Abstract
Purpose
Compartmental load-sensing technology has been used in the attempt to achieve optimal soft tissue balance during total knee arthroplasty (TKA). This study was conducted to investigate the validity of such use of intraoperative sensing technology.
Methods
Ninety-three knees scheduled to undergo total knee arthroplasty for knee osteoarthritis with a tibial sensor were prospectively enrolled. Measurements were divided into three groups according to the three different time points of intraoperative load testing: group Trial (with the trial components), group Final (with the definitive cemented implants and an open joint capsule), and group Closed (with the definitive cemented implants and a closed joint capsule). Load measurements and component rotational alignments were documented at 10°, 30°, 45°, 90°, and 120° of flexion for all three groups, and compared. One year postoperatively, the joint line obliquity angle was obtained radiographically in the valgus and varus stress views at 10° and 30° flexion to evaluate the clinical instability. The Knee Society, Hospital for Special Surgery, and Western Ontario McMaster Universities Osteoarthritis Index scores were used to determine functional outcomes. The correlations of the above outcomes with intraoperative load were evaluated.
Results
There were significant differences in medial and lateral loads at all flexion angles (except at a 120° lateral load) between group Trial and group Final (p < 0.05). Tibial trays were internally rotated to a significantly higher degree in group Final than in group Trial (p = 0.010). The lateral compartmental load significantly decreased after patellar inversion (p = 0.037). There were no correlations of intraoperative load with clinical instability and functional outcomes.
Conclusion
Significant variability was observed between the trial and final implant measurements and intraoperative sensing data were not correlated with instability or functional outcomes over a 1-year period. Therefore, intraoperative sensor technology provides limited feedback and clinical efficacy in the adjustment of the soft tissue balance during TKA.
Level of evidence
Level II.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Compartmental load-sensing technology was also introduced in the attempt to achieve optimal soft tissue balancing during TKA [4, 21]. A tibial sensor increases the accuracy of soft tissue balancing by quantifying the obscure feeling of load or tension and providing real-time data while the knee is taken through a range of motions (ROMs) during TKA [5]. This technology has yielded promising results related to improved objectivity in load measurement and improved clinical outcomes [3, 6, 13, 14].
However, many of the above-mentioned studies were conducted by the designers of the sensor technology, and the follow-up periods were relatively short [4, 21]. Furthermore, several issues related to the variability of measurements have been noted, such as movement of the sensor in the trial compartment and a mismatched point in the tibial contact location [2, 15, 16, 18]. Most of these studies also included a relatively low number of patients or biomechanical parameters. Hence, the validity of the tibial sensor in the assessment of soft tissue balance remains unknown. Therefore, this study was conducted with a relatively large number of patients to investigate the validity of such use of the tibial sensor by focussing on not only the presence of variability between measurements at different time points in the operation but also the clinical outcomes. The hypothesis of the present study was that the use of sensors would have limited validity in TKA.
Materials and methods
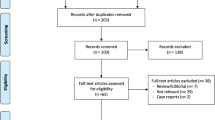
From May 2017 to February 2020, 93 knees scheduled to undergo TKA for knee osteoarthritis were enrolled, and the data were collected prospectively. Exclusion criteria were as follows: age < 50 years or > 85 years; rheumatoid or traumatic arthritis; previous knee surgery; severe sagittal or coronal deformity (preoperative flexion contracture > 20° or mechanical axis > 20°); neuromuscular disorders such as cerebral palsy, infarct, and parkinsonism (Fig. 1). Demographic characteristics of patients are summarised in Table 1.
The LPS standard posterior-stabilised system (Zimmer Biomet, Warsaw, IN) was used for all TKAs. The VERASENSE sensor device (OrthoSensor Inc., Dania Beach, FL) was used to measure the load and rotation between the tibial and femoral components. The VERASENSE device is a sensor-embedded tibial trial that can detect tibiofemoral contact points throughout the ROM to evaluate loads, component alignment, ligament tension, and rotation. OrthoSensor’s Link Station and cloud-based iQ software were used to wirelessly communicate with the sensor to provide real-time data and feedback during the surgical procedure.
Surgical procedure using the intraoperative sensor
One experienced, senior surgeon (M.C.L) performed all TKAs with routine procedures regardless of the measurement on the sensor device. The modified gap technique was performed in all cases, with the goal of neutral mechanical alignment. Briefly, an anterior midline skin incision was made and medial parapatellar arthrotomy was performed with a tourniquet. As a preliminary step, medial soft tissue, such as the semitendinosus and deep medial collateral ligament, was released with removal of the medial osteophytes. Both the cruciate ligaments were resected. Distal femoral cutting was performed using an intramedullary guide perpendicular to the mechanical axis. Proximal tibial cutting was performed with an extramedullary guide perpendicular to the mechanical axis. In the process of chamfer cutting, component rotation was determined with the modified gap technique, by tensioning the medial and lateral collateral ligaments in the direction of gravity. The Xcelerate Knee Balancer (Stryker Corporation, Kalamazoo, MI) was used to measure the tension of the medial and lateral soft tissue, and the femoral cutting block and alignment guide were used to measure the extension and flexion gaps. The medial and lateral gap differences were adjusted using a manual distraction device, and accepted when they were < 2 mm in size. Additional soft tissue, such as the semimembranosus and the popliteal tendon, was released to obtain equal-sized flexion and extension gaps and a rectangular flexion gap. The tibial rotation was determined by adjusting the tibial plate to the anterior border of the tibia and verifying its position upon knee extension when all trials (femur, insert, and tibia) were in position. Once the soft tissue balance was deemed adequate by the surgeon, the loads between the femoral and tibial trial components were measured by inserting the tibial sensor; these measurements were documented as ‘Trial’. Loads were recorded in two compartments (medial and lateral) while taking the knee through the ROM. In addition, the angles of rotation between the femoral and tibial trial components were recorded through the ROM. The tibial trial components were free floating without pin insertion during measurements to allow positioning according to their natural movement. Subsequently, all trial components were removed and irrigation was performed. Thereafter, the bone was dried and the real components were fixed with cement (Palacos R + G pro, Heraeus Kulzer GmbH, Hanau, Germany) using a pressurisation technique, according to a previously described protocol [8]. The loads and rotations were measured in exactly the same way as for the Trial measurements; these results were documented as ‘Final’. After resurfacing of the patella, the loads and rotations were measured again with relocation of the patella in the trochlear groove using two towel clips. These results were documented as ‘Closed’. The trial components were placed after the final releases were performed following femoral and tibial resections, and no additional procedures related to soft tissue balancing were performed between each measurement phase.
Postoperative management and rehabilitation
Standardised postoperative rehabilitation and pain management were applied according to a previously described protocol [10]. Briefly, the knee was immobilised for the first day after the operation, and machine-assisted passive motion of the knee joint was begun within 24 h of the operation. Patients started walking from 24 h after the operation. From 48 h after the operation, the patients were encouraged to start supervised passive and active ROM exercises.
Outcome assessments
A clinical investigator blinded to the study, gathered all data. Measurements were divided into three groups: group Trial (with the trial components), group Final (with the definitive cemented implants and an open joint capsule), and group Closed (with the definitive cemented implants and a closed joint capsule). Compartment loads were measured in pounds (lbs). ‘Quantifiably balanced’ soft tissue was defined as that with a load difference between the mediolateral compartments of ≤ 15 lbs [7]. Component rotational alignments were also recorded at 10°, 30°, 45°, 90°, and 120° of flexion for all three groups, as the angle (°) between the femoral and tibial components.
To investigate the clinical validity of the sensor measurements, radiographs were obtained in the valgus and varus stress views at 10° and 30° flexion, 1 year after the operation. The correlation between the joint line obliquity angle in each view and the medial/lateral loads at 10° and 30° flexion was evaluated. At least two orthopaedic surgeons who specialise in knee surgery, all blinded to the surgery, repeatedly measured the joint line obliquity angle together to improve the reliability of measurement.
Functional outcomes in the form of the Knee Society (KS) score, Hospital for Special Surgery (HSS) score, and Western Ontario McMaster Universities Osteoarthritis Index (WOMAC) score were evaluated 1 year after surgery [1, 9, 17]. The correlations between functional score and load difference between the medial and lateral compartments were evaluated to determine the load difference that could affect the clinical outcome.
Statistical analysis
Continuous variables are displayed as the mean ± standard deviation and were analysed with Student’s t test (load, rotational angle, joint line obliquity angle, and functional score). Correlation and linear regression analyses were used to evaluate the association of the compartmental load with the joint line obliquity angle and functional outcomes. Statistical analyses were conducted using IBM SPSS Statistics for Windows (version 23.0; IBM Corp., Armonk, NY). A two-sided p value < 0.05 was considered statistically significant. A priori power analysis was performed based on the results of a previous study, in which the mean percentage of unbalanced knees was 13% [7]. Seventy-nine patients were required at a compensated alpha level of 0.05 and power of 80% (two-sided). Therefore, 93 patients were recruited to account for a 15% loss of participants.
Results
There were significant differences in medial and lateral loads at all flexion angles (except at a 120° lateral load) between group Trial and group Final (Figs. 2, 3). Medial loads were significantly higher in group Trial (p = 0.016), whereas lateral loads were significantly higher in group Final (p = 0.028). Tibial trays were internally rotated to a significantly higher degree in group Final than in group Trial (p = 0.010) (Table 2). In addition, the tibial trays were positioned more internally with the progression of knee flexion in both groups (Fig. 4). The lateral compartmental load was significantly altered between group Final and group Closed, with a decreased lateral compartmental load after patellar inversion (p = 0.037) (Table 2, Fig. 5).
Only 90 patients were included for analyses because of loss to follow-up at 1 year after surgery (Fig. 1). No correlations between the intraoperative sensing data and any of the joint line obliquity angles were identified (Figs. 6, 7). In addition, there were no significant correlations between the load difference and the KS, HSS, or WOMAC scores (Fig. 8).
Discussion
The most important finding of the present study is that statistically significant variations between the trial and final implant measurements were observed when the intraoperative load sensor was used during TKA. The main aim of this study was to investigate the validity of intraoperative sensor technology by evaluating the difference between the loads obtained when the sensor is used with the trial components and when it is inserted after implant cementation. To this end, an experienced, senior surgeon performed all TKAs while being blinded to the measurements of the sensor. Trials were placed after final releases were performed, and no additional balancing was performed between each measurement phase.
We observed unpredictable motion between the trial tibial plate and the cut surface of the tibial bone when the knee was flexed during TKA. Specifically, when the sensor was attached to the trial tibial plate, the measured load changed as the knee was flexed. Most balancing procedures should be performed before cementation, as the range of balancing is limited after cementing. Furthermore, modification of the rotational alignment is impossible after cementation. Therefore, the results of our study call into question the validity of the use of an intraoperative tibial sensor in the assessment of soft tissue balance and component rotational alignment. Previous studies yielded similar results [2, 15, 16, 18]. Nodzo et al. [16] reported on a change in the load in the lateral compartment after cementation. Roche et al. [18] observed a statistically significant change in tibial rotational alignment after cementation. On the other hand, in another study, no statistically significant change in load was observed after cementation [23]. However, the authors of that study noted that the sample size might have been too small to detect such differences. In addition, a recently published biomechanical study revealed that the sensing area is smaller than the final tibial baseplate, which impair the accuracy of the trial measurement, as the contact location during flexion may be outside the sensing area [15].
The tibial trials were free floating without pin insertion during measurements to allow positioning according to their natural movement. Some surgeons recommend that a pin should be placed in either an anteromedial or an anterolateral position to stabilise the translational motion [7]. However, the unilateral insertion of a pin is not enough to fix the tibial; in fact, it is more likely to generate undesirable rotation along the axis of the pin. Furthermore, anterolateral pin placement may irritate the patellar tendon, affecting the load differences. On the other hand, pin insertion only on the anterior side may result in the lift-off of the posterior side of tibia plate during normal ROM.
The variability of the load in the lateral compartment was determined before and after patellar eversion in the present study. The lateral compartmental load statistically significantly increased after the patella was everted. After cement fixation, the expected results were obtained based on the physical position of the patella. Previous studies have yielded results similar to those of the present study [19, 20]. Furthermore, internal rotation of the tibial tray increased with flexion once the components were cemented in place, manifesting as lateral femoral condyle rollback and pivoting. These findings imply that firm fixation of the sensor to the tibial baseplate resulted in a more accurate reflection of the anatomical position of the knee and natural kinematics, justifying the results of the present study.
To the best of our knowledge, this is the first study in which the correlation between postoperative instability and intraoperative sensing data were investigated. The joint line obliquity angles were measured radiologically to assess the degree of instability. However, no clinical correlations were observed. This suggests that postoperative instability cannot be accurately predicted from the measurements obtained with the sensor. Moreover, none of the clinical scores we used was correlated with the intraoperative sensing data. Satisfactory soft tissue balance, as defined using intraoperative sensing data, was reportedly associated with positive clinical outcomes [6, 7, 14]. However, in multiple recently published studies, no specific correlations were reported [11, 12, 22]. One of the reasons for this discrepancy could be the fact that, other than the teams of the sensor technology designers, clinical research teams from around the world have also started to publish studies, providing abundant data for the objective evaluation and validation of the sensor technology.
This study has several limitations. First, the results may not be generalisable because of its single-centre nature and involvement of only one surgeon. The intraoperative sensing data may be biased because of the way in which the surgeon performed knee flexion, as an axial load from the ankle to the knee joint is inevitably generated during knee flexion. In the effort to overcome the limitations of the sensor during open-chain kinematic measurements, it is possible that the surgeon increased the axial load to the contact area, inducing an unexpected locking motion in the tibial baseplate. This motion could explain why the medial load was higher in group Trial. Furthermore, the higher lateral load in group Final suggests that the results of our procedure, conducted by one surgeon, may not be generalisable. We are currently working on solving this conundrum to ensure a balanced TKA. Further studies should be conducted to account for differences in the axial force applied by different surgeons. Second, the follow-up period was relatively short; therefore, long-term evaluation of knee stability and clinical outcomes was not possible. Specifically, instability may accumulate over time. Longer term studies of large sample size should be conducted to determine the correlation between intraoperative sensing data and clinical outcomes.
Conclusion
Most soft tissue balancing procedures should be completed before cementation; however, in the present study, statistically significant variability was observed between the trial and final implant measurements, and intraoperative sensing data were not correlated with the instability and functional outcome of TKA over a 1-year period. Therefore, intraoperative sensor technology provides limited feedback in adjusting soft tissue balance and clinical efficacy.
Abbreviations
- HSS:
-
Hospital for Special Surgery
- KS:
-
Knee Society
- ROM:
-
Range of motion
- TKA:
-
Total knee arthroplasty
- WOMAC:
-
Western Ontario McMaster Universities Osteoarthritis Index
References
Bellamy N, Buchanan WW, Goldsmith CH, Campbell J, Stitt LW (1988) Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J Rheumatol 15:1833–1840
Chow J, Wang K, Elson L, Anderson C, Roche M (2017) Effects of cementing on ligament balance during total knee arthroplasty. Orthopedics 40:e455–e459
Geller JA, Lakra A, Murtaugh T (2017) The Use of electronic sensor device to augment ligament balancing leads to a lower rate of arthrofibrosis after total knee arthroplasty. J Arthroplasty 32:1502–1504
Ghirardelli S, Bala A, Peretti G, Antonini G, Indelli PF (2019) Intraoperative sensing technology to achieve balance in primary total knee arthroplasty: a review of the literature. JBJS Rev 7:e4
Gustke KA (2014) Soft-tissue and alignment correction: the use of smart trials in total knee replacement. Bone Joint J 96-b:78–83
Gustke KA, Golladay GJ, Roche MW, Elson LC, Anderson CR (2014) A new method for defining balance: promising short-term clinical outcomes of sensor-guided TKA. J Arthroplasty 29:955–960
Gustke KA, Golladay GJ, Roche MW, Elson LC, Anderson CR (2014) Primary TKA patients with quantifiably balanced soft-tissue achieve significant clinical gains sooner than unbalanced patients. Adv Orthop 2014:628695
Han HS, Lee MC (2017) Cementing technique affects the rate of femoral component loosening after high flexion total knee arthroplasty. Knee 24:1435–1441
Insall JN, Dorr LD, Scott RD, Scott WN (1989) Rationale of the knee society clinical rating system. Clin Orthop Relat Res 1:13–14
Kim JK, Ro DH, Lee HJ, Park JY, Han HS, Lee MC (2020) Efficacy of systemic steroid use given one day after total knee arthroplasty for pain and nausea: a randomized controlled study. J Arthroplasty 35:69–75
Livermore AT, Erickson JA, Blackburn B, Peters CL (2020) Does the sequential addition of accelerometer-based navigation and sensor-guided ligament balancing improve outcomes in TKA? Bone Joint J 102-b:24–30
MacDessi SJ, Cohen DA, Wood JA, Diwan AD, Harris IA (2021) Does the use of intraoperative pressure sensors for knee balancing in total knee arthroplasty improve clinical outcomes? A comparative study with a minimum two-year follow-up. J Arthroplasty 36:514–519
Manning WA, Ghosh K, Blain A, Longstaff L, Deehan DJ (2017) Tibiofemoral forces for the native and post-arthroplasty knee: relationship to maximal laxity through a functional arc of motion. Knee Surg Sports Traumatol Arthrosc 25:1669–1677
McClelland JA, Webster KE, Ramteke AA, Feller JA (2017) Total knee arthroplasty with computer-assisted navigation more closely replicates normal knee biomechanics than conventional surgery. Knee 24:651–656
Nicolet-Petersen SJ, Howell SM, Hull M (2018) Force and contact location measurement errors of the VERASENSE. J Biomech Eng. https://doi.org/10.1115/1.4040601
Nodzo SR, Franceschini V, Gonzalez Della Valle A (2017) Intraoperative load-sensing variability during cemented, posterior-stabilized total knee arthroplasty. J Arthroplasty 32:66–70
Ranawat CS, Shine JJ (1973) Duo-condylar total knee arthroplasty. Clin Orthop Relat Res 94:185–195
Roche MW, Elson LC, Anderson CR (2015) A novel technique using sensor-based technology to evaluate tibial tray rotation. Orthopedics 38:e217-222
Schnaser E, Lee YY, Boettner F, Gonzalez Della Valle A (2015) The position of the patella and extensor mechanism affects intraoperative compartmental loads during total knee arthroplasty: a pilot study using intraoperative sensing to guide soft tissue balance. J Arthroplasty 30:1348-1353.e1343
Sculco P, Gruskay J, Nodzo S, Carrol K, Shanaghan K, Haas S et al (2018) The role of the tourniquet and patella position on the compartmental loads during sensor-assisted total knee arthroplasty. J Arthroplasty 33:S121-s125
Siddiqi A, Smith T, McPhilemy JJ, Ranawat AS, Sculco PK, Chen AF (2020) Soft-tissue balancing technology for total knee arthroplasty. JBJS Rev 8:e0050
Song SJ, Kang SG, Lee YJ, Kim KI, Park CH (2019) An intraoperative load sensor did not improve the early postoperative results of posterior-stabilized TKA for osteoarthritis with varus deformities. Knee Surg Sports Traumatol Arthrosc 27:1671–1679
van der Linde JA, Leong AKL (2019) Does bone cement influence soft tissue balancing in total knee arthroplasty? Knee 26:405–409
Funding
There is no funding source.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author(s) declare that they have no competing interests.
Ethical approval
This study obtained ethical approval from the institutional review board of Seoul National University Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kim, JK., Lee, D.W., Ro, D.H. et al. Variability between the trial and final implant measurements during the sensor-guided total knee arthroplasty. Knee Surg Sports Traumatol Arthrosc 30, 2846–2853 (2022). https://doi.org/10.1007/s00167-021-06795-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-021-06795-2