Abstract
Background
Intra-operative sensing technology is an alternative to standard techniques in total knee arthroplasty (TKA) for determining balance by providing quantitative analysis of loads and point of contact throughout a range of motion. We used intra-operative sensing (VERASENSE—OrthoSensor, Inc.) to examine pie-crusting release of the medial collateral ligament in knees with varus deformity (study group) in comparison to a control group where balance was obtained using a classic release technique and assessed using laminar spreaders, spacer blocks, manual stress, and a ruler.
Methods
The surgery was performed by a single surgeon utilizing measured resection and posterior-stabilized, cemented implants. Seventy-five study TKAs were matched 1:3 with 225 control TKAs. Outcome variables included the use of a constrained insert, functional- and knee-specific Knee Society score (KSS) at six weeks, four months, and one year post-operatively. Outcomes were analyzed in a multivariate model controlling for age, sex, BMI, and severity of deformity.
Results
The use of a constrained insert was significantly lower in the study group (5.3 vs. 13.8%; p = 0.049). The use of increased constraint was not significant between groups with increasing deformity. There was no difference in functional KSS and knee-specific KSS between groups at any follow-up interval.
Conclusion
An algorithmic pie-crusting technique guided by intra-operative sensing is associated with decreased use of constrained inserts in TKA patients with a pre-operative varus deformity. This may cause a positive shift in value and cost savings.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Adequate soft tissue balance at the time of total knee arthroplasty (TKA) prevents early failure [1]. In cases of varus deformity a progressive release of the medial soft tissue sleeve (MSS) from the proximal medial tibia is needed to achieve balance [2–4]. The “classic” medial soft tissue release technique, popularized by John Insall et al., consists of a sharp subperiosteal dissection from the proximal medial tibia that includes superficial and deep medial collateral ligament (MCL), semimembranosus tendon, posteromedial capsule, along with the pes anserinus tendons, if needed [5]. However, this technique allows little control over releases that selectively affect the flexion and extension gaps [6].
When severe deformity is present, extensive MSS release can cause iatrogenic medial instability [7]. If collateral ligament laxity and/or imbalance persist despite reconstruction of the joint line, mechanical alignment, component rotation, and attempt at ligament balancing; a constrained TKA implant may be necessary [8–10]. The use of a constrained implant may be associated with greater wear, osteolysis, aseptic loosening rates, and cost [11, 12].
The use of selective releases to elongate the MSS has been reported but has not been widely utilized [3, 13–16]. Elongation of the MSS in extension involves the release of the posterior oblique ligament, posteromedial capsule, and semimembranosus tendon insertion. In flexion, the anterior aspect of the medial collateral ligament (MCL) may be released [3]. Concerns have been voiced regarding potential post-operative disruption of the MCL when pie-crusting is performed leading to early joint instability [15, 17, 18].
We recently reported that the use of an algorithmic approach to pie-crusting the MSS while performing TKR in a patient with varus deformity was safe and has no detrimental effects in clinical results, joint function, and stability [11]. The study was limited in that the intra-operative assessment of ligament balance wase performed with laminar spreaders, spacer blocks, manual stress, and a ruler [11].
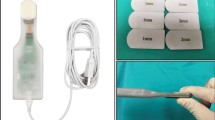
Intra-operative sensing technology is available as an alternative to the aforementioned techniques for determining balance [19–22]. Advantages include a quantitative analysis of loads and component tracking throughout knee range of motion. We chose to use Verasense (OrthoSensor Inc., Dania Beach, USA) which incorporates force plate sensors with a wireless link into a tibial insert “smart trial.” The insert replaces a standard trial insert and is specific to the implant system. Data is broadcast to a computer and a graphical interface shows load in pounds per square inch (PSI) and the point of contract between the insert and the femoral component in the medial and lateral compartments [23–25].
The aims of this study are to determine if the use of an algorithmic approach to perform medial soft tissue release that includes pie-crusting and is guided by intra-operative sensing— 1) reduces the need for constrained inserts in primary TKA and 2) results in improved functional outcomes. Additionally, we assessed if the use of constrained inserts is affected by the severity of pre-operative deformity.
Material and methods
We performed a retrospective review of 72 patients who underwent 75 primary TKAs (three bilateral) for osteoarthritis with a pre-operative varus deformity. All surgeries were performed by a single surgeon (*** Blinded ***). Patients were excluded during the study period if intra-operative sensing was not used. Cases were excluded if it was determined pre-operatively at the discretion of the surgeon that increased constraint would be necessary due to severity of varus deformity. All patients were followed for one year.
Knees in the study group were matched 1:3 with a historic group of TKAs operated with the “classic” subperiosteal release technique in order to achieve balance and without intra-operative sensing (control group). The control group time period was from 2005 to 2012 and the study group was from 2012 to 2014. Matching criteria included: age (+/− 5), BMI (+/− 3), sex (exact, female versus male), bilaterality (exact, unilateral versus bilateral TKA), pre-operative knee-specific Knee Society Score (KSS) (+/− 10), pre-operative functional KSS (+/− 10), and pre-operative gravity-assisted flexion (+/− 10). Criteria were further expanded to age (+/− 7), BMI (+/− 10), sex (allow mismatch), pre-operative knee-specific KSS (+/− 25), pre-operative functional KSS (+/− 30), and flexion (+/− 30) if three complete matches were not available (Table 1). Groups were also matched for degree of varus deformity. In the study group the average varus deformity (anatomic angle) was 10.7° (range: 5 to 20). In the control group the average varus deformity (anatomic angle) was 11° (range: 5 to 25) (p = 0.6).
Surgical technique remained constant from our previous study and involved a horizontal tibial cut using an extra-medullary alignment jig, distal femoral cut using an intramedullary alignment jig at 5° of valgus, measured resection technique, and posterior referencing instrumentation. Posterior-stabilized, cemented implants were used and the patella was routinely resurfaced. All knees in the study group were balanced with a combination of blocks and intra-operative sensing technology aiming at obtaining compartmental load differential within 15 PSI throughout the full range of motion. No absolute values were set, however, the knee was required to reach full extension. All knees in the control group were balanced using laminar spreaders, spacer blocks, manual stress, and a ruler.
The algorithmic approach to soft tissue release utilized in the study group included the resection of the posterior osteophytes as the initial balancing gesture. If additional MSS release was necessary to achieve a rectangular extension gap, a subperiosteal release of the posterior aspect of the MSS was performed with electrocautery, detaching the posterior aspect of the deep MCL, posteromedial capsule and semimembranosus tendon for the proximal and medial tibia [3]. Dissection did not extend more than 1.5 cm distal to the joint line. If additional release was necessary, the medial compartment was tensioned with a laminar spreader in extension, and multiple needle punctures (generally less than 6) were performed in the taut portion of the MSS using a 16G needle. After performing the posterior condylar resection, removing PCL and posterior osteophytes, the flexion gap was assessed using spacer blocks and laminar spreaders. The trial femoral and tibial components were placed followed by insertion of a tibial smart trial. Compartment loads were examined. If additional release was necessary to balance the gaps, multiple needle punctures in the taut portions of the MSS were performed. MCL pie-crusting was necessary to balance the extension or flexion gap in 41 of 75 patients. A TKA was considered well balanced when the load differential between compartments was within 15 PSI [22, 24–26], rectangular symmetric gaps were obtained, and medial and or lateral opening with the knee in extension and flexion did not exceed 3 mm when varus and valgus stresses were applied (Fig. 1). A posterior stabilized (PS) insert was used in well balanced knees. In the presence of residual ligament imbalance or irreconcilable flexion/extension mismatch, a constrained insert was implanted [9].
In the control group, the medial osteophytes were resected and the extension gap was assessed with spacer blocks. If lateral laxity was detected, a non-discriminatory, progressive subperiosteal dissection of the MSS was carried in an attempt at achieving a rectangular extension gap. Balance in flexion was also assessed with a spacer block and a lateralized extensor mechanism (dislocated patella). Additional releases on the MSS were performed if lateral laxity persisted in 90° of flexion. A constrained insert was used in knees with residual laxity as defined by greater than 3 mm of opening when varus and valgus stresses were applied in extension and flexion.
All implants utilized in this study were posterior stabilized and with the option of utilizing a constrained insert in a primary tibial and femoral component. Prosthetic design in the study group included Triathlon (Stryker, Mahwah, USA) in 42 knees, Vanguard (Zimmer Biomet, Warsaw, USA) in 24 knees, and Genesis 2 (Smith & Nephew, Memphis, USA) in nine knees. Prosthetic design in the control group included Triathlon (Stryker, Mahwah, USA) in 38 knees and Genesis 2 (Smith & Nephew, Memphis, USA) in 187 knees.
Intra-operative and peri-operative care remained consistent during the study period. Regional anesthesia was used. Patients were mobilized by post-operative day one. Each patient received standardized pain control, multimodal venous thromboembolism (VTE) prophylaxis [27], and medical co-management.
The analyzed pre-operative variables included: age, gender, BMI, deformity, functional KKS, and knee-specific KSS [28]. The anatomic varus deformity was measured on standing films and grouped according to KSS criteria (≤5°, 6–10°, 11–15°, and ≥16°). Four groups were defined based upon the severity of the deformity: mild (≤5°), moderate (6–10°), marked (11–15°), or severe (≥16°). The primary outcome measure was the use of a constrained insert. Secondary outcomes included functional KKS and knee-specific KSS at six weeks, four months, and a year post-operatively; symptomatic post-operative medial instability; and need for manipulation under anesthesia. Special attention was paid to the development of clinical symptoms of post-operative instability during the follow-up period.
Statistical analysis
Given that the primary aim of the study was to examine the influence of intra-operative sensing on the use of constrained inserts in primary TKA, we used this criterion to calculate our sample size. Constrained inserts were used in 5.3% of TKAs in our study group. Based on our previously reported experience, we estimated that the proportion of TKAs requiring a constrained insert would be 3.3 times higher. Utilizing a 2-sided significance level of 0.05, a 1:3 matching ratio results in a group sample sizes of 75 and 225 TKAs and achieves 80% power.
Descriptive statistics included for continuous variables and frequency and percentages for categorical variables. Demographic characteristics, functional KKS, and knee-specific KSS, and constrained insert use were compared between the groups using two-sample t-tests for continuous variables and Chi-square tests for categorical variables. A conditional logistic regression analysis was performed to compare the difference in the use of a constrained insert between the groups, controlling for age, gender, BMI, severity of varus deformity, preoperative functional KKS, and knee-specific KSS. The results were summarized using odds ratio (OR), 95% confidence intervals (95% CI), and p-values. Multiple regression analysis with the use of generalized estimating equation procedure was performed to examine if the use of intra-operative sensing results in improved functional outcomes, controlling for technique, insert type, manipulation, prophylaxis, severity of deformity, tibia size, insert size, tourniquet time, and length of hospital stay.
The comparison of constrained insert use between the study and the control groups was further stratified by the severity of the varus deformity. A Chi-square test was used to examine whether there’s a difference in the proportion of constrained insert use between the study and control groups, after controlling for severity of varus deformity. All tests were two-sided with a significance level of 0.05. All analyses were conducted using SAS for Windows 9.3 (SAS Institute Inc., Cary, NC).
Results
There were no significant differences in pre-operative age, gender, BMI, range of motion, functional KSS, and severity of varus deformity between the study and control groups. Differences in the pre-operative knee-specific KSS scores for the study and control groups were 34.1 and 31.2, respectively (p = 0.04) (Table 1). The tourniquet time was similar between study and control patients (53 min –range: 30 to 71; and 56 min – range: 7 to 103, respectively – p = 0.33).
The use of constrained inserts was significantly lower in the study than in the control group (5.3 vs. 13.8%, respectively) (p = 0.049). The difference in the use of constrained inserts was not significant in the univariate analysis of study and control patients with mild, moderate, marked, or severe deformity (p = 0.99, 0.75, 0.11, and 0.25, respectively). There was no difference in the functional KKS and knee-specific KSS, and range of motion between groups at all follow-up intervals. The severity of the varus deformity was not associated with constrained insert use. No difference in flexion instability was noticed.
There was an increase in the functional KSS and knee-specific KSS at all post-operative time intervals when compared to the pre-operative values (p < 0.0001). However, the changes in the functional KSS and knee-specific KSS were similar in study and control patients (p = 0.48 and p = 0.35 for functional KSS and knee-specific KSS, respectively). At six weeks, four months, and one year post-operatively, there was no new onset valgus instability indicating post-operative MCL rupture. No patients underwent manipulation under anesthesia in the study group, whereas 3.5% (eight patients) in the control group were manipulated (p = 0.21).
Discussion
With classic release technique [5], the portion of varus knees in which a constrained insert is necessary to address residual ligament instability can be up to 18% [9, 10]. An algorithmic approach to releasing the MSS that includes pie-crusting the taut structures in flexion or extension has been advocated by Verdonk et al. [3] as an alternative technique to achieve balance without the previously mentioned risks of the classic release technique. Our previous study supports the safety in pie-crusting the MSS without the concern of progressive stretching and post-operative failure [11].
Ligament balance is subjective, as is the decision to move from a PS to a constrained insert. Traditionally, a combination of laminar spreaders, spacer blocks, and manual stress are used to determine when rectangular flexion and extension spaces have been obtained. In this study, an algorithmic approach including pie-crusting was utilized for all patients, and the control group utilized a “classic” release technique advocated by pioneers of TKA surgery and using traditional balancing tools. The study group utilized intra-operative sensing technology to assess balance. Quantitative data through an arc of motion may provide a quantitative assessment of balance, which is unavailable with conventional instruments like laminar spreaders and blocks. As in previous studies, we looked for a difference of less than 15 PSI between medial and lateral sides of the joint [21].
We found that there was no significant difference in the post-operative functional KKS and knee-specific KSS scores between groups. Additionally there was no statistical difference in the use of constrained inserts based on severity of pre-operative deformity. There was however, a statistically significant decrease in the number of constrained implants used in those knees balanced with smart trials (5.3 vs. 13.8% p = 0.049).
Despite our previous report demonstrating that increased constraint does not seem to affect the early functional outcome and ROM following primary TKA [11], the use of increased constraint during primary TKA is not without risks. Fewer constrained inserts are desirable from a longevity standpoint as the design of the constrained tibial post increases resultant stresses transferred to the locking mechanism securing the liner to the tray, and to the fixation interfaces. Some implant designs requiring adjuvant, modularity, stem fixation, and greater bone resections. The conforming cam-post mechanism increases the surface area for adhesive and abrasive wear [29]. In our institution, Nam et al. compared the outcomes of 190 primary TKA that received a constrained insert and 140 with a non-constrained insert. Constrained TKA implanted with Palacos cement (Zimmer Inc., Warsaw, USA) failed at a high rate [30].
In addition, constrained constructs for TKA can be up to 205% more expensive than non-constrained ones [Courtesy of OR Material Management]. The use of the previously discussed pie-crusting technique combined with intra-operative sensing technology may result in implant cost savings. In order to assess if these savings would result in a reduction of the cost of care during hospitalization, the price of the sensing device should be considered.
In our study, we observed no MCL failure or development of medial laxity at 1 year of follow up, again supporting the safety of the procedure. Longer follow up and further studies may elucidate sub-populations that will have the greatest benefit from intra-operative sensing technology.
Studies reporting on the use of intra-operative sensing technology are being increasingly published. Verasense has been used to study a variety of different aspects of TKA including how variation in bone cuts and soft tissue release affect load across the knee [27], accuracy of current techniques for determining tibial rotation [31], and effects on post-operative functional scores [22, 24, 26]. We previously reported the effect of the extensor mechanism and patella lateralization on the compartmental loads in 56 well balanced TKAs [21].
This study has several limitations. First, it is a retrospective investigation of prospectively collected data. However, surgical indications and technique as well as peri-operative care was constant. Our matching methodology and strict comparison of pre-operative variables indicate that the study and control groups are comparable. It should be noted that some patients included in this study overlap with others previously reported by us. Specifically 12 TKAs in our control group were part of a study examining the safety of pie-crusting [11] and 22 in the study group were included in our previous study analyzing the effect of patellar position in the intra-operative compartmental loads [21]. The post-operative outcome of these patients has never been reported. Second, the prosthetic design changed throughout the study period reflecting the fact that the sensing device is available for a limited number of implants. However, all implants utilized in the study have a conventional posterior-stabilized design and allow the use of a constrained insert in a primary tibial and femoral component. The criteria for using a constrained insert remained constant throughout the study period. Third, all measurements with smart trials were done during trial reduction rather than after final implantation. Our recent investigations showed that there is variability in the loads recorded during trial reduction and those recorded after final prosthetic implantation. However, the state of adequate or inadequate balance did not change in 95% of cases [32]. Fourth, ligament balancing although quantitatively measured with intra-operative sensing, remains elusive and subjective to both intra and inter-observer variation. Fifth, long leg films to assess post-operative alignment and implant position were unavailable. Post-operative alignment in standing anteroposterior views of the knee revealed no significant difference in knee anatomic alignment (Study group: mean 4.6° of valgus – range 2 to 7 – SD: 1.5° – Control group: mean 4.7° of valgus – range 2 to 7 – SD: 1.6° – p = 0.81). Sixth, no assessment of long-term survivorship was performed. However, there is agreement in the literature that a minimum one-year follow up may be sufficient to assess the clinical safety of pie-crusting technique [14]. And finally, the results of a single experienced arthroplasty surgeon (*** Blinded ***) cannot be extrapolated to others.
Conclusion
Intraoperative sensing is a relatively new technology with potential to improve the balance and functional outcomes in TKA. An algorithmic approach to medial soft tissue balance that uses MCL pie-crusting and intraoperative sensing has resulted in diminished use of constrained TKA constructs. Further studies using intra-operative sensing technology are necessary to determine if the quantitative balancing of the knee translates to long-term improvements in function or implant survivorship.
References
Sharkey PF, Hozack WJ, Rothman RH, Shastri S, Jacoby SM (2002) Why are total knee arthroplasties failing today? Clin Orthop Relat Res 404:7–13. doi:10.1097/01.blo.0000036002.13841.32
Matsumoto T, Muratsu H, Kubo S, Matsushita T, Kurosaka M, Kuroda R (2011) The influence of preoperative deformity on intraoperative soft tissue balance in posterior-stabilized total knee arthroplasty. J Arthroplasty 26(8):1291–1298. doi:10.1016/j.arth.2011.01.003
Verdonk PCM, Pernin J, Pinaroli A, Si Selimi TA, Neyret P (2009) Soft tissue balancing in varus total knee arthroplasty: an algorithmic approach. Knee Surg Sports Traumatol Arthrosc 17(6):660–666. doi:10.1007/s00167-009-0755-7
Bellemans J, Vandenneucker H, Vanlauwe J, Victor J (2010) The influence of coronal plane deformity on mediolateral ligament status: an observational study in varus knees. Knee Surg Sports Traumatol Arthrosc 18(2):152–156. doi:10.1007/s00167-009-0903-0
Insall J, Scott N, Ranawat CS (1979) The total condylar knee prosthesis. A report of two hundred and twenty cases. J Bone Joint Surg Am 61(2):173–180
Krackow KA, Mihalko WM (1999) The effect of medial release on flexion and extension gaps in cadaveric knees: implications for soft-tissue balancing in total knee arthroplasty. Am J Knee Surg 12(4):222–228
Mullaji A, Sharma A, Marawar S, Kanna R (2009) Quantification of effect of sequential posteromedial release on flexion and extension gaps. J Arthroplasty 24(5):795–805. doi:10.1016/j.arth.2008.03.018
Hood RW, Vanni M, Insall JN (1981) The correction of knee alignment in 225 consecutive total condylar knee replacements. Clin Orthop Relat Res 160:94–105
King B, Gladnick B, Lee L, Lyman S, Gonzalez Della Valle A (2014) Range of motion and function are not affected by increased post constraint in patients undergoing posterior stabilized total knee arthroplasty. Knee 21(1):194–198. doi:10.1016/j.knee.2013.07.002
Callaghan JJ, O’Rourke MR, Liu SS (2005) The role of implant constraint in revision total knee arthroplasty. J Arthroplasty 20(4 suppl):41–43. doi:10.1016/j.arth.2005.03.008
Goudarz Mehdikhani K, Morales Moreno B, Reid JJ, de Paz Nieves A, Lee YY, Gonzalez Della Valle A (2016) An algorithmic, pie-crusting medial soft tissue release reduces the need for constrained inserts in patients with severe varus deformity undergoing total knee arthroplasty. J Arthroplasty 31(7):1465–1469. doi:10.1016/j.arth.2016.01.006
Lachiewicz PF, Soileau ES (2006) Ten-year survival and clinical results of constrained components in primary total knee arthroplasty. J Arthroplasty 21(6):803–808. doi:10.1016/j.arth.2005.09.008
Bellemans J, Vandenneucker H, Van Lauwe J, Victor J (2010) A new surgical technique for medial collateral ligament balancing: multiple needle puncturing. J Arthroplasty 25(7):1151–1156. doi:10.1016/j.arth.2010.03.007
Kim MW, Koh IJ, Kim JH, Jung JJ, In Y (2015) Efficacy and safety of a novel three-step medial release technique in varus total knee arthroplasty. J Arthroplasty 30(9):1542–1547. doi:10.1016/j.arth.2015.03.037
Koh IJ, Kwak D-S, Kim TK, Park IJ, In Y (2014) How effective is multiple needle puncturing for medial soft tissue balancing during total knee arthroplasty? A cadaveric study. J Arthroplasty 29(12):2478–2483. doi:10.1016/j.arth.2013.11.004
Meftah M, Blum YC, Raja D, Ranawat AS, Ranawat CS (2012) Correcting fixed varus deformity with flexion contracture during total knee arthroplasty: the “inside-out” technique. J Bone Joint Surg Am 94(10):e66. doi:10.2106/JBJS.K.01444
Meneghini RM, Daluga AT, Sturgis LA, Lieberman JR (2013) Is the pie-crusting technique safe for MCL release in varus deformity correction in total knee arthroplasty? J Arthroplasty 28(8):1306–1309. doi:10.1016/j.arth.2013.04.002
Mihalko WM, Woodard EL, Hebert CT, Crockarell JR, Williams JL (2015) Biomechanical validation of medial pie-crusting for soft-tissue balancing in knee arthroplasty. J Arthroplasty 30(2):296–299. doi:10.1016/j.arth.2014.09.005
Wasielewski RC, Galat DD, Komistek RD (2005) Correlation of compartment pressure data from an intraoperative sensing device with postoperative fluoroscopic kinematic results in TKA patients. J Biomech 38(2):333–339
Roche M, Elson L, Anderson C (2014) Dynamic soft tissue balancing in total knee arthroplasty. Orthop Clin North Am 45(2):157–165. doi:10.1016/j.ocl.2013.11.0011
Schnaser E, Lee Y-Y, Boettner F, Gonzalez Della Valle A (2015) The position of the patella and extensor mechanism affects intraoperative compartmental loads during total knee arthroplasty. A pilot study using intraoperative sensing to guide soft tissue balance. J Arthroplasty 30(8):1348–1353. doi:10.1016/j.arth.2015.03.004
Gutske KA, Golladay GJ, Roche MW, Elson LC, Anderson CR (2014) A new method for defining balance: promising short-term clinical outcomes of sensor-guided total knee arthroplasty. J Arthroplasty 29(5):955–960. doi:10.1016/j.arth.2013.10.020
Gustke KA (2012) Use of smart trials for soft-tissue balancing in total knee replacement surgery. Bone Joint J 94(11 Supple A):147–150. doi:10.1302/0301-620X.94B11.30621
Gustke KA, Golladay GJ, Roche MW, Jerry GJ, Elson LC, Anderson CR (2014) Increased satisfaction after total knee replacement using sensor-guided technology. Bone Joint J 96(10):1333–1338. doi:10.1302/0301-620X.96B10.34068
Gustke KA (2014) Soft-tissue and alignment correction: the use of smart trials in total knee replacement. Bone Joint J 96(11 Supple A):78–83. doi:10.1302/0301-620X.96B11.34339
Walker PS, Meere PA, Bell CP (2014) Effects of surgical variables in balancing of total knee replacements using an instrumented tibial trial. Knee 21(1):156–161. doi:10.1016/j.knee.2013.09.002
Gesell M, González Della Valle A, Bartolomé Garcia S, Memtsoudis SG, Ma Y, Haas S, Salvati EA (2013) Safety and efficacy of multimodal thromboprophylaxis following primary total knee arthroplasty: a comparative study of preferential aspirin vs. routine Coumadin chemoprophylaxis. J Arthroplasty 28(4):575–579. doi:10.1016/j.arth.2012.08004
Insall JN, Dorr LD, Scott RD, Scott WN (1989) Rationale of the Knee Society clinical rating system. Clin Orthop Relat Res 148:13–14
Wright TM (2005) Polyethylene in knee arthroplasty. Clin Orthop Relat Res 440:141–148
Nam D, Umunna BP, Cross MB, Cornell CN (2012) Clinical results and failure mechanisms of a nonmodular constrained knee without stem extensions. HSS J 8(2):96–102
Roche MW, Elson LC, Anderson CR (2015) A novel technique using sensor-based technology to evaluate tibial tray rotation. Orthopedics 38(3):e217–e222. doi:10.3928/01477447-20150305-60
Nodzo S, Francescini V, Gonzalez Della Valle A (2016) Intraoperative load sensing variability during cemented, posterior stabilized total knee arthroplasty. J Arthroplasty. doi:10.1016/j.arth.2016.06.029
Acknowledgements
This study was partially funded by the generous donation of Mr. Glenn Bergenfield and the Sidney Milton and Leoma Simon Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
One of the authors is a paid consultant for OrthoSensor. There are no other conflicts of interest.
Funding
There is no funding source.
Ethical approval
This study was approved by the institution’s IRB.
Informed consent
Informed consent waived by the IRB due to the retrospective nature of the study.
Rights and permissions
About this article
Cite this article
Amundsen, S., Lee, Yy. & González Della Valle, A. Algorithmic pie-crusting of the medial collateral ligament guided by sensing technology affects the use of constrained inserts during total knee arthroplasty. International Orthopaedics (SICOT) 41, 1139–1145 (2017). https://doi.org/10.1007/s00264-017-3413-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00264-017-3413-6