Abstract
Cotton is the chief crop and main pillar of textile industry. Its fiber and seed have significant economic importance. However, salinity interferes with the normal growth functioning and results in halted growth and declined yield of fiber and seed. Salinity effects are more obvious at early growth stages of cotton, limiting final yield. Salt decreases boll formation per plant which ultimately gives decreased fiber yield and poor lint quality. Salinity is a global issue increasing every year due to uncontrolled measures and improper land management. Application of saline irrigation water is adding increments to already existing salts and deteriorating the productive soil. Arid regions are totally dependent upon rain for growth of cotton. Salt problem is more in arid regions due least availability of moisture and water for flushing salts from cotton root zone. Moreover, higher temperature favors excessive evaporation under arid conditions and leaving salt on the upper surface of soil. Salts at the surface soil impede cotton seed germination. In this chapter, we discussed formation of saline soils and their sources which deter cotton growth. Physiological changes, oxidative stress caused due to salinity, role of molecular transporters involved in detoxification and specific gene expression is also illuminated.
Access provided by Autonomous University of Puebla. Download chapter PDF
Similar content being viewed by others
Keywords
19.1 Introduction
Soil salinity is foremost burden on agricultural lands, becoming hurdles for productive exploitation of agricultural lands for vigorous crop growth (Haque 2006; Lobell et al. 2007). Globally utilization of natural resources is increasing day by day and burgeoning population which severely influencing agriculture and different factors contributing toward worse soil conditions creating saline environment (Shahbaz and Ashraf 2013). Generally saline soils exhibit the characteristics of 4 dS m−1 and 15% exchangeable sodium percentage that inhibits the functioning of crops and ultimately result in retard growth and loss in yield (Munns 2005; Shahzad et al. 2017). Worldwide salt-affected soils cover the area of 20% of cultivable lands, and the problem on agricultural lands is 33%; moreover, it is increasing on annual basis due to change in climate. Several factors are currently under investigation which includes decreased supply of water and higher evaporation rate, weathering of rocks, saline water irrigation, and mismanagement of cultural practices (Jamil et al. 2011). No continent is free from saline problem (Table 19.1); it is estimated that 800 million lands are salt affected (FAO 2019), and salt affected land area would be increased by the year 2050 unless no precautionary measures cannot be taken with proper amendments (Ashraf 2009). It mostly occurs in arid and semiarid regions of the world which exist in all continents except Antarctica (Rengasamy 2006). Cotton (Gossypium sp.) is a cash crop named as white gold and king of fiber according its economic importance (Moseley 2001; Ahmad et al. 2014, 2017, 2018; Abbas and Ahmad 2018; Ahmad and Raza 2014; Ali et al. 2011, 2013a, b, 2014a, b). In addition to its fiber, its seed contains 15–20% oil contents, and seed cake is a rich source of protein for animal feed (Kothari et al. 2016). Cotton seed cake is used as manure having 6.5%, 3.0%, and 2.3% NPK. Cotton has significant importance for textile industry, and it is the oldest known crop employed for fiber (Amin et al. 2017, 2018; Bakhsh et al. 2012; Rahman et al. 2018; Tariq et al. 2017, 2018; Usman et al. 2009). Salinity obstructs its growth and quality traits (Ashraf 2010). Cotton is one of the salt-tolerant crops, but it is sensitive at early stages to salt stress, i.e., germination and seedling emergence as compared to later growth stages (Ahmad et al. 2002). Cotton has the ability of retaining sodium contents more than 95% in its tissues (Gouia et al. 1994). It is also reported to retain Na+ accumulation in leaves and roots of salt-tolerant cultivars (Sun and Liu 2001). Accumulation of salt in soils deters seed germination and plant growth and creates osmotic imbalance and toxicity leading to poor stand establishment (Ahmad et al. 2002; Bednarz et al. 2002). Under high salinity stress, it halts the physiological functioning of cotton by limiting photosynthesis and respiration which result in lower boll formation and poor fiber quality (Brugnoli and Lauteri 1991). Cotton is considered as moderately salt tolerant; however, it is depicted that its tolerance level varies from cultivars to cultivars (Leidi and Saiz 1997). Its tolerance level is also up to 7.7 dS m−1 (Maas and Hoffman 1977). Cotton salt tolerance level is dependent upon its ability of regulating sodium ion homeostasis in its tissues to minimize detrimental effects of cytotoxicity and by adjusting osmotic balance (Munns and Tester 2008). Salt stress disrupts photosynthetic machinery by firstly closing stomata of plants (Brugnoli and Lauteri 1991). Moreover, several reactive oxygen species (ROS) are formed under stress that further aggravates the oxidative stress to cotton (Meloni et al. 2003). On the other hand, contemporary modern approaches are available to cope with salt stress conditions to avail higher yield returns (Qadir et al. 2000; Gao et al. 2009; Shahzad et al. 2017). Saline soils can be reclaimed with plenty of good quality irrigation water (Murtaza et al. 2006). Chemical approach includes application of gypsum that improves the physical structure of soil and efficiently removes the soil from root zone (Murtaza et al. 2013). Arbuscular mycorrhizal fungi (AMF) are widely used to improve salinity tolerance in crops (Wu et al. 2010; Hajiboland et al. 2010; He et al. 2007). Improvements in cotton against salt stress tolerance is also conferred through biotechnological techniques (Zhang et al. 2011a, b, 2007; Ashraf et al. 2018a, b). It has broadened our horizon to understand the genes responsible for contributing stress tolerance (Liang et al. 2018).
19.2 Saline Soil Genesis and Distribution
Salinization originate from number of variety sources, and primary sources include weathering of rocks and minerals in the earth crust which contributes soluble salt concentration to soil and sea (Van Breemen and Buurman 2002). Weathering of earth crusts is also distributing salts to ocean and other water bodies in its surrounding. Weathering of rocks is the primary origin of salinization, while secondary origins are irrigation with saline water (Bui 2013). Saline water irrigation deposits salts in soils under harch climatic conditions. Water evaporation increases resulting in accumulation of salts on the surface of the soil which give rise to saline soil (Fig. 19.1). Underground water utilized for irrigating crops is a principal source of soil sodication because it also contains sodium fairly in high concentration (Bauder et al. 2011). The other important process of saline soil genesis is the rise in ground table in some parts around the globe (Fan et al. 2013). Groundwater table is increasing at the rate of 1–2 m annually. It is mineralized with salts, and due to capillary action, water is moving upward and enhancing salts in the root zone of the plant and leave behind on the surface after evaporation (Xie et al. 2011). Fossil salts often forms in arid region due to earlier depositions of salts in the form of marine deposits (Thomas 2011). Seepage of salts from upper soil parts to the lower soil layer which ultimately finish their journey in underground water contaminates it with accumulation of excessive salt. Agricultural lands in the vicinity of ocean and seas absorb salt either via wind or groundwater movement (Ondrasek et al. 2011). It is chiefly responsible for movement of salts up to many kilometers in the surrounding area. It was observed that rate of deposition by this mean is 20–100 kg ha−1 per year in coastal areas (Rengasamy 2010). Anthropogenic activities disturb the natural environment and intentionally increasing salinity of soil. It comprises construction of roads, dams, canals, and other irrigation system on saline strata (Metternicht and Zink 2003; Oldeman et al. 1991). Likely all these human practices block the movement of water which causes severe increase to rise in water table creates water logging and finally salinization of land (Day Jr et al. 2013). Moreover, mostly farmers are unaware of the water requirement of the crops, and they tend to over-irrigate the crops which result in increase in water table (Carreira et al. 2014). Fertilization is another practice which works as a double-edged sword to increase yield of crop while on the other hand is harmful for soil health and properties leading toward contribution of salt to soil (Savci 2012). Poor waste management practices and dumping of animal waste on farm increase salt content for agricultural land. Salt stress declined agricultural productivity in many parts of world (Rozema and Flowers 2008). It was observed salinity covers more than 397 million hectares of land globally (FAO 2005). Salinity problem is exacerbated especially in arid regions because of less availability of moisture, and these regions also receive less rainfall (Pasternak and De Malach 1994; Villa-Castorena et al. 2003). Irrigated lands receive salts in the form of saline irrigated water with least leaching and poor drainage practices. Approximately 20% of the world’s land is irrigated with saline water (Sumner 1999). Almost 75 countries have been marked in the red zone of salinity stress with a moderate to severe salinity problems covering a total area of 831 million hectare, which was productive for crops in the past (Martinez-Beltran and LiconaManzur 2005; Qadir et al. 2000). Land coverage of salinity in across different continents is illustrated in Fig. 19.2 (Hoang et al. 2016).
19.3 Production of Cotton on Saline Soil
Cotton production halts due to presence of excessive salt concentration in soil, disturbing uptake of other essential nutrients for the growth and yield of crop (Jafri and Rafiq 1994). The main mineral ion that causes salinity stress is sodium chloride and sodium sulfate (Reich et al. 2017). Salinity influences soil nutrients for cotton by osmotic effect by increasing its concentration in the vicinity of cotton roots for uptake by plant and likely in the roots (Wang et al. 2015). On another hand NaCl deposition in roots creates dehydration by pulling out water from the roots (Younis et al. 2014). Moreover, it also disrupts the ion solution inside the plant cells that results in hindrance of physiological processes of cotton (Ashraf et al. 2017). Under saline environment, the nutrients that becomes inhibited are nitrogen, potassium, phosphorus, and zinc, so it is imperative to consider the application of these nutrients on such soils for ensuring better supply of these nutrients to combat with toxic effects of sodium and providing maintenance strategy to keep the other function of cotton to normal limits (Hu and Schmidhalter 2005; Dong et al. 2010c). Cotton is sensitive to salt at early growth stages such as during seed germination, emergence, and establishment in comparison to mature plant (Chen et al. 2010). It was suggested to take extensive measures during early growth stages to ensure robust growth for final fruitful yield (Ashraf 2010). Delayed and non-uniformity in emergence rate are the first gesture of salinity after sowing cotton seeds in saline soil (Dong et al. 2009). Decrease in germination of seed is obvious with the proportional concentration of salt concentration in soil (Ashraf et al. 2002). Complete failure of germination was observed at 16 dS m−1 (Kahlown and Azam 2002). Gossypium barbadense genotypes exhibited higher tolerance toward salinity as compared to Gossypium hirsutum and Gossypium arboreum cotton (Ahmad et al. 2002). Salinity has also mild effect on root length at low concentration, while higher concentration of sodium affects its root length (Chen et al. 2018a, b). Decreased root length and delayed secondary root growth have been reported (Cramer et al. 1987). Sodium is also a competitor of calcium to limit its uptake by cotton roots (Byrt et al. 2018). Cotton is salt tolerant, but its vegetative growth is severely affected on saline soil. Shoot is more sensitive to salt than roots. Decrease in leaf area per plant, stem thickness, and shoot and root weight reduction are important morphology traits significantly influenced by uptake of salt and higher accumulation in plants via roots (Anjum et al. 2005). On the other hand, application of calcium is beneficial for limiting Na+ uptake by plants (Reid and Smith 2000). Biomass production of cotton reduces with adverse salt stress. Boll formation also reduces with less number of fruits per plant (Gandahi et al. 2017). Salinization results in boll shedding and senescence of leaves (Rathert 1982; Brugnoli and Björkman 1992). Cotton fiber yield and length are important traits for textile industry; however, it results in poor lint quality due to interference of salts (Soares et al. 2018). Its seed also holds significant position for feed and oil sector which also becomes affected with saline conditions (Ashraf 2010). Cotton seed is a good source of oil. Decrease of cotton seed yield causes decrease in seed oil contents. It was reported that 50% decrease in seed yield was with a salinity level of 16.75 dS m−1 (Ali et al. 1986). Morphologically decrease in cotton was also reported with salinity conditions as enhanced sodicity hampered growth with a significant decrease in root length, fruit number, and ultimately lint yield (Dodd et al. 2013). Cotton is deemed to tolerate salt moderately within the range of 7.7 dS m−1 (Maas 1986), thereby it is efficient candidate crop against salt-affected soils for its growth (Ahmad et al. 2002). However, reduction in its growth and yield traits has been reported with the increase in its salinity threshold level (Khan et al. 2001; Dong 2012b; Higbie et al. 2010). Moreover, tolerance level varies among different genotypes of cotton (Ashraf 2010; Hanif et al. 2008). Cotton grown on saline soil for years exhibited elevation of Na+ concentration and decrease in phosphorus and potassium concentration in plant tissues (Rochester 2010). Similar negative correlation was observed from young and mature leaves at different growth stages of cotton (Dodd 2007). Its tolerance mechanism is dependent on genotypes for different growth stages. In order to make it tolerant, it is essential to have basic knowledge of cotton regarding tolerance at varying growth stages (Ashraf and Ahmad 2000). General perception is that halophyte plant accumulates enormous quantity of NaCl ions in tissues to adapt themselves to saline conditions, while mesophytes restrict the entry of these ions (Flowers and Colmer 2015). Higher Na+ concentration perturbs other nutrient in plants as mentioned earlier that it disturbs osmotic balance, thereby disturbed K+/Na+ uptake and its interference with each other for uptake is important mechanism for considering tolerance among cultivars (Leidi and Saiz 1997). Negative correlation of these ions uptake confers positive correlation toward salinity tolerance such as higher K+ allows lower Na+ ions uptake (Cramer et al. 1987). Moreover Na+ exclusion also resulted in tolerance, and it was also observed in tolerant cultivars that K+ concentration was measured to be higher in leaves (Ahmad et al. 2002). Oxidative stress also triggered with the entry of Na+ ion and disturbing other ions, so ROS needs to be eliminated or their effects needs to be suppressed; in this regard ROS scavenger comes to play their role which is mentioned later in this chapter. Another important strategy that is being adapted by breeders is development of salt-tolerant cotton cultivars to improve the characteristics of their local high-yielding cultivar (Harshavardhan et al. 2018). Breeding for salinity tolerance has reported potential results for improving yield of crops (Blum 2018). Cotton conventional breeding improved tolerance against salinity with a 7.4% increase in yield (Ledbetter 1987; Ashraf and Akram 2009). Later selection method was exploited which was also potential method for developing salt-tolerant cultivars (Da Silva et al. 1992).
19.4 Physiological Changes and Role of Antioxidant Enzymes
Salt stress is an environmental factor that interrupts physiological and biochemical changes in cotton (Meloni et al. 2003; Zhang et al. 2014). Salinity stress affects physiological processes by increasing respiration rate and disturbing mineral ion distribution especially displacement of calcium and potassium with Na+, sometimes leading to ion toxicity (Kinraide 1999). Salinity limits the photosynthesis process as well as cell growth (Munns et al. 2006). It directly inhibits CO2 availability due to limited diffusion via stomata, mesophyll, and disrupting metabolism of photosynthesis (Lawlor and Cornic 2002; Flexas et al. 2007). Salt interfere the photosynthetic machinery of cotton by decreasing its photosynthetic rate (Meloni et al. 2003). It results from the decline in chlorophyll contents of plants under adverse conditions (Jaleel et al. 2008). Foremost mechanism during salinity stress is the adjustment of stomata to limits transpiration and maintains cell turgor (Miller et al. 2010). Stomatal closure is the response against dehydration with the impaired supply of CO2 to shifting plants to water-saving strategy (Brodribb and Holbrook 2003). In this regard abscisic acid (ABA) plays a crucial role as a signaling molecule from its production site (roots) to the leaves for closure of stomata (Wilkinson and Davies 2002). Moreover, under saline conditions Na+ uptake is increased which halts the uptake of other essential nutrients such as potassium, calcium, and manganese (Hasegawa et al. 2000). Salt uptake in tissues of plant brutally influences older leaves with higher accumulation of salts (Munns 2002; Chaves et al. 2003). Decrease in nutrient uptake and impaired photosynthetic process was observed in cotton in response to salinity (Liu et al. 2014a, b). Salinity stress causes excessive production of ROS (You and Chan 2015). ROS are produced in different cell compartments of plant, generally the sites of its production are chloroplast and mitochondria (Jubany-Marí et al. 2009). It includes oxygen free radicals, i.e., singlet oxygen (1O2), superoxide anions (O2 •−), hydrogen peroxide (H2O2), and hydroxyl radicals (•OH) (Zheng et al. 2009). These ROS species have detrimental effects on plant functioning by causing damage to DNA and protein (Foyer and Noctor 2005). Oxidative damages are alleviated by ROS-scavenging enzymes (Hussain et al. 2018). Antioxidant enzymes are superoxide dismutase (SOD), catalase (CAT), peroxidases (POD), and ascorbate peroxidase (APX) (Hossain and Dietz 2016). These enzymes work in a sequence to alleviate oxidative stress of cotton under salinity stress (Garratt et al. 2002). SOD is the first ROS-scavenger enzyme to start its function for alleviating ROS species; it dismutases O2 − into H2O2 and O2 (Azarabadi et al. 2017). Immediately after that POD starts catalyzing the H2O2 to H2O and O2 (Waszczak et al. 2018). Later CAT and APX capture H2O2 and convert in to H2O (Mittler 2017). Antioxidant enzymes work as indicator of salt-tolerant and susceptible cultivars (Ashraf and Harris 2004). It was observed that enzymes activities increased during salinity stress to cope with ROS species and aids in tolerance to stress (Koca et al. 2007). It was reported from studies on cotton that tolerant cultivar exhibited a higher level of antioxidant enzymes (Zhang et al. 2014; Liu et al. 2014a, b; Ibrahim et al. 2017). Increase in SOD activity in cotton alleviated oxidative stress caused by salinity (Xie et al. 2008). Lipid peroxidation results from ROS species where malondialdehyde is the product formed due to lipid peroxidation, and it represents how much damage has occurred to the plant in stress condition (Sharma et al. 2012). MDA is a marker for observing oxidative damage to the plants (Davey et al. 2005). Cotton exhibited increment in its amount due to salinity stress (Tang et al. 2007; Meloni et al. 2003). It also assists in differentiating the tolerant and susceptible cultivars (Liu et al. 2014a, b). Proline also accumulates during salinity stress to work in osmotic adjustment, alleviates free radicals, and maintains cellular redox potential (Ashraf and Foolad 2007). It accumulates normally in cytosol for osmotic regulations to cytoplasm (Ashraf et al. 2018a, b). Higher concentration of proline is correlated with tolerance of cultivar as tolerant cultivar depicts increase level of proline during salinity stress (Hayat et al. 2012). Furthermore, exogenous application also conferred significant results against salinity tolerance (Heuer 2003). Influence of proline for osmotic adjustment was observed in cotton (Meloni et al. 2001). Cotton was subjected to salt stress conditions, and marked increase proline concentration was observed to tolerate the stress (Golan-Goldhirsh et al. 1990). Proline is also associated for improving fiber quality of cotton (Xu et al. 2013).
19.5 Genetic Engineering and Molecular Biological Tool
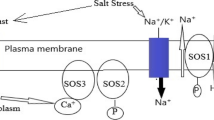
Salts in high amount in soil result in higher uptake by plants and ultimately causing disruption of membrane functions and inhibit cell division, photosynthesis, and development of plants (Flowers 1999; Horie and Schroeder 2004). Plants as being immobile to move to favorable growth conditions thus have to survive under existing environmental conditions. First plant organs that come in contact with saline environment are root hairs from where it is taken up and transported to epidermis and cortex of plants (Cao et al. 2017). Sodium is transported to the shoots via transpiration stream in xylem, and rarely it is returned to the roots through phloem (Wu et al. 2018). Therefore, it is observed that its movement is unidirectional and results in higher accumulation of sodium in shoots (Ishikawa and Shabala 2019). Sodium accumulates in higher concentration in shoots as compared to roots, and from shoots it is transported to the leaves (Farooq et al. 2015). Potassium is a crucial nutrient for plant especially under salt stress because it is the competitor of sodium; if K+ concentration is higher, it inhibits the entry of Na+ in the plant cell and protects it from detrimental effects of Na+ (Adams and Shin 2014). Potassium is also essential for photosynthetic apparatus to aid in its functioning (Lu et al. 2016). Concentration of Na+ in high quantity disrupts the membrane functions causing disturbance in ion homeostasis which result in stunted growth and sometimes lead to death of cell (Flowers et al. 2014). Plants both halophytes and glycophytes utilize an identical strategy of regulating and maintaining Na+ ion homeostasis by coordinated functions of ion transporters for controlling the flow into the plants (Wang et al. 2017). Moreover, there are numerous selective pumps that favor the uptake of K+ compared to Na+ (Zhang et al. 2018). High-affinity potassium transporter (HKT) proteins are reported in numerous plants for playing selective role of K+ uptake against Na+ (Fairbairn et al. 2000; Golldack et al. 2002; Horie et al. 2001; Sunarpi et al. 2005). HKT was the first potassium-selective transporter found in plants (Schachtman and Schroeder 1994). It also plays function in Na+ exclusion from leaves and maintains K+ homeostasis in leaves (Horie et al. 2005). Currently Na+/H+ transporter located in tonoplast was identified that play role in outward movement of Na+ from cytosol to apoplast or vacuole (Zeng et al. 2018). However, it is an energy-consuming process for cell, and proton pumps give force for transporting Na+ contrary to electrochemical gradient (Blumwald et al. 2000). It was reported in cotton roots that Na+ concentration was lower due to higher influx of H+ via potential role of Na+/H+ antiporter (Kong et al. 2011). Another transporter is salt overly sensitive pathway (SOS); it also works as an exchanger in plasma membrane (Qiu et al. 2002). It becomes activated and plays crucial role in Na+ exclusion mechanism to render plant salt tolerance (Zhu 2000). Mechanism of Na+ transport by different pumps is depicted in Fig. 19.3. Engineered transgenic cotton is also worth mentioning for their contribution toward development of salt-tolerant cotton cultivars (Liu et al. 2014a, b). Transgenic plants contain any foreign DNA that plant does not contain naturally to improve traits and quality of plants (Rao et al. 2009). Transgenic cotton exhibited higher root development and minimized the transpiration rate; in that way it became tolerant to survive on saline soil conditions (Liu et al. 2014a, b). Cotton plants were transformed with H+-PPase gene exhibited tolerance in transgenic cotton lines by improving vegetative growth and higher photosynthetic rate resultantly lower ion leakage from the plants under salt stress as compared with non-transgenic plants (Bock 2010). Overexpression of Arabidopsis vacuolar pyrophosphatase gene (AVP1) in cotton contributed to 20% increase in fiber due to a number of boll formations under salt stress (Zhang et al. 2011a, b). Senescence results from decline in cytokinin contents in plant under stress conditions thereby isopentenyl transferase (IPT) gene has potential role in supplementing cytokinin and enhancing chlorophyll contents and delayed in senescence was reported in transgenic cotton to survive on salt affected soils (Liu et al. 2012a, b). Numerous studies have been accomplished successfully in manipulating cotton for salinity stress tolerance (Shen et al. 2015; Zhang et al. 2007; Yu et al. 2016; Cheng et al. 2018; Song et al. 2018). Plants sense stress environment and generate signals to try to adapt and adjust themselves to the salinity stress (Zhu 2016). Principal factor is regulation of genes that response under salinity stresses (Egea et al. 2018). However, it is a complex set of many genes that regulate to make the plant tolerant, while some genes are down regulated (Albaladejo et al. 2018). Salt stress induces gene expression; it was reported that the expression of salt overly sensitive (SOS2) gene and plasma membrane H+ ATPase (PMA2) gene was observed to be higher in cotton (Peng et al. 2016). In another study conducted on cotton gene expression profiles were documented for salt stress by exploiting microarray technique, and all the observed genes conferred cotton tolerant to salinity stress, furthermore some transporters also exhibited their role in rendering tolerance (Zhu et al. 2013). Genetic transporter expression is efficient for tolerance, and they also improve quality traits of cotton under adverse saline conditions (He et al. 2005). Some of the genes and their potential role under salt stress in cotton are given in Table 19.2.
Illustration of Na+ transport mechanisms against salt stress response and role of molecular transporters. Sodium concentration increases under saline conditions for uptake via plant roots. High-affinity potassium transporter (HKT1) unloads Na+ from the xylem. Na+/H+ exchanger (NHX) and salt overly sensitive (SOS1) assist in detoxification mechanism by decreasing the concentration of Na+ with H+ ion concentration
19.6 Agronomic Practices to Circumvent Salinity for Cotton
Proper sowing method and good cultivation practices circumvent the effect of salt stress to cotton (Anjum et al. 2005). Cultivation methods such as mulching maintain moisture and protect from evaporation (Dong 2012b). Mulching practice to protect cotton from salinity effect is not new (Sandoval and Benz 1966). Straw mulching for 3 years reduced soil salinity (Benz et al. 1967). Mulching of cotton burr at the rate of 90 t ha−1 with intermittent sprinkling assisted in removing salts from root zone (Carter and Fanning 1964). This mulching technique is adapted for last many decades for growing cotton (Liu et al. 2012a, b). Plastic mulching with polyethylene is a general practice among cotton growers to protect seeds from complete failure of seed germination and safeguard early emergence on salt affected soils (Dong et al. 2009). Plastic mulching improved growth and lint yield of cotton by raising temperature of the soil (Dong et al. 2007). Benefits of conserving moisture are vital after its adoption that aided in control of saline environment in root zone (Bezborodov et al. 2010). Moreover, mulching is documented for unequal dispersion of salts which imparted suitable growth of roots and reduced damage of salt (Bezborodov et al. 2010). Sowing method has positive effect to overcome salinity stress (Sarangi et al. 2017). Cotton crops sown on ridges showed better growth and development as compared to crop sown on flat beds (Dong et al. 2010b). It was inferred that by exploiting this ridge sowing method there was non-uniform distribution of salts across the field as well as reduced deposition of Na+ in the root zone (Dong et al. 2008). Time of sowing is also important factor as cotton sown during normal growing season in temperate regions exhibited weak stand establishment and resulted in late maturity (Dong et al. 2007). However, late sowing efficiently enhanced seedling emergence and vigorous stand establishment due to rise in temperature with a declined Na+ contents in tissues of cotton (Dong et al. 2010a). Planting density also played a role in mitigating saline conditions. Cotton yield increase was observed by increasing planting density because enormous amount of salts might reduce plant size (Feinerman 1983). Increasing population density under severe salinity improved cotton seed yield (Dong 2012a). Plant density increased vegetative growth production, and it had positive effect for seed yield of cotton (Zhang et al. 2012a, b).
19.7 Reclamation Options and Fertilizer Management of Cotton for Saline Soils
Saline soils with excessive accumulated salts exacerbate soil physic-chemical properties which ultimately create harmful growth conditions for plants (Chabra 1996). Salinity deteriorates the soil physical structure by creating dispersion of particles, soil erosion, and sometimes water logged conditions (Warrence et al. 2002). In order to amend these soils, different reclamation strategies are in practice nowadays (Qadir et al. 2000). Reclamation can be done by considering different features appropriate in the selection of site, soil depth, and presence of hard pan and finally the most important presence of salt (Murtaza et al. 2006; Ghafoor et al. 2004). Freshwater availability free from salt can be used for removing salts from root zone and upper surface of soil with considerable leaching characteristics of soil (Bezborodov et al. 2010). It should also be considered that leaching should not be too much in order to protect the groundwater table form salts and also the amount of water applied for removal of salts. Proper irrigation management practices also contribute their role in minimizing salinity both for cotton crop and reclaiming soil to some extent (Wang et al. 2012). Sprinkler and drip irrigation are the preferred ways to remove the excessive salt from root zone of plant as well as protecting the groundwater table by limiting excessive leaching of salts as compared to flood irrigation (Karlberg and de Vries 2004). Leaching of soluble salts is an effective way of protecting rhizosphere from its toxic effects. It was documented from one study that drip irrigations conferred fruitful results for sustaining cotton productivity with the alleviation of sodium ion from roots of cotton at different growth stages (Kang et al. 2012). Chemical amendments are also beneficial of removing exchangeable sodium from cation exchange sites (Sahin et al. 2002). Gypsum is the most commonly used chemical amendment because it is cheap and easy availability (Ilyas et al. 1997). Organic amendments are also deployed for remediation of saline soils. Different organic amendments manure, mulch, and compost proved to have reclaiming characteristics of saline soil (Diacono and Montemurro 2015; Suzuki 1999). Emergence of cotton seedling increased on saline soil with organic manure application (da Costa et al. 2016). These amendments have multifaceted role in soil by enhancing aggregation of particles, improving water holding capacity to protect soil from drying via evaporation which causes accumulation of more Na+ in soil pockets (Lu et al. 2015). Moreover, they also provide nutrient that are essential to combat with Na+ for uptake by plant (Zhang et al. 2015). Therefore, fertilizer selection is crucial for saline soils. Potassium is normally applied as potassium chloride (muriate of potash) which is not suitable for saline soil. Potassium magnesium sulfate fertilizer is effective to cope with NaCl stress (Khare et al. 2015). Furthermore, nitrate has also potential to alleviate influence of higher chloride concentration in soil (Shrivastava and Kumar 2015). Management of fertilizers is a good option for cotton growing on salinized soils. Application of nitrogen to soil enhanced uptake by plant and confined Na+ uptake in cotton (Kawakami et al. 2010). Potassium and nitrogen foliar sprays are direct source of absorption by cotton leaves and bolls for sturdy growth of vegetative parts (Jabeen and Ahmad 2009). Interference of salt with nitrogen was illustrated by varying their levels and concluded that higher salt contents had profound effects on cotton plants (Chen et al. 2010). Good supply of nitrogen to the bolls either by foliar or basal resulted in higher lint yield of cotton on saline soils (Zhang et al. 2012a, b). Fertigation is another method for simultaneous application of water and fertilizers to crops (Castellanos et al. 2012). It is a modern agriculture approach for reduction of environmental pollution and feasible for cumulative chemical fertilizer usage efficiency (Hagin et al. 2002). It was employed for cotton growth to eradicate salt and give salt-free environment to the roots of cotton (Min et al. 2016). The benefits of fertigation for cotton crop was proved in another study that irrigation with suitable nitrogen fertilizer eradicated the devastating effecting of Na+ and provided nitrogen nutrient to the cotton for higher growth and yield (Min et al. 2017).
19.8 Conclusion
Salinity is an emerging global issue; cotton is being grown in more than 80 countries and plays a key role in the economy of various countries. Salinity is causing soil degradation at an alarming rate. Cotton productivity is severely hindered by degradation of soil. Cotton needs sustainable development under this situation; in order to cope with salinity, there are various mitigation tactics which can lead to sustainable productivity of cotton. Development of adaptable cotton cultivars is a basic step to face this challenge in the long run. Various scientists are keen to develop those techniques to enable crop to battle with various hazards faced during their life cycle. Adaptation in plants can be developed using various biotechnological tools. Development of appropriate surrounding situations for optimum development of crop is also considered; reclamation of soil, the use of appropriate fertilizer application, and good quality irrigation water are essential to minimize saline conditions and provide better growth of cotton.
Saline soils are formed; some decades now, the interference of human activities is worsening the already existing salt-affected soils. Moreover, adding increment to the salinized land area, proper control measures should be taken to control such activities polluting our natural environment and water resources. Salinity issues should be addressed, and awareness needs to create among farming communities directly involved for agriculture on such soils. Proper reclamation strategies potentially improved salt-affected soils and eradicating salts from root zone for improving growth of cotton. Irrigation water analysis should be done for irrigation saline soils. High EC water should not be applied to salt-affected soil. Sowing method for cotton is an efficient way for overcoming salinity problem. Likely density and plant methods should be considered. Fertilizer management is also important to supplement essential nutrients that become limited due to presence of sodium. High salt-tolerant cotton genotypes are suitable for growth on saline soil. Proper selection of cultivar also confers higher yield returns. If you don’t have any local salt-tolerant cultivar, breeding approaches should be exploited for improving traits of your local cultivar. The conventional breeding assist in fixing traits in susceptible but high yielding cotton genotypes it renders susceptible cotton genotype a tolerant one after several crossing for breeding. Physiological changes also occur in cotton due to the intake of Na+. Molecular approaches paved the path for improving antioxidant enzyme activities to combat salinity problem in cotton tissues. Development of transgenic also exhibited tolerance to salt stress which can be used growing cotton on salt-affected soil.
Abbreviations
- ABA:
-
Abscisic acid
- AMF:
-
Arbuscular mycorrhizal fungi
- APX:
-
Ascorbate peroxidase
- CAT:
-
Catalase
- H2O2 :
-
Hydrogen peroxide
- IPT:
-
Isopentenyl transferase
- 1O2 :
-
Singlet oxygen
- O2 •− :
-
Superoxide anions
- •OH:
-
Hydroxyl radicals
- POD:
-
Peroxidases
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
References
Abbas Q, Ahmad S (2018) Effect of different sowing times and cultivars on cotton fiber quality under stable cotton-wheat cropping system in southern Punjab, Pakistan. Pak J Life Soc Sci 16:77–84
Adams E, Shin R (2014) Transport, signaling, and homeostasis of potassium and sodium in plants. J Integr Plant Biol 56(3):231–249
Ahmad S, Raza I (2014) Optimization of management practices to improve cotton fiber quality under irrigated arid environment. J Food Agric Environ 2(2):609–613
Ahmad S, Khan N, Iqbal MZ, Hussain A, Hassan M (2002) Salt tolerance of cotton (Gossypium hirsutum L.). Asian J Plant Sci 1(6):715–719
Ahmad S, Raza I, Ali H, Shahzad AN, Atiq-ur-Rehman, Sarwar N (2014) Response of cotton crop to exogenous application of glycinebetaine under sufficient and scarce water conditions. Braz J Bot 37(4):407–415
Ahmad S, Abbas Q, Abbas G, Fatima Z, Atique-ur-Rehman, Naz S, Younis H, Khan RJ, Nasim W, Habib urRehman M, Ahmad A, Rasul G, Khan MA, Hasanuzzaman M (2017) Quantification of climate warming and crop management impacts on cotton phenology. Plants 6(7):1–16
Ahmad S, Iqbal M, Muhammad T, Mehmood A, Ahmad S, Hasanuzzaman M (2018) Cotton productivity enhanced through transplanting and early sowing. Acta Sci Biol Sci 40:e34610
Albaladejo I, Egea I, Morales B, Flores FB, Capel C, Lozano R, Bolarin MC (2018) Identification of key genes involved in the phenotypic alterations of res (restored cell structure by salinity) tomato mutant and its recovery induced by salt stress through transcriptomic analysis. BMC Plant Biol 18(1):213
Ali A, Ahmad N, Makhdum MI, Gill KH (1986) Effect of soil salinity on cotton (Gossypium hirsutum) on early stage of growth. Pak J Sci 1:37–40
Ali H, Afzal MN, Ahmad F, Ahmad S, Akhtar M, Atif R (2011) Effect of sowing dates, plant spacing and nitrogen application on growth and productivity on cotton crop. Int J Sci Eng Res 2(9):1–6
Ali H, Abid SA, Ahmad S, Sarwar N, Arooj M, Mahmood A, Shahzad AN (2013a) Integrated weed management in cotton cultivated in the alternate-furrow planting system. J Food Agric Environ 11(3&4):1664–1669
Ali H, Abid SA, Ahmad S, Sarwar N, Arooj M, Mahmood A, Shahzad AN (2013b) Impact of integrated weed management on flat-sown cotton (Gossypium hirsutum L.). J Anim Plant Sci 23(4):1185–1192
Ali H, Hameed RA, Ahmad S, Shahzad AN, Sarwar N (2014a) Efficacy of different techniques of nitrogen application on American cotton under semi-arid conditions. J Food Agric Environ 12(1):157–160
Ali H, Hussain GS, Hussain S, Shahzad AN, Ahmad S, Javeed HMR, Sarwar N (2014b) Early sowing reduces cotton leaf curl virus occurrence and improves cotton productivity. Cer Agron Moldova XLVII(4):71–81
Amin A, Nasim W, Mubeen M, Nadeem M, Ali L, Hammad HM, Sultana SR, Jabran K, Habib urRehman M, Ahmad S, Awais M, Rasool A, Fahad S, Saud S, Shah AN, Ihsan Z, Ali S, Bajwa AA, Hakeem KR, Ameen A, Amanullah, Rehman HU, Alghabar F, Jatoi GH, Akram M, Khan A, Islam F, Ata-Ul-Karim ST, Rehmani MIA, Hussain S, Razaq M, Fathi A (2017) Optimizing the phosphorus use in cotton by using CSM-CROPGRO-cotton model for semi-arid climate of Vehari-Punjab, Pakistan. Environ Sci Pollut Res 24(6):5811–5823
Amin A, Nasim W, Mubeen M, Ahmad A, Nadeem M, Urich P, Fahad S, Ahmad S, Wajid A, Tabassum F, Hammad HM, Sultana SR, Anwar S, Baloch SK, Wahid A, Wilkerson CJ, Hoogenboom G (2018) Simulated CSM-CROPGRO-cotton yield under projected future climate by SimCLIM for southern Punjab, Pakistan. Agric Syst 167:213–222
Anjum R, Ahmed A, Ullah R, Jahangir M, Yousaf M (2005) Effect of soil salinity/sodicity on the growth and yield of different varieties of cotton. Int J Agric Biol 7(4):606–608
Ashraf M, Ahmad S (2000) Influence of sodium chloride on ion accumulation, yield components and fibre characteristics in salt-tolerant and salt-sensitive lines of cotton (Gossypium hirsutum L.). Field Crop Res 66:115–127
Ashraf M (2009) Biotechnological approach of improving plant salt tolerance using antioxidants as markers. Biotechnol Adv 27:84–93
Ashraf M, Akram NA (2009) Improving salinity tolerance of plants through conventional breeding and genetic engineering: an analytical comparison. Biotechnol Adv 27(6):744–752
Ashraf M (2010) Salt tolerance of cotton: some new advances. Crit Rev Plant Sci 21(1):1–30
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59(2):206–216
Ashraf M, Harris PJC (2004) Potential biochemical indicators of salinity tolerance in plants. Plant Sci 166(1):3–16
Ashraf MY, Sarwar G, Ashraf M, Afaf R, Sattar A (2002) Salinity induced changes in α-amylase activity during germination and early cotton seedling growth. Biol Plantarum 45(4):589–591
Ashraf M, Shahzad SM, Imtiaz M, Rizwan MS, Iqbal MM (2017) Ameliorative effects of potassium nutrition on yield and fiber quality characteristics of cotton (Gossypium hirsutum L.) under NaCl stress. Soil Environ 36(1):51–58
Ashraf J, Zuo D, Wang Q, Malik W, Zhang Y, Abid MA, Cheng H, Yang Q, Song G (2018a) Recent insights into cotton functional genomics: progress and future perspectives. Plant Biotechnol J 16(3):699–713
Ashraf MA, Iqbal M, Rasheed R, Hussain I, Perveen S, Mahmood S (2018b) Dynamic proline metabolism: importance and regulation in water-limited environments. In: Plant metabolites and regulation under environmental stress. Academic, Cambridge, MA, pp 323–336
Azarabadi S, Abdollahi H, Torabi M, Salehi Z, Nasiri J (2017) ROS generation, oxidative burst and dynamic expression profiles of ROS-scavenging enzymes of superoxide dismutase (SOD), catalase (CAT) and ascorbate peroxidase (APX) in response to Erwinia amylovora in pear (Pyrus communis L.). Eur J Plant Pathol 147(2):279–294
Bakhsh A, Rao AQ, Shahid AA, Husnain T (2012) Spatio temporal expression pattern of an insecticidal gene (cry2A) in transgenic cotton lines. Not Sci Biol 4:115–119.
Bauder TA, Waskom RM, Sutherland PL, Davis JG, Follett RH, Soltanpour PN (2011) Irrigation water quality criteria. Service in Action; No. 0.506
Bednarz CW, Shurley WD, Anthony WS (2002) Losses in yield, quality, and profitability of cotton from improper harvest timing. Agron J 94(5):1004–1011
Benz LC, Sandoval FM, Willis WO (1967) Soil salinity changes with fallow and a straw mulch on fallow. Soil Sci 104:63–68
Bezborodov GA, Shadmanov DK, Mirhashimov RT, Yuldashev T, Qureshi AS, Noble AD, Qadir M (2010) Mulching and water quality effects on soil salinity and sodicity dynamics and cotton productivity in Central Asia. Agric Ecosyst Environ 138(1–2):95–102
Blum A (2018) Plant breeding for stress environments. CRC Press, Taylor & Francis Group, Boca Raton, FL, p 231
Blumwald E, Aharon GS, Apse MP (2000) Sodium transport in plant cells. Biochim Biophys Acta 1465:140–151
Bock R (2010) The give-and-take of DNA: horizontal gene transfer in plants. Trends Plant Sci 15:11–22
Brodribb TJ, Holbrook NM (2003) Stomatal closure during leaf dehydration, correlation with other leaf physiological traits. Plant Physiol 132(4):2166–2173
Brugnoli E, Björkman O (1992) Growth of cotton under continuous salinity stress-influence on allocation pattern, stomatal and nonstomatal components of photosynthesis and dissipation of excess light energy. Planta 187:335–347
Brugnoli E, Lauteri M (1991) Effects of salinity on stomatal conductance, photosynthetic capacity, and carbon isotope discrimination of salt-tolerant (Gossypium hirsutum L.) and salt-sensitive (Phaseolus vulgaris L.) C3 non-halophytes. Plant Physiol 95(2):628–635
Bui EN (2013) Soil salinity: a neglected factor in plant ecology and biogeography. J Arid Environ 92:14–25
Byrt CS, Munns R, Burton RA, Gilliham M, Wege S (2018) Root cell wall solutions for crop plants in saline soils. Plant Sci 269:47–55
Cao D, Lutz A, Hill CB, Callahan DL, Roessner U (2017) A quantitative profiling method of phytohormones and other metabolites applied to barley roots subjected to salinity stress. Front Plant Sci 7:2070
Carreira PM, Marques JM, Nunes D (2014) Source of groundwater salinity in coastline aquifers based on environmental isotopes (Portugal): natural vs. human interference. A review and reinterpretation. Appl Geochem 41:163–175
Carter DL, Fanning CD (1964) Combining surface mulches and periodic water applications for reclaiming saline soils. Soil Sci Soc Am J 28:564–567
Castellanos MT, Cabello MJ, Cartagena MC, Tarquis AM, Arce A, Ribas F (2012) Nitrogen uptake dynamics, yield and quality as influenced by nitrogen fertilization in ‘Piel de sapo’ melon. Span J Agric Res 10(3):756–767
Chabra R (1996) Soil salinity and water quality. CRC Press, Taylor & Francis Group, Balkema Publishers, Brookfield, VT
Chaves MM, Maroco JP, Pereira JS (2003) Understanding plant responses to drought—from genes to the whole plant. Funct Plant Biol 30(3):239–264
Chen W, Hou Z, Wu L, Liang Y, Wei C (2010) Effects of salinity and nitrogen on cotton growth in arid environment. Plant Soil 326(1–2):61–73
Chen W, Jin M, Ferré TPA, Liu Y, Xian Y, Shan T, Ping X (2018a) Spatial distribution of soil moisture, soil salinity, and root density beneath a cotton field under mulched drip irrigation with brackish and fresh water. Field Crop Res 215:207–221
Chen W, Wang Z, Jin M, Ferré TPA, Wang J, Huang J, Wang X (2018b) Effect of sodium chloride and manganese in irrigation water on cotton growth. Agron J 110:900–909
Cheng C, Zhang Y, Chen X, Song J, Guo Z, Li K, Zhang K (2018) Co-expression of AtNHX1 and TsVP improves the salt tolerance of transgenic cotton and increases seed cotton yield in a saline field. Mol Breed 38(2):19
Cramer GR, Lynch J, Läuchli A, Epstein E (1987) Influx of Na+, K+, and Ca2+ into roots of salt-stressed cotton seedlings: effects of supplemental Ca2+. Plant Physiol 83(3):510–516
da Costa MML, Gomes Nobre R, Soares de Lima G, Raj Gheyi H, Wesley Alves Pinheiro F, Sudário Dias A, Almeida dos AnjosSoares L (2016) Saline-sodic soil and organic matter addition in the cultivation of the colored cotton ‘BRS Topázio’. Semina: Ciências Agrárias 37(2):701–704
Da Silva MJ, De Souza JG, Neto MB, Da Silva JV (1992) Selection on 3 cotton cultivars for tolerance to germination under saline conditions. Pesquisa Agropec Brasileira 27:655–659
Davey MW, Stals E, Panis B, Keulemans J, Swennen RL (2005) High-throughput determination of malondialdehyde in plant tissues. Anal Biochem 347(2):201–207
Day JW Jr, Yanez-Arancibia A, Kemp WM, Crump BC (2013) Introduction to estuarine ecology. In: Day JW, Crump BC, Kemp WM, Yáñez-Arancibia A (eds) Estuarine ecology, 2nd edn. Wiley-Blackwell, Hoboken NJ, p 550
Diacono M, Montemurro F (2015) Effectiveness of organic wastes as fertilizers and amendments in salt-affected soils. Agriculture 5(2):221–230
Dodd K (2007) Characterising the soil and plant interactions that affect the growth and nutrition of cotton in sodic Vertosols. PhD Thesis, The University of New England, Armidale, NSW, Australia
Dodd K, Guppy CN, Lockwood PV, Rochester IJ (2013) The effect of sodicity on cotton: does soil chemistry or soil physical condition have the greater role? Crop Pasture Sci 64(8):806–815
Dong H (2012a) Technology and field management for controlling soil salinity effects on cotton. Aus J Crop Sci 6(2):333
Dong HZ (2012b) Underlying mechanisms and related techniques of stand establishment of cotton on coastal saline-alkali soil. China J Appl Ecol 23(2):566–572
Dong H, Li W, Tang W, Li Z, Zhang D (2007) Enhanced plant growth, development and fiber yield of Bt transgenic cotton by an integration of plastic mulching and seedling transplanting. Ind Crop Prod 26:298–306
Dong H, Li W, Tang W, Zhang D (2008) Furrow seeding with plastic mulching increases stand establishment and lint yield of cotton in a saline field. Agron J 100:1640–1646
Dong H, Li W, Tang W, Zhang D (2009) Early plastic mulching increases stand establishment and lint yield of cotton in saline fields. Field Crop Res 111:269–275
Dong H, Li W, Xin C, Tang W, Zhang D (2010a) Late planting of short-season cotton in saline fields of the Yellow River Delta. Crop Sci 50:292–300
Dong H, Kong X, Luo Z, Li W, Xin C (2010b) Unequal salt distribution in the root zone increases growth and yield of cotton. Eur J Agron 33:285–292
Dong H, Kong X, Li W, Tang W, Zhang D (2010c) Effects of plant density and nitrogen and potassium fertilization on cotton yield and uptake of major nutrients in two fields with varying fertility. Field Crop Res 119(1):106–113
Egea I, Pineda B, Ortíz-Atienza A, Plasencia FA, Drevensek S, García-Sogo B, Yuste-Lisbona FJ, Barrero-Gil J, Atarés A, Flores FB, Barneche F, Angosto T, Capel C, Salinas J, Vriezen W, Esch E, Bowler C, Bolarín MC, Moreno V, Lozano R (2018) The SlCBL10 calcineurin B-like protein ensures plant growth under salt stress by regulating Na+ and Ca2+ homeostasis. Plant Physiol 176(2):1676–1693
Fairbairn DJ, Liu W, Schachtman DP, Gomez-Gallego S, Day SR, Teasdale RD (2000) Characterisation of two distinct HKT1-like potassium transporters from Eucalyptus camaldulensis. Plant Mol Biol 43:515–525
Fan Y, Li H, Miguez-Macho G (2013) Global patterns of groundwater table depth. Science 339(6122):940–943
FAO (2005) Global network on integrated soil management for sustainable use of salt effected soils. http://www.fao.org/ag/AGL/agll/spush/intro.htm
FAO (2019) Management of salt affected soils. Soil Sci Soc Am J. http://www.fao.org/soils-portal/soil-management/management-of-some-problem-soils/salt-affected-soils/more-information-on-salt-affected-soils/en/. Accessed 21 June 2019
Farooq M, Hussain M, Wakeel A, Siddique KHM (2015) Salt stress in maize: effects, resistance mechanisms, and management. A review. Agron Sustain Dev 35(2):461–481
Feinerman E (1983) Crop density and irrigation with saline water. West J Agric Econ 8:134–140
Flexas J, Diaz-Espejo A, Galmés J, Kaldenhoff R, Medrano H, Ribas-Carbo M (2007) Rapid variations of mesophyll conductance in response to changes in CO2 concentration around leaves. Plant Cell Environ 30(10):1284–1298
Flowers TJ (1999) Salinisation and horticultural production. Sci Hortic 78:1–4
Flowers TJ, Colmer TD (2015) Plant salt tolerance: adaptations in halophytes. Ann Bot 115(3):327–331
Flowers TJ, Munns R, Colmer TD (2014) Sodium chloride toxicity and the cellular basis of salt tolerance in halophytes. Ann Bot 115(3):419–431
Foyer CH, Noctor G (2005) Redox homeostasis and antioxidant signaling: a metabolic interface between stress perception and physiological responses. Plant Cell 17:1866–1875
Gandahi AW, Kubar A, Sarki MS, Talpur N, Gandahi M (2017) Response of conjunctive use of fresh and saline water on growth and biomass of cotton genotypes. J Basic Appl Sci 13:326–334
Gao SQ, Chen M, Xia LQ, Xiu HJ, Xu ZS, Li LC, Zhao CP, Cheng XG, Ma YZ (2009) A cotton (Gossypium hirsutum) DRE-binding transcription factor gene, GhDREB, confers enhanced tolerance to drought, high salt, and freezing stresses in transgenic wheat. Plant Cell Rep 28(2):301–311
Gao W, Xu FC, Guo DD, Zhao JR, Liu J, Guo YW, Singh PK, Ma XN, Long L, Botella JR, Song CP (2018) Calcium-dependent protein kinases in cotton: insights into early plant responses to salt stress. BMC Plant Biol 18(1):15
Garratt LC, Janagoudar BS, Lowe KC, Anthony P, Power JB, Davey MR (2002) Salinity tolerance and antioxidant status in cotton cultures. Free Radic Biol Med 33(4):502–511
Ghafoor A, Qadir M, Murtaza G (2004) Salt-affected soils: principles of management, vol xxv. Allied Book Centre, Lahore, Pakistan. 304p
Golan-Goldhirsh A, Hankamer B, Lips SH (1990) Hydroxyproline and proline content of cell walls of sunflower, peanut and cotton grown under salt stress. Plant Sci 69(1):27–32
Golldack D, Su H, Quigley F, Kamasani UR, Munoz-Garay C, Balderas E, Popova OV, Bennett J, Bohnert HJ, Pantoja O (2002) Characterization of a HKT-type transporter in rice as a general alkali cation transporter. Plant J 31:529–542
Gouia H, Ghorbal MH, Touraine B (1994) Effects of NaCl on flows of N and mineral ions and on NO3-reduction rate within whole plants of salt-sensitive bean and salt-tolerant cotton. Plant Physiol 105(4):1409–1418
Hagin J, Sneh M, Lowengart-Aycicegi A (2002) Fertigation – fertilization through irrigation. IPI Research Topics No. 23. Ed. by Johnston AE. International Potash Institute, Basel, Switzerland
Hajiboland R, Aliasgharzadeh N, Laiegh SF, Poschenrieder C (2010) Colonization with arbuscular mycorrhizal fungi improves salinity tolerance of tomato (Solanum lycopersicum L.) plants. Plant Soil 331(1–2):313–327
Hanif M, Noor E, Murtaza N, Qayyum A, Malik W (2008) Assessment of variability for salt tolerance at seedling stage in Gossypium hirsutum L. J Food Agric Environ 6:134–138
Haque SA (2006) Salinity problems and crop production in coastal regions of Bangladesh. Pak J Bot 38(5):1359–1365
Harshavardhan VT, Govind G, Kalladan R, Sreenivasulu N, Hong CY (2018) Cross-protection by oxidative stress: improving tolerance to abiotic stresses including salinity. In: Kumar V, Wani SH, Suprasanna P, Tran LP (eds) Salinity responses and tolerance in plants, vol 1. Springer, Berlin, pp 283–305
Hasegawa PM, Bressan RA, Zhu JK, Bohnert HJ (2000) Plant cellular and molecular response to high salinity. Annu Rev Plant Physiol Plant Mol Biol 51:463–499
Hayat S, Hayat Q, Alyemeni MN, Wani AS, Pichtel J, Ahmad A (2012) Role of proline under changing environments: a review. Plant Signal Behav 7(11):1456–1466
He C, Yan J, Shen G, Fu L, Holaday AS, Auld D, Blumwald E, Zhang H (2005) Expression of an Arabidopsis vacuolar sodium/proton antiporter gene in cotton improves photosynthetic performance under salt conditions and increases fiber yield in the field. Plant Cell Physiol 46(11):1848–1854
He Z, He C, Zhang Z, Zou Z, Wang H (2007) Changes of antioxidative enzymes and cell membrane osmosis in tomato colonized by arbuscular mycorrhizae under NaCl stress. Colloids Surf B: Biointerfaces 59(2):128–133
Heuer B (2003) Influence of exogenous application of proline and glycinebetaine on growth of salt-stressed tomato plants. Plant Sci 165(4):693–699
Higbie SM, Wang F, Stewart JM, Sterling TM, Lindemann WC, Hughs E, Zhang J (2010) Physiological response to salt (NaCl) stress in selected cultivated Tetraploid cottons. Int J Agron 2010:1–12
Hoang TML, Tran TN, Nguyen TKT, Williams B, Wurm P, Bellairs S, Mundree S (2016) Improvement of salinity stress tolerance in rice: challenges and opportunities. Agronomy 6(4):54
Horie T, Schroeder JI (2004) Sodium transporters in plants. Diverse genes and physiological functions. Plant Physiol 136:2457–2462
Horie T, Yoshida K, Nakayama H, Yamada K, Oiki S, Shinmyo A (2001) Two types of HKT transporters with different properties of Na+ and K+ transport in Oryza sativa. Plant J 27:129–138
Horie ST, Motoda J, Kubo M, Yang H, Yoda K, Horie R, Chan WY, Leung HY, Hattori K, Konomi M, Osumi M (2005) Enhanced salt tolerance mediated by AtHKT1 transporter-induced Na+ unloading from xylem vessels to xylem parenchyma cells. Plant J 44(6):928–938
Hossain MS, Dietz KJ (2016) Tuning of redox regulatory mechanisms, reactive oxygen species and redox homeostasis under salinity stress. Front Plant Sci 7:548
Hu Y, Schmidhalter U (2005) Drought and salinity: a comparison of their effects on mineral nutrition of plants. J Plant Nutr Soil Sci 168(4):541–549
Hussain S, Khalid MF, Saqib M, Ahmad S, Zafar W, Rao MJ, Morillon R, Anjum MA (2018) Drought tolerance in citrus rootstocks is associated with better antioxidant defense mechanism. Acta Physiol Plant 40(8):135
Ibrahim W, Ahmed IM, Chen X, Wu F (2017) Genotype-dependent alleviation effects of exogenous GSH on salinity stress in cotton is related to improvement in chlorophyll content, photosynthetic performance, and leaf/root ultrastructure. Environ Sci Pollut Res 24(10):9417–9427
Ilyas M, Qureshi RH, Qadir MA (1997) Chemical changes in a saline-sodic soil after gypsum application and cropping. Soil Technol 10(3):247–260
Ishikawa T, Shabala S (2019) Control of xylem Na+ loading and transport to the shoot in rice and barley as a determinant of differential salinity stress tolerance. Physiol Plant 165(3):619–631
Jabeen R, Ahmad R (2009) Alleviation of the adverse effects of salt stress by foliar application of sodium antagonistic essential minerals of cotton (Gossypium hirsutum L.). Pak J Bot 41(5):2199–2208
Jafri AZ, Rafiq A (1994) Plant growth and ionic distribution in cotton (Gossypium hirsutum L.) under saline environment. Pak J Bot 26:105
Jaleel CA, Sankar B, Sridharan R, Panneerselvam R (2008) Soil salinity alters growth, chlorophyll content, and secondary metabolite accumulation in Catharanthusroseus. Turk J Biol 32(2):79–83
Jamil A, Riaz S, Ashraf M, Foolad MR (2011) Gene expression profiling of plants under salt stress. Crit Rev Plant Sci 30(5):435–458
Jubany-Marí T, Munné-Bosch S, López-Carbonell M, Alegre L (2009) Hydrogen peroxide is involved in the acclimation of the Mediterranean shrub, Cistus albidus L., to summer drought. J Exp Bot 60:107–120
Kahlown MA, Azam M (2002) Individual and combined effect of waterlogging and salinity on crop yields in the Indus basin. Irrig Drain 51(4):329–338
Kang Y, Wang R, Wan S, Hu W, Jiang S, Liu S (2012) Effects of different water levels on cotton growth and water use through drip irrigation in an arid region with saline ground water of Northwest China. Agric Water Manage 109:117–126
Karlberg L, de Vries FWTP (2004) Exploring potentials and constraints of low-cost drip irrigation with saline water in sub-Saharan Africa. Phys Chem Earth 29:1035–1042
Kawakami EM, Oosterhuis DM, Snider JL (2010) Effect of salinity on cotton nitrogen uptake and assimilation of urea applied with N-(n-Butyl) thiophosphorctriamide and dicyandiaminde. Summaries Arkansas Cotton Research. AAES Res Ser 589:40–45
Khan TM, Saeed M, Mukhtar MS, Khan AM (2001) Assesment of variation for salinity tolerance in some hybrids of cotton (Gossypium hirsutum L.). Int J Agric Biol 3:167–170
Khare T, Kumar V, Kishor PB (2015) Na+ and Cl− ions show additive effects under NaCl stress on induction of oxidative stress and the responsive antioxidative defense in rice. Protoplasma 252(4):1149–1165
Kinraide TB (1999) Interactions among Ca2+, Na+ and K+ in salinity toxicity: quantitative resolution of multiple toxic and ameliorative effects. J Exp Bot 50(338):1495–1505
Koca H, Bor M, Özdemir F, Türkan I (2007) The effect of salt stress on lipid peroxidation, antioxidative enzymes and proline content of sesame cultivars. Environ Exp Bot 60(3):344–351
Kong X, Luo Z, Dong H, Eneji AE, Li W (2011) Effects of non-uniform root zone salinity on water use, Na+ recirculation, and Na+ and H+ flux in cotton. J Exp Bot 63(5):2105–2116
Kothari N, Campbell BT, Dever JK, Hinze LL (2016) Combining ability and performance of cotton germplasm with diverse seed oil content. Crop Sci 56(1):19–29
Lawlor DW, Cornic G (2002) Photosynthetic carbon assimilation and associated metabolism in relation to water deficits in higher plants. Plant Cell Environ 25(2):275–294
Ledbetter CA (1987) Heritability of salt tolerance during germination and emergence in short staple cotton. Diss Abstr Int Sci Eng 47(11):113
Leidi EO, Saiz JF (1997) Is salinity tolerance related to Na accumulation in upland cotton (Gossypium hirsutum) seedlings? Plant Soil 190(1):67–75
Liang W, Ma X, Wan P, Liu L (2018) Plant salt-tolerance mechanism: a review. Biochem Biophys Res Commun 495(1):286–291
Liu MX, Yang JS, Li XM, YU M, Wang J (2012a) Effects of irrigation water quality and drip tape arrangement on soil salinity, soil moisture distribution, and cotton yield (Gossypium hirsutum L.) under mulched drip irrigation in Xinjiang, China. J Integr Agric 11(3):502–511
Liu YD, Yin ZJ, Yu JW, Li J, Wei HL, Han XL, Shen FF (2012b) Improved salt tolerance and delayed leaf senescence in transgenic cotton expressing the Agrobacterium IPT gene. Biol Plantarum 56(2):237–246
Liu G, Li X, Jin S, Liu X, Zhu L, Nie Y, Zhang X, Zhang J (2014a) Overexpression of rice NAC gene SNAC1 improves drought and salt tolerance by enhancing root development and reducing transpiration rate in transgenic cotton. PLoS One 9(1):e86895
Liu S, Dong Y, Xu L, Kong J (2014b) Effects of foliar applications of nitric oxide and salicylic acid on salt-induced changes in photosynthesis and anti-oxidative metabolism of cotton seedlings. Plant Growth Regul 73(1):67–78
Liu JF, Zhang SL, Tang HL, Wu LZ, Dong LJ, Liu LD, Che WL (2015) Overexpression of an Aeluropuslittoralis Parl. potassium transporter gene, AlHAK1, in cotton enhances potassium uptake and salt tolerance. Euphytica 203(1):197–209
Lobell DB, Ortiz-Monasterio JI, Gurrola FC, Valenzuela L (2007) Identification of saline soils with multiyear remote sensing of crop yields. Soil Sci Soc Am J 71(3):777–783
Lu H, Lashari MS, Liu X, Ji H, Li L, Zheng J, Kibue GW, Joseph S, Pan G (2015) Changes in soil microbial community structure and enzyme activity with amendment of biochar-manure compost and pyroligneous solution in a saline soil from Central China. Eur J Soil Biol 70:67–76
Lu Z, Lu J, Pan Y, Lu P, Li X, Cong R, Ren T (2016) Anatomical variation of mesophyll conductance under potassium deficiency has a vital role in determining leaf photosynthesis. Plant Cell Environ 39(11):2428–2439
Maas EV (1986) Salt tolerance of plants. Appl Agric Res 1:12–26
Maas EV, Hoffman GJ (1977) Crop salt tolerance–current assessment. J Irr Drain Div 103:115–134
Martinez-Beltran J, LiconaManzur C (2005) Overview of salinity problems in the world and FAO strategies to address the problem. In: International salinity forum managing saline soils and water: science, technology and social issues, Riverside Convention Center, Riverside, California, USA, 25–28 April 2005, pp 311–314
Meloni DA, Oliva MA, Ruiz HA, Martinez CA (2001) Contribution of proline and inorganic solutes to osmotic adjustment in cotton under salt stress. J Plant Nutr 24(3):599–612
Meloni DA, Oliva MA, Martinez CA, Cambraia J (2003) Photosynthesis and activity of superoxide dismutase, peroxidase and glutathione reductase in cotton under salt stress. Environ Exp Bot 49(1):69–76
Metternicht GI, Zink JA (2003) Remote sensing of soil salinity: potential and constraints. Remote Sens Environ 85:1–20
Miller G, Suzuki N, Ciftci-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ 33(4):453–467
Min W, Guo H, Zhou G, Zhang W, Ma L, Ye J, Hou Z, Wu L (2016) Soil salinity, leaching, and cotton growth as affected by saline water drip irrigation and N fertigation. Acta Agric Scand B Soil Plant Sci 66(6):489–501
Min W, Guo H, Hu Z, Zhang H, Ye J, Hou Z (2017) Cotton growth and the fate of N fertilizer as affected by saline water irrigation and N fertigation in a drip-irrigated field. Acta Agric Scand B Soil Plant Sci 67(8):712–722
Mittler R (2017) ROS are good. Trends Plant Sci 22(1):11–19
Moseley WG (2001) Sahelian ‘white gold’ and rural poverty-environment interactions: the political ecology of cotton production, environmental change, and household food economy in Mali. Doctoral dissertation, University of Georgia
Munns R (2002) Comparative physiology of salt and water stress. Plant Cell Environ 25(2):239–250
Munns R (2005) Genes and salt tolerance: bringing them together. New Phytol 167:645–663
Munns R, Tester M (2008) Mechanisms of salinity tolerance. Annu Rev Plant Biol 59:651–681
Munns R, James RA, Läuchli A (2006) Approaches to increasing the salt tolerance of wheat and other cereals. J Exp Bot 57(5):1025–1043
Murtaza G, Ghafoor A, Qadir M (2006) Irrigation and soil management strategies for using saline-sodic water in a cotton–wheat rotation. Agric Water Manage 81(1–2):98–114
Murtaza G, Murtaza B, Usman HM, Ghafoor A (2013) Amelioration of saline-sodic soil using gypsum and low quality water in following sorghum-berseem crop rotation. Int J Agric Biol 15(4):640–648
Oldeman LR, Hakkeling RTA, Sombroek WG (1991) World map of the status of human-induced soil degradation: an explanatory note. United Nations Environment Programme - I11. Global Assessment of Soil Degradation GLASOD, October 1990. International Soil Reference and Information Centre, Nairobi
Ondrasek O, Rengel Z, Veres S (2011) Soil salinisation and salt stress in crop production. InTech, Croatia. https://doi.org/10.5772/22248
Pasapula V, Shen G, Kuppu S, Paez-Valencia J, Mendoza M, Hou P, Chen J, Qiu X, Zhu L, Zhang X, Auld D, Blumwald E, Zhang H, Gaxiola R, Paytn P (2011) Expression of an Arabidopsis vacuolar H+ pyrophosphatase gene (AVP1) in cotton improves drought and salt tolerance and increases fibre yield in the field conditions. Plant Biotechnol J 9(1):88–99
Pasternak D, De Malach Y (1994) Crop irrigation with saline water. Handbook of plant and crop stress. Marcel Dekker, New York, pp 599–622
Peng Z, He S, Sun J, Pan Z, Gong WF, Lu Y, Du X (2016) Na+ compartmentalization related to salinity stress tolerance in upland cotton (Gossypium hirsutum) seedlings. Sci Rep 6:34548
Qadir M, Ghafoor A, Murtaza G (2000) Amelioration strategies for saline soils: a review. Land Degrad Dev 11:501–521
Qiu QS, Guo Y, Dietrich MA, Schumaker KS, Zhu JK (2002) Regulation of SOS1, a plasma membrane Na+/H+ exchanger in Arabidopsis thaliana, by SOS2 and SOS3. Proc Natl Acad Sci U S A 99(12):8436–8441
Rahman MH, Ahmad A, Wang X, Wajid A, Nasim W, Hussain M, Ahmad B, Ahmad I, Ali Z, Ishaque W, Awais M, Shelia V, Ahmad S, Fahad S, Alam M, Ullah H, Hoogenboom G (2018) Multi-model projections of future climate and climate change impacts uncertainty assessment for cotton production in Pakistan. Agric For Meteorol 253–254:94–113
Rathert G (1982) Influence of extreme K/Na ratios and high substrate salinity on plant metabolism of crops differing in salt tolerance. VI. Mineral distribution variability among different salt-tolerant cotton varieties. J Plant Nutr 5:183–193
Rao AQ, Bakhsh A, Kiani S, Shahzad K, Shahid AL, Husnain T, Riazuddin S (2009) The myth of plant transformation. Biotechnol Adv 27(6):753–763
Reich M, Aghajanzadeh T, Helm J, Parmar S, Hawkesford MJ, De Kok LJ (2017) Chloride and sulfate salinity differently affect biomass, mineral nutrient composition and expression of sulfate transport and assimilation genes in Brassica rapa. Plant Soil 411(1–2):319–332
Reid RJ, Smith FA (2000) The limits of sodium/calcium interactions in plant growth. Funct Plant Biol 27(7):709–715
Rengasamy P (2006) World salinization with emphasis on Australia. J Exp Bot 57:1017–1023
Rengasamy P (2010) Soil processes affecting crop production in salt-affected soils. Funct Plant Biol 37(7):613–620
Rochester IJ (2010) Phosphorus and potassium nutrition of cotton: interaction with sodium. Crop Pasture Sci 61:825–834
Rozema J, Flowers T (2008) Crops for a salinized world. Science 322(5907):1478–1480
Sahin U, Anapali O, Hanay A (2002) The effect of consecutive applications of leaching water applied in equal, increasing or decreasing quantities on soil hydraulic conductivity of a saline–sodic soil in the laboratory. Soil Use Manage 18:152–154
Sandoval FM, Benz LC (1966) Effect of bare fallow, barley and grass on salinity of a soil over a saline water table. Soil Sci Soc Am J 30:392–397
Sarangi SK, Maji B, Mandal UK, Mahanta KK, Mandal S, Sharma PC (2017) Raised bed sowing – a climate change adaptive maize cultivation practice for coastal saline region. Paper presented orally at 5th National Seminar of Indian Society of Soil Salinity and Water Quality (ISSSWQ), Swami Keshwanand Rajasthan Agricultural University, Bikaner, 21–23 January 2017, pp 71–72
Savci S (2012) An agricultural pollutant: chemical fertilizer. Int J Environ Sci Dev 3(1):77–80
Schachtman DP, Schroeder JI (1994) Structure and transport mechanism of a high affinity potassium uptake transporter from higher plants. Nature 370:655–658
Shahbaz M, Ashraf M (2013) Improving salinity tolerance in cereals. Crit Rev Plant Sci 32:237–249
Shahzad S, Khan MY, Zahir ZA, Asghar HN, Chaudhry UK (2017) Comparative effectiveness of different carriers to improve the efficacy of bacterial consortium for enhancing wheat production under salt affected field conditions. Pak J Bot 49(4):1523–1530
Sharma P, Jha AB, Dubey RS, Pessarakli M (2012) Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions. J Bot 2012:217037
Shen G, Wei J, Qiu X, Hu R, Kuppu S, Auld D, Blumwald E, Gaxiola R, Payton P, Zhang H (2015) Co-overexpression of AVP1 and AtNHX1 in cotton further improves drought and salt tolerance in transgenic cotton plants. Plant Mol Biol Rep 33(2):167–177
Shrivastava P, Kumar R (2015) Soil salinity: a serious environmental issue and plant growth promoting bacteria as one of the tools for its alleviation. Saudi J Biol Sci 22(2):123–131
Soares LA, Fernandes PD, de Lima GS, Suassuna JF, Brito MEB, da Sá FV (2018) Growth and fiber quality of colored cotton under salinity management strategies. Rev Bras Eng Agríc Ambient 22(5):332–337
Song J, Zhang R, Yue D, Chen X, Guo Z, Cheng C, Hu M, Zhang J, Zhang K (2018) Co-expression of ApGSMT2g and ApDMT2g in cotton enhances salt tolerance and increases seed cotton yield in saline fields. Plant Sci 274:369–382
Sumner ME (ed) (1999) Handbook of soil science. CRC Press, Boca Raton, FL
Sun X, Liu Y (2001) Test on criteria of evaluating salt tolerance of cotton cultivars. Zuo Wu Xue Bao 27(6):794–801
Sunarpi, Horie T, Motoda J, Kubo M, Yang H, Yoda K, Horie R, Chan WY, Leung HY, Hattori K, Konomi M, Osumi M, Yamagami M, Schroeder JI, Uozumi N (2005) Enhanced salt tolerance mediated by AtHKT1 transporter-induced Na+ unloading from xylem vessels to xylem parenchyma cells. Plant J 44(6):928–938
Suzuki K (1999) Effect of continuous compost application on water-stable soil macroaggregation in a field subjected to double cropping. Soil Sci Plant Nutr 45:1003–1007
Szabolcs I (1989) Salt-affected soils. CRC Press, Boca Raton, FL
Tang W, Luo Z, Wen SM, Dong HZ, Xin CS, Li WJ (2007) Comparison of inhibitory effects on leaf photosynthesis in cotton seedlings between drought and salinity stress. Cotton Sci 19:28–32
Tariq M, Yasmeen A, Ahmad S, Hussain N, Afzal MN, Hasanuzzaman M (2017) Shedding of fruiting structures in cotton: factors, compensation and prevention. Trop Subtrop Agroecosyst 20(2):251–262
Tariq M, Afzal MN, Muhammad D, Ahmad S, Shahzad AN, Kiran A, Wakeel A (2018) Relationship of tissue potassium content with yield and fiber quality components of Bt cotton as influenced by potassium application methods. Field Crop Res 229:37–43
Thomas DSG (ed) (2011) Arid zone geomorphology: process, form and change in drylands. Wiley, Hoboken, NJ, p 648
Usman M, Ahmad A, Ahmad S, Irshad M, Khaliq T, Wajid A, Hussain K, Nasim W, Chattha TM, Trethowan R, Hoogenboom G (2009) Development and application of crop water stress index for scheduling irrigation in cotton (Gossypium hirsutum L.) under semiarid environment. J Food Agric Environ 7(3&4):386–391
Van Breemen N, Buurman P (2002) Soil formation. Springer Science & Business Media, New York
Villa-Castorena M, Ulery AL, Catalán-Valencia EA, Remmenga MD (2003) Salinity and nitrogen rate effects on the growth and yield of Chile pepper plants. Soil Sci Soc Am J 67(6):1781–1789
Wang R, Kang Y, Wan S, Hu W, Liu S, Jiang S, Liu S (2012) Influence of different amounts of irrigation water on salt leaching and cotton growth under drip irrigation in an arid and saline area. Agric Water Manage 110:109–117
Wang R, Kang Y, Wan S (2015) Effects of different drip irrigation regimes on saline–sodic soil nutrients and cotton yield in an arid region of Northwest China. Agric Water Manage 153:1–8
Wang N, Qi H, Qiao W, Shi J, Xu Q, Zhou H, Yan G, Huang Q (2017) Cotton (Gossypium hirsutum L.) genotypes with contrasting K+/Na+ ion homeostasis: implications for salinity tolerance. Acta Physiol Plant 39(3):77
Warrence NJ, Bauder JW, Pearson KE (2002) Basics of salinity and sodicity effects on soil physical properties. Bozeman, MT, Department of Land Resources and Environmental Sciences, Montana State University, pp 1–29
Waszczak C, Carmody M, Kangasjärvi J (2018) Reactive oxygen species in plant signaling. Annu Rev Plant Biol 69:209–236
Wei Y, Xu Y, Lu P, Wang X, Li Z, Cai X, Zhou Z, Wang Y, Zhang Z, Lin Z, Liu F, Wang K (2017) Salt stress responsiveness of a wild cotton species (Gossypium klotzschianum) based on transcriptomic analysis. PLoS One 12(5):e0178313
Wilkinson S, Davies WJ (2002) ABA-based chemical signalling: the co-ordination of responses to stress in plants. Plant Cell Environ 25(2):195–210
Wu CA, Yang GD, Meng QW, Zheng CC (2004) The cotton GhNHX1 gene encoding a novel putative tonoplast Na+/H+ antiporter plays an important role in salt stress. Plant Cell Physiol 45(5):600–607
Wu QS, Zou YN, He XH (2010) Contributions of arbuscular mycorrhizal fungi to growth, photosynthesis, root morphology and ionic balance of citrus seedlings under salt stress. Acta Physiol Plant 32(2):297–304
Wu H, Zhang X, Giraldo JP, Shabala S (2018) It is not all about sodium: revealing tissue specificity and signalling roles of potassium in plant responses to salt stress. Plant Soil 431(1–2):1–17
Xie Z, Duan L, Tian X, Wang B, Eneji AE, Li Z (2008) Coronatine alleviates salinity stress in cotton by improving the antioxidative defense system and radical-scavenging activity. J Plant Physiol 165(4):375–384
Xie T, Liu X, Sun T (2011) The effects of groundwater table and flood irrigation strategies on soil water and salt dynamics and reed water use in the Yellow River Delta, China. Ecol Model 222(2):241–252
Xu WL, Zhang DJ, Wu YF, Qin LX, Huang GQ, Li J, Li L, Li XB (2013) Cotton PRP5 gene encoding a proline-rich protein is involved in fiber development. Plant Mol Biol 82(4–5):353–365
You J, Chan Z (2015) ROS regulation during abiotic stress responses in crop plants. Front Plant Sci 6:1092
Younis A, Riaz A, Ahmed I, Siddique MI, Tariq U, Hameed M, Nadeem M (2014) Anatomical changes induced by NaCl stress in root and stem of Gazania harlequin L. Agric Commun 2:8–14
Yu LH, Wu SJ, Peng YS, Liu RN, Chen X, Zhao P, Xu P, Zhu JB, Jiao GL, Pei Y, Xiang CB (2016) Arabidopsis EDT 1/HDG 11 improves drought and salt tolerance in cotton and poplar and increases cotton yield in the field. Plant Biotechnol J 14(1):72–84
Zeng Y, Li Q, Wang H, Zhang J, Du J, Feng H, Blumwald E, Yu L, Xu G (2018) Two NHX-type transporters from Helianthus tuberosus improve the tolerance of rice to salinity and nutrient deficiency stress. Plant Biotechnol J 16(1):310–321
Zhang HJ, Dong H, Shi YJ, Chen SY, Zhu YH (2007) Transformation of cotton (Gossypium hirsutum) with AhCMO gene and the expression of salinity tolerance. Acta Agron Sin 33:1073–1078
Zhang H, Dong H, Li W, Sun Y, Chen S, Kong X (2009) Increased glycine betaine synthesis and salinity tolerance in AhCMO transgenic cotton lines. Mol Breed 23(2):289–298
Zhang H, Shen G, Kuppu S, Gaxiola R, Payton P (2011a) Creating drought-and salt tolerant cotton by overexpressing a vacuolar pyrophosphatase gene. Plant Signal Behav 6(6):861–863
Zhang X, Zhen J, Li Z, Kang D, Yang Y, Kong J, Hua J (2011b) Expression profile of early responsive genes under salt stress in upland cotton (Gossypium hirsutum L.). Plant Mol Biol Rep 29(3):626–637
Zhang D, Li W, Xin C, Tang W, Eneji AE, Dong H (2012a) Lint yield and nitrogen use efficiency of field-grown cotton vary with soil salinity and nitrogen application rate. Field Crop Res 138:63–70
Zhang HJ, Dong HZ, Li WJ, Zhang DM (2012b) Effects of soil salinity and plant density on yield and leaf senescence of field-grown cotton. J Agron Crop Sci 198(1):27–37
Zhang L, Ma H, Chen T, Pen J, Yu S, Zhao X (2014) Morphological and physiological responses of cotton (Gossypium hirsutum L.) plants to salinity. PLoS One 9(11):e112807
Zhang T, Wang T, Liu KS, Wang L, Wang K, Zhou Y (2015) Effects of different amendments for the reclamation of coastal saline soil on soil nutrient dynamics and electrical conductivity responses. Agric Water Manage 159:115–122
Zhang K, Song J, Chen X, Yin T, Liu C, Li K, Zhang J (2016) Expression of the Thellungiella halophila vacuolar H+-pyrophosphatase gene (TsVP) in cotton improves salinity tolerance and increases seed cotton yield in a saline field. Euphytica 211(2):231–244
Zhang M, Cao Y, Wang Z, Wang ZQ, Shi J, Liang X, Song W, Chen Q, Lai J, Jiang C (2018) A retrotransposon in an HKT1 family sodium transporter causes variation of leaf Na+ exclusion and salt tolerance in maize. New Phytol 217(3):1161–1176
Zheng C, Jiang D, Liu F, Dai T, Jing Q, Cao W (2009) Effects of salt and water logging stresses and their combination on leaf photosynthesis, chloroplast ATP synthesis, and antioxidant capacity in wheat. Plant Sci 176:575–582
Zhu JK (2000) Genetic analysis of plant salt tolerance using Arabidopsis. Plant Physiol 124(3):941–948
Zhu JK (2016) Abiotic stress signaling and responses in plants. Cell 167(2):313–324
Zhu YN, Shi DQ, Ruan MB, Zhang LL, Meng ZH, Liu J, Yang WC (2013) Transcriptome analysis reveals crosstalk of responsive genes to multiple abiotic stresses in cotton (Gossypium hirsutum L.). PLoS One 8(11):e80218
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Singapore Pte Ltd.
About this chapter
Cite this chapter
Ahmed, N., Chaudhry, U.K., Ali, M.A., Ahmad, F., Sarfraz, M., Hussain, S. (2020). Salinity Tolerance in Cotton. In: Ahmad, S., Hasanuzzaman, M. (eds) Cotton Production and Uses. Springer, Singapore. https://doi.org/10.1007/978-981-15-1472-2_19
Download citation
DOI: https://doi.org/10.1007/978-981-15-1472-2_19
Published:
Publisher Name: Springer, Singapore
Print ISBN: 978-981-15-1471-5
Online ISBN: 978-981-15-1472-2
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)