Abstract
Purpose
Dual-tracer, 18F-fluorodeoxyglucose and 18F-fluorodeoxythymidine (18F-FDG/18F-FLT), dual-modality (positron emission tomography and computed tomography, PET/CT) imaging was used in a clinical trial on differentiation of pulmonary nodules. The aims of this trial were to investigate if multimodality imaging is of advantage and to what extent it could benefit the patients in real clinical settings.
Methods
Seventy-three subjects in whom it was difficult to establish the diagnosis and determine management of their pulmonary lesions were prospectively enrolled in this clinical trial. All subjects underwent 18F-FDG and 18F-FLT PET/CT imaging sequentially. The images were interpreted with different strategies as either individual or combined modalities. The pathological or clinical evidence during a follow-up period of more than 22 months served as the standard of truth. The diagnostic performance of each interpretation and their impact on clinical decision making was investigated.
Results
18F-FLT/18F-FDG PET/CT was proven to be of clinical value in improving the diagnostic confidence in 28 lung tumours, 18 tuberculoses and 27 other benign lesions. The ratio between maximum standardized uptake values of 18F-FLT and 18F-FDG was found to be of great potential in separating the three subgroups of patients. The advantage could only be obtained with the full use of the multimodality interpretation. Multimodality imaging induced substantial change in clinical management in 31.5% of the study subjects and partial change in another 12.3%.
Conclusion
Multimodality imaging using 18F-FDG/18F-FLT PET/CT provided the best diagnostic efficacy and the opportunity for better management in this group of clinically challenging patients with pulmonary lesions.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Modern medical imaging techniques like computed tomography (CT), magnetic resonance imaging (MRI), ultrasound and 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography (PET) play an important role in the clinical assessment and management of a wide variety of tumours [1]. However, because of the complex and unpredictable nature of the tumour biology, it is now recognized that combining different imaging modalities, in particular with radiolabelled probes other than 18F-FDG, each revealing different aspects of tumour biology, can be a better way of characterizing a tumour [2–6]. However, certain new questions arise along with multi-tracer imaging, such as whether the new tracers have an incremental value over or a potential to replace the classic 18F-FDG, or how much the information derived from those new tracers can be of real benefit to the patients. Therefore, we designed and conducted a prospective clinical trial in a group of patients with different pulmonary lesions in order to address the feasibility and clinical impact of dual-tracer, 18F-FDG and 18F-fluorodeoxythymidine (18F-FLT), and dual-modality (PET/CT) imaging. The questions to be answered in the current study include: (1) whether multimodality imaging using dual tracers and dual imaging (PET/CT) can improve the diagnosis of pulmonary lesions; (2) whether it is necessary to have all the modalities (i.e. whether any imaging modality could be omitted); and (3) whether and how multimodality imaging can affect the clinician’s decision and consequently influence the patients’ management.
Materials and methods
Subject enrolment and exclusion
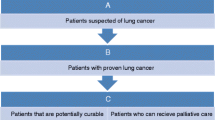
The patients enrolled in the current study were selected from consecutive patients with pulmonary lesions, who were referred to our PET/CT centre from either respiratory or thoracic surgery departments from January 2006 to July 2009. The criteria for including a subject in the study were: pulmonary lesion(s) was found by chest X-ray or CT; the nature of the lung lesion could not be elucidated from available imaging and clinical data upon admission; no previous history of lung disorder or any treatment directed toward a lung lesion; willing to join the trial and follow the protocol, provide relevant information with written consent; with physical and financial capability to complete the trial.
Some patients were excluded from entering the study due to the following reasons: (1) the diagnosis having been defined by other means before PET/CT imaging; (2) being in a critical situation or with metabolic abnormalities such as hyperglycaemia, which precluded the dual-tracer PET/CT study; (3) refusal or unable to provide relevant data; and (4) doubt concerning their compliance in the trial or their diagnosis during and at the end of follow-up. A few cases withdrawn from the trial due to various reasons were also excluded from the final analysis.
The clinical trial
The trial was designed in a prospective way. Once determined to participate in the trial, a code number was given to each subject to mask their real name for the entire period of the trial. Then all patients were imaged twice with 18F-FDG and 18F-FLT according to the standardized protocols. The images were immediately interpreted and reported by a group of nuclear medicine physicians and radiologists independently. The interpretations were provided to the referring doctors/surgeons, and an inquiry form about their clinical decision was collected by an independent organizer. Surgical procedures, operation or biopsy, documented therapeutic response or imaging/clinical data by a follow-up of at least 22 months served as the standard of truth in this trial. The image interpretation, diagnostic performance and the clinical decision were compared with the standard of truth. The diagnostic contribution of the imaging modalities and their impact on the clinical decision making were retrospectively analysed.
The imaging protocols
The order of PET/CT imaging using either 18F-FDG or 18F-FLT was randomly determined. The second PET/CT imaging with the other radiotracer was preformed within 7 days after the first imaging. The radiopharmaceuticals were synthesized with the same type of synthesizer (TRACERlab FX F-N, GE Healthcare, Waukesha, WI, USA) following the standard operating procedure (SOP). Patients were required to fast for at least 4 h prior to each PET/CT scan. The images were acquired 60 min after the administration of 300 ∼ 400 MBq radiotracer with a similar model of PET/CT scanner (Discovery ST, GE Healthcare, Waukesha, WI, USA). A low-dose CT (LDCT) scan was undertaken at 120 kV, ∼100 mAs, 0.8 s rotation with a 1.25-mm slice width. Pitch was 0.9. PET data were acquired in 3-D mode for 2.5 min/bed and three or seven bed positions, covering the entire chest or the trunk from the bottom of the pelvis to the chin. The PET images were reconstructed by the Fourier rebinning (FORE) ordered subset expectation maximization (OSEM) algorithm with attenuation correction.
Image interpretation
Four readers (two radiologists and two nuclear medicine physicians) participated in our trial. The images were randomly rebinned into seven sets and then read with different interpretation strategies, i.e. 18F-FDG, 18F-FLT, CT alone, combination of two image modalities and full multimodality, such as dual-tracer PET and CT altogether. The diagnosis was simply recorded as “M” (malignant) or “B” (benign). The CT images were displayed as 5-mm axial slices. The morphological features (such as the size, density, cavity, calcification, notched or speculated margin and plural contraction) of the nodule(s) were checked with the CT value measured. The uptake of 18F-FDG and 18F-FLT was assessed from a circular region of interest (ROI) over the entire lesion and expressed as the maximum standardized uptake value (SUVFDG and SUVFLT). A lesion was considered malignant if more than three morphological features were defined on CT, and SUVFDG was ≥ 2.5, SUVFLT was ≥ 1.4 and the ratio of SUVFLT to SUVFDG was between 0.4 and 0.9 on PET [7]. In cases of more than one lesion, the maximum values of SUVs among all lesions were chosen as representative. The results in this trial are presented and analysed based on subject rather than on lesion.
Inquiry form on clinical decision
In order to obtain a management plan for each patient, a standardized inquiry form was sent to the referring physician or surgeon at two time points. The first inquiry form was sent before the PET/CT imaging. The second form was sent after complete reading of the two PET/CT studies. The inquiry form contained three categories: “aggressive treatment” (including surgery, radiotherapy and cytotoxic chemotherapy); “conservative treatment” (including a regimen of anti-inflammation or anti-tuberculosis medication and clinical observation); and “unable to decide”. Under the former two categories, three more checkboxes were listed as “definitive”, “most likely” and “possible” to reflect the decision maker’s confidence in choosing the corresponding management. The inquiry form was immediately collected after its completion by the organizer and served as the basis for clinical impact analysis.
End-point of the trial
Once a subject finished his/her dual-tracer PET/CT imaging session, the patient was treated according to the clinician’s decision (such as surgery, anti-tumour, anti-tuberculosis, anti-inflammation or clinical observation without specific therapy). The trial on each case ended as having either the pathological evidence obtained from the surgical processes or the imaging or laboratory proof of his/her lesion’s nature, such as definite therapeutic response to a specific regimen. A lesion was considered benign if it remained unchanged in a 22-month or longer follow-up.
Statistics
Statistical analysis was conducted by SPSS 11.0 with a dedicated software package (MINITABLE for Six Sigma, GE Healthcare, Waukesha, WI, USA). The analysis was undertaken on the diagnostic performance of image interpretation strategies and the contribution from each imaging modality or combination of the modalities. A statistician took an active part in the design, data verification and final analysis in this trial.
Results
Subject demographic features
A total of 73 subjects with documented pathological or clinical evidence were collected during the trial period from January 2006 to July 2009. Table 1 shows the relevant information of those 73 subjects.
These subjects, 46 men and 27 women, aged 17–85, had a solitary pulmonary nodule (n = 42) or 2–3 lesions (n = 31), with the majority of lesions smaller than 30 mm (n = 46). The final diagnosis was confirmed by surgery (n = 42) and clinical evidence (n = 31), respectively. It was confirmed at the end of the trial that among the subjects 28 had lung cancer, 18 tuberculoses (TB) and 27 other benign lesions (inflammation, pseudotumour, granuloma and other benign conditions).
PET/CT imaging
Except for a small number of inflammatory cases, high uptake of 18F-FDG was observed in almost all lesions, while the uptake of 18F-FLT of those lesions was generally weaker. Although the image quality of LDCT was not as good as that of the ordinary diagnostic CT because of the lower tube current and free breathing during PET/CT acquisition, the image quality of the so-called LDCT was sufficient to reveal the major morphological features of most lesions. Therefore, the LDCT helped the PET image interpretation by providing more precise ROI localization and some morphological information suggesting the lesion’s nature.
In a quantitative analysis, both SUVFDG and SUVFLT in malignant tumours (8.32 ± 4.80, 3.38 ± 1.92) (Fig. 1) were higher than those of TB (6.71 ± 4.55, 1.99 ± 1.54) (Fig. 2) and inflammation (4.51 ± 3.74, 1.86 ± 1.52) (Fig. 3). Although analysis of variance (ANOVA) indicated a significant difference in those SUVs among all patient groups (F2,70 = 5.265, p = 0.007; and F2,70 = 6.507, p = 0.003), the Games-Howell test did not detect any significant difference in SUVFDG between tumour (8.32 ± 4.80) and TB (6.71 ± 4.55, p = 0.489). The difference of SUVFLT among tumour, TB and inflammation was significant (p = 0.006 ∼ 0.026) using the Games-Howell test, but not between TB and other benign lesions (p = 0.963).
The diagnostic performance of imaging modalities
As defined by the study protocol, the PET/CT images of each subject were immediately interpreted by the physicians and radiologists independently. When 18F-FDG PET images were read alone, the diagnostic sensitivity for lung tumour was 89.3%, while the specificity was as low as 26.7%. 18F-FLT PET alone had almost the same sensitivity as 18F-FDG, detecting 24 of 28 (85.7%) malignant tumours, and a little better specificity of 40%. The LDCT had almost the same diagnostic accuracy as 18F-FLT. When two imaging modalities were combined, i.e. 18F-FDG and 18F-FLT, or 18F-FDG and CT or 18F-FLT and CT together, the diagnostic performance improved, but was still worse than the full multimodality interpretation that provided the best sensitivity and specificity. Table 2 shows the diagnostic performance of different image interpretation strategies.
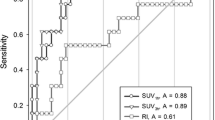
The three subgroups could be clearly distinguished by the SUVFLT to SUVFDG ratio (Fig. 4). Among 39 lesions with an SUVFLT to SUVFDG ratio lower than 0.40, 31 were benign and 8 malignant. All nine lesions with an SUVFLT to SUVFDG ratio over 0.90 were inflammatory. Among 25 lesions with an SUVFLT to SUVFDG ratio between 0.40 and 0.90, 20 were malignant. The difference of the SUVFLT to SUVFDG ratio among the three groups was statistically significant (F2,70 = 4.348, p = 0.017).
The clinical impact
Since the inclusion criteria precluded any subject with definite diagnosis from entering into the trial, it was learned from the first inquiry form that most subjects had no clear management plan at coming to our PET/CT centre. When the dual-tracer PET/CT diagnosis was provided, the referring physicians/surgeons made substantial (cross category) changes in the clinical management plan in 23 (31.5%) of the subjects, i.e. from “unable” to “treatment” or from “aggressive” to “conservative” or vice versa. Partial changes (confidence change within the same category) in clinical decision, such as prolonged observation or enhanced medication, were made in another nine (12.3%) subjects.
In follow-up, most of the changes in clinical management were proven correct. However, in six patients, the decision alteration after PET/CT imaging was ultimately found to be incorrect. Clinical follow-up identified one tumour mistreated as TB, and surgical operation and clinical follow-up revealed that another five benign lesions were incorrectly treated as malignant tumours.
Discussion
A pulmonary nodule is a type of very common but clinically challenging disorder [7, 8]. In China as well as in many other countries, lung cancer is the number one life-threatening tumour among all adult malignancies. The limitations of 18F-FDG in the evaluation of lung tumours have been well documented [9]. Nonspecific uptake by other tissues resulted in a positive predictive value (PPV) of 18F-FDG PET in pulmonary tumours as low as 44.6% [8]. Since the complexity of the biological characteristics of tumours is being more comprehensively understood, systematic use of information from various investigations becomes mandatory in clinical oncology. It has been widely recognized that integration of morphological, functional and metabolic information derived from different imaging modalities, especially with additional radiotracers other than 18F-FDG, provides information concerning different aspects of tumour biology [2, 10, 11], such as tissue hypoxia [12], angiogenesis [13], hormone dependence [6], other altered function or metabolic pathways [14], thus drawing a greater interest in clinical practice. In the present study, we attempted to assess the feasibility of adding 18F-FLT to 18F-FDG PET/CT as multimodality imaging and the effect on the diagnosis, differentiation and therapy assistance in a group of patients with pulmonary lesions. Our data indicated that 18F-FDG was quite sensitive in detecting pulmonary malignant lesions, but the specificity (26.7%) was even lower than that of most previous reports. This was probably due to a higher percentage of TB cases among our study population. 18F-FLT is introduced as the surrogate tracer for in vivo assessment of DNA synthesis. It shows a good correlation with other molecular markers of the S-phase of the cellular cycle (such as Ki-67 and proliferating-cell nuclear antigen), and it provides insight with respect to tumour proliferation, staging and therapy planning and monitoring [15–18]. However, as indicated by the false-positive cases, 18F-FLT is neither as specific as initially expected, nor is it “related exclusively to malignant tumors” [19]. Previous studies have also pointed out the nonspecific 18F-FLT uptake in interstitial pneumonia [20], inflammatory cells [21] and germinal centre of activated lymph nodes [22]. CT alone provided very limited evidence for the differential diagnosis in the current study. However, it is worthy of mentioning that the inclusion criteria precluded any subject with typical radiological features from this trial, and no diagnostic CT (such as standard tube current setting, breath-holding and contrast enhancement) was employed in the current study. As a result, the diagnostic features of CT presented in this study might be underestimated. In the present study, we clearly demonstrated that integrating all the imaging modalities together could best meet the clinical desire for correct diagnosis and proper management. Table 2 shows that omitting any modality might reduce the overall diagnostic performance in our study population given the rather heterogeneous underlying disease states.
In comparison with other criteria, the SUVFLT to SUVFDG ratio was very useful in distinguishing the nature of the pulmonary pathologies. A ratio within 0.40–0.90 correctly depicted most tumours, and this finding is consistent with some previous studies [19–21]. This ratio is diagnostically valuable in differentiating both active TB and inflammation from tumour, which is hard to accomplish with either 18F-FDG or 18F-FLT alone, as evidenced by the false-positive results in our study occurring in quite a number of TB and inflammatory lesions. However, most of our TB and inflammations could be easily distinguished from lung tumours using the SUVFLT to SUVFDG ratio. It remains unclear why there is a fixed SUVFLT to SUVFDG ratio for malignancy. Regardless of the underlying biological reason, the difference between tumour and non-tumour could be identified only when dual tracers were used. The most impressive feature of the SUVFLT to SUVFDG ratio is that it is quantitative, independent of subjective judgments of the image readers. Given the proper drawing of the ROI, the ratio can be objectively calculated, which might make the diagnostic parameter more promising in the future computer-assisted diagnostic system [23].
The improvement in diagnostic accuracy of multimodality imaging had consequently introduced better clinical care for our patients. We demonstrated that nearly half of our patients had their original management altered with additional information provided by the dual-tracer PET/CT imaging. In a prospective study with a larger scale investigation, Hillner et al. also found that FDG PET is associated with a 36.5% change in the treatment or no-treatment decision [24]. But it should be mentioned that in our trial, the impact on the clinical decision was achieved after dual-tracer imaging on the basis of any single tracer, either FDG or FLT. The difference might indicate further improvement of the clinical confidence in making a proper decision. Moreover, it might highlight our previous notion that complementary information from all available sources is necessary for the best diagnostic performance, at least in clinically difficult cases as in our cohort of subjects. More significantly, the alteration of management in the majority of those 43.8% patients was proven correct and brought proper and better clinical outcome for most of them. It could be more meaningful if the long-term follow-up, in particular the survival issue, is investigated, but unfortunately the observation was not included in the current study.
With dual-tracer imaging, the increased radiation dose should be considered. For a whole-body FLT scan, the patient received about 11.47 mSv radiation, similar to that of FDG (10.73 mSv) [25]. Added together with the irradiation dose from LDCT, the patient would totally receive about 28–30 mSv, which was still acceptable, considering the clinical gain for those highly selected, clinically difficult cases. However, under real clinical conditions, considering the radiation dose and the cost, we highly recommend that the second tracer, 18F-FLT, be restricted to those patients who are still difficult to diagnose and treat after the first-line 18F-FDG PET/CT has been completed.
In conclusion, the questions posed at the beginning of this article could be addressed as follows: (1) 18F-FDG and 18F-FLT, PET and CT are complementary to one another, providing information concerning different aspects of lesion biology. Therefore, the integration of multimodality imaging indeed enhances the diagnostic confidence for lung diseases. (2) The diagnostic gains of multimodality imaging are further supported by their positive influence on clinical decision making. Nevertheless, the conclusions should be treated with caution because of the limited number of incorrect decisions and the rather heterogeneous composition of our study subjects. (3) The diagnostic gains could be achieved only when dual tracers (metabolism and proliferation) and dual techniques (functional probes and morphological imaging) were fully used. (4) The benefit brought by multimodality imaging for our study subjects is real, but further studies in a bigger cohort with longer follow-up of survival, cost-effectiveness and radiation burden are needed in future investigations.
Conclusion
In the current randomized prospective clinical trial including 73 subjects with heterogeneous pulmonary disorders, dual-tracer (18F-FDG and 18F-FLT) and dual-modality (PET and CT) imaging improved the diagnostic performance. In this small group of subjects, the improvement could only be obtained when the modalities were fully used, and the improvement was further demonstrated via a positive impact on the clinical decision making. The technique and the diagnostic criteria established in the current study warrant further verification and application.
References
Newsline. Molecular imaging in the spotlight at RSNA 2007. J Nucl Med 2008;49(1):19N.
Rajendran JG, Mankoff DA. Beyond detection: novel applications for PET imaging to guide cancer therapy. J Nucl Med 2007;48(6):855–6.
van Waarde A, Cobben DC, Suurmeijer AJ, Maas B, Vaalburg W, de Vries EF, et al. Selectivity of 18F-FLT and 18F-FDG for differentiating tumor from inflammation in a rodent model. J Nucl Med 2004;45(4):695–700.
Ho CL, Chen SR, Yeung DWC, Cheng TK. Dual-tracer PET/CT imaging in evaluation of metastatic hepatocellular carcinoma. J Nucl Med 2007;48:902–9.
Rischin D, Hicks RJ, Fisher R, Binns D, Corry J, Porceddu S, et al. Prognostic significance of [18F]-misonidazole positron emission tomography-detected tumor hypoxia in patients with advanced head and neck cancer randomly assigned to chemoradiation with or without tirapazamine: a substudy of Trans-Tasman Radiation Oncology Group Study 98.02. J Clin Oncol 2006;24:2098–104.
Linden HM, Stekhova SA, Link JM, Gralow JR, Livingston RB, Ellis GK, et al. Quantitative fluoroestradiol positron emission tomography imaging predicts response to endocrine treatment in breast cancer. J Clin Oncol 2006;24:2793–9.
Tian JH, Yang XF, Yu LJ, Chen P, Xin J, Ma L, et al. A multicenter clinical trial on the diagnostic value of dual-tracer PET/CT in pulmonary lesions using 3′-deoxy-3′-18F-fluorothymidine and 18F-FDG. J Nucl Med 2008;49(2):186–94.
Bunyaviroch T, Coleman RE. PET evaluation of lung cancer. J Nucl Med 2006;47(3):451–69.
Strauss LG. Fluoro-18 deoxyglucose and false-positive results: a major problem in the diagnostics of oncological patients. Eur J Nucl Med 1996;23:1409–15.
Schöder H, Yeung HWD, Larson SM. CT in PET/CT: essential features of interpretation. J Nucl Med 2005;46:1249–51.
Kim SK, Allen-Auerbach M, Goldin J, Fueger BJ, Dahlbom M, Brown M, et al. Accuracy of PET/CT in characterization of solitary pulmonary lesions. J Nucl Med 2007;48:214–20.
Beck R, Röper B, Carlsen JM, Huisman M, Lebschi J, Andratschke N, et al. Pretreatment 18F-FAZA PET predicts success of hypoxia-directed radiochemotherapy using tirapazamine. J Nucl Med 2007;48:973–80.
Hsu AR, Cai W, Veeravagu A, Mohamedali KA, Chen K, Kim S, et al. Multimodality molecular imaging of glioblastoma growth inhibition with vasculature-targeting fusion toxin VEGF121/rGel. J Nucl Med 2007;48:445–54.
Wagnar HN. From molecular imaging to molecular medicine. J Nucl Med 2006;47(7):13N–39.
Vesselle H, Grierson J, Muzi M, Pugsley JM, Schmidt RA, Rabinowitz P, et al. In vivo validation of 3′deoxy-3′-[(18)F]fluorothymidine ([(18)F]FLT) as a proliferation imaging tracer in humans: correlation of [(18)F]FLT uptake by positron emission tomography with Ki-67 immunohistochemistry and flow cytometry in human lung tumors. Clin Cancer Res 2002;8(11):3315–23.
Grierson JR, Shields AF. Radiosynthesis of 3′-deoxy-3′-[(18)F]fluorothymidine: [(18)F]FLT for imaging of cellular proliferation in vivo. Nucl Med Biol 2000;27(2):143–56.
Reske SN, Deisenhofer S. Is 3′-deoxy-3′-(18)F-fluorothymidine a better marker for tumour response than (18)F-fluorodeoxyglucose? Eur J Nucl Med Mol Imaging 2006;33 Suppl 1:38–43.
Wieder HA, Geinitz H, Rosenberg R, Lordick F, Becker K, Stahl A, et al. PET imaging with [18F]3′-deoxy-3′-fluorothymidine for prediction of response to neoadjuvant treatment in patients with rectal cancer. Eur J Nucl Med Mol Imaging 2007;34(6):878–83.
Buck AK, Halter G, Schirrmeister H, Kotzerke J, Wurziger I, Glatting G, et al. Imaging proliferation in lung tumors with PET: 18F-FLT versus 18F-FDG. J Nucl Med 2003;44(9):1426–31.
Yap CS, Czernin J, Fishbein MC, Cameron RB, Schiepers C, Phelps ME, et al. Evaluation of thoracic tumors with 18F-fluorothymidine and 18F-fluorodeoxyglucose-positron emission tomography. Chest 2006;129:393–401.
Yamamoto Y, Nishiyama Y, Ishikawa S, Nakano J, Chang SS, Bandoh S, et al. Correlation of 18F-FLT and 18F-FDG uptake on PET with Ki-67 immunohistochemistry in non-small cell lung cancer. Eur J Nucl Med Mol Imaging 2007;34(10):1610–6.
Troost EGC, Vogel WV, Merkx MAW, Slootweg PJ, Marres HAM, Peeters WJM, et al. 18F-FLT PET does not discriminate between reactive and metastatic lymph nodes in primary head and neck cancer patients. J Nucl Med 2007;48(5):726–35.
Porenta G. Being right for the right reason: better than just being right? J Nucl Med 2007;48(3):335–6.
Hillner BE, Siegel BA, Liu D, Shields AF, Gareen IF, Hanna L, et al. Impact of positron emission tomography/computed tomography and positron emission tomography (PET) alone on expected management of patients with cancer: initial results from the National Oncologic PET Registry. J Clin Oncol 2008;26(13):2155–61.
Vesselle H, Grierson J, Peterson LM, Muzi M, Mankoff DA, Krohn KA. 18F-Fluorothymidine radiation dosimetry in human PET imaging studies. J Nucl Med 2003;44(9):1482–8.
Acknowledgements
This work was partly supported by grants from the National Natural Science Foundation of China (30770607). The contributions of Drs. Zhoushe Zhao and Shuang Wang of GE, China, and all staff in the imaging centre are deeply appreciated.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xu, B., Guan, Z., Liu, C. et al. Can multimodality imaging using 18F-FDG/18F-FLT PET/CT benefit the diagnosis and management of patients with pulmonary lesions?. Eur J Nucl Med Mol Imaging 38, 285–292 (2011). https://doi.org/10.1007/s00259-010-1625-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-010-1625-8