Abstract
Syngas fermentation has been successfully implemented in commercial-scale plants and can enable the biochemical conversion of the driest fractions of biomass through synthesis gas (H2, CO2, and CO). The process relies on optimized acetogenic strains able to reach and maintain high productivity of ethanol and acetate. In parallel, microbial communities have shown to be the best choice for the production of valuable medium-chain carboxylates through anaerobic fermentation of biomass, demanding low technical complexity and being able to realize simultaneous hydrolysis of the substrate. Each of the two technologies benefits from different strong points and has different challenges to overcome. This review discusses the rationales for merging these two seemingly disparate technologies by analyzing previous studies and drawing opinions based on the lessons learned from such studies. For keeping the technical demands of the resulting process low, a case is built for using microbial communities instead of pure strains. For that to occur, a shift from conventional syngas-based to “syngas-aided” anaerobic fermentation is suggested. Strategies for tackling the intricacies of working simultaneously with communities and syngas, such as competing pathways, and thermodynamic aspects are discussed as well as the stoichiometry and economic feasibility of the concept. Overall, syngas-aided anaerobic fermentation seems to be a promising concept for the biorefinery of the future. However, the effects of process parameters on microbial interactions have to be understood in greater detail, in order to achieve and sustain feasible medium-chain carboxylate and alcohol productivity.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Biorefineries based on waste biomass are appealing technologies that can support efforts of moving to a new bio-based, circular economy through the production of platform chemicals and fuels (de Jong et al. 2012). Among the many candidate technologies, chain elongation (CE) and syngas fermentation (SF) are two bioprocesses performed by anaerobic bacteria that might have a place in the integrated biorefineries of the future (Bengelsdorf and Dürre 2017; Chen et al. 2017; De Groof et al. 2019; Latif et al. 2014). Nevertheless, both CE and SF face their own challenges to achieve feasibility as industrial-scale processes. For instance, although CE is able to produce high-value medium-chain carboxylates (MCC) from waste biomass, in most lab-scale experiments to date, refined ethanol or lactate had to be co-fed to achieve extractable product concentrations (Chen et al. 2017). While the first demonstration plants of SF have started operating with CO-rich waste gas from steel mills, their main products are mostly limited to commodities that can also be produced by already existing biorefinery technologies, in particular ethanol and acetate (Takors et al. 2018). Thus, since 2008, when Steinbusch et al. (2008) purposely co-fed H2 to a microbial community to steer production to carboxylates and alcohols, many studies have been trying to merge SF and CE as a way of complementing the limitations of one technology with the other’s advantages.
Recent reviews have covered anaerobic fermentation for CE with microbial communities (Angenent et al. 2016; De Groof et al. 2019) and production of chemicals through SF (Bengelsdorf et al. 2018; Fernández-Naveira et al. 2016; Liew et al. 2016b; Molitor et al. 2017). This review is focusing on the use of syngas with such communities (sometimes also referred to as consortia, reactor microbiota, mixed cultures, or open cultures) to aid or to completely sustain production of medium-chain carboxylates and alcohols. Some studies with Clostridium carboxidivorans and acidogenic-carboxydotrophic co-cultures are also covered to establish a comparison ground with syngas-aided anaerobic fermentation by microbial communities.
General overview of chain elongation
Through a pathway known as reverse β-oxidation (RBO), short-chain monocarboxylates (SCC, e.g., acetate, propionate, n-butyrate) are elongated by two carbons to MCC (e.g., n-valerate, n-caproate) with the help of an electron donor (Fig. 1a). Once a broth is rich in MCC and electron donors are available, it is possible to trigger bacterial solventogenesis metabolism by changing reactor operating conditions to produce medium-chain alcohols (MCA, e.g., n-pentanol, n-hexanol) from their MCC counterpart, thereby expanding the product spectrum of CE (Ganigué et al. 2016).
Most relevant metabolic arsenal of an anaerobic microbial community for syngas-aided chain elongation for MCC and MCA production. a Different metabolic routes can be realized by a single cell or by two or more species with extracellular transfer of intermediates. i-Butyrate production is likely from bio-isomerization of n-butyrate and is a coproduct receiving increasing attention; i-caproate is another possible product (not shown) (de Leeuw et al. 2019; de Smit et al. 2019). Each arrow represents one reactional step realized by an enzyme, phosphorylation steps are omitted for conciseness. The direction of the arrow suggests the most favorable direction of the reaction during MCC and MCA production. Unidirectional arrows do not mean, necessarily, irreversible reactions. Dashed lines illustrate simplified pathways. Lines connected without an arrow indicate a summing reaction. For instance, one acetyl-CoA and one butyryl-CoA are used to form caproyl-CoA. Use of electron carriers and ATP coupling for n-valerate, n-caproate, n-heptanoate, and n-caprylate occur analogously to n-butyrate formation. Different species may use different electron carriers than those proposed here. b Both the WLP and RBO pathways are intimately connected to energy conservation, electron cycling, and the ionic homeostasis of the cell. Some of the most relevant soluble and membrane-bound enzymes for CE and SF are shown with exemplary stoichiometry values. The arrow directions indicate the normal reaction direction during autotrophic growth or carboxylate production. These reactions are generally reversible under the physiological conditions of the cell. Schemes elaborated based on Angenent et al. (2016); Buckel and Thauer (2013); Liew et al. (2016a); Schuchmann and Müller (2014); Bengelsdorf et al. (2018); Costa and Leigh (2014); Kremp et al. (2018); Spirito et al. (2014); Weghoff et al. (2015); Weimer and Moen (2013)
Limitation by electron donors
Despite its potential to convert waste biomass into higher-value MCC, CE is mainly limited by the electron donor. Ethanol and—more recently—lactate have been mostly used as electron donors (Angenent et al. 2016; Cavalcante et al. 2017; Zhu et al. 2015). As most industrial waste streams have lower ethanol or lactate concentrations than ideally needed to promote high yield CE, considerable amounts of these chemicals have to be procured to the fermenter feed.
Ethanol and lactate supplementation to improve MCC has consequences on the economics and sustainability of the process:
- 1.
Large-scale streams of diluted ethanol and lactate, e.g., corn beer from bioethanol plants or acid whey from dairy production lines can be concentrated to the commodity-grade chemical by inexpensive, mature technologies (e.g., pressure swing adsorption for ethanol and evaporation/crystallization for lactate) (Chahal and Starr 2006; Kosaric et al. 2000). This means that the feasibility of using ethanol or lactate for MCC production would be restricted to niche waste streams—streams that for certain reasons cannot be used for commodity ethanol or lactate production. Some of these streams have been already studied, such as wine lees with diluted ethanol (Kucek et al. 2016c) or lactate from ensiled crop by-products (Lambrecht et al. 2019; Scarborough et al. 2018b; Sträuber et al. 2018).
- 2.
Considering the environmental impact, ethanol use was identified as the biggest accountable factor for the environmental impact in terms of global warming potential, acidification potential, and eutrophication potential during n-caproate production from waste biomass (Chen et al. 2017). The impact of supplemented lactate in the MCC production life cycle remains to be assessed. However, it can be anticipated that the environmental impact of procured lactate is not too different from that of ethanol since commercial lactate is also produced by sugar fermentation or by chemical synthesis from fossil derivatives (Chahal and Starr 2006; Endres and Siebert-Raths 2009).
In order to improve process feasibility, it is highly desirable to supplement the waste streams feed with more affordable electron donors than ethanol and lactate. Novel strategies are studied to diversify ethanol and lactate usage in CE. Among them, the usage of strains that produce MCC from sugars or methanol that could come from waste biomass (Chen et al. 2016; Jankowska et al. 2018; Jeon et al. 2016), bioelectrochemical systems (Jourdin et al. 2018; Vassilev et al. 2019), phototrophic organisms (Doud et al. 2017), or, ultimately, approaches using the reductive power of H2 and CO (Liew et al. 2016b; Steinbusch et al. 2011) have been investigated.
Medium-chain carboxylates and alcohols of special interest
MCC and MCA are potential platform chemicals and biofuels that could meet many market needs that nowadays are met by fossil resources. Among MCC, n-caproate (C6) and n-caprylate (C8) have received special attention in CE research. This is attributed, among other reasons, to higher C/O ratios, higher energy density, and easiness to extract them from water in comparison to n-butyrate.
Regarding alcohols, similar reasoning justifies research focus on n-butanol and n-hexanol production among MCA (Fernández-Naveira et al. 2017). The highly reduced and almost water-insoluble n-octanol would be also a desired product from acidogenic-solventogenic fermenters, but its production in anaerobic fermentations has been restricted to trace amounts up to now (Richter et al. 2016a).
Production of even-numbered MCC depends on the presence of an electron donor that generates acetyl-CoA (i.e., ethanol or lactate) and even-numbered SCC (e.g., acetate, n-butyrate), which are more commonly present in acidogenic reactors than the odd-numbered counterparts, i.e., n-propanol or propionate, which are needed to form n-valerate and n-heptanoate (Fig. 1a) (Bengelsdorf et al. 2018). Since Clostridium kluyveri produces n-valerate when fed by propanol and acetate (Kenealy and Waselefsky 1985), one way to extend product selectivity to odd-numbered MCC is to use n-propanol as an electron donor. The reason for it is shown in the metabolic network in Fig. 1a where propionyl-CoA, produced from n-propanol, condenses with acetyl-CoA during CE forming the odd-numbered n-valerate through pentanoyl-CoA (Kenealy and Waselefsky 1985; Marounek et al. 1989). Since the RBO pathway can really be intermediated by acetyl-CoA together with propionate (propionyl-CoA), it is also possible to produce odd-numbered MCC from lactate and acetate by a bacterium such as Megasphaera elsdenii that possesses both the RBO and the acrylate pathways, the latter being a lactate-consuming pathway intermediated by propionyl-CoA, as shown in Fig. 1a (Weimer and Moen 2013). Methanol is another strategic electron donor for being a C1 compound able to be incorporated into the MCC and MCA pool. Mostly even-numbered MCC are produced from methanol and acetate fed to a mixed culture (Chen et al. 2016). Methanol could in principle be oxidized to formaldehyde that enters the Wood-Ljungdahl pathway (WLP) or have its methyl group transferred to THF to yield methyl-THF. Downstream, methyl-THF is converted with carbon monoxide into acetyl-CoA by a microorganism able to grow autotrophically, like Eubacterium limosum (de Smit et al. 2019; Kremp et al. 2018; Pacaud et al. 1985).
Syngas as an alternative source of electron donors
With the capacity to convert the lignin fraction, biomass gasification technologies may also have a place in the integrated biorefinery of the future. Gasification products are considered third-generation substrates; among them, syngas (i.e., H2, CO, and CO2) and water-shifted gas (i.e., H2 and CO2) can be an extra source of electrons and carbon for MCC production (Cueto-Rojas et al. 2015; Liew et al. 2016b). Besides, H2 gas from electrochemical processes could also be fed to the anaerobic fermenter as long as a carbon source is provided (Rabaey and Rozendal 2010; Vassilev et al. 2018). For this review, water-shifted gas and H2 gas are all referred as syngas for simplicity.
The gateway for incorporating syngas into the carboxylate pool is the WLP (Fig. 1a). In the WLP of conventionally known homoacetogens, H2, CO2, and CO are fixed into acetyl-CoA with consumption of ATP. Downstream, acetyl-CoA can either be converted to acetate, returning the ATP investment, or to ethanol, oxidizing NAD(P). As shown in Fig. 1b, the pathway depends on the interconversion of these electron carriers through hydrogenases in the cytosol and cation export complexes such as the Rnf complex (e.g., in Clostridium ljungdahlii and Acetobacterium woodii) or the Ech complex (e.g., in Moorella thermoacetica and Rhodospirillum rubrum) (Schuchmann and Müller 2014). Net ATP gain in autotrophically grown homoacetogens is still possible thanks to the ion-motive force maintained by these ion export complexes (i.e., Rnf or Ech) and ion import made by transmembrane ATP synthases (Fig. 1b). There is in-depth literature covering current knowledge of SF by anaerobic bacteria (Bengelsdorf et al. 2018; Diender et al. 2015; Liew et al. 2016b; Schuchmann and Müller 2014).
Other, less common products of syngas-fermenting bacteria are formate, 2,3-butanediol, n-butyrate, i-butyrate, n-caproate, n-butanol, and n-hexanol (Bengelsdorf et al. 2018). Up to date, C. carboxidivorans and E. limosum are the only two strains reported to be able to form n-caproate from syngas, and C. carboxidivorans is the only known strain able to form n-hexanol from syngas (Lindley et al. 1987; Phillips et al. 2015).
When C. carboxidivorans was not found in syngas-fed microbiota, it was assumed that n-caproate/n-caprylate production occurred via a multi-species synergy with conventional CE intermediated by acetate (or n-butyrate) and ethanol from the WLP (Ding et al. 2010; Ganigué et al. 2015). Conversely, typical ethanol-based chain-elongating species, such as C. kluyveri, have not always been found in studies where n-caproate and n-caprylate were produced (Grimalt-Alemany et al. 2018; Kucek et al. 2016b; Nzeteu et al. 2018).
Microbial communities and pure cultures
Syngas can be swiftly consumed by pure strains producing a limited range of short-chain chemicals such as acetate and ethanol (Molitor et al. 2017). C. carboxidivorans P7 (Phillips et al. 2015; Ramachandriya et al. 2013) is a remarkable exception because, to date, no other pure culture has been confirmed to produce the C6 carboxylate and alcohol when fed only with syngas. Yet, even after media optimization for strain P7, concentrations of C6 produced from syngas remained one order of magnitude lower (i.e., ~ 1 g/L) than those produced in conventional CE reactors (i.e., ~ 10 g/L of n-caproate or about the solubility limit of n-caproate at the working pH) (Fernández-Naveira et al. 2019; Grootscholten et al. 2014; Ramió-Pujol et al. 2015; Reddy et al. 2018). Even though there might be other strains producing MCC from syngas (e.g., the syngas-fermenting species E. limosum that can also produce C6 from methanol), their product titers will hardly increase by an order of magnitude (Bengelsdorf et al. 2018; Lindley et al. 1987; Wade 2015). Thus, without considering the use of genetic engineering, some studies have adopted two-culture approaches with homoacetogenic and acidogenic strains (Diender et al. 2016; Gildemyn et al. 2017; Richter et al. 2016a).
Microbial communities share the low C6 and C8 titers of C. carboxidivorans when fed only by syngas (Molitor et al. 2017). Still, mixed cultures can have productivities and concentrations of MCC comparable with pure cultures of chain-elongating species in lactate- or ethanol-based acidogenic reactors and in a broader range of pH (De Groof et al. 2019). Benchmarking open mixed cultures with C. carboxidivorans and co-cultures (e.g., C. kluyveri/C. ljungdahlii) for syngas-aided CE is limited since microbial communities are the only option to directly convert complex biomass (e.g., lignocellulose) to the MCC pool. Therefore, a co-feeding strategy of syngas and degradable types of biomass (in particular with feedstock proven successful for CE) can make mixed cultures feasible for syngas-fermenting reactors. In other words, open cultures may excel in syngas-aided (and not in syngas-based) CE.
The use of open mixed cultures can further add simplifications to the bioprocess of MCC and MCA production. It is known from anaerobic digestion and fermentation research that microbial communities can operate steadily in unsterile reactors (Werner et al. 2011; Agler et al. 2014; Sträuber et al. 2018), which can help lower process capital and operating costs in comparison with monoseptic conditions. Besides, it is expected that communities can better handle the inhibitors and contaminants typically found in syngas. Aromatics, tars, HCN, sulfur oxides, and many other compounds - besides H2, CO, and CO2—can be found in real syngas and some are known to negatively affect performance of the best syngas fermenters (Oswald et al. 2018; Sikarwar et al. 2016). Open mixed cultures have shown robustness and resilience in hydrolysis and fermentative reactor operation and are used to degrade biomass of varying quality despite the presence of natural inhibitors in the substrate, such as alkaloids (Popp et al. 2016) and phenolic compounds (Chapleur et al. 2016). This characteristic resilience to substrate quality fluctuations has been decisive for the success of technologies in anaerobic digestion and wastewater treatment (Werner et al. 2011). However, to the best of the authors’ knowledge, no study has tested yet this assumed robustness of microbial communities with real syngas.
The biggest hurdles for the application of open mixed cultures may be instability of the community and lack of knowledge of the microbial interactions in these systems (Arslan et al. 2016; Werner et al. 2011). It is not trivial to establish a common ground for comparing kinetics between pure and mixed cultures. However, it can be observed that when operating with mixed cultures fed with syngas, longer fermentation times (in the order of dozens and hundreds of days) are generally needed to achieve steady production rates in comparison with pure cultures, the latter having more reproducible and definable kinetics. The study done by Ganigué et al. (2015) can be taken as an example, where batch trials with syngas-fed C. carboxidivorans and with an enriched carboxydotrophic mixed culture were realized for 4 and 20 days with the pure culture and the microbial consortium, respectively. Dynamics and fermentation time to achieve stable conversion rates can also be in the order of dozens of days for microbial communities degrading solid substrates as shown by Sträuber et al. (2016), where a 200-day pre-cultivated community was used to degrade corn silage in semi-continuous reactors. Fortunately, studies on acidogenic bacterial communities do not have to start from scratch because much of the knowledge developed with methanogenic communities in anaerobic digesters is useful for MCC- and MCA-producing communities (Agler et al. 2014, 2012; Spirito et al. 2014). In the next years, the remaining knowledge gaps on acidogenic bacterial communities may be tackled with the advancement of high-throughput omics approaches and increasing accessibility of cell-level analytical techniques. Notwithstanding, deeper understanding in microbial ecology will offer applied insights on what happens and what does not within such fermentative communities.
Process rationales for syngas-aided and syngas-based strategies
The concept of merging SF and CE in order to take profit from its synergies has been a topic in several previous studies. Main rationales together with example studies are stated in Table 1. Figure 2 summarizes the process flow strategies adopted in the studies that attempted to join CE and SF. Figure 2 a and d are syngas-based strategies while Fig. 2 b, c, and e are syngas-aided strategies.
Process rationales for merging syngas fermentation (SF) and chain elongation (CE). Ethanol can be substituted by lactate as an electron donor for CE. Virtually all organic fractions of feedstock biomass can be incorporated into SF by gasification, although not all biomass types are feasible. Fractions of biomass can be incorporated to CE by microbial hydrolysis and fermentation. MCC: medium-chain carboxylates; MCA: medium-chain alcohols
The scheme depicted in Fig. 2a shows SF in the first process step and a CE reactor receives the effluent from the first reactor, preferably with a high ethanol:acetate ratio. The first reactor depends on solventogenic carboxydotrophic bacteria and the CE reactor needs, therefore, bacteria able to realize ethanol-based CE. As shown in Table 1, studies that adopted this strategy opted out for a pure strain for the SF step and for C. kluyveri or a mixed culture for the CE step.
Figure 2 b shows CE as the first step and SF in series for reducing MCC to MCA. Richter et al. (2013) built the case for lowering medium costs by assuming prices for laboratory-grade, non-bulk nutrients. The authors acknowledged that laboratory-grade nutrient prices are higher than industrial-grade ones but sustained the point that the estimate is still valid to prove the need for lower nutrient costs. This nutrient cost overestimation is sometimes orders of magnitude higher than from industrial-grade ingredients, thus raising a flag about the claim that medium costs are indeed such a big hindrance for viability of SF and CE as bioprocesses.
The strategy depicted in Fig. 2c is the least frequently studied among the five strategies. Such strategy allows the incorporation of feedstock biomass to the substrate pool and can work with two differently acclimatized microbial communities. It is a possible answer to the incompatibilities of process conditions of CE, SF, and solventogenesis reported previously (Ganigué et al. 2016; Richter et al. 2016a). The first stage operates as a hydrolysis and CE reactor and the second stage converts carboxylates to alcohols through syngas-based solventogenesis. However, one caveat against this strategy is that, in general, microbial communities have not excelled in solvent production.
Figure 2 d represents the scheme for simultaneous SF and CE to MCC and MCA. A strain like C. carboxidivorans P7 can produce MCC and MCA from syngas in a single process step. Despite C. carboxidivorans has been reported to be able to grow on cellulose, cellobiose, and pectin (Liou et al. 2005), the authors did not find any reports about its performance in mixotrophic growth with significant MCC and MCA production. Therefore, it is assumed here that C. carboxidivorans is not a viable culture for simultaneous degradation of real lignocellulosic waste and SF. Without biomass hydrolysis, the only way to use waste biomass in this process configuration is by a preceding conversion to syngas (Fig. 2). When using communities, MCC and MCA titers from using such configuration have remained low to date. Reactors that allow high mass transfer such as hollow-fiber membrane biofilm reactors have been used to successfully increase selectivity to n-caproate and n-caprylate. However, the maximum concentration of n-caproate or n-hexanol of around 1 g/L seems to be a hard cap for the optimization of such strategy (Ganigué et al. 2016; Shen et al. 2018; Zhang et al. 2013).
Anaerobic fermentation of syngas with a community for MCC/MCA production works analogously to Fig. 2d, but with the possibility of biomass co-feeding (Fig. 2e). Such “syngas-aided anaerobic fermentation” uses biomass that is preferably rich in an electron donor such as lactate or ethanol. Similarly to conventional ensiling, lactic acid bacteria can convert fractions of the lignocellulose to lactate in situ as electron donors that are subsequently consumed during CE. In parallel, the reductive power of H2 and CO could lower lactate or ethanol consumption, lowering process costs or increasing total conversion to MCC/MCA. Several studies that follow this strategy are listed in Table 1. Interestingly, product concentrations are not limited as in setups that follow Fig. 2d and studies with the adequate controls indicate that the added syngas can increase concentration and selectivity to the more reduced chemicals, i.e., the longer-chain carboxylates and alcohols. As one remarkable example for the scheme in Fig. 2e for syngas-aided CE, batch test results presented by Nzeteu et al. (2018) suggested that a lactate-based CE community had synergy with hydrogenotrophic activity to produce about 130% more n-caproate (totaling 10.4 g/L) in comparison with the H2-free fermentation. A similar synergy was recently reported by Wu et al. (2019), where 44% more n-caproate (totaling 5.5 g/L) was obtained from a lactate-based CE reactor with H2 co-feeding. Interestingly, such synergy was also found by Steinbusch et al. (2011)—though to a lesser extent of about 10% increase of n-caproate, to 8.2 g/L—when bubbling H2 to a mixed culture that was performing ethanol-based CE.
Syngas-aided anaerobic fermentation with microbial communities
Competing pathways in microbial communities
Together with the advantages brought by highly diversified bacterial communities, it is likely that more competing pathways to those important for MCC and MCA production will coexist in the reactor. Many of these pathways have been studied and strategies to steer them towards the desired process have been proposed. Table 2 sums up these generally undesired pathways when producing MCC and MCA by mixed cultures with the help of syngas. The effect of each pathway is discussed and known strategies that are able to inhibit partially or completely these pathways are also presented.
Partial pressures of H2 usually present in syngas are more than enough to inhibit excessive oxidation of alcohols and carboxylates. This feature has, indeed, motivated studies about syngas-based and syngas–aided CE and some of these studies are listed in Table 1. As a trade-off, the continuous presence of H2 means that the sulfate-reducing and methanogenic members of the community can also grow abundantly, taking advantage of higher energy-yielding reactions than, for instance, homoacetogenesis and CE.
Regarding the competing pathways that are not inhibited by H2, the acrylate pathway (AP) has not been found to be of concern for lactate consumption in most studies on lactate-based CE (Cavalcante et al. 2017; Lambrecht et al. 2019), and even when it occurred, it was overcome by adequately managing pH values and lactate loading rates. Besides, AP is not always considered a competing pathway. Due to propionate formation, AP could be a desired pathway in the acidogenic community, since it is a way to expand the product spectrum to odd-numbered MCC and MCA (such as n-valerate, n-heptanoate, and n-pentanol) in CE systems based on lactate and acetate (Wu et al. 2018). The same cannot be said about hydrogenotrophic methanogenesis (HM). Resilient HM is arguably the most challenging pathway to be tackled in syngas-aided MCA and MCC production with an open mixed community (Zhang et al. 2018). Differently from acetoclastic methanogenesis (AM), HM has been found to be persistent at pH values as low as 5.2 (Savant et al. 2002) and the most common way to selectively inhibit it in laboratory studies—using relatively high concentrations of 2-bromoethanosulfonate (50 mM)—would be too expensive for a refinery-scale process. Additionally, no single proposed action in Table 2 is able to completely counter HM alone without also compromising MCC and MCA yields. Thus, new cost-effective and selective ways to hinder HM still need to be studied if syngas-based fermentation with microbial communities is to become a biorefinery process. Despite the fact that sulfate-reducing bacteria (SRB) are able to outcompete even methanogens for consumption of H2 and acetate (Plugge et al. 2011), competition from SRB can be avoided in a relatively simply fashion by keeping sulfate concentrations sufficiently low and using reduced sulfur supplements as sulfur source (Hu et al. 2015).
Thermodynamic aspects
A myriad of metabolic pathways are possible to be realized by anaerobic communities in the ranges of pH 4.5 to 7.5 and temperatures of 28 to 37 °C, ranges in which CE and SF are also found to perform best (González-Cabaleiro et al. 2015). When the community is not limited by the lack of a gene, its expression or by kinetic phenomena, it is still ultimately limited by thermodynamics. It has been shown that such thermodynamic limitation holds true for many metabolic routes in anaerobes (Heimann et al. 2009; Kleerebezem and Stams 2000; Richter et al. 2016b). In the case of syngas-aided CE, relevant catabolic reactions that are subject to thermodynamic limitation are shown in Table 3. The Gibbs free energy of the reactions was calculated for biochemical standard conditions (T = 298 K; 100 kPa; pH 7.0; 1 M of each reactant and product) as well as for conditions of temperature, pH, and chemical concentrations closer to those of a bioreactor operating for syngas-aided MCC and MCA production (pH = 5.5; T = 310 K; 100 mM acetate; PH2 = PCO2 = PCO = 30 kPa; 10 mM or 1 kPa for other reactants and products).
As shown in Table 3, some strategies are conceivable to selectively favor MCC and MCA formation in a syngas-fermenting community. As seen in reactions 8 and 13 (Table 3), high acetate concentrations in the reactor favor reactions that accumulate longer-chain carboxylates. In practice, this was verified in various experiments with acidogenic reactors, as reported by Arslan et al. (2016), and can give a selective advantage to acidogenic bacteria over methanogenic archaea at a defined pH (Zhang et al. 2018). As a trade-off, SCC concentrations higher than 50 mM generally increase the lag-phase of anaerobic cultures (Jaros et al. 2012).
It is generally assumed that MCC production from H2 and CO2 by pure or mixed cultures does not occur by a specific pathway with H2 as a direct electron donor. Instead, it has been proposed that the n-butyrate, n-caproate, and n-caprylate titers seen in syngas-based fermentations were intermediated by ethanol and acetate (Ding et al. 2010; González-Cabaleiro et al. 2015). According to González-Cabaleiro et al. (2013), the unfeasibility of acetate reduction to n-butyrate with H2 (reaction 13 Table 3) may be due to a kinetic bottleneck during the condensation of two acetyl-CoA into acetoacetyl-CoA, which becomes more unfavorable the higher the H2 partial pressure is, despite the exergonic character of the overall reaction. This limitation imposes that ethanol (or lactate) needs to be present for the formation of n-butyrate and longer chain carboxylates from acetate to take place.
Ethanol can be formed by acetate reduction and can be assisted with H2 consumption in the near-equilibrium reaction 5 or with CO consumption in the more exergonic reaction 6 (Table 3) being both reactions highly exothermic (with standard enthalpy of reactions of − 83 and − 88 kJ/reaction, respectively). Biologically, however, autotrophic ethanol production from H2/CO2 or CO seems to be less frequently observed than autotrophic acetogenesis in microbial communities since not every acetogen is able to couple such ethanol formation with net ATP gain (Molitor et al. 2017). As an example, when A. woodii is fed by H2:CO2, ethanol formation would lead to net ATP loss. In acetogens, such as C. ljundgdahlii, the ATP yield of autotrophic ethanol formation depends on which of the two possible routes for ethanol production from acetyl-CoA is active (Fig. 1a). Thanks to the acetaldehyde:ferredoxin oxidoreductase (also known as AOR), acetaldehyde can be formed from acetate reduction. In this case, and if reduced ferredoxin is not limiting, ethanol formation has more favorable energetics since the ATP balance is zeroed in the WLP before solventogenesis starts. AOR is absent in organisms like A. woodii but present in solventogenic strains like Clostridium autoethanogenum and C. ljungdahlii. C. autoethanogenum is able to sustainably produce ethanol from H2:CO2 regardless the way how acetaldehyde is formed (Köpke et al. 2010; Mock et al. 2015; Steinbusch et al. 2008). The AOR enzyme uses reduced ferredoxin for the reduction of acetate to acetaldehyde and CO oxidation generates just that (Fig. 1).
Put simply, CO removes kinetic hindrances of the WLP and solventogenesis by preserving the ferredoxin pool reduced. Thermodynamically, acetogenesis and solventogenesis from CO are more favorable than the hydrogenotrophic reactions (reactions 2, 4, and 6 Table 3). Nevertheless, the use of CO from syngas in a community can be seen as a double-edged sword. CO is a substrate that can increase autotrophic activity, promote cell biomass formation, and sustain solventogenesis, but it has nonselective inhibitory and toxic effects on microorganisms. To add to the complexity, resistance to CO varies among different microbial species and though it is possible to acclimatize communities to it, high CO partial pressures limit the microbial diversity (Esquivel-Elizondo et al. 2017; Guiot et al. 2011). The toxicity mechanisms of CO in bacteria, though still not completely clear, are assumed to be through irreversible inhibition of metalloenzymes such as the ferredoxin-dependent hydrogenases (Ragsdale 2004; Yasin et al. 2015). Since hydrogenases and other enzymes involved in the electron transport are metalloenzymes in general, this mechanism can explain the observed hindrance of H2 and CO2 consumption by autotrophs in the presence of CO in some studies (Diender et al. 2015). Whatsoever the predicted consequences of CO on communities performing biomass degradation, SF, or CE are, studies that try to understand these trade-offs could add much to this topic (Bengelsdorf et al. 2018; Chakraborty et al. 2019; Diender et al. 2015; Esquivel-Elizondo et al. 2018; Molitor et al. 2016; Sipma et al. 2004).
H2 is a product of conventional ethanol-based CE (4 times reaction 8 coupled with a reversed reaction 5, Table 3), formed during n-butyrate production from sugars and in conventional anaerobic fermentation of sugar to acetate (reactions 14 and 15, Table 3) (Arslan et al. 2012; Schoberth and Gottschalk 1969). Such clearly exergonic reactions are limited by kinetics rather than by thermodynamic equilibrium and even high partial pressures of H2 are not enough to cause a noticeable inhibition effect on them (González-Cabaleiro et al. 2015). A study done by Arslan et al. (2012) found that higher H2 partial pressure actually favored conversion of carbohydrate-rich waste and increased carboxylate yield by a microbial community. The higher yields of acetate, n-butyrate, and n-caproate in the reactors with added H2 could be explained by a combination of 1) homoacetogenic sugar fermentation, as described by Schuchmann and Müller (2016), where 1 mol of glucose is converted to 3 mol of acetate by acetogenic bacteria able to grow mixotrophically (reactions 14 and 1 in series, Table 3); and 2) H2:CO2-aided CE, as described before, possibly intermediated by ethanol (reaction 5 in series with reactions 8 and 9, Table 3). Results of some studies suggest that similar synergies with mixotrophs and parallel H2:CO2-aided CE might exist although their exact mechanisms were not systematically tested yet (Arslan et al. 2012; Ding et al. 2010; Nzeteu et al. 2018; Steinbusch et al. 2011).
The intermediation of lactate in mixed cultures producing MCC from H2 and SCC is yet a poorly studied possibility (Scarborough et al. 2018a). Small concentrations of lactate can be found in anaerobic fermentation systems even in the absence of organic substrates as a result of the natural pyruvate concentrations in the active cells (Garvie 1980). As seen in reactions 11 and 12 Table 3, CE of SCC with low lactate concentrations is highly exergonic as long as high SCC concentrations are kept. Analogously to CE realized by C. kluyveri, the recently isolated lactate-based MCC-producing strain Ruminococcaceae bacterium CPB6 also needs to oxidize part of the lactate into SCC for ATP generation and cell growth producing H2 as consequence (reaction 23 Table 3) (Wang et al. 2018; Zhu et al. 2017). The coupling of reactions 11, 12, and 23 (Table 3) also happens in lactate-based n-caproate production by mixed cultures (Zhu et al. 2015), producing 2 moles of H2 and 3 moles of CO2 per produced mol of n-caproate from 3 moles of lactate. In principle, if active hydrogenotrophs are present in the community, the produced H2/CO2 can be reincorporated to the carboxylate pool through the WLP (reaction 1 Table 3).
It is worth pointing that in MCC/MCA-producing systems, the predictive power of such thermodynamic analysis is limited to the operation states where inhibitory effects of products and substrates on the community are not significant. The longer the chain of the MCC (and MCA) the higher is the inhibiting effect on a mass basis. MCC inhibition is especially strong in acidic media, where more MCC is found undissociated. Such forms of MCC can jeopardize the pH homeostasis of cells by passively crossing the cellular membrane or even by dissolving it. Accordingly, MCA inhibition occurs due to the solvent, hydrophobic behavior.
The thermodynamic effects of temperature also need to be taken into consideration in syngas-aided CE. Besides the clear effect of temperature on microbial kinetics, temperature changes the solubility of gases in water and the Gibbs free energy of possible reactions realized by the microbial community. Figure 3 depicts these thermodynamic effects of temperature in a range from 25 to 55 °C. Figure 3 a shows particular strong effects of temperature on the solubility of CO2 and CO, while solubility of H2 barely changes. Even under conditions when CO2 and CO are not the limiting substrates, lowering the temperature slightly to increase their solubility can be worth considering, since experience has shown that increasing the availability of these substrates significantly impacts the process performance (Roghair et al. 2018a). It is worth noticing, however, that diffusion coefficients increase with temperature; thus, such conclusions—i.e., lowering temperature for higher gas solubility—should only be applicable to systems that are not limited by gas-liquid mass-transfer (Diender et al. 2015).
Lower temperatures increase the solubility of gaseous substrates in water (a), thermodynamically favor gas-consuming reactions, and have little effect on CE reactions (b). Gas solubility values calculated for unbuffered water using Henry’s law with temperature correction according to the Van’t Hoff equation. \( \Delta {G}_r^{\prime 0} \) is the Gibbs free energy of reaction with activity of chemical species equal to 1, at 25 °C and pH = 7.0
In general, lower temperatures increase favorability of gas-consuming reactions with a particularly strong effect on solventogenesis and hydrogenotrophic methanogenesis (Fig. 3b). This might help explain better yields of n-caproate and n-hexanol obtained by Ramió-Pujol et al. (2015) when growing C. carboxidivorans on syngas at 25 °C in comparison with cultivations at 37 °C. On the other hand, due to particularly stronger effects on kinetics of some methanogens, lower temperatures do not necessarily give a selective advantage to hydrogenotrophic methanogenesis in practice. In fact, temperatures lower than 25 °C have been even proposed as a tool to avoid methanogenic activity in acidogenic reactors (Liu et al. 2018). As a caveat, the fact that some industrial-scale anaerobic digesters can operate successfully at temperatures around 25 °C (Liebetrau et al. 2019) suggests that this strategy alone (i.e., operating closer to psychrophilic temperatures) might not be sufficient to outcompete methanogens.
Reactions able to generate ATP through substrate-level phosphorylation, i.e., ethanol and lactate oxidation, are thermodynamically disfavored at lower temperature. However, this might not have a prohibiting effect of the overall CE reaction since most of the ATP is generated through the electron bifurcation-ATP synthase system (Angenent et al. 2016). In any case, it must be carried in mind that temperature has also strong effects on kinetic phenomena.
Adapting syngas fermentation reactors to communities
Be it in a bubbled column, gas-lift, or in stirred reactors, SF depends on high bubbling flow rates to overcome gas-liquid mass transfer limitations. Intensive bubbling in stirred reactors also offers the secondary advantage of lowering the power input of stirring (Takors et al. 2018). At first glance, such high gas flow rate seems to be incompatible with the typically low consumption rates of syngas by mixed cultures when high H2 and CO conversion is desired (Molitor et al. 2017). Syngas might be consumed at even lower rates when used as a co-substrate with lactate or ethanol. Nevertheless, excess H2 and CO in the broth is supposed to affect oxidative-reductive potentials and ratios of reduced electron carriers in the cell, ultimately affecting the metabolic pathways and steering product pools (de Kok et al. 2013; Esquivel-Elizondo et al. 2018). In that sense, recirculating gas—in contraposition to the one-pass strategy—can fit well with mixed culture reactors (Fig. 4) despite still few examples (such as in the setups used by Richter et al. (2016a) and Guiot et al. (2011)) among recent studies. This is partly due to the fact that SF reactor systems have been usually developed for pure cultures, as seen in the reactor schemes described by Asimakopoulos et al. (2018).
When adopting current knowledge of SF (mostly obtained with pure cultures) to fermentation with microbial communities, process strategies have to be adapted: conversely to the conventional single-pass gas strategy in (a), gas recirculation and substrate biomass co-feeding (b) can be more compatible to the typically lower gas consumption rates of open communities
With a gas recirculation strategy as illustrated in Fig. 4b, the syngas flow rate can, in principle, be kept high regardless of the community’s gas consumption rate. By this way, nearly complete H2 and CO conversion can be achieved without lowering the gas-liquid transfer coefficient, kla, described in Eq. 2.
Operation with gas recirculation means that gas composition fluctuates naturally along time. Thus, possible accumulation of inhibitors, inert gases, and methane, and the intermittent feeding and purging cycles would need to be studied. Also as a consequence, partial pressures of H2 and CO are expected to decrease along each gas restocking cycle.
The first reason to avoid operating at too low H2 partial pressures during gas recirculation is that it can selectively favor HM considering that hydrogenotrophic methanogens are known to have higher affinity to H2 (Heimann et al. 2009). The other reason is that the amount of gaseous substrate in the aqueous phase in the equilibrium, \( {C}_g^{\ast } \), depends directly on the partial pressure of gas, pg, according to Henry’s law as seen in Eq. 1. In the kinetic regime, the de facto available gaseous substrate to microorganisms, Cg, also depends on \( {C}_g^{\ast } \) according to the rate Eq. 2, described for a 1st-order one-dimensional diffusion. Consequently, a low pg of the substrate gas also lowers mass transfer rates. Such transient characteristic of gas recirculation and its effect on the community metabolism would also have to be taken into account (Yasin et al. 2015).
For Eqs. 1 and 2: \( {C}_g^{\ast } \) is the equilibrium concentration of gas in the liquid, in M; H is the Henry coefficient for the gas, in M/atm; pg is the partial pressure of the gas, in atm; kla is the coefficient of gas-liquid transfer per area times the interfacial gas-liquid area per volume, in 1/h; and Cg is the concentration of gas in the liquid, in M. kla depends mainly on the gas flow rate, on the strategy for liquid-gas contact in the reactor and on the gas diffusivities.
CO2 is not commonly the limiting gaseous substrate; nevertheless, the concentration of this gas can become limiting in some cases in acidogenic systems (Vasudevan et al. 2014). The solubility of CO2 is controlled mainly by chemical equilibria of the carbonate system. As a consequence, CO2 solubility is highly dependent on the pH for values between 5.0 and 8.0 as seen in Fig. 5. Most acidogenic bioreactors work within this pH range of greatest CO2 solubility changes. At pH values lower than 5.0, CO2 solubility is lowest, though water still harbors a significant fraction of CO2 (roughly 45%) in the form of aqueous CO2.
Percentages of CO2 species in the gas and in the aqueous phase as function of pH at 25°C for a closed system with 50% headspace and a diluted aqueous phase. For not too high CO2 partial pressures (< 1000 kPa), this pattern is independent of the total amount of CO2 in the system (Diamond and Akinfiev 2003). The carbonic acid species is present in very small amounts and, therefore, it is not visible in the graph. Calculations done similarly to Seinfeld and Pandis (2016). Henry’s law and dissociation constants obtained from Sander (2015) and Greenwood and Earnshaw (2012), respectively
Product downstream processing
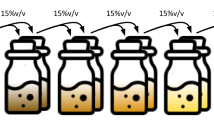
Traditionally, anaerobic fermentation research has taken profit from lessons learned in anaerobic digestion research (Agler et al. 2014). Nevertheless, one specific challenge of anaerobic fermentations is product downstream processing (DSP). Unlike methane, the desired product in anaerobic digestion, linear MCC and MCA are liquids under ambient conditions and form aqueous solutions. Thus, the DSP technology classically adopted in the ethanol and acetate industry, distillation, could be considered as an alternative for purification of their longer-chain counterparts. However, high operational costs could incur on distillation of MCC and MCA because their concentrations in the broth are generally lower than 2% w/v (i.e., 20 g/L) (Arslan et al. 2016), in comparison with the typical concentrations in ethanol fermentation of at least 6% w/v (de S Dias et al. 2015).
the solubility of MCC and MCA in water decreases the longer the hydrophobic part of the carbon chain is. As a consequence, in an industrial scale, n-caproate and n-caprylate could have lower DSP costs due to their lower solubility at a defined pH, in comparison with n-butyrate, which is miscible in water along the entire pH range. This feature was explored in previous studies where the bioreactor was coupled to a pertraction (liquid-liquid extraction) technology (Agler et al. 2014; Ge et al. 2015). Differently to organic acids, solubility of alcohols cannot be altered by changing pH and their DSP would be restricted, essentially, to the techniques of traditional solvent-water extraction, such as ‘salting out’. This factor might be decisive to favor MCC production, while further processing for production of MCA could adopt non-biological conversion routes. The bigger ease of MCC purification can be a central factor for choosing them as a chemical platform for MCA and alkanes production (Pham et al. 2010). Further processing of MCC is a promising topic and the case for alkane production through Kolbe hydrolysis of MCC produced by CE, as shown by Urban et al. (2017), can be taken as a good example.
Stoichiometry and economic feasibility of the process
The most fundamental limitation of feedstock biomass conversion is imposed by the overall stoichiometry of the process. The stoichiometry of the process depends on the substrate, on the final product, and on the pathways used to reach it. Neglecting the inorganic fraction of dry lignocellulosic biomass and assuming the chemical formula of cellulose (C6H10O5), Fig. 6 compares the maximum stoichiometric yields for some carboxylates, alcohols, and methane. Without considering the intricacies of the conversion of recalcitrant fractions, both major processes for making biomass bioavailable (i.e., hydrolysis and biomass gasification) present equivalent theoretical stoichiometric yields. Big differences in the maximum stoichiometric conversion appear depending on the ability of the biocatalyst to realize autotrophy or not, as seen for acetate in Fig. 6. Cultures that grow only heterotrophically do not reincorporate H2 and CO2 produced in the fermentation of biomass-derived sugar and have, therefore, lower maximum conversion to carboxylates (Schuchmann and Müller 2016). For methane production, stoichiometric conversion from biomass (cellulose) is inherently low regardless if it is produced through hydrogenotrophic, acetotrophic, or methylotrophic methanogenesis.
Stoichiometric ‘hard caps’ of biomass derivatives. The chemical formula of cellulose is assumed for the feedstock biomass. 1 mol of cellulose consumes 1 mol of water through hydrolysis or through gasification producing water-shifted syngas (H2/CO2). The reactions for n-butyrate, n-caproate, and n-caprylate are for bioprocesses, in which the produced H2 and CO2 during substrate biomass conversion can be reincorporated into the carboxylate pool, such as syngas-aided CE
It is also evident from Fig. 6 that the potential for carbon fixation and the maximum stoichiometric conversion lowers with increasing chain length of the carboxylate or alcohol. Still, production of chemicals beyond acetate and ethanol from syngas is a promising concept as a bioprocess and its success as an industrial process depends basically on ensuring a sufficient conversion of syngas to chemicals in the bioreactor by using inherently low-cost processes. Considering dry biomass as the raw material (with the cellulose chemical formula) and the same assumptions adopted for calculating the maximum stoichiometric conversions, Fig. 7 presents the cost gaps for each product of different bioprocess concepts, in comparison with other more mature processes.
Different alternative uses of dry, feedstock biomass in US$ (2016) on a kg of biomass basis. The chart considers maximum stoichiometric conversions together with market prices on a single basis of kg of biomass (cellulose). A generic cost of 0.05 $/kg of dry biomass is assumed. *Considering 1 kWh per 1 kg of dry biomass and 0.12 $/kWh. Elaborated based on information from Biddy et al. (2016); de Medeiros et al. (2017); DOE (2016); Granda (2015); Kleerebezem and van Loosdrecht (2007)
Each kilogram of dry biomass, when converted to syngas, has the highest margin for conversion loss and process costs if used to produce n-caproate, 1.61 $/kg dry biomass, in comparison with 0.40 $/kg dry biomass when used to produce ethanol. This is due to the actual high market price of n-caproate of around 3.00 $/kg (Granda 2015). However, it is worth pointing out that prices per kilogram for commodity-grade chemicals like ethanol are generally much lower than specialty chemicals like n-caproate (de Medeiros et al. 2017). Despite many possible applications, its market is still small in comparison with acetate, ethanol, and butanol. If a MCC, such as n-caproate, eventually becomes a chemical platform, its price would also have to lower to attend a broader range of applications.
Open questions and future perspectives
The mechanisms of syngas-based n-caproate and n-caprylate production by single species like C. carboxidivorans or E. limosum are still not completely clear. Although the complete WLP was found in these species, their CE pathways and how electrons can be shifted from H2 and CO to C6 and C8 compounds are still poorly understood (Bengelsdorf et al. 2018; Ganigué et al. 2016; Zhang et al. 2016). Such knowledge could be useful, for instance, to manage these strains in mixotrophic growth, steering carbon and electrons from multiple low-cost sources into the desired product.
The knowledge gaps regarding syngas-aided CE in microbial communities are plenty. Undiscovered syngas-fermenting, MCC-producing strains could explain some cases where C6 and C8 compounds were produced in significant amounts from syngas. Besides, interspecies ethanol transfer remains the most popular assumption for the observed phenomenon even though it was not yet systematically tested. More detailed studies on the mechanisms underlying the reported n-caproate and n-caprylate formation can reveal overlooked metabolic shifts facilitated by co-fed H2 and CO. Specifically, the ethanol-intermediated assumption is not enough to explain the effect of syngas on mixed cultures specialized in lactate-based CE. Further studies about the effect of syngas addition in lactate-based CE communities are needed and the transcriptome analysis done by Scarborough et al. (2018a) is a good example of that.
Formation of odd-numbered MCC also remains a topic that deserves more study. More understanding on the necessary operational parameters to incorporate propionate and propionyl-CoA to the RBO pathway could extend the product spectrum of MCC and MCA to their odd-numbered counterparts.
Overall, there are still few studies about syngas-aided CE with microbial communities and even fewer studies considering the use of real waste substrates or focusing on the process optimization. This may be partially due to the inherent difficulty to conciliate two types of substrate with very different consumption kinetics in anaerobic communities (e.g., H2 opposed to ethanol or lactate). Since syngas consumption rates by communities are generally low, we propose here a simple reactor operation strategy, with gas recycling, for coupling the two types of substrate (liquid and syngas substrate) to simultaneously keep syngas conversion high while avoiding limitation by gas-liquid mass transfer. Future technical studies should also analyze how competing pathways (such as hydrogenotrophic methanogenesis) could be managed in an up-scalable manner when feeding the reactor with real waste biomass. Moreover, testing process scalability—with and without in-line product extraction—could contribute greatly to understand further challenges and synergies of combining the syngas and carboxylate platforms with undefined mixed cultures.
References
Agler MT, Werner JJ, Iten LB, Dekker A, Cotta MA, Dien BS, Angenent LT (2012) Shaping reactor microbiomes to produce the fuel precursor n-butyrate from pretreated cellulosic hydrolysates. Environ Sci Technol 46(18):10229–10238. https://doi.org/10.1021/es302352c
Agler MT, Spirito CM, Usack JG, Werner JJ, Angenent LT (2014) Development of a highly specific and productive process for n-caproic acid production: applying lessons from methanogenic microbiomes. Water Sci Technol 69(1):62–68. https://doi.org/10.2166/wst.2013.549
Angenent LT, Richter H, Buckel W, Spirito CM, Steinbusch KJ, Plugge CM, Strik DP, Grootscholten TI, Buisman CJ, Hamelers HV (2016) Chain elongation with reactor microbiomes: open-culture biotechnology to produce biochemicals. Environ Sci Technol 50(6):2796–2810. https://doi.org/10.1021/acs.est.5b04847
Arslan D, Steinbusch KJ, Diels L, De Wever H, Buisman CJ, Hamelers HV (2012) Effect of hydrogen and carbon dioxide on carboxylic acids patterns in mixed culture fermentation. Bioresour Technol 118:227–234. https://doi.org/10.1016/j.biortech.2012.05.003
Arslan D, Steinbusch KJJ, Diels L, Hamelers HVM, Strik DPBTB, Buisman CJN, De Wever H (2016) Selective short-chain carboxylates production: a review of control mechanisms to direct mixed culture fermentations. Crit Rev Environ Sci Technol 46(6):592–634. https://doi.org/10.1080/10643389.2016.1145959
Asimakopoulos K, Gavala HN, Skiadas IV (2018) Reactor systems for syngas fermentation processes: a review. Chem Eng J 348:732–744. https://doi.org/10.1016/j.cej.2018.05.003
Bengelsdorf FR, Dürre P (2017) Gas fermentation for commodity chemicals and fuels. Microb Biotechnol 10(5):1167–1170. https://doi.org/10.1111/1751-7915.12763
Bengelsdorf FR, Beck MH, Erz C, Hoffmeister S, Karl MM, Riegler P, Wirth S, Poehlein A, Weuster-Botz D, Dürre P (2018) Bacterial anaerobic synthesis gas (syngas) and CO2+H2 fermentation. Adv Appl Microbiol 103:143–221. https://doi.org/10.1016/bs.aambs.2018.01.002
Biddy MJ, Scarlata C, Kinchin C (2016) Chemicals from biomass: a market assessment of bioproducts with near-term potential. National Renewable Energy Lab (NREL), Golden, CO (United States)
Björnsson L, Murto M, Mattiasson B (2000) Evaluation of parameters for monitoring an anaerobic co-digestion process. Appl Microbiol Biotechnol 54(6):844–849
Buckel W, Thauer RK (2013) Energy conservation via electron bifurcating ferredoxin reduction and proton/Na(+) translocating ferredoxin oxidation. Biochim Biophys Acta 1827(2):94–113. https://doi.org/10.1016/j.bbabio.2012.07.002
Cavalcante WA, Leitão RC, Gehring TA, Angenent LT, Santaella ST (2017) Anaerobic fermentation for n-caproic acid production: a review. Process Biochem 54:106–119. https://doi.org/10.1016/j.procbio.2016.12.024
Chahal SP, Starr J (2006) Lactic Acid, Ullmann’s Encyclopedia of Industrial Chemistry. Wiley-VCH, GmbH & Co. KGaA, Weinheim
Chakraborty S, Rene ER, Lens PNL, Veiga MC, Kennes C (2019) Enrichment of a solventogenic anaerobic sludge converting carbon monoxide and syngas into acids and alcohols. Bioresour Technol 272:130–136. https://doi.org/10.1016/j.biortech.2018.10.002
Chapleur O, Madigou C, Civade R, Rodolphe Y, Mazeas L, Bouchez T (2016) Increasing concentrations of phenol progressively affect anaerobic digestion of cellulose and associated microbial communities. Biodegradation 27(1):15–27. https://doi.org/10.1007/s10532-015-9751-4
Chen WS, Ye Y, Steinbusch KJJ, Strik DPBTB, Buisman CJN (2016) Methanol as an alternative electron donor in chain elongation for butyrate and caproate formation. Biomass Bioenergy 93:201–208. https://doi.org/10.1016/j.biombioe.2016.07.008
Chen WS, Strik D, Buisman CJN, Kroeze C (2017) Production of caproic acid from mixed organic waste: an environmental life cycle perspective. Environ Sci Technol 51(12):7159–7168. https://doi.org/10.1021/acs.est.6b06220
Costa KC, Leigh JA (2014) Metabolic versatility in methanogens. Curr Opin Biotechnol 29:70–75. https://doi.org/10.1016/j.copbio.2014.02.012
Cueto-Rojas HF, van Maris AJ, Wahl SA, Heijnen JJ (2015) Thermodynamics-based design of microbial cell factories for anaerobic product formation. Trends Biotechnol 33(9):534–546. https://doi.org/10.1016/j.tibtech.2015.06.010
De Groof V, Coma M, Arnot T, Leak DJ, Lanham AB (2019) Medium chain carboxylic acids from complex organic feedstocks by mixed culture fermentation. Molecules 24(3). https://doi.org/10.3390/molecules24030398
de Jong E, Higson A, Walsh P, Wellisch M (2012) Bio-based chemicals value added products from biorefineries. IEA Bioenergy, Task42 Biorefinery:34
de Kok S, Meijer J, van Loosdrecht MC, Kleerebezem R (2013) Impact of dissolved hydrogen partial pressure on mixed culture fermentations. Appl Microbiol Biotechnol 97(6):2617–2625. https://doi.org/10.1007/s00253-012-4400-x
de Leeuw K, Buisman CJN, Strik DPBTB (2019) Branched medium chain fatty acids: iso-caproate formation from iso-butyrate broadens the product spectrum for microbial chain elongation. Environ Sci Technol 53:7704–7713. https://doi.org/10.1021/acs.est.8b07256
de Medeiros EM, Posada JA, Noorman H, Osseweijer P, Filho RM (2017) Hydrous bioethanol production from sugarcane bagasse via energy self-sufficient gasification-fermentation hybrid route: Simulation and financial analysis. J Clean Prod 168:1625–1635. https://doi.org/10.1016/j.jclepro.2017.01.165
de S Dias MO, Maciel Filho R, Mantelatto PE, Cavalett O, Rossell CEV, Bonomi A, Leal MRLV (2015) Sugarcane processing for ethanol and sugar in Brazil. Environ Dev 15:35–51. https://doi.org/10.1016/j.envdev.2015.03.004
de Smit SM, de Leeuw KD, Buisman CJN, Strik D (2019) Continuous n-valerate formation from propionate and methanol in an anaerobic chain elongation open-culture bioreactor. Biotechnol Biofuels 12:132. https://doi.org/10.1186/s13068-019-1468-x
de Vrieze J, Coma M, Debeuckelaere M, Van der Meeren P, Rabaey K (2016) High salinity in molasses wastewaters shifts anaerobic digestion to carboxylate production. Water Res 98:293–301. https://doi.org/10.1016/j.watres.2016.04.035
Diamond LW, Akinfiev NN (2003) Solubility of CO2 in water from −1.5 to 100 °C and from 0.1 to 100 MPa: evaluation of literature data and thermodynamic modelling. Fluid Phase Equilib 208(1-2):265–290. https://doi.org/10.1016/s0378-3812(03)00041-4
Diender M, Stams AJ, Sousa DZ (2015) Pathways and bioenergetics of anaerobic carbon monoxide fermentation. Front Microbiol 6:1275. https://doi.org/10.3389/fmicb.2015.01275
Diender M, Stams AJ, Sousa DZ (2016) Production of medium-chain fatty acids and higher alcohols by a synthetic co-culture grown on carbon monoxide or syngas. Biotechnol Biofuels 9:82. https://doi.org/10.1186/s13068-016-0495-0
Ding HB, Tan GY, Wang JY (2010) Caproate formation in mixed-culture fermentative hydrogen production. Bioresour Technol 101(24):9550–9559. https://doi.org/10.1016/j.biortech.2010.07.056
DOE (2016) Biomass for electricity generation. United States Department of Energy. https://www.wbdg.org/resources/biomass-electricity-generation. Accessed 25 Jan 2019
Doud DFR, Holmes EC, Richter H, Molitor B, Jander G, Angenent LT (2017) Metabolic engineering of Rhodopseudomonas palustris for the obligate reduction of n-butyrate to n-butanol. Biotechnol Biofuels 10:178. https://doi.org/10.1186/s13068-017-0864-3
Endres H-J, Siebert-Raths A (2009) Technische Biopolymere. Rahmenbedingungen, Marktsituation, Herstellung, Aufbau und Eigenschaften 1st edn München: Hanser
Esquivel-Elizondo S, Delgado AG, Rittmann BE, Krajmalnik-Brown R (2017) The effects of CO2 and H2 on CO metabolism by pure and mixed microbial cultures. Biotechnol Biofuels 10:220. https://doi.org/10.1186/s13068-017-0910-1
Esquivel-Elizondo S, Miceli J 3rd, Torres CI, Krajmalnik-Brown R (2018) Impact of carbon monoxide partial pressures on methanogenesis and medium chain fatty acids production during ethanol fermentation. Biotechnol Bioeng 115(2):341–350. https://doi.org/10.1002/bit.26471
Fernández-Naveira Á, Abubackar HN, Veiga MC, Kennes C (2016) Efficient butanol-ethanol (B-E) production from carbon monoxide fermentation by Clostridium carboxidivorans. Appl Microbiol Biotechnol 100(7):3361–3370. https://doi.org/10.1007/s00253-015-7238-1
Fernández-Naveira Á, Veiga MC, Kennes C (2017) H-B-E (hexanol-butanol-ethanol) fermentation for the production of higher alcohols from syngas/waste gas. J Chem Technol Biotechnol 92(4):712–731. https://doi.org/10.1002/jctb.5194
Fernández-Naveira Á, Veiga MC, Kennes C (2019) Selective anaerobic fermentation of syngas into either C2-C6 organic acids or ethanol and higher alcohols. Bioresour Technol 280:387–395. https://doi.org/10.1016/j.biortech.2019.02.018
Ganigué R, Ramió-Pujol S, Sanchez P, Baneras L, Colprim J (2015) Conversion of sewage sludge to commodity chemicals via syngas fermentation. Water Sci Technol 72(3):415–420. https://doi.org/10.2166/wst.2015.222
Ganigué R, Sánchez-Paredes P, Bañeras L, Colprim J (2016) Low fermentation pH is a trigger to alcohol production, but a killer to chain elongation. Front Microbiol 7:702. https://doi.org/10.3389/fmicb.2016.00702
Garvie EI (1980) Bacterial lactate dehydrogenases. Microbiol Rev 44(1):106
Ge S, Usack JG, Spirito CM, Angenent LT (2015) Long-term n-caproic acid production from yeast-fermentation beer in an anaerobic bioreactor with continuous product extraction. Environ Sci Technol 49(13):8012–8021. https://doi.org/10.1021/acs.est.5b00238
Gildemyn S, Molitor B, Usack JG, Nguyen M, Rabaey K, Angenent LT (2017) Upgrading syngas fermentation effluent using Clostridium kluyveri in a continuous fermentation. Biotechnol Biofuels 10:83. https://doi.org/10.1186/s13068-017-0764-6
González-Cabaleiro R, Lema JM, Rodríguez J, Kleerebezem R (2013) Linking thermodynamics and kinetics to assess pathway reversibility in anaerobic bioprocesses. Energy Environ Sci 6(12):3780. https://doi.org/10.1039/c3ee42754d
González-Cabaleiro R, Lema JM, Rodríguez J (2015) Metabolic energy-based modelling explains product yielding in anaerobic mixed culture fermentations. PLoS One 10(5):e0126739. https://doi.org/10.1371/journal.pone.0126739
Granda CB Production of highly pure fatty acids from anaerobic digestion. In: World Congress on Industrial Biotechnology, Montréal, 2015
Greenwood NN, Earnshaw A (2012) Chemistry of the Elements. Elsevier
Grimalt-Alemany A, Lezyk M, Lange L, Skiadas IV, Gavala HN (2018) Enrichment of syngas-converting mixed microbial consortia for ethanol production and thermodynamics-based design of enrichment strategies. Biotechnol Biofuels 11(1):198. https://doi.org/10.1186/s13068-018-1189-6
Grootscholten TI, Steinbusch KJ, Hamelers HV, Buisman CJ (2013) Improving medium chain fatty acid productivity using chain elongation by reducing the hydraulic retention time in an upflow anaerobic filter. Bioresour Technol 136:735–738. https://doi.org/10.1016/j.biortech.2013.02.114
Grootscholten TIM, Strik DPBTB, Steinbusch KJJ, Buisman CJN, Hamelers HVM (2014) Two-stage medium chain fatty acid (MCFA) production from municipal solid waste and ethanol. Appl Energy 116:223–229. https://doi.org/10.1016/j.apenergy.2013.11.061
Guiot SR, Cimpoia R, Carayon G (2011) Potential of wastewater-treating anaerobic granules for biomethanation of synthesis gas. Environ Sci Technol 45(5):2006–2012. https://doi.org/10.1021/es102728m
He P, Han W, Shao L, Lu F (2018) One-step production of C6-C8 carboxylates by mixed culture solely grown on CO. Biotechnol Biofuels 11:4. https://doi.org/10.1186/s13068-017-1005-8
Heimann A, Jakobsen R, Blodau C (2009) Energetic constraints on H2-dependent terminal electron accepting processes in anoxic environments: a review of observations and model approaches. Environ Sci Technol 44(1):24–33
Hu Y, Jing Z, Sudo Y, Niu Q, Du J, Wu J, Li YY (2015) Effect of influent COD/SO42- ratios on UASB treatment of a synthetic sulfate-containing wastewater. Chemosphere 130:24–33. https://doi.org/10.1016/j.chemosphere.2015.02.019
Jankowska E, Duber A, Chwialkowska J, Stodolny M, Oleskowicz-Popiel P (2018) Conversion of organic waste into volatile fatty acids – the influence of process operating parameters. Chem Eng J 345:395–403. https://doi.org/10.1016/j.cej.2018.03.180
Jaros A, Rova U, Berglund K (2012) Effect of acetate on fermentation production of butyrate. Cellul Chem Technol 46(5-6):341–347
Jeon BS, Choi O, Um Y, Sang BI (2016) Production of medium-chain carboxylic acids by Megasphaera sp. MH with supplemental electron acceptors. Biotechnol Biofuels 9:129. https://doi.org/10.1186/s13068-016-0549-3
Jourdin L, Raes SMT, Buisman CJN, Strik DPBTB (2018) Critical biofilm growth throughout unmodified carbon felts allows continuous bioelectrochemical chain elongation from CO2 up to caproate at high current density. Front Energy Res 6. https://doi.org/10.3389/fenrg.2018.00007
Kenealy WR, Waselefsky DM (1985) Studies on the substrate range of Clostridium kluyveri; the use of propanol and succinate. Arch Microbiol 141(3):187–194
Kleerebezem R, Stams AJ (2000) Kinetics of syntrophic cultures: a theoretical treatise on butyrate fermentation. Biotechnol Bioeng 67(5):529–543
Kleerebezem R, van Loosdrecht MC (2007) Mixed culture biotechnology for bioenergy production. Curr Opin Biotechnol 18(3):207–212. https://doi.org/10.1016/j.copbio.2007.05.001
Kleerebezem R, Van Loosdrecht MCM (2010) A generalized method for thermodynamic state analysis of environmental systems. Crit Rev Environ Sci Technol 40(1):1–54. https://doi.org/10.1080/10643380802000974
Köpke M, Held C, Hujer S, Liesegang H, Wiezer A, Wollherr A, Ehrenreich A, Liebl W, Gottschalk G, Dürre P (2010) Clostridium ljungdahlii represents a microbial production platform based on syngas. Proc Natl Acad Sci U S A 107(29):13087–13092. https://doi.org/10.1073/pnas.1004716107
Kosaric N, Duvnjak Z, Farkas A, Sahm H, Bringer-Meyer S, Goebel O, Mayer D (2000) Ethanol. In: Ullmann’s encyclopedia of industrial chemistry. Wiley-VCH, GmbH & Co. KGaA, Weinheim
Koster IW, Koomen E (1988) Ammonia inhibition of the maximum growth rate (μm) of hydrogenotrophic methanogens at various pH-levels and temperatures. Appl Microbiol Biotechnol 28(4-5):500–505
Kremp F, Poehlein A, Daniel R, Müller V (2018) Methanol metabolism in the acetogenic bacterium Acetobacterium woodii. Environ Microbiol 20(12):4369–4384. https://doi.org/10.1111/1462-2920.14356
Kucek LA, Nguyen M, Angenent LT (2016a) Conversion of L-lactate into n-caproate by a continuously fed reactor microbiome. Water Res 93:163–171. https://doi.org/10.1016/j.watres.2016.02.018
Kucek LA, Spirito CM, Angenent LT (2016b) High n-caprylate productivities and specificities from dilute ethanol and acetate: chain elongation with microbiomes to upgrade products from syngas fermentation. Energy Environ Sci 9(11):3482–3494. https://doi.org/10.1039/c6ee01487a
Kucek LA, Xu J, Nguyen M, Angenent LT (2016c) Waste conversion into n-caprylate and n-caproate: resource recovery from wine lees using anaerobic reactor microbiomes and in-line extraction. Front Microbiol 7:1892. https://doi.org/10.3389/fmicb.2016.01892
Lambrecht J, Cichocki N, Schattenberg F, Kleinsteuber S, Harms H, Müller S, Sträuber H (2019) Key sub-community dynamics of medium-chain carboxylate production. Microb Cell Factories 18(1):92. https://doi.org/10.1186/s12934-019-1143-8
Latif H, Zeidan AA, Nielsen AT, Zengler K (2014) Trash to treasure: production of biofuels and commodity chemicals via syngas fermenting microorganisms. Curr Opin Biotechnol 27:79–87. https://doi.org/10.1016/j.copbio.2013.12.001
Liebetrau J, Weinrich S, Sträuber H, Kretzschmar J (2019) Anaerobic fermentation of organic material: biological processes and their control parameters. energy from organic materials (biomass). Encyclopedia of Sustainability Science and Technology Series. Springer, New York, pp 779–807
Liew F, Henstra AM, Winzer K, Köpke M, Simpson SD, Minton NP (2016a) Insights into CO2 fixation pathway of Clostridium autoethanogenum by targeted mutagenesis. MBio 7(3). https://doi.org/10.1128/mBio.00427-16
Liew F, Martin ME, Tappel RC, Heijstra BD, Mihalcea C, Kopke M (2016b) Gas fermentation - a flexible platform for commercial scale production of low-carbon-fuels and chemicals from waste and renewable feedstocks. Front Microbiol 7:694. https://doi.org/10.3389/fmicb.2016.00694
Lindley N, Loubiere P, Pacaud S, Mariotto C, Goma G (1987) Novel products of the acidogenic fermentation of methanol during growth of Eubacterium limosum in the presence of high concentrations of organic acids. Microbiology 133(12):3557–3563
Liou JS, Balkwill DL, Drake GR, Tanner RS (2005) Clostridium carboxidivorans sp. nov., a solvent-producing clostridium isolated from an agricultural settling lagoon, and reclassification of the acetogen Clostridium scatologenes strain SL1 as Clostridium drakei sp. nov. Int J Syst Evol Microbiol 55(Pt 5):2085–2091. https://doi.org/10.1099/ijs.0.63482-0
Liu H, Wang J, Wang A, Chen J (2011) Chemical inhibitors of methanogenesis and putative applications. Appl Microbiol Biotechnol 89(5):1333–1340. https://doi.org/10.1007/s00253-010-3066-5
Liu K, Atiyeh HK, Stevenson BS, Tanner RS, Wilkins MR, Huhnke RL (2014) Mixed culture syngas fermentation and conversion of carboxylic acids into alcohols. Bioresour Technol 152:337–346. https://doi.org/10.1016/j.biortech.2013.11.015
Liu C, Luo G, Wang W, He Y, Zhang R, Liu G (2018) The effects of pH and temperature on the acetate production and microbial community compositions by syngas fermentation. Fuel 224:537–544. https://doi.org/10.1016/j.fuel.2018.03.125
Marounek M, Fliegrova K, Bartos S (1989) Metabolism and some characteristics of ruminal strains of Megasphaera elsdenii. Appl Environ Microbiol 55(6):1570–1573
Mock J, Zheng Y, Mueller AP, Ly S, Tran L, Segovia S, Nagaraju S, Köpke M, Dürre P, Thauer RK (2015) Energy conservation associated with ethanol formation from H2 and CO2 in Clostridium autoethanogenum involving electron bifurcation. J Bacteriol 197(18):2965–2980. https://doi.org/10.1128/JB.00399-15
Molitor B, Richter H, Martin ME, Jensen RO, Juminaga A, Mihalcea C, Angenent LT (2016) Carbon recovery by fermentation of CO-rich off gases - Turning steel mills into biorefineries. Bioresour Technol 215:386–396. https://doi.org/10.1016/j.biortech.2016.03.094
Molitor B, Marcellin E, Angenent LT (2017) Overcoming the energetic limitations of syngas fermentation. Curr Opin Chem Biol 41:84–92. https://doi.org/10.1016/j.cbpa.2017.10.003
Nzeteu CO, Trego AC, Abram F, O’Flaherty V (2018) Reproducible, high-yielding, biological caproate production from food waste using a single-phase anaerobic reactor system. Biotechnol Biofuels 11:108. https://doi.org/10.1186/s13068-018-1101-4
Oswald F, Zwick M, Omar O, Hotz EN, Neumann A (2018) Growth and product formation of Clostridium ljungdahlii in presence of cyanide. Front Microbiol 9:1213. https://doi.org/10.3389/fmicb.2018.01213
Pacaud S, Loubiere P, Goma G (1985) Methanol metabolism by Eubacterium limosum B2: Effects of pH and carbon dioxide on growth and organic acid production. Curr Microbiol 12(5):245–250
Pham V, Holtzapple M, El-Halwagi M (2010) Techno-economic analysis of biomass to fuel conversion via the MixAlco process. J Ind Microbiol Biotechnol 37(11):1157–1168. https://doi.org/10.1007/s10295-010-0763-0
Phillips JR, Atiyeh HK, Tanner RS, Torres JR, Saxena J, Wilkins MR, Huhnke RL (2015) Butanol and hexanol production in Clostridium carboxidivorans syngas fermentation: Medium development and culture techniques. Bioresour Technol 190:114–121. https://doi.org/10.1016/j.biortech.2015.04.043
Plugge CM, Zhang W, Scholten JC, Stams AJ (2011) Metabolic flexibility of sulfate-reducing bacteria. Front Microbiol 2:81. https://doi.org/10.3389/fmicb.2011.00081
Popp D, Harms H, Sträuber H (2016) The alkaloid gramine in the anaerobic digestion process-inhibition and adaptation of the methanogenic community. Appl Microbiol Biotechnol 100(16):7311–7322. https://doi.org/10.1007/s00253-016-7571-z
Prabhu R, Altman E, Eiteman MA (2012) Lactate and acrylate metabolism by Megasphaera elsdenii under batch and steady-state conditions. Appl Environ Microbiol 78(24):8564–8570. https://doi.org/10.1128/AEM.02443-12
Rabaey K, Rozendal RA (2010) Microbial electrosynthesis - revisiting the electrical route for microbial production. Nat Rev Microbiol 8(10):706–716. https://doi.org/10.1038/nrmicro2422
Ragsdale SW (2004) Life with carbon monoxide. Crit Rev Biochem Mol Biol 39(3):165–195. https://doi.org/10.1080/10409230490496577
Ramachandriya KD, Kundiyana DK, Wilkins MR, Terrill JB, Atiyeh HK, Huhnke RL (2013) Carbon dioxide conversion to fuels and chemicals using a hybrid green process. Appl Energy 112:289–299. https://doi.org/10.1016/j.apenergy.2013.06.017
Ramió-Pujol S, Ganigué R, Bañeras L, Colprim J (2015) Incubation at 25°C prevents acid crash and enhances alcohol production in Clostridium carboxidivorans P7. Bioresour Technol 192:296–303. https://doi.org/10.1016/j.biortech.2015.05.077
Reddy MV, ElMekawy A, Pant D (2018) Bioelectrochemical synthesis of caproate through chain elongation as a complementary technology to anaerobic digestion. Biofuels Bioprod Biorefin 12(6):966–977. https://doi.org/10.1002/bbb.1924
Richter H, Loftus SE, Angenent LT (2013) Integrating syngas fermentation with the carboxylate platform and yeast fermentation to reduce medium cost and improve biofuel productivity. Environ Technol 34(13-16):1983–1994. https://doi.org/10.1080/09593330.2013.826255
Richter H, Molitor B, Diender M, Sousa DZ, Angenent LT (2016a) A narrow pH range supports butanol, hexanol, and octanol production from syngas in a continuous co-culture of Clostridium ljungdahlii and Clostridium kluyveri with in-line product extraction. Front Microbiol 7:1773. https://doi.org/10.3389/fmicb.2016.01773
Richter H, Molitor B, Wei H, Chen W, Aristilde L, Angenent LT (2016b) Ethanol production in syngas-fermenting Clostridium ljungdahlii is controlled by thermodynamics rather than by enzyme expression. Energy Environ Sci 9(7):2392–2399. https://doi.org/10.1039/c6ee01108j
Roghair M, Hoogstad T, Strik D, Plugge CM, Timmers PHA, Weusthuis RA, Bruins ME, Buisman CJN (2018a) Controlling ethanol use in chain elongation by CO2 loading rate. Environ Sci Technol 52(3):1496–1505. https://doi.org/10.1021/acs.est.7b04904
Roghair M, Liu Y, Adiatma JC, Weusthuis RA, Bruins ME, Buisman CJN, Strik D (2018b) Effect of n-caproate concentration on chain elongation and competing processes. ACS Sustain Chem Eng 6(6):7499–7506. https://doi.org/10.1021/acssuschemeng.8b00200
Sander R (2015) Compilation of Henry's law constants (version 4.0) for water as solvent. Atmos Chem Phys 15(8):4399–4981. https://doi.org/10.5194/acp-15-4399-2015
Savant DV, Shouche YS, Prakash S, Ranade DR (2002) Methanobrevibacter acididurans sp. nov., a novel methanogen from a sour anaerobic digester. Int J Syst Evol Microbiol 52(Pt 4):1081–1087. https://doi.org/10.1099/00207713-52-4-1081
Scarborough MJ, Lawson CE, Hamilton JJ, Donohue TJ, Noguera DR (2018a) Metatranscriptomic and thermodynamic insights into medium-chain fatty acid production using an anaerobic microbiome. mSystems 3(6):e00221-18
Scarborough MJ, Lynch G, Dickson M, McGee M, Donohue TJ, Noguera DR (2018b) Increasing the economic value of lignocellulosic stillage through medium-chain fatty acid production. Biotechnol Biofuels 11:200. https://doi.org/10.1186/s13068-018-1193-x
Schoberth S, Gottschalk G (1969) Considerations on the energy metabolism of Clostridium kluyveri. Arch Microbiol 65(4):318–328
Schuchmann K, Müller V (2014) Autotrophy at the thermodynamic limit of life: a model for energy conservation in acetogenic bacteria. Nat Rev Microbiol 12(12):809–821. https://doi.org/10.1038/nrmicro3365
Schuchmann K, Müller V (2016) Energetics and application of heterotrophy in acetogenic bacteria. Appl Environ Microbiol 82(14):4056–4069. https://doi.org/10.1128/AEM.00882-16
Seinfeld JH, Pandis SN (2016) Atmospheric chemistry and physics: from air pollution to climate change. Wiley, Hoboken
Shen N, Dai K, Xia X-Y, Zeng RJ, Zhang F (2018) Conversion of syngas (CO and H2) to biochemicals by mixed culture fermentation in mesophilic and thermophilic hollow-fiber membrane biofilm reactors. J Clean Prod 202:536–542. https://doi.org/10.1016/j.jclepro.2018.08.162
Sikarwar VS, Zhao M, Clough P, Yao J, Zhong X, Memon MZ, Shah N, Anthony EJ, Fennell PS (2016) An overview of advances in biomass gasification. Energy Environ Sci 9(10):2939–2977. https://doi.org/10.1039/c6ee00935b
Sipma J, Meulepas RJ, Parshina SN, Stams AJ, Lettinga G, Lens PN (2004) Effect of carbon monoxide, hydrogen and sulfate on thermophilic (55°C) hydrogenogenic carbon monoxide conversion in two anaerobic bioreactor sludges. Appl Microbiol Biotechnol 64(3):421–428. https://doi.org/10.1007/s00253-003-1430-4
Spirito CM, Richter H, Rabaey K, Stams AJ, Angenent LT (2014) Chain elongation in anaerobic reactor microbiomes to recover resources from waste. Curr Opin Biotechnol 27:115–122. https://doi.org/10.1016/j.copbio.2014.01.003
Steinbusch KJ, Hamelers HV, Buisman CJ (2008) Alcohol production through volatile fatty acids reduction with hydrogen as electron donor by mixed cultures. Water Res 42(15):4059–4066. https://doi.org/10.1016/j.watres.2008.05.032
Steinbusch KJJ, Hamelers HVM, Plugge CM, Buisman CJN (2011) Biological formation of caproate and caprylate from acetate: fuel and chemical production from low grade biomass. Energy Environ Sci 4(1):216–224. https://doi.org/10.1039/c0ee00282h
Sträuber H, Lucas R, Kleinsteuber S (2016) Metabolic and microbial community dynamics during the anaerobic digestion of maize silage in a two-phase process. Appl Microbiol Biotechnol 100(1):479–491. https://doi.org/10.1007/s00253-015-6996-0
Sträuber H, Bühligen F, Kleinsteuber S, Dittrich-Zechendorf M (2018) Carboxylic acid production from ensiled crops in anaerobic solid-state fermentation - trace elements as pH controlling agents support microbial chain elongation with lactic acid. Eng Life Sci 18(7):447–458. https://doi.org/10.1002/elsc.201700186
Takors R, Kopf M, Mampel J, Bluemke W, Blombach B, Eikmanns B, Bengelsdorf FR, Weuster-Botz D, Dürre P (2018) Using gas mixtures of CO, CO2 and H2 as microbial substrates: the do's and don'ts of successful technology transfer from laboratory to production scale. Microb Biotechnol 11(4):606–625. https://doi.org/10.1111/1751-7915.13270
Urban C, Xu J, Sträuber H, dos Santos Dantas TR, Mühlenberg J, Härtig C, Angenent LT, Harnisch F (2017) Production of drop-in fuels from biomass at high selectivity by combined microbial and electrochemical conversion. Energy Environ Sci 10(10):2231–2244. https://doi.org/10.1039/c7ee01303e
Vassilev I, Hernández PA, Batlle-Vilanova P, Freguia S, Krömer JO, Jr K, Ledezma P, Virdis B (2018) Microbial electrosynthesis of isobutyric, butyric, caproic acids, and corresponding alcohols from carbon dioxide. ACS Sustain Chem Eng 6(7):8485–8493
Vassilev I, Kracke F, Freguia S, Keller J, Kromer JO, Ledezma P, Virdis B (2019) Microbial electrosynthesis system with dual biocathode arrangement for simultaneous acetogenesis, solventogenesis and carbon chain elongation. Chem Commun (Camb) 55(30):4351–4354. https://doi.org/10.1039/c9cc00208a
Vasudevan D, Richter H, Angenent LT (2014) Upgrading dilute ethanol from syngas fermentation to n-caproate with reactor microbiomes. Bioresour Technol 151:378–382. https://doi.org/10.1016/j.biortech.2013.09.105
Wade WG (2015) Eubacterium. In: Whitman WB, Rainey F, Kämpfer P, Trujillo M, Chun J, DeVos P, Hedlund B, Dedysh S (eds) Bergey's Manual of Systematics of Archaea and Bacteria. https://doi.org/10.1002/9781118960608.gbm00629
Wang H, Li X, Wang Y, Tao Y, Lu S, Zhu X, Li D (2018) Improvement of n-caproic acid production with Ruminococcaceae bacterium CPB6: selection of electron acceptors and carbon sources and optimization of the culture medium. Microb Cell Factories 17(1):99. https://doi.org/10.1186/s12934-018-0946-3
Weghoff MC, Bertsch J, Müller V (2015) A novel mode of lactate metabolism in strictly anaerobic bacteria. Environ Microbiol 17(3):670–677. https://doi.org/10.1111/1462-2920.12493
Weimer PJ, Moen GN (2013) Quantitative analysis of growth and volatile fatty acid production by the anaerobic ruminal bacterium Megasphaera elsdenii T81. Appl Microbiol Biotechnol 97(9):4075–4081. https://doi.org/10.1007/s00253-012-4645-4
Werner JJ, Knights D, Garcia ML, Scalfone NB, Smith S, Yarasheski K, Cummings TA, Beers AR, Knight R, Angenent LT (2011) Bacterial community structures are unique and resilient in full-scale bioenergy systems. Proc Natl Acad Sci U S A 108(10):4158–4163. https://doi.org/10.1073/pnas.1015676108
Wu Q, Guo W, Bao X, Meng X, Yin R, Du J, Zheng H, Feng X, Luo H, Ren N (2018) Upgrading liquor-making wastewater into medium chain fatty acid: Insights into co-electron donors, key microflora, and energy harvest. Water Res 145:650–659. https://doi.org/10.1016/j.watres.2018.08.046
Wu Q, Guo W, You S, Bao X, Luo H, Wang H, Ren N (2019) Concentrating lactate-carbon flow on medium chain carboxylic acids production by hydrogen supply. Bioresour Technol 291:121573. https://doi.org/10.1016/j.biortech.2019.121573
Yasin M, Jeong Y, Park S, Jeong J, Lee EY, Lovitt RW, Kim BH, Lee J, Chang IS (2015) Microbial synthesis gas utilization and ways to resolve kinetic and mass-transfer limitations. Bioresour Technol 177:361–374. https://doi.org/10.1016/j.biortech.2014.11.022
Zhang F, Ding J, Zhang Y, Chen M, Ding ZW, van Loosdrecht MC, Zeng RJ (2013) Fatty acids production from hydrogen and carbon dioxide by mixed culture in the membrane biofilm reactor. Water Res 47(16):6122–6129. https://doi.org/10.1016/j.watres.2013.07.033
Zhang J, Taylor S, Wang Y (2016) Effects of end products on fermentation profiles in Clostridium carboxidivorans P7 for syngas fermentation. Bioresour Technol 218:1055–1063. https://doi.org/10.1016/j.biortech.2016.07.071
Zhang W, Dai K, Xia XY, Wang HJ, Chen Y, Lu YZ, Zhang F, Zeng RJ (2018) Free acetic acid as the key factor for the inhibition of hydrogenotrophic methanogenesis in mesophilic mixed culture fermentation. Bioresour Technol 264:17–23. https://doi.org/10.1016/j.biortech.2018.05.049
Zhu X, Tao Y, Liang C, Li X, Wei N, Zhang W, Zhou Y, Yang Y, Bo T (2015) The synthesis of n-caproate from lactate: a new efficient process for medium-chain carboxylates production. Sci Rep 5:14360. https://doi.org/10.1038/srep14360
Zhu X, Zhou Y, Wang Y, Wu T, Li X, Li D, Tao Y (2017) Production of high-concentration n-caproic acid from lactate through fermentation using a newly isolated Ruminococcaceae bacterium CPB6. Biotechnol Biofuels 10:102. https://doi.org/10.1186/s13068-017-0788-y
Zinder SH, Anguish T, Cardwell SC (1984) Selective inhibition by 2-bromoethanesulfonate of methanogenesis from acetate in a thermophilic anaerobic digestor. Appl Environ Microbiol 47(6):1343–1345
Funding
The study was funded by the Helmholtz Association, Research Program Renewable Energies. Financial support was also received from the CAPES – Brazilian Federal Agency for Support and Evaluation of Graduate Education within the Ministry of Education of Brazil (No. 88887.163504/2018-00) and from the BMBF - German Federal Ministry of Education and Research (No. 01DQ17016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baleeiro, F.C.F., Kleinsteuber, S., Neumann, A. et al. Syngas-aided anaerobic fermentation for medium-chain carboxylate and alcohol production: the case for microbial communities. Appl Microbiol Biotechnol 103, 8689–8709 (2019). https://doi.org/10.1007/s00253-019-10086-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-019-10086-9