Abstract
The total mercury (Hg) content of the soft tissues of cultured oysters of the genus Crassostrea obtained during the dry and rainy seasons at sampling sites of NW Mexico with different degrees of urbanization, was determined by cold vapor atomic absorption spectrophotometry. Hg levels ranged from 0.05 to 0.37 µg/g (dry weight) and no significant differences (p > 0.05) related to season and sampling site were observed. The values did not exceed the limit of 1.0 µg/g (wet weight) established by Mexican legislation and by the Food and Drug Agency (FDA), and the hazard quotient was between 0.001 and 0.002. The estimated hazard quotient for MeHg ranged approximately from 0.002 to 0.01.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Mercury (Hg) enters the environment through volcanism, fossil fuel and waste burning and from other human as well as natural processes. It has a high environmental cost due to its non-biodegradable nature and long biological half-life (Hylander and Goodsite 2006). It is a cause of ecological concern, because it accumulates in sediments and in aquatic organisms and is biomagnified along aquatic food chains (Pan and Wang 2011; Jara-Marini et al. 2012; Kim et al. 2012). Among the inorganic and organic Hg species of different toxicity, methylated Hg (MeHg) is well known for its neurotoxicity and because it affects several physiological processes (Ipolyi et al. 2004; Apeti et al. 2012). Although it may originate from human sources, methylation by bacterial activities is its most common and important source.
Oysters of the genus Crassostrea have been used as biomonitors of contamination of the aquatic environment, because of their adaptability to environmental conditions, tolerance to contamination and wide geographic distribution (Vaisman et al. 2005). This genus is represented along the Pacific Mexican coasts by four species. Three of these (C. corteziensis, C. iridescens, C. palmula) are native, and one (C. gigas) was introduced for aquaculture (Rangel-Dávalos 1990). Among these, C. corteziensis and C. gigas are cultured for human consumption along the NW coast of Mexico, although most sections of the coastline are impacted by the discharges of urban, industrial, mining and agriculture activities.
In this study we determined the concentration of total mercury in oysters of this genus obtained at different sampling sites of three coastal states of NW Mexico during the dry and the rainy season, and used this information to assess human health risk, considering the average rate of consumption in Mexico.
Methods and Materials
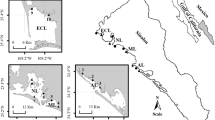
Samples were obtained in August and November 2010 (rainy season) and February and May 2011 (dry season) from twelve sites (two visits/season in each station) located on the coastal area of NW Mexico (Fig. 1). All oysters (20/sample) were of commercial size (shell height >5 cm) and those of each sample had similar height. After collection, oysters were washed with seawater, transported within 24 h to the laboratory in clean polyethylene bags kept in coolers, and stored at −20°C until processing for metal analysis (Vaisman et al. 2005). Samples from Sonora State were cultured Pacific oyster C. gigas (mean size: 9.5 ± 1.6 cm), those from Sinaloa and Nayarit States were pleasure oysters C. corteziensis cultured with different techniques (clutchless oysters in Sinaloa, with mean size: 8.9 ± 1.4 cm; traditional culture on mother shell collectors in Nayarit: mean size: 6.8 ± 1.2 cm). Those obtained at Mármol station were wild C. iridescens (rock oyster, mean size: 9.5 ± 1.1 cm), collected from their natural habitat.
After thawing, oysters were shucked with a stainless steel knife, the whole soft bodies of the 20 specimens of each sample were freeze dried (72 h at 80 × 10−3mbar, −50°C), ground and homogenized in a Teflon mortar to obtain a composite sample and 0.25 g of the homogenate were pre-digested overnight at room temperature with 5 mL of concentrated nitric acid (Baker, trace metal grade) (Rojas de Astudillo et al. 2002). Digestion was performed in closed Teflon bombs at 120°C for 3 h, and samples were diluted to 25 mL with MilliQ water. All materials and glassware used in sampling and metal analysis were acid cleaned (Moody and Lindstrom 1977).
Total Hg was determined by cold vapor atomic absorption spectrophotometry (Buck Scientific) after reduction with SnCl2 in a mercury analyzer (Buck Scientific) (Ruelas-Inzunza et al. 2004). For QA/QC all samples were analyzed in triplicate, the accuracy of the analytical method was evaluated with certified reference material (DORM 3) with a mean recovery of 104.3 % ± 1.0 % and parallel reagent blanks were used to check contamination. The limit of detection was 0.003 µg/g.
The values obtained in the dry and in the rainy season at each sampling station were compared with a two ways ANOVA test with α = 0.05 using site and sampling season as independent variables. The test was run after rank transformation, because the data were not homoscedastic (Conover and Iman 1981; Zar 1999; Conover 2012). The risk to human health due to oyster consumption was estimated according to Newman and Unger (2002), using the equation HQ = E/RfD, in which HQ is the hazard quotient, E and RfD are the level of Hg exposure and the reference dose for total Hg (0.5 µg/kg/day: FDA 2006), respectively, and the level of exposure E is calculated as E = C I/W where C is the total concentration of Hg (in µg/g wet weight) of the species tested, I is its apparent daily consumption (0.39 kg/person/year = 1.07 g/person/day: CONAPESCA 2011). W is the assumed weight of an adult consumer (70 kg).
The risk due to MeHg content of the oysters was estimated with the same equations, using 0.1 as RfD (EPA 2001) and the average MeHg/total Hg ratio calculated using the data available for oysters in Claisse et al. (2001), Pan and Wang (2011), and Apeti et al. (2012).
Results and Discussion
Hg values ranged from 0.05 to 0.34 µg/g (dry weight), determined during the rainy season at Santa Barbara and Tóbari, respectively. This range is comparable to that found by Apeti et al. (2012) in the Northern Gulf of Mexico. There were no significant differences due to site or sampling season. The annual mean values ranged between 0.12 and 0.27 µg/g (station Dautillos, Sinaloa and station Bachoco, Sonora, respectively), and the overall mean concentration, calculated using all the data obtained at each station was 0.172 ± 0.034 µg/g (Table 1).
This lack of difference may be explained by the Hg sources shared by the three states: although the production of the mining industry of Sonora is far higher than those of Sinaloa and Nayarit, the freshwater inputs to the coastal waters of these states flow from the mineral-rich Sierra Madre Occidental (states of Chihuahua, Durango and Zacatecas) where >70,000 ton of Hg are still present in mine tailings as residues of the >190,000 tons of Hg used from colonial times until the early 1900s in the amalgamation process involved in silver and gold extraction (Castro-Díaz 2013).
Important additional sources are the effluents from the 3.5 × 106 ha of fertile Pacific coastal plains dedicated to intensive agriculture, since Hg-based fungicides are still used for pest control of several crops (Osuna-Martínez et al. 2010). Other inputs are atmospheric deposition, because of Hg volatility which allows easy transport even to areas far from the geographic source (Vaisman et al. 2005; Apeti et al. 2012), and the Hg content of seawater, because of the continuous water renovation of the coastal lagoons and embayments (Osuna-Martínez et al. 2010; Harris et al. 2012).
There were no differences between the values determined in the rainy and dry season at each sampling site, but values tended to be higher in the rainy season. This might explain the higher metal levels (including Hg) found in oysters during the rainy than the dry season by Osuna-Martínez et al. (2010) in some coastal lagoons of Sinaloa State, who related this difference to increased continental runoff and concurrent high terrigenous sediment transport from metal-rich watersheds because of natural processes, such as rock weathering and leaching, or from metal-impacted surrounding areas (Yesudhason et al. 2013).
A comparison of the Hg levels in the soft tissues of oysters from several parts of the world show that, apart from the extremely high values reported for Minamata Bay, the range of data found in this study (0.05–0.37 µg/g) lies within the limits of most values found in literature, with the exception of those reported in Zhejiang, China and in the transition zone between the Persian Gulf and Arabian Sea (Oman and Qeshm Island, Iran), although with a tendency to be lower than the values reported for Villa Clara, Cuba and for the coast of Croatia. As to previous studies in NW Mexico, the range found by García-Rico et al. (2010) was similar to that of this study, the upper limits of those obtained by Osuna-Martínez et al. (2010) for C. gigas and C. corteziensis were between twice and three times higher, whereas the upper limit of the Hg content of the wild oysters of Urías lagoon (Jara-Marini et al. 2012) was lower than the lowest of our mean values (Table 2).
Due to its high toxicity even at low concentrations, to the higher toxicity and high affinity to biological systems of the end products of its biotransformation, especially of methyl-mercury (Clarkson and Magos 2006), risk assessments of Hg exposure should be carried out in all world´s coastal zones. For instance, Shirneshan et al. (2012) reported no risk by human consumption in view of the low values detected in Qeshm Island (Persian Gulf), and a similar result was obtained by Bogdanović et al. (2014) in their biomonitoring survey along the coast of Croatia. In the case of this study, all HQ values were <1 (Table 1) indicating no risk for oyster consumption in NW Mexico.
In general, because of their low trophic position, bivalves have lower Hg content than fishes (Jara-Marini et al. 2012; Kim et al. 2012). However their contents may reach toxic levels for top predators because of biomagnification through the food web, which is of ecological as well as human health concern (Fang et al. 2004; Ochoa et al. 2013). The oysters used for this study did not exceed the 1.0 µg/g (wet weight) limit established by Mexican legislation (SSA 1995) and FDA (2006) and the calculated HQ was <1. These values indicate that at least for the time being the Hg concentration of cultivated oysters is not a matter of concern although, in view of increasing urbanization and industrialization of NW Mexico, the Hg content of aquatic organisms should be monitored in order to prevent human health risk.
References
Aguilar CA, Montalvo C, Rodríguez L, Cerón JG, Cerón RM (2012) American oyster (Crassostrea virginica) and sediments as a coastal zone pollution monitor by heavy metals. Int J Environ Sci Technol 9:579–586
Apeti DA, Lauenstein GG, Evans DW (2012) Recent status of total mercury and methyl mercury in the coastal waters of the northern Gulf of Mexico using oysters and sediments from NOAA´s mussel watch program. Mar Pollut Bull 64:2399–2408
Bogdanović T, Ujević I, Sedak M, Listeś E, Śimat V, Petričević S, Poljak V (2014) As, Cd, Hg and Pb in four edible shellfish species from breeding and harvesting areas along the eastern Adriatic coast, Croatia. Food Chem 146:197–203
Castro-Díaz J (2013) An assessment of primary and secondary mercury supplies in Mexico. Commission for environmental cooperation, Montreal
Claisse D, Cossa D, Bretaudeau-Sanjuan J, Touchard G, Bombled B (2001) Methylmercury in molluscs along the French coast. Mar Pollut Bull 42:329–332
Clarkson TW, Magos L (2006) The toxicology of mercury and its chemical compounds. Crit Rev Toxicol 36:609–662
CONAPESCA (2011) Anuario estadístico de acuacultura y pesca. Comisión Nacional de Pesca, Mazatlán
Conover WJ (2012) The rank transformation-an easy and intuitive way to connect many nonparametric methods to their parametric counterparts for seamless teaching introductory statistics courses. WIREs Comp Stat 4:432–438
Conover WJ, Iman RL (1981) Rank transformations as a bridge between parametric and nonparametric statistics. Am Stat 35:124–133
Eisler R (1987) Mercury hazards to fish, wildlife and invertebrates: a synoptic review. Biological report 85, Contaminant hazard reviews 10, US Fish and Wildlife Service
EPA (2001) Water quality criterion for the protection of human health: methylmercury. Washington DC. http://water.epa.gov/scitech/swguidance/standards/criteria/health/upload/2009_01_15_criteria_methylmercury_mercury-criterion.pdf
Fang J, Wang KX, Tang JL, Wang M, Ren SJ, Wu HY, Wang J (2004) Copper, lead, zinc, cadmium, mercury, and arsenic in marine products of commerce from Zheijiang coastal area, China. Bull Environ Contam Toxicol 73:583–590
FDA (2006): Mercury levels in commercial fish and shellfish. Washington DC. www.cfsan.fda.gov/~frf/sea-mehg.html
García-Rico L, Tejeda-Valenzuela L, Burgos-Hernández A (2010) Seasonal variations in the concentrations of metals in Crassostrea corteziensis from Sonora, Mexico. Bull Environ Contam Toxicol 85:209–213
Harris R, Pollman C, Landing W, Evans D, Axelrad D, Hutchinson D, Morey SL, Rumbold D, Dukhovskoy D, Adams DH, Vijayaraghavan K, Holmes C, Atkinson RD, Myers T, Sunderland E (2012) Mercury in the Gulf of Mexico: sources to receptors. Environ Res 119:42–52
Hylander LD, Goodsite ME (2006) Environmental cost of mercury pollution. Sci Total Environ 368:352–370
Ipolyi I, Massanisso P, Sposato S, Fodor P, Morabito R (2004) Concentration levels of total and methylmercury in mussel samples collected along the coasts of Sardinia Island (Italy). Anal Chim Acta 505:145–151
Jara-Marini ME, Soto-Jiménez MF, Páez-Osuna F (2012) Mercury transfer in a subtropical coastal lagoon food web (SE Gulf of California) under two contrasting climatic condition. Environ Toxicol 27:526–536
Kim E, Kim H, Shin KH, Kim MS, Kundu SR, Lee BG, Han S (2012) Biomagnification of mercury through the benthic food webs of a temperate estuary: Masan Bay Korea. Environ Toxicol Chem 31:1254–1263
Moody JR, Lindstrom RN (1977) Selection and cleaning of plastic containers for storage of trace elements samples. Anal Chem 49:2264–2267
Newman MC, Unger MA (2002) Fundamentals of ecotoxicology. Lewis Publishers, Boca Raton
Ochoa V, Barata C, Riva MC (2013) Heavy metal content in oysters (Crassostrea gigas) cultured in the Ebro delta in Catalonia, Spain. Environ Monit Assess 185:6783–6792
Olivares-Rieumont S, Lima L, Rivero S, Graham DW, Alonso-Hernandez C, Bolaño Y (2012) Mercury levels in sediments and mangrove oysters, Crassostrea rhizophorae from the North coast of Villa Clara, Cuba. Bull Environ Contam Toxicol 88:589–593
Osuna-Martínez CC, Páez-Osuna F, Alonso-Rodríguez R (2010) Mercury in cultured oysters (Crassostrea gigas Thunberg, 1793 and C. corteziensis Hertlein, 1951) from four coastal lagoons of the SE Gulf of California, Mexico. Bull Environ Contam Toxicol 85:339–343
Pan K, Wang WX (2011) Mercury accumulation in marine bivalves: influences of biodynamics and feeding niche. Environ Pollut 159:2500–2506
Rangel-Dávalos C (1990) El cultivo de moluscos marino en México. In: De la Lanza-Espino G, Arredondo-Figueroa JL (eds) La acuicultura en México: de los conceptos a la producción. UNAM, México DF, pp 107–138
Rojas de Astudillo L, Yen IC, Agard J, Bekele I, Hubbard R (2002) Heavy metals in green mussel (Perna viridis) and oysters (Crassostrea sp.) from Trinidad and Venezuela. Arch Environ Contam Toxicol 42:410–415
Ruelas-Inzunza J, García-Rosales SB, Páez-Osuna F (2004) Distribution of mercury in adult penaeid shrimp from Altata-Ensenada del Pabellón (SE Gulf California). Chemosphere 57:1657–1661
Shirneshan G, Bakhtiari AR, Kazemi A, Mohamadi M, Kheirabadi N (2012) Oyster Saccostrea cucullata as a biomonitor for Hg contamination and the risk to humans on the coast of Qeshm Island, Persian Gulf, Iran. Bull Environ Contam Toxicol 88:962–966
SSA (1995) Norma Oficial Mexicana NOM-031-SSA1-1993. Bienes y servicios. Productos de la pesca. Moluscos bivalvos frescos-refrigerados y congelados. Especificaciones sanitarias. Secretaria de Salud. Diario Oficial de la Federación, 6 de marzo 1995
Vaisman AG, Marins RV, Lacerda LD (2005) Characterization of the mangrove oyster, Crassostrea rhizophorae, as a biomonitor for mercury in tropical estuarine systems, Northeast Brazil. Bull Environ Contam Toxicol 74:582–588
Yesudhason P, Al-Busaidi M, Al-Rahabi W, Al-Waili AS, Al-Nakhaili AK, Al-Mazrooei NA, Al-Habsi SH (2013) Distribution patterns of toxic metals in the marine oyster Saccostrea cucullata from the Arabian Sea in Oman: spatial, temporal, and size variations. SpringerPlus 2:1–11
Zar JH (1999) Biostatistical analysis. Prentice Hall, Upper Saddle River
Acknowledgments
Supported by projects Red Delfín, PROFAPI-UAS 2014/065; PROMEP/103.5/13/9354 and CONACYT INFRA 2012-01-188029. The assistance of G. Ramírez, K. Sánchez and P. Rosas is gratefully aknowledged. The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Delgado-Álvarez, C.G., Ruelas-Inzunza, J., Osuna-López, J.I. et al. Total Mercury Content in Cultured Oysters from NW Mexico: Health Risk Assessment. Bull Environ Contam Toxicol 94, 209–213 (2015). https://doi.org/10.1007/s00128-014-1430-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-014-1430-3