Abstract
Purpose
To assess the utility of magnetic resonance imaging (MRI) in patients with clinically suspected ectopic pregnancy (EP).
Methods
We retrospectively reviewed MRIs of 26 consecutive patients who were clinically suspected of having an EP. The diagnostic utility of MRI features of EP was analyzed retrospectively as follows: (1) Direct sign detection of ectopic gestational sac (GS); (2) Indirect signs tubal dilatation with hemosalpinx, adnexal hematoma, and hemorrhagic ascites. The diagnostic accuracy of each sign and their combination was compared to surgical records. The MRI findings of an ectopic GS were reviewed as follows: size, shape, signal intensity, and enhancement pattern.
Results
Of 26 patients, 24 had a tubal pregnancy; 22 of these 24 patients (92%) had a direct sign (sensitivity: 91.3%; specificity: 100%; positive predictive value: 100%). The diagnostic accuracy of the direct sign was 92%; this was more accurate than that of any single indirect sign (39%, 54%, and 50%, respectively). However, the diagnostic accuracy of EP increased to 100% when diagnostic criteria required the presence of a direct sign or at least two indirect signs.
Conclusions
MRI is an effective modality for diagnosing EP with a high detection rate of extrauterine GSs. The combination of direct and indirect signs is useful for establishing the correct diagnosis.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
An ectopic pregnancy (EP) is a common clinical condition in which a fertilized ovum implants and grows in an area other than the uterine cavity. The frequency of EP is increasing due to the increase in sexually transmitted disease as well as the growth of assisted reproductive technology [1]. Diagnosis and management of EP has shifted from emergency laparotomy to more conservative and less invasive methods. The detection of an extrauterine gestational sac (GS) is a definite indicator of EP; following identification, an appropriate course of therapy should be chosen. Although transvaginal sonography (TVUS) is the initial modality of choice, it may occasionally be difficult to image the implantation site. For complicated cases, magnetic resonance imaging (MRI) is reported to be another diagnostic modality [2, 3]. The purpose of this study was to both define the MRI findings of EP and assess the utility of MRI in patients with clinically suspected EP.
Materials and methods
Between September 2007 and March 2012, we retrospectively reviewed the medical records and MRI results of 26 consecutive patients who were clinically suspected of having an EP at Nara Medical University. All patients underwent TVUS before MRI and the results were indeterminate. The gold standard for final diagnosis was a surgical procedure [laparoscopy, surgery, or dilatation and curettage (D&C)]. Based on the surgical procedure, 24 patients were diagnosed with EP; one patient suffered a spontaneous abortion and another suffered a missed abortion with an underdeveloped intrauterine GS. All EP patients were tubal pregnancies: 19 ampullar, 3 interstitial, and 2 isthmic. The mean patient age was 33 years (range 25–38). Mean estimated gestational age (based on the last menstrual period) was 7 weeks (range 5–10). The serum β-subunit of human chorionic gonadotropin (β-hCG) levels was measured and was found to be elevated in all patients (range 604–32473 mIU/mL).
MR imaging
Pelvic MRI was performed using a 1.5-T MR unit (MAGNETOM Avanto; Siemens, Germany; n = 22) or 3-T MR unit (MAGNETOM Verio; Siemens, Germany; n = 4). A phased-array body coil was used. All images were obtained with the parallel imaging technique. MRI sequences included axial and sagittal T2-weighted fast spin echo, axial T1-weighted spin echo, and diffusion-weighted images; all patients underwent imaging (Table 1). Dynamic contrast-enhanced study was obtained in 19 cases and T2*-weighted imaging was obtained in 20 cases. Dynamic contrast images with volumetric interpolated breath-hold examination (VIBE) were obtained in the axial plane every 30, 90, and 150 s after intravenous administration of 0.1 mmol of gadolinium diethylene-triamine-pentaacetic acid (Magnevist; Beyel, Osaka, Japan). The sequences of MRI studies are summarized in Table 1.
MRI image review and analysis
MRI of 26 consecutive patients who were clinically suspected of having an EP were retrospectively reevaluated by two expert radiologists (A.T. and J.T.) by consensus without any information regarding clinical results. For diagnosis of an EP, the following findings were analyzed: extrauterine GS; adnexal hematoma; hemorrhagic ascites; and tubal dilatation. An extrauterine GS was defined as a sac-like cystic lesion surrounded by a thick peripheral area with distinct low signal intensity on T2-weighted images [4, 5]. In cases with contrast-enhanced study, the existence of marked ring-like or dot-like peripheral enhancement was also considered as trophoblastic tissue of a GS [4, 6]. The consistency and the accuracy of the MRI findings regarding a GS were verified with surgical records. Since we focused on the extrauterine GS findings, the following imaging features of GS in each case were also assessed: size; shape; signal intensity on unenhanced scans; and enhancement pattern on dynamic enhanced images. The correlation coefficient between the size of the GS and estimated gestational age was also evaluated. Ovarian corpus lutein cysts were also evaluated.
MRI findings were categorized into two groups comprising direct signs (clear evidence of extrauterine GS) and indirect signs (adnexal hematoma, hemorrhagic ascites, and tubal dilatation). According to the imaging results, the diagnostic accuracy of MRI for the diagnosis of EP using direct or indirect signs was analyzed.
Statistical analysis
The performance of MRI was evaluated in terms of sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV).
The correlation coefficient between the size of the GS and estimated gestational age was calculated with Graphpad Prism Software (Graphpad Prism for Mac, version 5.0c; Graphpad Software, La Jolla, CA, USA).
Results
Of 26 patients clinically suspected of having an EP, an extrauterine GS was observed in 22, an adnexal hematoma in 12, hemorrhagic ascites in 11, and tubal dilatation in 8. In 22 patients in whom a GS was imaged via MRI, the site of the GS noted on surgical records was compared to the MRI findings. Table 2 summarizes MRI findings regarding a GS. The size of the GS ranged from 10 to 42 mm (mean: 18 mm); no correlation was found between GS size and gestational age (r = 0.1703; Fig. 1). The shape of sac-like structure was round in 17 cases and oval in 5 cases. Signal intensity on T2-weighted images was high in 18 cases, and low in 4 cases. On T1-weighted images, 8 cases exhibited low intensity, 11 cases exhibited iso-intensity, and 3 exhibited high intensity. On diffusion-weighted images, 20 cases exhibited high signal intensity; 8 exhibited ring-like signal intensity; and 12 exhibited dot-like intensity. A dynamic study was obtained for 16 cases; the enhancement pattern was ring-like in 12 cases and dot-like in 4 cases. Corpus luteum cysts coexisted in 12 patients (Table 3).
Table 4 summarizes direct and indirect signs of EP on MRI. Based on the gold standard, the diagnostic accuracy of a direct sign on MRI for the diagnosis of EP was 92%. The diagnostic accuracy of indirect signs was less: tubal dilatation (39%), adnexal hematoma (54%), and hemorrhagic ascites (50%).
Discussion
EP is a clinical condition in which a fertilized ovum implants on any tissue other than the endometrial lining. Recognized risk factors include the use of an intrauterine device, a history of pelvic inflammatory disease (especially Chlamydia trachomatis), previous tubal surgery, previous EP, and assisted reproductive technology (ART) [7, 8]. The incidence of EP is between 1% and 2% of all pregnancies and has increased due to growing number of sexually transmitted disease and employment of ART [9]. In developed nations, the mortality of EP is decreasing due to advances in the use of diagnostic tools, surgical techniques, and medical treatment. The introduction of sensitive β-hCG assays and high-resolution TVUS facilitate making a diagnosis before the onset of peritoneal hemorrhage. Surgical techniques have also developed and improved, and management of EP has shifted from emergency laparotomy to non-invasive and conservative methods. Medical treatment with methotrexate has contributed to the employment of nonsurgical and fertility-preserving treatment options; furthermore, it reportedly achieves results comparable to surgery in the treatment of appropriately selected patients [8, 10]. For these reasons, despite an increase in incidence of EP in recent years, morbidity and mortality have substantially decreased [11].
Some factors related to the patient and the disease itself may affect the therapeutic outcome, including the size of the GS, the presence or the absence of embryonic cardiac activity, pretreatment serum β-hCG level, or other biomarkers [8, 12]. For the detection of an ectopic GS, the selection of the appropriate imaging modality is necessary.
TVUS is the imaging modality of choice for diagnosing EP and the sonographic findings of EP are well-described [6, 13, 14]. TVUS has high accuracy for the diagnosis of an EP with a sensitivity of 69%–99% and a specificity of 84%–99.9% [5]. Sonographic diagnosis is only definite when a live embryo is detected in an extrauterine location; however, this finding is rare. An extrauterine live embryo (100% specificity), the most specific finding of EP, is known to lack sensitivity [13]. GS findings on US are also known to have a PPV 100%; however, they also have low sensitivity (26%) [15]. The combination of intrauterine finding (the lack of normal intrauterine pregnancy) and extrauterine findings (i.e., cystic or solid adnexal mass, dilated and thick-walled fallopian tube, echogenic peritoneal free fluid, and extra-uterine GS with a yolk sac and with or without an embryo) have diagnostic value [16]. In our institute, the first choice for diagnosis of an EP is TVUS and all patients undergo TVUS by gynecologists. MRI is applied for EP patients with an unclear implantation site on TVUS.
Some previous studies reported the utility of MR imaging when the results of TVUS are insufficient or inconclusive, especially for a specific diagnosis of a rare or complicated form of EP [4, 5]. Reported MRI findings of EP include GS-like structure, an adnexal or abdominal hematoma, tubal dilatation due to hemosalpinx, and tubal wall enhancement (Figs. 2, 3). In these previous studies, the concrete MRI features of a GS were not discussed.
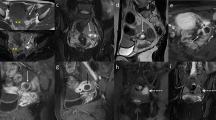
Right tubal pregnancy in a 30-year-old woman at 6 weeks’ gestation. A Axial T2-weighted MRI images a GS-like structure surrounded by a peripheral area of heterogeneous signal intensity (arrow). Dilated right tube of low signal intensity indicates hemosalpinx. B Axial T1-weighted MRI images pelvic fluid exhibit with a slightly increased intensity indicative of hemorrhagic ascites (asterisk). C Axial post-contrast T1-weighted MR image with fat suppression clearly demonstrates the GS with heterogeneous ring-like enhancement (arrow) associated with tubal wall enhancement (arrowhead).
Left tubal pregnancy in a 30-year-old woman at 7 weeks’ gestation. A Axial T2-weighted MRI images as hematoma of heterogeneous signal intensity (asterisk) surrounding the dilated left tube (arrow). GS-like cystic structure is unclear in this case. B Axial T1-weighted MRI images pelvic fluid with a slightly increased intensity indicative of hemorrhagic ascites (double asterisk). C Axial post-contrast T1-weighted MRI with fat suppression demonstrates thickened wall of a dilated tube (arrow).
In our study, the extrauterine GS was detected in 22 of 24 EP cases (92%) with high accuracy (92%). The size of the GS ranged from 10 to 42 mm and did not correlate with gestational weeks. This suggested that the GS of EP is usually mal-developed due to abnormal implantation. A GS-like structure is defined as sac-like cystic structure with low signal intensity on T1WI and high signal intensity on T2WI [4, 5]. In our study, a GS was detected as a round or oval image and exhibited low to iso-intensity on T1-weighted images (86%) and high intensity on T2-weighted (82%) (Fig. 4). The GS also includes an area of intermediate or high signal intensity on T1-weighed images and distinct low intensity on T2-weighed images, which are indicative of fresh hemorrhage [4].
Left tubal pregnancy in a 34-year-old woman at 7 weeks’ gestation. A Axial T2-weighted MRI images a GS-like structure surrounded by a peripheral area of heterogeneous low signal intensity (arrow). Hemosalpinx and hematoma; hemorrhagic ascites is unclear in this case. B Axial T1-weighted MRI images homogeneous hypointense thick wall of GS-like cystic structure (arrow). C Axial post-contrast T1-weighted MRI with fat suppression demonstrates the GS with a thick and heterogeneous ring-like enhancement (arrow). D Diffusion-weighted image demonstrates the GS with ring-like high signal intensity (arrow).
Although Yoshigi et al. [1] previously reported the utility of T2*-weighted imaging, a GS with low signal intensity on T2* weighted imaging was observed in only 50% of EP cases in our study. This may due to early stage cases of EP without hemorrhage. In contrast, a ring-like or dot-like high signal intensity on DWI was observed in 91% of EPs.
Contrast-enhanced MRI has also been reported to be useful for the diagnosis of EP because it demonstrates enhancement in the GS and the affected tubal wall [4]. The villi were reported to image as papillary solid components within the dilated tube [5]. In our study, enhancement patterns were divided into either ring-like or dot-like; these findings were observed in 15 of 16 GSs with contrast-enhanced study. When non-contrast images are indeterminate, contrast-enhanced images allow more accurate detection of a GS structure because they exhibit a thick, enhanced wall. The cystic wall of the GS typically exhibits high intensity on T2-weighed images; areas of distinct low signal intensity with marked dot-like enhancement are present (Figs. 4, 5). This structure reflects the trophoblastic tissue in the GS.
Right tubal pregnancy in a 36-year-old woman at 6 weeks’ gestation. Axial T2-weighted MRI images sac-like structure surrounded by a thick wall of homogeneous intermediate intensity indicative of a theca lutein cyst (arrowhead). GS- and sac-like structure surrounded by thick wall with spotty low signal intensity is located in the opposite side (arrow). Axial T1-weighted MRI images a homogeneously hypo intense thick wall (arrow). Axial post-contrast T1-weighted MRI with fat suppression clearly shows a difference in enhanced pattern between GS (arrow) and theca lutein cyst (arrowhead).
The differential diagnosis of a GS is a corpus luteum cyst (Fig. 5). A corpus luteum cyst is present in the ovary and typically has a thick wall exhibiting a slightly increased intensity on T1-weighted images and relatively low intensity on T2-weighted images. On TVUS, a corpus luteum images as a cystic structure with the ring of fire on Color Doppler imaging, which resembles the feature of EP. On MRI, the distinction may be facilitated by focusing on the signal intensity of the peripheral area, location (intra-ovarian or extra-ovarian), and enhancement pattern. A corpus luteum images as an intraovarian cystic-like structure with a thickened wall and homogeneous enhancement. Although a GS also images as a cystic-like structure, it usually locates extra-ovarian and the dot-like enhancement pattern is specific for a GS.
The finding of a dilated tubal wall is another key MRI finding; this is suspicious for a tubal pregnancy and may reflect increased vascularity of the tubal wall following implantation (Fig. 3). Hemorrhagic ascites, which typically exhibit higher signal intensity than that of the urine in the bladder on T1WI, has also been reported to be an indirect sign of EP. A small amount of hemorrhagic fluid can be observed as a localized adnexal hematoma and is not necessarily indicative of tubal rupture; furthermore, it has low sensitivity. Although these features are important signs of the EP, the sensitivity was not adequate in our study. The indication for a MRI for the diagnosis of an EP is a clinically unclear case. Therefore, the more obvious ruptured EP cases were excluded and these signs were not observed in the unruptured cases without hemorrhage.
The identification of an extrauterine GS is the most specific radiologic finding of EP [5]. In our study, a sac-like structure with thick, low signal intensity on T2WI was the most specific sign of EP when compared to indirect signs. When employing diagnostic criteria, which required the presence of either one direct or at least two indirect signs, accuracy increased to 100%. The combination of direct and indirect signs is, of course, important on both TVUS and MRI. Inasmuch as the majority of ectopic pregnancies are currently identified at an early stage in an unruptured condition recently [17], the detection of a GS is considered to be more important.
Our study was a retrospective one and contained a relatively small sample size. Based on the results of this study, we concluded that MRI is a feasible modality for diagnosing EP. It has a high detection rate of an extrauterine GS. When TVUS findings are indeterminate, MRI may provide superior delineation of the presence of an EP.
References
Yoshigi J, Yashiro N, Kinoshita T, O’Uchi T, Kitagaki H (2006) Diagnosis of ectopic pregnancy with MRI: efficacy of T2*-weighted imaging. Magn Reson Med Sci 5(1):25–32
Kinoshita T, Ishii K, Higashiiwai H (1999) MR appearance of ruptured tubal ectopic pregnancy. Eur J Radiol 32(2):144–147
Ha HK, Jung JK, Kang SJ, et al. (1993) MR imaging in the diagnosis of rare forms of ectopic pregnancy. AJR Am J Roentgenol 160(6):1229–1232
Kataoka ML, Togashi K, Kobayashi H, et al. (1999) Evaluation of ectopic pregnancy by magnetic resonance imaging. Hum Reprod 14(10):2644–2650
Tamai K, Koyama T, Togashi K (2007) MR features of ectopic pregnancy. Eur Radiol 17(12):3236–3246
Atri M, Leduc C, Gillett P, et al. (1996) Role of endovaginal sonography in the diagnosis and management of ectopic pregnancy. Radiographics 16(4):755–774 ((discussion 75))
Lubner M, Menias C, Rucker C, et al. (2007) Blood in the belly: CT findings of hemoperitoneum. Radiographics 27(1):109–125
Skubisz MM, Tong S (2012) The evolution of methotrexate as a treatment for ectopic pregnancy and gestational trophoblastic neoplasia: a review. ISRN Obstet Gynecol 2012:637094
Banz C, Chalvatzas N, Kelling K, et al. (2010) Laparoscopic management of ectopic pregnancy during a 9-year period. Fertil Steril 94(7):2780–2782
Farquhar CM (2005) Ectopic pregnancy. Lancet 366(9485):583–591
Luciano DE, Jain A, Roy G, Solima E, Luciano AA (2004) Ectopic pregnancy—from surgical emergency to medical management. J Am Assoc Gynecol Laparosc 11(1):107–121 (quiz 22)
Rabischong B, Tran X, Sleiman AA, et al. (2011) Predictive factors of failure in management of ectopic pregnancy with single-dose methotrexate: a general population-based analysis from the Auvergne Register, France. Fertil Steril 95(1):401-404, 4 e1.
Levine D (2007) Ectopic pregnancy. Radiology 245(2):385–397
Laing FC (1990) Sonographic determination of tubal rupture in patients with ectopic pregnancy: is it feasible? Radiology 177(2):330–331
Tongsong T, Pongsatha S (1993) Transvaginal sonographic features in diagnosis of ectopic pregnancy. Int J Gynaecol Obstet 43(3):277–283
Pham H, Lin EC (2007) Adnexal ring of ectopic pregnancy detected by contrast-enhanced CT. Abdom Imaging 32(1):56–58
Nishino M, Hayakawa K, Kawamata K, Iwasaku K, Takasu K (2002) MRI of early unruptured ectopic pregnancy: detection of gestational sac. J Comput Assist Tomogr 26(1):134–137
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takahashi, A., Takahama, J., Marugami, N. et al. Ectopic pregnancy: MRI findings and clinical utility. Abdom Imaging 38, 844–850 (2013). https://doi.org/10.1007/s00261-012-9969-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-012-9969-0