Abstract
The incidence of fragility fractures begins to increase in middle age. We investigated prospectively risk factors for low-energy fractures in men and women, and specifically for forearm, proximal humerus, vertebral, and ankle fractures. The population-based Malmö Preventive Project consists of 22,444 men and 10,902 women, mean age 44 and 50 years, respectively, at inclusion. Baseline assessment included multiple examinations and lifestyle information. Mean follow-up was 19 and 15 years for men and women, respectively, regarding incident fractures. Fractures were ascertained from radiographic files. At least one low-energy fracture occurred in 1,262 men and 1,257 women. In men, the risk factors most strongly associated with low-energy fractures were diabetes [relative risk (RR) 2.38, confidence interval (CI) 95% 1.65–3.42] and hospitalization for mental health problems (RR 1.92, CI 95% 1.47–2.51). Factors associated with mental health and lifestyle significantly increased the fracture risk in most of the specific fracture groups: hospitalizations for mental health problems (RR 2.28–3.38), poor appetite (RR 3.05–3.43), sleep disturbances (RR 1.72–2.95), poor self-rated health (RR 1.80–1.83), and smoking (RR 1.70–2.72). In women, the risk factors most strongly associated with low-energy fractures were diabetes (RR 1.87, CI 95% 1.26–2.79) and previous fracture (RR 2.00, CI 95% 1.56–2.58). High body mass index (BMI) significantly increased the risk of proximal humerus and ankle fractures (RR 1.21–1.33) while, by contrast, lowering the risk of forearm fractures (RR 0.88, CI 95% 0.81–0.96). Risk factors for fracture in middle-aged men and women are similar but with gender differences for forearm, vertebral, proximal humerus, and hip fracture whereas risk factors for ankle fractures differ to a certain extent. The risk-factor pattern indicates a generally impaired health status, with mental health problems as a major contributor to fracture risk, particularly in men.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Over recent decades, the incidence of fractures has increased substantially, especially among the elderly [1, 2]. Different fracture types dominate at different ages, and the fracture pattern varies between women and men [1]. In middle age, forearm and ankle fractures are the most frequent whereas in the elderly, hip fractures become the predominating problem. Fracture incidence is usually greater among men than women before the age of 50 years, but at higher ages, women suffer more fractures [3]. In general, 75% of all fractures among men occur before the age of 45 whereas in women, 75% of all fractures occur after the age of 45 [4].
Although the consequences of a limb fracture are usually not as devastating as those of a hip fracture, the suffering, inconvenience, and temporary or sometimes permanent disability are strong enough reasons to motivate a closer epidemiological investigation of these fractures. Fracture-related costs to society are substantial; the cost for direct medical care of osteoporotic fractures other than hip fractures in the USA was US $5.1 billion in 1995 [5]. Prevention of these fractures would free resources for other areas of health care and avoid unnecessary suffering.
Having a history of a previous fracture is a known risk factor predisposing for future fractures in the elderly [6–8]. Identification of factors associated with fracture risk already in middle age, when a fracture is still a rare event, has not been extensively investigated. Increased knowledge in this area would ultimately enable us to initiate preventive measures when most appropriate.
The primary objective of this study was to investigate risk-factor pattern for fractures commonly occurring in women and men in middle age: 48–68 years. A secondary objective was to identify differences in risk between men and women.
Material and methods
The Malmö Preventive Project is a prospective, population-based, cardiovascular screening study consisting of 33,346 probands, 22,444 men and 10,902 women, representing 72% of the invited population [9]. Mean age at baseline investigation was 44 (range 27–61) years for men and 48 (range 28–58) years for women. The inclusion period for men was 1974–1984 (10 years) and for women 1977–1992 (15 years). The primary objective of the Malmö Preventive Project was to describe cardiovascular risk factors and related conditions in a middle-aged population and to improve the scientific basis for cardiovascular disease prevention. Over and above this, the study provides data for evaluation of other common public health conditions, such as fracture and diabetes. The probands were followed prospectively till the end of 1999, with a mean follow-up of 19 (range 7–25) years for men and 15 (7–22) years for women for incident fractures and mortality.
Physical examination
Baseline physical investigation included height and body weight measurement as well as triceps skinfold thickness. Blood pressure and pulse rate were measured twice after a 10-min rest, and the mean was recorded. Simple spirometry (Spirotron, Drägerwerke, Germany) was performed, and pulmonary function parameters were calculated. In a subgroup of women, bone mineral density (BMD) of the distal radius was measured and has been reported previously [10].
Questionnaire
At baseline, participants completed a questionnaire regarding health-related and lifestyle issues, previously described in detail [11]. During the extended inclusion period, certain questions were added while others were withdrawn. The total number of questions used in the questionnaire were 417, 78 of which were core questions asked throughout the inclusion period. Mean response rate for women was about 98% and for men 72% for core questions. In the early period of this study (the beginning of the 1970s), the computer technique was still young, and due to computer problems, some questionnaire results were lost for 6,368 men. The response rate was 99% for the remaining results. Questions added during the inclusion period had a response rate of almost 100% for those who had the opportunity to respond. The amount of data is extensive, thus, only variables of biological interest and with possible impact on fracture risk were selected for this report.
Laboratory investigation
Morning blood samples were collected after an overnight fast. The following analyses were performed and used in this substudy: hemoglobin, erythrocyte sedimentation rate (ESR), serum creatinine, fasting blood glucose, serum γ-glutamyl transferase, serum triglycerides, serum total cholesterol, serum uric acid, and serum phosphate. Due to skewed distribution of serum γ-glutamyl transferase, data were logarithmically transformed when used in the analyses.
Fracture identification
Fracture data were obtained by linking the probands included in the Malmö Preventive Project with the register at the Department of Diagnostic Radiology at Malmö University Hospital. In the city of Malmö, all emergency radiographic examinations are performed at the Department of Diagnostic Radiology at Malmö University Hospital, fractures are recorded, and films are stored and saved permanently. The unique ten-digit personal identification number, based on birth date and issued to every Swedish citizen, makes identification of cases easy and precise. Fractures identified were confirmed through manual search of medical and radiological files. Previous studies have shown that at least 97% of all fractures experienced by citizens of Malmö can be identified this way [12]. Fractures registered were classified under the following categories: forearm, vertebral, proximal humerus, and ankle (Table 1). Data on hip fractures, previously reported [11], are included for comparison. Regarding vertebral fractures, only those coming to clinical attention and described in radiographic reports or accidentally found on chest or abdominal X-rays for other causes and described in the radiographic report were included. The degree of deformity was not quantified.
Fractures were classified as high- or low-energy trauma depending on the cause of fracture. Fractures caused by falling from standing height or less were classified as low-energy fractures, and those caused by high-energy trauma as high-energy fractures. Classification was based on information given in the radiographic reports. Of all fractures, 4,098 (97.7%) were described with adequate information about the degree of trauma in the radiology reports. The 98 fractures with insufficient information concerning trauma were classified as low-energy fractures based on the experience that it is highly unlikely that information about high-energy accidents is not reported. Fractures caused by high-energy trauma were excluded from the analyses, as were pathological fractures caused by cancer or other bone diseases.
Statistics
For this substudy, relevant data were extracted from the Malmö Preventive Project main database. The population was divided into those with and those without fracture. Individuals suffering exclusively fractures caused by high-energy trauma were excluded from the fracture population and included in the nonfracture population. Individuals with at least one low-energy-incident fracture were classified into the low-energy fracture group even if a previous or subsequent high-energy fracture was recorded during the study period. The first fracture of each fracture type was the one included in the calculation even if the individual had suffered more than one fracture of the same type. If an individual suffered more than one type of fracture, the individual was included in the calculations for each fracture type, albeit under different dates for the fracture incident. Fracture incidence was calculated as the number of fractures per 1,000 person years. Baseline descriptive data of the whole cohort and fracture subgroups were presented as percentages, means, and standard deviations (SD). Due to the skewed distribution of serum γ-glutamyl transferase, it was logarithmically transformed when used in the analyses.
Variables of biological interest were chosen from the database. Z-scores were calculated for continuous variables to permit comparison of different variables. Variables were analyzed, one by one, in an age-adjusted Cox proportional hazard model. To reduce the risk of mass significance through multiple testing, we chose a p-value of <0.01 as the level of significance. A final multiple regression model was constructed consisting of 11 (women) and ten (men) variables. Variables were selected from the age-adjusted Cox proportional hazard model based on the strength of their association with the different fracture types. Among variables with possible interdependence, we chose the one with the strongest association and highest response rate. Two questions, one regarding previous fracture history and another regarding hormone replacement therapy, were not included throughout the study, thus generating an incomplete data set. Despite their significant impact on fracture risk, we chose to exclude these questions from the final Cox analysis. The statistics program used was SPSS 12.0 for Windows (Statistics Package for Social Sciences, Chicago, IL, USA).
Results
Descriptives
Women
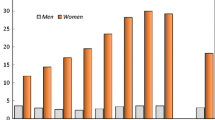
During the 11-year follow-up, 1,878 incident fractures were registered affecting 1,292 women, i.e., 12% of the female study population. Of these, 1,257 (97%) were classified as suffering low-energy fractures. The number of women with only one low-energy fracture was 894, representing 71% of women with low-energy fractures. Of the total number of 1,805 low-energy fractures recorded, 662 were forearm, 155 proximal humerus, 160 vertebral, 223 ankle, and 141 hip fractures (Table 1). Fracture incidence rates for each type are reported in Fig. 1. The age effect is most pronounced in women.
Baseline characteristics are reported for the entire cohort and for major fracture types (Table 2). Women suffering fractures were slightly older at study start than the nonfracture population, with the exception of women with ankle fractures. Mean body weight was 3.5 kg greater at inclusion in women with ankle and proximal humerus fractures compared with the nonfracture population and 2.5 kg less in women with a hip fracture. Prevalence of diabetes seemed higher in women with ankle, vertebral, and hip fractures. Women with vertebral and hip fractures also appeared to suffer from more comorbidities than the nonfracture population.
Men
During the 16-year follow-up, 2,422 incident fractures were registered affecting 1,505 men, i.e., 6.7% of the male study population. Of these, 1,262 (84%) were classified as suffering low-energy fractures. The number of men with only one low-energy fracture was 822, representing 65% of the men with low-energy fractures. Of the total number of 1,975 low-energy fractures recorded, 330 were forearm, 123 proximal humerus, 168 vertebral, 259 ankle, and 174 hip fractures (Table 1). Fracture incidence rates were half those in women, and the age effect was seen only in the highest age group (Fig. 1). Men with fractures were slightly older at the start of the study than the nonfracture population, which was especially apparent in men with a hip fracture (Table 2). Mean body weight and body mass index (BMI) were slightly lower in all separate fracture groups except for ankle fractures, where it was slightly higher. Smoking was more common among men suffering from vertebral, proximal humerus, and hip fractures. Risk factors for low-energy fractures overall and for specified types of fracture, for women and for men, are reported separately. Factors evaluated are presented as means and SD in Tables 3 and 4. In the following, we highlight results for women and men in the uni- and multivariate models presented in Table 5, 6, 7 and 8.
Risk factors in women
Low-energy fractures
The risk factor with the largest impact on low-energy fractures in general was a history of previous fracture, doubling the fracture risk (RR 2.00, CI 95% 1.56–2.58; p=0.001). Diabetes was associated with a similar risk increase (RR 1.95, 1.33–2.86; p=0.001) while high serum γ-glutamyl transferase and poor self-rated health gave smaller risk increases. A relative risk decrease was seen for those using hormone replacement therapy (HRT). In the final Cox model, diabetes remained a significant risk factor (Tables 5 and 7).
Forearm fractures
The risk factor with the largest impact on forearm fractures was a history of previous fracture, doubling the risk (RR 2.00, 1.40–2.85; p=0.001), whereas diabetes was not associated with an increased risk. High BMI was associated with decreased risk.
Vertebral fractures
Diabetes was the risk factor with the largest impact on vertebral fractures, increasing the risk more than three times (RR 3.56, 1.75–7.23; p=0.001). A history of previous fracture (RR 3.13, 1.67–5.85; p=0.001) also increased risk, as did high serum γ-glutamyl transferase, smoking, and decreased lung capacity [forced vital capacity (FVC)], but to a lesser extent. ESR as an indicator of inflammation was also associated with a small risk increase (RR 1.16, 1.04–1 29; p=0.009).
Proximal humerus fractures
In contrast to most other fracture groups, the risk of proximal humerus fracture was not affected by previous fracture history, diabetes, smoking, or high serum γ-glutamyl transferase. The factor most strongly associated with proximal humerus fracture was being in the higher age segment of the study population, with RR 2.27 (1.76–2.93; p=0.001). High body weight, high BMI, and poor self-rated health also increased the risk.
Ankle fractures
A history of previous fracture doubled the risk of ankle fracture (RR 2.11, 1.17–3.80; p=0.001) whereas the variable most strongly associated with ankle fractures was diabetes (RR 3.36, 1.58–7.15; p=0.002). High serum γ-glutamyl transferase as well as high body weight and BMI increased the risk.
Risk factors in men
Low energy fractures
The factor associated with the strongest risk effect on low-energy fractures in general was diabetes, which more than doubled the risk (RR 2.50, 1.75–3.57; p=0.001). A history of hospitalization for mental disorders (RR 1.92, 1.47–2.51; p=0.001) and having a poor appetite (RR 1.72, 127–2.32; p=0.001) also increased the risk. Smoking, and high serum γ-glutamyl transferase, an indirect measure of an alcohol effect, were associated with moderately increased risks. High BMI was associated with a decreased risk and is a protective attribute. In the final Cox model, the associations remained chiefly the same (Tables 6 and 8).
Forearm fractures
The factor associated with the strongest risk effect on forearm fractures was hospitalization for mental disorders (RR 2.28, 1.37–3.78; p=0.001). The only other factor significantly associated with forearm fractures was high BMI (RR 0.85, 0.75–0.96; p=0.007), which decreased the risk.
Vertebral fractures
A history of hospitalization for mental disorders increased fracture risk significantly (RR 2.34, 1.36–4.03; p=0.002). Other strong risk factors were having a poor appetite (RR 3.05, 1.80–5.17; p=0.001) and being on sick leave at the time of the baseline investigation (RR 2.98, 1.83–4.85; p=0.001). Smoking and a high serum γ-glutamyl transferase were also associated with increased risks.
Proximal humerus fractures
For risk of proximal humerus fractures, several factors related to mental health were significant contributors, each increasing the risk three-fold: prior hospitalization for mental disorders, poor appetite, and premature awakening. Smoking and high serum γ-glutamyl transferase were also associated risk factors. A high BMI decreased the risk. In the final Cox model, a high level of serum triglycerides was associated with a decreased risk.
Ankle fractures
Significant risk factors for ankle fractures were to some extent similar to other peripheral fractures but with a lower impact; prior hospitalization for mental disorders (RR 2.74, 1.62–4.66; p=0.002), premature awakening, and high serum γ-glutamyl transferase. In the final Cox model, only the association of high serum γ-glutamyl transferase remained.
Discussion
The etiology of fractures is multifaceted and complex, making fracture studies and reporting difficult and never complete. Our aim was to determine and examine a range of factors that affect fracture risk in middle age —low energy fractures in general, and, in more detail, fractures of the forearm, vertebrae, proximal humerus, and ankle. Low-energy fractures in general were strongly associated with diabetes and previous fracture history in women and with diabetes and mental health problems in men. For the specified fracture groups, the risk-factor pattern was to some extent different, but certain factors affected almost all subgroups: in women, being in the higher age segment of the study population, a history of previous fracture, and diabetes; and in men, being in the higher age segment of the study population, low BMI, high serum γ-glutamyl transferase, smoking, sleep disturbances, and hospitalization for mental disorders, were all factors identified as significant contributors to risk in most fracture subgroups. Our findings suggest that individuals in middle age with impaired physical and mental health are at high risk of experiencing peripheral and vertebral fractures.
Fracture incidences were calculated for the specified fracture groups. Results are similar to those of a study of the Geelong population in Victoria, Australia, except for forearm fractures [13]. Discrepancies may be attributable to the low number of fractures in some categories. Our incidence curves are similar to the results of a register study by Kanis and coworkers performed on the total population of Malmö through fracture registers although our incidence rates are lower [14]. However, our incidence rates are similar to earlier clinical studies in Malmö [1] but, as previously recognized, the rates for some fractures, forearm fractures in particular, are higher than in other large epidemiological studies [15]. The fact cannot be excluded that this study, as well as other population-based cohort studies, attract a somewhat more health-conscious segment of the population and who thus sustain fewer fractures in contrast to a register study that includes all citizens.
In both men and women, high body weight and BMI lowered the risk of forearm and hip fractures and of proximal humerus fractures in men. In contrast, in women, high body weight and BMI increased the risk of proximal humerus and ankle fractures. This further elicits the complexity of fracture etiology when both being overweight and being thin affects fracture risk, albeit different types of fracture. One could speculate that there is an association between poor nutrition and poor bone quality, as well as between high body weight and clumsiness with an increased tendency to fall and a higher impact of a fall even from standing height. These findings suggest two different types of individuals, both prone to fracture but with different risk factors predominating. A case-control study of 448 men and women over 45 years of age found that high BMI as well as frequent falling were risk factors for proximal humerus fracture [16]. This is consistent with our findings in women. Proximal humerus fractures, at least in elderly subjects, have been associated with low BMD [17] and considered a type fracture of osteoporosis. A study by Olsson and coworkers of both middle-aged and elderly subjects showed associations between humerus fractures and previous fracture history, as well as increased risk of future fracture [18]. This further supports our hypothesis about subgroups within the fracture population.
High body weight and BMI increased the risk of ankle fractures in women but decreased the risk of forearm and hip fractures. In the age-adjusted Cox model, we found other risk factors common to these fracture types, but in the final Cox model, only BMI remained a significant risk factor for ankle fractures and a factor decreasing risk of hip fractures. The age effect was no longer significant for ankle fractures in contrast to the other fracture types. This is consistent with previous reports [10, 19–22] suggesting that risk factors for ankle fractures are related with lifestyle, including a physically active life, whereas the risk associated with forearm fractures is more dependent on bone density. Our results indicate that ankle fractures differ in risk-factor profile compared with fractures commonly associated with poor bone quality, and it is thus questionable to include these in an analysis of osteoporotic fractures, a point further supported by a report by Kanis and coworkers [4].
Diabetes in both men and women was strongly associated with an increased relative risk of low-energy and hip fractures, as well as with vertebral and ankle fractures in women, a result consistent with previous studies [23–27]. In the multivariate analysis, the association between ankle fractures and diabetes in women disappeared. This is probably due to the small number of cases of diabetes found in each specific fracture group and not to a lack of impact on ankle fractures. Where diabetes is of long duration, multiple organ systems of the body are affected, leading to angiopathy, neuropathy, and nephropathy, with effects on bone mass and the tendency to fall. In a previous study [11], we have shown that diabetes has a large impact on hip fracture risk by middle age, suggesting more fragile bone structure in individuals with diabetes.
In recent studies, blood lipids have been associated with BMD, reporting a positive correlation between serum triglycerides and BMD and a negative correlation between serum cholesterol and BMD [28, 29]. In this study, we found that in both men and women of middle age, high levels of serum triglycerides are associated with a decreased risk of some peripheral fracture types. A high level of blood lipids may possibly represent a good status concerning nutrition and good health, protecting the individual from fracture. The true correlation between blood lipids and fracture risk remains to be explored.
Elevated levels of serum γ-glutamyl transferase may be an indicator of alcohol overconsumption [30–32]. Also in the population studied alcohol overconsumption has been shown to be the main cause of elevated levels of γ-glutamyl transferase [33]. However, indicators of the metabolic syndrome, including liver-derived serum lipids, may influence the levels of γ-glutamyl transferase, and the likelihood of interaction is high. Nevertheless, alcohol was still a major contributor [34]. In this study, elevated serum γ-glutamyl transferase was associated with an increased risk of low-energy fractures in general in both men and women for most of the specified fracture subtypes. Studies have shown that alcohol overconsumption increases the risk of fracture through metabolic effects as well as alcohol-related falls and a more hazardous life style in general [35–37]. Fracture pattern of men in this study, with a high proportion of high-energy fractures as well as hand and skull fractures, may indicate an unsafe life style, possibly in connection with alcohol abuse. In women, elevated serum γ-glutamyl transferase levels were associated with an increased risk of vertebral and ankle fractures. One could speculate that the effect on ankle fractures may be a result of acute alcohol intoxication since ankle fractures seem to be more associated with lifestyle factors than poor bone quality, and the effect on vertebral fractures seem more related to long-term effects of alcohol consumption. Unfortunately the study did not include specific questions on alcohol consumption; thus, we rely on this indirect but nevertheless objective measure.
In many previous reports, smoking has been found to be a risk factor for fractures in general and hip fractures in particular [38–40], with a negative effect on BMD [41, 42]. In this study, smoking was associated with increased risk of vertebral fractures in women . In men, it was associated with low-energy fractures in general and vertebral, proximal humerus, and hip fractures specifically.
Several variables regarding psychological well-being are recorded, allowing us to evaluate the impact on fracture risk. In men, one variable was a significant risk factor for all types of fracture, namely, previous hospitalization for mental health problems. When we compared the individuals reporting mental health problems with the nonfracture population, these individuals appeared to suffer from more comorbidities than the nonfracture population. A study from Tromsö, Norway, reported psychiatric disease in men as a risk factor for all fracture types and especially for forearm and proximal humerus fractures, in agreement with our findings [43]. Individuals with mental health problems may lead a more irregular life, with possible alcohol and drug abuse affecting their health, creating a frailer body constitution, and also with an increased risk of falling. Similar reasoning may be applicable regarding the increased risk of vertebral fractures in those currently on sick leave, assuming that sick leave is a sign of comorbidity.
This study has some limitations. Since the original study was not designed to investigate fractures and fracture risk and because of computer error, certain data sets are incomplete, and information is accessible only for the major parts of the study population. Nevertheless, the overall response rate is excellent. BMD measurements were performed only on a small part of the population [10], thus, the level of prevalent osteoporosis cannot be determined.
A strength of this study is that, to our knowledge, this is one of the first prospective studies evaluating a broader range of risk factors of common fracture types in middle age in both men and women. Furthermore, we have been able to evaluate factors that are not commonly studied, which should be regarded as a strength. The study is large and with a long follow-up, which makes the results more reliable.
This study has identified and described many risk factors for low-energy fractures in middle age and their impact on different fracture types, which confirms the multitude and complexity of factors affecting fracture risk. The study indicates similarities in risk-factor patterns between forearm, vertebral, proximal humerus, and hip fractures whereas ankle fractures, to a certain extent, have a different risk profile. A middle-aged person of generally impaired physical and mental health status with subsequent impaired bone strength as a possible background factor is, according to this study, at high risk of a peripheral fracture. Identification of risk factors in younger age groups gives us a unique opportunity to implement preventive strategies at a time when intervention may still prevent irreversible bone loss.
References
Obrant KJ et al (1989) Increasing age-adjusted risk of fragility fractures: a sign of increasing osteoporosis in successive generations? Calcif Tissue Int 44(3):157–167
Kannus P et al (2002) Why is the age-standardized incidence of low-trauma fractures rising in many elderly populations? J Bone Miner Res 17(8):1363–1367
van Staa TP et al (2001) Epidemiology of fractures in England and Wales. Bone 29(6):517–522
Kanis JA, Pitt FA (1992) Epidemiology of osteoporosis. Bone 13(Suppl 1):S7–S15
Melton LJ 3rd et al (2003) Cost-equivalence of different osteoporotic fractures. Osteoporos Int 14(5):383–388
Ettinger B et al (2003) Limb fractures in elderly men as indicators of subsequent fracture risk. Arch Intern Med 163(22):2741–2747
Klotzbuecher CM et al (2000) Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res 15(4):721–739
Johnell O et al (2004) Fracture risk following an osteoporotic fracture. Osteoporos Int 15(3):175–179
Berglund G et al (1996) Cardiovascular risk groups and mortality in an urban Swedish male population: the Malmo Preventive Project. J Intern Med 239(6):489–497
Holmberg A et al (2004) Forearm bone mineral density in 1294 middle-aged women: a strong predictor of fragility fractures. J Clin Densitom 7(4):419–423
Holmberg AH et al (2005) Risk factors for hip fractures in a middle-aged population: a study of 33,000 men and women. Osteoporos Int 16(12):2185–2194
Jonsson B et al (1993) Life-style and different fracture prevalence: a cross-sectional comparative population-based study. Calcif Tissue Int 52(6):425–433
Sanders KM et al (1999) Age- and gender-specific rate of fractures in Australia: a population-based study. Osteoporos Int 10(3):240–247
Kanis JA et al (2000) Long-term risk of osteoporotic fracture in Malmo. Osteoporos Int 11(8):669–674
Melton LJ 3rd et al (1998) Long-term trends in the incidence of distal forearm fractures. Osteoporos Int 8(4):341–348
Chu SP et al (2004) Risk factors for proximal humerus fracture. Am J Epidemiol 160(4):360–367
Nguyen TV et al (2001) Risk factors for proximal humerus, forearm, and wrist fractures in elderly men and women: the Dubbo Osteoporosis Epidemiology Study. Am J Epidemiol 153(6):587–595
Olsson C, Nordqvist A, Petersson CJ (2004) Increased fragility in patients with fracture of the proximal humerus: a case control study. Bone 34(6):1072–1077
Guggenbuhl P, Meadeb J, Chales G (2005) Osteoporotic fractures of the proximal humerus, pelvis, and ankle: epidemiology and diagnosis. Jt Bone Spine 72(5):372–375
Honkanen R et al (1998) Relationships between risk factors and fractures differ by type of fracture: a population-based study of 12,192 perimenopausal women. Osteoporos Int 8(1):25–31
Valtola A et al (2002) Lifestyle and other factors predict ankle fractures in perimenopausal women: a population-based prospective cohort study. Bone 30(1):238–242
Seeley DG et al (1996) Predictors of ankle and foot fractures in older women. The Study of Osteoporotic Fractures Research Group. J Bone Miner Res 11(9):1347–1355
Ahmed LA et al (2005) Diabetes mellitus and the risk of non-vertebral fractures: the Tromso study. Osteoporos Int :1–6
Forsen L et al (1999) Diabetes mellitus and the incidence of hip fracture: results from the Nord-Trondelag Health Survey. Diabetologia 42(8):920–925
Ottenbacher KJ et al (2002) Diabetes mellitus as a risk factor for hip fracture in Mexican American older adults. J Gerontol A Biol Sci Med Sci 57(10):M648–M653
Schwartz AV et al (2001) Older women with diabetes have an increased risk of fracture: a prospective study. J Clin Endocrinol Metab 86(1):32–38
Vestergaard P, Rejnmark L, Mosekilde L (2005) Relative fracture risk in patients with diabetes mellitus, and the impact of insulin and oral antidiabetic medication on relative fracture risk. Diabetologia 48(7):1292–1299
Adami S et al (2004) Relationship between lipids and bone mass in 2 cohorts of healthy women and men. Calcif Tissue Int 74(2):136–142
Lidfeldt J et al (2002) The influence of hormonal status and features of the metabolic syndrome on bone density: a population-based study of Swedish women aged 50 to 59 years. The women’s health in the Lund area study. Metabolism 51(2):267–270
Conigrave KM et al (2003) Traditional markers of excessive alcohol use. Addiction 98(Suppl 2):31–43
Conigrave KM, Saunders JB, Whitfield JB (1995) Diagnostic tests for alcohol consumption. Alcohol Alcohol 30(1):13–26
Banciu T et al (1983) Serum gamma-glutamyltranspeptidase assay in the detection of alcohol consumers and in the early and stadial diagnosis of alcoholic liver disease. Med Interne 21(1):23–29
Trell E, Kristenson H, Fex G (1984) Alcohol-related problems in middle-aged men with elevated serum gamma-glutamyltransferase: a preventive medical investigation. J Stud Alcohol 45(4):302–309
Yokoyama H et al (2003) Association between gamma-glutamyl transpeptidase activity and status of disorders constituting insulin resistance syndrome. Alcohol Clin Exp Res 27(8 Suppl):22S–25S
Kanis J et al (1999) Risk factors for hip fracture in men from southern Europe: the MEDOS study. Mediterranean Osteoporosis Study. Osteoporos Int 9(1):45–54
Johnell O et al (1995) Risk factors for hip fracture in European women: the MEDOS Study. Mediterranean Osteoporosis Study. J Bone Miner Res 10(11):1802–1815
Clark MK et al (2003) Bone mineral density and fractures among alcohol-dependent women in treatment and in recovery. Osteoporos Int 14(5):396–403
Kanis JA, Johnell O (2005) Requirements for DXA for the management of osteoporosis in Europe. Osteoporos Int 16(3):229–238
Law MR, Hackshaw AK (1997) A meta-analysis of cigarette smoking, bone mineral density and risk of hip fracture: recognition of a major effect. BMJ 315(7112):841–846
Huopio J et al (2000) Risk factors for perimenopausal fractures: a prospective study. Osteoporos Int 11(3):219–227
Kiel DP et al (1996) The effect of smoking at different life stages on bone mineral density in elderly men and women. Osteoporos Int 6(3):240–248
Gerdhem P, Obrant KJ (2002) Effects of cigarette-smoking on bone mass as assessed by dual-energy X-ray absorptiometry and ultrasound. Osteoporos Int 13(12):932–936
Ahmed LA et al (2006) Self-reported diseases and the risk of non-vertebral fractures: the Tromso study. Osteoporos Int 17(1):46–53
Acknowledgements
The Swedish Research Council Project K2003-73X-11610-08A, The Kock Foundation, The Herman Järnhardt Foundation, Malmö University Hospital Funds, and regional research grants supported this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
An erratum to this article can be found at http://dx.doi.org/10.1007/s00198-006-0164-4
Rights and permissions
About this article
Cite this article
Holmberg, A.H., Johnell, O., Nilsson, P.M. et al. Risk factors for fragility fracture in middle age. A prospective population-based study of 33,000 men and women. Osteoporos Int 17, 1065–1077 (2006). https://doi.org/10.1007/s00198-006-0137-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-006-0137-7