Abstract
Purpose
Most early publications on integrated positron emission tomography/computed tomography (PET/CT) devices have reported the new scanner generation to be superior to conventional PET. However, few of these studies have analysed the situation where, in addition to PET, a current CT scan is available for side-by-side viewing. This fact is important, because combined PET/CT or a software-based fusion of the two modalities may improve diagnosis only in cases where side-by-side reading of PET and CT data does not lead to a definitive diagnosis. The aim of this study was to analyse which patients will profit from integrated PET/CT in terms of lesion characterization.
Methods
A total of 328 consecutively admitted patients referred for PET in whom a current CT scan was available were included in the study. The localization of all pathological PET lesions, as well as possible infiltration of adjacent anatomical structures, was assessed.
Results
Of 467 pathological lesions, 94.0% were correctly assessed with respect to localization and infiltration by either conventional PET alone (51.6%) or combined reading of PET and the already existing CT scans (42.4%). Hence, in only 6.0% of all lesions, affecting 6.7% of all patients, could evaluation have profited from integrated PET/CT.
Conclusion
We conclude that side-by-side viewing of PET and CT scans is essential, as in 42.4% of all cases, combined viewing was important for a correct diagnosis in our series. In up to 6.7% of patients, integrated PET/CT might have given additional information, so that in nearly 50% of patients some form of combined viewing of PET and CT data is needed for accurate lesion characterization.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Positron emission tomography (PET) and computed tomography (CT) are well-established imaging modalities which are widely employed in modern oncology [1, 2]. As a new development in medical imaging, integrated PET/CT scanners are currently being introduced into clinical routine, permitting the acquisition of morphological and metabolic data in a single examination. Judging from the promising results of early studies, this innovative scanner generation is likely to bring substantial advances in oncological imaging [3–14]. Even so, it remains unclear which kind of patients will benefit the most from this new technique. As far as the above-mentioned studies are concerned, the diagnostic accuracies of CT, PET and integrated PET/CT have been analysed by assessing each modality separately [9–14]. None of the papers has taken into account the fact that many patients suspected of having a malignancy will already have undergone CT before referral to PET. In this respect, it must be kept in mind that integrated PET/CT may yield a diagnostic profit only for patients with pathological lesions which cannot be correctly identified by the combined reading of PET and already existing CT scans.
In the case of patients who are referred for PET without prior CT, the value of integrated PET/CT is beyond controversy. From the patient’s point of view, the preparatory procedures and acquisition protocols of PET and CT examinations are often considered extremely arduous. With integrated PET/CT, both procedures are distinctly shortened, which will certainly affect patient compliance positively. From the physician’s point of view, integrated PET/CT devices are advantageous because they yield metabolic as well as morphological data in a single session. In addition, CT and PET scans are fused automatically without complex image processing, and attenuation correction of the emission scans is done by using CT data so that no further transmission measurements are required. All these factors help to economize on the two most valuable resources: time and manpower.
The aim of the present study was to analyse which patients will benefit the most from integrated PET/CT in terms of lesion characterization. Since neither PET nor CT is able to differentiate benign from malignant lesions with absolute certainty, tumour staging according to the TNM classification was considered inappropriate to answer this question. Instead, the localization of PET lesions was chosen as the criterion for the present analysis. In addition, special care was taken to assess potential infiltration of adjacent organs or anatomical structures. Integrated PET/CT was considered advantageous for lesions that could be localized correctly neither by PET alone nor by combined side-by-side interpretation of PET and existing CT scans. Additionally, integrated PET/CT was considered advantageous when infiltration of adjacent structures could not be conclusively assessed using the available PET and CT scans. Clinical aspects like patient management, therapeutic outcome, prognosis and survival rate were not included in the study design because the focus was strictly on lesion characterization. Since both benign and malignant lesions were analysed using the same criteria—localization and potential infiltration—the cause of the increased 18F-fluorodeoxyglucose (FDG) uptake was not verified by surgical intervention, histopathology or clinical follow-up.
Materials and methods
Subjects
From March to December 2003, a total of 576 patients suspected of having malignant disease or a relapse were referred for PET examinations with FDG for staging purposes. In 342 patients (59.4%), a current CT scan performed within the preceding 4 weeks was available at the time of the examination. Of these 342 patients, 14 (4.1%) had to be excluded because a therapeutic intervention (radiotherapy, chemotherapy, surgery) had been initiated during the interval between CT and PET. Hence, 328 of 576 patients (56.9%) could be included in the study (223 males, 105 females, age range 31–79 years). This prospective study was designed according to the regulations of the local board for protection of data privacy and confidentiality.
Positron emission tomography
PET scans were acquired 59±6 min after intravenous administration of 305±26 MBq FDG with a dedicated full-ring PET scanner (Siemens ECAT Exact 922/47, Siemens-CTI, Knoxville, Tennessee, USA). Imaging was done from the base of the skull to the proximal femora. All patients fasted for at least 6 h prior to examination, as verified by determining the blood glucose level. Acquisition time was 12 min per bed position, with a transmission time of 4 min each. Both emission and transmission scans were done in two-dimensional mode. The transmission was acquired as “hot transmission” without repositioning of the patient using 68Ga/68Ge rod sources. After segmentation of the transmission data, empirical attenuation coefficients were employed for lung tissue (0.028 cm−1), bone (0.107 cm−1) and soft tissue (0.095 cm−1). Following normalization and scatter correction, the emission scan was reconstructed with and without attenuation correction using a weighted iterative ordered subsets expectation maximization algorithm (OSEM, 6 iterations, 16 substeps). In a final step, a three-dimensional isotropic Gauss filter was applied (FWHM 8 mm). Transverse, coronal and sagittal slices of 7 mm thickness were reconstructed with and without attenuation correction. Image interpretation was done while taking into account these images and three-dimensional maximum intensity projections.
Computed tomography
Spiral CT scans were acquired with different multi-slice and single-slice scanners after intravenous administration of a water-soluble contrast medium containing iodine. For CT scans of the abdomen, contrast medium was also administered orally. Standard acquisition protocols were followed for the different anatomical regions. Depending on the scan protocol, reconstruction algorithm and scanner type, axial slices with an effective slice thickness between 5.0 and 7.0 mm were reconstructed for printouts on film.
Assessment
Evaluation of scans was done according to the flowchart shown in Fig. 1 and the classification given in Table 1. All PET scans were analysed by two experienced and board-certified referees who were blinded to the results of other examinations and the clinical data. In a first step, the PET scans were assessed for pathological lesions. No differentiation between malignant and benign findings was done since only localization of the lesions and possible infiltration of adjacent structures mattered. All scans without pathological PET lesions were assigned to class A. If a pathological lesion was found, the two referees tried to assess its localization and potential infiltration of adjacent structures. A consensus between the referees was reached by discussing each case. Lesions which could be correctly assessed regarding localization and infiltration by PET alone were assigned to class B. The gold standard used to confirm or disprove the decision was the CT scan, which was evaluated by two experienced and board-certified radiologists. In the event that either the localization and/or the infiltration could not be correctly assessed with the PET scan alone, the CT scan was consulted. Lesions where the existing CT scan turned out to be conclusive for localization and infiltration were assigned to class C while those with an inconclusive CT scan for either or both criteria were assigned to class D. CT lesions without increased FDG uptake were not included in the study. PET/CT was considered advantageous for all lesions assigned to class D. As a final step, a patient-based analysis was derived from the lesion-based one. In the patient-based analysis, the lesion with the most disadvantageous class regarding combined reading of PET and existing CT was considered decisive (sequence: class D > class C > class B).
Results
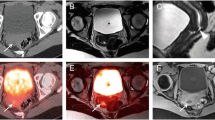
A total of 467 lesions were diagnosed in the 328 patients included in the study. For 28 of these 467 lesions (6.0%), the combined reading of PET and existing CT scans was inconclusive regarding localization and/or infiltration, so that integrated PET/CT was considered advantageous. The lesions were localized in the following regions: head/neck 7/28 (25.0%), thorax 3/28 (10.7%), abdomen 9/28 (32.1%), pelvis 5/28 (17.9%) and spine 4/28 (14.3%). Patient-based analysis revealed that a total of 22 patients (6.7%) were affected by these 28 lesions. Eleven lesions (39.3%) in ten patients (3.0%) were assigned to class D because they were not localized within the field of view of the CT scan. In seven (25%) of the remaining 17 lesions, no morphological correlate to the PET findings could be detected in the CT scans. Six of the seven lesions were small abdominal PET findings with a diameter of ≤2 cm on the PET scan; the seventh lesion—found in the bone marrow of the spine—had a diameter of about 2.5 cm. Regarding the remaining ten lesions (35.7%) assigned to class D, infiltration of adjacent anatomical structures could not be determined conclusively. Of these ten lesions, three were pulmonary findings where infiltration of the chest wall could be neither excluded nor confirmed, three occurred in patients with head and neck cancer with uncertain infiltration of the mandible, three were detected in the liver with uncertain infiltration of the abdominal wall or diaphragm, and one was found in a patient with colorectal cancer with unclear infiltration of the psoas. Detailed results of the lesion-based and patient-based analyses are given in Table 2. Figure 2 shows a lesion which could be easily assessed by PET, and Fig. 3 displays a lesion where CT was necessary to determine the exact anatomical localization and possible infiltration of adjacent structures. The frequency distribution of the different tumour types is given in Table 3.
Discussion
The recent literature indicates that integrated PET/CT devices will substantially refine oncological diagnostics by establishing a direct link between morphological and metabolic imaging [3–15]. Although several technical aspects of the new scanner generation are still subject to discussion [16–24], early clinical studies have yielded very promising results [8–14]. However, some authors object that expectations may be set too high [25, 26], something quite common in early studies dealing with new technical developments. The aim of our study was to investigate which patients may benefit from integrated PET/CT regarding lesion characterization, based on identification of those who are sufficiently well diagnosed by PET alone or by side-by-side reading of PET and already existing CT scans.
In our cohort, the majority of patients underwent a CT scan which could be used for side-by-side evaluation before referral of the patient for PET (328/576, or 56.9%). Of this subgroup, 94.0% of all lesions (439/467) and 93.3% of all patients (306/328) were correctly evaluated with either PET alone (lesion-based analysis: 51.6%; patient-based analysis: 48.8%) or combined reading of PET and CT (lesion-based analysis: 42.4%; patient-based analysis: 44.5%). Accordingly, only 6.0% of all lesions (28/467) and 6.7% of all patients (22/328) would possibly have profited from integrated PET/CT, with the small abdominal lesions turning out to be the critical ones.
In view of these data, having CT scans available from a separate study and correlating them with the PET data is an acceptable alternative to PET/CT imaging. We determined an upper threshold of usefulness of PET/CT at around 6.7%. As there is still wide variation in the protocols with which CT data are acquired in PET/CT, it is unlikely that all the cases that remained controversial after side-by-side viewing of PET and CT could have been diagnosed definitively by means of PET/CT [4, 5, 7, 8, 10–13, 17, 18, 20–22]. On the other hand, the option of instantaneous image fusion without time lag or repositioning of the patient on a second scanner is definitely advantageous, especially for anatomical regions where organs show a certain degree of mobility. Furthermore, side-by-side reading of PET and CT can be difficult, time-consuming and arduous, particularly for subtle lesions. Such small lesions may be missed by this approach—an assumption confirmed by the fact that six of the seven PET lesions without a morphological correlate on CT were small abdominal lesions.
Regarding diagnostic accuracy, there is no question that combined reading of PET and CT yields more accurate results compared with a separate evaluation of either modality: in our study, 42.4% of all PET lesions could only be assessed correctly with respect to localization and/or infiltration when CT results were also taken into account . These findings support the notion that PET and CT are synergistic methods which complement each other. Whenever possible, CT scans should be taken into consideration when analysing PET studies. PET/CT scanners as “one-stop shop” devices are surely advantageous in this regard.
A limitation of our study is that the structure of our cohort was somewhat unbalanced, as 50.9% of all patients were referred for PET owing to lung cancer. Although patients with lung cancer predominated in our cohort, those with malignant melanoma and lymphoma profited the most from integrated PET/CT regarding lesion characterization [malignant melanoma 3/23 (13.0%), lymphoma 3/24 (12.5%), lung cancer 11/166 (6.6%)]. In this context it must be kept in mind that only localization and infiltration of PET lesions were assessed. Further information available on CT, e.g. concerning density, lesion configuration or contrast enhancement, was not taken into consideration. Another limitation of our study concerns the assessment of possible infiltration of the chest wall by lesions located adjacent to the pleura. Since no definite CT evaluation criteria exist for such lesions save the detection of osseous destruction in the ribs or vertebrae [27, 28], doubtful cases were assigned to class D.
In conclusion, it can be said that the combined reading of PET and already existing CT scans proved highly effective for accurate lesion characterization. The complementary and synergistic character of PET and CT is confirmed by our results, and we therefore strongly recommend that all available CT scans be taken into consideration when interpreting PET studies. Furthermore, our data show that, compared with side-by-side reading of PET and CT, integrated PET/CT devices might provide additional information in about 6–7% of all lesions. Although this would not be a dramatic increase, it must be borne in mind that PET and CT have an excellent accuracy for many tumour types, and so even a relatively small improvement can be seen as substantial.
References
Gambhir SS, Czernin J, Schwimmer J, Silverman DH, Coleman RE, Phelps ME. A tabulated summary of the FDG PET literature. J Nucl Med 2001;42(Suppl):1S–93S.
Miles KA. Functional computed tomography on oncoloegy. Eur J Cancer 2002;38:2079–84.
Schoder H, Erdi YE, Larson SM, Yeung HW. PET/CT: a new imaging technology in nuclear medicine. Eur J Nucl Med Mol Imaging 2003;30:1419–37.
Steinert HC, von Schulthess GK. Initial clinical experience using a new integrated in-line PET/CT system. Br J Radiol 2002;S36–8.
Freudenberg LS, Antoch G, Schutt P, Beyer T, Jentzen W, Muller SP, et al. FDG-PET/CT in re-staging of patients with lymphoma. Eur J Nucl Med Mol Imaging 2004;31:325–9.
Ciernik IF, Dizendorf E, Baumert BG, Reiner B, Burger C, Davis JB, et al. Radiation treatment planning with an integrated positron emission and computer tomography (PET/CT): a feasibility study. Int J Radiat Oncol Biol Phys 2003;57:853–63.
Antoch G, Stattaus J, Nemat AT, Marnitz S, Beyer T, Kuehl H, et al. Non-small cell lung cancer: dual-modality PET/CT in preoperative staging. Radiology 2003;229:526–33.
Lardinois D, Weder W, Hany TF, Kamel EM, Korom S, Seifert B, et al. Staging of non-small-cell lung cancer with integrated positron-emission tomography and computed tomography. N Engl J Med 2003;348:2500–7.
Antoch G, Vogt FM, Freudenberg LS, Nazaradeh F, Goehde SC, Barkhausen J, et al. Whole-body dual-modality PET/CT and whole-body MRI for tumor staging in oncology. J Am Med Assoc 2003;290:3199–206.
Bar-Shalom R, Yefremov N, Guralnik L, Gaitini D, Frenkel A, Kuten A, et al. Clinical performance of PET/CT in evaluation of cancer: additional value for diagnostic imaging and patient management. J Nucl Med 2003;44:1200–9.
Pannu HK, Bristow RE, Cohade C, Fishman EK, Wahl RL. PET-CT in recurrent ovarian cancer: initial observations. Radiographics 2004;24:209–23.
Cohade C, Osman M, Leal J, Wahl RL. Direct comparison of 18F-FDG PET and PET/CT in patients with colorectal carcinoma. J Nucl Med 2003;44:1797–803.
Bristow RE, del Carmen MG, Pannu HK, Cohade C, Zahurak ML, Fishman EK, et al. Clinically occult recurrent ovarian cancer: patients selection for secondary cytoreductive surgery using combined PET/CT. Gynecol Oncol 2003;90:519–28.
Hany TF, Steinert HC, Goerres GW, Buck A, von Schulthess GK. PET diagnostic accuracy: improvement with in-line PET-CT system: initial results. Radiology 2002;225:575–81.
Schöder H, Larson SM, Yeung HWD. PET/CT in oncology: integration into clinical management of lymphoma, melanoma, and gastrointestinal malignancies. J Nucl Med 2004;45:72S–81S.
De Juan R, Seifert B, Berthold T, von Schulthess GK, Goerres GW. Clinical evaluation of a breathing protocol for PET/CT. Eur Radiol 2004;14:1118-23.
Nehmeh SA, Erdi YE, Kalaigian H, Kolbert KS, Pan T, Yeung H, et al. Correction for oral contrast artifacts in CT attenuation-corrected PET images obtained by combined PET/CT. J Nucl Med 2003;44:1940–4.
Antoch G, Freudenberg LS, Beyer T, Bockisch A, Debatin JF. To enhance or not to enhance? 18F-FDG and CT contrast agents in dual-modality 18F-FDG PET/CT. J Nucl Med 2004;45:56S–65S.
Kamel EM, Burger C, Buck A, von Schulthess GK, Goerres GW. Impact of metallic dental implants on CT-based attenuation correction in a combined PET/CT scanner. Eur Radiol 2003;13:724–8.
Cohade C, Osman M, Nakamoto Y, Marshall LT, Links JM, Fishman EK, Wahl RL. Initial experience with oral contrast in PET/CT: phantom and clinical studies. J Nucl Med 2003;44:412–6.
Dizendorf E, Hany TF, Buck A, von Schulthess GK, Burger C. Cause and magnitude of the error induced by oral CT contrast agent in CT-based attenuation correction of PET emission studies. J Nucl Med 2003;44:732–8.
Nakamoto Y, Chin BB, Kraitchman DL, Lawler LP, Marshall LT, Wahl RL. Effects of nonionic intravenous contrast agents at PET/CT imaging: phantom and canine studies. Radiology 2003;227:817–24.
Goerres GW, Ziegler SI, Burger C, Berthold T, von Schulthess GK, Buck A. Artifacts at PET and PET/CT caused by metallic hip prosthetic material. Radiology 2003;226:577–84.
Halpern BS, Dahlbom M, Waldherr C, Yap CS, Schiepers C, Silverman DH, et al. Cardiac pacemakers and central venous lines can induce focal artifacts on CT-corrected PET images. J Nucl Med 2004;45:290–3.
Schiepers C. PET/CT in colorectal cancer. J Nucl Med 2003;44:1804–5.
Vogel WV, Oyen WJG, Barentsz JO, Kaanders JHAM, Corstens FHM. PET/CT: panacea, redundancy, or something in between? J Nucl Med 2004;45:15S–24S.
Uhrmeister P, Allmann KH, Wertzel H, Altehoefer C, Laubenberger J, Hasse J, Langer M. Chest wall infiltration by lung cancer: value of thin-sectional CT with different reconstruction algorithms. Eur Radiol 1999;9:1304–9.
Roberts JR, Blum MG, Arildsen R, Drinkwater DC Jr, Christian KR, Powers TA, Merrill WH. Prospective comparison of radiologic, thoracoscopic, and pathologic staging in patients with early non-small cell lung cancer. Ann Thorac Surg 1999;68:1154–8.
Acknowledgements
Thanks are due to Alejandro Rodón and Birgit Reinartz for general and language editing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Reinartz, P., Wieres, FJ., Schneider, W. et al. Side-by-side reading of PET and CT scans in oncology: which patients might profit from integrated PET/CT?. Eur J Nucl Med Mol Imaging 31, 1456–1461 (2004). https://doi.org/10.1007/s00259-004-1593-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-004-1593-y