Abstract
Purpose
There is currently no single modality for accurate characterization of enlarged mediastinal lymph nodes into benign or malignant. Recently 18F-fluorothymidine (FLT) has been used as a proliferation marker. In this prospective study, we examined the role of 18F-fluorodeoxyglucose (18F-FDG) positron emission tomography/computed tomography (PET/CT) and 18F-FLT PET/CT in categorizing mediastinal lymph nodes as benign or malignant.
Materials and methods
A total of 70 consecutive patients with mediastinal lymphadenopathy detected on computed tomography (CT) or chest radiograph underwent whole body 18F-FLT PET/CT and 18F-FDG PET/CT (within 1 week of each other). Lymph nodal tracer uptake was determined by calculation of standardized uptake value (SUV) with both the tracers. Results of PET/CT were compared with histopathology of the lymph nodes.
Results
Histopathology results showed thirty-seven patients with sarcoidosis, seven patients with tuberculosis, nine patients with non-small cell lung cancer, five patients with Hodgkin’s lymphoma and twelve patients with non-Hodgkin’s lymphoma. The mean FDG SUVmax of sarcoidosis, tuberculosis, Hodgkin’s and non-Hodgkin’s lymphoma was 12.7, 13.4, 8.2, and 8.8, respectively, and the mean FLT SUVmax was 6.0, 5.4, 4.4, and 3.8, respectively. It was not possible to characterize mediastinal lymphadenopathy as benign or malignant solely based on FDG SUVmax values (p > 0.05) or FLT SUVmax values (p > 0.05). There was no significant difference in FDG uptake (p > 0.9) or FLT uptake (p > 0.9) between sarcoidosis and tuberculosis. In lung cancer patients, the FDG SUVmax and FLT SUVmax of those lymph nodes with tumor infiltration on biopsy was 6.7 and 3.9, respectively, and those without nodal infiltration was 6.4 and 3.7, respectively, and both the tracers were not able to characterize the nodal status as malignant or benign (p > 0.05).
Conclusion
Though 18F-FLT PET/CT and 18F-FDG PET/CT reflect different aspects of biology, i.e., proliferation and metabolism, respectively, neither tracer could provide satisfactory categorization of benign and malignant lymph nodes. The results of this study clearly suggest that differentiation of mediastinal nodes into benign and malignant solely based on SUVmax values cannot be relied upon, especially in settings where tuberculosis and sarcoidosis are common.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Mediastinal lymphadenopathy (MNL) can occur from a wide range of pathologies. Common benign conditions include sarcoidosis and granulomatous infectious conditions including tuberculosis, histoplasmosis, coccidioidomycosis, and others [1–3]. Malignant causes include lymphoma and metastatic nodal disease. Thus, MNL comprises a broad spectrum of diseases including entities for which immediate therapy may not be required, e.g., sarcoidosis. However, for malignant conditions such as lymphoma, the outcome may be considerably influenced by timely initiation of specific treatment. Hence, differentiating between malignant and benign lymph nodes is of considerable clinical importance. Currently, there exists no ideal imaging modality and neither anatomical nor functional imaging can accurately characterize lymph nodes. Computed tomography (CT) and magnetic resonance imaging (MRI) are the most widely used modalities for evaluation of mediastinal lymph nodes but both have low sensitivity and specificity in detecting malignant nodes [4–7]. Further, the size of lymph nodes is not a reliable criterion [8].
The accurate diagnosis of benign and malignant lymph nodal involvement requires invasive procedures including transthoracic needle aspirate (TTNA), transbronchial needle aspirate (TBNA), endobronchial ultrasound guided TBNA (EBUS-TBNA), which are not free of complications [9–12]. There is therefore a need for non-invasive methods that can accurately differentiate between benign and malignant lymphadenopathy.
Positron emission computed tomography/computed tomography (PET/CT) has been found to be a good tool for the staging of lung cancer. Although the negative predictive value of 18F-fluorodeoxyglucose (FDG) PET/CT is high, the specificity to differentiate benign from malignant lymph nodal involvement is low [13]. 18F-fluorothymidine (FLT) PET/CT was introduced as the surrogate tracer for in vivo assessment of DNA synthesis. It shows a good correlation with other molecular markers of the S-phase of the cellular cycle and thus is thought to be tumor specific as shown by different studies [14]. FLT has been recently used to image proliferation in different malignant conditions [15]. Being a biomarker for proliferation FLT have higher specificity, accuracy and, positive predictive value than FDG for nodal staging in non-small cell lung cancer patients [16, 17]. Utility of this tracer has been studied for characterization of pulmonary nodules, in which malignant nodules showed a higher uptake value compared to benign lung nodules [18]. For cervical nodal metastases evaluation in head and neck cancers, FLT has been found to have a lesser false positive involvement as compared to FDG and hence less over-staging of disease [19]. FLT is reported to be superior to FDG for overall positive and negative predictive values for detection of nodal metastases in some cancers in head and neck cancers [20]. In colorectal cancers, FLT has been found to be at par with FDG to detect nodal disease lymph nodal metastasis [21]. In this study, we evaluate the ability of 18F-FDG PET/CT and 18F-FLT PET/CT to characterize the mediastinal lymph nodes as either benign or malignant.
Materials and methods
This was a prospective study conducted between July 2012 and June 2014. The protocol was approved by the Institutional Ethics Committee and a written informed consent was obtained from all patients. Consecutive treatment and diagnosis naïve patients with mediastinal lymphadenopathy were included in the study. Patients with pregnancy, uncontrolled hyperglycemia, and uncorrected coagulopathy were excluded. Since this was a prospective study, the pathological status of the lymph node as to whether benign or malignant was not known and biopsy was performed after the PET scans.
18F-FDG and 18F-FLT synthesis
18F-FDG and 18F-FLT were synthesized in-house by standard procedures using reagent kits (ABX, Germany) in the institute cyclotron facility (Pettrace 4, 16.5 meV, GE Healthcare, USA).
PET imaging protocols
Whole body (base of skull to mid thigh) 18F-FDG PET/CT scan followed by regional (thorax only) 18F-FLT PET/CT were performed in all patients using a dedicated hybrid PET/CT scanner (Discovery STE-16, GE Healthcare, Milwaukee, USA). Both the investigations were performed within 7 days interval. For 18F-FDG PET/CT imaging, patients fasted for at least 4 h and blood glucose level was ensured to be <150 mg/dl. Images were acquired 45–60 min post intravenous administration of 370 MBq F-18FDG. CT acquisition parameters were120 kV, 350 mA, rotation time of 0.5 s, and slice thickness of 3.75 mm, 512 × 512 pixels matrix and pixel size of about 1 mm. PET acquisition was done using 128 × 128 pixels matrix with a slice thickness of 3.25 mm. CT based attenuation correction of the emission images was employed. 18F-FLT PET/CT imaging was performed 45–60 min after intravenous administration of 370 MBq of 18 F-FLT. For 18F-FLT PET/CT, low dose (20 mA) current was used in CT acquisition to minimize radiation exposure.
Image evaluation
Data obtained from both the studies was reconstructed using iterative reconstruction, ordered subset expectation maximization (OSEM). Transaxial, sagittal, and coronal images were generated after reconstruction and evaluated qualitatively for positive findings. Images were interpreted visually by two experienced nuclear medicine physicians on per patient basis. Circular regions of interest (ROI) 0.9–1 cm diameter were placed on areas with increased tracer uptake corresponding to lymph nodes on CT. SUVmax values were calculated from each region of interest.
Disease diagnosis
All patients had a histopathologic/cytologic examination of specimen obtained from the lymph nodes. This served as the gold standard as to whether the lymph nodes were benign or malignant against which the results of PET/CT was compared. Patients who had peripherally enlarged lymph nodes along with mediastinal lymphadenopathy had FNAC/excision biopsy of the accessible peripheral lymph node to determine the pathology. Same pathology of peripheral LN and the mediastinal LN was considered, as the enlarged mediastinal and the biopsied peripheral lymph node showed a similar value of FDG and FLT uptake.
In case where peripheral lymph nodes were not accessible or when peripherally sampled nodes did not yield a diagnosis, patients underwent flexible bronchoscopy and TBNA, either conventional or EBUS-guided. Endobronchial biopsy (EBB)/transbronchial lung biopsy (TBLB) and broncho-alveolar lavage (BAL) samples were obtained as and when indicated. Specimens (both EBB and TBLB) were fixed with formalin and stained with hematoxylin and eosin for morphology and Ziehl–Neelsen stain for mycobacteria. Patients with benign lymph nodes showing granulomas on cytology/biopsy were all followed up for 6 months after initiation of appropriate therapy.
Histopathologic samples containing malignant cells or lymphomatous cells were considered diagnostic of malignancy and lymphoma, respectively. The final diagnosis of lymphoma and its type was based on the histopathology and immuno-histochemistry of lymph nodes. Staining for Cyclin D1 positivity was done in those cases if the histological pattern was suspicious for mantle cell lymphoma. A final diagnosis of sarcoidosis was made on the presence of all the following criteria: (a) clinical features of sarcoidosis along with mediastinal lymphadenopathy; (b) demonstration of non-necrotizing granulomas on either TBNA, TBLB or EBB along with negative acid-fast bacilli and fungal stains, and no growth of mycobacteria on cultures; and (c) clinical and radiological response after treatment with glucocorticoids. A diagnosis of tuberculosis was based on the demonstration of all the following: (a) necrotizing granulomatous inflammation or presence of acid-fast bacilli (AFB) on microscopy or a positive culture for Mycobacterium tuberculosis; and, (b) clinic-radiological response to anti-tuberculosis treatment.
Statistical analysis
Qualitative and quantitative data were compared using Chi-square test, student’s T-test (or Mann–Whitney U test), as appropriate. Data are presented as mean ± standard deviation. A p value <0.05 was considered as significant.
Results
Subject demographic features
A total of 70 patients (43 males and 27 females, age range 23–58 years) with mediastinal lymphadenopathy were included. Table 1 shows the demographic and relevant clinical information of those subjects. A final diagnosis of benign and malignant lymph nodal enlargement was made in 50 and 20 patients, respectively, based on the histopathology from biopsy samples. Among the benign lymph nodal pathologies, 37 were sarcoidosis, seven were tuberculosis and six were lung cancer patients with no tumor infiltration into lymph nodes. Among the malignant nodal cases, five were Hodgkin’s lymphoma and 12 were non-Hodgkin’s lymphoma and three lung cancer patients with malignant nodal involvement.
18F-FDG PET/CT and 18F-FLT PET/CT imaging
The measured size of the lymph nodes in all the disease were all greater than 1.0 cm in short axis diameter. In a quantitative analysis, the FDG SUVmax of the sarcoidosis nodes ranged from 4.6 to 27.0 with a mean ± standard deviation of 12.7 ± 5.5 and FLT SUVmax of the sarcoid nodes ranged from 1.8 to 14.8 with a mean ± standard deviation of 6.0 ± 2.5 (Fig. 1). The FDG SUVmax of tubercular lymph nodes ranged from 9.0 to 17.6 with a mean ± standard deviation of 13.4 ± 2.6 and the FLT SUVmax of the tubercular nodes ranged from 4.0 to 6.3 with a mean ± standard deviation of 5.4 ± 0.7 (Fig. 2). The Tukey HSD test did not reveal any significant difference in the SUVmax of FDG (p value = 0.9) or SUVmax FLT significant (p value = 0.9) between sarcoidosis and tuberculosis.
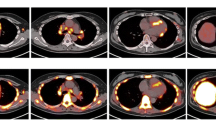
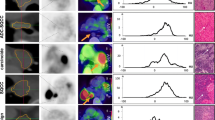
Transaxial fused 18F-FDG PET/CT (a) and 18F-FLT PET/CT (b), CT only (c), FDG PET only (d) and FLT PET only (e) showing increased tracer uptake in paratracheal lymph nodes in a sarcoidosis patient. Lymph node biopsy shows a well formed epithelioid cell granuloma with Langhan type cell giant cells (H n E ×400) (f). The lymph nodal SUVmax FDG was 16.3 and SUVmax FLT was 6.0
Transaxial fused 18F-FDG PET/CT (a) and 18F-FLT PET/CT (b), CT only (c), FDG PET (d) and FLT PET (e) showing increased tracer uptake in subcarinal and bilateral hilar lymph nodes in a tuberculosis patient. Biopsy of lymph node showing necrosis and epithelioid cells (H n E ×400) (f). The subcarinal lymph node has SUVmax FDG 14.4 and SUVmax FLT 4.7
The mean SUVmax of the nodes with Hodgkin’s lymphoma (Fig. 3) for FDG and FLT was 8.2 ± 3.1 and 4.4 ± 2.1, respectively. Similarly, mean SUVmax of the nodes with non-Hodgkin’s lymphoma (Fig. 4) was 8.8 ± 4.0 for FDG and 3.8 ± 1.9 for FLT (range 1.8–7.1). The SUVmax of FDG and FLT to differentiate between the Hodgkin’s lymphoma, non-Hodgkin’s lymphoma and malignant lymph nodal involvement in lung cancer patients was statistically insignificant (p value >0.05).
Transaxial fused 18F-FDG PET/CT (a) and 18F-FLT PET/CT (b), FDG PET (c) and FLT PET (d) showing increased tracer uptake in paratracheal and aorto-pulmonary (AP) window lymph nodes in a Hodgkin lymphoma patient. Lymph node biopsy shows lymphocytes, few eosinophils, occasional plasma cells and larger, Reed-Sternberg cells interspersed in between (H n E, ×200) (e) CD30 positive Reed-Sternberg cells (anti CD30, ×200) (f). The aorto-pulmonary window node has SUVmax FDG 4.9 and SUVmax FLT 3.8
Transaxial fused 18F-FDG PET/CT (a) and 18F-FLT PET/CT (b), FDG PET only (c) and FLT PET only (d) showing increased tracer uptake in bilateral hilar lymph nodes in a Mantle cell lymphoma patient. Lymph node biopsy showing sheets of lymphoma cells (H and E, ×200) (e): Cyclin D1 positive lymphoma cells (anti cyclin D1, ×200) (f) The right hilar lymph node has SUVmax FDG was 6.0 and SUVmax FLT 4.0
In NSCLC patients mean SUVmax of the nodes without infiltration was 6.4 ± 4.4 (FDG) and 3.7 ± 2.9 (FLT). In patients, mean SUVmax of the nodes with infiltration from NSCLC was 6.7 ± 3.3 and 3.9 ± 0.8 for FDG and FLT, respectively. Further, neither FDG SUVmax nor FLT SUVmax was able to differentiate between six patients who had benign nodal enlargement and three patients who had malignant lymph nodal involvement (Figs. 5, 6).
Transaxial fused 18F-FDG PET/CT (a) and 18F-FLT PET/CT (b), CT only (c), FDG PET only (d) and FLT PET only (e) showing increased tracer uptake in paratracheal lymph node in a lung cancer patient. Transbronchial needle aspirate from the lymph node shows clusters of malignant cells with features of adenocarcinoma (Papanicolaou stain, ×200) (f). The paratracheal lymph node has SUVmax FDG 9.0 and SUVmax FLT 4.4
Discussion
In this clinical study, we directly compared two tracers, 18F-FDG PET and 18F-FLT reflecting metabolism and proliferation, respectively, in the characterization of mediastinal lymph nodes into malignant and benign pathologies. It would be of great benefit to patients if an imaging modality could characterize enlarged lymph nodes in the mediastinum into benign or malignant. Although, high uptake of FDG has been found in malignant lymph nodes, it is not tumor specific. A study conducted to evaluate mediastinal lymph nodes using 18F-FDG PET/CT scan showed that in benign disease like tuberculosis, the SUVmax ranged from 2.3 to 11.8 with a mean ± SD of 5.3 ± 3.4 [22].
Factors responsible for inaccuracies in staging the mediastinal nodes in lung cancer by 18F-FDG PET/CT include adenomatous hyperplasia, rheumatoid arthritis, pneumonia [23]. Hence, histopathology is needed in 18F-FDG PET/CT positive cases [24]. In a study using 18F-FDG PET/CT, Kumar et al. reported mean SUVmax of 4.6 in lymph nodes of patients with sarcoidosis [22] and with a cut off value of SUVmax of 6.2 found FDG to be 87 % sensitive and 70 % specific for differentiation of malignant and benign lymph nodes. Our study had high mean FDG SUVmax value of sarcoid lymph nodes of 12.7 a value consistent with other studies [25–28] thus proving that FDG may not be ideal for such differentiation. This difference in FDG uptake with their study might be attributed to small number of patients with sarcoidosis in their study. FLT is considered as a marker of malignancy and is correlates with the cell proliferation as shown by monoclonal antibody Ki67, which binds to nuclear antigens expressed by cells in the GI, G2, M, and S proliferative phases and theoretically uptake should be more in case of malignant disease.
The uptake of FLT was also high in cases of sarcoidosis and tuberculosis in the present study. The high FLT uptake in sarcoidosis could be attributed to high proliferation of lymphocytes and macrophages around granulomas in the lymph nodes [29, 30]. The high FLT uptake in sarcoidosis due to increased proliferation in our study would have been further validated if immune-staining with Ki-67 was done. A recent study showed high 18F-FLT PET/CT accumulation in pyogenic abscess site indicating that 18F-FLT is not a specific tumor tracer, since active inflammation also resulted in the uptake of this compound indicating that proliferation, irrespective whether tumor related or bacterial infection related leads to high accumulation of 18 F-FLT [31]. The same study group showed that the due to the accelerated DNA synthesis in the bacterial reproductive period the FLT uptake is high but during the non reproductive or the period of chronic bacterial inflammation the 18F-FLT uptake is low. Therefore, granulomatous lesions with mycobacterium tuberculosis may exhibit different FLT SUVmax values as seen in our study, ranging from 4 to 6.3. Though FLT uptake has been reported in histologically sarcoid like granulomas [32], studies with large number of diagnosis proven sarcoidosis patients is lacking and our study has shown considerable nodal FLT uptake in such patients with mean SUVmax FLT of 6.0. The mean SUVmax of the nodes with Hodgkin’s lymphoma for FLT was 4.4 ± 2.1 in our study. This value is similar as reported by Wang et al. where 21 patients with Hodgkin’s disease where mean SUVmax value of 4.5 ± 1.9 was observed [33]. In patients with non-Hodgkin’s lymphoma our study showed mean SUVmax FLT of 3.8 ± 1.9 (range 1.8–7.1). Similar to this range, previous studies have reported FLT SUVmax levels in different non-Hodgkin’s lymphoma [33, 34] ranging from 1.3 to 8.1 in indolent lymphoma to 5.0–17.0 in aggressive non- Hodgkin’s lymphomas [34]. It is clear from the above observations of FLT uptake in tuberculosis, sarcoidosis, and lymphoma that FLT would not be an ideal tracer to characterize nodes in the mediastinum as benign or malignant.
In countries like India where sarcoidosis is increasingly being diagnosed and tuberculosis is highly prevalent [35], it becomes clear from the current study that the mean SUVmax values of neither FDG nor FLT can be relied upon for characterizing mediastinal lymph nodes into malignant and benign causes. Further, both the tracers cannot differentiate between the two benign granulomatous conditions.
Conclusion
In this prospective study, PET/CT using both18F-FLT PET/CT and 18F-FDG PET/CT could not accurately characterize malignant and benign lymph nodal enlargement. Though 18F-FLT and 18F-FDG PET reflect different aspects of biology i.e., proliferation and metabolism, neither tracer could provide satisfactory diagnosis. From the current study it is clear that differentiation of mediastinal nodes into benign and malignant etiology solely based on SUVmax values may not hold true and especially in countries like India where tuberculosis and sarcoidosis are common; high SUVmax values may not always reflect malignancy. It is clear from the current study that neither the SUVmax values of FDG nor FLT can be relied upon for differentiating granulomatous disease like sarcoidosis and tuberculosis. From our study the only advantage of FDG over 18F-FLT PET/CT would be that, 18F-FDG PET/CT produces high contrast images as compared to 18F-FLT PET/CT.
Limitation
In our study, sarcoidosis and tuberculosis were the diseases in the benign category both of which are granulomatous conditions and showed high 18-F FLT uptake. Our study did not have any non-granulomatous benign diseases. The uptake of 18F-FLT could be different in such cases, which needs further evaluation.
References
Goodwin RA, Shapiro JL, Thurman GH, Thurman SS, Des Prez RM. Disseminated histoplasmosis: clinical and pathologic correlations. Medicine (Baltimore). 1980;59(1):1–33.
Sagel SS. Common fungal diseases of the lungs. I. Coccidioidomycosis. Radiol Clin North Am. 1973;11(1):153–61.
Manns BJ, Baylis BW, Urbanski SJ, Gibb AP, Rabin HR. Paracoccidioidomycosis: case report and review. Clin Infect Dis. 1996;23(5):1026–32.
Petrillo R, Balzarini L, Bidoli P, Ceglia E, D’Ippolito G, Tess JD, et al. Esophageal squamous cell carcinoma: MRI evaluation of mediastinum. Gastrointest Radiol. 1990;15(4):275–8.
Zerhouni EA, Rutter C, Hamilton SR, Balfe DM, Megibow AJ, Francis IR, et al. CT and MR imaging in the staging of colorectal carcinoma: report of the Radiology Diagnostic Oncology Group II. Radiology. 1996;200(2):443–51.
Sohn KM, Lee JM, Lee SY, Ahn BY, Park SM, Kim KM. Comparing MR imaging and CT in the staging of gastric carcinoma. AJR Am J Roentgenol. 2000;174(6):1551–7.
Curtin HD, Ishwaran H, Mancuso AA, Dalley RW, Caudry DJ, McNeil BJ. Comparison of CT and MR imaging in staging of neck metastases. Radiology. 1998;207(1):123–30.
Puri SK, Fan CY, Hanna E. Significance of extracapsular lymph node metastases in patients with head and neck squamous cell carcinoma. Curr Opin Otolaryngol Head Neck Surg. 2003;11(2):119–23.
Savage C, Zwischenberger JB. Image-guided fine needle aspirate strategies for staging of lung cancer. Clin Lung Cancer. 2000;2(2):101–10 (discussion 11–12).
Milman N, Faurschou P, Munch EP, Grode G. Transbronchial lung biopsy through the fibre optic bronchoscope. Results and complications in 452 examinations. Respir Med. 1994;88(10):749–53.
Pereira W Jr, Kovnat DM, Snider GL. A prospective cooperative study of complications following flexible fiberoptic bronchoscopy. Chest. 1978;73(6):813–6.
Varela-Lema L, Fernandez-Villar A, Ruano-Ravina A. Effectiveness and safety of endobronchial ultrasound-transbronchial needle aspiration: a systematic review. Eur Respir J. 2009;33(5):1156–64.
Strauss LG. Fluorine-18 deoxyglucose and false-positive results: a major problem in the diagnostics of oncological patients. Eur J Nucl Med. 1996;23(10):1409–15.
Buck AC, Schirrmeister HH, Guhlmann CA, Diederichs CG, Shen C, Buchmann I, et al. Ki-67 immunostaining in pancreatic cancer and chronic active pancreatitis: does in vivo FDG uptake correlate with proliferative activity? J Nucl Med. 2001;42(5):721–5.
Tehrani OS, Shields AF. PET imaging of proliferation with pyrimidines. J Nucl Med. 2013;54(6):903–12.
Yamamoto Y, Nishiyama Y, Kimura N, Ishikawa S, Okuda M, Bandoh S, et al. Comparison of (18)F-FLT PET and (18)F-FDG PET for preoperative staging in non-small cell lung cancer. Eur J Nucl Med Mol Imaging. 2008;35(2):236–45.
Yang W, Zhang Y, Fu Z, Yu J, Sun X, Mu D, et al. Imaging of proliferation with 18F-FLT PET/CT versus 18F-FDG PET/CT in non-small-cell lung cancer. Eur J Nucl Med Mol Imaging. 2010;37(7):1291–9.
Tian J, Yang X, Yu L, Chen P, Xin J, Ma L, et al. A multicenter clinical trial on the diagnostic value of dual-tracer PET/CT in pulmonary lesions using 3′-deoxy-3′-18F-fluorothymidine and 18F-FDG. J Nucl Med. 2008;49(2):186–94.
Hoshikawa H, Kishino T, Mori T, Nishiyama Y, Yamamoto Y, Inamoto R, et al. Comparison of (18) F-FLT PET and (18) F-FDG PET for detection of cervical lymph node metastases in head and neck cancers. Acta Otolaryngol. 2012;132(12):1347–54.
Schaefferkoetter JD, Carlson ER, Heidel RE. Can 3′-deoxy-3′-((18)F) fluorothymidine out perform 2-deoxy-2-((18)F) fluoro-d-glucose positron emission tomography/computed tomography in the diagnosis of cervical lymphadenopathy in patients with oral/head and neck cancer? J Oral Maxillofac Surg. 2015;73(7):1420–8.
Nakajo M, Kajiya Y, Jinguji M, Nishimata N, Shimaoka S, Nihara T, et al. Diagnostic performance of (1)(8)F-fluorothymidine PET/CT for primary colorectal cancer and its lymph node metastasis: comparison with (1)(8)F-fluorodeoxyglucose PET/CT. Eur J Nucl Med Mol Imaging. 2013;40(8):1223–32.
Kumar A, Dutta R, Kannan U, Kumar R, Khilnani GC, Gupta SD. Evaluation of mediastinal lymph nodes using F-FDG PET-CT scan and its histopathologic correlation. Ann Thorac Med. 2011;6(1):11–6.
Al-Sarraf N, Aziz R, Doddakula K, Gately K, Wilson L, McGovern E, et al. Factors causing inaccurate staging of mediastinal nodal involvement in non-small cell lung cancer patients staged by positron emission tomography. Interact CardioVasc Thorac Surg. 2007;6(3):350–3.
Takamochi K, Yoshida J, Murakami K, Niho S, Ishii G, Nishimura M, et al. Pitfalls in lymph node staging with positron emission tomography in non-small cell lung cancer patients. Lung Cancer. 2005;47(2):235–42.
Carbone RG, Penna D, Baughman RP, Lower EE. Accuracy of serial PET-CT imaging in systemic sarcoidosis. J Clin Imaging Sci. 2014;4:21.
Sobic-Saranovic DP, Grozdic IT, Videnovic-Ivanov J, Vucinic-Mihailovic V, Artiko VM, Saranovic DZ, et al. Responsiveness of FDG PET/CT to treatment of patients with active chronic sarcoidosis. Clin Nucl Med. 2013;38(7):516–21.
Sobic-Saranovic D, Grozdic I, Videnovic-Ivanov J, Vucinic-Mihailovic V, Artiko V, Saranovic D, et al. The utility of 18F-FDG PET/CT for diagnosis and adjustment of therapy in patients with active chronic sarcoidosis. J Nucl Med. 2012;53(10):1543–9.
Maturu VN, Agarwal R, Aggarwal AN, Mittal BR, Bal A, Gupta N, et al. Dual-time point whole-body 18F-fluorodeoxyglucose PET/CT imaging in undiagnosed mediastinal lymphadenopathy: a prospective study of 117 patients with sarcoidosis and TB. Chest. 2014;146(6):e216–20.
Chilosi M, Menestrina F, Capelli P, Montagna L, Lestani M, Pizzolo G, et al. Immunohistochemical analysis of sarcoid granulomas. Evaluation of Ki67+ and interleukin-1+ cells. Am J Pathol. 1988;131(2):191–8.
Semenzato G, Pezzutto A, Pizzolo G, Chilosi M, Ossi E, Angi MR, et al. Immunohistological study in sarcoidosis: evaluation at different sites of disease activity. Clin Immunol Immunopathol. 1984;30(1):29–40.
Tan Y, Liang J, Liu D, Zhu F, Wang G, Ding X, et al. F-FLT PET/CT imaging in a Wister rabbit inflammation model. Exp Ther Med. 2014;8(1):69–72.
Zhao S, Kuge Y, Kohanawa M, Takahashi T, Zhao Y, Yi M, et al. Usefulness of 11C-methionine for differentiating tumors from granulomas in experimental rat models: a comparison with 18F-FDG and 18F-FLT. J Nucl Med. 2008;49(1):135–41.
Wang R, Zhu H, Chen Y, Li C, Li F, Shen Z, et al. Standardized uptake value based evaluation of lymphoma by FDG and FLT PET/CT. Hematol Oncol. 2014;32(3):126–32.
Buck AK, Bommer M, Stilgenbauer S, Juweid M, Glatting G, Schirrmeister H, et al. Molecular imaging of proliferation in malignant lymphoma. Cancer Res. 2006;66(22):11055–61.
Sharma SK, Mohan A. Sarcoidosis in India: not so rare! JIACM. 2004;5(1):12–21.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Rayamajhi, S.J., Mittal, B.R., Maturu, V.N. et al. 18F-FDG and 18F-FLT PET/CT imaging in the characterization of mediastinal lymph nodes. Ann Nucl Med 30, 207–216 (2016). https://doi.org/10.1007/s12149-015-1047-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12149-015-1047-6