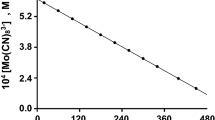
Abstract
Crude extracts or supernatants of broken cells of Clostridium formicoaceticum reduce unbranched, branched, saturated and unsaturated carboxylates at the expense of carbon monoxide to the corresponding alcohols. The presence of viologens with redox potentials varying from E 0′=-295 to-650 mV decreased the rate of propionate reduction. The more the propionate reduction was diminished the more formate was formed from carbon monoxide. The lowest propionate reduction and highest formate formation was observed with methylviologen. The carbon-carbon double bond of E-2-methyl-butenoate was only hydrogenated when a viologen was present. Formate as electron donor led only in the presence of viologens to the formation of propanol from propionate. The reduction of propionate at the expense of a reduced viologen can be followed in cuvettes. With respect to propionate Michaelis Menten behavior was observed. Experiments are described which lead to the assumption that the carboxylates are reduced in a non-activated form. That would be new type of biological reduction.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Abbreviations
- glc:
-
Gas liquid chromatography
- HPLC:
-
high performance liquid chromatography
- RP:
-
reverse phase; Mediators (the figures in parenthesis of the mediators are redox potentials E 0′ in mV)
- CAV2+ :
-
carbamoylmethylviologen, 1,1′-carbamoyl-4,4′-dipyridinium dication (E 0′=-296 mV)
- BV2+ :
-
benzylviologen, 1,1′-dibenzyl-4,4′-dipyridinium dication (E 0′=-360 mV)
- MV:
-
methylviologen, 1,1′-dimethyl-4,4′-dipyridinium-dication (E 0′=-444 mV)
- DMDQ2+ :
-
dimethyldiquat, 4,4′-dimethyl-2,2′-dipyridino-1,1′-ethylendication (E 0′=-514 mV)
- TMV2+ :
-
tetramethylviologen, 1,1′,4,4′-tetramethyl-4,4′-dipyridinium dication (E 0′=-550 mV)
- PDQ2+ :
-
propyldiquat, 2,2′-dipyridino-1,1′-propenyl dication (E 0′=-550 mV)
- DMPDQ2+ :
-
dimethylpropyldiquat, 4,4′-dimethyl-2,2′-dipyridino-1,1′-propenyl dication (E 0′=-656 mV)
- PN:
-
productivity number=mmol product (obtained by the uptake of one pair of electrons) x (biocatalyst (dry weight) kg)-1×h-1
References
Andreesen, JR, Gottschalk G, Schlegel HG (1970) Clostridium formicoaceticum nov. spec. Isolation, description, and distinction from C. aceticum and C. thermoaceticum. Arch Mikrobiol 72:154–174
Diekert GB, Thauer RK (1978) Carbon monoxide oxidation by Clostridium thermoaceticum and Clostridium formicoaceticum. J Bacteriol 136:597–606
Fuchs G (1986) CO2 fixation in acetogenic bacteria: variation on a theme. FEMS Microbiol Rev 39:181–213
Loach PA (1976) Oxidation-Reduction potentials, absorbance bands and molar absorbance of compounds used in biochemical studies. In: Fasman GD (ed) Handbook of biochemistry and molecular biology, 3rd edn, Physical and chemical data, vol 1. CRC Press, Cleveland Ohio, pp 122–130
Read SM, Northcote DH (1981) Minimization of variation in the response of different proteins of the Coomassie blue G dye binding assay for protein. Anal Biochem 116:53–64
Schaller KH, Triebig G (1984) Determination with formate dehydrogenase. In: Bergmeyer HU (ed) Methods of enzymatic analysis, vol 6. Verlag Chemie, Weinheim, pp 668–672
Simon H, White H, Lebertz H, Thanos I (1987) Reduktion von 2-Enoaten und Acylaten mit Kohlenmonoxid oder Formiat, Viologenen, und Clostridium thermoaceticum zu gesättigten Säuren und ungesättigten bzw. gesättigten Alkoholen. Angew Chem 99:785–787. Angew Chem Int Ed Engl 26:785–787
White H, Lebertz H, Thanos I, Simon H (1987) Clostridium thermoaceticum forms methanol from carbon monoxide in the presence of viologens or cobalt sepulchrate. FEMS Microbiol Lett 43:173–176
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Fraisse, L., Simon, H. Observations on the reduction of non-activated carboxylates by Clostridium formicoaceticum with carbon monoxide or formate and the influence of various viologens. Arch. Microbiol. 150, 381–386 (1988). https://doi.org/10.1007/BF00408311
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00408311