Abstract
The emergence of modern microbial technology and product biotechnology had significantly made over the way, scientists and researchers view differently the microbes and the product they biosynthesize. Pullulan is one of the most potent bio-compatible polymers which is basically synthesized by the Aureobasidium pullulans. This microbial pullulan acts as the promising biomaterial that is currently used for packaging of readily oxidized food materials, controlled drug delivery, tissue engineering and can also function as artificial molecular chaperones. Commercial pullulan is expensive. It was also estimated that the cost of pullulan is three times higher than the other polysaccharides. However, the cost of the raw materials required for pullulan production accounts for 30% of the total production costs. Therefore, it is essential to search out the cheapest substrate for the production of pullulan. From 1999 till date, different groups of researcher had found and reported various carbon source and nitrogen source from the waste products to be utilized for pullulan production. This review attempts to critically appraise the current literature on ‘pullulan’ considering its microbial sources and utilization of various agro-industrial wastes for pullulan production.
Access provided by CONRICYT-eBooks. Download chapter PDF
Similar content being viewed by others
Keywords
1 Introduction
Research studies in the field of carbohydrates have revealed that pullulan is one of the most emerging biopolymers, basically synthesized by Aureobasidium pullulans. This polymer is consisting of maltotriose units connected by α(1 → 4) glycosidic bond. The consecutive maltotriose units are inter-linked with α(1 → 6) glycosidic bond (Bender and Wallenfels 1961). This alternation of α(1 → 4) and α(1 → 6) bonds, is the main cause of its structural flexibility and hydrophilic nature. The daily intake of pullulan should be up to 10 g per day as per the FDA (Singh and Saini 2008). The major constraint prevailing on the use of pullulan is its cost that is three times more than the price of other polysaccharides. The general structure of pullulan has been illustrated in the Fig. 1.
(Singh and Saini 2008)
Dry pullulan powders are white, crystalline or amorphous and non-hygroscopic and dissolve quickly in the cold or hot water. The pullulan shows its resistance to the mammalian amylases. Hence, it provides fewer calories and can be treated as dietary fibre. The employment and application of pullulan in biomedical and tissue engineering field are emerging owing to its biocompatible, non-toxic, non-immunogenic and inert nature. In comparison to dextran, the degradation rate of pullulan in blood serum is much quicker. It was found that the degradation index was 0.7 for pullulan with the intervals of 48 h of incubation while it was of 0.05 for the dextran in the same condition (Bruneel and Schacht 1995). Pullulan is suitable for all consumer groups because of its non-animal origin nature. It is a slow digesting macromolecule, which is colourless, odourless and hence it is used as very low calorie food additives. This is used as a texturizer and glazing agent in chewing gum and bubble gum. It is also used as foaming agent in milk-based desserts. Pullulan contributes major four areas of applications commercially like food, pharmaceutical, biomedical and other miscellaneous as described in Fig. 2.
2 Microbial Sources of Pullulan
Although it is well-known fact that A. pullulans is a key producer of pullulan, but not all the strains of A. pullulans have the potential to synthesize pullulan (Ueda et al. 1963; Leathers et al. 1988).
Different types of microbial strains apart from the A. pullulans having the ability to produce pullulan have been summarized in Table 1.
3 Fermentative Production of Pullulan
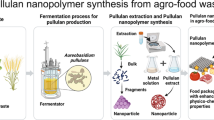
In the industrial scale, pullulan is produced from the liquefied starch by using non-toxigenic and non-pathogenic strain under specific conditions. The upstream part of pullulan production by the fermentation deals with the initial step of the process like selection of the potential strain, inoculum preparation, medium development, growth kinetics, optimization etc., in order to get the product with maximum yield. In submerged fermentation, the nature of the liquid medium is changed drastically during production of pullulan. Initially, the liquid follows the Newtonian behaviour as the viscosity of the medium is very close to pure water. After sometimes, when the polymer is synthesized, the behaviour of the liquid medium changes to non-Newtonian due to increase in the apparent viscosity (Lazaridou et al. 2002).
Downstream processing is essential to meet the purity and quality of the final product from the fermentation broth. There are several steps which are mainly composed of separation of biomass from the culture broth, removal of melanin pigments, proteins and finally precipitation of pullulan with the appropriate organic solvent. Co-production of melanin pigment along with pullulan is a major problem. It was reported that the melanin is produced in the pentaketide pathway at the end of the fermentation (Siehr 1981; Dharmendra et al. 2003). However, pullulan is also synthesized during the late log phase or early stationary phase of the growth of the microorganism (Longfa et al. 2011). After the pigment separation, the main step is to do the precipitation of polymer with suitable organic solvent. Generally, two phases are developed after the precipitation. The deposition of the precipitated pullulan in the solvent system depends upon the molecular weight of the polymer. An appropriate and cost-effective downstream processing is the major requirement and necessary for the production of pullulan. The schematic representation of pullulan production has been illustrated in the Fig. 3.
All the major research in the process of pullulan production was carried out with A. pullulans as the yield is very high as comparison to other microbial sources. Pullulan production efficiency of different strains of A. pullulans has been summarized in Table 2.
4 Production of Pullulan from Agro-industrial Wastes
Production of pullulan from agro-industrial wastes is both feasible and ecologically sound. Therefore, it would be prudent to search for the available and suitable substrate and select the proper strain of the pullulan-producing organism to produce a valuable product, while protecting the environment from pollution. Pollution problems associated with accumulation of agro-industrial wastes and by-products increased the demand for bioconversion of the plant biomass to value-added compounds by economically feasible ways (Table 3).
The cost of pullulan production currently is relatively high as compared to other carbohydrate. Hence, it is very essential to search for inexpensive carbon and nitrogen sources, which are nutritionally rich enough to support the growth of the microorganism as well as the production of pullulan.
5 Sweet Potatoes
Sweet potato (Ipomoea batatas) is a cheap and easily available agriculture product. The major constituent of sweet potato is starch and little sugar. Sweet potato has been used as a suitable carbon source for various industrial fermentations. Wu et al. (2009) investigated the utilization of sweet potato waste by A. pullulans AP329 for pullulan production in submerged fermentation. Generally, starch should be hydrolysed to sugars before starting the fermentation of pullulan production. But, there is probably no need to add expensive β-amylase when sweet potato is used as carbon source in the fermentation. Because, it contains considerable amount of highly active β-amylase, which is the primary saccharifying enzyme in sweet potato.
6 Asian Palm Kernel
Research-based studies were done by Sugumaran et al. (2014a) in which four agro-wastes namely, wheat bran, rice bran, coconut kernel, and palm kernel were evaluated as a low carbon source the pullulan production by A. pullulans in solid state for fermentation at 50% moisture content. Palm kernel emerged to be the best carbon source among the four agro-wastes and yield 16 g/L pullulan. Later, the same group of researchers had studied the optimization of the process parameters for the pullulan production considering the carbon source as Asian Palm kernel with Response Surface Methodology (RSM). The pullulan yield was increased to 30.4 g/L. Thus, palm kernel appears to be a potential low-cost carbon source for the production of the pullulan.
7 Soybean by-Products
Soybean pomace is a major agro-industrial by-product from the soy sauce industry. It consists of carbohydrates and proteins as major component. Although soybean pomace has very essential utility, it is discarded as waste because of its high content of sodium chlorite (NaCl). This creates very serious environmental problems. Moreover, considering soybean pomace as a waste is a huge loss to the natural resources as it is a rich source of carbohydrates and proteins. A research-based study was done by Seo et al. (2004a, b), in which yeast extract was replaced by soybean pomace as nitrogen source for production of pullulan by A. pullulan HP-2001.
8 Brewery Wastes
Spent grain liquor is a significant source of waste in the brewing industry. It is the liquor resulting from the final separation of the wort from spent grain in a brewing plant. It consists of water, proteins, mineral salts and total sugars as glucose. A number of brewery wastes were examined as substrates for fungal growth (Shannon and Stevenson 1975). Hang et al. (1977) reported that Aspergillus niger produces a significant amount of citric acid during the fermentation of brewery spent grain liquor. Pullulan production from brewery waste by A. pullulans has been investigated by Roukas (1999).
9 Coconut By-product
Coconut water is a naturally occurring clear liquid at the fruit’s centre. It consists of easily digested carbohydrate in the form of simple sugars and electrolytes. Coconut milk is a liquid which is obtained from the grated meat of mature coconut. Especially, various factories producing desiccated coconut, copra and other coconut meat products (Coco sauce, coconut honey, coconut chips, roasted young coconut, coconut cream, coconut candy, coconut flour) generate the waste product in the form of coconut water and coconut milk. Due to higher Biological Oxygen Demand (BOD) of coconut by-product, it is considered as an active pollutant in the environment. This environmental problem has increased the interest of current researchers in coconut by-product and motivated its utilization in the production of such an industrially important product. Thirumavalavan et al. (2009) have studied and used both coconut water and coconut milk for the pullulan production. The coconut milk found to be more efficient for pullulan production as compared to coconut water as C/N ratio is higher in case of coconut milk.
10 Jackfruit Seed
Jackfruit (Artocarpus heterophyllus) tree is an evergreen tree that is mainly found in Florida, Central and Eastern African nations, India, Australia and most of the Pacific islands. Generally, only fleshy pulp of jackfruit is eaten and seeds are considered as the by-product and discarded as waste products. It was found that these seeds contributed 8–15% of the total fruit weight. These seeds contain both proteins and carbohydrates content (Bobbio et al. 1978). The potential of jackfruit seeds as carbon and nitrogen source was studied by Sugumaranc et al. (2013) for pullulan production (34.22 g/L) in solid state fermentation. The optimal concentration of different factors for pullulan production was as follows: NaCl−1.69 g/L, KH2PO4 −4.16 g/L, ZnSO4·5H2O−0.052 g/L and moisture content −47.9%, w/w.
11 Jaggery
Jaggery, a concentrated sugar cane juice with or without prior purification produced by cottage industries (also known as gur), containing 75–85% sucrose is widely used in India as a substitute of white and refined cane sugar. Vijayendra et al. (2001) and Ganduri et al. (2016) had used jaggery as carbon source for the production of pullulan with A. pullulans CFR-77 and by A. pullulans MTCC 2195 respectively.
12 Beet Molasses
Few researchers (Yekta et al. 2004; Lazaridou et al. 2002) had investigated about the production of pullulan by utilizing beet molasses as carbon source with a strain of A. pullulans P56 (a melanin-deficient strain). Maximum sugar utilization was found to be more than 90%. Overall, beet molasses were proven as an attractive fermentation medium for the production of pullulan by A. pullulans.
13 Cassava By-products
Cassava bagasse disposed from sago industry causes high pollution owing to its organic content and more biodegradability (Singh et al. 2008). As a result, biotransformation of these industrial solid wastes to value added products is helpful to our society to minimize the environmental pollution as well as cost of fermentation. Sugumaranb et al. (2014) have used Cassava bagasse for pullulan production in solid state fermentation with A. pullulans MTCC2670. Approximately about 600–650 tonnes per day of cassava bagasse are disposed from sago industry in India (Srivas and Anatharaman 2005). Ray et al. (2007) had investigated the production of pullulan by using cassava starch residues by A. pullulans MTCC1991. Cassava by-products a low-cost and easily available waste material from starch extraction from cassava could provide an economic advantage as a solid substrate as well as a carbon source for production of the emerging pullulan elaborated by the organism A. pullulans.
14 Corn Steep Liquor
Corn steep liquor is a high-protein (42%) feed ingredient, which is obtained from the soluble parts of the corn kernel in the steeping process. Sharma et al. (2013) have investigated about five different agricultural wastes (rice bran oil cake, soya bean oil cake, cotton seed oil cake, mustard seed oil cake and corn steep liquor) as nitrogen source for the production of pullulan by A. pullulans. Corn steep liquor was found to be best one, which can yield 77.92 g/L of pullulan. This fermentation process was also validated in a 7-L fermenter and a process economics was analysed with respect to cost of pullulan production. It was found that, corn steep liquor can make threefold reduction cost of raw materials for pullulan production in comparison to a conventional process. These observations can be useful in development of a cost-effective pullulan production.
15 De-Oiled Jatropha Seed Cake
Jatropha seed oil is used extensively for the bio-diesel production from last decades. Generally, after the bio-diesel production the seed cake has to be discarded as waste product. The de-oiled seed is very toxic to both human and animal health. Hence its use is restricted. In addition to that, the de-oiled jatropha seed cake comprises of 55–65% of biomass used for the production of bio-diesel. Hence it causes major environmental problem for its disposal. The potential of de-oiled jatropha seed cake used as a substrate for pullulan production was investigated by Chaudhury et al. (2012). It was found that, the yield of pullulan was 83.98 g/L in a 5L laboratory scale fermenter.
16 Potatoes Starch
The cells of root tuber of the potato contain starch grain. Large amount of waste residue has been released in the potato starch industry, which includes effluents and potato residues. This causes serious environmental problems. The major constituents of the potato starch waste consist of carbohydrates. These effluents are having COD (chemical oxygen demand) more than 30 g/L, hence it is rich with biodegradable components (starch, cellulose and proteins) and can be utilized by the microorganism. Yekta et al. (2011) had investigated the utility of potatoes starch waste for pullulan production by A. pullulan P56. The potato starch was liquefied by pullulanase and amyloglucosidase enzymes (Ca-alginate immobilized form) in a packed bed bioreactor. Maximum pullulan production was found to be 19.2 g/L and after optimization of various process parameters, the production was increased by 20% as compared to initial state.
The mixtures of potato starch hydrolysate and sucrose were also utilized for the production of pullulan (Chao et al. 2017). It was found that the mixtures of potato starch hydrolysate and sucrose could promote pullulan synthesis and possibly that a small amount of sucrose stimulated the enzyme responsible for pullulan synthesis and promoted effective potato starch hydrolysate conversion effectively. Thus, mixed sugars in potato starch hydrolysate and sucrose fermentation might be a promising alternative for the economical production of pullulan.
The utilization of raw potato starch hydrolysates for pullulan production was also investigated. The maximum pullulan production was found to be 36.17 g/L after 96 h of the fermentation. A comparative study was performed with glucose and sucrose as carbon source for pullulan production, which resulted 22.07 and 31.42 g/L of pullulan respectively (Shengjun et al. 2016). These results infer that raw potato starch hydrolysates can be utilized as carbon source for pullulan production in a cost-effective manner.
17 Rice Hull Hydrolysate
Rice hull is one of the most widely available agricultural by-products in many rice producing countries, which generates 120 million metric tonnes per year. The physic-chemical pre-treatment is required for efficient conversion of rice hull into the fermentable sugars. Hydrolysis process with dilute sulphuric acid has been used to recover usable sugars from the rice hull in efficient manner. A research based studies was done by Dahui et al. (2014) for the production of pullulan from rice hull hydrolysate by A. pullulans CCTCC M 201259. They found that acetic acid presents in the hydrolysate exert a negative effect on pullulan production. Hence, a mutagenic study was performed with the A. pullulans CCTCC M 201259 and a new mutated strain (designated as A. pullulans ARH-1) was isolated in a medium containing acetic acid. The maximum pullulan production was found to be 22.2 g/L by A. pullulans ARH-1 after 48 h, while that obtained by the parental strain (A. pullulans CCTCC M 201259) was 15.6 g/L after 60 h of fermentation.
18 Strain Improvement for Pullulan Production
Pollock et al. (1992) had investigated the mutagenic study with respect to Aureobasidium strain for the first time by using Ethidiumbromide as the chemical mutagen. The mutated strain produced higher molecular weight pullulan as compared to parent strain.
Gniewosz and Duszkiewicz-Reinhard (2008) had screened a mutant strain of A. pullulans A.p.-3 with reduced pigmentation by the physical mutagenesis for the higher production of pullulan. In one of the study, a pullulan-producing strain of Aspergillus japonicus-VITSB1 was mutated by UV rays and EMS mutagenesis. The resulted mutant strain showed an increased molecular weight, less pigmentation with higher yield of pullulan (Mishra and Suneetha 2014a, b).
Periyasamy et al. (2015) had mutated a strain of Aureobasidium mousonni (NCIM 1226) through UV ray treatment for enhanced pullulan production. It was found that the mutated strain could produce 1.4 times more pullulan than that of parent strain. Most of the research in this orientation had carried out to find a melanin deficient strain for pullulan production.
Xiaoliu et al. (2012) had screened a mutant strain of A. pullulans SZU 1001 deficient in pigment production and was screened by complex UV and γ-ray mutagenesis studies. Medium composition optimization for increased pullulan molecular weight and production was conducted using this mutant.
19 Metabolic Engineering
There are very few reports available regarding the metabolic engineering for pullulan production. Wang et al. (2013) investigated the effect of ATP/ADP ratio for biosynthesis of pullulan by A. pullulans. They found that production of pullulan is directly proportional to the ATP concentration in the cell.
Ma et al. (2015) studied the about production of pullulan by a newly isolated marine yeast of Aureobasidium melanogenum P16 from inulin. They have integrated INU1 gene (from Kluyveromyces maximum KM) into the genomic DNA of A. melanogenum P16. As there was not any gene encoding inulinase in this yeast, so inulin can be directly converted to pullulan.
It was also found that homologous expression of ApUGPase in A. pullulans NRRLY-12974 can effectively improve the yield of extracellular pullulan. The yield of pullulan in engineering strain was improved to 1.3-fold time of parent strain (Haifeng et al. 2016). Recent study showed that knock-out of PKSIII gene in A. pullulans decreasing the production of melanin and pullulan and knock-in of gltP gene leads to increased production of heavy oil and pullulan (Jian et al. 2017).
20 Conclusion
Current research shows that production of pullulan from agro-industrial wastes is feasible and ecologically sound. Utilization of agro-industrial waste for the production of pullulan is a challenging. Enhancement of both purity and productivity of pullulan could be achieved by the employment of proper downstream processing and improving the C/N ratio of the substrate. Selection of proper substrate and microbial strain with optimized conditions can yield higher amount of pullulan.
References
Bender H, Wallenfels K (1961) Investigations on pullulan. II. Specific degradation by means of a bacterial enzyme. Biochem Z 334:79–95
Bobbio FO, Dash AA, Bobbio PA, Rodrigues LR (1978) Isolation and characterization of the physic-chemical properties of starch of jackfruit seeds (Artocarpus heterophyllus). Cereal Chem 55:505–511
Bruneel D, Schacht E (1995) End group modification of pullulan. Polymer 36:169–172
Chao A, Sai-jian M, Fan C, Wen-jiao X (2017) Efficient production of pullulan by Aureobasidium pullulans grown on mixtures of potato starch hydrolysate and sucrose 48:180–185
Chaudhury AR , Nishat S, Prasad GS (2012) Deoiled Jtropha seed cake is a useful nutrient for pullulan production Microbial Cell Factrories 11:33 doi: 10.1186/1475-2859-11-39
Chen J, Wu S, Pan S (2012) Optimization of medium for pullulan production using a novel strain of Auerobasidium pullulans isolated from sea mud through response surface methodology. Carbohydr Polym 87:771–774
Cheng KC, Demirci A, Catchmark JM (2011) Continuous pullulan fermentation in a biofilm reactor. Appl Microbiol Biotechnol 90:921–927
Chi Z, Zhao S (2003) Optimization of medium and cultivation conditions for pullulan production by a new pullulan-producing yeast strain. Enzyme Microb Technol 33:206–211
Dahui W, Xiaomin J, Donghai Z, Gongyuan W (2014) Efficient production of pullulan using rice hull hydrolysate by adaptive laboratory evolution of Aureobasidium pullulans. Biores Technol 164:12–19
Delben F, Forabosco A, Guerrini M, Liut G, Torri G (2006) Pullulans produced by strains of Cryphonectria parasitica-II. Nucl Magn Reson Evid Carbohydr Polym 63:545–554
Dharmendra KK, Paramita B, Rekha SS (2003) Studies on downstream processing of pullulan. Carbohydr Polym 52:25–28
Forabosco A, Bruno G, Sparapano L, Liut G, Marino D, Delben F (2006) Pullulans produced by strains of Cryphonectria parasitica-I. Production and characterization of the exopolysaccharides. Carbohydr Polym 63:535–544
Fraser CG, Jennings HJ (1971) A glucan from Tremella mesenterica NRRL-Y6158 49:1804–1807
Ganduri VSRK, Sambasiva Rao KRS, Usha Kiranmayi M, Vijaya Lakahmi M, Sudhakar P (2016) Production of Pullulan using jaggery as substrate by Aureobasidium pullulans MTCC 2195. Curr Trends Biotechnol Pharma 10:153–160
Gao W, Chung CH, Li J, Lee JW (2011) Application of statistical experimental design for optimization of physiological factors and their influences on production of pullulan by Aureobasidium pullulans HP-2001 using an orthogonal array method. Korean J Chem Engg 28:2184–2189
Gniewosz M, Duszkiewicz-Reinhard W (2008) Comparative studies on pullulan synthesis, melanin synthesis and morphology of white mutant Aureobasidium pullulans B-1 and parent strain A.p.-3. Carbohydr Polym 72:431–438
Haifeng L, Yunyi Z, Yunyun G, Yuanyang L, Xiaopu Y, Lifeng H (2016) Characterization of UGPase from Aureobasidium pullulans NRRLY-12974 and application in enhanced pullulan production. Appl Biochem Biotechnol 178:1141–1153. http://dx.doi.org/10.1007/s12010-015-1934-2
Hang YD, Splittstoesser DF, Woodamss EE, Sherman RM (1977) Citric acid fermentation of brewery waste. J Food Sci 42:383–385
Jian G, Yuanhua W, Baozhong L, Siyao H, Yefu C, Xuewu G, Dongguang X (2017) Development of a one-step gene knock-out and knock- in method for metabolic engineering of Aureobasidium pullulans. J Biotechnol 251:145–150
Lazaridou A, Roukas T, Billiaderis CG, Vaikousi H (2002) Characterization of pullulan produced from beet molasses by Aureobasidium pullulans in stirred tank reactor under varying agitation. Enzyme Microb Technol 31:122–132
Leathers TD (2003) Biotechnological production, applications of pullulan. Appl Microbiol Biotechnol 62:468–473
Leathers TD, Nofsinger GW, Kurtzman CP, Bothast RJ (1988) Pullulan production by color variant strains of Aureobasidium pullulans. J Ind Microbiol 3:231–239
Liva EM, Cirelli AF, Lederkremer RMD (1986) Characterization of a pullulan in Cyttaria darwinii. Carbohydr Res 158:262–267
Longfa J (2010) Optimization of fermentation conditions for pullulan production by Aureobasidium pullulans using response surface methodology. Carbohydr Polym 79:414–417
Longfa J, Shengjun W, Kim JM (2011) Effect of different nitrogen sources on activities of UDPG-pyrophosphorylase involved in pullulan synthesis and pullulan production by Aureobasidium pullulans. Carbohydr Polym 86:1085–1088
Ma Z, Nan-Nan L, Zhe C, Guang-Lei L, Zhen-Ming C (2015) Genetic Modification of the Marine-Isolated Yeast Aureobasidium melanogenum P16 for Efficient Pullulan Production from Inulin. Mar Biotechnol 17:511–522
Mishra B, Suneetha V (2014a) Biosynthesis and hyper production of Pullulan by a newly isolated strain of Aspergillus japonicus-VIT-SB1. World J Microbiol Biotechnol 30:2045–2052
Mishra B, Suneetha V (2014b) Strain improvement and statistical analysis of pullulan producing strain of Aspergillus japonicus-VIT-SB1 for maximum yield. J Pure Appl Microbiol 8:1535–1545
Periyasamy B, Manchinella G (2015) Ramanathan A (2015) Strain improvement of Aureobasidium mausonii NCIM 1226 for Pullulan production and its applications. Inventi Impact: Pharm Tech 4:178–182
Pollock JT, Thorne L, Armentront RW (1992) Isolation of new Aureobasidium strains that produce high molecular weight pullulan with reduced pigmentation. Appl Environ Microbiol 58:877–883
Prasongsuk S, Berhow MA, Dunlap CA, Weisleder D, Leathers TD, Eveleigh DE, Punnapayak H (2007) Pullulan production by tropical isolates of Aureobasidium pullulans. J Ind Microbiol Biotechnol 34:55–61
Ray RC, Moorthy SN (2007) Exopolysaccharide (pullulan) production from cassava starch residue by Aureobasidium pullulans strain MTCC 1991. J Sci Ind Res 66:252–255
Reis RA, Tischer CA, Gorrin PAJ, Iacomini M (2002) A new pullulan and a branched (1→3), (1→6)-linked β-glucan from the lichenised ascomycete Teloschistes flavicans. FEMS Microbiol Lett 210:1–5
Roukas T (1999) Pullulan production from brewery wastes by Aureobasidium pullulans. World J Microbiol Biotechnol 15:447–450
Sena RF, Costelli MC, Gibson LH, Coughlin RW (2006) Enhanced production of pullulan by two strains of Aureobasidium pullulans with different concentrations of soybean oil in sucrose solution in batch fermentations. Braz J of Chem Engg 23:507–515
Seo H, Son C, Chung C, Jung D, Kim S, Gross RA, Kaplan DL, Lee J (2004) Production of high molecular weight pullulan by Aureobasidium pullulans HP-2001 with soybean pomace as a nitrogen source. Bioresour Technol 95:293–299
Shannon LJ, Stevenson KE (1975) Growth of Fungi and BOD reduction in selected brewery wastes. J Food Sci 40:826–828
Sharma N, Prasad GS, Choudhury AR (2013) Utilization of corn steep liquor for biosynthesis of pullulan, an important exopolysaccharide. Carbohydr Polym 93:95–101
Shengjun W, Mingsheng L, Jing C, Yaowei F, Leilei W, Yan X, Shujun W (2016) Production of pullulan from raw potato starch hydrolysates by a new strain of Auerobasidium Pullulans. Int J Biol Macromolec 82:740–743
Siehr DJ (1981) Melanin biosynthesis in Aureobasidium pullulans. J Coating Technol 53:23–25
Singh R, Gaur R, Tiwari S, Gaur MK (2012) Production of pullulan by a thermo tolerant Aureobasidium pullulans strain in non-stirred fed batch fermentation process. Braz J Microbiol 43:1042–1050
Singh RS, Saini GK (2008) Pullulan-hyperproducing color variant strain of Aureobasidium pullulans FB-1 newly isolated from phylloplane of Ficus sp. Bioresour Technol 9:3896–3899
Srivas T, Anatharaman M (2005) Cassava marketing system in India, Technical Bull ser No. 43. Central Tuber Crops research Institute, Thiruvananthapuram, pp 102
Sugumarana KR, Shobana P, Mohan Balaji P, Ponnusami V, Gowdhaman D (2014) Statistical optimization of pullulan production from Asian palmkernel and evaluation of its properties. Int J Biol Macromol 66:229–235
Sugumaranb KR, Jothi P, Ponnusami V (2014) Bioconversion of industria solid waste cassava bagasse for pullulan production in solid state fermentation. Carbohydr Polym 99:22–30
Sugumaranc KR, Sindhu RV, Sukanya S, Aiswarya N, Ponnusami V (2013) Statistical studies on high molecular weight pullulan production in solid state fermentation using jack fruit seed. Carbohydr Polym 98:854–860
Thirumavalavan K, Manikkadan TR, Dhanasekar R (2009) Pullulan production from coconut by-products by Aureobasidium pullulans. Afr J Biotechnol 8:254–258
Ueda S, Fujit K, Komatsu K, Nakashima Z (1963) Polysaccharide produced by the genus Pullularia. I. production of polysaccharide by growing cells. Appl Microbiol 11:211–215
Vijayendra SVN, Bansal D, Prasad MS, Nand K (2001) Jaggery: a novel substrate for pullulan production by Aureobasidium pullulans CFR-77. Process Biochem 37:359–364
Xiaoliu Y,Yulei W, Gongyuan W, Yingying D (2012) Media optimization for elevated molecular weight and mass production of pigment-free pullulan Carbohydr Polym 89:928–934
Waksman N, Lederkremer RMD, Cerezo AS (1977) The structure of an α-D-glucan from Cyttaria harioti Fischer. Carbohydr Res 59:505–515
Wang D, Yu X, Gongyuan W (2013) Pullulan production and physiological characteristics of Aureobasidium pullulans under acid stress. Appl Microbiol Biotechnol 97:8069–8077
Wang D, Xiaomin J, Donghai Z, Gongyuan W (2014) Efficient production of pullulan using rice hull hydrolysate by adaptive laboratory evolution of Aureobasidium pullulans Bioresource Technol 164:12–19
Wu S, Chen J, Pan S (2012) Optimization of fermentation conditions for the production of pullulan by a new strain of Aureobasidium pullulans isolated from sea mud and its characterization. Carbohydr Polym 87:1696–1700
Wu S, Jin Z, Kim JM, Tong Q, Chen H (2009) Downstream processing of pullulan from fermentation broth. Carbohydr Polym 77:750–753
Yekta G, Asli U, Ulgar G (2004) Production of Pullulan From Beet Molasses and Synthetic Medium by Aureobasidium pullulans. Turk. J Biol 28:23–30
Yekta G, Purlen U, Seval D (2011) Optimization of pullulan production from hydrolysed potato starch waste by response surface methodology. Carbohydr Polym 83:1330–1337
Youssef F, Roukas T, Biliaderis CG (1999) Pullulan production by a non-pigmented strain of Aureobasidium pullulans using batch and fed-batch culture. Process Biochem 34:355–366
Yu X, Wang Y, Wei G, Dong Y (2012) Media optimization for elevated molecular weight and mass production of pigment-free pullulan. Carbohydr Polym 89:928–934
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Mishra, B., Zamare, D., Manikanta, A. (2018). Selection and Utilization of Agro-industrial Waste for Biosynthesis and Hyper-Production of Pullulan: A Review. In: Varjani, S., Parameswaran, B., Kumar, S., Khare, S. (eds) Biosynthetic Technology and Environmental Challenges. Energy, Environment, and Sustainability. Springer, Singapore. https://doi.org/10.1007/978-981-10-7434-9_6
Download citation
DOI: https://doi.org/10.1007/978-981-10-7434-9_6
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-10-7433-2
Online ISBN: 978-981-10-7434-9
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)