13C derived from 13CO2. Prior to analysis for soil 13C, about 20 mg of soil was freeze-dried and ground to a fine powder. The stable C isotope ratios were measured by an isotope ratio mass spectrometer (IsoPrime 100, ElementarAnalysensys-teme, Hanau, Germany).
Statistical analysis
All statistical analyses were performed using SPSS version 20 and one-way analysis of variance (ANOVA) followed by Duncan’s multiple range test to check for the differences in soil physiochemical properties and functional gene abundance between treatments. P < 0.05 was considered to be statistically significant. Redundancy analysis (RDA) was used to determine the relationship between community composition of microbes and environmental factors. VEGAN of R language was implemented to do the above two analyses. All the figures were prepared by Origin (OriginPro 9.0 for Windows).
Results
Soil physicochemical properties
Soil properties were affected by the different stocking rates of grazing (Table 1). Soil pH was significantly higher in the SR3 treatment and significantly lower in the SR6 and SR9 treatments compared with that in the SR0 treatment. Bulk densities were significantly higher in the SR6 and SR9 soils than those in the SR0 and SR3 soils. Soil SOM and TC contents were significantly higher in the SR3 treatment than under other stocking rates. Soil AK showed a rising trend with increasing stocking rates. Olsen P was significantly higher in the SR3 treatment than in the SR0 and SR9 treatments. Soil mineral N (exchangeable NH4 +-N and NO3 −-N) contents were significantly lower in the SR9 soil than in the SR0 soil.
Change of 13C amounts between day 0 and day 56 in soils
The 13C incorporation into the soils was analyzed at day 0 and day 56 to detect if 13C-CO2 was assimilated by autotrophic ammonia oxidizers (Fig. S1). The total 13C at day 0 were 1.08, 1.09, 1.11, and 1.08%, whereas those at day 56 were 1.34, 1.32, 1.24, and 1.28% in the SR0, SR3, SR6, and SR9 soils, respectively. We assumed that all added 13C derived from 13C-urea was maintained in the soils after incubation for 56 days, and the 13C value was 1.11% in the soils with 13C-urea amendment at day 56, significantly lower than the total 13C values at day 56.
Soil nitrification activity and mineral N
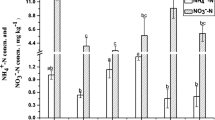
In microcosms with no acetylene added, NO3 − concentrations increased continuously, while exchangeable NH4 + concentrations increased slightly, in the different stocking rates throughout the 56-day incubation period (Fig. S2). Acetylene application completely inhibited nitrification, leading to the accumulation of exchangeable NH4 + while the NO3 − concentration remained stable throughout the incubation period (Fig. S2). There was no significant difference in nitrification rates between 13CO2-labeled and 12CO2 control microcosms at day 56 (Fig. 1a). However, the nitrification rate in the SR3 soil was 13.45 μg NO3 −-N g−1 soil per day, which was significantly higher than in the SR0 soil with a nitrification rate of 12.23 μg NO3 −-N g−1 soil per day. In contrast, it was 10.62 μg NO3 −-N g−1 soil per day in the SR9 soil, significantly lower than in the SR0 soil (Fig. 1a). A significant quadric polynomial regression (R 2 = 0.97, P < 0.001) was established between stocking rates and the nitrification activity (Fig. S3).
Changes in nitrification activity (a) and community composition of nitrifiers in soil microcosms over an incubation period of 56 days. The nitrification activity (a) was assessed by difference between soil NO3 −-N content on day 56 and that on day 0. The abundances of AOA (b) and AOB (c) amoA genes were determined by qPCR. The relative abundances of AOA (d), AOB (e), and NOB (f) were determined by high-throughput fingerprinting of 16S rRNA genes. The ratios were calculated by targeted 16S rRNA gene reads to the total microbial 16S rRNA gene reads in each microcosm. The error bars represent the standard errors of the mean of triplicate microcosms. The different letters above the columns indicate a significant difference (P < 0.05) based on the analysis of variance
Abundance and community composition of soil-nitrifying communities
Abundance of archaeal amoA genes increased significantly in the 12C/13C-CO2 microcosms after 56 days of incubation in the SR0 and SR6 soils, but no significant increase was observed in the SR3 and SR9 treatments (Fig. 1b). In contrast, bacterial amoA gene abundance increased significantly in the 12C/13C-CO2 microcosms in all four treatments after 56 days of incubation. Generally, the AOB amoA gene copy numbers increased from 1.12 × 106, 9.75 × 105, 5.77 × 106, and 2.02 × 106 g−1 soil at day 0 to 1.68 × 108, 1.03 × 108, 8.68 × 107, and 6.35 × 107 g−1 soil at day 56 in the SR0, SR3, SR6, and SR9 soils, representing 150-, 106-, 15-, and 31-fold increases, respectively (Fig. 1c). Acetylene application completely inhibited the growth of both AOA and AOB in the four treatments (Fig. 1b, c), suggesting chemolithoautotrophic lifestyles of active nitrifying communities (Zhao et al. 2015).
High-throughput fingerprinting of 16S rRNA genes at the whole community level was performed by MiSeq of the total 16S rRNA genes in SIP microcosms at day 0 and day 56 in the labeled microcosms (Fig. 1d–f). Approximately 896,860 high-quality sequence reads were obtained (Table S2). Relative abundance of bacterial 16S rRNA genes ranged from 88.6 to 99.4%, and this was much higher than that of archaeal 16S rRNA genes, which ranged from 0.6 to 11.4%. MiSeq data provided details of the relative abundance of targeted microbial 16S rRNA gene sequences in the total microbial community. The AOA relative abundance increased from 1.4, 1.3, and 1.0% at day 0 to 5.0, 2.3, and 3.7% at day 56 in the SR0, SR3 and SR9 soils, respectively (Fig. 1d). No significant changes were found in AOA relative abundance in the SR6 soil. The 56-day incubation resulted in a 7.4-, 4.9-, 2.7-, and 2.6-fold increase in AOB relative abundance in the SR0, SR3, SR6, and SR9 soils, respectively (Fig. 1e). Similar results were observed for NOB in the SR0 and SR3 soils (Fig. 1f). The relative abundance of NOB 16S rRNA gene sequences in the total microbial community increased from 0.5 and 0.6% at day 0 to 1.1 and 1.0% at day 56 in the SR0 and SR3 soils, respectively. In the SR6 and SR9 soils, however, NOB showed no significant differences after 56 days of incubation.
The 16S rRNA genes affiliated with AOA, AOB, and NOB were selected for phylogenetic analysis from the total MiSeq reads in labeled microcosms at day 0 and day 56 (Figs. S4 and S5). All the archaeal 16S rRNA genes were affiliated with soil group І.1b lineage, containing seven distinct OTUs within six clusters: Nitrososphaera viennensis cluster, N. viennensis-associated cluster, 29i4 cluster, 29i4-associated cluster, 54d9 cluster, and 54d9-associated cluster (Fig. S4). With respect to AOB, the bacterial 16S rRNA genes were classified into eight OTUs, three of which were unclassified (Fig. S5). The other five OTUs fell into four clusters: Nitrosomonas oligotropha, Nitrosomonas marina, Nitrosospira cluster 3, and Nitrosococcus watsonii. As for NOB, eight distinct OTUs were selected and phylogenetically assigned to Nitrospira marina, Nitrospira defluvii, Nitrospira moscoviensis, and Nitrobacter vulgaris, while two OTUs were unclassified (Fig. S6).
SIP of active nitrifying communities
Isopycnic gradient centrifugation was performed on the total DNA from each treatment to determine which putative autotrophic ammonia oxidizers were labeled by 13C-CO2 during the active nitrification in four soils after 56 days of incubation. The ultracentrifugation led to 14 fractions, and the buoyant density ranged from 1.701 to 1.764 g mL−1 from the top to the bottom of the tube. The abundances of archaeal and bacterial amoA genes in the 14 fractions were determined by qPCR (Fig. 2). The distribution of archaeal amoA gene abundance in microcosms with 12CO2 and 13CO2 was maximal in lighter fractions (1.70 to 1.74 g mL−1), except a slight shift in the buoyant density (1.74 to 1.76 g mL−1) under the SR3 treatment with 13CO2 (Fig. 2a). By comparison, the abundances of bacterial amoA genes in all 13C-spiked microcosms peaked in heavy fractions (1.74 to 1.76 g mL−1), while those in the presence of 12CO2 were observed in fractions with buoyant densities of 1.70 to 1.74 g mL−1 (Fig. 2b). In 13CO2 + C2H2 microcosms, the relative abundances of archaeal and bacterial amoA genes in the heavy fractions were close to the limit of detection (Fig. 2). MiSeq of total 16S rRNA genes in the fractionated DNA showed significant enrichment of target nitrifying communities in the heavy fractions from labeled microcosms compared with the control microcosms (Table 2). Rarefaction curves of total DNA (Fig. S7a) and fractionated DNA (Fig. S7b) were built to certify the coverage of soil microbial diversity. The number of unique operational taxonomic units (sharing ≥ 97% sequence identity) per total reads for each sample was shown by the rarefaction curves. The cumulative number of species reached an asymptote (Fig. S7). AOA, AOB, and NOB were enriched by up to 69.0, 63.7, and 32.2%, respectively.
Quantitative distribution of the archaeal (a) and bacterial (b) amoA genes across the entire buoyant density gradient of the fractionated DNA from soil microcosms incubated with 12CO2 and 13CO2 in the presence or absence of C2H2 after incubation for 56 days. The normalized data are the ratios of the gene copy numbers in each DNA gradient fraction to the sum of the amoA genes across the entire gradient of DNA fractions for each treatment. The error bars represent the standard errors of the mean of triplicate microcosms
Phylogenetic analysis of active ammonia oxidizers
Phylogenetic analysis of the labeled archaeal 16S rRNA gene sequence reads as well as clone library construction of 13C-labeled archaeal amoA genes showed that the active archaeal amoA genes retrieved from the labeled microcosms fell exclusively within group І.1b (Fig. 3a, b). The 13C-labeled 16S rRNA gene revealed that the 29i4 cluster dominated active archaeal amoA genes in the SR0, SR6, and SR9 soils, accounting for up to 82.9, 62.3, and 86.1% of active AOA populations in the SR0, SR6, and SR9 soils, respectively. Sequences affiliated with the N. viennensis cluster comprised 52.7% of the active archaeal community in the SR3 soil (Fig. 3a). Clone library construction of 13C-labeled archaeal amoA genes (Fig. 3b) further demonstrated that N. viennensis cluster encompassed 86% of the active archaeal genes in the SR3 soil. No sequences in archaeal clone library were affiliated with the 29i4-associated cluster.
Phylogenetic analysis of the 16S rRNA (a) and amoA genes (b) of AOA in 13C-labeled DNA from the labeled microcosms after an incubation period of 56 days. The designation “HF” indicates the 13C-DNA in the active fraction after the ultracentrifugation of the total DNA extract from the labeled microcosms. The designation “SR0-AOA-16SrRNA-HF-OTU-1-731(5.5%)” indicates that OTU-1 contains 731 reads with > 97% sequence similarity, accounting for 5.5% of the total archaeal AOA 16S rRNA gene reads in the 13C-DNA from the SR0 soil microcosms. The designation “SR0-AOA-amoA-clone-HF-OTU-1-16(84%)” indicates that OTU-1 contains 16 sequences with > 97% sequence similarity, accounting for 84% of the total archaeal AOA amoA gene clones in the 13C-DNA from the SR0 soil microcosms. The scale bars represent 0.5 and 2% nucleic acid sequences divergence for the 16S rRNA and amoA genes, respectively
With respect to active AOB (Fig. 4), up to 89.0, 80.4, 91.4, and 93.5% of labeled bacterial 16S rRNA gene sequences were closely related to the Nitrosospira cluster 3 lineage in the SR0, SR3, SR6, and SR9 soils, respectively (Fig. 4a). About 11.0% of bacterial amoA genes in the heavy fractions fell into N. oligotropha in the SR0 soil, and 11.2% of active AOB populations were grouped into N. marina in the SR3 soil. Interestingly, clone library construction of bacterial amoA gene revealed that all the active AOB populations were clustered with Nitrosospira (Fig. 4b). Nitrosospira cluster 3 dominated the active bacterial amoA genes in the SR3, SR6, and SR9 soils, accounting for 63, 100, and 70%, respectively.
Phylogenetic analysis of the 16S rRNA genes (a) and amoA (b) of AOB in 13C-labeled DNA from the 13CO2-treated microcosms after an incubation period of 56 days. The designation “HF” indicates the 13C-DNA in the active fraction after the ultracentrifugation of the total DNA extract from the labeled microcosms. The designation “SR3-AOB-16SrRNA-HF-OTU-1-79(1.2%)” indicates that OTU-1 contains 79 reads with > 97% sequence similarity, accounting for 1.2% of the total bacterial AOB 16SrRNA gene reads in the 13C-DNA from the SR3 soil microcosms. The designation “SR3-AOB-amoA-clone-HF-OTU-1-3(19%)” indicates that OTU-1 contains 3 sequences with > 97% sequence similarity, accounting for 19% of the total archaeal AOB amoA gene clones in the 13C-DNA from the SR3 soil microcosms. The scale bars represent 1 and 2% nucleic acid sequences divergence for the 16S rRNA and amoA genes, respectively
The communities of NOB (Fig. 5) were members within N. defluvii, accounting for 92.1, 75.0, 69.5, and 65.6% of the 13C-labeled 16S rRNA genes in the SR0, SR3, SR6, and SR9 soils, respectively. Furthermore, about 6.3, 21.8, 15.2, and 24.8% of active NOB were classified into N. marina lineage in the SR0, SR3, SR6, and SR9 soils, respectively.
Phylogenetic analysis of the 16S rRNA genes of NOB in the 13C-labeled DNA from 13CO2-treated microcosms after incubation for 56 days. The designation “SR0-NOB-16SrRNA-HF-OTU-1-147 (2.5%)” indicates that OTU-1 contains 147 reads with > 97% sequence similarity, accounting for 2.5% of the total NOB 16S rRNA gene reads in the 13C-DNA from the SR0 soil microcosms. Bootstrap values higher than 50% are indicated at the branch nodes. The scale bars represent 5% nucleic acid sequence divergence in the 16S rRNA genes
We also analyzed the proportional changes of active nitrifying phylotypes in soil microcosms to assess the effects of stocking rates on nitrifier activity (Fig. S8). Stocking rates significantly changed the proportions of active AOA, AOB, and NOB phylotypes (Fig. S8). As for AOA (Fig. S8a), the 29i4 clusters dominated the active AOA in the SR0, SR6, and SR9 soils. The proportion of N. viennensis clusters significantly increased, while the proportion of 29i4 clusters decreased in the SR3 soil. With respect to AOB (Fig. S8b), the majority of active AOB were placed within cluster 3 in the four soils. The proportion of Nitrosomonas increased with increasing stocking rates. Similar results were observed for NOB (Fig. S8c); N. defluvii made up a much higher proportion of active NOB than other species.
Redundancy analysis (RDA) between nitrifier composition and soil properties
Mantel test was performed to analyze the major environmental factors responsible in shaping active nitrifying community structure, and RDA was used to identify the major environmental variables controlling the active nitrifying community structure. The results of Mantel tests and RDA analyses of all measured physicochemical parameters showed that SOM, bulk density, pH, exchangeable NH4 +-N, and NO3 −-N in combination yielded the best model explaining the highest percentage of variance in active nitrifying communities (Fig. 6). The first RDA axis was represented by SOM (r = − 0.83, P < 0.01), bulk density (r = 0.57, P < 0.05), pH (r = −0.59, P < 0.05), and exchangeable NH4 +-N contents (r = − 0.41, P < 0.05) and could explain 56.5% of the variance in the active nitrifying phylotypes under in situ conditions. The second RDA axis was represented by NO3 −-N contents (r = − 0.47, P < 0.05) and explained 21.4% of the variance.
Redundancy analysis (RDA) between nitrifiers and physiochemical characteristics of the grassland soils tested. The phylotypes of AOA, AOB, and NOB were pooled from the 16S rRNA genes in the 13C-labeled DNA from 13CO2-treated microcosms after incubation for 56 days. A1–A5 represent the OTU1–OTU5 of active AOA amoA genes, B1–B5 represent the OTU1–OTU5 of active AOB amoA genes, and N1–N8 represent the OTU1–OTU8 of active NOB. Single asterisk and double asterisk mark significance at P < 0.05 and 0.01, respectively, based on 999 Monte Carlo permutations
Discussion
Nitrogen is the limiting factor which regulates net plant primary production in many ecosystems, including grassland (Vitousek and Howarth 1991), and nitrification is an important N-transformation process that can affect N availability and has environmental consequences (Di and Cameron 2016). This study, therefore, focused on the effects of gradient grazing on nitrification activity and the active nitrifying community, and suggested that stocking rates had a major impact on nitrification activity and phylogenetically distinct active nitrifiers. To our knowledge, this study is the first to distinguish nitrification activity and active nitrifiers under different stocking rates in grassland soils. An improved understanding of the relationships between different stocking rates and nitrification activity and active nitrifiers is critically important for providing deeper insights into N cycling in grazed grassland soils.
According to Di and Cameron (2002), about 70–90% of the N ingested by the grazing animal is returned to the grassland soils in animal excreta, particularly in urine. Most of the N in the urine is released as ammonium in the soil which is then subjected to nitrification. In addition, different stocking rates may also lead to differential soil physical conditions (e.g., bulk density and aeration status) (Li et al. 2008; Steffens et al. 2008) and soil chemical fertility (e.g., pH and organic matter content) (Li et al. 2008; Steffens et al. 2008). Soil bulk densities significantly increased under the SR6 and SR9 treatments compared with those under the SR0 and SR3 treatments, because of animals trampling the soil. However, compared with a significant decline in TN concentration with increasing grazing intensity in a previous report (Han et al. 2008), the TN content was not affected directly by 10 years of gradient grazing in our study, while NO3 −-N and exchangeable NH4 +-N contents decreased significantly in the SR9 soil (Table 1). After incubation for 56 days, the NO3 −-N contents in the SR9 soils remained significantly lower than those in soils under other stocking rates (Fig. S2a-b), suggesting that external 100 μg urea-N g−1 d.w.s application did not affect the indigenous nitrification in the present study. Besides, the highest accumulation of exchangeable NH4 +-N contents after incubation further proved that nitrification rates in the SR9 soil were significantly lower than soils under the other three stocking rates (Fig. S2d-f). Soil TC and SOM contents were significantly higher in the lightly grazed (SR3) soil than the un-grazed (SR0) and heavier grazed (SR6 and SR9) soils. This was because light-to-moderate levels of grazing could lead to a greater diversity of plant species, which would have dense fibrous rooting systems benefiting soil organic matter formation and soil C sequestration (Reeder et al. 2001). Heavy grazing, on the other hand, would decrease plant species diversity (Reeder and Schuman 2002) and accelerate soil erosion because of the severe grazing and trampling actions of the grazing animals, resulting in a loss of soil organic matter (Soane 1990). Olsen P was generally higher in the lightly (SR3) grazed soil compared with un-grazed and heavier grazed soils. According to Frizano et al. (2002), soil organic matter is important for maintaining the labile P pools.
The total 13C atom % enrichments in the soils with 13CO2 and 13C-urea at day 56 were much higher than those at the start of incubation (day 0). As some nitrifiers might have assimilated urea-derived carbon (Li et al. 2014), we assumed that all 13C-carbon derived from 13C-urea was maintained in the soils throughout the 56-day incubation period. However, the 13C values calculated of soils with 13C-urea amendment was still much lower than those with 13CO2 and 13C-urea at day 56. These strongly demonstrated the assimilation of 13CO2 by autotrophic nitrifiers.
The quantitative analysis after fractionation showed that AOB were labeled in the four soils in the 13C-CO2 microcosms, and AOA were labeled solely in the SR3 soils (Fig. 2). Surprisingly, the percentage of AOA reads to the total 16S rRNA gene reads at the whole microbial community accounted for up to 50.5, 25.9, 27.1, and 37.1% in the heavy fractions in the SR0, SR3, SR6, and SR9 soils, respectively (Table 2), which suggested the autotrophic growth of AOA in the grazed soils. This discrepancy resulted from one OTU which was grouped into the soil fosmid 29i4 cluster (Fig. 3, Fig. S4). It is possible that the qPCR failed to amplify the archaeal amoA genes grouped with the soil fosmid 29i4 cluster, because of possible biases of the PCR primers (Alves et al. 2013). The fact that both AOA and AOB were labeled would suggest the importance of both AOA and AOB in the nitrification processes in the grassland soil. Nevertheless, the ratios of labeled cells of AOA/AOB may be used as a parameter to indicate the relative importance of AOA and AOB during active soil nitrification (Hai et al. 2009; Trias et al. 2012; Wang et al. 2015). In this study, the labeled AOA to AOB ratios were 0.12, 1.15, 0.21, and 0.24 in the SR0, SR3, SR6, and SR9 soils, respectively (Table S3). The sequencing summary of the total 16S rRNA genes in the fractionated DNA isolated from each treatment after 56-day incubation further showed the higher AOA/AOB 16S rRNA gene ratio for the SR3 treatment, while lower ratios were found for the SR6 and SR9 soils compared with the SR0 soil (Table 2). These results suggested the importance of AOA in nitrification in the light grazing soil but AOB in nitrification in the heavy grazing soils.
The phylogenetic analysis of archaeal 16S rRNA (Fig. 3a) showed that the fosmid 29i4 cluster dominated the active archaeal amoA genes in the SR0, SR6, and SR9 soils, whereas up to 52.7% of active AOA was grouped within the N. viennensis cluster in the SR3 soil (Fig. 3a). The N. viennensis lineage has been shown to be well adapted to low-temperature arctic soils (Alves et al. 2013) and microaerophilic environments (Wang et al. 2015). Results from this study indicate that they also play an important role in ammonia oxidation in lightly grazed alkaline grassland soils. Tourna et al. (2011) proposed that N. viennensis as an ammonia-oxidizing archaeon from soil, using ammonia or urea as an energy source, might be also be adapted to mixotrophic growth. The SOM in the SR3 soil was the highest of the four different stocking rates, up to 30.42 g kg−1, and this might have been responsible for the high proportion of active N. viennensis lineage of AOA in the SR3 treatment. As for AOB, the phylogenetic analysis of 16S rRNA genes revealed that more than 80% of active AOB fell within Nitrosospira cluster 3 in the four treatments (Fig. 4a). Predominance of Nitrosospira cluster 3 in the AOB community, as observed in the present study, was also observed in the studies targeting Chinese agricultural soil (Xia et al. 2011), grassland soils (Di et al. 2009; Pan et al. 2016), and acidic upland soils (Wang et al. 2015). Some studies suggested that members of cluster 3 are usually dominant at relatively high ammonium concentrations (Kowalchuk et al. 2000). Other studies, however, have shown the presence of this cluster in both low and high N environments (Webster et al. 2002; Avrahami et al. 2003; Chu et al. 2007). The present study, however, shows that the predominant role of Nitrosospira cluster 3 of AOB was not affected by the different stocking rates. In addition, up to 11.0 and 18.5% of active AOB were phylogenetically related to the Nitrosomonas lineage in the SR0 and SR3 treatments. This might have been because the O2 availability decreased significantly with increasing grazing intensity as animal tramping significantly increased soil bulk density (Table 1), while Nitrosomonas were dominant in oxic or micro-oxic soils (Wang et al. 2009). It is worth mentioning that nitrification activity was significantly increased by light grazing (SR3), while it was dramatically decreased by heavy grazing (SR9) (Fig. 1a). Enhanced nitrification in response to grazing has been reported in many other grassland soils, such as Yellowstone grasslands (Frank et al. 2000) and seminatural grasslands (Le Roux et al. 2003; Patra et al. 2005). Patra et al. (2005) attributed the enhancement of microbial driven N processes by grazing per se to the increased cell numbers and the changes in community composition. The nitrification activity in the SR9 soil was significantly related to the bacterial amoA gene abundance (r = 0.877, p < 0.05) and relative abundance of 16S rRNA genes of AOB in total reads (r = 0.943, P < 0.05), but not to those of archaea. These results strongly suggested that heavy grazing (SR9) significantly decreased the nitrification activity resulting from reducing the abundance of active AOB, which further indicated a slow N cycling rate in heavier grazing than lighter grazing soils.
Nitrospira and Nitrobacter are regarded as two main participants of nitrite-oxidizing bacteria (NOB) in nitrification in terrestrial ecosystems. Phylogenetic analysis in this study indicated that Nitrospira-like sequences rather than Nitrobacter-like ones were predominant of the active NOB in the four soils (Fig. 5). Other studies have reported similar results in agricultural soils (Xia et al. 2011), and paddy soils (Zhao et al. 2015; Wang et al. 2015). It was suggested that Nitrospira-like NOB preferred nutrient-limiting environments, whereas Nitrobacter-like NOB positively responded to increased N and oxygen levels (Schramm et al. 1999). It is, therefore, not surprising that Nitrospira-like NOB were found to be dominant in this extensively grazed low fertility grassland soil. Nitrospira defluvii are mesophilic aerobic chemolithoautotrophic nitrite oxidizers, which can assimilate pyruvate and have a strong tendency to aggregate (Spieck et al. 2006). In this study, the proportions of N. defluvii were decreased with increasing stocking rates (92.1, 75.0, 69.5, and 65.6% in the SR0, SR3, SR6, and SR9 treatments, respectively). We speculate that N. defluvii tend to aggregate in high-fertility and aerated environments as soil aeration and fertility are significantly decreased with increasing stocking rates (Steffens et al. 2008). In contrast, in our work, the lowest relative abundance of the N. marina-like bacteria accounted for only up to 6.3% in the SR0 soil, but up to 21.8, 15.2, and 24.8% in the SR3, SR6, and SR9 soils, respectively. This community might have benefited from the complex C and N sources in low concentration in grazed soils (Watson et al. 1986). In addition, N. moscoviensis were exclusively found in the SR6 and SR9 soils. This may be related to the optimal pH of this cluster which is 7.6–8.0, and the cells of this cluster were inhibited by high organic matter concentrations (Ehrich et al. 1995). The pH and organic matter content in the SR0 and SR3 soils were much higher than the optimal ranges for this cluster (Table 1).
Stocking rates significantly changed the proportions of active AOA, AOB, and NOB phylotypes (Fig. S8). Redundancy analysis (RDA) showed that soil SOM, bulk density, pH, exchangeable NH4 +-N, and NO3 −-N were the key factors in shaping the phylogenetic distribution of active nitrifying communities (Fig. 6). Therefore, different stocking rates may have affected the distribution of active nitrifying communities by affecting the soil physicochemical characteristics, such as SOM, bulk density, pH, exchangeable NH4 +-N, and NO3 −-N contents. It is also worth mentioning that the incubation period lasted just for 56 days, and it would be a little bit short to cover the annual change. Accordingly, an experiment which lasted for a longer time would be considered in the near future to understand N cycling and control nitrification in grazed grassland. Moreover, as a major disturbance associated with grazing, compaction would lead to alterations in soil porosity, which would affect the air permeability (Ball et al. 2012) and thus affect nitrifier activities. Therefore, further study taking the factor into consideration during incubation study is required.
Conclusions
Results from this study indicate that stocking rates significantly alter the distributions of the active nitrifying communities in the grazed grassland soil by impacting soil physicochemical characteristics (e.g., SOM, bulk density, pH, exchangeable NH4 +-N, and NO3 −-N). The dominant player in ammonia oxidation was the N. viennensis lineage of AOA in the lightly grazed soil, but it was the Nitrosospira cluster 3 of AOB in the heavily grazed soil. The stocking rates significantly shifted the nitrification activity and distribution of active nitrifying communities. The stocking rate of SR3 significantly enriched the N. viennensis lineage of AOA and the Nitrosomonas lineage of AOB and significantly increased the nitrification activity, while heavy grazing (SR9) significantly decreased the abundance of active AOB and resulted in the lowest nitrification activity. Nitrospira defluvii of NOB was a dominant player in nitrite oxidation, and grazing increased the proportions of N. marina within functional active NOB. These results provide new insights into our understanding of the N cycle in grazed grassland soils.
References
Avrahami S, Liesack W, Conrad R (2003) Effects of temperature and fertilizer on activity and community structure of soil ammonia oxidizers. Environ Microbiol 5:691–705
Alawi M, Lipski A, Sanders T, Spieck E (2007) Cultivation of a novel cold-adapted nitrite oxidizing beta proteobacterium from the Siberian Arctic. ISME J 1:256–264
Attard E, Poly F, Commeaux C, Laurent F, Terada A, Smets BF, Recous S, Roux XL (2010) Shifts between Nitrospira-and Nitrobacter-like nitrite oxidizers underlie the response of soil potential nitrite oxidation to changes in tillage practices. Environ Microbiol 12:315–326
Alves RJE, Wanek W, Zappe A, Richter A, Svenning MM, Schleper C, Urich T (2013) Nitrification rates in Arctic soils are associated with functionally distinct populations of ammonia-oxidizing archaea. ISME J 7:1620–1631
Bartosch S, Hartwig C, Spieck E, Bock E (2002) Immunological detection of Nitrospira-like bacteria in various soils. Microbial Ecol 43:26–33
Bai Y, Wu J, Clark CM, Naeem S, Pan Q, Huang J, Zhang L, Han X (2010) Tradeoffs and thresholds in the effects of nitrogen addition on biodiversity and ecosystem functioning: evidence from inner Mongolia grasslands. Glob Chang Biol 16:358–372
Ball BC, Cameron KC, Di HJ, Moore S (2012) Effects of trampling of a wet dairy pasture soil on soil porosity and on mitigation of nitrous oxide emissions by a nitrification inhibitor, dicyandiamide. Soil Use Manage 28:194–201
Cebron A, Garnier J (2005) Nitrobacter and Nitrospira genera as representatives of nitrite-oxidizing bacteria: detection, quantification and growth along the lower Seine River (France). Water Res 39:4979–4992
Chu H, Fujii T, Morimoto S, Lin X, Yagi K, Hu J, Zhang J (2007) Community structure of ammonia-oxidizing bacteria under long-term application of mineral fertilizer and organic manure in a sandy loam soil. Appl Environ Microb 73:485–491
Conrad R (1996) Soil microorganisms as controllers of atmospheric trace gases (H2, CO, CH4, OCS, N2O, and NO). Microbiol Rev 60:609–640
Di HJ, Cameron KC (2002) Nitrate leaching in temperate agroecosystems: sources, factors and mitigating strategies. Nutr Cycl Agroecosys 64:237–256
Di HJ, Cameron KC, Shen JP, Winefield CS, O’Callaghan M, Bowatte S, He JZ (2009) Nitrification driven by bacteria and not archaea in nitrogen-rich grassland soils. Nat Geosci 2:621–624
Daims H, Lücker S, Le Paslier D, Wagner M (2011) Diversity, environmental genomics, and ecophysiology of nitrite-oxidizing bacteria[M]//nitrification. Am Soc Microbiol 2011:295–322
Di HJ, Cameron KC, Podolyan A, Robinson A (2014) Effect of soil moisture status and a nitrification inhibitor, dicyandiamide, on ammonia oxidizer and denitrifier growth and nitrous oxide emissions in a grassland soil. Soil Biol Biochem 73:59–68
Di HJ, Cameron KC (2016) Inhibition of nitrification to mitigate nitrate leaching and nitrous oxide emissions in grazed grassland: a review. J Soils Sediments 16:1401–1420
Ehrich S, Behrens D, Lebedeva E, Ludwig W, Bock E (1995) A new obligately chemolithoautotrophic, nitrite-oxidizing bacterium, Nitrospira moscoviensis sp. nov. and its phylogenetic relationship. Arch Microbiol 164:16–23
Frank DA, Groffman PM, Evans RD, Tracy BF (2000) Ungulate stimulation of nitrogen cycling and retention in Yellowstone Park grasslands. Oecologia 123:116–121
Frizano J, Johnson AH, Vann DR, Scatena FN (2002) Soil phosphorus fractionation during forest development on landslide scars in the Luquillo Mountains, Puerto Rico. Biotropica 34:17–26
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai Z, Freney JR, Martinelli LA, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Sci 320:889–892
He NP, Zhang YH, Yu Q, Chen QS, Pan QM, Zhang GM, Han XG (2011) Grazing intensity impacts soil carbon and nitrogen storage of continental steppe. Ecosphere 2:1–10
Han G, Hao X, Zhao M, Wang M, Ellert BH, Willms W, Wang M (2008) Effect of grazing intensity on carbon and nitrogen in soil and vegetation in a meadow steppe in Inner Mongolia. Agric Ecosyst Environ 125:21–32
Hai B, Diallo NH, Sall S, Haesler F, Schauss K, Bonzi M, Assigbetse K, Chotte J, Munch JC, Schloter M (2009) Quantification of key genes steering the microbial nitrogen cycle in the rhizosphere of sorghum cultivars in tropical agroecosystems. Appl Environ Microb 75:4993–5000
Jia Z, Conrad R (2009) Bacteria rather than Archaea dominate microbial ammonia oxidation in an agricultural soil. Environ Microbiol 11:1658–1671
Jiang L, Wang S, Pang Z, Wang C, Kardol P, Zhou X, Rui Y, Lan Z, Wang Y, Xu X (2016) Grazing modifies inorganic and organic nitrogen uptake by coexisting plant species in alpine grassland. Biol Fert Soils 52:211–221
Kalembasa SJ, Jenkinson DS (1973) A comparative study of titrimetric and gravimetric methods for the determination of organic carbon in soil. J Sci Food Agr 24:1085–1090
Kowalchuk GA, Stienstra AW, Stephen JR, Woldendorp JW (2000) Changes in the community structure of ammonia-oxidizing bacteria during secondary succession of calcareous grasslands. Environ Microbiolenviron 2:99–110
Könneke M, Bernhard AE, José R, Walker CB, Waterbury JB, Stahl DA (2005) Isolation of an autotrophic ammonia-oxidizing marine archaeon. Nat 437:543–546
Lampurlanés J, Cantero-Martinez C (2003) Soil bulk density and penetration resistance under different tillage and crop management systems and their relationship with barley root growth. Agron J 95:526–536
Le Roux X, Bardy M, Loiseau P, Louault F (2003) Stimulation of soil nitrification and denitrification by grazing in grasslands: do changes in plant species composition matter? Oecologia 137:417–425
Lal R (2004) Soil carbon sequestration impacts on global climate change and food security. Sci 304:1623–1627
Le Roux X, Poly F, Currey P, Commeaux C, Hai B, Nicol GW, Prosser JM, Schloter M, Attard E, Klumpp K (2008) Effects of aboveground grazing on coupling among nitrifier activity, abundance and community structure. ISME J 2:221–232
Li C, Hao X, Zhao M, Han G, Willms WD (2008) Influence of historic sheep grazing on vegetation and soil properties of a Desert Steppe in Inner Mongolia. Agric Ecosyst Environ 128:109–116
Lu L, Han W, Zhang J, Wu Y, Wang B, Lin X, Zhu J, Cai Z, Jia Z (2012) Nitrification of archaeal ammonia oxidizers in acid soils is supported by hydrolysis of urea. ISME JT 6:1978–1984
Li Y, Watanabe T, Murase J, Asakawa S, Kimura M (2014) Abundance and composition of ammonia oxidizers in response to degradation of root cap cells of rice in soil microcosms. J Soils Sediments 14:1587–1598
Morimoto S, Hayatsu M, Takada Hoshino Y, Nagaoka K, Yamazaki M, Karasawa T, Takenaka M, Akiyama H (2011) Quantitative analyses of ammonia-oxidizing archaea (AOA) and ammonia-oxidizing bacteria (AOB) in fields with different soil types. Microbes Environ 26:248–253
McSherry ME, Ritchie ME (2013) Effects of grazing on grassland soil carbon: a global review. Glob Chang Biol 19:1347–1357
Nan Z (2005) The grassland farming system and sustainable agricultural development in China. Grassl Sci 51:15–19
Noll M, Matthies D, Frenzel P, Derakshani M, Liesack W (2005) Succession of bacterial community structure and diversity in a paddy soil oxygen gradient. Environ Microbiol 7:382–395
Olsen SR (1954) Estimation of available phosphorus in soils by extraction with sodium bicarbonate. In: United States Department Of Agriculture; Washington
Patra AK, Abbadie L, Clays-Josserand A, Degrange V, Grayston SJ, Loiseau P, Louault F, Mahmood S, Nazaret S, Philippot L, Poly F, Prosser JI, Richaume A, Le Roux X (2005) Effects of grazing on microbial functional groups involved in soil N dynamics. Ecol Monogr 75:65–80
Patra AK, Abbadie L, Clays-Josserand A, Degrange V, Grayston SJ, Guillaumaud N, Loiseau P, Louault F, Mahmood S, Nazaret S, Philippot L, Poly F, Prosser JI, Le Roux X (2006) Effects of management regime and plant species on the enzyme activity and genetic structure of N-fixing, denitrifying and nitrifying bacterial communities in grassland soils. Environ Microbiol Environm 8:1005–1016
Pan H, Li Y, Guan X, Li J, Xu X, Liu J, Zhang Q, Xu J, Di H (2016) Management practices have a major impact on nitrifier and denitrifier communities in a semiarid grassland ecosystem. J Soils Sediments 16:896–908
Ryden JC, Ball PR, Garwood EA (1984) Nitrate leaching from grassland. Nat 311:50–53
Reeder JD, Franks CD, Milchunas DG (2001) Root biomass and microbial processes. In: Follett RF, Kimble JM, Lal R (eds) The potential of US grazing lands to sequester carbon and mitigate the greenhouse effect. Lewis Publishers, Boca Raton FL, pp 139–166
Reeder JD, Schuman GE (2002) Influence of livestock grazing on C sequestration in semi-arid mixed-grass and short-grass rangelands. Environ Pollut 116:457–463
Ren H, Han G, Ohm M, Schönbach P, Gierus M, Taube F (2015) Do sheep grazing patterns affect ecosystem functioning in steppe grassland ecosystems in Inner Mongolia? Agric Ecosyst Environ 213:1–10
Soane BD (1990) The role of organic matter in soil compactibility: a review of some practical aspects. Soil Till Res 16:179–201
Schramm A, de Beer D, van den Heuvel JC, Ottengraf S, Amann R (1999) Microscale distribution of populations and activities of Nitrosospira and Nitrospira spp. along a macroscale gradient in a nitrifying bioreactor: quantification by in situ hybridization and the use of microsensors. Appl Environ Microb 65:3690–3696
Spieck E, Hartwig C, McCormack I, Maixner F, Wagner M, Lipski A, Daims H (2006) Selective enrichment and molecular characterization of a previously uncultured Nitrospira-like bacterium from activated sludge. Environ Microbiol 8:405–415
Steffens M, Kölbl A, Totsche KU, Kögel-Knabner I (2008) Grazing effects on soil chemical and physical properties in a semiarid steppe of Inner Mongolia (PR China). Geoderma 143:63–72
Schloss PD, Westcott SL, Ryabin T, Hall JR, Hartmann M, Hollister EB, Lesniewski RA, Oakley BB, Parks DH, Robinson CJ, Sahl JW, Stres B, Thallinger GG, Van Horn DJ, Weber CF (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microb 75:7537–7541
Sorokin DY, Lücker S, Vejmelkova D, Kostrikina NA, Kleerebezem R, Rijpstra WIC, Damsté JSS, Paslier DL, Muyzer G, Wagner M, van Loosdrecht MC, Daims H (2012) Nitrification expanded: discovery, physiology and genomics of a nitrite-oxidizing bacterium from the phylum Chloroflexi. ISME J 6:2245–2256
Szukics U, Hackl E, Zechmeister-Boltenstern S, Sessitsch A (2012) Rapid and dissimilar response of ammonia oxidizing archaea and bacteria to nitrogen and water amendment in two temperate forest soils. Microbiol Res 167:103–109
Sterngren AE, Hallin S, Bengtson P (2015) Archaeal ammonia oxidizers dominate in numbers, but bacteria drive gross nitrification in N-amended grassland soil. Front Microbiol 6
Tong C, Xi FJ, Yang JR (2003) Remote sensing monitoring on degraded steppe and determination of reasonable grazing intensity for the restoration of steppe in middle reach of Xilin river basin. Acta Prataculturae Sinica 12:78–83
Tourna M, Stieglmeier M, Spang A, Könneke M, Schintlmeister A, Urich T, Engel M, Schloter M, Wagner M, Richter A, Schleper C (2011) Nitrososphaera viennensis, an ammonia oxidizing archaeon from soil. P Natl Acad Sci USA Proc 108:8420–8425
Trias R, García-Lledó A, Sánchez N, López-Jurado JL, Hallin S, Bañeras L (2012) Abundance and composition of epiphytic bacterial and archaeal ammonia oxidizers of marine red and brown macroalgae. Appl Environ Microb 78:318–325
Vitousek PM, Howarth RW (1991) Nitrogen limitation on land and in the sea: how can it occur? Biogeochemistry 13:87–115
Watson SW, Bock E, Valois FW, Waterbury JB, Schlosser U (1986) Nitrospira marina gen. nov. sp. nov.: a chemolithotrophic nitrite-oxidizing bacterium. Arch Microbiol 144:1–7
Whitehead DC (1986) Sources and transformations of organic nitrogen in intensively managed grassland soils[M]//Nitrogen fluxes in intensive grassland systems. Springer, Dordrect, the Netherlands: 47-58
Webster G, Embley TM, Prosser JI (2002) Grassland management regimens reduce small-scale heterogeneity and species diversity of β-proteobacterial ammonia oxidizer populations. Appl Environ Microb 68:20–30
Wang Y, Ke X, Wu L, Lu Y (2009) Community composition of ammonia-oxidizing bacteria and archaea in rice field soil as affected by nitrogen fertilization. Syst Appl Microbiol 32:27–36
Wang B, Zhao J, Guo Z, Ma J, Xu H, Jia Z (2015) Differential contributions of ammonia oxidizers and nitrite oxidizers to nitrification in four paddy soils. ISME J 9:1062–1075
Wang Q, Zhang LM, Shen JP, Du S, Han LL, He JZ (2016) Nitrogen fertiliser-induced changes in N2O emissions are attributed more to ammonia-oxidising bacteria rather than archaea as revealed using 1-octyne and acetylene inhibitors in two arable soils. Biol Fert Soils 52:1163–1171
Xia W, Zhang C, Zeng X, Feng Y, Weng J, Lin X, Zhu J, Xiong Z, Xu J, Cai Z, Jia Z (2011) Autotrophic growth of nitrifying community in an agricultural soil. ISME J 5:1226–1236
Xie Z, Le Roux X, Wang C, Gu Z, An M, Nan H, Chen B, Li F, Liu Y, Du G, Feng H, Ma X (2014) Identifying response groups of soil nitrifiers and denitrifiers to grazing and associated soil environmental drivers in Tibetan alpine meadows. Soil Biol Biochem 77:89–99
Zhang LM, Offre PR, He JZ, Verhamme DT, Nicol GW, Prosser JI (2010) Autotrophic ammonia oxidation by soil thaumarchaea. P Natl Acad Sci USA 107:17240–17245
Zhong L, Du R, Ding K, Kang X, Li F, Bowatte S, Hoogendoorn C, Wang Y, Rui Y, Jiang LL, Wang S (2014) Effects of grazing on N2O production potential and abundance of nitrifying and denitrifying microbial communities in meadow-steppe grassland in northern China. Soil Biol Biochem 69:1–10
Zhao J, Wang B, Jia Z (2015) Phylogenetically distinct phylotypes modulate nitrification in a paddy soil. Appl Environ Microb 81:3218–3227
Zhong W, Bian B, Gao N, Min J, Shi W, Lin X, Shen W (2016) Nitrogen fertilization induced changes in ammonia oxidation are attributable mostly to bacteria rather than archaea in greenhouse-based high N input vegetable soil. Soil Biol Biochem 93:150–159
Zhou G, Zhou X, He Y, Shao J, Hu Z, Liu R et al (2017) Grazing intensity significantly affects belowground carbon and nitrogen cycling in grassland ecosystems: a meta-analysis. Glob Chang Biol 23:1167–1179
Acknowledgements
We thank the Inner Mongolia Grassland Ecosystem Research Station, Institute of Botany, the Chinese Academy of Sciences, for access to the soil sampling plots. This work was financially supported by the National Key Basic Research Program of China (2014CB138801) and Natural Science Foundation of China (41301254 and 41271272).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
ESM 1
(PPTX 1.26 MB)
Rights and permissions
About this article
Cite this article
Pan, H., Xie, K., Zhang, Q. et al. Archaea and bacteria respectively dominate nitrification in lightly and heavily grazed soil in a grassland system. Biol Fertil Soils 54, 41–54 (2018). https://doi.org/10.1007/s00374-017-1236-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00374-017-1236-7