Abstract
Drought is a conspicuous stress-causing deleterious effect on plant growth and productivity. In order to compensate the yield loss due to drought, efficient and sustainable strategies are required for its management. Drought stress tolerance is complex trait involving clusters of genes; hence, genetic engineering to generate drought-resistant varieties is a challenging task. In this context, the application of plant growth-promoting microbes (PGPM) to mitigate drought stress is gaining attention as an attractive and cost-effective alternative strategy. PGPM have envisaged a plethora of mechanisms to overcome drought stress in plants which encompasses ACC (1-aminocyclopropane-1-carboxylate) deaminase activity, production of exopolysaccharide (EPS) and volatile organic compounds (VOCs), osmolyte and antioxidant production, enhanced uptake of mineral nutrients, phytohormones production, and modulation. These mechanisms either individually or collectively bestow the PGPRs to combat drought stress in plants. The association of arbuscular mycorrhizal fungi (AMF) with the roots of crop plants can significantly promote water and nutrient uptake by host plants and induce tolerance to drought stress. The inoculation of PGPM in crop plants is also capable of modulating host transcriptome for induced drought tolerance. Further, efforts are needed to develop proficient microbial consortia for enhancing plant growth under drought stress. Thus, the application of PGPM/AMF represents a promising approach to increase nutrient availability and expedite the development of sustainable agriculture.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
12.1 Introduction
Plants being sessile are subjected to a wide array of environmental stresses such as salinity, drought, heavy metals, waterlogging, chilling, and high temperature. Furthermore, climate change is influencing the austerity of abiotic stresses, particularly high temperature and drought. Intense drought is mostly associated with the global climate change which is having a major impact of crop productivity (Etesami and Maheshwari 2018). It is being projected that drought will affect more than 50% of the arable lands worldwide by the year 2050 (Vurukonda et al. 2016). Among these stresses, drought is a conspicuous stress-causing deleterious effect on plant growth and productivity. This stress adversely affects many physiological and biochemical processes of plants such as phytosynthesis, respiration, transpiration, carbohydrates metabolism and nutrient uptake, translocation, and assimilation (Khan et al. 2018). In order to compensate the yield loss due to drought, efficient and sustainable strategies are required for its management. From past years, mainly two strategies are mostly focused to combat the drought stress in plants such as traditional breeding methods and genetic engineering of crop for drought-resistant (Naveed et al. 2014). The major constraint for application of these techniques is time taking, tedious, and expensive. Furthermore, the acceptance of a transgenic crop is uncertain in the market regarding the consumer response to genetically modified plant products which varies from country to country (Ullah et al. 2019a).
In addition to several ethical issues, genetic engineering of all crops is not feasible (Etesami and Maheshwari 2018). Besides, drought stress tolerance is complex trait involving clusters of genes; hence, genetic engineering to generate drought-resistant varieties is a challenging task (Nautiyal et al. 2013; Saikia et al. 2018). Another sustainable strategy to limit drought stress is soil resource management by application of mulching, crop residues, crop cover, non-crop mulch material (plastic foil, geotextile), etc. These methods tend to reduce runoff and evaporation from soil surfaces. Besides, use of crop residue as mulch can increase the soil organic matter which in turn increases the soil water storage capacity and its availability to crops during drought conditions (Bodner et al. 2015).
In this context, the use of plant growth-promoting microbes (PGPM) to mitigate drought stress is gaining attention as an attractive and cost-effective alternative strategy. There are various studies which suggest the beneficial role of plant growth-promoting rhizobacteria (PGPR), actinomycetes, and mycorrhiza in boosting plant growth and endurance toward drought stress (Gontia-Mishra et al. 2016; Saikia et al. 2018; Zade et al. 2019). This chapter summarizes the present understanding toward drought stress and their physiological and molecular responses in plants. The later section of the chapter highlights the imperative mechanisms of PGPM for mitigation of drought stress in plants. In the present chapter, we have also attempted to comprehend the perceptive use of PGPM to alleviate the drought stress in crop plant by fine-tuning the metabolic, signaling, and molecular pathways, thereby enhancing crop productivity.
12.2 Impact of Drought on Crop Plants
Similar to other abiotic stresses, drought is also multidimensional in its effects and has various deleterious impacts on plant. The plants respond to drought stress at physiological, biochemical, and molecular levels, from seed germination to maturity and senescence (Tiwari et al. 2017). Nonetheless, adaptability of plants to drought is determined by the severity and time of exposure to stress, plant species as well as the developmental stages (Kaur and Asthir 2017). Under water scarcity, plant exhibits various events to acclimatize such as morphological (reduced biomass and altered root structure), physiological (reduced photosynthesis and altered transpiration and stomatal activity), and biochemical changes (accumulation of osmolytes, increased oxidative enzyme activity) (Conesa et al. 2016).
Excessive loss of water in drought condition leads to closed stomata and controlled gas exchange and desiccation which lead to complete metabolism and cellular structure disruption; this gradually ends in interruption in enzymatic reactions (Jaleel et al. 2007). Aboveground parts of plant are more susceptible to drought stress than their counterparts. During water-deficit conditions, plants respond by shrinking leaf area, spiraling, and in some cases by shedding their leaves. Leaf size reduction is an important strategy to drought stress as it can directly influence the rate of transpiration. However, reduction in leaf size leads to marked decline in the photosynthetic activity of plants. Drought stress in plants is noted by declined leaf water potential, stomatal closure, and marked reduction in cell growth (Farooq et al. 2009). The first plant organs to feel and react to water deficiency are the roots. Under moderate drought stress, the root length is increased which is attributed to the plants need to utilize the groundwater (Forni et al. 2017), but severe drought conditions can retard root growth. Characteristics related to root traits like biomass, root length, root density, and depths of roots have been identified as the major drought avoidance traits under drought environment (Kashiwagi et al. 2006). A decrease in growth is the most obvious plant response to water stress, which results from decrease in water uptake by roots.
Plant growth and morphology are proportional to enlargement, division, and differentiation of cell. Drought is also reported to inhibit mitotic division of cells along with elongation and expansion which results in growth retardation of plants (Hussain et al. 2008). Various physiological changes occur in plants to overcome drought stress as the early response of plant to stress can make the plant to survive. The basic mechanism to show drought response is to decrease the osmotic potential of plant cells; as a result, the turgor potential gets maintained for routine metabolic processes (Levitt 1980). Drought stress leads to an imbalance between antioxidant defenses and the amount of Reactive Oxygen Species (ROS) resulting in oxidative stress. ROS are required to trigger the signaling but eventually at high concentration can cause impairment of plant organelles especially chloroplasts (Smirnoff 1993). Later on, ROS can initiate lipid peroxidation and degradation of vital proteins, lipids, and nucleic acids (Kaushal and Wani 2016).
Plants under drought stress starts synthesis of new metabolites for their proper functioning. Plants can accumulate biomolecules such as dehydrins (DHNs), heat shock proteins (HSPs), late embryogenesis abundant (LEA) proteins (Lipiec et al. 2013), osmolytes like proline, trehalose, and sugars (Ilhan et al. 2015), glycine, and betaine (Chen and Murata 2011). Changes in membrane fluidity, fatty acid, and protein composition of membranes help to maintain the cellular integrity of plants under drought stress (Bohnert et al. 1995). The osmolytes like ectoine, glycine, and betaine interplay in protein solubilization, and the uncharged solutes like mannitol, trehalose, and pinitol play an important role as scavengers of ROS (Ashraf and Foolad 2007). Plants have envisaged several enzymatic mechanisms including superoxide dismutase (SOD), catalase (CAT), ascorbate peroxidase (APX), glutathione reductase (GR) and non-enzymatic components such as cysteine, glutathione, and ascorbic acid, to aid them fight against the oxidative damage caused by ROS (Kaushal and Wani 2016).
Nitric oxide also protects plants under water-deficit condition from oxidative stress. Most of these processes are regulated through a complex network governed by abscisic acid (ABA), ion transport system, and various transcription factors. Water stress in the root tips results in release of a stress-signaling hormone ABA, which is transported from root zone to the aerial parts of plants. It is directly involved in regulation of stomatal aperture closure. Various reports argued that early interaction of drought signals is mediated by transmembrane protein like histidine kinase which functions like osmosensor (Posas et al. 1996; Urao et al. 1999). Other membrane proteins like aquaporins also involved in regulation of cell volume and turgor homeostasis. In addition, some genes are also identified which regulates the synthesis of osmolytes in the cytoplasm in order to maintain the osmotic potential at water stress condition. Other mechanism involved in sensing of drought stress is changed in the fluidity of membrane lipids (Knight and Knight 2001). Phospholipase C and D along with phosphatidyl-4,5-phosphate 5-kinase are found to be involved in this mechanism which leads to control ion and water channels (Mikami et al. 1998).
12.3 PGPM with Special Context to Drought Stress Management
Almost every parts of the plant are colonized by microbes, but the rhizosphere (soil in the proximity of roots) represents the main source of bacteria with plant-beneficial activities. The microbial community residing in the rhizosphere is comparatively different than its surroundings due to the presence of root exudates (Vejan et al. 2016). These bacteria largely utilize root exudates a source of nutrients for their growth and survival, hence termed as PGPR (Kolepper and Schroth 1978; Verma et al. 2015a, b; Yadav et al. 2015). They interact with the plant roots and influence plant growth and yield as well as enhance soil fertility. Besides PGPR, association of arbuscular mycorrhizal fungi (AMF) with the roots of crop plants can significantly promote water and nutrient uptake by host plants and induce tolerance to drought stress (Xu et al. 2018; Hashem et al. 2019; Yadav et al. 2019b, c, d). Many researchers have advocated the positive impact of PGPR and AMF to promote plant growth under drought stress; hence, it is an effective developing technology (Compant et al. 2010; Carmen et al. 2016; Wu et al. 2019; Verma et al. 2017; Yadav et al. 2018a, b). Crop plants in association with PGPRs persuade morphological and biochemical adjustments leading to increased tolerance to drought by eliciting induced systemic tolerance (IST) (Naveed et al. 2014).
It is determined that PGPR can intercede the drought stress in host plant by increasing accumulation of osmolytes (compatible solutes like proline, glycine betaine, polyamines, sugars like trehalose and polyols), improved uptake of nutrients and modulating the activities of antioxidant enzymes (Barnawal et al. 2019; Kour et al. 2019c; d). Phytohormones are known to control signaling of many abiotic and biotic stresses in plants. It is an established fact that phytohormone synthesis and signaling have a great significance in response to extreme environmental conditions (Tiwari et al. 2017; Kour et al. 2019b; Yadav et al. 2019a). Interestingly, this approach is adopted by PGPR to induce drought tolerance in host plant by regulating the level of phytohormones, like ABA, salicylic acid (SA), and ethylene, therefore directly affecting plant signaling networks and altering drought-responsive genes (Lu et al. 2018). Similarly, inoculation of AMF in host plants can accelerate plant growth and yield under drought stress by increased water and nutrient uptake, modulating stress-responsive genes and cell membrane fatty acid composition and degree of unsaturation (Xu et al. 2018; Wu et al. 2019). The use of various PGPR and AMF for alleviation of drought stress and their positive impact on host plants is presented in Tables 12.1 and 12.2.
12.4 Ways Out by Which PGPM Handle Drought Stress
PGPM have envisaged a plethora of mechanisms to overcome drought stress in plants which encompasses ACC (1-aminocyclopropane-1-carboxylate) deaminase activity, production of exopolysaccharide (EPS) and volatile organic compounds (VOCs), osmolyte production, uptake of mineral nutrients (N, P, and K), phytohormones production/modulation [auxin/indole acetic acid (IAA), cytokinins, abscisic acid (ABA), salicylic acid (SA) and jasmonic acid (JA)], and eliciting the activity antioxidant enzymes in host plants. These mechanisms either individually or collectively bestow the PGPRs to combat drought stress in plants.
12.4.1 ACC Deaminase Activity
Ethylene is a crucial plant hormone which controls a number of plant processes ranging from seed germination, fruit ripening, abscission of leaves, and plant senescence (Sapre et al. 2019). It is also produced in plant in response to several environmental stresses including drought stress (Gontia-Mishra et al. 2014). Consequently, the ethylene generated under stressful condition is often termed as “stress ethylene” (Glick 2014). The ethylene produced during stress can subsequently induce the defoliation, retarded root, and stem growth along with the expression of genes leading to plant senescence, leading to inferior crop performance (Vejan et al. 2016). Interestingly, it is noted that ACC works as precursor for ethylene biosynthesis (Shaharoona et al. 2006). Among the different suggested strategies for improvement in plant growth under drought stress, the most plausible one is alteration in the endogenous levels of ethylene caused by the PGPR (Kumar et al. 2019a, b).
Furthermore, many PGPR possessing the enzyme ACC deaminase catalyze the conversion of ACC to ammonia and α-ketobutyrate, which indirectly decrease the ethylene concentration in plants under drought stress (Glick et al. 1998). By facilitating the development of longer roots, these PGPR may enhance the survival of seedlings, which help in combating the effect of stress ethylene. The root elongation plants under drought stress can allow a better access to water and uptake of nutrients. Ethylene is also known to compromise the nodule formation and nitrogen fixation in legume (Sapre et al. 2019). Rhizobium with ACC deaminase activity can diminish the deleterious effect of ethylene under drought stress by increasing the nodulation and nitrogen fixation in its symbiotic legume partner (Belimov et al. 2009). In this regard, numerous researchers have documented the application of ACC deaminase-producing PGPR in ameliorating drought stress in crop plant such as chickpea (Tiwari et al. 2016), mung bean (Sarma and Saikia 2014), wheat (Gontia-Mishra et al. 2016; Barnawal et al. 2017), rice (Tiwari et al. 2017), foxtail millet (Niu et al. 2018), and other tropical crop plants (Kumar et al. 2019a, b; Kour et al. 2019a; Yadav et al. 2017a; b; Yadav and Yadav 2018).
12.4.2 EPS Production
PGPR have the unique ability to produce exopolysaccharide (EPS)/extracellular polymeric substances. The biofilm formation and EPS production by PGPR are important mechanisms to tolerate drought stress in the environment. The EPS has multifarious function in bacterial cells ranging from quorum-sensing signals, development, survival, and host colonization (Nocelli et al. 2016). The EPS largely constitutes of high-molecular-weight macromolecules like polysaccharide along with smaller proportions of protein, lipids, and uronic acid (Naseem et al. 2018). The EPS-producing PGPR can better clamp and colonize the root surface under adverse conditions (Ali et al. 2014).
It can protect PGPR and its host plant under drought stress by enhancing water retention than the surrounding environment (Hepper 1975; Vurukonda et al. 2016). The EPS secreted by PGPR into the soil can be absorbed by soil particle due to their different anionic functional groups (e.g., sulfhydryl, carboxyl, hydroxyl, sulfonate, amine, and amide) and increase the water-holding capacity of soil and improve physicochemical properties of soil under prolong desiccation conditions (Sandhya et al. 2009). Hence, plants inoculated with EPS-producing PGPR can maintain the higher water potential, boost root-adhering soil/root tissue ratio, and accelerate the uptake of nutrients by plant, thereby enhancing plant growth and yield under drought stress (Selvakumar et al. 2012; Rolli et al. 2014; Kaushal 2019). Many researchers have suggested the use of EPS-producing rhizobacteria in alleviating drought stress in important crop plants such as maize (Vardharajula et al. 2011), sunflower (Sandhya et al. 2009), wheat (Timmusk et al. 2014), and foxtail millet (Niu et al. 2018).
12.4.3 Production of VOCs
Unlike plants, soil bacteria produce a range of volatile compounds, which have specific function in their life cycles as well as interplay with other microbes and plants (Sharifi and Ryu 2018). The bacterial VOCs are chemically characterized as alkenes, ketones, and alcohols. PGPR can stimulate plant growth by synthesizing and releasing volatile compounds, which is now known as an essential mechanism of plant–microorganism interactions (Froni et al. 2017). The role of these VOCs is largely associated as activator against plant pathogens, leading to induced systemic resistance in plants (Ruzzi and Aroca 2015). Remarkably, it is noted that besides their role in biotic stress tolerance, these compounds can actively alleviate several abiotic stresses including drought (Timmusk et al. 2014). The VOCs produced by PGPRs can promote plant growth by increasing photosynthesis, carbon assimilation, enhancing mineral uptake, altering root structure, and intensive phytohormone signaling under abiotic stress conditions (Sharifi and Ryu 2018).
It was demonstrated that a PGPR Pseudomonas chlororaphis releases characterized as 2R, 3R-butanediol, can induce drought tolerance in Arabidopsis thaliana (Cho et al. 2008). Moreover, few studies suggest the role of VOCs (produced by PGPR) in modulation of the transcript levels in plants, resulting in enhanced biosynthesis of choline and glycine betaine which in turn to shielded A. thaliana plants from drought stress (Cho et al. 2008; Zhang et al. 2010). It was noted that VOCs produced by soil bacteria such as acetic acid can stimulate the formation of biofilms/EPS, which can indirectly influence drought stress in plants (Chen et al. 2015). Another study of bacterial inoculation with Bacillus thuringiensis in wheat seedlings under drought stress markedly decreased the stress-induced volatile compounds emitted by plants and increased plant biomass and photosynthesis (Timmusk et al. 2014). VOCs producing PGPR are potential for application as bio-stimulants to improve plant health under drought stress. The mechanism of PGPR-induced VOCs in ameliorating drought stress in crop plants is limited and requires to be explored extensively.
12.4.4 Phytohormones Production
The production of phytohormones in plants is essential because of their physiological effects on its growth. The phytohormones such as auxins, cytokinins, gibberellins and ethylene, and abscisic acid (ABA) have a particular function in the regulation of plant growth and development (Vurukonda et al. 2016). PGPR have been widely known to produce these phytohormones which can help in promoting plant growth under stressed conditions by stimulating cell growth and division (Kaushal 2019). One of the important aspects of the bacterial–plant interaction that has received worldwide attention is the bacterial production of IAA/auxins. Production of IAA, a plant hormone that does not any apparent function as a hormone in bacterial cells, may have evolved in bacteria because of its significance in the bacterium–plant relationship (Patten and Glick 2002). A positive correlation is noted between the in vitro production of auxins by PGPR strains and their growth promotion effects (Jha et al. 2012). The auxin-producing PGPR can influence root proliferation and formation of lateral and adventitious roots, which results in an increased mineral and water uptake by the plants rotoscoping them against drought stress (Gontia-Mishra et al. 2016). Besides, some of the PGPR have known to modulate the expression of auxin-responsive genes in host–plant roots (Lakshmanan et al. 2013) resulting in stress tolerance in plants. Several authors have suggested the IAA production by PGPR as a mechanism to drought tolerance enhancement in crop plants such as maize, wheat, mung bean, etc. (Naveed et al. 2014; Sarma and Saikia 2014; García et al. 2017).
Gibberellins have a crucial function in plant growth such as stem elongation, germination, flowering, and senescence (Kaushal 2019). In contrast, cytokinins play a vital role in regulation of cell division and nutrient allocation, and maintain photosynthetic activity under drought stress (Ullah et al. 2019b). The application of cytokinin-producing PGPR, Bacillus subtilis in Platycladus orientalis, conferred drought stress tolerance by increasing shoot growth (Liu et al. 2013). Similarly, maize and soybean plants inoculated with gibberellin-producing PGPRs (Pseudomonas putida and Azospirillum lipoferum) registered improved plant growth under drought stress (Cohen et al. 2009; Kang et al. 2014). Consequently, there are evidences from many studies which advocate that PGPM have the ability to positively alter phytohormone levels of plant, leading to drought stress tolerance (Saakre et al. 2017; Ali et al. 2018).
12.4.5 Osmolytes Production and Alteration in Antioxidant Enzyme Activities
Water-deficit conditions result in altered osmotic balance in plants; hence, the water-absorbing capacity of plants get reduced and plant tries to change various physiological and biochemical processes for adaptation under stressed environment. Under these conditions, plants tend to produce a wide range of osmolytes or commonly known as osmoprotectants. Some cellular events like protein and membrane stabilization are supported by osmoprotectants (Nahar et al. 2016). Due to their diverse chemical properties, osmolytes also protect plant cells from oxidative stress by attacking on ROS (Zhu 2002). There are several osmoregulators like proteins, sugars, and free amino acids reported to play a key role in balancing osmotic pressure in plant cells (Hasegawa et al. 2000). It is reported in various studies that microbes residing in soil also produce and secrete osmolytes when encounter to drought stress. These osmolytes function as produced by plants. Maize inoculated with Pseudomonas fluorescence growing under drought has potential for accumulating proline and resulted in increased water content and plant biomass (Ansary et al. 2012). Soil microbes also reported to alter the amount of osmoprotectants and antioxidant enzyme in plants (Kaushal 2019).
Plants in response to drought also generate various types of ROS oxidative damage by reacting with importing biomolecules like protein and lipids of cell. To protect from this oxidative damage, cell has developed antioxidant defense system which includes enzymatic as well as non-enzymatic pathway (Miller et al. 2010). Inoculation of plants with beneficial microbes suppresses the activity of antioxidant enzyme activity to alleviate the drought stress. Various species of Pseudomonas and Bacillus inoculated with maize are reported to reduce the activity of antioxidant enzymes when compared to un-inoculated plants under drought (Sandhya et al. 2010; Vardharajula et al. 2011).
12.4.6 Nutrient Availability
Water deficit can reduce the uptake of nutrients in the soil due to poor soil structure. PGPR and AMF have the capability to increase the accessibility of nutrient in the rhizosphere either by fixing nutrients (N2) or by solubilizing insoluble minerals such as P, K, and Zn into the soluble form (Vejan et al. 2016). The explanation for enhanced nutrient status in AMF inoculation in plant has increased absorption surface of extraradical hyphae for extensive acquisition of nutrients from soil. The extraradical hyphae of AMF have a deep network into the soil readily absorption of nutrients which in turn is transported to arbuscules in cortical cells and are finally released into the apoplast to ameliorate nutrient deficiency caused by stress (Zhao et al. 2015; Kaushal 2019). Several reports suggest that PGPR inoculation can improve uptake of nutrients especially P under drought stress (Sandhya et al. 2010; Timmusk et al. 2014). In addition, there are evidences of increased mobility of nutrients (P, N, K, Ca, and Zn) in plants inoculated with AMF during drought stress (Gholamhoseini et al. 2013; Zhao et al. 2015; Abdel-Salam et al. 2018).
12.5 Modulation of Host Transcriptome by PGPR Inoculation
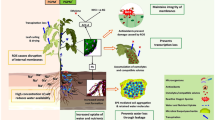
The PGPRs have been efficiently utilized in crops to alleviate disease stress. Nevertheless, they can also be used as potential targets for inducing drought tolerance in crop species. Physiological studies in few crop species suggest an efficient application of PGPRs to reduce drought stress (Khan et al. 2018; Niu et al. 2018) (Fig. 12.1). Drought stress is a complex trait which influences various cross-linked signaling between biotic and abiotic stresses. Hence, the beneficial effects of PGPRs in biotic stress can indirectly induce tolerance to drought stress. This mode of genetic improvement comprises understanding the PGPR-induced biochemical differential regulation. These biochemical changes are governed by differential expression of genes involved in induced systemic response (ISR), which are ultimately triggered through salicylic acid (SA)-signaling pathway (Zhang et al. 2002). The PGPR inoculation greatly modifies the transcriptome of the plant species, regulating expression of several genes (Rekha et al. 2018). Microarrays and RNA-seq studies have been employed in certain crop species to understand the molecular mechanism of PGPR-induced drought tolerance.
PGPRs often colonize the root surface and may induce production of several phytochemicals that regulate phytohormone signaling like auxins and ABA (Srivastava et al. 2012). Systemic acquired resistance (SAR) is strongly regulated by phytohormones and their in planta levels. The inoculation of PGPR Pseudomonas sp. in rice plants induced the expression of LEA (late embryogenesis abundant) genes, which encodes IAA amido synthetases resulting in drought stress tolerance (Yasmin et al. 2017). Similarly, the inoculation of A. thaliana with PGPR Paenibacillus polymyxa induced drought tolerance through enhanced transcription of Early Responsive to Dehydration 15 (ERD15) gene (Timmusk and Wagner 1999). The drought alleviation by PGPR is mediated primarily through differential regulation of ABA-responsive pathway. The ABA-responsive signaling pathway transcription factor genes bZIP1, COC1, and Hsp20 proteins were overexpressed in susceptible rice cultivar upon PGPR Pseudomonas fluorescens inoculation, conferring drought tolerance (Saakre et al. 2017).
DHNs are a class of proteins of group 2 LEA proteins and proportionately related to their active accumulation during water stress tolerance. Microarray studies of barley seedlings overexpressing DHNs genes were drought-tolerant (Rodriguez et al. 2005). Plants expressing drought-tolerant CaDHN gene were highly expressed in pepper plant inoculated with PGPR Bacillus licheniformis (Lim and Kim 2013). DHN genes are interacting members of the SA-induced SAR response (Jing et al. 2016), commonly observed in PGPR-associated stress alleviation. The inoculation of water-stressed A. thaliana roots with Pseudomonas chlororaphis O6 induced an overexpression of LEA and dehydrin genes to over 100-folds (Cho et al. 2013). Trehalose is a nonreducing disaccharide, which is actively synthesized in bacteroids of Rhizobium sp. (Streeter 1985). It is an osmoprotectant and plays an essential role as a signaling molecule (Paul et al. 2008) during water stress management. The transformed Rhizobium etli and Azospirillum brasilense mutant with an overexpressing trehalose-6-phosphate synthase gene inoculated to Phaseolus vulgaris and maize, respectively, resulted in the enhanced expression of drought tolerance genes (Suárez et al. 2008; Rodríguez-Salazar et al. 2009).
To identify the transcriptional regulation of plants with drought stress in the presence of PGPRs, few transcriptomic studies have been undertaken. The PGPR inoculation improves drought stress tolerance by repressing the enhanced expression of abiotic stress response genes, viz., ABA and ethylene. The transcriptome study of sugarcane plants colonizing Gluconacetobacter diazotrophicus identified a reverse regulation of drought stress genes to that of stressed non-inoculated roots. The DREB1A/CBF3, DREB1B/CBF1, and NCED3 homologs were down-regulated in water-stressed plants treated with G. diazotrophicus. Although the auxin metabolism and ABA pathway were equally enriched in both the inoculated and non-inoculated plants, cytokinin hormone pathway up-regulation was observed only in the inoculated plants. However, the auxin, ABA, and ethylene-mediated stress-responsive signaling were mainly down-regulated (Vargas et al. 2014). Similar results have been demonstrated through transcriptome analysis of maize plants inoculated with Pseudomonas putida strain FBKV2 displayed drought stress alleviation, by Ali et al. (2018).
The expressions of ABA and ethylene signaling pathway genes were down-regulated, including down-regulation of bZIP transcription factor (TFs), and 1-aminocyclopropane-1-carboxylate synthase2 and ethylene-responsive TFs, respectively. The transcriptome of A. thaliana treated with PGPR P. chlororaphis O6 identified remarkable up-regulation of calmodulin and calcium-binding proteins. These genes play important role in cell-to-cell communication. The stress-responsive down-regulated genes in PGPR inoculation also include class of MYB and AP2 domain transcription factors (Cho et al. 2013). This clearly presents that plant growth-promoting bacteria reduce the drought stress by reducing the expression of stress-induced molecules, keeping optimum environment to plants. It is evident from several studies that PGPRs prime the stress-responsive pathway and following subjection of plants to drought stress reduces the production or biosynthesis of stress molecules.
12.6 Concluding Remarks and Future Prospects
Agricultural productivity is largely dependent on climatic conditions. Climate change is expected to reduce water accessibility for agriculture in coming years. Drought has the noxious effects on growth and development of plant. Hence, it is the need of the hour to search for the effectual solution to overcome the problem of drought stress in plants. Moreover, drought is a complex trait, so developing transgenic plants resistant to drought stress is also a challenging task. Under such stressful conditions, the interaction of plant and beneficial microorganisms is of great importance. The application of drought-tolerant PGPM has gained abundant attention as an alternative and eco-friendly option to mitigate drought stress in crop plants. Another effective option to combat drought stress in plant is the exogenous application of PGPM in combination with either the plant growth regulators (SA, JA, Trinexapacethyl, and ABA), polyamine-like putrescine, biochar (organic carbon), silicon nanoparticles, or seaweed extracts (Ali et al. 2017; Khan et al. 2019; Hashem et al. 2019).
In the current scenario, research must be concentrated to increase the number and diversity of effective and competitive drought-tolerant PGPM from drought-stricken agricultural ecosystems. The drought-tolerant PGPM could be useful to design new bio-inoculants/biofertilizers, especially for arid regions. Additionally, the performance of potential PGPR strains should be essentially assessed under field conditions, as plants usually face cyclic drought conditions rather than continuous drought. Further efforts are needed to develop proficient microbial consortia for enhancing plant growth under drought stress. Regardless of several findings, on the adaptation of plants under drought stress and their association with PGPM for mitigation of stress, substantial efforts are required to explore the underlying molecular mechanisms of interplay between plant and PGPM in soil to hasten the process of stress amelioration in crops. Thus, it could be concluded that the use of PGPM represents a promising approach to increase nutrient availability and expedite the development of sustainable agriculture under drought stress.
References
Abdel-Salam E, Alatar A, El-Sheikh MA (2018) Inoculation with arbuscular mycorrhizal fungi alleviates harmful effects of drought stress on damask rose. Saudi J Biol Sci 25:1772–1780
Ali SkZ, Sandhya V, Rao LV (2014) Isolation and characterization of drought-tolerant ACC deaminase and exopolysaccharide-producing fluorescent Pseudomonas sp. Ann Microbiol 64:493–502
Ali F, Bano A, Fazal A (2017) Recent methods of drought stress tolerance in plants. Plant Growth Regul 82:363–375
Ali SkZ, Vardharajula S, Vurukonda SS (2018) Transcriptomic profiling of maize (Zea mays L.) seedlings in response to Pseudomonas putida stain FBKV2 inoculation under drought stress. Ann Microbiol 68:331–349
Ansary MH, Rahmani HA, Ardakani MR, Paknejad F, Habibi D, Mafakheri S (2012) Effect of Pseudomonas fluorescent on proline and phytohormonal status of maize (Zea mays L.) under water deficit stress. Ann Biol Res 3:1054–1062
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59:206–216
Barnawal D, Bharti N, Pandey SS, Pandey A, Chanotiya CS, Kalra A (2017) Plant growth-promoting rhizobacteria enhance wheat salt and drought stress tolerance by altering endogenous phytohormone levels and TaCTR1/TaDREB2 expression. Physiol Plantarum 161:502–514
Barnawal D, Singh R, Singh RP (2019) Role of plant growth promoting rhizobacteria in drought tolerance: regulating growth hormones and osmolytes. In: Singh AK, Kumar A, Singh PK (eds) PGPR amelioration in sustainable agriculture. Woodhead Publishing, pp 107–128
Belimov AA, Dodd IC, Hontzeas N, Theobald JC, Safronova VI, Davies WJ (2009) Rhizosphere bacteria containing 1-aminocyclopropane-1-carboxylate deaminase increase yield of plants grown in drying soil via both local and systemic hormone signaling. New Phytol 181:413–423
Bodner G, Nakhforoosh A, Kaul HP (2015) Management of crop water under drought: a review. Agron Sustain Dev 35:401–442
Bohnert HJ, Nelson DE, Jensen RG (1995) Adaptations to environmental stresses. Plant Cell 7:1099–1111
Boyer LR, Brain P, Xu X, Jeffries P (2015) Inoculation of drought-stressed strawberry with a mixed inoculum of two arbuscular mycorrhizal fungi: effects on population dynamics of fungal species in roots and consequential plant tolerance to water deficiency. Mycorrhiza 25:215–227
Carmen CA, Patricia P, Rubén B, Victoria SM (2016) Plant-rhizobacteria interaction and drought stress tolerance in plants. In: Hossain MA, Wani SH, Bhattacharjee S, Burritt DJ, Tran LSP (eds), Drought stress tolerance in plants, vol 1. Springer, Cham, pp 287–308
Chandra D, Srivastava R, Gupta VV, Franco CM, Sharma AK (2019) Evaluation of ACC-deaminase-producing rhizobacteria to alleviate water-stress impacts in wheat (Triticumaestivum L.) plants. Can J Microbiol 65:387–403
Chen TH, Murata N (2011) Glycine betaine protects plants against abiotic stress: mechanisms and biotechnological applications. Plant Cell Environ 34:1–20
Chen Y, Gozzi K, Yan F, Chai Y (2015) Acetic acid acts as a volatile signal to stimulate bacterial biofilm formation. MBio 6:e00392
Cho SM, Kang BR, Han SH, Anderson AJ, Park JY, Lee YH et al (2008) 2R, 3R-butanediol, a bacterial volatile produced by Pseudomonas chlororaphis O6, is involved in induction of systemic tolerance to drought in Arabidopsis thaliana. Mol Plant Microb Interact 21:1067–1075
Cho SM, Kang BR, Kim YC (2013) Transcriptome analysis of induced systemic drought tolerance elicited by Pseudomonas chlororaphis O6 in Arabidopsis thaliana. Plant Pathol J 29:209–220
Cohen AC, Travaglia CN, Bottini R, Piccoli PN (2009) Participation of abscisic acid and gibberellins produced by endophytic Azospirillum in the alleviation of drought effects in maize. Botanique 87:455–462
Compant E, van der Heijden MGA, Sessitsch A (2010) Climate change effects on beneficial-plant microorganism interactions. FEMS Microbiol Ecol 73:197–214
Conesa MR, Rosa JM, Domingo R, Banon S, Perez-Pastor A (2016) Changes induced by water stress on water relations, stomatal behaviour and morphology of table grapes (cv. Crimson seedless) grown in pots. SciHort 202:9–16
Etesami H, Maheshwari DK (2018) Use of plant growth promoting rhizobacteria (PGPRs) with multiple plant growth promoting traits in stress agriculture: action mechanisms and future prospects. Ecotoxicol Environ Saf 156:225–246
Farooq M, Wahid A, Kobayashi N, Fujita D, Basra SMA (2009) Plant drought stress: effects, mechanisms and management. Agron Sustain Dev 29:185–212
Forni C, Duca D, Glick BR (2017) Mechanisms of plant response to salt and drought stress and their alteration by rhizobacteria. Plant Soil 410:335–356
Gagné-Bourque F, Mayer BF, Charron J-B, Vali H, Bertrand A, Jabaji S (2015) Accelerated growth rate and increased drought stress resilience of the model grass Brachypodium distachyon colonized by Bacillus subtilis B26. PLoS ONE 10:e0130456
García JE, Maroniche G, Creus C, Suárez-Rodríguez R, Ramirez-Trujillo JA, Groppa MD (2017) In vitro PGPR properties and osmotic tolerance of different Azospirillum native strains and their effects on growth of maize under drought stress. Microbiol Res 202:21–29
Gholamhoseini M, Ghalavand A, Dolatabadian A, Jamshidi E, Khodaei-Joghan A (2013) Effects of arbuscular mycorrhizal inoculation on growth, yield, nutrient uptake and irrigation water productivity of sunflowers grown under drought stress. Agric Water Manag 117:106–114
Ghosh D, Gupta A, Mohapatra S (2019) A comparative analysis of exopolysaccharide and phytohormone secretions by four drought-tolerant rhizobacterial strains and their impact on osmotic-stress mitigation in Arabidopsis thaliana. World J Microbiol Biotechnol 35:90
Glick BR (2014) Bacteria with ACC deaminase can promote plant growth and help to feed the world. Microbiol Res 169:30–39
Glick BR, Penrose DM, Li J (1998) A model for the lowering of plant ethylene concentrations by plant growth promoting bacteria. J Theor Biol 190:63–68
Gontia-Mishra I, Sapre S, Kachare S, Tiwari S (2017) Molecular diversity of 1-aminocyclopropane-1-carboxylate (ACC) deaminase producing PGPR from wheat (Triticum aestivum L.) rhizosphere. Plant Soil 414:213–227
Gontia-Mishra I, Sapre S, Sharma A, Tiwari S (2016) Amelioration of drought tolerance in wheat by the interaction of plant growth-promoting rhizobacteria. Plant Biol 18:992–1000
Gontia-Mishra I, Sasidharan S, Tiwari S (2014) Recent developments in use of 1-amino cyclopropane-1-carboxylate (ACC) deaminase for conferring tolerance to biotic and abiotic stress. Biotechnol Lett 36:889–898
Gusain YS, Singh US, Sharma AK (2015) Bacterial mediated amelioration of drought stress in drought tolerant and susceptible cultivars of rice (Oryza sativa L.). Afr J Biotechnol 14:764–773
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular responses to high salinity. Ann Rev Plant Physiol Plant Mol Biol 51:463–499
Hashem A, Kumar A, Al-Dbass AM et al (2019) Arbuscular mycorrhizal fungi and biochar improves drought tolerance in chickpea. Saudi J Biol Sci 26:614–624
Hepper CM (1975) Extracellular polysaccharides of soil bacteria. In: Walker N (ed) Soil microbiology, a critical review. Wiley, New York, pp 93–111
Hussain M, Malik MA, Farooq M, Ashraf MY, Cheema MA (2008) Improving drought tolerance by exogenous application of glycine betaine and salicylic acid in sunflower. J Agron Crop Sci 194:193–199
Ilhan S, Ozdemir F, Bor M (2015) Contribution of trehalose biosynthetic pathway to drought stress tolerance of Capparis ovata Desf. Plant Biol 17:402–407
Jaleel CA, Manivannan P, Sankar B, Kishorekumar A, Gopi R, Somasundaram R, Panneerselvam R (2007) Induction of drought stress tolerance by ketoconazole in Catharanthus roseus is mediated by enhanced antioxidant potentials and secondary metabolite accumulation. Colloids Surf, B 60:201–206
Jha B, Gontia I, Hartmann A (2012) The roots of the halophyte Salicornia brachiata are a source of new halotolerant diazotrophic bacteria with plant growth-promoting potential. Plant Soil 356(1–2):265–277
Jing H, Li C, Ma F, Ma JH, Khan A, Wang X, Zhao LY, Gong ZH, Chen RG (2016) Genome-wide identification, expression diversication of dehydrin gene family and characterization of CaDHN3 in pepper (Capsicum annuum L.). PloS ONE. 11:e0161073
Kang SM, Radhakrishnan R, Khan AL, Kim MJ, Park JM, Kim BR, Shin D-H, Lee I-J (2014) Gibberellin secreting rhizobacterium Pseudomonas putida H-2-3 modulates the hormonal and stress physiology of soybean to improve the plant growth under saline and drought conditions. Plant Physiol Biochem 84:115–124
Kashiwagi J, Krishnamurthy L, Crouch JH, Serraj R (2006) Variability of root length density and its contributions to seed yield in chickpea (Cicer arietinum L.) under terminal drought stress. Field Crops Res 95:171–181
Kaur G, Asthira B (2017) Molecular responses to drought stress in plants. Biol Plantarum 61:201–209
Kaushal M (2019) Microbes in cahoots with plants: MIST to hit the jackpot of agricultural productivity during drought. Int J Mol Sci 20:1769
Kaushal M, Wani SP (2016) Plant-growth-promoting rhizobacteria: drought stress alleviators to ameliorate crop production in drylands. Ann Microbiol 66:35–42
Khan N, Bano A, Rahman MA, Guo J, Kang Z, Babar MA (2019) Comparative physiological and metabolic analysis reveals a complex mechanism involved in drought tolerance in chickpea (Cicer arietinum L.) induced by PGPR and PGRs. Sci Rep 9:2097
Khan N, Zandi P, Ali S, Mehmood A, Adnan Shahid M (2018) Impact of salicylic acid and PGPR on the drought tolerance and phytoremediation potential of Helianthus annus. Front Microbiol 9:2507
Kloepper JW, Schroth M (1978) Plant growth promoting rhizobacteria on radishes. In: Proceedings of the 4th international conference on plant pathogenic bacteria, Angers, vol 2, 879–882
Knight H, Knight M (2001) Abiotic stress signalling pathways: specificity and cross-talk. Trends Plant Sci 6:262–267
Kour D, Rana KL, Kumar A, Rastegari AA, Yadav N, Yadav AN, Gupta VK (2019a) Extremophiles for hydrolytic enzymes productions: biodiversity and potential biotechnological applications. In: Molina G, Gupta VK, Singh BN, Gathergood N (eds) Bioprocessing for biomolecules production. Wiley, USA, pp 321–372
Kour D, Rana KL, Yadav AN, Yadav N, Kumar V, Kumar A, Sayyed RZ, Hesham AE-L, Dhaliwal HS, Saxena AK (2019b) Drought-tolerant phosphorus-solubilizing microbes: biodiversity and biotechnological applications for alleviation of drought stress in plants. In: Sayyed RZ, Arora NK, Reddy MS (eds) Plant growth promoting rhizobacteria for sustainable stress management, Volume 1: Rhizobacteria in Abiotic Stress Management. Springer, Singapore, pp 255–308. https://doi.org/10.1007/978-981-13-6536-2_13
Kour D, Rana KL, Yadav N, Yadav AN, Kumar A, Meena VS, Singh B, Chauhan VS, Dhaliwal HS, Saxena AK (2019c) Rhizospheric microbiomes: biodiversity, mechanisms of plant growth promotion, and biotechnological applications for sustainable agriculture. In: Kumar A, Meena VS (eds) Plant growth promoting rhizobacteria for agricultural sustainability: from theory to practices. Springer, Singapore, pp 19–65. https://doi.org/10.1007/978-981-13-7553-8_2
Kour D, Rana KL, Yadav N, Yadav AN, Singh J, Rastegari AA, Saxena AK (2019d) Agriculturally and industrially important fungi: current developments and potential biotechnological applications. In: Yadav AN, Singh S, Mishra S, Gupta A (eds) Recent advancement in white biotechnology through fungi, Volume 2: Perspective for Value-Added Products and Environments. Springer International Publishing, Cham, pp 1–64. doi:https://doi.org/10.1007/978-3-030-14846-1_1
Kumar A, Patel JS, Meena VS, Ramteke PW (2019a) Plant growth-promoting rhizobacteria: strategies to improve abiotic stresses under sustainable agriculture. J Plant Nutr 42:1402–1415
Kumar M, Saxena R, Rai PK, Tomar RS, Yadav N, Rana KL, Kour D, Yadav AN (2019) Genetic diversity of methylotrophic yeast and their impact on environments. In: Yadav AN, Singh S, Mishra S, Gupta A (eds) Recent advancement in white biotechnology through fungi: Volume 3: Perspective for Sustainable Environments. Springer International Publishing, Cham, pp 53–71. https://doi.org/10.1007/978-3-030-25506-0_3
Lakshmanan V, Castaneda R, Rudrappa T, Bais HP (2013) Root transcriptome analysis of Arabidopsis thaliana exposed to beneficial Bacillus subtilis FB17 rhizobacteria revealed genes for bacterial recruitment and plant defense independent of malate efflux. Planta 238:657–668
Levitt J (1980) Response of plants to environmental stress, vol 2. Academic Press, New York
Li H, Guo Q, Jing Y et al (2019a) Application of Streptomyces pactum Act12 enhances drought resistance in wheat. J Plant Growth Regul. https://doi.org/10.1007/s00344-019-09968-z
Li J, Meng B, Chai H, Yang X, Song W, Li S, Lu A, Zhang T, Sun W (2019b) Arbuscularmycorrhizal fungi alleviate drought stress in C3 (Leymus chinensis) and C4 (Hemarthria altissima) grasses via altering antioxidant enzyme activities and photosynthesis. Front Plant Sci 10:499
Li Y, Shi H, Zhang H, Chen S (2019c) Amelioration of drought effects in wheat and cucumber by the combined application of super absorbent polymer and potential biofertilizer. Peer J 7:e6073
Lim JH, Kim SD (2013) Induction of drought stress resistance by multi-functional PGPR Bacillus licheniformis K11 in pepper. Plant Pathol J 29:201–208
Lipiec J, Doussan C, Nosalewicz A, Kondracka K (2013) Effect of drought and heat stresses on plant growth and yield: a review. Int Agrophys 27:463–477
Liu CY, Zhang F, Zhang DJ, Srivastava AK, Wu QS, Zou YN (2018) Mycorrhiza stimulates root-hair growth and IAA synthesis and transport in trifoliate orange under drought stress. Sci Rep 8:1978
Liu F, Xing S, Ma H, Du Z, Ma B (2013) Cytokinin producing, plant growth promoting rhizobacteria that confer resistance to drought stress in Platycladus orientalis container seedlings. Appl Microbiol Biotechnol 97:9155–9164
Lu X, Liu S-F, Yue L, Zhao X, Zhang Y-B, Xie Z-K, Wang R-Y (2018) Epsc involved in the encoding of exopolysaccharides produced by Bacillus amyloliquefaciens FZB42 act to boost the drought tolerance of Arabidopsis thaliana. Int J Mol Sci 19:3795
Mikami K, Katagiri T, Iuchi S, Yamaguchi-Shinozaki K, Shinozaki K (1998) A gene encoding phosphatidylinositol-4-phosphate-5- kinase is induced by water stress and abscisic acid in Arabidopsis thaliana. Plant J 15:563–568
Miller G, Susuki N, Ciftci-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ 33:45–467
Nahar K, Hasanuzzaman M, Fujita M (2016) Roles of osmolytes in plant adaptation to drought and salinity. Osmolytes and plants acclimation to changing environment: emerging omics technologies. Springer, New Delhi, pp 37–68
Naseem H, Ahsan M, Shahid MA, Khan N (2018) Exopolysaccharides producing rhizobacteria and their role in plant growth and drought tolerance. J Basic Microbiol 58:1009–1022
Nautiyal CS, Srivastava S, Chauhan PS, Seem K, Mishra A, Sopory SK (2013) Plant growth-promoting bacteria Bacillus amyloliquefaciens NBRISN13 modulates gene expression profile of leaf and rhizosphere community in rice during salt stress. Plant Physiol Biochem 66:1–9
Naveed M, Hussain MB, Zahir ZA, Mitter B, Sessitsch A (2014) Drought stress amelioration in wheat through inoculation with Burkholderia phytofirmans strain PsJN. Plant Growth Regul 73:121–131
Niu X, Song L, Xiao Y, Ge W (2018) Drought-tolerant plant growth-promoting rhizobacteria associated with foxtail millet in a semi-arid agroecosystem and their potential in alleviating drought stress. Front Microbiol 8:2580
Nocelli N, Bogino PC, Banchio E, Giordano W (2016) Roles of extracellular polysaccharides and biofilm formation in heavy metal resistance of rhizobia. Materials 9:418
Patten CL, Glick BR (2002) Role of Pseudomonas putida indoleacetic acid in development of host plant root system. Appl Environ Microbiol 48:3795–3801
Paul MJ, Primavesi LF, Jhurreea D, Zhang Y (2008) Trehalose metabolism and signaling. Ann Rev Plant Biol 59:417–441
Posas F, Wurgler-Murphy SM, Maeda T, Witten EA, Thai TC, Saito H (1996) Yeast HOG1 MAP kinase cascade is regulated by a multistep phosphorelay mechanism in the SLN1-YPD1-SSK1 “two-component” osmosensor. Cell 86:865–875
Quiroga G, Erice G, Aroca R, Chaumont F, Ruiz-Lozano JM (2017) Enhanced drought stress tolerance by the arbuscular mycorrhizal symbiosis in a drought-sensitive maize cultivar is related to a broader and differential regulation of host plant aquaporins than in a drought-tolerant cultivar. Front Plant Sci 8:1056
Quiroga G, Erice G, Aroca R, Zamarreño AM, García-Mina JM, Ruiz-Lozano JM (2018) Arbuscular mycorrhizal symbiosis and salicylic acid regulate aquaporins and root hydraulic properties in maize plants subjected to drought. Agric Water Manag 202:271–284
Rekha K, Kumar RM, Ilango K, Rex A, Usha B (2018) Transcriptome profiling of rice roots in early response to Bacillus subtilis (RR4) colonization. Botany 96:749–765
Rodriguez EM, Svensson JT, Malatrasi M, Choi DW, Close TJ (2005) Barley Dhn13 encodes a KS-type dehydrin with constitutive and stress responsive expression. Theo Appl Genet 110:852–858
Rodríguez-Salazar J, Suárez R, Caballero-Mellado J, Iturriaga G (2009) Trehalose accumulation in Azospirillum brasilense improves drought tolerance and biomass in maize plants. FEMS Microbiol Lett 296:52–59
Rolli E, Marasco R, Vigani G, Ettoumi B, Mapelli F, Deangelis ML, Gandolfi C, Casati E, Previtali F, Gerbino R et al (2014) Improved plant resistance to drought is promoted by the root-associated microbiome as a water stress-dependent trait. Environ Microbiol 17:316–331
Ruzzi M, Aroca R (2015) Plant growth-promoting rhizobacteria act as biostimulants in horticulture. Sci Hort 196:124–134
Saakre M, Baburao TM, Salim AP, Ffancies RM, Achuthan VP, Thomas G, Sivarajan SR (2017) Identification and characterization of genes responsible for drought tolerance in rice mediated by Pseudomonas fluorescens. Rice Sci 24:291–298
Saikia J, Sarma RK, Dhandia R, Yadav A, Bharali R, Gupta VK, Saikia R (2018) Alleviation of drought stress in pulse crops with ACC deaminase producing rhizobacteria isolated from acidic soil of Northeast India. Sci Rep 8(1)
Saleem AR, Brunetti C, Khalid A, Della Rocca G, Raio A, Emiliani G et al (2018) Drought response of Mucuna pruriens (L.) DC. inoculated with ACC deaminase and IAA producing rhizobacteria. PLoS ONE 13:e0191218
Sanchez-Romera B, Ruiz-Lozano JM, Zamarreno AM, Garcia-Mina JM, Aroca R (2016) Arbuscular mycorrhizal symbiosis and methyl jasmonate avoid the inhibition of root hydraulic conductivity caused by drought. Mycorrhiza 26:111–122
Sandhya V, SkZ Ali, Grover M, Reddy G, Venkateswaralu B (2010) Effect of plant growth promoting Pseudomonas spp. on compatible solutes antioxidant status and plant growth of maize under drought stress. Plant Growth Regul 62:21–30
Sandhya V, Ali SKZ, Grover M, Reddy G, Venkateswarlu B (2009) Alleviation of drought stress effects in sunflower seedlings by the exopolysaccharides producing Pseudomonas putida strain GAP-P45. Biol Fertil Soils 46:17–26
Sapre S, Gontia-Mishra I, Tiwari S (2019) ACC deaminase producing bacteria: a key player in alleviating abiotic stresses in plants. In: Kumar A, Meena VS (eds) Plant growth promoting rhizobacteria for agricultural sustainability-from theory to practices. Springer Nature, pp 267–291
Sarma RK, Saikia R (2014) Alleviation of drought stress in mung bean by strain Pseudomonas aeruginosa GGRJ21. Plant Soil 377:111–126
Selvakumar G, Panneerselvam P, Ganeshamurthy AN (2012) Bacterial mediated alleviation of abiotic stress in crops. In: Maheshwari DK (ed) Bacteria in agrobiology: stress management. Springer-Verlag, Berlin, pp 205–224
Shaharoona B, Bibi R, Arshad M, Zahir ZA, Hassan Z (2006) 1-aminocylopropane-1-carboxylate (ACC)-deaminase rhizobacteria extenuates ACC-induced classical triple response in etiolated pea seedlings. Pak J Bot 38:1491–1499
Sharifi R, Ryu C-M (2018) Revisiting bacterial volatile-mediated plant growth promotion: lessons from the past and objectives for the future. Ann Bot 122:349–358
Smirnoff N (1993) The role of reactive oxygen in the response of plants to water deficit and desiccation. J New Phytol 125:27–30
Srivastava S, Chaudhry V, Mishra A, Chauhan PS, Rehman A, Yadav A, Tuteja N, Nautiyal CS (2012) Gene expression profiling through microarray analysis in Arabidopsis thaliana colonized by Pseudomonas putida MTCC5279, a plant growth promoting rhizobacterium. Plant Sig Behav 7:235–245
Streeter JG (1985) Accumulation of alpha, alpha-trehalose by Rhizobium bacteria and bacteroids. J Bacteriol 164:78–84
Suárez R, Wong A, Ramírez M, Barraza A, Orozco MD, Cevallos MA, Lara M, Hernández G, Iturriaga G (2008) Improvement of drought tolerance and grain yield in common bean by overexpressing trehalose-6-phosphate synthase in rhizobia. Mol Plant-Microbe Interact 21:958–966
Sukkasem P, Kurniawan A, Kao TC, Chuang HW (2018) A multifaceted rhizobacterium Bacillus licheniformis functions as a fungal antagonist and a promoter of plant growth and abiotic stress tolerance. Environ Exper Bot 155:541–551
Sun X, Shi J, Ding G (2017) Combined effects of arbuscular mycorrhiza and drought stress on plant growth and mortality of forage sorghum. Appl Soil Ecol 119:384–391
Symanczik S, Lehmann MF, Wiemken A, Boller T, Courty P (2018) Effects of two contrasted arbuscular mycorrhizal fungal isolates on nutrient uptake by Sorghum bicolor under drought. Mycorrhiza 28:779–785
Timmusk S, El-Daim IAA, Copolovici L, Tanilas T, Kännaste A, Behers L, Nevo E, Seisenbaeva G, Stenström E, Niinemets Ü (2014) Drought-tolerance of wheat improved by rhizosphere bacteria from harsh environments: enhanced biomass production and reduced emissions of stress volatiles. PLoS ONE 9:e96086
Timmusk S, Wagner EGH (1999) The plant-growth-promoting rhizobacterium Paenibacillus polymyxa induces changes in Arabidopsis thaliana gene expression a possible connection between biotic and abiotic stress responses. Mol Plant-Microbe Interact 12:951–959
Tiwari S, Lata C, Chauhan PS, Nautiyal CS (2016) Pseudomonas putida attunes morphophysiological, biochemical and molecular responses in Cicer arietinum L. during drought stress and recovery. Plant Physiol Biochem 99:108–117
Tiwari S, Prasad V, Chauhan PS, Lata C (2017) Bacillus amyloliquefaciens confers tolerance to various abiotic stresses and modulates plant response to phytohormones through osmoprotection and gene expression regulation in rice. Front Plant Sci 8:1510
Ullah A, Manghwar H, Shaban M, Khan AH, Akbar A, Ali U, Fahad S (2019a) Phytohormones enhanced drought tolerance in plants: a coping strategy. Environ Sci Pollut Res 25:33103–33118
Ullah A, Nisar M, Ali H, Hazrat A, Hayat K, Keerio AA, Ihsan M, Laiq M, Ullah S, Fahad S, Khan A (2019b) Drought tolerance improvement in plants: an endophytic bacterial approach. Appl Microbiol Biotechnol 103:7385–7397
Urao T, Yakubova B, Satoha R, Yamaguchi-Shinozakia K, Sekib M, Hirayamab T, Shinozakib K (1999) A transmembrane hybrid-type histidine kinase in Arabidopsis functions as an osmosensor. Plant Cell 11:1743–1754
Vaishnav A, Choudhary DK (2019) Regulation of drought-responsive gene expression in Glycine max L. (merrill) is mediated through Pseudomonas simiae strain AU. J Plant Grow Regul 38:333–342
Vardharajula S, Zulfikar Ali S, Grover M, Reddy G, Bandi V (2011) Drought-tolerant plant growth promoting Bacillus spp.: effect on growth, osmolytes, and antioxidant status of maize under drought stress. J Plant Interact 6:1–14
Vargas L, Santa Brigida AB, MotaFilho JP et al (2014) Drought tolerance conferred to sugarcane by association with Gluconacetobacter diazotrophicus: a transcriptomic view of hormone pathways. PLoS ONE 9:e114744
Vejan P, Abdullah R, Khadiran T, Ismail S, Nasrulhaq Boyce A (2016) Role of plant growth promoting rhizobacteria in agricultural sustainability-a review. Molecules 21:573
Verma P, Yadav AN, Khannam KS, Panjiar N, Kumar S, Saxena AK, Suman A (2015a) Assessment of genetic diversity and plant growth promoting attributes of psychrotolerant bacteria allied with wheat (Triticum aestivum) from the northern hills zone of India. Ann Microbiol 65:1885–1899
Verma P, Yadav AN, Kumar V, Singh DP, Saxena AK (2017) Beneficial plant-microbes interactions: biodiversity of microbes from diverse extreme environments and its impact for crop improvement. In: Singh DP, Singh HB, Prabha R (eds) Plant-microbe interactions in agro-ecological perspectives: Volume 2: Microbial Interactions and Agro-Ecological Impacts. Springer, Singapore, pp 543–580.
Verma P, Yadav AN, Shukla L, Saxena AK, Suman A (2015b) Alleviation of cold stress in wheat seedlings by Bacillus amyloliquefaciens IARI-HHS2-30, an endophytic psychrotolerant K-solubilizing bacterium from NW Indian Himalayas. Natl J Life Sci 12:105–110
Vigani G, Rolli E, Marasco R, Dell’Orto M, Michoud G, Soussi A, Raddadi N, Borin S, Sorlini C, Zocchi G, Daffonchio D (2018) Root bacterial endophytes confer drought resistance and enhance expression and activity of a vacuolar H+ -pumping pyrophosphatase in pepper plants. Environ Microbiol 21:3212–3228.
Vurukonda SSKP, Vardharajula S, Shrivastava M, SkZ A (2016) Enhancement of drought stress tolerance in crops by plant growth promoting rhizobacteria. Microbiol Res 184:13–24
Wu QS, He JD, Srivastava AK, Zou YN, Kuča K (2019) Mycorrhiza enhances drought tolerance of citrus by altering root fatty acid compositions and their saturation levels. Tree Physiol 39:1149–1158
Wu HH, Zou YN, Rahman MM, Ni QD, Wu QS (2017) Mycorrhizas alter sucrose and proline metabolism in trifoliate orange exposed to drought stress. Sci Rep 7:42389
Xie W, Hao Z, Zhou X, Jiang X, Xu L, Wu S, Zhao A, Zhang X, Chen B (2018) Arbuscular mycorrhiza facilitates the accumulation of glycyrrhizin and liquiritin in Glycyrrhiza uralensis under drought stress. Mycorrhiza 28:285–300
Xie Z, Chu Y, Zhang W, Lang D, Zhang X (2019) Bacillus pumilus alleviates drought stress and increases metabolite accumulation in Glycyrrhiza uralensis Fisch. Environ Exper Bot 158:99–106
Xu L, Li T, Wu Z, Feng H, Yu M, Zhang X, Chen B (2018) Arbuscular mycorrhiza enhances drought tolerance of tomato plants by regulating the 14-3-3 genes in the ABA signaling pathway. Appl Soil Ecol 125:213–221
Yadav AN, Kour D, Sharma S, Sachan SG, Singh B, Chauhan VS, Sayyed RZ, Kaushik R, Saxena AK (2019a) Psychrotrophic microbes: biodiversity, mechanisms of adaptation, and biotechnological implications in alleviation of cold stress in plants. In: Sayyed RZ, Arora NK, Reddy MS (eds) Plant growth promoting rhizobacteria for sustainable stress management: Volume 1: Rhizobacteria in Abiotic Stress Management. Springer, Singapore, pp 219–253. https://doi.org/10.1007/978-981-13-6536-2_12
Yadav AN, Kumar V, Prasad R, Saxena AK, Dhaliwal HS (2018a) Microbiome in crops: diversity, distribution and potential role in crops improvements. In: Prasad R, Gill SS, Tuteja N (eds) Crop improvement through microbial biotechnology. Elsevier, USA, pp 305–332
Yadav AN, Mishra S, Singh S, Gupta A (2019b) Recent advancement in white biotechnology through fungi Volume 1: Diversity and Enzymes Perspectives. Springer International Publishing, Cham
Yadav AN, Singh S, Mishra S, Gupta A (2019c) Recent advancement in white biotechnology through fungi. Volume 2: Perspective for Value-Added Products and Environments. Springer International Publishing, Cham
Yadav AN, Singh S, Mishra S, Gupta A (2019d) Recent advancement in white biotechnology through fungi. Volume 3: Perspective for Sustainable Environments. Springer International Publishing, Cham
Yadav AN, Verma P, Kumar M, Pal KK, Dey R, Gupta A, Padaria JC, Gujar GT, Kumar S, Suman A, Prasanna R, Saxena AK (2015) Diversity and phylogenetic profiling of niche-specific Bacilli from extreme environments of India. Ann Microbiol 65:611–629
Yadav AN, Verma P, Kumar S, Kumar V, Kumar M, Singh BP, Saxena AK, Dhaliwal HS (2018b) Actinobacteria from rhizosphere: molecular diversity, distributions and potential biotechnological applications. In: Singh B, Gupta V, Passari A (eds) New and future developments in microbial biotechnology and bioengineering. USA, pp 13–41. https://doi.org/10.1016/b978-0-444-63994-3.00002-3
Yadav AN, Verma P, Kumar V, Sachan SG, Saxena AK (2017a) Extreme cold environments: a suitable niche for selection of novel psychrotrophic microbes for biotechnological applications. Adv Biotechnol Microbiol 2:1–4
Yadav AN, Verma P, Sachan SG, Saxena AK (2017b) Biodiversity and biotechnological applications of psychrotrophic microbes isolated from Indian Himalayan regions. EC Microbiol ECO 01:48–54
Yadav N, Yadav A (2018) Biodiversity and biotechnological applications of novel plant growth promoting methylotrophs. J Appl Biotechnol Bioeng 5:342–344
Yasmin H, Nosheen A, Naz R, Bano A, Keyani R (2017) l-tryptophan-assisted PGPR-mediated induction of drought tolerance in maize (Zea mays L.). J Plant Interact 12:567–578
Zade NSE, Sadeghi A, Moradi P (2019) Streptomyces strains alleviate water stress and increase peppermint (Mentha piperita) yield and essential oils. Plant Soil 434:441–452
Zhang H, Murzello C, Sun Y, Kim MS, Xie X, Jeter RM, Zak JC, Dowd SE et al (2010) Choline and osmotic-stress tolerance induced in Arabidopsis by the soil microbe Bacillus subtilis (GB03). Mol Plant Microbe Interact 23:1097–1104
Zhang S, Moyne AL, Reddy MS, Kloepper JW (2002) The role of salicylic acid in induced systemic resistance elicited by plant growth-promoting rhizobacteria against blue mold of tobacco. Biol Cont 25:288–296
Zhao R, Guo W, Bi N, Guo J, Wang L, Zhao J, Zhang J (2015) Arbuscular mycorrhizal fungi affect the growth, nutrient uptake and water status of maize (Zea mays L.) grown in two types of coalmine spoils under drought stress. Appl Soil Ecol 88:41–49
Zhu JK (2002) Salt and drought stress signal transduction in plants. Ann Rev Plant Biol 53:247–273
Acknowledgements
The author I. Gontia-Mishra acknowledges the funding provided by Science and Engineering Research Board, New Delhi, India, grant number PDF/2017/001001.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Gontia-Mishra, I., Sapre, S., Deshmukh, R., Sikdar, S., Tiwari, S. (2020). Microbe-Mediated Drought Tolerance in Plants: Current Developments and Future Challenges. In: Yadav, A., Singh, J., Rastegari, A., Yadav, N. (eds) Plant Microbiomes for Sustainable Agriculture. Sustainable Development and Biodiversity, vol 25. Springer, Cham. https://doi.org/10.1007/978-3-030-38453-1_12
Download citation
DOI: https://doi.org/10.1007/978-3-030-38453-1_12
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-38452-4
Online ISBN: 978-3-030-38453-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)