Abstract
Summary
Switching weekly ALN or RIS to monthly MIN in patients with RA, of whom two-thirds were treated with low-dose PSL, significantly decreased bone turnover markers and increased BMD at 12 months, suggesting that monthly MIN may be an effective alternative treatment option of oral bisphosphonate treatment.
Introduction
The aim of this prospective, observational study was to evaluate the effects of switching weekly alendronate (ALN 35 mg) or risedronate (RIS 17.5 mg) to monthly minodronate (MIN 50 mg) in patients with rheumatoid arthritis (RA).
Methods
Patient characteristics were as follows: n = 172; 155 postmenopausal women, age 65.5 (44–87) years; T-score of lumbar spine (LS), −1.4; total hip (TH), −1.8; femoral neck (FN), −2.1; dose and rate of oral prednisolone (2.3 mg/day), 69.1 %; prior duration of ALN or RIS, 46.6 months; were allocated, based on their preference, to either the (1) continue group (n = 88), (2) switch-from-ALN group (n = 44), or (3) switch-from-RIS group (n = 40).
Results
After 12 months, increase in BMD was significantly greater in group 3 compared to group 1: LS (4.1 vs 1.2 %; P < 0.001), TH (1.9 vs −0.7 %; P < 0.01), and FN (2.7 vs −0.5 %; P < 0.05); and in group 2 compared to group 1: LS (3.2 vs 1.2 %; P < 0.05) and TH (1.5 vs −0.7 %; P < 0.01). The decrease in bone turnover markers was significantly greater in group 3 compared to group 1: TRACP-5b (−37.3 vs 2.5 %; P < 0.001), PINP (−24.7 vs −6.2 %; P < 0.05), and ucOC (−39.2 vs 13.0 %; P < 0.05); and in group 2 compared to group 1: TRACP-5b (−12.5 vs 2.5 %; P < 0.05) at 12 months.
Conclusions
Switching weekly ALN or RIS to monthly MIN in patients with RA may be an effective alternative treatment option of oral bisphosphonate treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Increased risk of fractures in patients with rheumatoid arthritis (RA) compared to non-RA controls has been reported, with risk ratios (RR) varying from 2.0 to 3.0 at the hip and 2.4 to 6.2 at the spine [1–3]. Pro-inflammatory cytokines, such as tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1, IL-6, and IL-17, are strongly involved in the pathogenesis of RA and also concerned with osteoclastogenesis and consequent bone loss [4–7]. Indeed, high bone turnover and inflammation is associated with bone loss of the femoral neck (FN) in postmenopausal RA patients [8]. Moreover, glucocorticoids are often used to treat RA, which induce apoptosis of osteoblasts and osteocytes, and result in increased fracture risk [9, 10]. Minodronate (MIN) is an oral nitrogen-containing bisphosphonate (BP) developed in Japan which has a stronger inhibitory effect on farnesyl pyrophosphate synthase in osteoclasts compared with alendronate (ALN) or risedronate (RIS) [11]. It has been shown that switching daily or weekly BP (mainly ALN and RIS) to monthly MIN increased bone mineral density (BMD) of the lumbar spine (LS) and distal radius, and also decreased bone turnover markers in patients with osteoporosis [12]. There are still considerable number of patients who desire oral osteoporosis treatment, and we hypothesized that MIN can be a convenient candidate of alternative oral BP treatment in patients with RA treated by ALN and RIS, which may be more effective in decreasing bone turnover and increasing BMD. The aim of this prospective study was to clarify the effect of switching weekly ALN (35 mg) or RIS (17.5 mg) to monthly minodronate (50 mg) in patients with RA.
Materials and methods
Study design and subjects
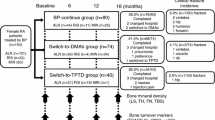
This 12-month observational study was conducted based on a two-center, prospective, open-label design. A total of 172 patients with RA who were treated with oral weekly ALN or RIS in proportion to the Japanese guidelines for prevention and treatment of osteoporosis 2011 [13] and the guidelines on the management and treatment of glucocorticoid-induced osteoporosis of the Japanese Society for Bone and Mineral Research 2004 [14], were enrolled in the study (Fig. 1). RA was diagnosed based on the 1987 revised American College of Rheumatology (ACR) criteria [15]. C-reactive protein (CRP), matrix metalloproteinase-3 (MMP-3), and the Disease Activity Score assessing 28 joints with CRP (DAS28-CRP) were evaluated as the parameters reflecting inflammation as well as the disease activity of RA [16, 17]. Registered patients were asked their preference for a change to monthly oral BP treatment and were allocated based on their preferences to either the “continue” group (n = 88), consisting of patients who wanted to continue their current therapies, or the “switch-from-ALN” group (n = 44) or “switch-from-RIS” group (n = 40), consisting of patients who were willing to switch over to MIN 50 mg from their current therapies. Other combined osteoporosis treatments, such as active vitamin D, vitamin K2, and calcium were continued during the study period. Patients’ treatment persistence and satisfaction levels with the therapies were assessed using a self-administered questionnaire at 12 months (Table 1). Patients were asked for their drug adherence every time visiting outpatient clinic (every 1–3 months), and patients who did not take their medications more than twice of their interval (more than 2 weeks for weekly ALN or RIS and more than 2 months for monthly MIN) were considered as dropout.
This observational study was conducted in accordance with the ethical standards of the Declaration of Helsinki and was approved by ethical review boards at the clinical center (approval number 11273-2; Osaka University, Graduate School of Medicine). Written informed consent was obtained from individual patients included in the study.
BMD assessment
A real BMD in the LS (L2–L4), total hip (TH), and femoral neck (FN) were assessed by dual-energy X-ray absorptiometry (Discovery A, Hologic, Inc., Waltham, MA, USA) at baseline and after 6 and 12 months of treatment. Regions of severe scoliosis, vertebral fracture, and operated sites were excluded from BMD measurements as previously described [18].
Biochemical markers of bone turnover
Bone turnover markers were measured in serum obtained from each patient at approximately the same time in the morning after overnight fasting. The bone formation marker, N-terminal type I procollagen propeptide (PINP); inter-assay coefficient of variation (CV), 3.2 %–5.2 %, (Intact UniQ assay, Orion Diagnostica, Espoo, Finland), and bone resorption marker, isoform 5b of tartrate-resistant acid phosphatase (TRACP-5b); inter-assay CV, 5.0 %–9.0 %, (Immunodiagnostic Systems Ltd., Boldon, UK) were measured by ELISA as previously described [19]. Levels of undercarboxylated osteocalcin (ucOC) were measured by a solid-phase enzyme immunoassay kit; inter-assay CV, 5.2 %–8.3 %, (Takara Bio, Shiga, Japan) with a sensitivity of 0.25 ng/mL. UcOC reflects not only vitamin K deficiency, but also total bone turnover, as it is released from both osteoblasts and absorbed bone extracellular matrix by osteoclast as previously described [20, 21]. Intact parathyroid hormone (PTH) was measured using a two-site immunoradiometric assay; inter-assay CV 8.4 %, (Nichols Institute Diagnostics, Valencia, USA).
Statistical analysis
The normal distributions of the data were examined by the Shapiro-Wilk test. Differences between each study group were tested using analysis of variance for normally distributed data and the nonparametric Kruskal-Wallis test was used for non-normally distributed data. Changes in BMD and ranked bone turnover marker data from baseline to specified time points within each study group were compared using the nonparametric Wilcoxon signed-rank test. Results are expressed as the mean ± standard error. A P value <0.05 indicated statistical significance. All tests were performed using IBM SPSS Statistics version 22 software (IBM, Armonk, NY, USA).
Results
Baseline characteristics are shown in Table 2. Of the 172 study patients, 84 (48.8 %) were willing to switch to MIN 50 mg. No significant differences were observed in the baseline age, combined dose, and prescription rate of active vitamin D or vitamin K2 or calcium or prednisolone (PSL), BMD, or disease activity of RA between the groups.
Duration of prior BP therapy at baseline was significantly longer in the switch-from-ALN group (57.2 months) compared to the continue group (43.6 months; P < 0.05) and the switch-from-RIS group (41.0 months; P < 0.05). Baseline serum TRACP-5b levels in the switch-from-ALN group were significantly lower compared to the switch-from-RIS group (244.5 vs 309.8 mU/dL; P < 0.05). Eventually, 95.5 % (84/88) of patients in the continue group (2 patients were lost to follow up and 2 patients desired to change the medication) and 94.0 % (79/84) of patients in the switch group (3 patients were lost to follow up and 2 patients desired to change the medication) completed the 12-month trial (Fig. 1).
Change in BMD
BMD was monitored every 6 months (Fig. 2). Both the switch groups showed a significant increase in LS and TH BMD from baseline to 6 and 12 months, while only the switch-from-RIS group showed a significant increase in FN BMD from baseline to 6 and 12 months. Moreover, the switch-from-RIS group showed a significantly greater increase compared to the continue group in the LS from 6 months (2.3 vs 0.6 %; P < 0.05) to 12 months (4.1 vs 1.2 %; P < 0.001), in the TH from 6 months (1.8 vs –0.5 %; P < 0.01) to 12 months (2.0 vs –0.7 %; P < 0.01), and in the FN from 6 months (2.0 vs –0.4 %; P < 0.05) to 12 months (2.7 vs –0.5 %; P < 0.05), respectively. On the other hand, the switch-from-ALN group showed a significantly greater increase compared to the continue group in LS BMD at 12 months (3.2 vs 1.2 %; P < 0.05) and in the TH from 6 months (1.2 vs –0.5 %; P < 0.01) to 12 months (1.5 vs –0.7 %; P < 0.01). The switch-from-RIS group showed a significantly greater increase compared to the switch-from-ALN group in the FN from 6 months (2.1 vs –0.3 %; P < 0.05) to 12 months (2.7 vs –0.6 %; P < 0.05).
Mean ± standard error (SE) change from baseline in bone mineral density (BMD) at the lumbar spine (a), total hip (b), and femoral neck (c). *P < 0.05, **P < 0.01, ***P < 0.001 change from baseline within each treatment group. # P < 0.05, ## P < 0.01, ### P < 0.001 continue group versus switch-from-RIS group. † P < 0.05, switch-from-ALN group versus switch-from-RIS group
Bone turnover markers
Percent changes in bone turnover markers from baseline are shown in Fig. 3. The switch-from-RIS group showed a significantly greater decrease compared to the continue group in TRACP-5b levels from 6 months (–35.8 vs 1.3 %; P < 0.001) to 12 months (–37.3 vs 2.5 %; P < 0.001), in PINP levels from 6 months (–22.2 vs –3.3 %; P < 0.05) to 12 months (–24.7 vs –6.2 %; P < 0.05), and in ucOC levels from 6 months (–22.2 vs 12.4 %; P < 0.05) to 12 months (–39.2 vs 13.0 %; P < 0.05). On the other hand, the switch-from-ALN group showed a significantly greater decrease compared to the continue group only in TRACP-5b levels from 6 months (–14.6 vs 1.3 %; P < 0.01) to 12 months (–12.5 vs 2.5 %; P < 0.05). The switch-from-RIS group showed a significantly greater decrease than the minimum significant change of serum TRACP-5b, PINP, and ucOC levels, while the switch-from-ALN group showed only in the serum TRACP-5b at 12 months. There were no greater changes than the minimum significant change of serum TRACP-5b, PINP, and ucOC levels in the continue group. The absolute value of bone turnover markers are shown in Fig. 4. The average value of TRACP-5b, PINP, and ucOC in all the groups were all within the reference value.
Mean ± standard error (SE) change from baseline in serum concentration of bone turnover markers TRAP-5b (a), PINP (b), and ucOC (c). TRAP-5b, isoform 5b of tartrate-resistant acid phosphatase; PINP, type I collagen N-terminal propeptide; ucOC, undercarboxylated osteocalcin; # P < 0.05, ## P < 0.01, ### P < 0.001 continue group versus each switch group. *P < 0.05, **P < 0.01 switch-from-ALN group versus switch-from-RIS group
Mean ± standard error (SE) absolute value of bone turnover markers TRAP-5b (a), PINP (b), and ucOC (c). TRAP-5b isoform 5b of tartrate-resistant acid phosphatase, PINP type I collagen N-terminal propeptide, ucOC undercarboxylated osteocalcin. # P < 0.05, ## P < 0.01, ### P < 0.001 continue group versus each switch group. *P < 0.05 switch-from-ALN group versus switch-from-RIS group
Rate of fragility fracture
During the 12-month period, the continue group patients experienced three vertebral and one non-vertebral clinical fragility fractures (4.5 %). The switch-from-ALN group experienced one vertebral and one non-vertebral clinical fragility fractures (4.5 %), and no clinical fragility fracture was observed in the switch-from-RIS group (0.0 %). No statistically significant difference in the total clinical fragility fracture rate was observed between the groups.
Patient preference after switching to MIN 50 mg
Patient preference after switching to monthly MIN 50 mg is shown in Fig. 5. The questionnaire revealed that 80.8 % of patients were satisfied with the switch to monthly therapy and 88.7 % preferred to continue the monthly treatment. The main reasons for desiring continuation of monthly dosing was both the decreased frequency (69.8 %) and less worry about forgetting doses (47.2 %), thus a perception of less overall burden.
Discussion
In this study, we have demonstrated for the first time that in patients with RA, of whom two-thirds were treated with low-dose PSL (<10 mg/day), switching from weekly ALN or RIS to monthly MIN was effective in increasing BMD and decreasing bone turnover markers at 12 months. In addition, no previous studies have demonstrated the difference of the effects of switching by the difference of prior BP therapies.
In nitrogen-containing BP treatment, mineral-binding affinities may influence their distribution within bone and the period till anti-fracture effects are shown, and inhibition of farnesyl diphosphate synthase (FPPS) may affect their antiresorptive effects by inducing apoptosis of osteoclasts [22].
It has been shown that ALN possesses a stronger binding affinity to hydroxyapatite compared to RIS, while RIS possesses a stronger FPPS inhibition compared to ALN [22]. Consequently, weekly ALN (70 mg) showed a greater increase in BMD and decrease in bone turnover markers compared to weekly RIS (35 mg) in patients with postmenopausal osteoporosis [23], while RIS showed lower rates of hip and non-vertebral fractures than ALN during the first year of therapy [24].
Previous reports have demonstrated that MIN showed stronger FPPS inhibition [11] and a weaker binding affinity to hydroxyapatite compared to ALN and RIS [25], which suggests that MIN inhibits bone resorption more strongly and is more quickly distributed within the bone compared to ALN and RIS. Indeed, MIN suppressed bone remodeling of cancellous and cortical bone more strongly than ALN in vitro [26] as well as in ovariectomized cynomolgus monkeys in vivo [27]. In the previous human study, switching ALN or RIS to monthly MIN for 6 months increased BMD +1.1 % in LS, and the reduction rate of serum TRACP-5b was approximately 35 % in the switching from RIS group at 6 months [12], which were consistent with our study.
Finally, glucocorticoids have been shown to induce apoptosis of osteocytes, and BPs inhibit osteocyte apoptosis in vitro [28] as well as in glucocorticoid-treated animals [29]. A systematic review and meta-analysis revealed that BPs can preserve bone mass and reduce the incidence of vertebral fractures in patients with rheumatic disease, mainly for those who are being treated with glucocorticoids [30], and both ALN and RIS strongly decreased the fracture risk associated with glucocorticoid-induced osteoporosis (GIO) [31, 32]. In this study, monthly MIN 50 mg resulted in a greater BMD increase and bone turnover decrease when patients were switched from ALN or RIS, which suggests its effectiveness not only in primary osteoporosis, but also in GIO.
There are several limitations to this study. Due to the small number of subjects, fracture risk comparisons should be assessed in a randomized, larger cohort. As most of the patients showed remission or low disease activity in this study, the effects of switching on high disease activity patients should be assessed in further study. Although most patients were postmenopausal, some male patients were included in this study. Concerning medication, the dose of ALN and RIS allowed in Japan is the half of Caucasians, and the duration of prior BP therapy was significantly longer in switch-to-ALN group compared to other groups. In addition, only a small number of patients were combined with calcium formulation, and total calcium intake could not be monitored.
In conclusion, switching weekly ALN or RIS to monthly MIN in patients with RA, of whom two-thirds were treated with low-dose PSL, significantly decreased bone turnover markers and increased BMD at 12 months, suggesting that monthly MIN may be an effective alternative treatment option of oral BP treatment.
References
Peel NF, Moore DJ, Barrington NA, Bax DE, Eastell R (1995) Risk of vertebral fracture and relationship to bone mineral density in steroid treated rheumatoid arthritis. Ann Rheum Dis 54:801–806
van Staa TP, Geusens P, Bijlsma JW, Leufkens HG, Cooper C (2006) Clinical assessment of the long-term risk of fracture in patients with rheumatoid arthritis. Arthritis Rheum 54:3104–3112
Wright NC, Lisse JR, Walitt BT, Eaton CB, Chen Z (2011) Arthritis increases the risk for fractures—results from the women’s health initiative. J Rheumatol 38:1680–1688
Braun T, Schett G (2012) Pathways for bone loss in inflammatory disease. Curr Osteoporos Rep 10:101–108
Ebina K, Oshima K, Matsuda M et al (2009) Adenovirus-mediated gene transfer of adiponectin reduces the severity of collagen-induced arthritis in mice. Biochem Biophys Res Commun 378:186–191
Kaneshiro S, Ebina K, Shi K, Higuchi C, Hirao M, Okamoto M, Koizumi K, Morimoto T, Yoshikawa H, Hashimoto J (2014) IL-6 negatively regulates osteoblast differentiation through the SHP2/MEK2 and SHP2/Akt2 pathways in vitro. J Bone Miner Metab 32:378–392
Noguchi T, Ebina K, Hirao M et al (2015) Progranulin plays crucial roles in preserving bone mass by inhibiting TNF-alpha-induced osteoclastogenesis and promoting osteoblastic differentiation in mice. Biochem Biophys Res Commun 465:638–643
Cortet B, Guyot MH, Solau E, Pigny P, Dumoulin F, Flipo RM, Marchandise X, Delcambre B (2000) Factors influencing bone loss in rheumatoid arthritis: a longitudinal study. Clin Exp Rheumatol 18:683–690
Kanis JA, Johansson H, Oden A et al (2004) A meta-analysis of prior corticosteroid use and fracture risk. J Bone Miner Res 19:893–899
Van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C (2000) Use of oral corticosteroids and risk of fractures. J Bone Miner Res 15:993–1000
Dunford JE, Thompson K, Coxon FP, Luckman SP, Hahn FM, Poulter CD, Ebetino FH, Rogers MJ (2001) Structure-activity relationships for inhibition of farnesyl diphosphate synthase in vitro and inhibition of bone resorption in vivo by nitrogen-containing bisphosphonates. J Pharmacol Exp Ther 296:235–242
Sakai A, Ikeda S, Okimoto N et al (2014) Clinical efficacy and treatment persistence of monthly minodronate for osteoporotic patients unsatisfied with, and shifted from, daily or weekly bisphosphonates: the BP-MUSASHI study. Osteoporos Int 25:2245–2253
Orimo H, Nakamura T, Hosoi T et al (2012) Japanese 2011 guidelines for prevention and treatment of osteoporosis—executive summary. Arch Osteoporos 7:3–20
Nawata H, Soen S, Takayanagi R et al (2005) Guidelines on the management and treatment of glucocorticoid-induced osteoporosis of the Japanese Society for Bone and Mineral Research (2004). J Bone Miner Metab 23:105–109
Arnett FC, Edworthy SM, Bloch DA et al (1988) The American rheumatism association 1987 revised criteria for the classification of rheumatoid arthritis. Arthritis Rheum 31:315–324
Ebina K, Shi K, Hirao M, Kaneshiro S, Morimoto T, Koizumi K, Yoshikawa H, Hashimoto J (2013) Vitamin K2 administration is associated with decreased disease activity in patients with rheumatoid arthritis. Mod Rheumatol 23:1001–1007
Matsui T, Kuga Y, Kaneko A, Nishino J, Eto Y, Chiba N, Yasuda M, Saisho K, Shimada K, Tohma S (2007) Disease activity score 28 (DAS28) using C-reactive protein underestimates disease activity and overestimates EULAR response criteria compared with DAS28 using erythrocyte sedimentation rate in a large observational cohort of rheumatoid arthritis patients in Japan. Ann Rheum Dis 66:1221–1226
Ebina K, Noguchi T, Hirao M, Kaneshiro S, Tsukamoto Y, Yoshikawa H (2015) Comparison of the effects of 12 months of monthly minodronate monotherapy and monthly minodronate combination therapy with vitamin K or eldecalcitol in patients with primary osteoporosis. J Bone Miner Metab
Ebina K, Hashimoto J, Shi K, Kashii M, Hirao M, Yoshikawa H (2014) Comparison of the effect of 18-month daily teriparatide administration on patients with rheumatoid arthritis and postmenopausal osteoporosis patients. Osteoporos Int 25:2755–2765
Booth SL, Centi A, Smith SR, Gundberg C (2013) The role of osteocalcin in human glucose metabolism: marker or mediator? Nat Rev Endocrinol 9:43–55
Ebina K, Hashimoto J, Shi K, Kashii M, Hirao M, Yoshikawa H (2014) Undercarboxylated osteocalcin may be an attractive marker of teriparatide treatment in RA patients: response to Mokuda. Osteoporos Int
Russell RG, Watts NB, Ebetino FH, Rogers MJ (2008) Mechanisms of action of bisphosphonates: similarities and differences and their potential influence on clinical efficacy. Osteoporos Int 19:733–759
Reid DM, Hosking D, Kendler D et al (2008) A comparison of the effect of alendronate and risedronate on bone mineral density in postmenopausal women with osteoporosis: 24-month results from FACTS-International. Int J Clin Pract 62:575–584
Silverman SL, Watts NB, Delmas PD, Lange JL, Lindsay R (2007) Effectiveness of bisphosphonates on nonvertebral and hip fractures in the first year of therapy: the risedronate and alendronate (REAL) cohort study. Osteoporos Int 18:25–34
Ebetino FH, Hogan AM, Sun S et al (2011) The relationship between the chemistry and biological activity of the bisphosphonates. Bone 49:20–33
Tsubaki M, Komai M, Itoh T et al (2014) Nitrogen-containing bisphosphonates inhibit RANKL- and M-CSF-induced osteoclast formation through the inhibition of ERK1/2 and Akt activation. J Biomed Sci 21:10
Yamagami Y, Mashiba T, Iwata K, Tanaka M, Nozaki K, Yamamoto T (2013) Effects of minodronic acid and alendronate on bone remodeling, microdamage accumulation, degree of mineralization and bone mechanical properties in ovariectomized cynomolgus monkeys. Bone 54:1–7
Plotkin LI, Manolagas SC, Bellido T (2006) Dissociation of the pro-apoptotic effects of bisphosphonates on osteoclasts from their anti-apoptotic effects on osteoblasts/osteocytes with novel analogs. Bone 39:443–452
Follet H, Li J, Phipps RJ, Hui S, Condon K, Burr DB (2007) Risedronate and alendronate suppress osteocyte apoptosis following cyclic fatigue loading. Bone 40:1172–1177
Feng Z, Zeng S, Wang Y, Zheng Z, Chen Z (2013) Bisphosphonates for the prevention and treatment of osteoporosis in patients with rheumatic diseases: a systematic review and meta-analysis. PLoS One 8:e80890
Adachi JD, Saag KG, Delmas PD et al (2001) Two-year effects of alendronate on bone mineral density and vertebral fracture in patients receiving glucocorticoids: a randomized, double-blind, placebo-controlled extension trial. Arthritis Rheum 44:202–211
Wallach S, Cohen S, Reid DM et al (2000) Effects of risedronate treatment on bone density and vertebral fracture in patients on corticosteroid therapy. Calcif Tissue Int 67:277–285
Acknowledgments
The authors thank Dr. Kenrin Shi for his excellent cooperation in conducting the study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This observational study was conducted in accordance with the ethical standards of the Declaration of Helsinki and was approved by ethical review boards at the clinical center (approval number 11273-2; Osaka University, Graduate School of Medicine). Written informed consent was obtained from individual patients included in the study.
Conflicts of interest
This research was funded by Astellas Pharma, Inc. The funder had no role in the study design, data collection, data analysis, decision to publish, or preparation of the manuscript. Kosuke Ebina, Takaaki Noguchi, Makoto Hirao, Jun Hashimoto, Shoichi Kaneshiro, Masao Yukioka, and Hideki Yoshikawa declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Ebina, K., Noguchi, T., Hirao, M. et al. Effects of switching weekly alendronate or risedronate to monthly minodronate in patients with rheumatoid arthritis: a 12-month prospective study. Osteoporos Int 27, 351–359 (2016). https://doi.org/10.1007/s00198-015-3369-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-015-3369-6